3.1. Ageing Results
The results obtained from the data analysis are presented together in
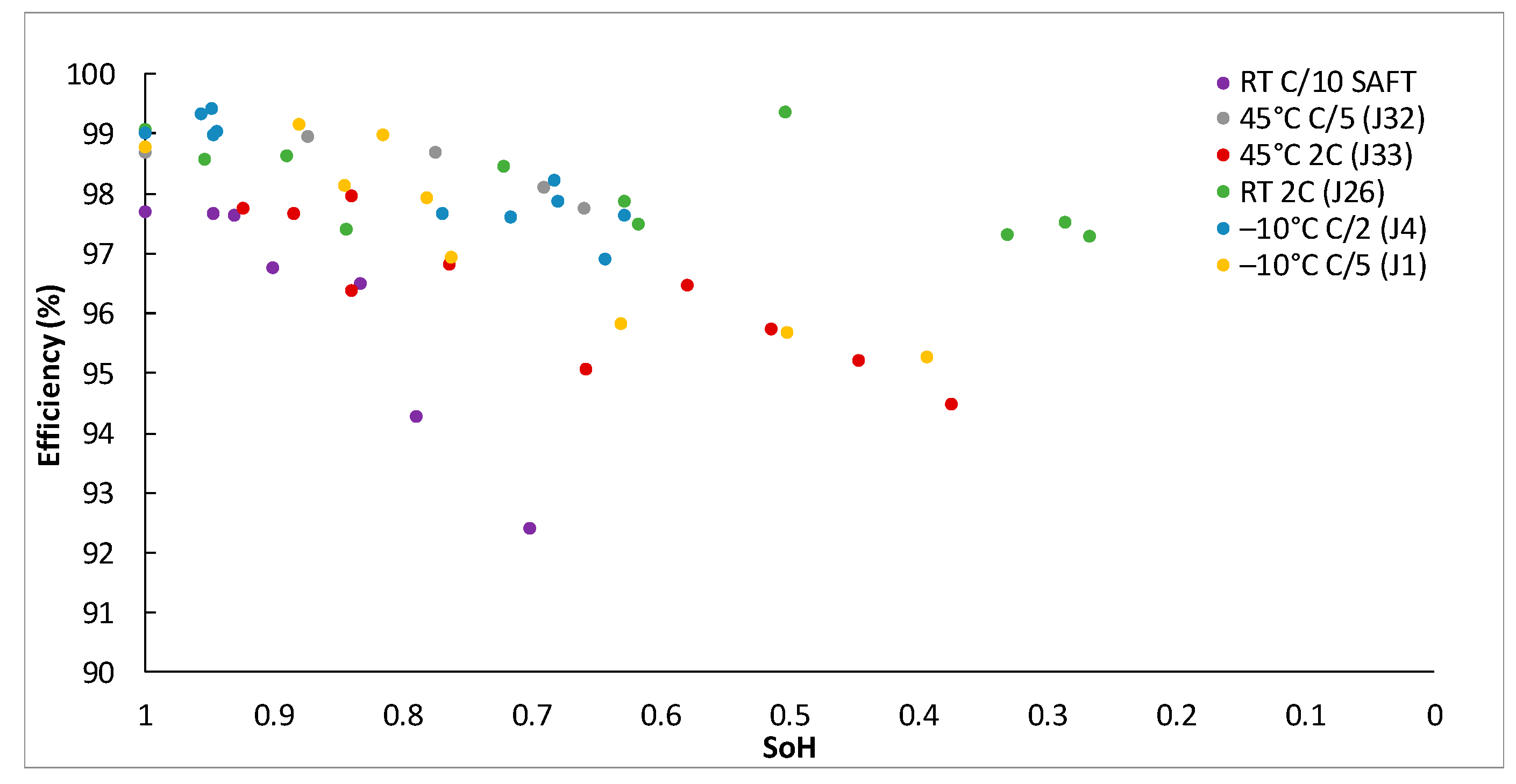
Figure 5 for all the endurance tests to highlight the similarity of the degradation trends (SoH reduction) that all cells follow independently of the cell.
