Geographic and Socioeconomic Disparity of Gastric Cancer Patients in Canada
Abstract
1. Introduction
2. Materials and Methods
2.1. Data Extraction
2.2. Data Analysis
2.3. Mapping Analysis
2.4. SES/Ethnicity Analysis
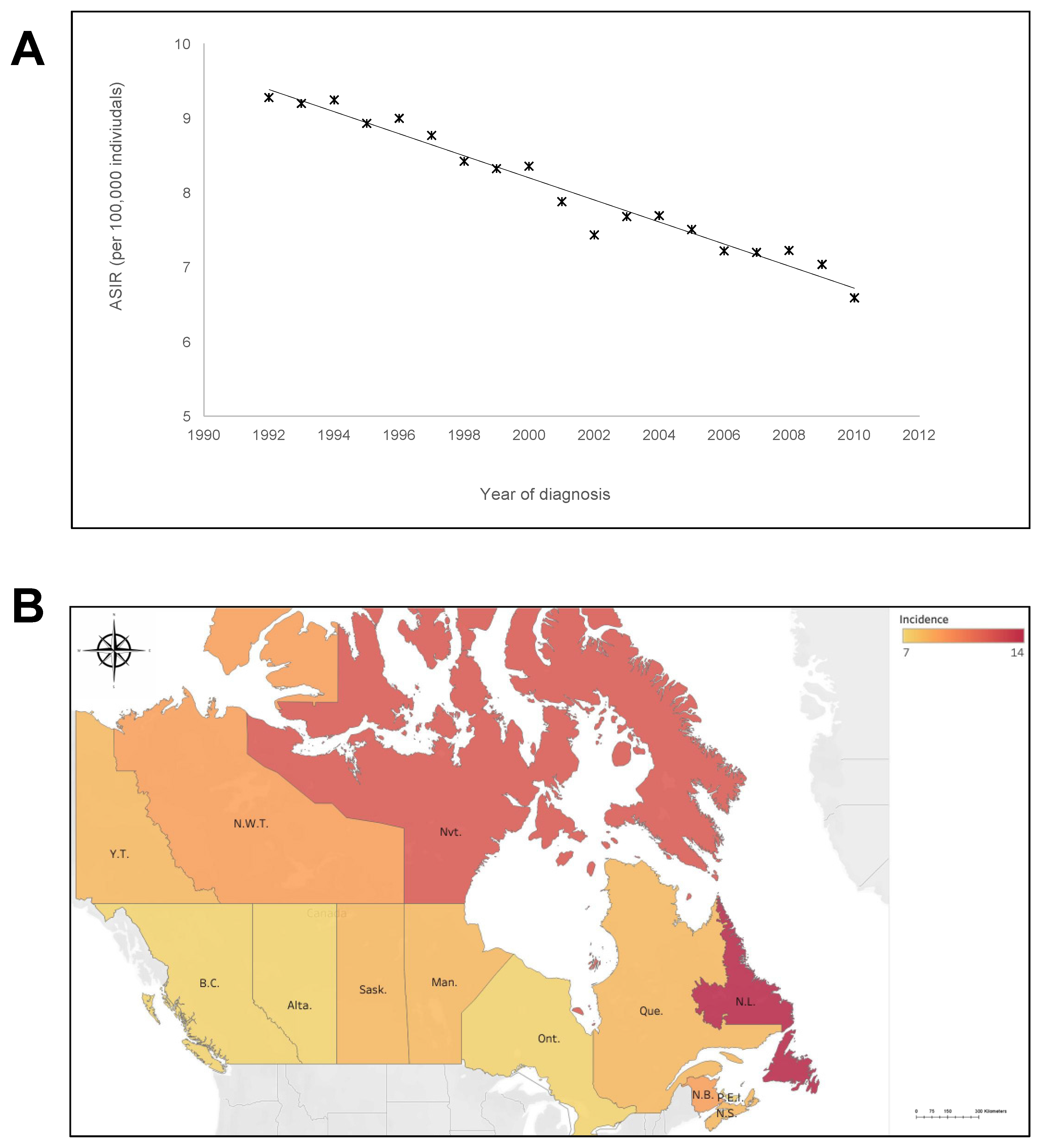
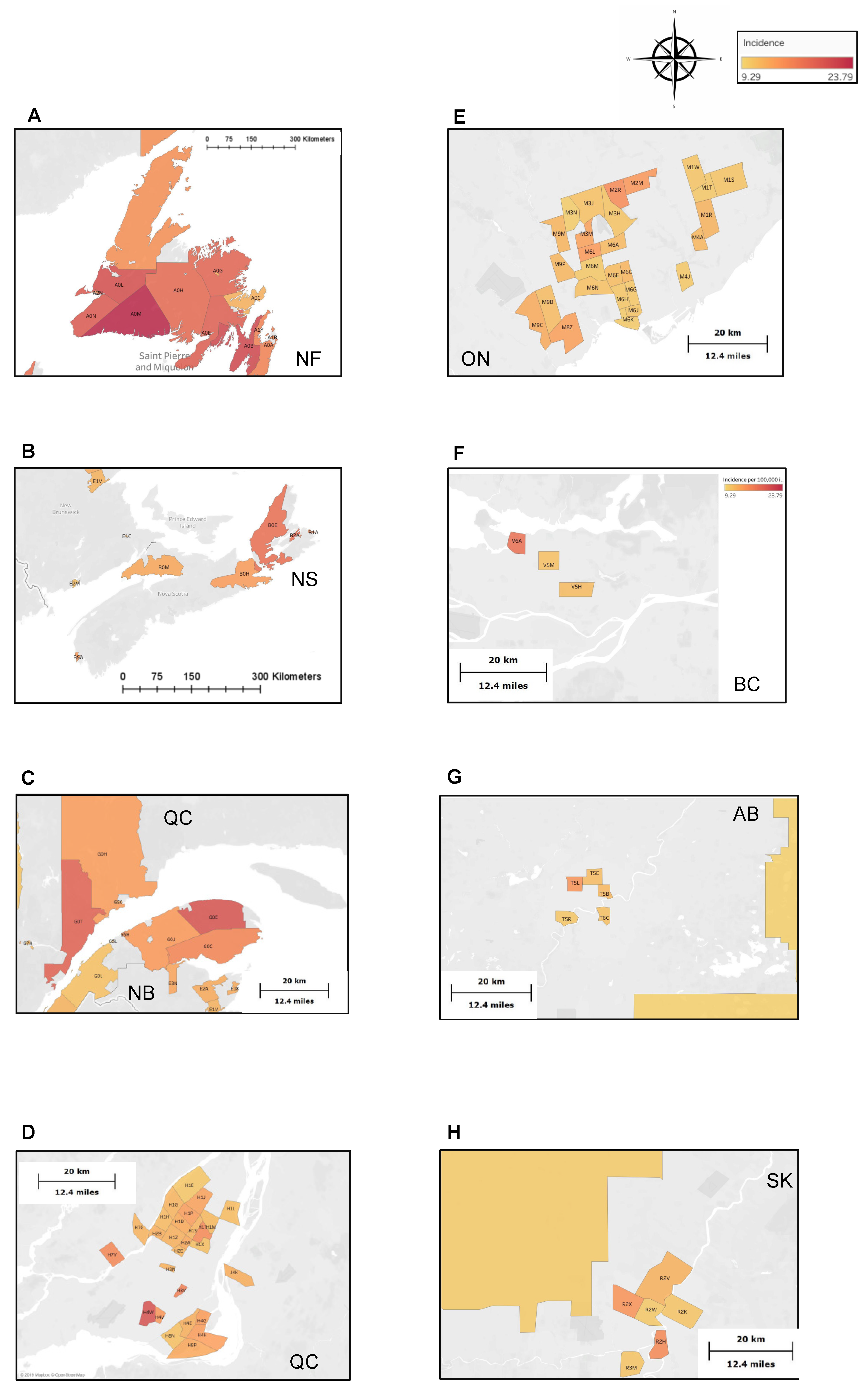
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Canadian Cancer Statistics Advisory Committee. Canadian Cancer Statistics; Canadian Cancer Society: Toronto, ON, Canada, 2019. [Google Scholar]
- Karimi, P.; Islami, F.; Anandasabapathy, S.; Freedman, N.D.; Kamangar, F. Gastric Cancer: Descriptive Epidemiology, Risk Factors, Screening, and Prevention. Cancer Epidemiol. Biomark. Prev. 2014, 23, 700–713. [Google Scholar] [CrossRef]
- Rawla, P.; Barsouk, A. Epidemiology of gastric cancer: Global trends, risk factors and prevention. Gastroenterol. Rev. 2019, 14, 26–38. [Google Scholar] [CrossRef]
- Kim, S.; Hwang, J. Assessment of trends in socioeconomic inequalities in cancer screening services in Korea, 1998–2012. Int. J. Equity Health 2016, 15, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Kim, G.H.; Liang, P.S.; Bang, S.J.; Hwang, J.H. Screening and surveillance for gastric cancer in the United States: Is it needed? Gastrointest. Endosc. 2016, 84, 18–28. [Google Scholar] [CrossRef]
- Ang, T.L.; Fock, K.M. Clinical epidemiology of gastric cancer. Singap. Med. J. 2014, 55, 621–628. [Google Scholar] [CrossRef]
- Casamayor, M.; Morlock, R.; Maeda, H.; Anjani, J. Targeted literature review of the global burden of gastric cancer. Ecancermedicalscience 2018, 12, 883. [Google Scholar] [CrossRef]
