Digital Innovation in Oncological Primary Treatment for Well-Being of Patients: Psychological Caring as Prompt for Enhancing Quality of Life
Abstract
:1. Introduction
2. Method
2.1. Ethical Approval
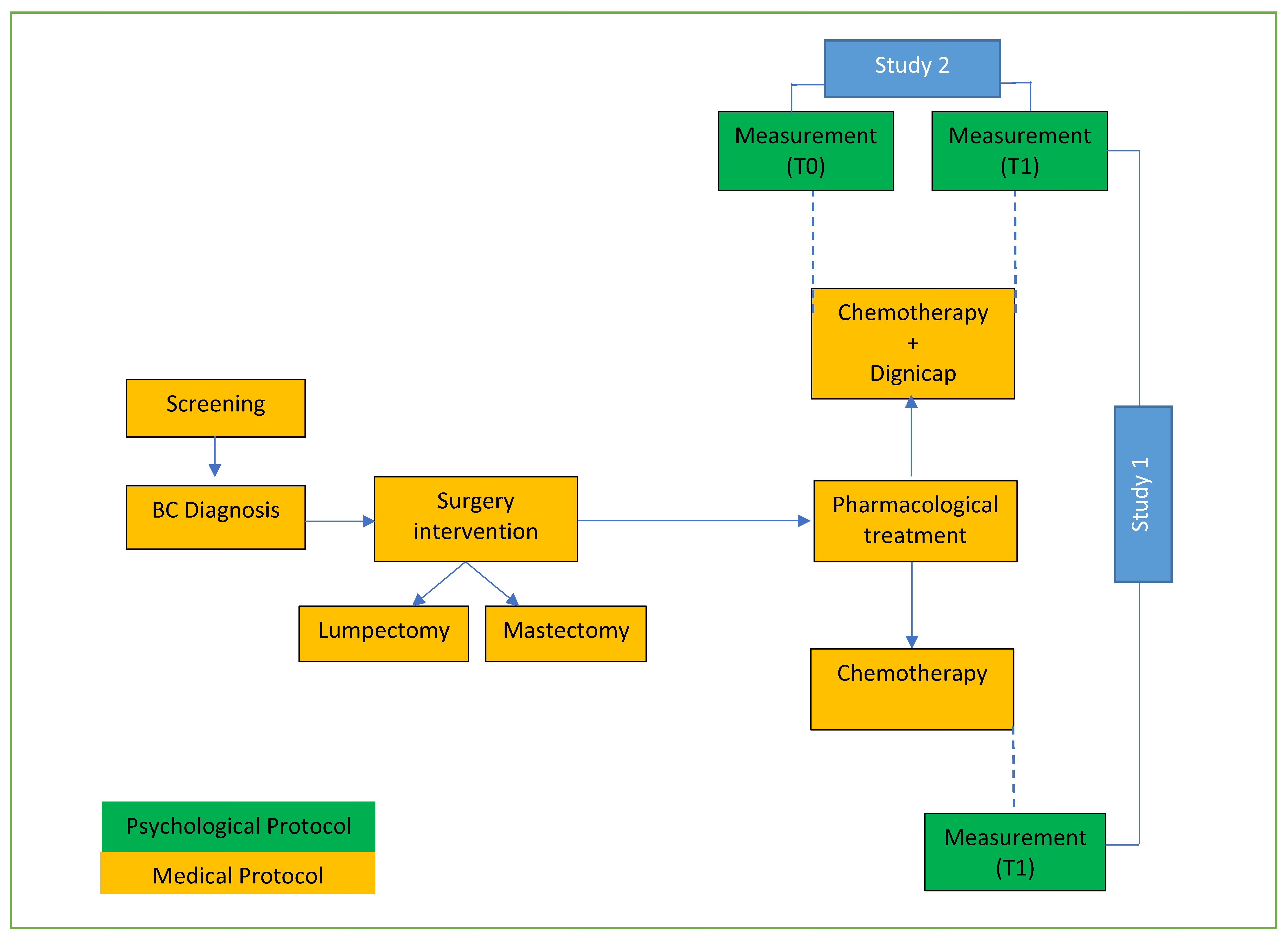
2.2. Study Design
3. Research Protocol 1
3.1. Objective
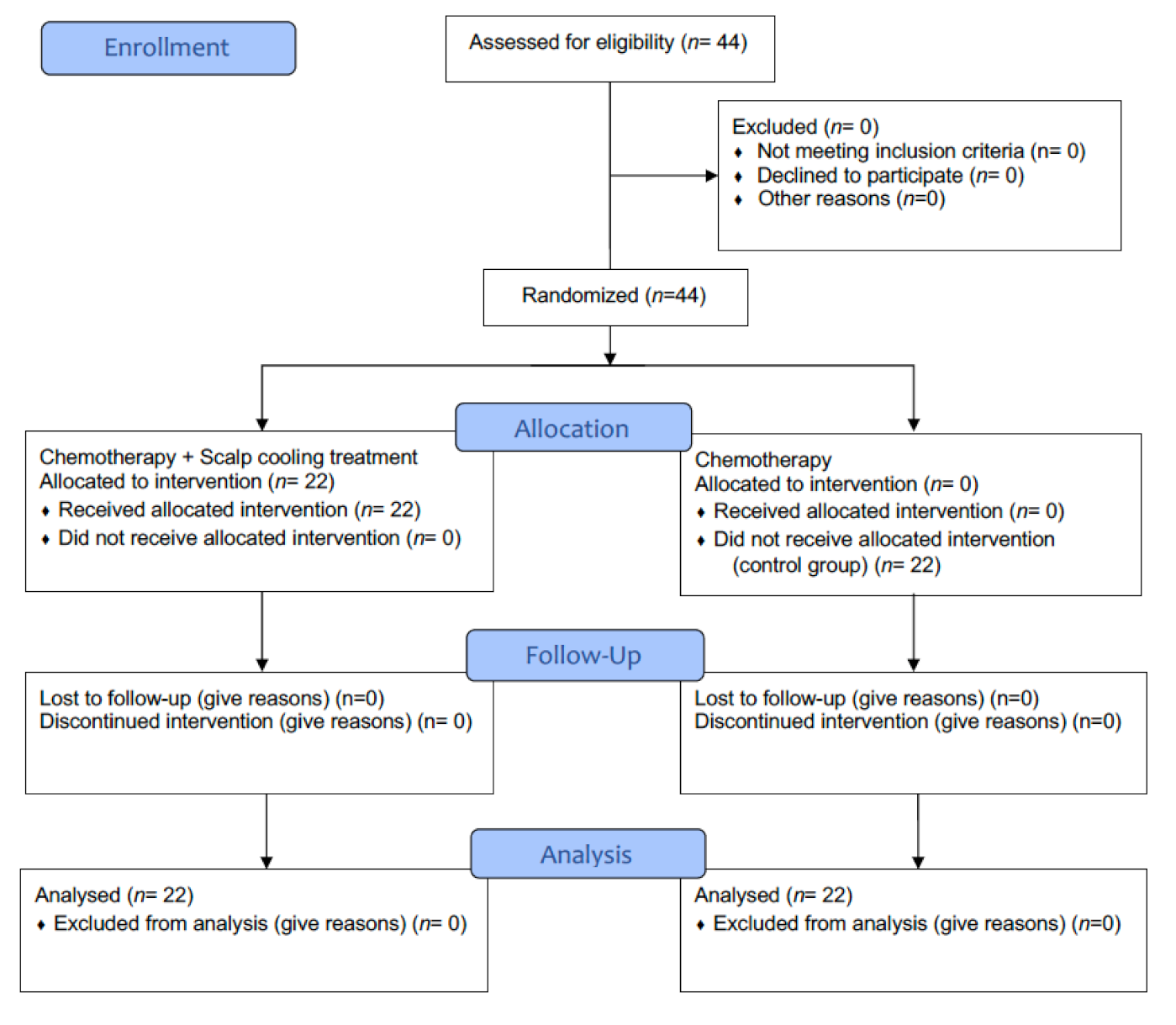
3.2. Participants
3.3. Scalp Cooling Technology Treatment
3.4. Measurement
3.5. Procedure
3.6. Statistical Analysis
3.7. Results
4. Research Protocol 2
4.1. Objective
4.2. Participants
4.3. Measurements
4.4. Procedure
4.5. Statistical Analysis
4.6. Results
5. Discussion
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mehnert, A.; Hartung, T.J.; Friedrich, M.; Vehling, S.; Brähler, E.; Härter, M.; Keller, M.; Schulz, H.; Wegscheider, K.; Weis, J.; et al. One in two cancer patients is significantly distressed: Prevalence and indicators of distress. Psychooncology 2018, 27, 75–82. [Google Scholar] [CrossRef]
- Di Giacomo, D.; Cannita, K.; Ranieri, J.; Cocciolone, V.; Passafiume, D.; Ficorella, C. Breast cancer and psychological resilience among young women. J. Psychopathol. 2016, 22, 191–195. [Google Scholar]
- Ranieri, J.; Guerra, F.; Di Giacomo, D. Role of Metacognition Thinking and Psychological Traits in Breast Cancer Survivorship. Behav. Sci. 2020, 10, 135. [Google Scholar]
- Nangia, J. Quality of Life Matters: It Is Time to Integrate Scalp Cooling in Routine Clinical Practice. J. Oncol. Pract. 2018, 14, 157–158. [Google Scholar] [CrossRef]
- Orlando, L.; Loparco, D.; Fedele, P.; Schiavone, P.; Quaranta, A.; Caliolo, C.; Cinefra, M.; Rizzo, P.; Calvani, N.; Morleo, A.; et al. Final results of prospective study of scal cooling in preventing chemotherapy-induced alopecia. Future Oncol. 2019, 15. [Google Scholar] [CrossRef] [PubMed]
