Using a Weekly Patient-Reported Outcome Questionnaire to Track Acute Toxicity in Patients Undergoing Pelvic Radiotherapy for Gynecologic Cancers
Abstract
:1. Introduction
2. Materials and Methods
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kotronoulas, G.; Kearney, N.; Maguire, R.; Harrow, A.; Di Domenico, D.; Croy, S.; MacGillivray, S. What is the value of the routine use of patient-reported outcome measures toward improvement of patient outcomes, processes of care, and health service outcomes in cancer care? A systematic review of controlled trials. J. Clin. Oncol. 2014, 32, 1480–1501. [Google Scholar] [CrossRef] [PubMed]
- Velikova, G.; Booth, L.; Smith, A.B.; Brown, P.M.; Lynch, P.; Brown, J.M.; Selby, P.J. Measuring quality of life in routine oncology practice improves communication and patient well-being: A randomized controlled trial. J. Clin. Oncol. 2004, 22, 714–724. [Google Scholar] [CrossRef] [PubMed]
- Basch, E.; Barbera, L.; Kerrigan, C.L.; Velikova, G. Implementation of Patient-Reported Outcomes in Routine Medical Care. Am. Soc. Clin. Oncol. Educ. Book 2018, 38, 122–134. [Google Scholar] [CrossRef] [PubMed]
- Olson, R.A.; Howard, F.; Lapointe, V.; Schellenberg, D.; Nichol, A.; Bowering, G.; Curtis, S.; Walter, A.; Brown, S.; Thompson, C.; et al. Provincial development of a patient-reported outcome initiative to guide patient care, quality improvement, and research. Healthc. Manag. Forum. 2018, 31, 13–17. [Google Scholar] [CrossRef] [Green Version]
- Barbera, L.; Olson, R.; Bird, L.; Brown, E.; Brundage, M.; Caissie, A.; Davis, C.A.; Milosevic, M.; O’Donnell, J. Canadian Partnership for Quality Radiotherapy: Guidance on the Use of Patient Reported Outcomes for Canadian Radiation Treatment Programs. Available online: http://www.pcqr.ca/wp-content/uploads//2020/09/PRO.2020.09.01.pdf (accessed on 28 April 2022).
- Nout, R.A.; Smit, V.T.; Putter, H.; Jurgenliemk-Schulz, I.M.; Jobsen, J.J.; Lutgens, L.C.; Van der Steen-Banasik, E.M.; Mens, J.W.; Slot, A.; Kroese, M.C.; et al. PORTEC Study Group Vaginal brachytherapy versus pelvic external beam radiotherapy for patients with endometrial cancer of high-intermediate risk (PORTEC-2): An open-label, non-inferiority, randomised trial. Lancet 2010, 375, 816–823. [Google Scholar] [CrossRef]