- Charalampakis, N.; Economopoulou, P.; Kotsantis, I.; Tolia, M.; Schizas, D.; Liakakos, T.; Elimova, E.; Ajani, J.A.; Psyrri, A. Medical management of gastric cancer: A 2017 update. Cancer Med. 2017, 7, 123–133. [Google Scholar] [CrossRef]
- Correa, P. Gastric cancer: Overview. Gastroenterol. Clin. N. Am. 2013, 42, 211–217. [Google Scholar] [CrossRef]
- Guggenheim, D.E.; Shah, M.A. Gastric cancer epidemiology and risk factors. J. Surg. Oncol. 2012, 107, 230–236. [Google Scholar] [CrossRef] [PubMed]
- Choi, C.E.; Sonnenberg, A.; Turner, K.; Genta, R.M. High Prevalence of Gastric Preneoplastic Lesions in East Asians and Hispanics in the USA. Dig. Dis. Sci. 2015, 60, 2070–2076. [Google Scholar] [CrossRef]
- Kim, Y.; Park, J.; Nam, B.-H.; Ki, M. Stomach cancer incidence rates among Americans, Asian Americans and Native Asians from 1988 to 2011. Epidemiol. Health 2015, 37, e2015006. [Google Scholar] [CrossRef]
- Gupta, S.; Tao, L.; Murphy, J.D.; Camargo, M.C.; Oren, E.; Valasek, M.A.; Gomez, S.L.; Martinez, M.E. Race/Ethnicity-, Socioeconomic Status-, and Anatomic Subsite-Specific Risks for Gastric Cancer. Gastroenterology 2019, 156, 59–62.e4. [Google Scholar] [CrossRef]
- Petrelli, A.; Di Napoli, A.; Sebastiani, G.; Rossi, A.; Rossi, P.G.; Demuru, E.; Costa, G.; Zengarini, N.; Alicandro, G.; Marchetti, S.; et al. Italian Atlas of mortality inequalities by education level. Epidemiol. Prev. 2019, 43, 1–120. [Google Scholar] [PubMed]