- Kruse, M.; Abraham, J. Management of chemotherapy-induced alopecia with scalpcooling. J. Oncol. Pract. 2018, 14, 149–154. [Google Scholar] [CrossRef] [PubMed]
- Zdenkowski, N.; Tesson, S.; Lombard, J.; Lovell, M.; Hayes, S.; Francis, P.A.; Dhillon, H.M.; Boyle, F.M. Supportive care of women with breast cancer: Key concerns and practical solutions. Med. J. Aust. 2016, 205, 471–475. [Google Scholar] [CrossRef] [PubMed]
- Linden, W.; Vodermaier, A.; Mackenzie, R.; Greig, D. Anxiety and depression after cancer diagnosis: Prevalence rates by cancer type, gender, and age. J. Affect. Disord. 2012, 141, 343–351. [Google Scholar] [CrossRef]
- Seiler, A.; Jenewein, J. Resilience in Cancer Patients. Front. Psychiatry 2019, 10, 208. [Google Scholar] [CrossRef] [Green Version]
- Di Giacomo, D.; Ranieri, J.; Perilli, E. Psychological impact of clinical treatment after breast cancer diagnosis in younger patients (38–50 age range): An explorative 3-year observational study. Neurol. Psychiatry Brain Res. 2019, 32, 85–90. [Google Scholar] [CrossRef]
- Giarratano, T.; Frezzini, S.; Zanocco, M.; Giorgi, C.A.; Mioranza, E.; Miglietta, F.; Griguolo, G.; Falci, C.; Faggioni, G.; Tasca, G.; et al. Use of scalp cooling device to prevent alopecia for early breast cancer patients receiving chemotherapy: A prospective study. Breast J. 2019, 100, 1–6. [Google Scholar] [CrossRef]
- Bajpai, J.; Kagwade, S.; Chandrasekharan, A.; Dandekar, S.; Kanan, S.; Kembhavi, Y.; Ghosh, J.; Banavali, S.D.; Gupta, S. Randomised controlled trial of scalp cooling for the prevention of chemotherapy induced alopecia. Breast 2020, 49, 187–193. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Silva, G.D.B.; Ciccolini, K.; Donati, A.; Hurk, C.V.D. Scalp cooling to prevent chemotherapy-induced alopecia. An. Bras. Dermatol. 2020, 95, 631–637. [Google Scholar] [CrossRef] [PubMed]