- ASTEC/EN 5 Study Group; Blake, P.; Swart, A.M.; Orton, J.; Kitchener, H.; Whelan, T.; Lukka, H.; Eisenhauer, E.; Bacon, M.; Tu, D.; et al. Adjuvant external beam radiotherapy in the treatment of endometrial cancer (MRC ASTEC and NCIC CTG EN.5 randomised trials): Pooled trial results, systematic review, and meta-analysis. Lancet 2009, 373, 137–146. [Google Scholar] [CrossRef] [Green Version]
- Johnson, T.P.; Wislar, J.S. Response rates and nonresponse errors in surveys. JAMA 2012, 307, 1805–1806. [Google Scholar] [CrossRef]
- Flores, L.T.; Bennett, A.V.; Law, E.B.; Hajj, C.; Griffith, M.P.; Goodman, K.A. Patient-Reported Outcomes vs. Clinician Symptom Reporting During Chemoradiation for Rectal Cancer. Gastrointest. Cancer Res. 2012, 5, 119–124. [Google Scholar]
- Fromme, E.K.; Eilers, K.M.; Mori, M.; Hsieh, Y.C.; Beer, T.M. How accurate is clinician reporting of chemotherapy adverse effects? A comparison with patient-reported symptoms from the Quality-of-Life Questionnaire C30. J. Clin. Oncol. 2004, 22, 3485–3490. [Google Scholar] [CrossRef]
- Klopp, A.H.; Yeung, A.R.; Deshmukh, S.; Gil, K.M.; Wenzel, L.; Westin, S.N.; Gifford, K.; Gaffney, D.K.; Small, W.; Thompson, S.; et al. Patient-Reported Toxicity During Pelvic Intensity-Modulated Radiation Therapy: NRG Oncology-RTOG 1203. J. Clin. Oncol. 2018, 36, 2538–2544. [Google Scholar] [CrossRef]
- Dröge, L.H.; Von Sivers, F.F.; Schirmer, M.A.; Wolff, H.A. Conventional 3D conformal radiotherapy and volumetric modulated arc therapy for cervical cancer: Comparison of clinical results with special consideration of the influence of patient- and treatment-related parameters. Strahlenther. Onkol. 2021, 197, 520–527. [Google Scholar] [CrossRef]
- Vandecasteele, K.; Makar, A.; Van den Broecke, R.; Delrue, L.; Denys, H.; Lambein, K.; Lambert, B.; Van Eijkeren, M.; Tummers, P.; De Meerleer, G. Intensity-modulated arc therapy with cisplatin as neo-adjuvant treatment for primary irresectable cervical cancer. Toxicity, tumour response and outcome. Strahlenther. Onkol. 2012, 188, 576–581. [Google Scholar] [CrossRef]
- Chen, M.F.; Tseng, C.J.; Tseng, C.C.; Kuo, Y.C.; Yu, C.Y.; Chen, W.C. Clinical outcome in posthysterectomy cervical cancer patients treated with concurrent Cisplatin and intensity-modulated pelvic radiotherapy: Comparison with conventional radiotherapy. Int. J. Radiat. Oncol. Biol. Phys. 2007, 67, 1438–1444. [Google Scholar] [CrossRef]
