Quality of Colon Cancer Care in Patients Undergoing Emergency Surgery
Abstract
1. Introduction
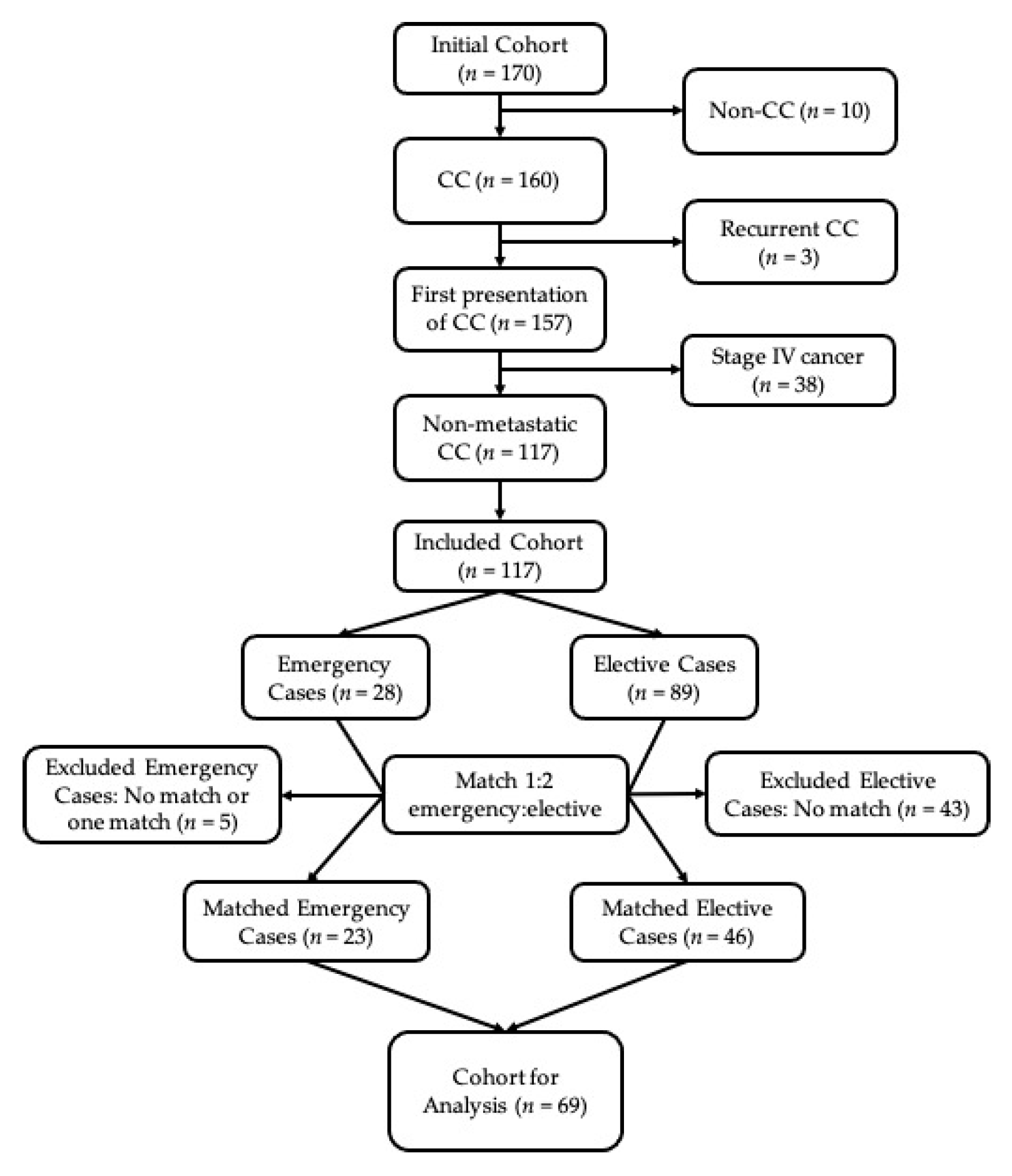
2. Materials and Methods
2.1. Study Setting and Design
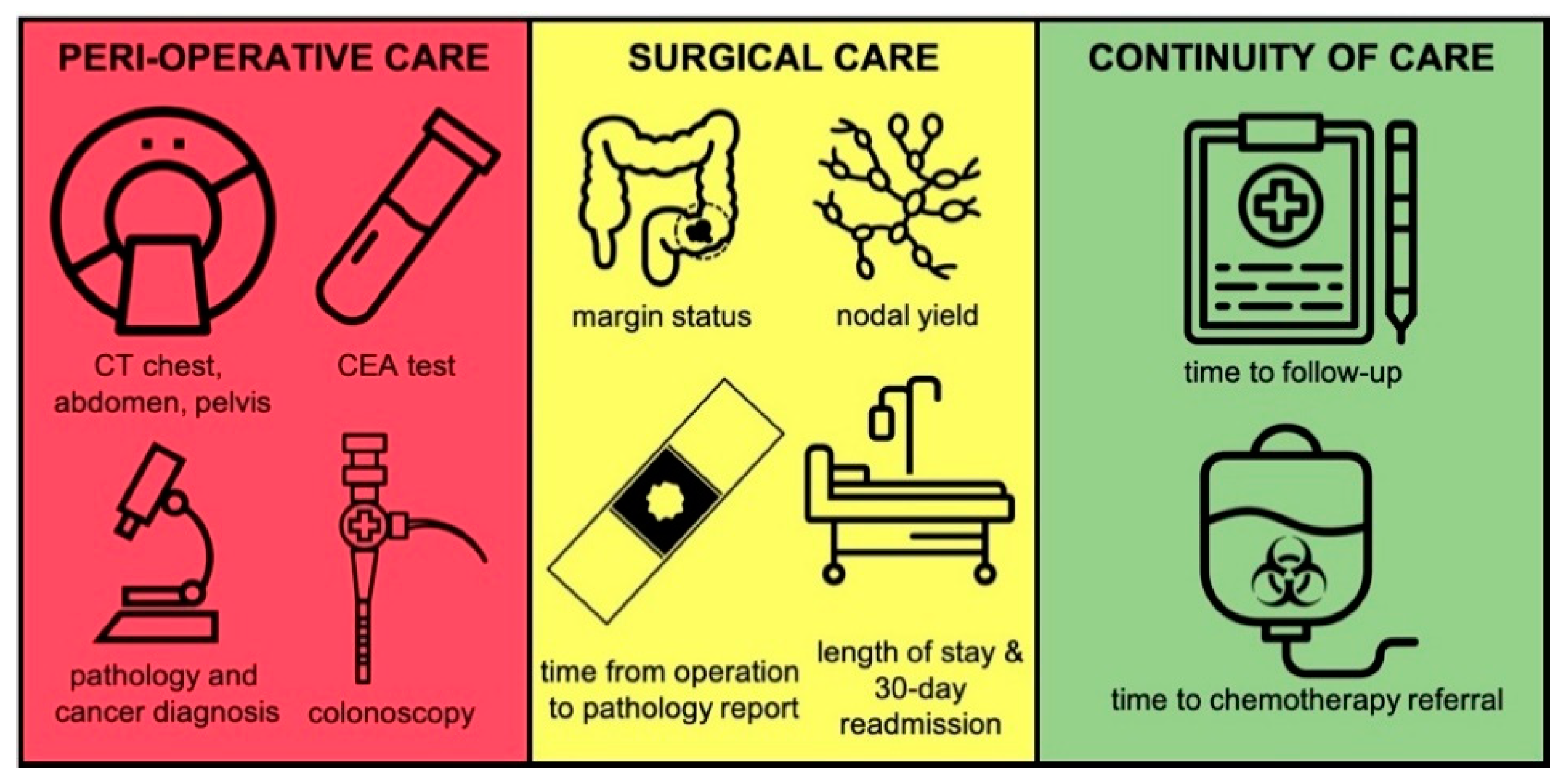
2.2. Outcomes and Baseline Variables
2.3. Statistical Analysis
2.4. Ethics Approval
3. Results
3.1. Baseline Variables
3.2. Outcome Comparison
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef]
- Zielinski, M.D.; Merchea, A.; Heller, S.F.; You, Y.N. Emergency management of perforated colon cancers: How aggressive should we be? J. Gastrointest. Surg. 2011, 15, 2232–2238. [Google Scholar] [CrossRef]
- Gunnarsson, H.; Holm, T.; Ekholm, A.; Olsson, L.I. Emergency presentation of colon cancer is most frequent during summer. Colorectal Dis. 2011, 13, 663–668. [Google Scholar] [CrossRef]
- Alvarez, J.A.; Baldonedo, R.F.; Bear, I.G.; Truán, N.; Pire, G.; Alvarez, P. Presentation, treatment, and multivariate analysis of risk factors for obstructive and perforative colorectal carcinoma. Am. J. Surg. 2005, 190, 376–382. [Google Scholar] [CrossRef]
- Amri, R.; Bordeianou, L.G.; Sylla, P.; Berger, D.L. Colon cancer surgery following emergency presentation: Effects on admission and stage-adjusted outcomes. Am. J. Surg. 2015, 209, 246–253. [Google Scholar] [CrossRef] [PubMed]
- Vogel, J.D.; Eskicioglu, C.; Weiser, M.R.; Feingold, D.L.; Steele, S.R. The American Society of Colon and Rectal Surgeons Clinical Practice Guidelines for the Treatment of Colon Cancer. Dis. Colon Rectum 2017, 60, 999–1017. [Google Scholar] [CrossRef] [PubMed]
- Biagi, J.J.; Raphael, M.J.; Mackillop, W.J.; Kong, W.; King, W.D.; Booth, C.M. Association between time to initiation of adjuvant chemotherapy and survival in colorectal cancer: A systematic review and meta-analysis. JAMA 2011, 305, 2335–2342. [Google Scholar] [CrossRef] [PubMed]