- Beck, A.T.; Brown, G.; Steer, R.A. Beck Depression Inventory II Manual; The Psychological Corporation: San Antonio, TX, USA, 1996. [Google Scholar]
- Morasso, G.; Costantini, M.; Baracco, G.; Borreani, C.; Capelli, M. Assessing psychological distress in cancer patients: Validation of a self-administered questionnaire. Oncology 1996, 53, 295–302. [Google Scholar] [CrossRef] [PubMed]
- Bottesi, G.; Ghisi, M.; Altoè, G.; Conforti, E.; Melli, G.; Sica, C. The Italian version of the Depression Anxiety Stress Scales-21: Factor structure and psychometric properties on community and clinical samples. Compr. Psychiatry 2015, 60, 170–181. [Google Scholar] [CrossRef]
- Grugnetti, A.M.; Arrigoni, C.; Fusco, M.A.; Magon, A.; Sgarella, A.; Gallotti, M.L.; Grugnetti, G.; Caruso, R. Versione italiana della Chemotherapy-induced Alopecia Distress Scale (I-CADS): Adattamento linguistico culturale, validitá di contenuto e valutazione delle proprietá psicometriche. Recenti Progress. Med. 2018, 109, 174–184. [Google Scholar]
- Cheli, S.; Agostini, A.; Herd-Smith, A.; Caligiani, L.; Martella, F.; Fioretto, L. The Italian version of body image scale—reliability and sensitivity in a sample of breast cancer patients. Psicoter. Cogn. Comport. 2016, 22, 65–79. [Google Scholar] [CrossRef]
- Carver, C.S.; Measure of Body Apperception (MBA). Measurement Instrument Database for the Social Science. 2013. Available online: https://www.midss.org/content/measure-body-apperception-mba (accessed on 13 March 2020).