- Kirwan, J.M.; Symonds, P.; Green, J.A.; Tierney, J.; Collingwood, M.; Williams, C.J. A systematic review of acute and late toxicity of concomitant chemoradiation for cervical cancer. Radiother. Oncol. 2003, 68, 217–226. [Google Scholar] [CrossRef]
- Wortman, B.G.; Post, C.C.B.; Powell, M.E.; Khaw, P.; Fyles, A.; D’Amico, R.; Haie-Meder, C.; Jürgenliemk-Schulz, I.M.; McCormack, M.; Do, V.; et al. Radiation Therapy Techniques and Treatment-Related Toxicity in the PORTEC-3 Trial: Comparison of 3-Dimensional Conformal Radiation Therapy Versus Intensity-Modulated Radiation Therapy. Int. J. Radiat. Oncol. Biol. Phys. 2022, 112, 390–399. [Google Scholar] [CrossRef]
- Gandhi, A.K.; Sharma, D.N.; Rath, G.K.; Julka, P.K.; Subramani, V.; Sharma, S.; Manigandan, D.; Laviraj, M.A.; Kumar, S.; Thulkar, S. Early clinical outcomes and toxicity of intensity modulated versus conventional pelvic radiation therapy for locally advanced cervix carcinoma: A prospective randomized study. Int. J. Radiat. Oncol. Biol. Phys. 2013, 87, 542–548. [Google Scholar] [CrossRef]
- Reijtenbagh, D.M.W.; Godart, J.; Mens, J.M.; Heijkoop, S.T.; Heemsbergen, W.D.; Hoogeman, M.S. Patient-reported acute GI symptoms in locally advanced cervical cancer patients correlate with rectal dose. Radiother. Oncol. 2020, 148, 38–43. [Google Scholar] [CrossRef]
- Howell, D.; Molloy, S.; Wilkinson, K.; Green, E.; Orchard, K.; Wang, K.; Liberty, J. Patient-reported outcomes in routine cancer clinical practice: A scoping review of use, impact on health outcomes, and implementation factors. Ann. Oncol. 2015, 26, 1846–1858. [Google Scholar] [CrossRef]
- Abbott-Anderson, K.; Kwekkeboom, K.L. A systematic review of sexual concerns reported by gynecological cancer survivors. Gynecol. Oncol. 2012, 124, 477–489. [Google Scholar] [CrossRef]
- Barker, C.L.; Routledge, J.A.; Farnell, D.J.; Swindell, R.; Davidson, S.E. The impact of radiotherapy late effects on quality of life in gynaecological cancer patients. Br. J. Cancer 2009, 100, 1558–1565. [Google Scholar] [CrossRef] [Green Version]
- Grover, S.; Hill-Kayser, C.E.; Vachani, C.; Hampshire, M.K.; DiLullo, G.A.; Metz, J.M. Patient reported late effects of gynecological cancer treatment. Gynecol. Oncol. 2012, 124, 399–403. [Google Scholar] [CrossRef]