Although there is a large dispersion in the measurements (
Figure 5), cells seem to follow a linear degradation of about –0.00057 SoH per cycle on average. This value is close to the values obtained by Shang et al. [
46] where, after a first rapid decrease, cells continued with a constant (0.000625 SoH per cycle) capacity decrease for over 200 cycles. Additional relevant information can be extracted from
Figure 5, for instance, there seems to be no clear relationship between the C-rate and the acceleration of cell degradation. This is demonstrated by the fact that the cell from SAFT (purple dots), which cycled at C/10 (the lowest current under test) and J33 (red dots), which cycled at 2C (20 times faster) follow almost the same trend. Similarly, temperature also does not appear to be a relevant factor that accelerates the ageing of Li-S cells. This can be observed in the figure by comparing the cells J4 (blue dots), SAFT (purple dots) and J33 (red dots) that cycled at –10 °C, room temperature (RT) and 45 °C respectively, and for which similar degradation trends are obtained.
To clearly state this first impression,
Table 5 presents the slope of the linear curve that best fits each cell submitted to endurance tests and the corresponding R square value. Effectively, these three cells (J4, J33 and SAFT) have a slope that is close to the average, reinforcing the idea that temperature and current have no relevant effect on ageing. Notice that the most rapid degradation occurs at C/5 at 45 °C (J32), however, there are too few points to ensure that this trend is going to be sustained after more cycles. In fact, it is worth mentioning that the behavior of the cell cycling at C/5 at –10°C (J4) initially had a similar degradation pattern but then stabilized for some cycles before again decreasing more rapidly. This behavior might indicate that the initial aggressive slope would be softened if cycled for a longer period. Similarly, after more than 600 cycles, cell 26 (cycling at 2C and RT) reaches the 60% SoH and then seems to suffer a substantial drop but in fact it re-stabilizes at 40% SoH.
The differences in capacity fading observed with Li-S cell cycling have also been noted in previous research. The trend that shows an initial linear capacity fade transitioning into a stabilization period and then continuing with a capacity loss was indicated in [
47] for cells cycled at C/10 and C/5, where stabilization periods of 150 to 350 cycles were observed. Furthermore, an increase of the capacity during the initial cycles, as observed for cell J33, has been highlighted in [
48] for Li-S cells cycled at C/10 and C/5 and temperatures of 20, 30 and 40 °C.
Thus, it seems reasonable to state that Li-S battery ageing does not behave like the Lithium ion in response to changes in temperature and C-rate. In fact, Lithium ion battery ageing is strongly affected by several factors that accelerate the ageing in different ways [
49,
50]. These factors are Temperature, State of Charge (SoC), C-rate and Depth of Discharge (DoD) [
51]. Typically, temperature has an exponential effect on ageing, meaning that the battery lifespan shortens as temperature increases [
52,
53]. The SoC and C-rate, on the other hand, follow first and second polynomial relations being more severe when the battery remains fully charged or suffers from higher intensive discharges [
54,
55]. The DoD follows a logarithmic behavior, which reverts in almost no ageing effect during small ripples or cycles that increases rapidly as the DoD increases, becoming relatively stable after 40% DoD [
56].
In comparison to the Li-ion ageing behavior, the endurance tests presented in this section indicate that Li-S ageing seems independent of changes in temperature and C-rate confirmed by the linear degradation trends with similar slopes under varying test conditions. However, this statement does not mean that the C-rate and temperature have no effect on Li-S battery performance. In fact, the tested coin cells displayed higher stability when working at higher temperatures and low C-rates but also presented poorer efficiency, as described later in this section. In addition, these results show that the sudden death or ageing knee that typically occurs in Lithium ion batteries [
57,
58] is not appreciable in Li-S batteries (some cells achieved 40% SoH and continued working).
There is another important aspect to look at related to battery ageing performance for traction purposes, which is the efficiency and loss of power. These two aspects are closely related to the internal resistance of the battery by the Ohm law, the higher the internal resistance, the higher the losses. Lithium ion batteries generally suffer an exponential internal resistance increase as SoH decreases, that is, the internal resistance increase is quite low at the beginning but is more and more noticeable as the battery ages. For instance, a study regarding the battery ageing of real electric vehicles using the internal resistance shows how at 88% SoH, the internal resistance of all the cells in the battery was slightly higher than at the beginning, but at 82% SoH their internal resistance was already 20% higher [
59] and it may rise even higher if the SoH goes beyond this point [
57], up to 200% at 60% SoH [
60].