- van den Hurk, C.J.; Breed, W.P.; Nortier, J.W. Short post-infusion scalp cooling time in the prevention of docetaxel-induced alopecia. Support. Care Cancer 2012, 20, 3255–3260. [Google Scholar] [CrossRef]
- Wang, X.; Wang, N.; Zhong, L.; Wang, S.; Zheng, Y.; Yang, B.; Zhang, J.; Lin, Y.; Wang, Z. Prognostic value of depression and anxiety on breast cancer recurrence and mortality: A systematic review and meta-analysis of 282,203 patients. Mol. Psychiatry 2020, 25, 3186–3197. [Google Scholar] [CrossRef]
- Peterson, L.L.; Lustberg, M.; Tolaney, S.M.; Ross, M.; Salehi, E.; Isakoff, S.J. Integration of Physician and Nursing Professional Efforts to Deliver Supportive Scalp Cooling Care to Oncology Patients at Risk for Alopecia. Oncol. Ther. 2020, 8, 325–332. [Google Scholar] [CrossRef]
- Gianotti, E.; Razzini, G.; Bini, M.; Crivellaro, C.; Righi, A.; Darecchio, S.; Lui, S.; Basiricò, M.L.; Cocconi, S.; Cagossi, K.; et al. Scalp Cooling in Daily Clinical Practice for Breast Cancer Patients Undergoing Curative Chemotherapy: A Multicenter Interventional Study. Asia Pac. J. Oncol. Nurs. 2019, 6, 277–282. [Google Scholar] [CrossRef] [PubMed]
| DEMOGRAPHIC INDEXES | POST CHEMOTHERAPY GROUPING | ||
|---|---|---|---|
| DG GROUP (N. 22) x 44.85 sd ± 6.82 | NDG GROUP (N.22) x 42.03 sd ± 5.84 | Total (N. 44) x 43.85 sd ± 6.45 | |
| EDUCATION | |||
| NO HIGH SCHOOL HIGH SCHOOL DEGREE UNDERGRADUATE DEGREE | 7.15% | 14.28% | 10% |