- Msaouel, P.; Oromendia, C.; Siefker-Radtke, A.O.; Tannir, N.M.; Subudhi, S.K.; Gao, J.; Wang, Y.; Siddiqui, B.A.; Shah, A.Y.; Aparicio, A.M.; et al. Evaluation of Technology-Enabled Monitoring of Patient-Reported Outcomes to Detect and Treat Toxic Effects Linked to Immune Checkpoint Inhibitors. JAMA Netw. Open 2021, 4, e2122998. [Google Scholar] [CrossRef]
- Lapen, K.; Sabol, C.; Tin, A.L.; Lynch, K.; Kassa, A.; Mabli, X.; Ford, J.; Cha, E.; Bernstein, M.B.; Braunstein, L.Z.; et al. Development and Pilot Implementation of a Remote Monitoring System for Acute Toxicity Using Electronic Patient-Reported Outcomes for Patients Undergoing Radiation Therapy for Breast Cancer. Int. J. Radiat. Oncol. Biol. Phys. 2021, 111, 979–991. [Google Scholar] [CrossRef]
- Møller, P.K.; Pappot, H.; Bernchou, U.; Schytte, T.; Dieperink, K.B. Development of patient-reported outcomes item set to evaluate acute treatment toxicity to pelvic online magnetic resonance-guided radiotherapy. J. Patient Rep. Outcomes 2021, 5, 47. [Google Scholar] [CrossRef] [PubMed]
| Variable | (n = 698) | |
|---|---|---|
| Age (years) | <50 50–69 ≥70 | 22% 55% 23% |
| Intent | Radical Post-op | 42% 58% |
| Primary tumor | Endometrial Cervical Ovarian Vulvar Vaginal Unknown | 50% 33% 9% 5% 3% 0.1% |
| Concurrent chemotherapy | No Yes | 65% 35% |
| Brachytherapy | None Intracavitary Interstitial Vaginal Vault | 59% 27% 0.7% 14% |
| Radiation Field | Local Only Whole Pelvis ± Boost | 3% 97% |
| Total Dose (Gy) | <45 45–50 >50 | 0.1% 76% 24% |
| Whole Pelvic Field Dose (Gy) | <45 45 45.1–50 | 0.4% 95% 4% |
| Whole Pelvic Field Dose Per Fraction (Gy) | 1.5–1.79 1.8–2 | 4% 96% |
| Technique | 3D-CRT IMRT/VMAT | 25% 75% |
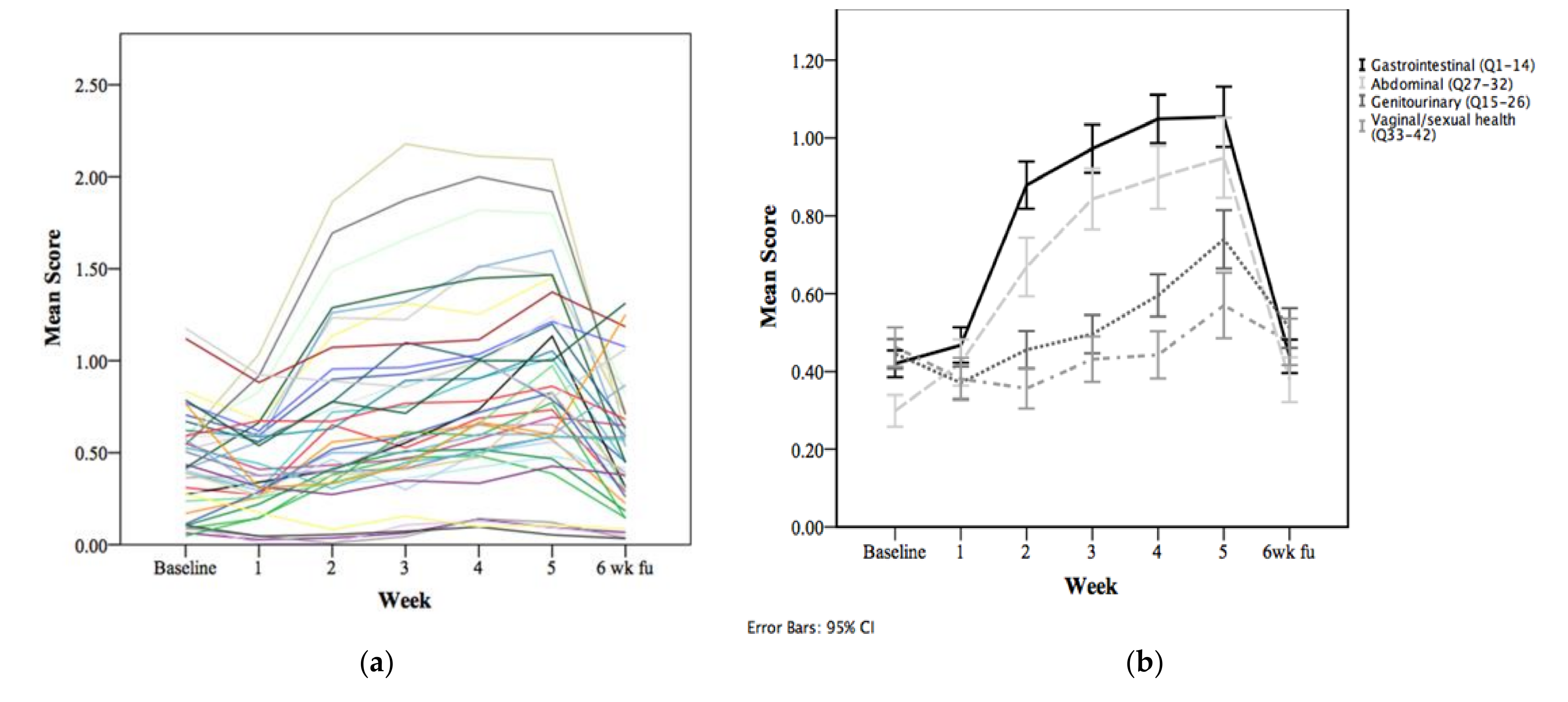
| Question | Mean Score (Δ Compared to Baseline) | ||||||
|---|---|---|---|---|---|---|---|
| Baseline | Wk1 | Wk2 | Wk3 | Wk4 | Wk5 | 6 Wk Fu | |
| Section 1–Bowel Functions | |||||||
| 1. How often have you had rectal urgency (felt like you had to pass stool, but did not) during the last 7 days? | 0.68 | 0.71 (+4%) | 0.74 (+9%) | 0.97 (+43%) | 0.92 (+35%) | 0.94 (+38%) | 0.49 (−28%) |
| 2. How often have you had uncontrolled leakage of stool or feces during the last 7 days? | 0.14 | 0.10 (−29%) | 0.25 (+79%) | 0.49 (+250%) | 0.43 (+207%) | 0.40 (+186%) | 0.23 (+64%) |
| 3. How often have you had stools (bowel movements) that were loose or liquid (no form, watery, mushy) during the last 7 days? | 0.58 | 0.59 (+2%) | 1.18 (+103%) | 1.92 (+231%) | 1.93 (+233%) | 1.93 (+233%) | 0.70 (+21%) |