To analyze what occurs with Li-S, the evolution of the efficiency measured for all cells during the endurance tests versus the SoH (Equation 1) was plotted (
Figure 6). Note that the dispersion is relevant due to the constant current cycling method, the particularities of the entrance into the second plateau, and the fact that the efficiency versus SoH is presented in the figure. However, it is difficult to identify any correlation between the evolution of efficiency versus SoH and the temperature or C-rate. The shuttle effect has an important impact on efficiency [
61], making Li-S cells less efficient at higher temperatures and at lower C-rates [
62]. It should be noted, however, that the cell with a lower efficiency and a quicker efficiency loss is the SAFT cell, which was manufactured following an industrial process. This cell was cycled at a lower C-rate where the self-discharge of the battery might noticeably interfere [
63]. Yet, the loss of efficiency through ageing is relatively low decreasing from 99% to 96% in all the cases except for the SAFT cell and cell J33. This is in accordance with the quite stable efficiency values presented in [
64], where three cells containing different separators to inhibit the shuttle effect were tested for more than 500 cycles. It further demonstrates that the internal resistance does not seem to increase in an exponential way as occurs with Li-ion batteries.
All these aforementioned issues indicate that, effectively, aged Li-S batteries perform similarly to new batteries but differ in initial capacity. Therefore, from a strictly ageing perspective, it seems that there should not be much difference in using brand new Li-S cells or aged cells apart from the bigger volume of the battery built from re-used cells and an uncertainty of a sudden malfunction of the cell corresponding to a shorter lifespan. However, some of the cells continued working well below a 50% SoH and, in one case (J26), 30% SoH was reached before the test was finally stopped. The stabilization of the capacity fade at below 50% SoH was also observed in previous research on Li-S pouch cells [
65], however the number of cycles achieved by these cells was reported to be significantly lower, reaching a 20% capacity fade before 50 cycles. The observed stabilization resulted from the inhibition of polysulfide diffusion caused by similar concentrations of sulfur/polysulfide in the electrolyte and carbon interface being reached [
65].
It should be noted that most applications will fail before such a low SoH is reached, and thus, a limit of 60% SoH at the end of the first and second life is acceptable, which is the value used for the LCA discussed in the following section. Furthermore, the preliminary ageing results presented for the few cells studied should be confirmed by future research.
The following recommendations should be considered when using Li-S batteries in stationary applications for both new or re-used batteries:
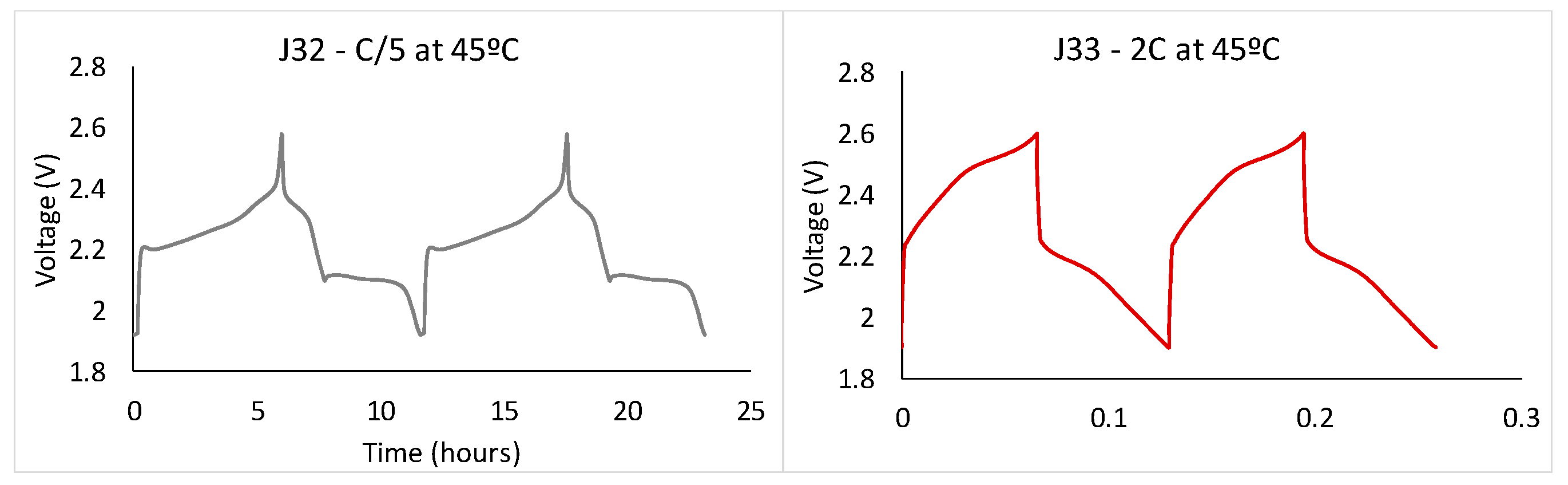
A loss of capacity occurs when cycled at high current rates (caused by the impossibility to enter the second plateau shown in
Figure 2).