| 67.85% | 57.15% | 62% | |
| 25% | 28.57% | 28% | |
| MARITAL STATUS | |||
| MARRIED/LIVING WITH PARTNER SINGLEDIVORCED/WIDOWED | 78.57% | 57.14% | 67.85% |
| 3.57% | 25% | 14.3% | |
| 17.85% | 17.85% | 17.85% | |
| OCCUPATION | |||
| UNEMPLOYED EMPLOYED SELF EMPLOYED | 14.29% | 42.85% | 28.57% |
| 60.71% | 53.58% | 57.15% | |
| 25% | 3.57% | 14.28% | |
| TNM | |||
| 0 1 2 3 | 3.58% | 8% | 5.4% |
| 35.71% | 47% | 42% | |
| 35.71% | 22.5% | 29.1% | |
| 25% | 22.5% | 23.5% | |
| LUMINAL TYPE | |||
| A B HER2 TRIPLE NEGATIVE | 10.51% | 26% | 18% |
| 7.42% | 66% | 68% | |
| 7.14% | 0% | 4% | |
| 10.71% | 8% | 10% | |
| TESTS | POST CHEMOTHERAPY GROUPING | F | P | |
|---|---|---|---|---|
| DG x sd | NDG x sd | |||
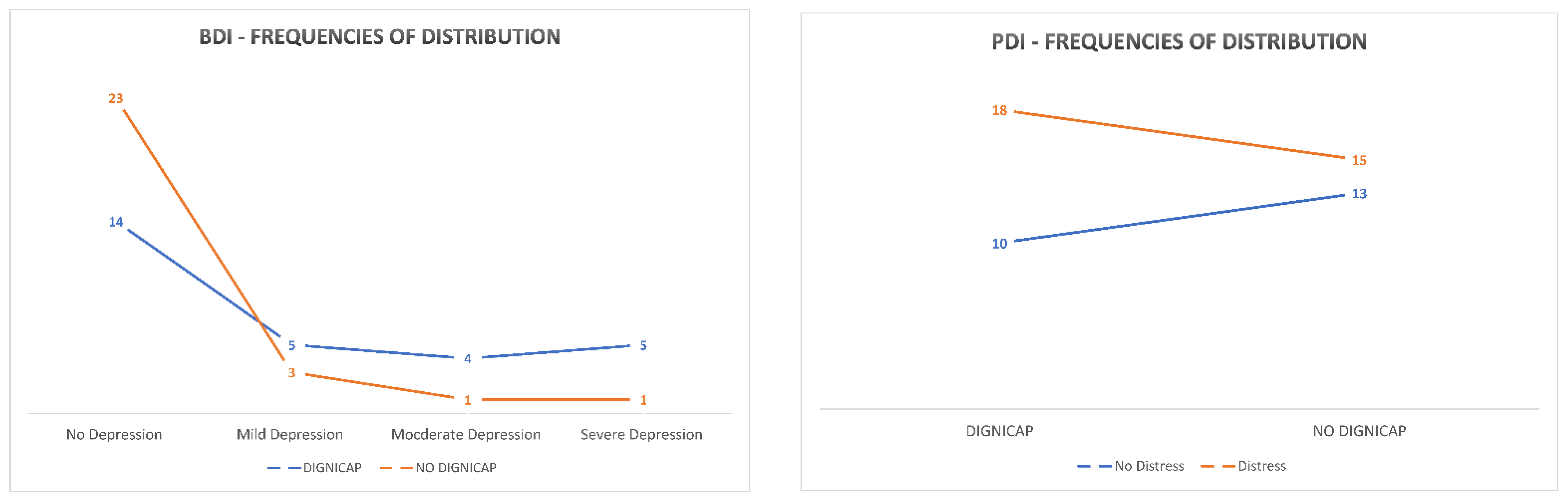
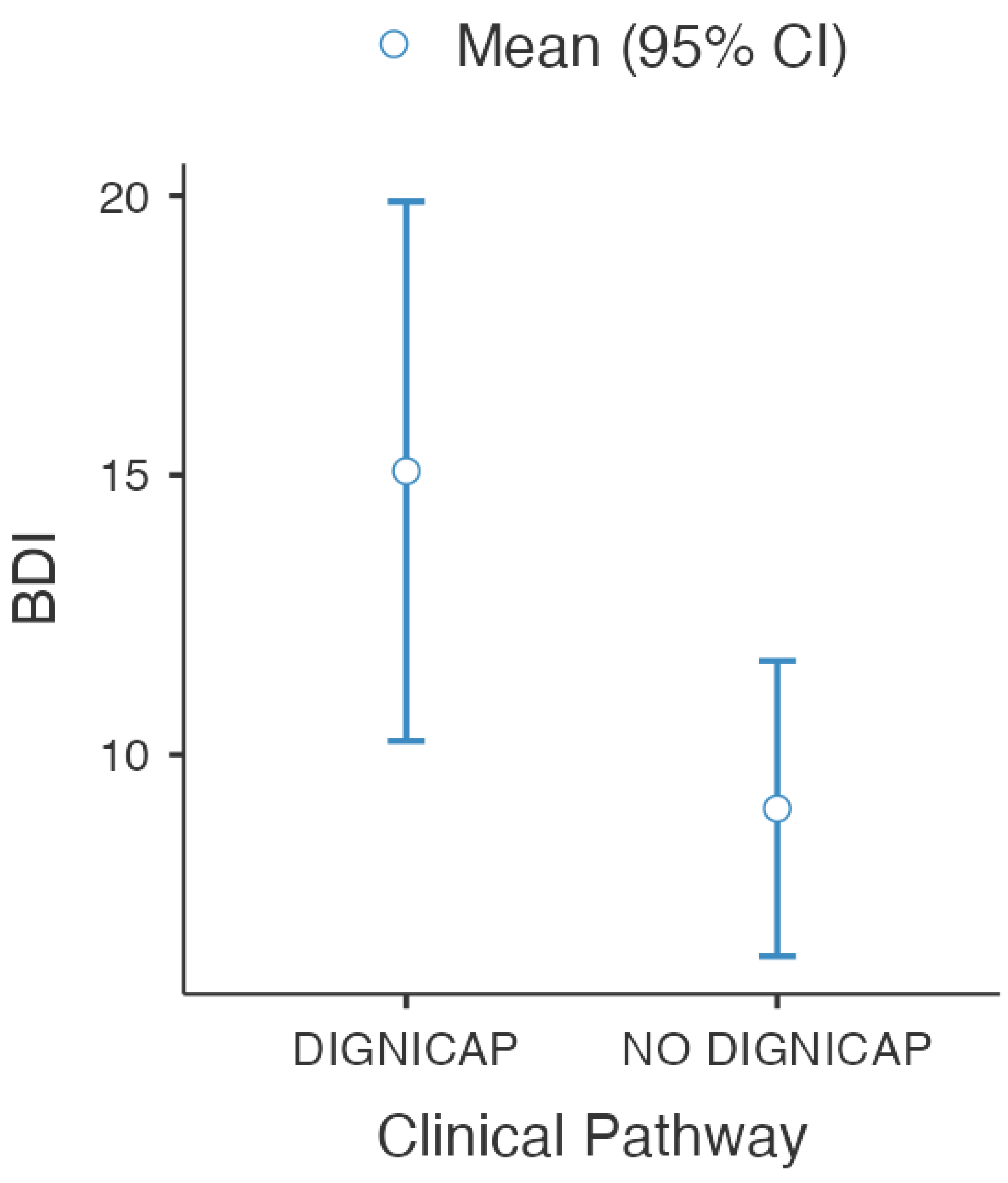
| PDI | 29.7 (±8.0) | 28.1 (±7.4) | 0.62 | 0.43 |
| BDI | 15.0 (±12.4) | 9.0 (±6.8) | 41.8 | 0.02 * |
| DEMOGRAPHIC INDEXES | PARTICIPANTS N(%) |
|---|---|
| EDUCATION | |
| NO HIGH SCHOOL | 2 (9.1%) |
| HIGH SCHOOL DEGREE | 16 (72.72%) |
| UNDERGRADUATE DEGREE | 4 (18.18%) |
| MARITAL STATUS | |
| MARRIED/LIVING WITH PARTNER | 17 (77.27%) |
| SINGLE | 1 (4.55%) |
| DIVORCED/WIDOWED | 4 (18.18%) |
| OCCUPATION | |
| UNEMPLOYED | 1 (4.55%) |
| EMPLOYED | 19 (86.35%) |
| SELF EMPLOYED | 2 (9.1%) |
| TNM | |
| 0 | 2 (9%) |
| 1 | 9 (40.9%) |
| 2 | 5 (22.77%) |
| 3 | 6 (27.33%) |
| LUMINAL TYPE | |