- Wanis, K.N.; Ott, M.; Van Koughnett, J.A.M.; Colquhoun, P.; Brackstone, M. Long-term oncological outcomes following emergency resection of colon cancer. Int. J. Colorectal Dis. 2018, 33, 1525–1532. [Google Scholar] [CrossRef] [PubMed]
- Arhi, C.S.; Burns, E.M.; Bouras, G.; Aylin, P.; Ziprin, P.; Darzi, A. Complications after discharge and delays in adjuvant chemotherapy following colonic resection: A cohort study of linked primary and secondary care data. Colorectal Dis. 2019, 21, 307–314. [Google Scholar] [CrossRef] [PubMed]
- Ha, G.S.; Kim, Y.W.; Choi, E.H. Factors Associated with the Lack of Adjuvant Chemotherapy Following Curative Surgery for Stage II and III Colon Cancer: A Korean National Cohort Study. Anticancer Res. 2017, 37, 915–922. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Becerra, A.Z.; Aquina, C.T.; Hensley, B.J.; Justiniano, C.F.; Boodry, C.; Swanger, A.A.; Arsalanizadeh, R.; Noyes, K.; Monson, J.R.; et al. Emergent Colectomy Is Independently Associated with Decreased Long-Term Overall Survival in Colon Cancer Patients. J. Gastrointest. Surg. 2017, 21, 543–553. [Google Scholar] [CrossRef] [PubMed]
- Ingraham, A.; Nathens, A.; Peitzman, A.; Bode, A.; Dorlac, G.; Dorlac, W.; Miller, P.; Sadeghi, M.; Wasserman, D.D.; Bilimoria, K. Assessment of emergency general surgery care based on formally developed quality indicators. Surgery 2017, 162, 397–407. [Google Scholar] [CrossRef] [PubMed]
- Weixler, B.; Warschkow, R.; Ramser, M.; Droeser, R.; von Holzen, U.; Oertli, D.; Kettelhack, C. Urgent surgery after emergency presentation for colorectal cancer has no impact on overall and disease-free survival: A propensity score analysis. BMC Cancer 2016, 16, 208. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.V.; Brackstone, M. Emergency surgery for colorectal cancer does not result in nodal understaging compared with elective surgery. Can. J. Surg. 2014, 57, 349–353. [Google Scholar] [CrossRef] [PubMed]
