1. Introduction
Lung cancer incidence and mortality rates continue to rank among the highest of all cancer types in the United States [
1]. Pulmonary nodule size and growth rate are known risk factors for lung cancer, [
2] with nodules ≥ 8 mm posing a higher likelihood of malignancy [
3]. Concerning scans typically warrant tissue sampling to assist with initial diagnosis [
4]. The most accessible sites are usually chosen for biopsy, [
5] as different approaches carry different yields [
6]. As it is not uncommon for tissue samples to be classified as having atypical cellularity, repeat biopsies or surgical interventions are further recommended [
7]. It remains unclear, however, how often a cytologic diagnosis of atypia is ultimately found to be a malignancy, and in what time frame this lung cancer diagnosis occurs. The purpose of this study was to evaluate the duration of time and potential risk factors associated with the development of lung cancer among patients with an initial diagnosis of atypia.
2. Materials and Methods
This study retrospectively examined the electronic medical records (EMR) of all patients seen by the Lung Cancer Evaluation Center (LCEC) at the Stony Brook Cancer Center, Stony Brook, NY, USA, between 1 January 2010 and 31 May 2020. The LCEC was established to evaluate, diagnose, treat, and monitor patients found to have concerning pulmonary nodules. The LCEC maintains a database which captures longitudinal data for each patient, beginning with their baseline visit. The surveillance component of the program enables an opportunity to evaluate patients who span the full spectrum of disease and includes those who are diagnosed with cancer at first presentation, those who present with benign nodules and develop cancer over time, and those who present with non-malignant lesions yet remain cancer-free while under care of the program.
Upon identification of a suspicious nodule on imaging, a PET is ordered for further evaluation. If found to be FDG avid, a biopsy is recommended. A biopsy that yields atypia is presented at the weekly tumor board. Upon consensus of the multidisciplinary care team, which includes representation from all program sub-specialties, an individualized patient plan is formulated. In most cases, if the images are suspicious and biopsy results are atypical, the team will recommend a repeat scan and possible repeat procedure within 1–6 months [
8].