| A | 1 (4.56%) |
| B | 17 (77.27%) |
| HER2 | 1 (4.56%) |
| TRIPLE NEGATIVE | 3 (13.63%) |
| TEST | TIMEPOINT | RAW SCORE DG | F | P | H2 | |
|---|---|---|---|---|---|---|
| χ | sd | |||||
| PDI | T0 | 28.2 | ±7.46 | 0.74 | 0.39 | - |
| T1 | 29.4 | ±8.72 | ||||
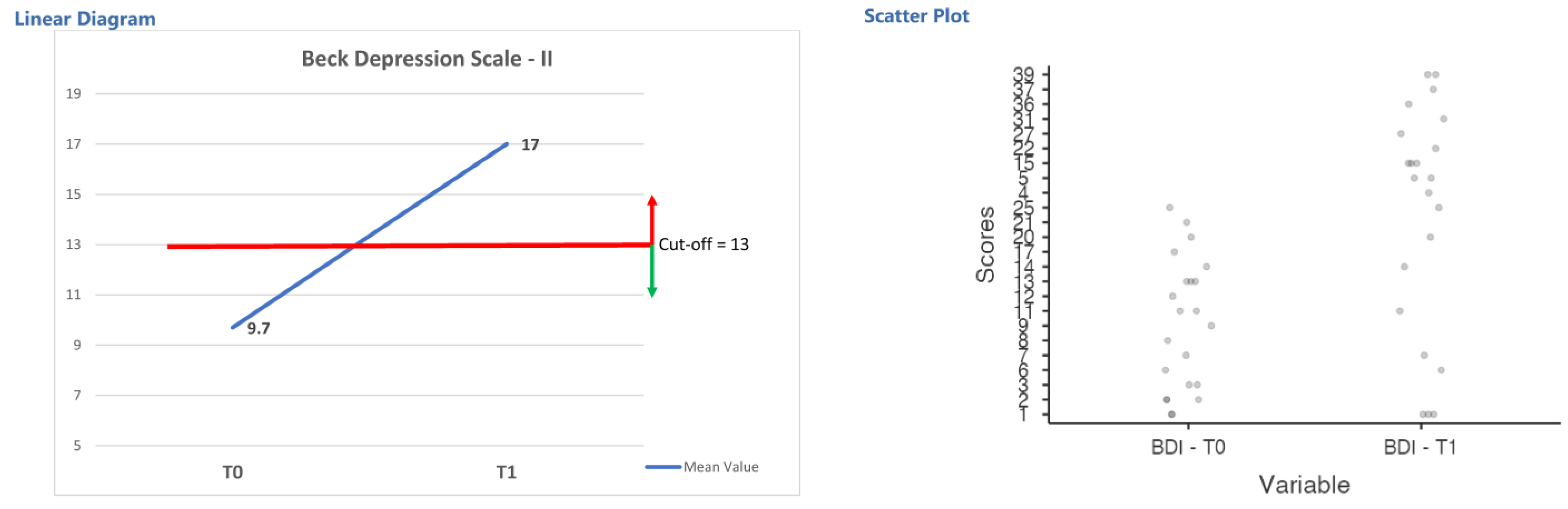
| BDI | T0 | 9.7 | ±6.98 | 12.65 | 0.001 * | 0.11 |
| T1 | 17.0 | ±13.13 | ||||
| ESDQ | T0 | 16.0 | ±3.39 | 1.42 | 0.24 | - |
| T1 | 17.0 | ±3.13 | ||||
| MBA (TOTAL) | T0 | 15.5 | ±5.19 | 2.70 | 0.11 | - |
| T1 | 18.0 | ±4.71 | ||||
| MBA (PA INDEX) | T0 | 9.4 | ±3.23 | 1.69 | 0.20 | - |
| T1 | 10.0 | ±2.84 | ||||
| MBA (BI INDEX) | T0 | 7.0 | ±2.80 | 1.08 | 0.31 | - |
| T1 | 7.9 | ±2.68 | ||||
| BIS | T0 | 9.0 | ±8.32 | 4.22 | 0.05 | - |
| T1 | 11.1 | ±8.10 | ||||
| DASS-A | T0 | 9.5 | ±9.19 | 0.05 | 0.81 | - |
| T1 | 9.1 | ±6.80 | ||||
| Tests | χ | sd | t | p | Cohen’s d |
|---|---|---|---|---|---|
| I-CADS Self-perception index | 16.1 | ±5.72 | 13.2 | 0.01 | 2.82 |
| I-CADS Emotionality index | 5.5 | ±2.32 | 11.82 | 0.01 | 2.36 |
| I-CADS Social Engagement index | 7.2 | ±3.23 | 10.4 | 0.01 | 2.17 |
| I-cads total score | 28.8 | ±9.81 | 13.7 | 0.01 | 2.88 |
| ESDQ T1 | DASS-A T1 | BDI - T1 | PDI T1 | MBA T1 | MBA IA T1 | MBA IBI T1 | I-CADS SP | I-CADS E | I-CADS- SE | I-CADS Tot | ||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ESDQ T1 | Pearson's r | — | ||||||||||
| p-value | — | |||||||||||