| 4. How often have you had bloody stools during the last 7 days? | 0.07 | 0.04 (−43%) | 0.05 (−29%) | 0.08 (+14%) | 0.13 (+86%) | 0.17 (+143%) | 0.08 (+14%) |
| 5. How often have you had crampy pain in your abdomen, pelvis, or rectum during the last 7 days? | 0.81 | 0.64 (−21%) | 0.80 (−1%) | 1.10 (+36%) | 1.19 (+47%) | 1.22 (+51%) | 0.74 (−9%) |
| 6. How many bowel movements have you had on a typical day during the last 7 days? | 0.23 | 0.24 (+4%) | 0.37 (+61%) | 0.58 (+152%) | 0.56 (+143%) | 0.64 (+178%) | 0.32 (+39%) |
| 7. How often have your bowel movements been painful during the last 7 days? | 0.39 | 0.36 (−8%) | 0.37 (−5%) | 0.64 (+64%) | 0.81 (+108%) | 0.87 (+123%) | 0.46 (+18%) |
| How big a problem, if any, has each of the following been for you during the last 7 days? | |||||||
| 8. Urgency to have a bowel movement | 0.58 | 0.54 (−7%) | 0.82 (+41%) | 1.29 (+122%) | 1.22 (+110%) | 1.30 (+124%) | 0.76 (+31%) |
| 9. Increased frequency of bowel movements | 0.37 | 0.38 (+3%) | 0.83 (+124%) | 1.33 (+259%) | 1.39 (+276%) | 1.34 (+262%) | 0.56 (+51%) |
| 10. Watery bowel movements | 0.42 | 0.38 (−10%) | 0.78 (+86%) | 1.40 (+233%) | 1.33 (+217%) | 1.34 (+219%) | 0.54 (+29%) |
| 11. Losing control of your stools | 0.19 | 0.13 (−32%) | 0.31 (+63%) | 0.58 (+205%) | 0.56 (+195%) | 0.54 (+184%) | 0.33 (+74%) |
| 12. Bloody stools | 0.10 | 0.07 (−30%) | 0.06 (−40%) | 0.07 (−30%) | 0.11 (+10%) | 0.15 (+50%) | 0.10 (0%) |
| 13. Abdominal/Pelvic/Rectal Pain | 0.72 | 0.58(−19%) | 0.66(−8%) | 0.87 (+21%) | 0.98 (+36%) | 1.00 (+39%) | 0.71 (−1%) |
| 14. Overall, how big a problem have your bowel habits been for you during the last 7 days? | 0.64 | 0.75 (+17%) | 1.05 (+64%) | 1.54 (+141%) | 1.59 (+148%) | 1.64 (+156%) | 0.82 (+28%) |
| Section 2–Urinary Functions | |||||||
| 15. Over the past 7 days, how often have you leaked urine? | 0.69 | 0.54 (−22%) | 0.52 (−25%) | 0.47 (−32%) | 0.53 (−23%) | 0.56 (−19%) | 0.66 (−4%) |
| 16. Over the past 7 days, how often have you urinated blood? | 0.12 | 0.08 (−33%) | 0.05 (−58%) | 0.11 (−8%) | 0.06 (−50%) | 0.10 (−17%) | 0.03 (−75%) |
| 17. Over the past 7 days, how often have you had pain or burning with urination? | 0.24 | 0.15 (−37%) | 0.27 (+13%) | 0.48 (+100%) | 0.68 (+183%) | 0.95 (+296%) | 0.33 (+38%) |
