Abstract
The thermal aging property of oil-paper insulation is a key factor affecting the service life of transformers. In this study, nano-Al2O3 was added to insulating paper to improve its anti-thermal aging property and delay the aging rate of the insulating oil. The composite paper containing 2% nano-Al2O3 had the highest tensile strength and therefore was selected for the thermal aging test. The composite and normal papers were treated with an accelerated thermal aging experiment at the temperature of 130 °C for 56 days. The variations of the degree of polymerization (DP) and tensile strength of the insulating papers with aging time were obtained. The characteristics of the insulating oil, including color, acid content, breakdown voltage, and dielectric loss were analyzed. The results revealed that compared with a plain paper, the composite paper maintained a higher DP, and its tensile strength decreased more slowly during the aging process. The oil-impregnated composite paper presented a lighter-colored oil, less viscosity changes, and a considerably lower quantity of thermal aging products. In addition, nano-Al2O3 can effectively adsorb copper compounds and keep part of the acid products and water away from the thermal aging process. This characteristic restrained the catalysis of copper compounds and H+ in the thermal aging reaction and reduced the thermal aging speed of both the insulating paper and the insulating oil.
1. Introduction
The power transformer is the core of power transmission and is one of the key units of equipment for power systems [1]. Oil-paper insulation is an insulating structure widely used in large power transformers. During long-term operation, aging and operation failures diminish the insulation performance of oil-paper insulation systems. The insulation performance of the insulating oil can be recovered by changing the oil, whereas the insulating paper cannot be replaced during the operation. Therefore, the performance of the insulating paper mainly determines the service life of a transformer [2].
Cellulose-insulating paper is subjected to thermal degradation, oxidation degradation, and hydrolysis during the operation of a transformer [3,4,5,6], during which its insulation performance gradually deteriorates, thus endangering the safe operation of the power system. To extend the service life of power transformers, scholars have studied the performance of cellulose-insulated paper for a long time. The following two methods have been recognized and successfully applied to industrial production [7,8]: (1) the introduction of more stable chemical groups to replace hydrophilic groups in cellulose, including cyanoethylation, and acetylation [4] and (2) the addition of thermal stabilizers, primarily amine compounds, including dicyandiamide, urea, and polyacrylamide [9], which can consume the water and acid produced in the aging process to delay the thermal aging.
Nanoparticles have various special effects because of their small size, large specific surface area, and so on [10]. Therefore, the use of inorganic nanoparticles in nanocomposite dielectrics to enhance the properties of insulating materials has been extensively studied in recent years [11,12]. J.K. Nelson first introduced nano-TiO2-doped epoxy resin (EP) [13]. The results showed that compared with the traditional micrometer doped composite dielectric, the dielectric constant of the nano-TiO2/EP composite was reduced, and its space charge was suppressed. The addition of nano-MgO to low-density polyethylene improved the breakdown voltage, volume resistivity, and space charge distribution under DC voltage [14]. The addition of ZnO and TiO2 to LDPE and epoxy resin, respectively, effectively improved the volume resistivity, breakdown, and dielectric properties of composites [15,16,17]. Several reports have shown that adding Al2O3 to polyimide can improve the breakdown electric field strength and corona resistance of composites, and that Al2O3 exerts a thermal stability effect [18,19,20]. The doping of inorganic nanomaterials during polymer modification can significantly improve the electrical properties of the polymer dielectric. It is also confirmed that the nanoparticles improve the dielectric properties of the mineral oil [21].
At present, few studies have explored the modification of cellulose-insulating paper by inorganic nanoparticles. Our previous study on nano-Al2O3 doping on the insulating paper focused on the mechanical, dielectric, and breakdown properties [19].
Expanding on our previous work, this paper examined the thermal aging properties of composite insulating paper doped with nano-Al2O3. Accelerated thermal aging tests revealed that the doping of nano-Al2O3 can effectively delay the degradation of the degree of polymerization (DP) and mechanical properties of the insulating paper in the aging process and delay the thermal aging rate of the insulating oil [22]. In this paper, nano-Al2O3 modified papers and plain papers were prepared for 56 days thermal aging experiment. The DP and tensile strength of the insulating paper were tested, since they are widely regarded as the parameters to evaluate the aging level. As there is no judging standard for the degree of aging of the insulating oil, the parameters of the insulating oil such as color, acid value, breakdown voltage, and dielectric loss with aging will be tested to reflect the change of deterioration of the insulating oil. Finally, the mechanism of the influence of nanoparticle doping on retarding the aging rate of oil-paper insulation was put forward, and verified by the test result of content of copper in oil during the aging.
2. Thermal Aging Experiment of Oil-Paper Insulation
2.1. Preparation of the Nano-Al2O3 Modified Paper
The experiment used paper made in the laboratory, and the raw material was unbleached Kraft softwood pulp imported from Russia. The nanoparticle used in the experiment was nano-Al2O3 which had a purity of ≥99.99%, an average particle size of 20 nm, and a specific surface area of 200 m2/g. Walley beater (XYTEST Co.,Ltd., Xianyang, Shanxi, China) was used to beat the pulping board to the specific beating degree (45° SR)to obtain the pulp. After filtration and dissociation, the slurry was poured into a fast paper sheet former (XYTEST Co.,Ltd., Xianyang, Shanxi, China), as shown as Figure 1a, to make the insulating paper sheet with a basis weight of 120 g/m2.

Figure 1.
(a) Fast paper sheet former; (b) Nano-Al2O3 surface treatment.
To increase the adhesion of inorganic nano-Al2O3 on the cellulose chains of the organic matrix and reduce the agglomeration of nanoparticles, the surface treatment of nano-Al2O3 was conducted using a silane coupling agent KH550. Five grams of nano-Al2O3 and 0.5 mL of KH550 were added to 500 mL of the dispersion medium (V(C2H5OH):V(H2O) = 19:1), and the surface-treated nano-Al2O3 solution was obtained by stirring and sonicating for 6 h at 40 °C, as shown in Figure 1b. The surface-treated nano-Al2O3 was doped into the slurry, and the mixture was evenly mixed. Then, the same procedure was used to prepare the composite insulating paper sheet.
2.2. Determination of Doping Amount
Studies have found that due to the interface effect of nanoparticles, doping with an appropriate amount of nanoparticles, normally 1 wt% to 3 wt%, depends on the kinds of matrix and nano-particles, and can effectively improve the electrical strength of the composite insulating paper [19]. However, many nanoparticles tend to agglomerate into larger-particle-sized inorganic particles during the papermaking of the insulating paper, such as Al2O3, MgO, SiO2, ZnO, and so on, thereby reducing the tensile strength of the composite insulating paper [20]. In this study, the nano-Al2O3 doping amount that can maximize the nano-effect was determined by testing the tensile strength of the composite insulating paper with different nano-doping amounts (as shown in Figure 1).
In this study, the AT-L-1 tensile testing machine (Annimat Instrument Co., Ltd., Jinan, Shandong, China) was used to measure the tensile strength of the insulating paper in accordance with the constant speed tensile method (ISO 1924-2-2008).