- Kye, B.H.; Lee, Y.S.; Cho, H.M.; Kim, J.G.; Oh, S.T.; Lee, I.K.; Kim, H.J. Comparison of Long-Term Outcomes between Emergency Surgery and Bridge to Surgery for Malignant Obstruction in Right-Sided Colon Cancer: A Multicenter Retrospective Study. Ann. Surg. Oncol. 2016, 23, 1867–1874. [Google Scholar] [CrossRef]
- Orive, M.; for the REDISSEC-CARESS/CCR group; Aguirre, U.; Gonzalez, N.; Lázaro, S.; Redondo, M.; Bare, M.; Anula, R.; Briones, E.; Escobar, A.; et al. Risk factors affecting hospital stay among patients undergoing colon cancer surgery: A prospective cohort study. Support Care Cancer 2019, 27, 4133–4144. [Google Scholar] [CrossRef] [PubMed]
- Kelly, M.; Sharp, L.; Dwane, F.; Kelleher, T.; Comber, H. Factors predicting hospital length-of-stay and readmission after colorectal resection: A population-based study of elective and emergency admissions. BMC Health Serv. Res. 2012, 12, 77. [Google Scholar] [CrossRef] [PubMed]
- Capsec, J.; Lefebvre, C.; Chupé, F.; Heitzmann, P.; Raveneau, C.; Dardaine-Giraud, V.; Sauger, C.; Lagasse, J.-P.; Kraft, K.; Linassier, C.; et al. Age and factors associated with access and time to post-operative adjuvant chemotherapy in colon cancer: A French epidemiological study. J. Gastrointest. Oncol. 2017, 8, 842–849. [Google Scholar] [CrossRef] [PubMed]
| Variable | Emergency Cases (n = 23) | Elective Cases (n = 46) | p-Value |
|---|---|---|---|
| Age (years), median (IQR) | 73 (64.4–79.7) | 73.7 (63.3–81.7) | 0.843 |
| Male sex (%) | 13 (56.5%) | 26 (56.5%) | 1.00 |
| ASA Class (%) | 0.01 | ||
| I | 0 (0%) | 1 (2.2%) | |
| II | 1 (4.4%) | 3 (6.5%) | |
| III | 9 (39.1%) | 34 (73.9%) | |
| IV | 13 (56.5%) | 8 (17.4%) | |
| V | 0 (0%) | 0 (0%) | |
| BMI, median (IQR) | 31.9 (27.4–34.7) | 27.9 (24.1–30.9) | 0.412 |
| Subspecialized Surgeon a (%) | 21 (91.3%) | 46 (100%) | 0.042 |
| Type of Operation | 0.046 | ||
| Open right hemicolectomy (%) | 7 (30.4%) | 4 (8.7%) | |