| 18. Which of the following best describes your urinary control during the last 7 days? | 0.51 | 0.36 (−29%) | 0.36 (−29%) | 0.38 (−25%) | 0.43 (−16%) | 0.44 (−14%) | 0.49 (−4%) |
| 19. How many pads or adult diapers per day did you usually use to control leakage during the last 7 days? | 0.39 | 0.37 (−5%) | 0.39 (0%) | 0.40 (+3%) | 0.48 (+23%) | 0.47 (+21%) | 0.40 (+3%) |
| How big a problem, if any, has each of the following been for you during the last 7 days? | |||||||
| 20. Dripping or leaking urine | 0.63 | 0.49 (−22%) | 0.47 (−25%) | 0.45 (−29%) | 0.47 (−25%) | 0.52 (−17%) | 0.60 (−5%) |
| 21. Pain or burning on urination | 0.22 | 0.17 (−23%) | 0.25 (+14%) | 0.40 (+82%) | 0.59 (+168%) | 0.76 (+245%) | 0.32 (+45%) |
| 22. Bleeding with urination | 0.14 | 0.09 (−36%) | 0.05 (−64%) | 0.11 (−21%) | 0.07 (−50%) | 0.09 (−36%) | 0.04 (−71%) |
| 23. Weak urine stream or incomplete emptying | 0.43 | 0.34 (−21%) | 0.33 (−23%) | 0.40 (−7%) | 0.46 (+7%) | 0.51 (+19%) | 0.36(−16%) |
| 24. Waking up to urinate | 1.19 | 0.98 (−18%) | 1.08 (−9%) | 1.13 (−5%) | 1.15 (−3%) | 1.31 (+10%) | 1.20 (+1%) |
| 25. Need to urinate frequently during the day | 0.83 | 0.70 (−16%) | 0.81 (−2%) | 0.92 (+11%) | 0.97 (+17%) | 1.11 (+34%) | 0.92 (+11%) |
| 26. Overall, how big a problem have your urinary function been for you during the last 7 days? | 0.72 | 0.55 (−24%) | 0.68 (−6%) | 0.72 (0%) | 0.92 (+28%) | 1.04 (+44%) | 0.82 (+14%) |
| Section 3–Abdominal Problems | |||||||
| 27. In the last 7 days, what was the severity of your pain in the abdomen (belly area) at its worst? | 0.64 | 0.56 (−12%) | 0.62 (−3%) | 0.73 (+14%) | 0.85 (+33%) | 0.89 (+39%) | 0.61 (−5%) |
| 28. In the last 7 days, how much did pain in the abdomen (belly area) interfere with your usual or daily activities? | 0.45 | 0.39 (−13%) | 0.40 (−11%) | 0.50 (+11%) | 0.55 (+22%) | 0.64 (+42%) | 0.43 (−4%) |
| 29. In the last 7 days, how often did you have loose or watery stools (diarrhea)? | 0.55 | 0.50 (−9%) | 1.07 (+95%) | 1.70 (+209%) | 1.76 (+220%) | 1.81 (+229%) | 0.78 (+42%) |
| 30. In the last 7 days, how often did you lose control of bowel movements? | 0.14 | 0.13 (−7%) | 0.25 (+79%) | 0.48 (+243%) | 0.50 (+257%) | 0.45 (+221%) | 0.26 (+86%) |
| 31. In the last 7 days, how much did loss of control of bowel movements interfere with your usual or daily activities | 0.14 | 0.14 (0%) | 0.27 (+93%) | 0.58 (+314%) | 0.66 (+371%) | 0.64 (+357%) | 0.30 (+114%) |