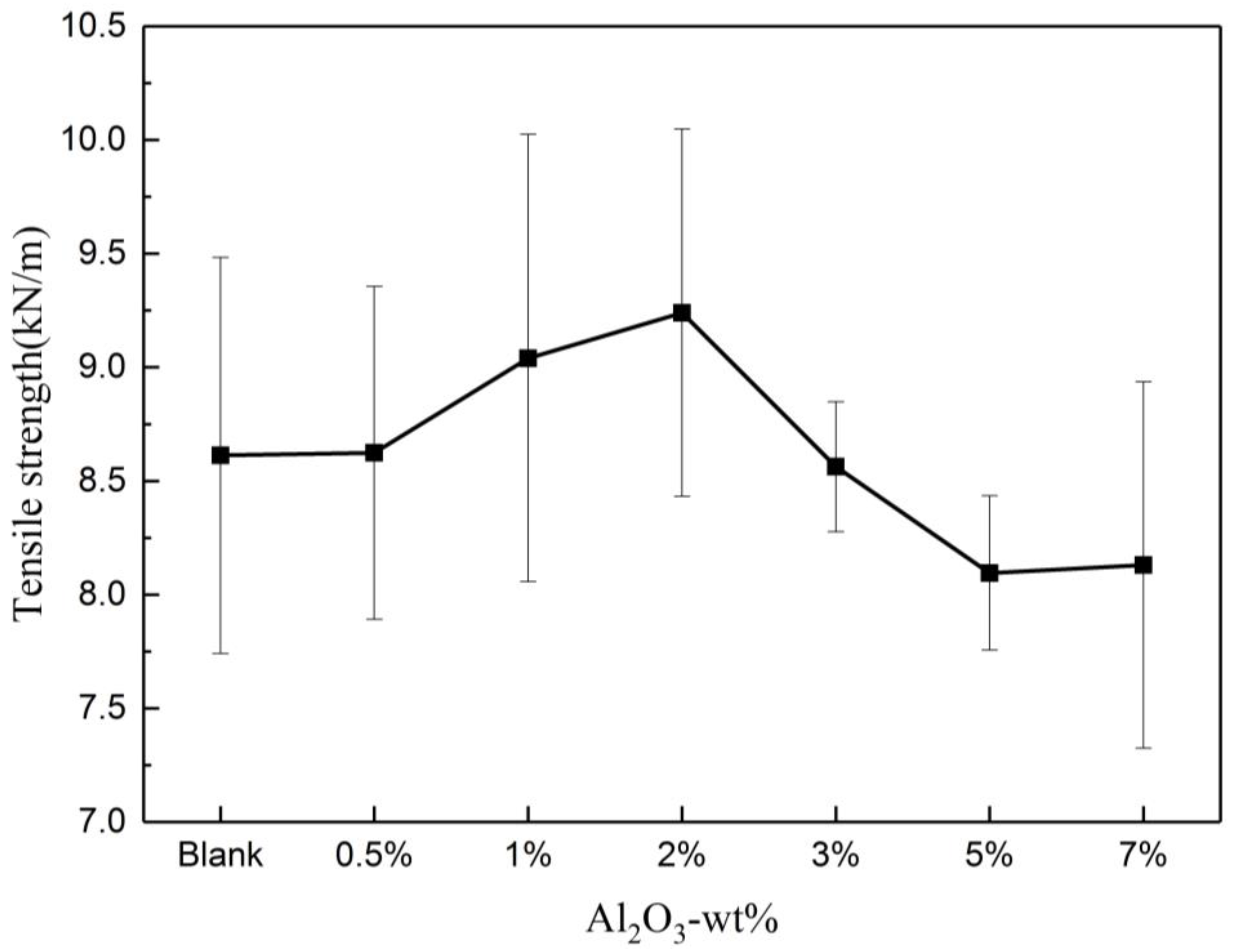
Figure 2 shows that the tensile strength of the composite insulating paper initially increased and subsequently decreased with the increase in the nano-Al2O3 doping content, reaching the highest when the doping amount was 2 wt%. Nano-Al2O3 can exert the best nano-effect when the doping amount was 2 wt% in the papermaking process, which means the minimal reunion. Therefore, a doping amount of 2 wt% composite insulating paper was adopted for the subsequent thermal aging test.
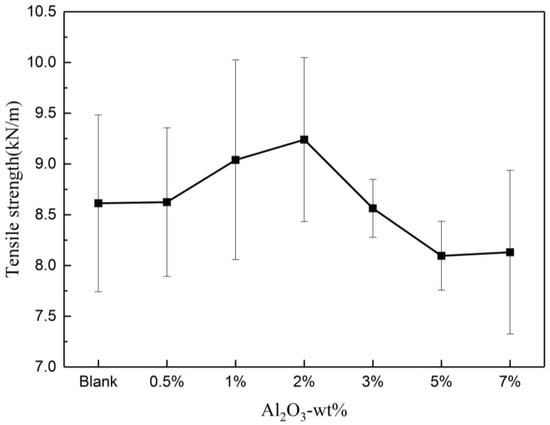
Figure 2.
Variation of tensile strength with Al2O3 content.
2.3. Thermal Aging Properties of Paper and Oil
To study the effect of nano-particles on the thermal aging performance of the oil-paper insulation, this paper tested and analyzed the thermal aging characteristics of the oil-impregnated insulating paper and oil through an accelerated thermal aging test. First, an ordinary insulating paper and a composite insulating paper were dried at 50 Pa and 90 °C for 48 h. Then, the dried paper was impregnated with 25# Xinjiang Karamay mineral insulating oil, which was degassed at 50 Pa and 40 °C for 24 h. The oil and the paper were placed in a ground glass jar under a nitrogen atmosphere to maintain the oil-paper mass ratio of 20:1. An appropriate amount of copper strip was added before the jar was sealed.
The samples were placed in a 130 °C aging chamber for accelerated thermal aging. Samples were collected at 0, 7, 14, 28, and 56 days to test the thermal aging parameters of the paper and the oil. The DP and tensile strength of the paper, and the color, acid content, breakdown voltage, and dielectric loss of the oil were chosen to characterize the degree of aging.
In order to facilitate the subsequent comparison and analysis, the ordinary insulating paper and nano-Al2O3 modified insulating paper were defend as P0 and P1, while the insulating oil with ordinary insulating paper and the insulating oil with modified insulating paper were defined as O0 and O1.
2.3.1. Degree of Polymerization of Insulating Paper
The DP is one of the most commonly used parameters for the characterization of insulating paper. The DP refers to the number of glucose repeating units constituting the long chain of fibers in the insulating paper. This paper used the NCY-2 automatic viscometer (Srida Scientific Instrument Co., Ltd., Shanghai, China) to measure the DP of the insulating paper in accordance with ASTM D4243-99. Figure 4 shows the variation of the DP with time for P0 and P1 at 130 °C.
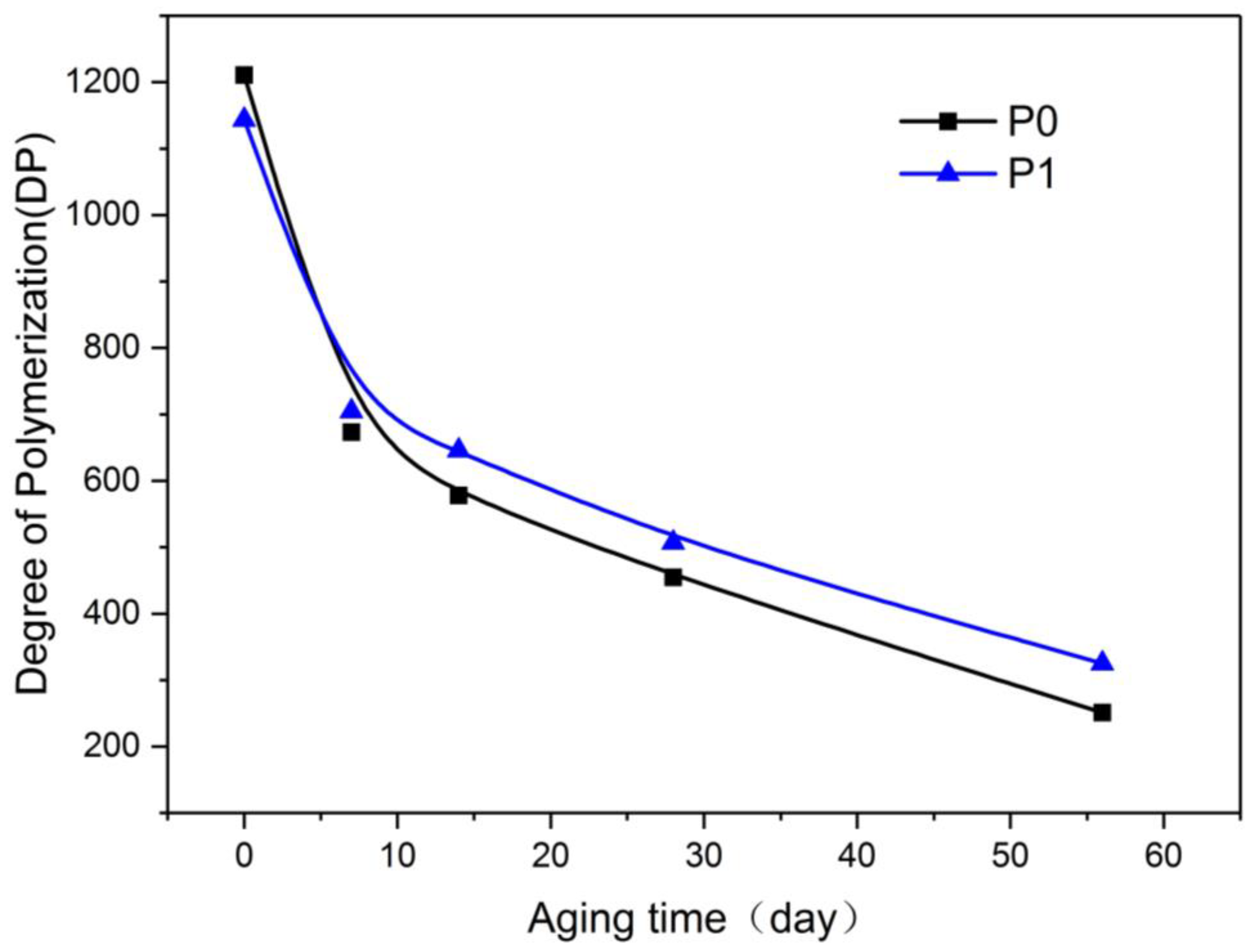
Figure 3 shows that at the beginning of aging, the DP of P1 was approximately 5.5% lower than that of P0. With prolonged aging time, the DP of P1 started to surpass that of P0. At the end of the aging test, the difference in the DP of P1 and P0 increased. After 56 days of aging, the DP of P0 was reduced to 251, suggesting that the aging was nearly over, whereas the DP of P1 was approximately 29.4% higher than that of P0. These findings demonstrated that the aging rate of P1 was lower than that of P0.