| DASS-A T1 | Pearson's r | −0.412 | — | |||||||||
| p-value | 0.057 | — | ||||||||||
| BDI - T1 | Pearson's r | −0.339 | 0.311 | — | ||||||||
| p-value | 0.122 | 0.159 | — | |||||||||
| PDI T1 | Pearson's r | −0.42 | 0.408 | 0.613 ** | — | |||||||
| p-value | 0.052 | 0.06 | 0.002 | — | ||||||||
| MBA T1 | Pearson's r | −0.591 ** | 0.413 | 0.235 | 0.624 ** | — | ||||||
| p-value | 0.004 | 0.056 | 0.292 | 0.002 | — | |||||||
| MBA IA T1 | Pearson's r | −0.568 ** | 0.23 | 0.038 | 0.43 * | 0.86 *** | — | |||||
| p-value | 0.006 | 0.302 | 0.867 | 0.046 | <0 .001 | — | ||||||
| MBA IBI T1 | Pearson's r | −0.435 * | 0.48 * | 0.372 | 0.637 ** | 0.842 *** | 0.45 * | — | ||||
| p-value | 0.043 | 0.024 | 0.088 | 0.001 | <0 .001 | 0.036 | — | |||||
| I-CADS SP | Pearson's r | −0.495 * | 0.482 * | 0.629 ** | 0.639 ** | 0.621 ** | 0.397 | 0.669 *** | — | |||
| p-value | 0.019 | 0.023 | 0.002 | 0.001 | 0.002 | 0.067 | < 0.001 | — | ||||
| I-CADS E | Pearson's r | −0.276 | 0.08 | 0.585 ** | 0.79 *** | 0.466 * | 0.417 | 0.375 | 0.566 ** | — | ||
| p-value | 0.213 | 0.723 | 0.004 | < 0.001 | 0.029 | 0.054 | 0.086 | 0.006 | — | |||
| I-CADS- SE | Pearson's r | −0.369 | 0.234 | 0.314 | 0.418 | 0.5 * | 0.52 * | 0.325 | 0.648 ** | 0.526 * | — | |
| p-value | 0.091 | 0.295 | 0.154 | 0.053 | 0.018 | 0.013 | 0.139 | 0.001 | 0.012 | — | ||
| I-CADS Tot | Pearson's r | −0.478 * | 0.379 | 0.617 ** | 0.7 *** | 0.652 ** | 0.512 * | 0.6 ** | 0.938 *** | 0.743 *** | 0.827 *** | — |