Data for this investigation, which were abstracted from both the LCEC database and EMR, included age, gender, atypia status, date of atypia diagnosis, smoking history (current, former, or never), comorbidities (history of diabetes, hypertension, and emphysema), family history of cancer, and nodule features including lesion size (in cm), mixed ground glass opacity type, spiculation, location (upper vs. lower lobe, right vs. left) and PET SUV. PET positivity was defined as PET SUV
2.5 [
9,
10]. Additionally, for those patients who were ultimately diagnosed with cancer, the date of cancer diagnosis was collected. For patients who remained disease-free during the observation period, the last date of examination was recorded.
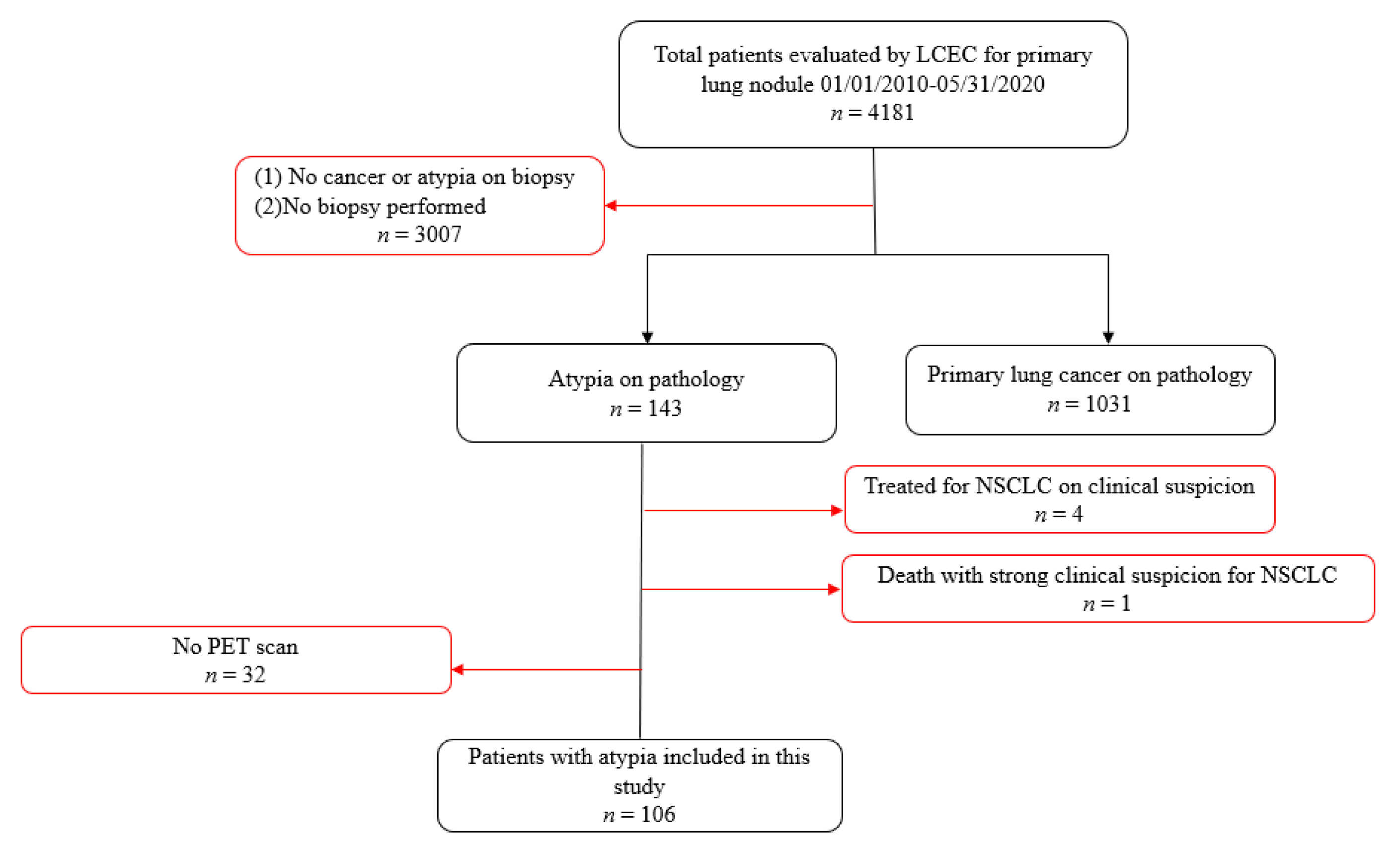
Figure 1 presents the inclusion and exclusion criteria for the study sample. As this study sought to evaluate cytology-proven outcomes among patients with atypia, those who did not have a lung biopsy or a diagnosis of atypia (
n = 3007) were excluded from the study, as were patients found to have biopsy-confirmed primary lung cancer without a previous diagnosis of atypia (
n = 1031). Among the 143 patients receiving a baseline diagnosis of atypia, 4 were treated as a result of a strong clinical suspicion of lung cancer despite the biopsy confirmation of only atypia without re-biopsy, and 1 died prior to such confirmation. Those patients were excluded from the study, along with 32 others who did not have a PET scan. The remaining 106 patients represent the basis for this investigation.
Statistical Analyses
Descriptive statistics were used to compare demographic and nodule characteristics between patients with atypia who were ultimately diagnosed with lung cancer and those who remained disease-free by the conclusion of the study. Quantitative variables are presented as mean standard deviation (SD), and differences between groups are evaluated using the Student’s t-test. Categorical factors are presented as percentages and compared using the chi-square test. The distribution of duration in time (in days) between the date of atypia diagnosis and the date of final outcome (defined as either diagnosis of lung cancer or disease-free at the close of the study) is presented in quartiles. Kaplan–Meier curves are provided, and patients for whom no event occurred within the specified time frame were right censored. Cox Proportional Hazard (CPH) Models, adjusted for age and gender, were used to assess the possible effect of any variable on outcome status. Factors found to be different between groups at the 0.10 level of significance in the univariate analyses were included in the CPH Models. Hazard Ratios (HR) and 95% Confidence Intervals (CI) are presented. SPSS version 21 (IBM Corporation, Armonk, NY, USA) was used to carry out the analyses for this study.