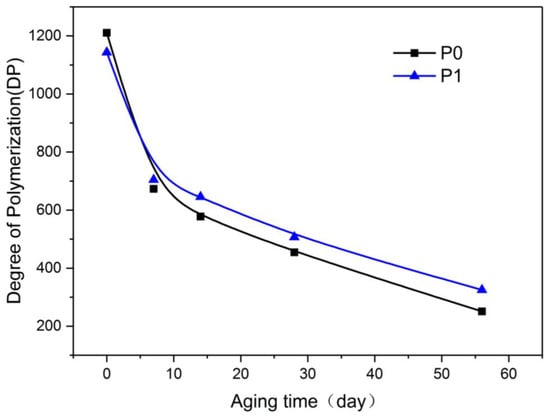
Figure 3.
Variation of degree of polymerization (DP) with aging process.
2.3.2. Tensile Strength of the Insulating Paper
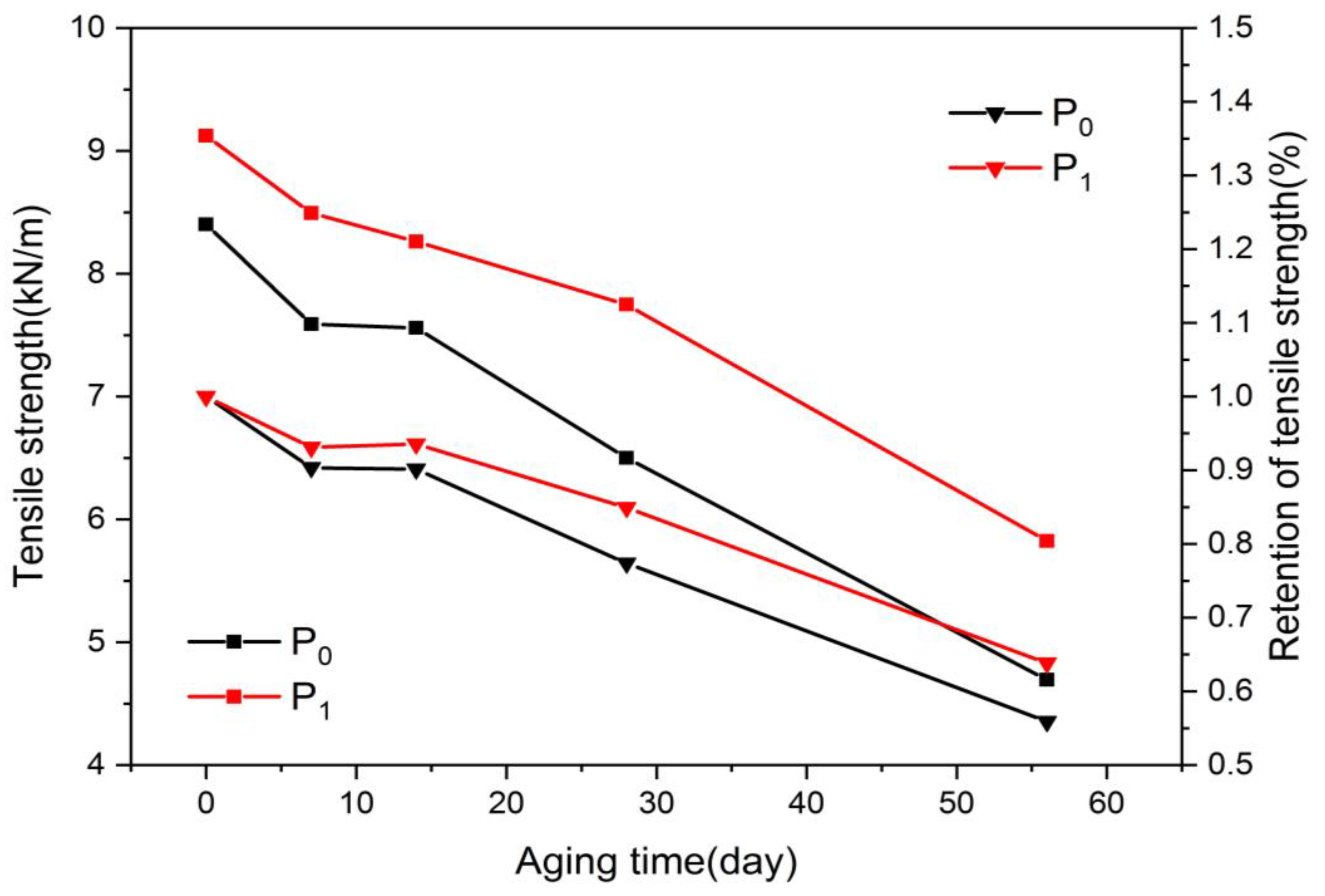
The tensile strength is the ability of a material or component to resist damage when pulled, and can act as an indicator of paper’s mechanical performance. The effect of the aging of oil-paper insulation on the mechanical properties of the insulating paper was mainly reflected in the change in the tensile strength. Figure 3 shows the variation of the tensile strength of insulating the paper, in which the tensile strength was retained during aging. On day 0 of aging, the tensile strength of P1 was higher than that of P0. The doping of nano-Al2O3 effectively improved the mechanical strength of the insulating paper, and this property was not provided by the chemical modification and the addition of the heat stabilizer.
As the thermal aging time progressed, both the modified paper and the plain paper gradually deteriorated, and the cellulose chain broke and cleaved. Therefore, the tensile strength of both kinds of insulating papers tended to decrease continuously. As shown in Figure 4, the tensile strength of the modified paper was always higher than that of the plain paper throughout the thermal aging process. Nano-Al2O3 doping can maintain the high mechanical strength of the insulating paper during the thermal aging process.
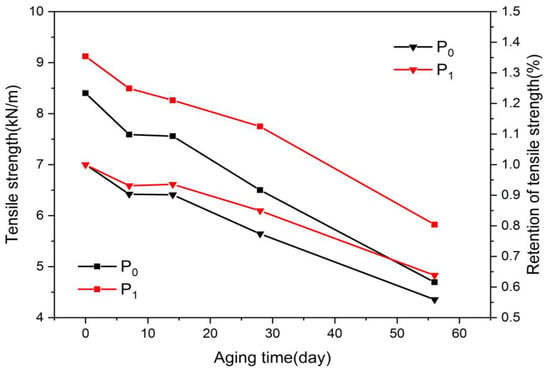
Figure 4.
Variation of tensile strength with aging process.
The tensile strength retention rate of the insulating paper is considered an important indicator of the remaining thermal life of the insulating paper in the thermal aging process. Figure 4 shows that the retention rate of the tensile strength of the modified paper was higher than that of the plain paper throughout the aging process. After 56 days of aging, the tensile strength retention rates of the modified paper and the plain paper were approximately 56% and 64%, respectively, indicating that the modified paper had a higher residual heat life and better heat aging resistance.
2.3.3. Color of Insulating Oil
In general, as the aging of the oil-paper insulation progressed, the accumulation of aging products of the insulating paper and the insulating oil increased, and the color of the insulating oil gradually deepened. Figure 4 shows the trend of the color of O0 and O1 with aging time. Figure 5a,b show the O0 and O1 on five different thermal aging period, 0, 7, 14, 28, and 56 days from left to right. As shown in Figure 4, as the degree of aging increased, the color of the two kinds of insulating oil gradually changed from colorless and transparent to yellow. The color of O1 was obviously lighter than that of O0 during the latter period of the aging test, indicating to some extent that the accumulation of aging products in O1 was less than that in O0.