Low temperatures result in a sudden decrease in performance of Li-S batteries (at temperatures below 0 °C)
High temperatures result in a loss in efficiency (due to an increase of the shuttle effect)
Very low C-rates or long durations without use result in a loss in efficiency (due to the shuttle effect)
3.2. Life Cycle Impact Assessment Results and Interpretation
The Life Cycle Impact Assessment results are presented and discussed in this section. For the system boundary defined in
Figure 3, the ageing test results from
Section 3.1 were used to define three scenarios for the analysis. From
Figure 5, the number of cycles achieved by the battery to reach 60% SoH can range from 552 to 912 cycles. The scenarios defined for the use phase are summarized in
Table 6. From this table, Scenario 2 is very close to and Scenario 3 exceeds the 150,000 km defined for most LCA studies on EVs, however, Scenario 1 does not quite meet this distance. Particularly for Scenario 1, the options for battery replacement may be considered depending on the End-of-Life of the EV.
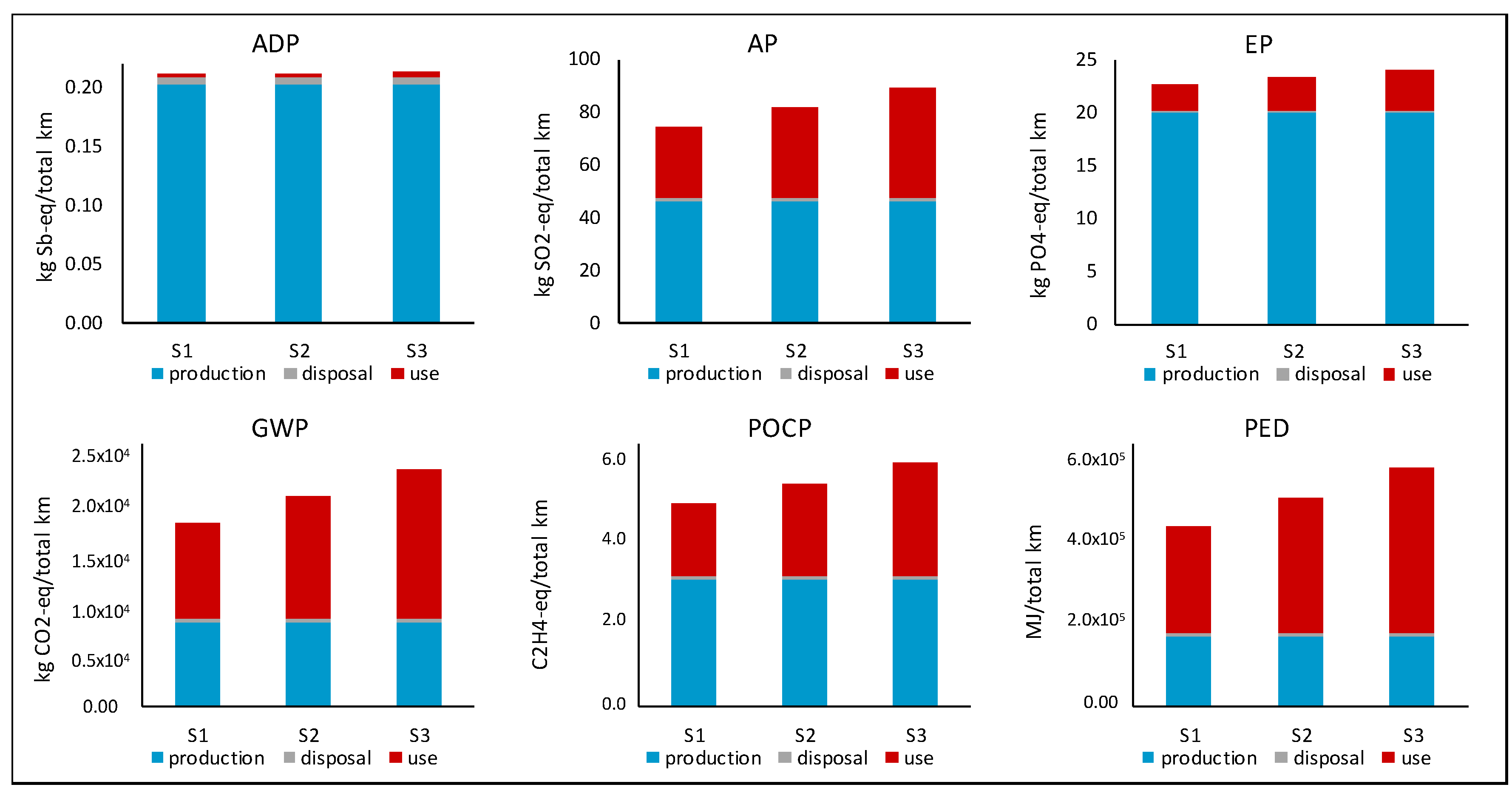
Figure 7 shows the impact per total kilometers achieved for each scenario defined in
Table 6 and for all impact categories assessed as described in