3. Results
Among 106 patients with a baseline diagnosis of atypia, 80 (75%) were diagnosed with lung cancer during the observation period. While adenocarcinoma (63%) and squamous cell (15%) were the primary cancer types subsequently diagnosed among cases, pneumonia (23%), fibrosis (19%), and chronic inflammation (12%) were the primary histologic diagnoses among patients who remained cancer-free.
Table 1 presents the demographic and nodule characteristics of the study sample, stratified by cancer outcome status. The average age of lung cancer among cases was 67.4 years and 47.5% were male. There were no significant differences in age or gender between patients who were diagnosed with cancer and those who remained disease-free by the close of the study. Likewise, there were no differences in smoking status, history of diabetes or hypertension, family history of cancer, or diagnosis of emphysema between the two groups. Cancer cases were more likely to be PET positive (
p = 0.02) and had marginally larger baseline nodules, on average, than patients without cancer (2.4 cm vs. 1.9 cm,
p = 0.08). Additionally, mixed ground glass opacities tended to be more common among patients diagnosed with lung cancer compared to non- cancer cases (
p = 0.06). As solid and mixed ground glass nodules represent different entities of disease, patients were further stratified by opacity type to determine if there were any differences between the two groups. Cases with mixed ground glass opacities were found to be older than those with solid nodules (73.6 ± 6.0 vs. 66.5 ± 8.9 years,
p = 0.02), however there were no other statistically significant differences with regard to any of the demographic factors or nodule characteristics under investigation.
The distribution of duration in days between the baseline diagnosis of atypia and the final outcome is presented in
Table 2, alongside the Kaplan–Meier survival curve (
Figure 2). Among lung cancer cases, the median amount of time to diagnosis after an initial indication of atypia was 35 days, and more than 75% of patients were diagnosed within 6 months. Less than 20% of cases were diagnosed after 1 year. In contrast, 75% of non-cases remained disease-free after 1 year. Additionally, similar conversion rates were found for patients with solid (75%) and mixed ground glass lesions (70%) at 6 months.
CPH Models were used to determine factors that may be predictive of lung cancer among patients with atypia. The results are presented in
Table 3 and indicate that PET positivity (HR = 1.74 (1.03, 2.94)), nodule size > 3.5 cm (HR = 2.83 (1.47, 5.45)), and mixed ground glass opacity type (HR = 2.15 (1.05, 4.43)) were significant predictors of cancer.
As more than 80% of cases were diagnosed within 12 months and 25% of non-cases were not under the care of the LCEC for a full year during the study period, additional CPH models were performed to address any potential misclassification biases. After excluding non-cases who did not participate in the program for a minimum of 365 days, the main findings remained unchanged.
4. Discussion
While the literature indicates that larger and more rapidly growing pulmonary nodules have an increased likelihood of carcinogenesis [
2] and that nodules > 8 mm in size pose a higher likelihood of malignancy, [
3] data are more limited as to whether (and in what capacity) atypical cells may represent precursors to cancer. Findings from the current investigation indicate that 75% of patients with an initial diagnosis of atypia were diagnosed with cancer. Of those, half were diagnosed within one month, and more than three-quarters were confirmed to have lung cancer within 6 months. Additionally, PET positive patients with mixed ground glass opacities > 3.5 cm in size were at significantly increased risk of developing lung cancer. The findings from this study suggest that patients with this profile who are found to have a result of “Atypia” on their biopsy may be considered as likely already having cancer, and this may warrant repeat attempts at establishing a definitive diagnosis to potentially reduce treatment delays.