Figure 5.
(a) Color of O0; (b) Color of O1.
2.3.4. Acid Content in Oil
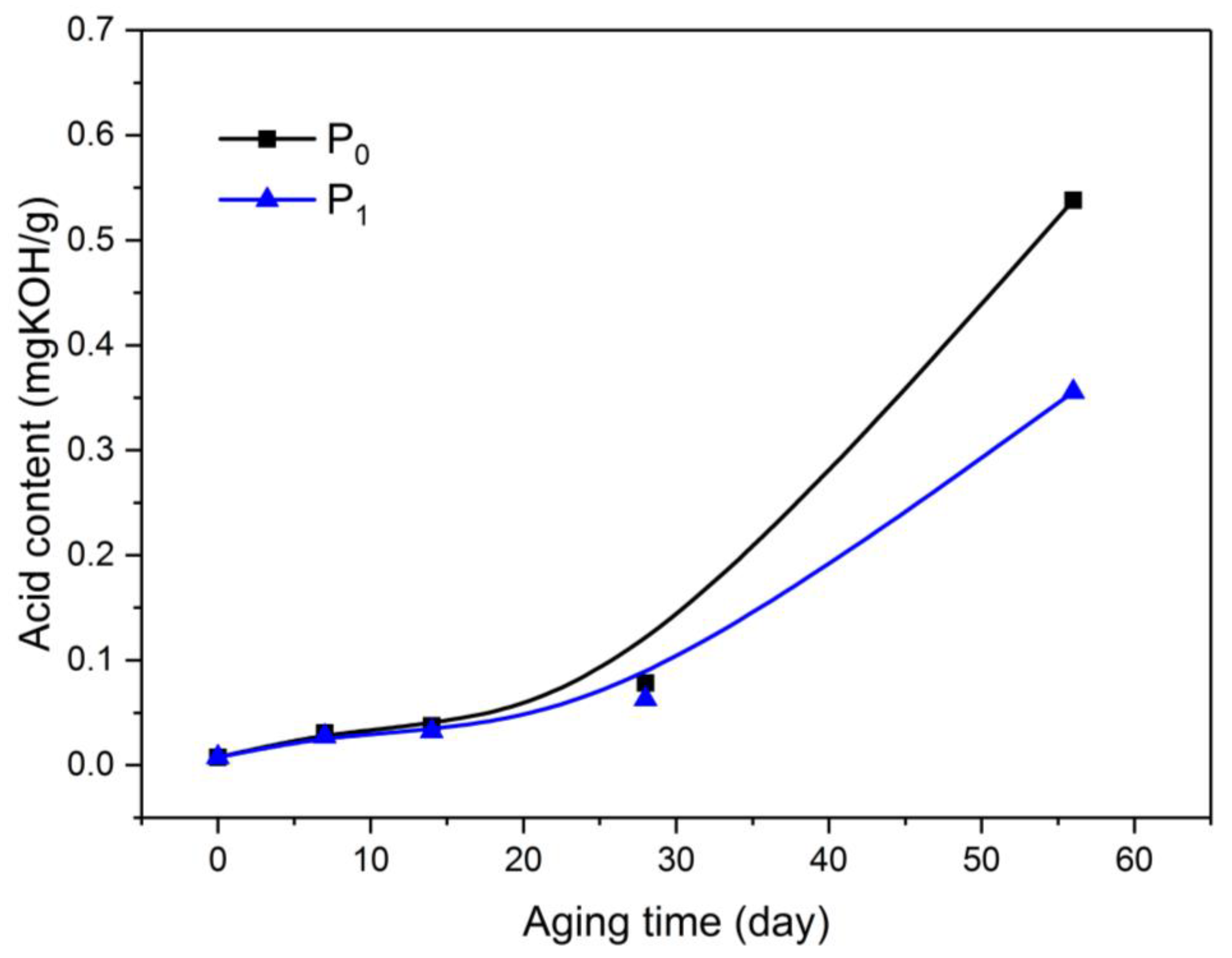
Among the acid substances produced by the thermal aging and decomposition of oil-paper insulation, small-molecule acids were mainly produced by the aging decomposition of insulating paper. The strong hydrophilicity was mainly adsorbed in the insulating paper, whereas the macromolecular acid was mainly decomposed by the aging of the insulating oil. The distribution of the acid in the insulating oil can reflect the aging of the oil [21]. Therefore, the acid content in oil is important for measuring the aging of the insulating oil. This article used the Metrohm 907 Titrino automatic potentiometric titrator (Metrohm Co., Ltd., Herisau, Swiss) to measure the acid content in the oil in accordance with IEC 62021-1-2003. Figure 6 shows the measurement of the acid content in the oil during the aging test.
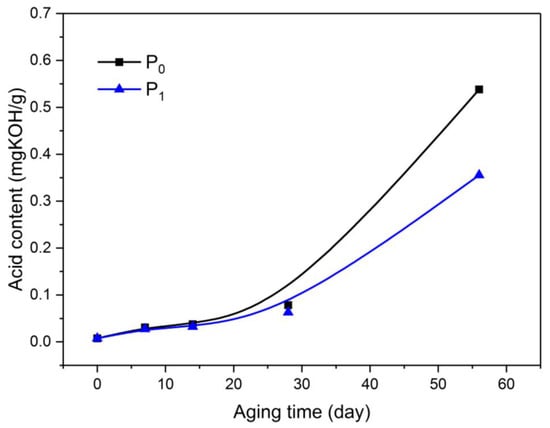
Figure 6.
Variation of acidity in oil with aging process.
At the initial stage of aging, the acid content in the insulating oil was low, and the two kinds of oil displayed no difference in acid content. As the aging progressed, the acid content in O1 was lower than that in O0 during the early aging period. In the middle of aging, the acidification rate of O0 was higher than that of O1. By the end of the aging test, the acid content in O1 was lower than that in O0 by 33.92%.
2.3.5. Breakdown Voltage of Oil
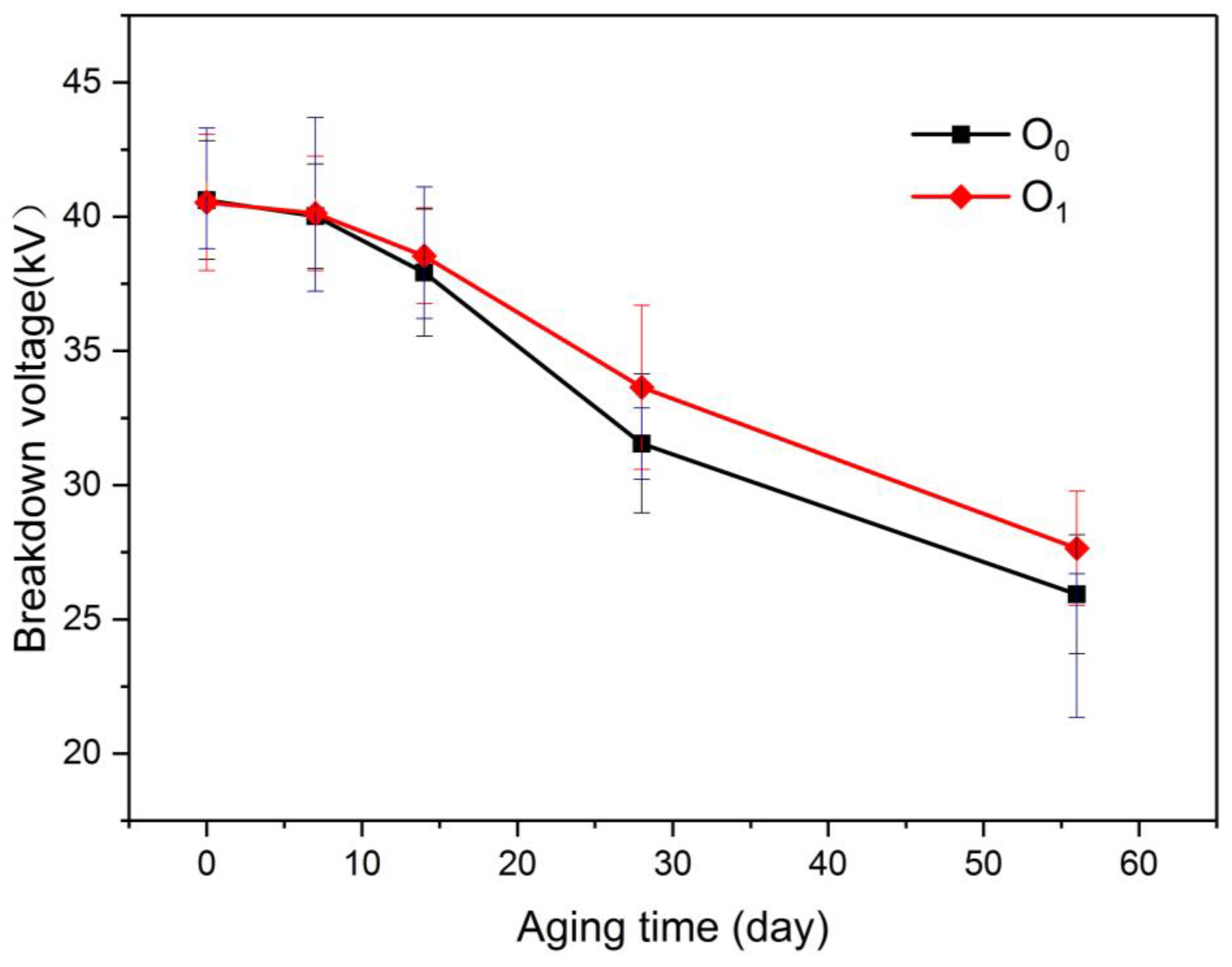
The power frequency breakdown voltage of the insulating oil, as the most direct electrical strength parameter of oil-paper insulation, is an important parameter for characterizing the electrical performance of liquid dielectric insulation. The breakdown voltage of insulating oil is measured according to IEC60156, using IJJD-80 automatic insulating oil dielectric strength tester (Ruixin Electrical Test Equipment Co., Ltd., Wuhan, Huibei, China). The breakdown voltage of two insulation oils at different aging stages was tested. The test results are shown in Figure 7.