| 32. In the last 7 days, how often on average have you taken an anti-diarrhea medication? | 0.06 | 0.07 (+17%) | 0.16 (+167%) | 0.49 (+717%) | 0.70 (+1067) | 0.74 (+1133) | 0.16 (+167%) |
| Section 4–Gynecologic Problems | |||||||
| 33. Have you had irritation or soreness in your vagina or vulva during the past 4 weeks? | 0.40 | 0.26 (−35%) | 0.28 (−30%) | 0.40 (0%) | 0.52 (+30%) | 0.59 (+48%) | 0.44 (+10%) |
| 34. Have you had discharge from your vagina during the past 4 weeks? | 0.57 | 0.50 (−12%) | 0.39 (−32%) | 0.35 (−39%) | 0.40 (−30%)) | 0.41 (−28%) | 0.42 (−26%) |
| 35. Have you had abnormal bleeding from your vagina during the past 4 weeks? | 0.39 | 0.24 (−38%) | 0.16 (−59%) | 0.17 (−56%) | 0.15 (−62%) | 0.14 (−64%) | 0.08 (−79%) |
| 36. Have you felt dissatisfied with your body during the past 4 weeks? | 0.68 | 0.61 (−10%) | 0.61 (−10%) | 0.71 (+4%) | 0.74 (+9%) | 0.78 (+15%) | 0.69 (+1%) |
| 37. Have you worried that sex would be painful during the past 4 weeks? | 0.53 | 0.48 (−9%) | 0.46 (−13%) | 0.39 (−26%) | 0.48 (−9%) | 0.52 (−2%) | 0.68 (+28%) |
| 38. Have you been sexually active during the past 4 weeks? | 0.18 | 0.25 (+39%) | 0.23 (+28%) | 0.18 (0%) | 0.18 (0%) | 0.16 (−11%) | 0.29 (+61%) |
| 39. Has your vagina felt dry during sexual activity during the past 4 weeks? | 0.99 | 0.87 (−12%) | 0.89 (−10%) | 0.91 (−8%) | 0.96 (−3%) | 0.92 (−7%) | 1.04 (+5%) |
| 40. Has your vagina felt short during the past 4 weeks? | 0.45 | 0.31 (−31%) | 0.40 (−11%) | 0.42 (−7%) | 0.56 (+24%) | 0.57 (+27%) | 0.88 (+96%) |
| 41. Has your vagina felt tight during the past 4 weeks? | 0.53 | 0.63 (+19%) | 0.66 (+25%) | 0.84 (+58%) | 0.77 (+45%) | 0.79 (+49%) | 1.10 (+108%) |
| 42. Have you had pain during sexual intercourse or other sexual activity during the past 4 weeks? | 0.65 | 0.48 (−26%) | 0.34 (−48%) | 0.43 (−34%) | 0.54 (−17%) | 0.67 (+3%) | 0.95 (+46%) |
| 43. Was sexual activity enjoyable for you during the past 4 weeks? | 1.91 | 1.85 (−3%) | 1.91 (0%) | 1.82 (−5%) | 1.87 (−2%) | 1.79 (−6%) | 1.40 (−27%) |
| Sub-Question Toxicity 1 | |||||||
|---|---|---|---|---|---|---|---|
| Positive | Negative | Sensitivity | Specificity | PPV | NPV | ||
| GI summary question (#14) * | Positive | 1957 | 33 | 69.2% | 93.2% | 98.3% | 34.4% |
| Negative | 869 | 455 | |||||
| GU summary question (#26) + | Positive | 1472 | 2 | 56.4% | 99.7% | 99.9% | 36.8% |