| Laparoscopic right hemicolectomy (%) | 9 (39.1%) | 23 (50.0%) | |
| Segmental colectomy (%) | 2 (8.7%) | 0 (0%) | |
| Laparoscopic segmental colectomy (%) | 3 (13.0%) | 9 (19.6%) | |
| Laparoscopic low anterior resection (%) | 1 (4.4%) | 8 (17.4%) | |
| Open low anterior resection (%) | 0 (0%) | 1 (2.2%) | |
| Laparoscopic low anterior resection + diversion (%) | 1 (4.4%) | 0 (0%) | |
| Laparoscopic Hartmann’s procedure (%) | 0 (0%) | 1 (2.2%) | |
| Approach | 0.001 | ||
| Open (%) | 9 (39.1%) | 3 (6.5%) | |
| Laparoscopic (%) | 9 (39.1%) | 39 (84.8%) | |
| Converted (%) | 2 (8.7%) | 2 (4.4%) | |
| Laparoscopic Assisted (%) | 3 (13.0%) | 2 (4.4%) | |
| Tumour Location | 0.468 | ||
| Right (%) | 15 (65.2%) | 27 (58.7%) | |
| Transverse (%) | 3 (13.0%) | 4 (8.7%) | |
| Left (%) | 2 (8.7%) | 2 (4.4%) | |
| Sigmoid (%) | 3 (13.0%) | 13 (28.3%) | |
| Cancer diagnosed pre-operatively | 0.108 | ||
| Yes | 20 (87%) | 45 (97.8%) | |
| No | 1 (4.4%) | 1 (2.2%) | |
| On differential but unconfirmed | 2 (8.7%) | 0 (0%) | |
| Pre-operative pathology status | 0.299 | ||
| Negative | 2 (8.7%) | 2 (4.4%) | |
| Positive | 14 (60.9%) | 40 (87.0%) | |
| Pre-operative colonoscopy | <0.001 | ||
| Yes | 16 (69.6%) | 44 (95.7%) | |
| No | 7 (30.4%) | 0 (0%) | |
| Attempted but uncompleted | 0 (0%) | 2 (4.4%) | |
| Pathologic T Stage | 0.092 | ||
| 1 (%) | 0 (0%) | 1 (2.2%) | |
| 2 (%) | 1 (4.4%) | 6 (13.0%) | |
| 3 (%) | 15 (65.2%) | 35 (76.1%) | |
| 4 (%) | 7 (30.4%) | 4 (8.7%) | |
| Pathologic N Stage | 0.255 | ||
| 0 (%) | 11 (47.8%) | 22 (47.8%) | |
| 1 (%) | 4 (17.4%) | 15 (32.6%) | |
| 2 (%) | 8 (34.8%) | 9 (19.6%) | |
| Pathologic M Stage | 1.000 | ||
| 0 (%) | 23 (100%) | 46 (100%) | |
| 1 (%) | 0 (0%) | 0 (0%) |
| Variable | Emergency Cases (n = 23) | Elective Cases (n = 46) | Odds Ratio (95% CI) | p-Value |
|---|---|---|---|---|
| CT staging within 30 days (%) | 20 (87%) | 46 (100%) | N/A | 0.012 |