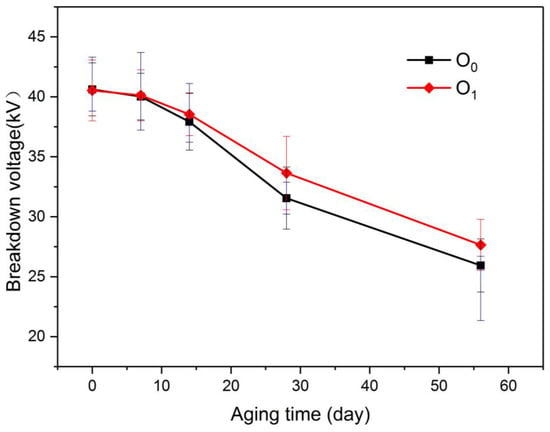
Figure 7.
Variation of breakdown of voltage in oil with aging process.
As depicted in the diagram, the power breakdown voltages of O0 and O1 were reduced as the degree of aging increased. During the entire aging process, the breakdown voltage of O1 was always higher than the power frequency breakdown voltage of O0, indicating that the insulation degree of aging of the oil-paper insulation with the modified paper was lower than that of the ordinary oil-paper insulation, although the gap between O1 and O0 at the same aging stage was insignificant.
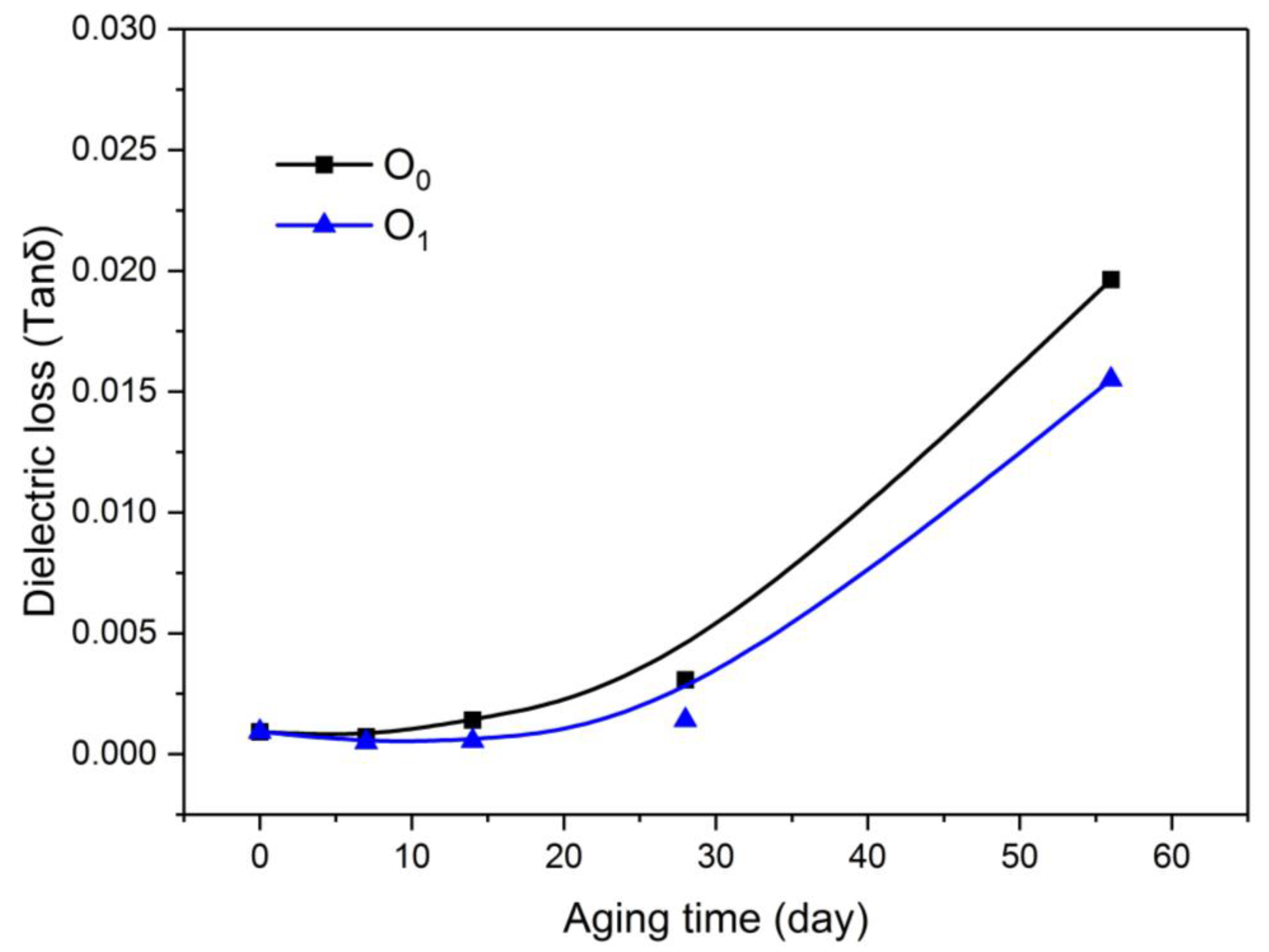
2.3.6. Dielectric Loss of Oil
A DTL C dielectric instrument (BAUR GmbH Co., Ltd., Sulz, Vorarlberg, Austria) was applied to measure dielectric properties of immersed insulating paper and insulating oil. The test temperature is 90°C and the test frequency is 50 Hz, according to IEC 60156:1995.
As shown in Figure 8, the dielectric loss factor (tanδ) of the two insulating oils increased with increasing aging time, and the dielectric loss factor of the insulating oil with the modified paper was lower than that of the oil with the unmodified paper.
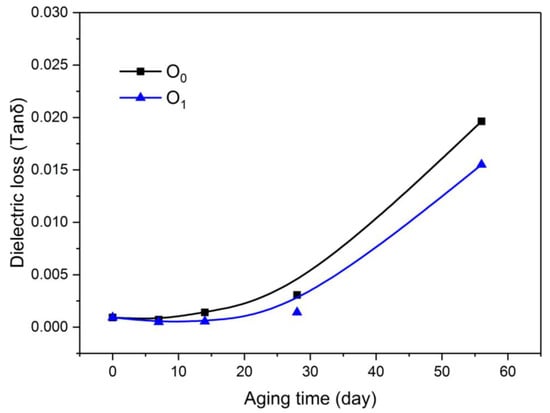
Figure 8.
Variation of oil dielectric loss with aging process.
In addition, various impurities, such as paper fibers that have been aged, diffused into the oil and increased the conductance loss of the insulating oil, causing the loss factor of the dielectric oil to increase rapidly with the aging time.