Many factors may contribute to a diagnosis of atypical cellularity [
11,
12] and the outcome is often the result of a limited number of cells captured for evaluation. Challenges in obtaining an adequate sample may result from a small or less than desirable location for the biopsy, patient body habitus, expertise of the proceduralist, and the number of times a procedure is performed. Additionally, multiple passes are known to reduce the quality and quantity of the specimens [
2,
13]. Sampling techniques, including fine needle aspiration, CT or ultrasound guided percutaneous procedures, and bronchoscopy [
4] can also play a role in the adequacy of the sample obtained. CT-guided transthoracic lung biopsies have been reported to produce a 77–90% diagnostic yield, [
14,
15] whereas bronchoscopy has been shown to have a 70–81% yield, and bronchial brushes a 56% yield [
16,
17]. The procedure type, as well as the size and location of the nodule, remain the main factors influencing sample collection. Lesions larger than 1 cm and at a location less than 4 cm from the needle path are thought to provide the most significant yield [
18].
Progression of benign nodules to cancer is often quantified in terms of doubling time. The literature indicates that cancerous nodules are typically expected to double in volume between 20 and 300 days [
19,
20,
21,
22]. This study found that 50% of patients with atypia who were diagnosed with lung cancer had biopsy-proven cancer within one month, and 75% within 6 months. The short interval of time between original atypia cytology and final cancer diagnosis noted in this investigation may be attributable, at least in part, to the multidisciplinary approach employed by the LCEC program. In most cases, if images are suspicious and the cell type is atypical, a repeat scan and possible repeat procedure will be recommended within 1–6 months. This tight surveillance practice may potentially reduce the time to diagnosis among this subgroup of patients.
This investigation is among the first to evaluate factors influencing the ultimate diagnosis of lung cancer after findings of atypical cytology in a lung nodule biopsy specimen, however it has several limitations. First, given its observational, retrospective design, this study lacked data for several important factors that may influence outcomes. Details were unavailable for biopsy yield and adequacy, as well as the molecular and cellular profiles of the sample. Without this information, it is difficult to evaluate the mechanistic drivers which may have influenced the cytologic determination of atypia. Secondly, the study was unable to capture and quantify variables such as the expertise of the proceduralist, the physical environment, transport conditions of the sample to the cytology lab, and the experience of the pathologist, all of which may have contributed to the resulting diagnosis of atypia. In this investigation, 98% of the biopsies were performed at Stony Brook and reviewed by experienced pathologists specializing in the lung. While the experience of the pathologists external to the Stony Brook Cancer Center is unknown and may introduce potential bias, the direction of such bias is unclear, and the low percentage of patients with an external review suggests that such bias would be minimal. Lastly, the study’s sample size was limited and the investigation was conducted within a single institution in the northeast, thereby potentially limiting the generalizability of the findings. Future studies are needed to validate the results of this investigation and further elucidate the mechanisms and factors that may influence the conversion of atypical cells to cancer.
5. Conclusions
In patients with lung nodules initially classified as atypia on biopsy, repeat efforts at establishing a diagnosis are warranted in order to cut treatment delays to a minimum due to the high likelihood of this population already having a cancer; particularly within the first 6 months of the baseline biopsy. As such, further consideration should be given to repeat biopsy. Patients with mixed ground glass pulmonary nodules > 3.5 cm in size and a positive PET scan may particularly benefit from tighter monitoring in the early months following a diagnosis of atypia. Targeting this subgroup of patients may help to maximize the potential for early detection and thereby improve patient outcomes.
Author Contributions
Conceptualization, D.A., M.F. and B.N.; Methodology, D.A., L.A.S., T.B. and B.N.; Software, Validation, B.N.; Formal analysis, B.N.; B.N.; Investigation, D.A. and B.N.; Resources, B.N.; Data curation, L.A.S., T.B. and S.N.; Writing—original draft, D.A., L.A.S., T.B., M.F., S.N. and B.N.; Writing—review & editing, D.A., L.A.S., T.B., M.F., S.N. and B.N; Visualization, D.A. and M.F.; Supervision, D.A., T.B., M.F. and B.N.; Project administration, D.A., L.A.S. and B.N.; All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
The study was conducted according to the guidelines of the Declaration of Helsinki, and approved by the Institutional Review Board of Stony Brook University (IRB2019-00596).