| CEA within 30 days (%) | 16 (70%) | 44 (97%) | 0.104 (0.020–0.553) | 0.020 |
| Longitudinal margin | 1.000 | |||
| Gross negative | 23 (100%) | 46 (100%) | N/A | |
| Microscopic negative | 23 (100%) | 46 (100%) | N/A | |
| Circumferential margin | ||||
| Gross negative | 23 (100%) | 46 (100%) | N/A | 1.000 |
| Microscopic negative | 20 (87%) | 45 (97.8%) | N/A | 0.069 |
| Nodal yield, median (IQR) | 28 (19–33.5) | 27 (20–37) | N/A | 0.819 |
| Time from operation to pathology report, median days (IQR) | 16 (13.5–19.5) | 13.5 (10.25–18.75) | N/A | 0.058 |
| Length of stay, days, median (IQR) | 9 (6–13) | 2.5 (1–5) | N/A | <0.001 |
| 30-day readmission (%) | 6 (26%) | 2 (4%) | 3.706 (0.928–14.803) | 0.053 |
| Medical oncology referral a | 16 (70%) | 39 (85%) | 0.737 (0.236–2.303) | 0.599 |
| Time from operation to medical oncology referral, days, median (IQR) | 30 (18–33) | 27 (21.5–34) | N/A | 0.346 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guidolin, K.; Withers, R.; Shariff, F.; Ashamalla, S.; Nadler, A. Quality of Colon Cancer Care in Patients Undergoing Emergency Surgery. Curr. Oncol. 2021, 28, 2079-2086. https://doi.org/10.3390/curroncol28030192
Guidolin K, Withers R, Shariff F, Ashamalla S, Nadler A. Quality of Colon Cancer Care in Patients Undergoing Emergency Surgery. Current Oncology. 2021; 28(3):2079-2086. https://doi.org/10.3390/curroncol28030192
Chicago/Turabian StyleGuidolin, Keegan, Rebecca Withers, Farhana Shariff, Shady Ashamalla, and Ashlie Nadler. 2021. "Quality of Colon Cancer Care in Patients Undergoing Emergency Surgery" Current Oncology 28, no. 3: 2079-2086. https://doi.org/10.3390/curroncol28030192
APA StyleGuidolin, K., Withers, R., Shariff, F., Ashamalla, S., & Nadler, A. (2021). Quality of Colon Cancer Care in Patients Undergoing Emergency Surgery. Current Oncology, 28(3), 2079-2086. https://doi.org/10.3390/curroncol28030192