3. Discussion
The test and analysis of the thermal aging parameters of the oil-paper insulation at various aging stages revealed the doping of nano-Al2O3 reduced the degree of deterioration of the oil-immersed composite insulating paper in the thermal aging process. Therefore, the doping of nano-Al2O3 with different crystal forms effectively retarded and inhibited the thermal aging of both the insulating paper and the insulating oil.
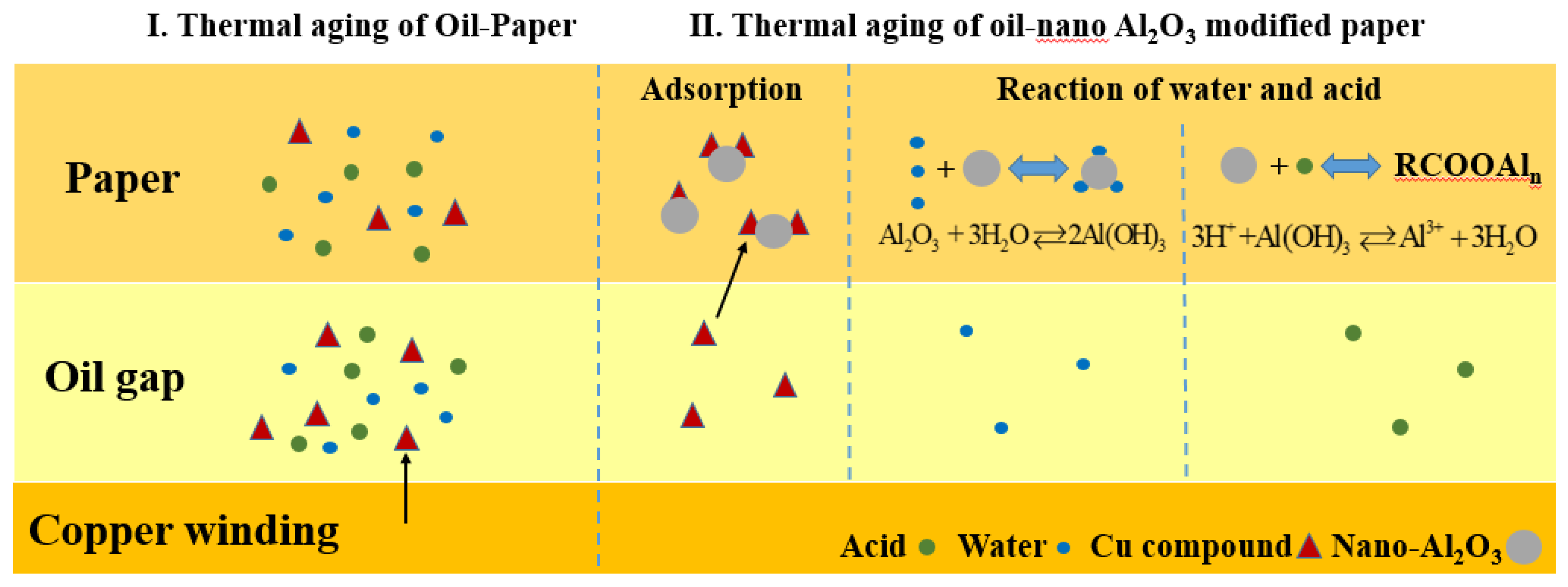
During the aging process of oil-paper insulation, the insulating paper and the insulating oil were hydrolyzed to produce water under the action of thermal stress. The insulating paper produced small molecular acids, such as formic acid and acetic acid [21], in the cracking reaction. The small molecular acids were dissolved in water to produce H+, which further catalyzed the hydrolysis process. This “positive feedback” effect implied that water and small molecular acids play important roles in the thermal aging reaction [10]. In view of the strong hydrophilicity of the insulating paper, most of the moisture of the oil-paper insulation was adsorbed on insulating paper. As the degree of aging increased, the moisture in the insulating paper gradually accumulated, and the catalytic activity was enhanced.
In addition, copper bars were added to simulate the actual condition of the transformer with a copper compound, thereby accelerating the aging of the insulating oil [23]. Copper compounds generate peroxide radicals by decomposing hydrogen peroxide, which promotes the oxidation of oil products. Therefore, an increase in the content of copper compound increased the aging rate of the transformer oil. At the same time, copper compound was produced during the aging process of the oil-paper insulation, and acidic aging products further accelerated the corrosion of copper and generated copper compounds. In the aging process of a transformer, in addition to the residual oxygen inside and the small amount of oxygen infiltrated from the outside, the aging products of the insulating oil also contained oxidants, which would accelerate the aging of the insulating paper and thus cannot be ignored [24].
Therefore, suppressing the small molecule acid in the oil-paper insulation and suppressing the copper element content in the oil are effective measures to delay the aging of the oil-paper insulation.
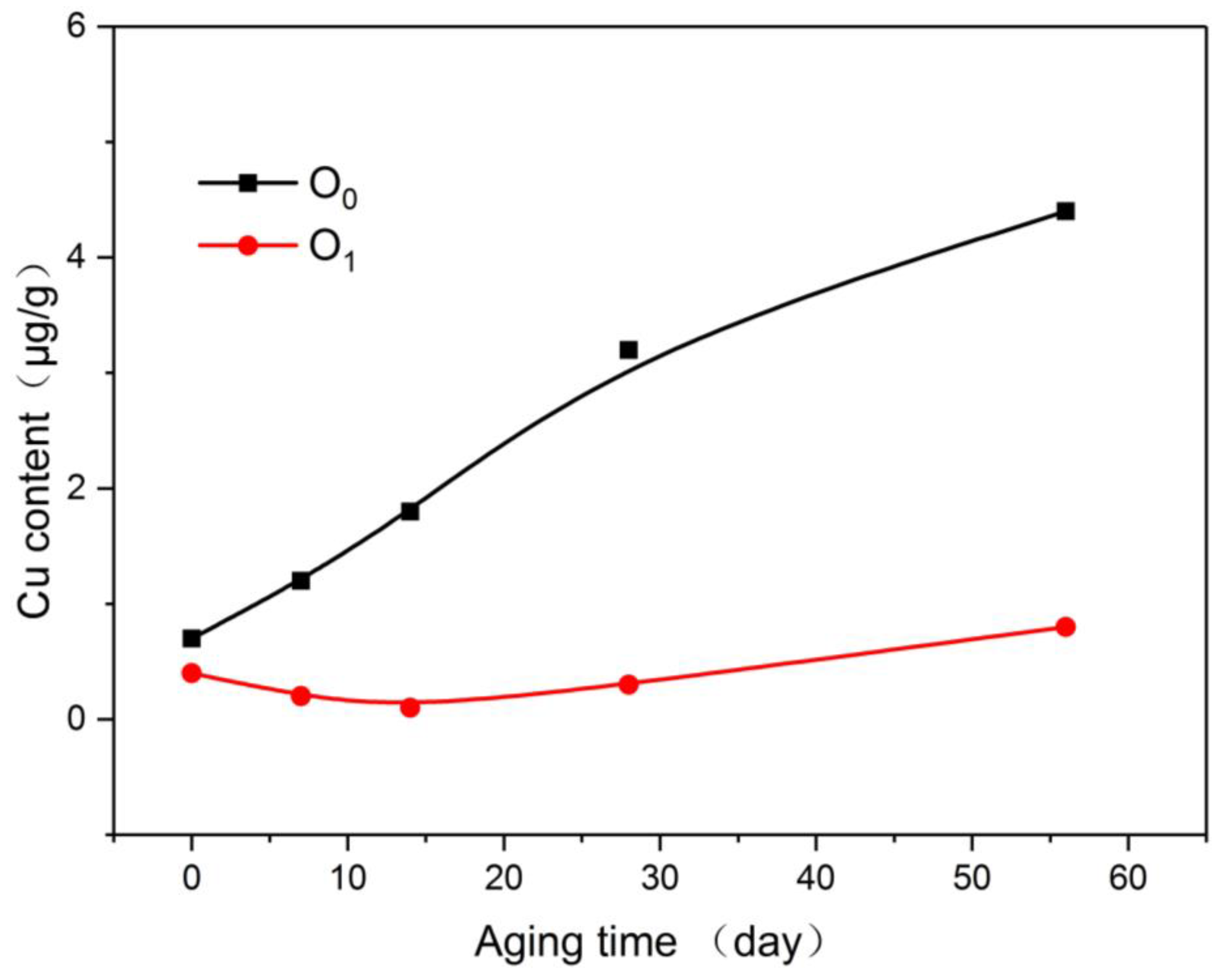
To further analyze the mechanism of the influence of nano-Al2O3 on the thermal stability of the oil-paper insulation, inductively coupled plasma mass spectrometry (ICAP-QC, Thermo Scientific Co., Ltd., Waltham, MA, USA) was used to quantitatively measure the copper content of the insulating paper and the insulating oil.