Informed Consent Statement
Patient consent was waived due to the retrospective nature of this study.
Data Availability Statement
The data presented in this study are available on request from the corresponding author. The data are not publicly available due to patient privacy.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2020. CA Cancer J. Clin. 2020, 70, 7–30. [Google Scholar] [CrossRef]
- Gould, M.K.; Donington, J.; Lynch, W.R.; Mazzone, P.J.; Midthun, D.E.; Naidich, D.P.; Wiener, R.S. Evaluation of individuals with pulmonary nodules: When is it lung cancer? Diagnosis and management of lung cancer, 3rd ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest 2013, 143, e93S–e120S. [Google Scholar] [CrossRef] [Green Version]
- Sanchez, M.; Benegas, M.; Vollmer, I. Management of incidental lung nodules <8 mm in diameter. J. Thorac. Dis. 2018, 10, S2611–S2627. [Google Scholar] [CrossRef]
- Idowu, M.O.; Powers, C.N. Lung cancer cytology: Potential pitfalls and mimics—A review. Int. J. Clin. Exp. Pathol. 2010, 3, 367–385. [Google Scholar] [PubMed]
- Ofiara, L.M.; Navasakulpong, A.; Ezer, N.; Gonzalez, A.V. The importance of a satisfactory biopsy for the diagnosis of lung cancer in the era of personalized treatment. Curr. Oncol. 2012, 19, S16–S23. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Deng, C.J.; Dai, F.Q.; Qian, K.; Tan, Q.Y.; Wang, R.W.; Deng, B.; Zhou, J.H. Clinical updates of approaches for biopsy of pulmonary lesions based on systematic review. BMC Pulm. Med. 2018, 18, 146. [Google Scholar] [CrossRef]
- Tongbai, T.; McDermott, S.; Kiranantawat, N.; Muse, V.V.; Wu, C.C.C.; Shepard, J.A.O.; Gilman, M.D. Non-Diagnostic CT-Guided Percutaneous Needle Biopsy of the Lung: Predictive Factors and Final Diagnoses. Korean J. Radiol. 2019, 20, 1515–1526. [Google Scholar] [CrossRef] [PubMed]
- Wagnetz, U.; Menezes, R.J.; Boerner, S.; Paul, N.S.; Wagnetz, D.; Keshavjee, S.; Roberts, H.C. CT screening for lung cancer: Implication of lung biopsy recommendations. AJR Am. J. Roentgenol. 2012, 198, 351–358. [Google Scholar] [CrossRef]
- Mosmann, M.P.; Borba, M.A.; Macedo, F.P.N.d.; Liguori, A.d.A.L.; Villarim Neto, A.; Lima, K.C.d. Solitary pulmonary nodule and 18F-FDG PET/CT. Part 1: Epidemiology, morphological evaluation and cancer probability. Radiol. Bras. 2016, 49, 35–42. [Google Scholar] [CrossRef]
- Hellwig, D.; Graeter, T.P.; Ukena, D.; Groeschel, A.; Sybrecht, G.W.; Schaefers, H.J.; Kirsch, C.M. 18F-FDG PET for mediastinal staging of lung cancer: Which SUV threshold makes sense? J. Nucl. Med. 2007, 48, 1761–1766. [Google Scholar] [CrossRef] [Green Version]
- Huang, C.T.; Tsai, Y.J.; Ho, C.C.; Yu, C.J. Atypical cells in pathology of endobronchial ultrasound-guided transbronchial biopsy of peripheral pulmonary lesions: Incidence and clinical significance. Surg. Endosc. 2019, 33, 1783–1788. [Google Scholar] [CrossRef]