| Negative | 1138 | 663 | |||||
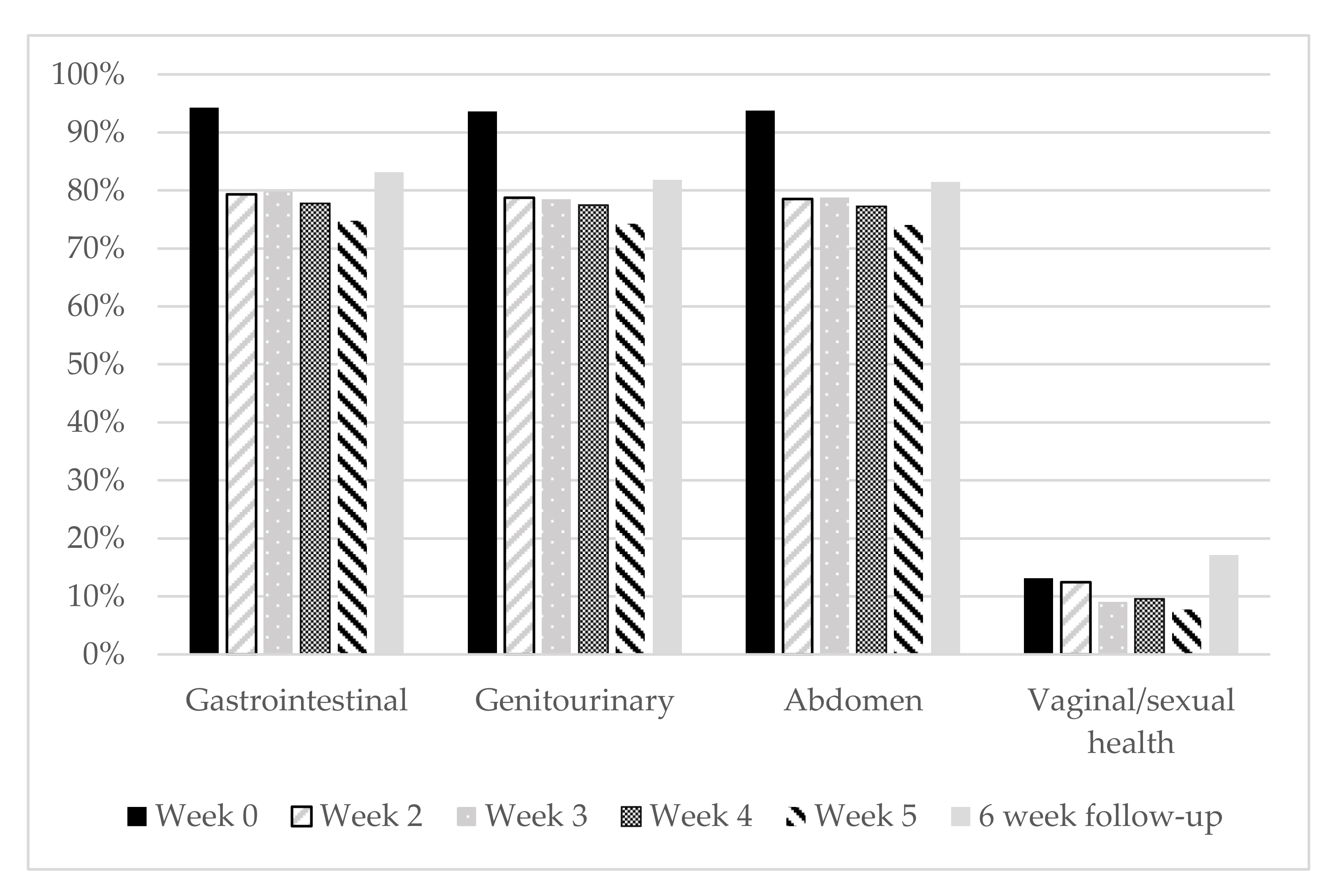
| Variable | Week 5 Score of 4 (Total Cases) | p-Value | |
|---|---|---|---|
| Technique | 4 Field IMRT VMAT | 11.6% (112) 10.0% (20) 5.5% (363) | 0.06 |
| Primary Site | Cervix Endometrium Ovary Vagina Vulva Other | 7.4% (163) 7.4% (242) 4.6% (44) 7.1% (14) 4.6% (22) 9.1% (11) | 0.98 |
| Concurrent Chemo | No Yes | 6.6% (333) 8.0% (163) | 0.58 |
| Brachytherapy | No Yes | 7.6% (303) 6.2% (193) | 0.60 |
| Total Dose | Mean (cGy) | 4830.6 (4814.6 *) | 0.88 |
| Variable | Week 5 Score of 4 (Total Cases) | p-Value | |
|---|---|---|---|
| Technique | 4 Field IMRT VMAT | 4.5% (111) 0% (20) 2.0% (357) | 0.28 |
| Primary Site | Cervix Endometrium Ovary Vagina Vulva Other | 10.1% (79) 4.5% (157) 0.8% (242) 0% (11) 15.4% (13) 0% (44) | 0.01 |
| Concurrent Chemo | No Yes | 1.5% (331) 4.4% (158) | 0.06 |
| Brachytherapy | No Yes | 2.7% (299) 2.1% (190) | 0.77 |
| Total Dose | Mean (cGy) | 4923.3 (4810.4 *) | 0.52 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chan, M.; Olson, R.; Lapointe, V.; Hamm, J.; Bachand, F.; Holloway, C.; Parsons, C.; Lim, P. Using a Weekly Patient-Reported Outcome Questionnaire to Track Acute Toxicity in Patients Undergoing Pelvic Radiotherapy for Gynecologic Cancers. Curr. Oncol. 2022, 29, 3306-3317. https://doi.org/10.3390/curroncol29050270
Chan M, Olson R, Lapointe V, Hamm J, Bachand F, Holloway C, Parsons C, Lim P. Using a Weekly Patient-Reported Outcome Questionnaire to Track Acute Toxicity in Patients Undergoing Pelvic Radiotherapy for Gynecologic Cancers. Current Oncology. 2022; 29(5):3306-3317. https://doi.org/10.3390/curroncol29050270
Chicago/Turabian StyleChan, Matthew, Robert Olson, Vincent Lapointe, Jeremy Hamm, Francois Bachand, Caroline Holloway, Christina Parsons, and Peter Lim. 2022. "Using a Weekly Patient-Reported Outcome Questionnaire to Track Acute Toxicity in Patients Undergoing Pelvic Radiotherapy for Gynecologic Cancers" Current Oncology 29, no. 5: 3306-3317. https://doi.org/10.3390/curroncol29050270
APA StyleChan, M., Olson, R., Lapointe, V., Hamm, J., Bachand, F., Holloway, C., Parsons, C., & Lim, P. (2022). Using a Weekly Patient-Reported Outcome Questionnaire to Track Acute Toxicity in Patients Undergoing Pelvic Radiotherapy for Gynecologic Cancers. Current Oncology, 29(5), 3306-3317. https://doi.org/10.3390/curroncol29050270