| p-value | 0.024 | 0.082 | 0.002 | <0 .001 | 0.001 | 0.015 | 0.003 | <0 .001 | < 0.001 | < 0.001 | — |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Di Giacomo, D.; Guerra, F.; Cannita, K.; Di Profio, A.; Ranieri, J. Digital Innovation in Oncological Primary Treatment for Well-Being of Patients: Psychological Caring as Prompt for Enhancing Quality of Life. Curr. Oncol. 2021, 28, 2452-2465. https://doi.org/10.3390/curroncol28040224
Di Giacomo D, Guerra F, Cannita K, Di Profio A, Ranieri J. Digital Innovation in Oncological Primary Treatment for Well-Being of Patients: Psychological Caring as Prompt for Enhancing Quality of Life. Current Oncology. 2021; 28(4):2452-2465. https://doi.org/10.3390/curroncol28040224
Chicago/Turabian StyleDi Giacomo, Dina, Federica Guerra, Katia Cannita, Anna Di Profio, and Jessica Ranieri. 2021. "Digital Innovation in Oncological Primary Treatment for Well-Being of Patients: Psychological Caring as Prompt for Enhancing Quality of Life" Current Oncology 28, no. 4: 2452-2465. https://doi.org/10.3390/curroncol28040224
APA StyleDi Giacomo, D., Guerra, F., Cannita, K., Di Profio, A., & Ranieri, J. (2021). Digital Innovation in Oncological Primary Treatment for Well-Being of Patients: Psychological Caring as Prompt for Enhancing Quality of Life. Current Oncology, 28(4), 2452-2465. https://doi.org/10.3390/curroncol28040224