- Travis, W.D.; Brambilla, E.; Noguchi, M.; Nicholson, A.G.; Geisinger, K.; Yatabe, Y.; Ishikawa, Y.; Wistuba, I.; Flieder, D.B.; Franklin, W.; et al. Diagnosis of lung cancer in small biopsies and cytology: Implications of the 2011 International Association for the Study of Lung Cancer/American Thoracic Society/European Respiratory Society classification. Arch. Pathol. Lab. Med. 2013, 137, 668–684. [Google Scholar] [CrossRef] [Green Version]
- Callister, M.E.; Baldwin, D.R.; Akram, A.R.; Barnard, S.; Cane, P.; Draffan, J.; Franks, K.; Gleeson, F.; Graham, R.; Malhotra, P.; et al. British Thoracic Society guidelines for the investigation and management of pulmonary nodules. Thorax 2015, 70 (Suppl. 2), ii1–ii54. [Google Scholar] [CrossRef] [Green Version]
- Padrao, E.; Rodrigues, M.; Guimaraes, S.; Caetano Mota, P.; Melo, N.; Souto Moura, C.; Morais, A.; Pereira, J.M. Diagnostic Yield of Computed Tomography-Guided Transthoracic Lung Biopsy in Diffuse Lung Diseases. Respiration 2018, 96, 455–463. [Google Scholar] [CrossRef] [PubMed]
- Zhao, G.; Shi, X.; Sun, W.; Liang, H.; Mao, X.; Wen, F.; Shan, M.; Wang, X.; Lu, Z. Factors affecting the accuracy and safety of computed tomography-guided biopsy of intrapulmonary solitary nodules ≤30 mm in a retrospective study of 155 patients. Exp. Ther. Med. 2017, 13, 1986–1992. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Acharya, K.V.; Unnikrishnan, B.; Shenoy, A.; Holla, R. Utility of Various Bronchoscopic Modalities in Lung Cancer Diagnosis. Asian Pac. J. Cancer Prev. 2017, 18, 1931–1936. [Google Scholar] [CrossRef] [PubMed]
- Herth, F.J.; Eberhardt, R.; Becker, H.D.; Ernst, A. Endobronchial ultrasound-guided transbronchial lung biopsy in fluoroscopically invisible solitary pulmonary nodules: A prospective trial. Chest 2006, 129, 147–150. [Google Scholar] [CrossRef] [PubMed]
- Ohno, Y.; Hatabu, H.; Takenaka, D.; Higashino, T.; Watanabe, H.; Ohbayashi, C.; Sugimura, K. CT-guided transthoracic needle aspiration biopsy of small (<or =20 mm) solitary pulmonary nodules. AJR Am. J. Roentgenol. 2003, 180, 1665–1669. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.P.; Chew, C.T.; Consigliere, D.T.; Heng, D.; Huang, D.T.; Khoo, J.; Khoo, K.S.; Low, J.; Lui, S.; Ooi, L.L.; et al. Ministry of health clinical practice guidelines: Cancer screening. Singap. Med. J. 2010, 51, 170–173. [Google Scholar]
- MacMahon, H.; Naidich, D.P.; Goo, J.M.; Lee, K.S.; Leung, A.N.C.; Mayo, J.R.; Mehta, A.C.; Ohno, Y.; Powell, C.A.; Prokop, M.; et al. Guidelines for Management of Incidental Pulmonary Nodules Detected on CT Images: From the Fleischner Society 2017. Radiology 2017, 284, 228–243. [Google Scholar] [CrossRef] [Green Version]
- Nathan, M.H.; Collins, V.P.; Adams, R.A. Differentiation of Benign and Malignant Pulmonary Nodules by Growth Rate. Radiology 1962, 79, 221–232. [Google Scholar] [CrossRef] [PubMed]
- Ost, D.E.; Gould, M.K. Decision making in patients with pulmonary nodules. Am. J. Respir. Crit. Care Med. 2012, 185, 363–372. [Google Scholar] [CrossRef] [PubMed]
| Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).