As shown in Figure 9, the plain paper can also adsorb part of the copper compound during the aging process, but it gradually became saturated as the aging process progressed. In comparison, the insulating paper doped with nano-Al2O3 displayed a stronger ability to adsorb copper, and the copper compounds absorbed by the modified paper at the early stage of aging were significantly greater than that by the plain paper. At the end of aging, the adsorption of copper compounds by the nano-Al2O3 modified paper was thrice that of the plain paper.
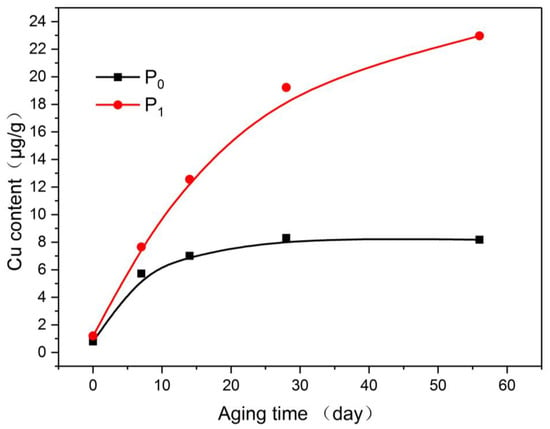
Figure 9.
Variation of copper content in paper with aging process.
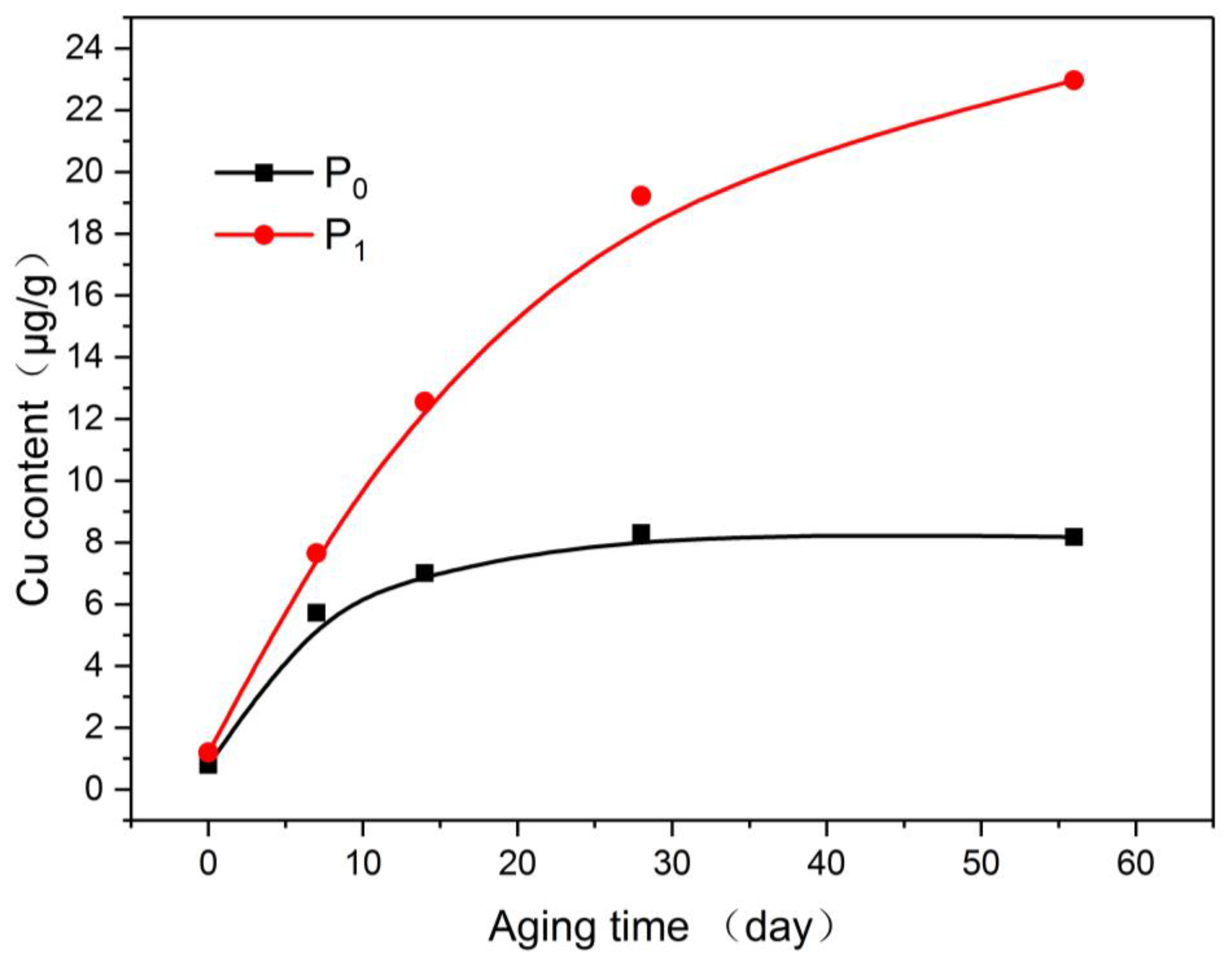
In Figure 10, the copper content in the insulating oil was consistent with the copper content in the insulating paper. The copper content of O0 increased throughout the aging period. The copper in O1 was at a very low levels during the initial period of aging and gradually increased after the middle of aging, although it was still much smaller than that of O0.
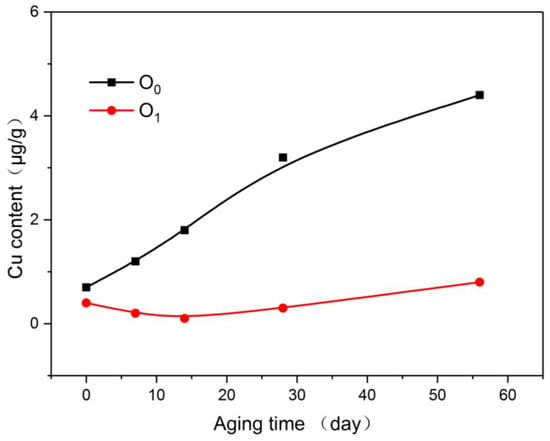
Figure 10.
Variation of copper content in oil with aging process.
The delaying mechanism of the thermal aging rate of the oil-paper insulation is shown in Figure 11. First, the nano-Al2O3 surface atoms can be combined with other metal ions by electrostatic interaction, which means nano-Al2O3 has a strong adsorption capacity for many transition metal ions, and it can achieve adsorption equilibrium in a short time. Different temperatures and pH value pairs had an insignificant effect on the nano-Al2O3 copper ion adsorption capacity [25]. In the aging process, the small molecule acid produced by the aging of the insulating paper, and the peroxide generated from the aging of the insulating oil promoted the formation of copper compounds and the aging of the insulating oil. The insulating paper doped with nano-Al2O3 adsorbed more copper compounds, delayed the aging of the insulating oil, reduced the aging products of the insulating oil, and neutralized small molecular acids, thereby retarding the aging of the insulating paper and the insulating oil.
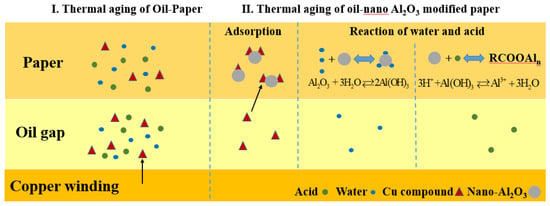
Figure 11.
Influence of nano-Al2O3 on thermal aging of insulating paper and oil.
Moreover, through nano-Al2O3 doping, the voids of the composite insulating paper fibers were filled with nano-Al2O3 containing hydroxyl groups. The hydroxyl group on the surface of nano-Al2O3 had a strong ability to adsorb water and neutralize the consumption of small molecules, such as formic acid, as defined in Equations (1) and (2):
In view of the two reversible reactions, some water and acidic products did not participate in the aging reaction of insulating paper and the insulating oil, thereby reducing their aging rates.