Table 4. The figure also shows the contribution of the production, use and disposal life cycle stages to the overall result. As can be seen, the production and disposal stages are the same for all scenarios. This is due to the fact that only one battery was considered for each scenario. For the use phase, however, the impact for each scenario differs depending on the quantified energy required as calculated with Equation (7). The energy required is dependent on the total number of cycles the battery is able to achieve before reaching its defined End-of-Life of 60% SoH. Therefore, the life cycle environmental impact of the Li-S battery changes based on the number of cycles (and hence the total kilometers) the battery is able to achieve. However, the amount of this change differs depending on the impact category being assessed. For example, the contribution of the use phase to the ADP elements (referred to as ADP from here forward) is insignificant compared to the production stage and therefore changes in the use phase will not significantly change the overall result.
For all the other impact categories assessed (AP, EP, GWP, POCP, PED), however, the use phase has a more significant contribution to the overall result than seen for ADP. Thus, the result for these impact categories increases with an increase in the number of cycles achieved, as defined in each scenario. It can further be seen for these impact categories that the production phase also has a contribution to the overall result, and thus both the production and use life cycle stages are important for quantification of the overall environmental impact for these impact categories.
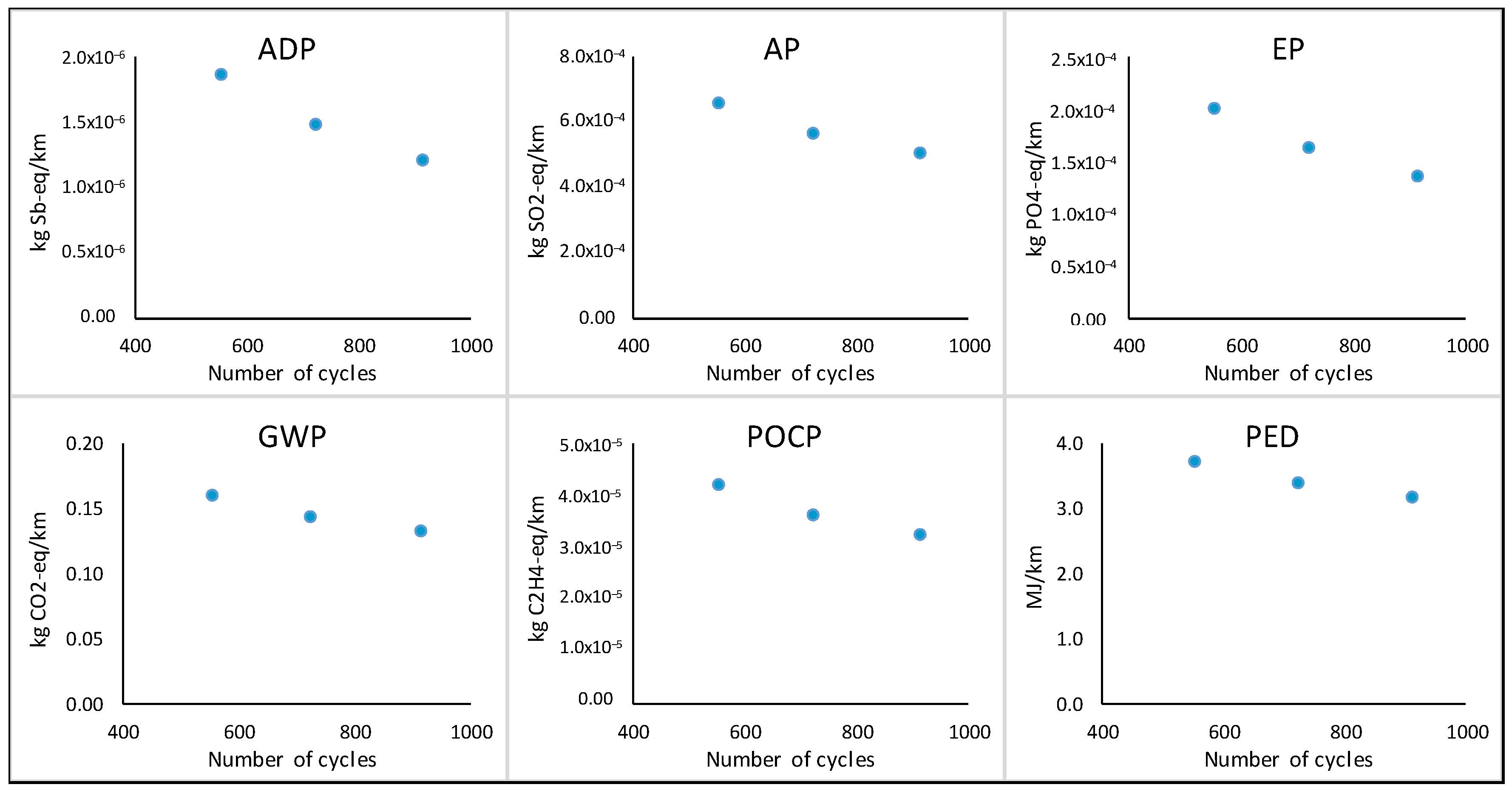
In order to see the environmental benefits of the extended lifespan of the battery due to more cycles being achieved, it is necessary to look at the results per functional unit of 1 km (
Figure 8). In
Figure 8, the trend clearly shows that as the number of cycles achievable by the battery improves from 552 towards 912 cycles, the impact per km also improves.
Table 7 further summarizes the results per functional unit (per km) and per reference flow (per total km for one battery).
Therefore, for the use phase, improvements in the environmental impact will come from extending the lifespan of the battery, as well as from the improvement in efficiencies and decarbonization of the electricity grid mix, as discussed in the introduction. Furthermore, it is shown that one 50 kWh Li-S battery, taking into consideration the effects from cycle ageing, has the potential to reach the 150,000 km reference that is often used in comparative LCA studies for EVs and ICEVs.
Since it was further found that the production phase also contributes to the results (
Figure 7), the contribution of the battery components to the production phase was further investigated.