Therefore, the nano-Al2O3, filled in the insulating paper, can adsorb copper compounds and keep part of the water and acid products away from the thermal aging process. Under such double action, the aging of the oil-paper insulation can be delayed.
4. Conclusions
This work investigated the thermal aging characteristics of oil-impregnated composite insulating paper and oil-impregnated general insulating paper at 130 °C. The DP, tensile strength, and copper of the insulating paper were measured, whereas various parameters, such as color, acid value, breakdown voltage, dielectric loss, and copper content in the oil were compared and analyzed to arrive at the following conclusions:
- In the papermaking process, 2 wt% nano-Al2O3 was doped into the insulating paper to prepare composite insulating paper. Its DP and tensile strength were always superior to those of the plain paper in the thermal aging process, indicating that the aging decomposition rate of the nano-Al2O3 modified insulating paper was significantly lower than that of the plain paper.
- The testing of the insulating oil revealed the color, acidity value, moisture, breakdown voltage, and dielectric loss of the insulating oil of the modified paper were better than those of the oil with the plain paper, indicating that the nano-Al2O3 modified insulating paper can significantly slow down the aging rate of the insulating oil.
- Nano-Al2O3 in the modified paper can effectively absorb copper compounds in the insulating oil to delay the aging of the oil and reduction of the aging product will also decrease the aging rate of the insulating paper. Simultaneously, Nano-Al2O3 in the modified paper can adsorb water molecules and small molecule acids, and inhibit the dissolution of the small molecules of acid in the composite insulating paper to delay the aging of the insulating paper and the insulating oil. In summary, the Al2O3-modified insulating paper has a good thermal stability and can delay the aging rate of the insulating oil.
Author Contributions
This article has five authors: N.L., R.L., Y.Y., M.X., Y.M. N.L. proposed the conception and wrote the manuscript; R.L. and Y.Y. gave their modification suggestions for the manuscript; M.X. and Y.M. carried out the experiment.
Funding
This research was supported by Nation Natural Science Foundation of China under Grant No.51437001.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Wu, Y.; Zhu, H.; Zhou, Z.; Li, X.; Xiong, H. Transformer condition assessment based on entropy fuzzy matter-element and principal component analysis. Power Syst. Protect. Control 2015, 43, 1–7. [Google Scholar]
- Liao, R.; Liu, T.; Zhang, F.; Yuan, Y.; Wu, W. Preparation and thermal aging properties of montmorillonite modified thermally-upgraded paper. Trans. China Electrotech. Soc. 2015, 30, 220–227. [Google Scholar]
- Li, S.; Lyons-hart, J.; Banyasz, J.; Shafer, K. Real-time evolved gas analysis by FTIR method: An experimental study of cellulose pyrolysis. Fuel 2001, 80, 1809–1817. [Google Scholar] [CrossRef]
- Lundgaard, L.E.; Hansen, W.; Linhjell, D.; Painter, T.J. Aging of oil-impregnated paper in power transformers. IEEE Trans. Power Deliv. 2004, 19, 230–239. [Google Scholar] [CrossRef]
- Ese, M.H.G.; Liland, K.B.; Lundgaard, L.E. Oxidation of paper insulation in transformers. IEEE Trans. Dielectr. Electr. Insul. 2010, 17, 939–946. [Google Scholar] [CrossRef]
- Lampe, W.; Spicar, E. The Oxygen-free Transformer: Reduced ageing by continuous degassing. Cigre Pap. 1976, 12, 17. [Google Scholar]
- Beavers, M.; Raab, E.; Raab, L.; Leslie, J.C. Permalex, a new insulation system. Power Appar. Syst. III Trans. Am. Inst. Electr. Eng. 1960, 79, 64–70. [Google Scholar] [CrossRef]
- Brummet, B.; Sadler, F. A new transformer insulation. Power Appar. Syst. III Trans. Am. Inst. Electr. Eng. 1962, 81, 754–759. [Google Scholar] [CrossRef]
- Liao, R.; Wu, W.; Nie, S.; Liu, T.; Lü, C.; Zhang, S. Study on the aynergistic anti-aging effect of composite thermal stabilizers on the insulation paper by molecular modeling. Trans. China Electrotech. Soc. 2015, 30, 330–337. [Google Scholar]
- Lewis, T.J. Nanometric dielectrics. IEEE Trans. Dielectr. Electr. Insul. 1994, 1, 812–825. [Google Scholar] [CrossRef]
- Zhang, L.; Liu, Q.; Sun, X. Research progress on free radical polymerization modification of nanoparticles. Chem. World 2012, 53, 381–384. [Google Scholar]
- Sang, S.; Yang, R. Prospects of nanotechnology. China New Technol. Prod. 2012, 6, 1. [Google Scholar]
- Nelson, J.K.; Fothergill, J.C.; Dissado, L.A.; Peasgood, W. Toward an Understanding of Nanometric Dielectrics. In Proceedings of the 2002 Annual Report Conference on Electrical Insulation and Dielectric Phenomena, Cancun, Mexico, 20–24 October 2002; Volume 1, pp. 295–298. [Google Scholar] [CrossRef]
- Murakami, Y.; Nemoto, M.; Okuzumi, S.; Masuda, S. DC conduction and electrical breakdown of MgO/LDPE nanocomposite. IEEE Trans. Dielectr. Electr. Insul. 2008, 15, 33–39. [Google Scholar] [CrossRef]
- Wang, X.; Cheng, X.; Chen, S.Q.; Zheng, X.Q.; Tu, D.M. Dielectric properties of the composites of nano-ZnO/low-density polyethylene. Proc. CSEE 2008, 28, 13–19. [Google Scholar]
- Li, S.; Yin, G.; Bai, S.; Li, J. A new potential barrier model in epoxy resin nanodielectrics. IEEE Trans. Dielectr. Electr. Insul. 2011, 18, 1535–1543. [Google Scholar] [CrossRef]
- Yang, W.; Yi, R.; Yang, X.; Xu, M. Effect of Particle Size and Dispersion on Dielectric Properties in ZnO/Epoxy Resin Composites. Trans. Electr. Electron. Mater. 2012, 13, 116–120. [Google Scholar] [CrossRef]
- Lundgaard, L.E.; Hansen, W.; Ingebrigtsen, S. Ageing of mineral oil impregnated cellulose by acid catalysis. IEEE Trans. Dielectr. Electr. Insul. 2008, 15, 540–546. [Google Scholar] [CrossRef]
- Tian, F.; Yang, C.; He, L.; Han, B.; Wang, Y. Recent research advancement in dielectric properties and the corresponding mechanism of polymer/inorganic nanocomposite. Trans. China Electrotech. Soc. 2011, 26, 1–12. [Google Scholar]
- Li, H.; Guo, L.; Liu, B.; Chen, W.; Chen, S.T. The dielectic properties of polyimide/nano-Al2O3 composites films. Proc. CSEE 2006, 26, 166–170. [Google Scholar]
- Velasco, J.; Frascella, R.; Albarracín, R.; Burgos, J.C.; Dong, M.; Ren, M.; Yang, L. Comparison of Positive Streamers in Liquid Dielectrics with and without Nanoparticles Simulated with Finite-Element Software. Energies 2018, 11. [Google Scholar] [CrossRef]
- He, L.; Liao, R.; Lv, Y.; Yang, L.; Zhao, X.; Yan, S. Effect of nano-Al2O3 on the thermal aging physicochemical properties of insulating paper. In Proceedings of the International Conference on Condition Monitoring and Diagnosis, Xi’an, China, 25–28 September 2016; pp. 254–257. [Google Scholar] [CrossRef]
- Zhou, X. Study on the Change of Molecular Weight and the Effect of Copper Compound on Aging of Oil-Impregnated Insulation Paper during Accelerated Thermal Aging. Master’s Thesis, Chongqing University, Chongqing, China, 2010. [Google Scholar]
- Emsley, A.; Xiao, X.; Heywood, R.; Ali, M. Degradation of cellulosic insulation in power transformers. Part 3: Effects of oxygen and water on ageing in oil. IET 2000, 147, 115–119. [Google Scholar] [CrossRef]
- Chang, G.; Jiang, Z.; Peng, T.; Hu, B. Preparation of High-Specific-Surface-Area Nanometer-sized Alumina by Sol-Gel Method and Study on Adsorption Behaviors of Transition Metal Ions on the Alumina Powder with ICP-AES. Acta Chim. Sin. 2003, 61, 100–103. [Google Scholar]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).