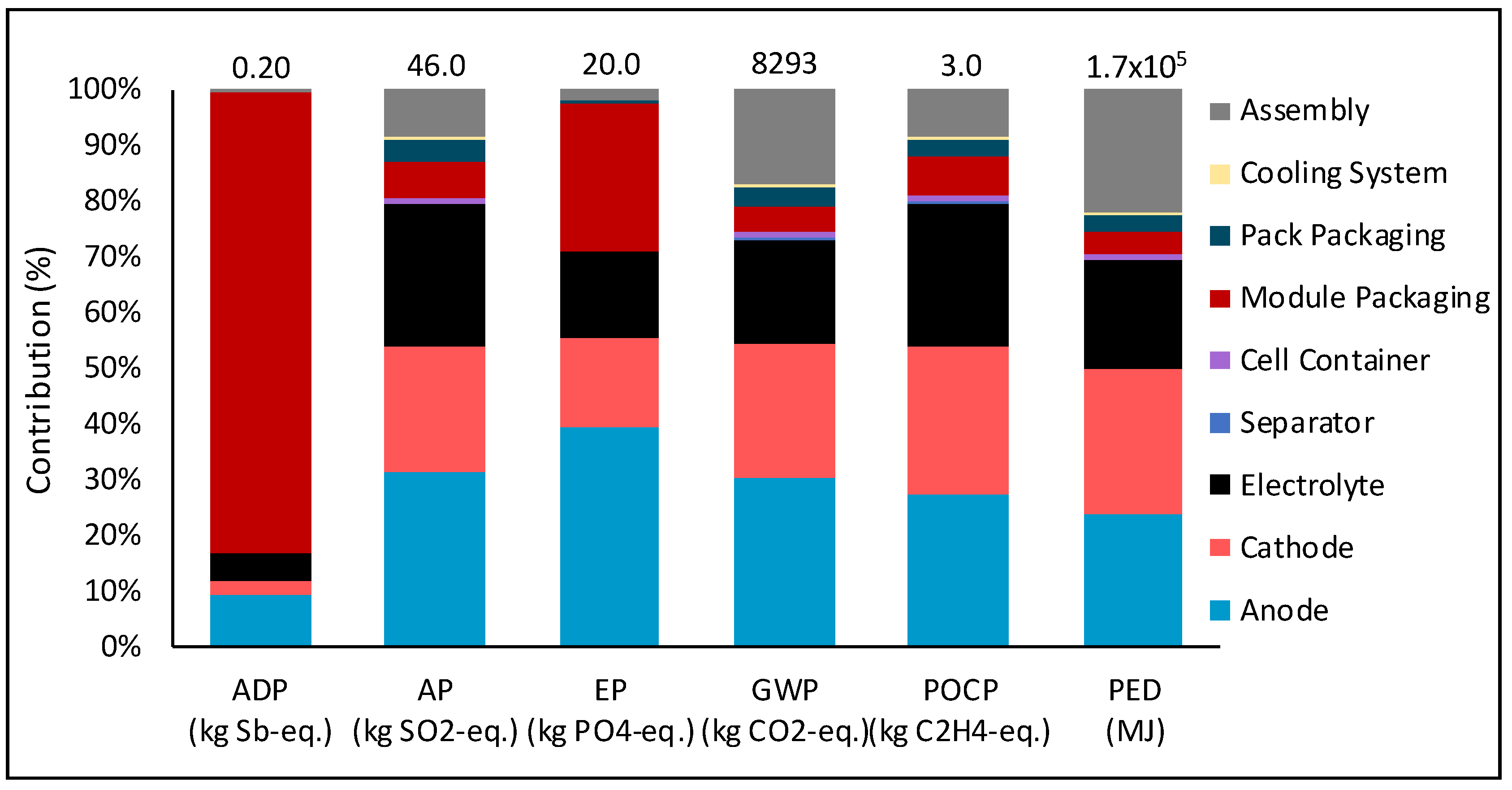
Figure 9 shows the percent contribution of each component to the total result due to production of the battery. The material components are defined in the bill of materials in
Table 3.
From
Figure 9., the module packaging contributes significantly to the ADP. The module packaging consists of electronic components, the contribution of which was found to range from 80% to 99% of the total impact of the module packaging production depending on the impact category (
Figure 10). A complete list of the components of the module packaging can be found in [
43]. Therefore, the main contributing component to the ADP, which was found to come from the production stage (
Figure 7), is due to the electronic components in the modular packaging (
Figure 9 and
Figure 10).
For all other impact categories, the contribution of the active material (cathode, anode and electrolyte) is greater than 70% to the total impact of battery production (
Figure 9). This material has been scaled from that in a coin cell with the use of Equations (3)–(6). Therefore, this data should be updated when more tests are conducted on Li-S batteries and more data is available for the composition and performance of larger batteries. This is important as the production of the battery contributes to the overall result for all impact categories, as was seen in
Figure 7. Therefore, improvements in this data will influence the overall result for each of the environmental impact categories assessed.