- Lugn, N.E. Global health care–bridging the gap. J. Telemed. Telecare 2006, 12, 109–110. [Google Scholar] [CrossRef]
- The Lancet Global Health. Bridging the global health gap. Lancet Glob. Health 2016, 4, e579. [Google Scholar] [CrossRef]
- Tsang, M.; Le, M.; Ghazawi, F.M.; Cyr, J.; Alakel, A.; Rahme, E.; Lagacé, F.; Netchiporouk, E.; Moreau, L.; Zubarev, A.; et al. Multiple myeloma epidemiology and patient geographic distribution in Canada: A population study. Cancer 2019, 125, 2435–2444. [Google Scholar] [CrossRef]
- Le, M.; Ghazawi, F.M.; Rahme, E.; Alakel, A.; Netchiporouk, E.; Savin, E.; Zubarev, A.; Glassman, S.J.; Sasseville, D.; Popradi, G.; et al. Identification of significant geographic clustering of polycythemia vera cases in Montreal, Canada. Cancer 2019, 125, 3953–3959. [Google Scholar] [CrossRef]
- Le, M.; Ghazawi, F.M.; Alakel, A.; Netchiporouk, E.; Rahme, E.; Zubarev, A.; Powell, M.; Moreau, L.; Roshdy, O.; Glassman, S.J.; et al. Incidence and Mortality Trends and Geographic Patterns of Follicular Lymphoma in Canada. Curr. Oncol. 2019, 26, 473–481. [Google Scholar] [CrossRef]
- Lagacé, F.; Ghazawi, F.M.; Le, M.; Savin, E.; Zubarev, A.; Powell, M.; Moreau, L.; Sasseville, D.; Popa, I.; Litvinov, I.V. Penile Invasive Squamous Cell Carcinoma: Analysis of Incidence, Mortality Trends, and Geographic Distribution in Canada. J. Cutan. Med. Surg. 2019, 24, 124–128. [Google Scholar] [CrossRef]
- Lagacé, F.; Ghazawi, F.M.; Le, M.; Rahme, E.; Savin, E.; Zubarev, A.; Alakel, A.; Sasseville, D.; Moreau, L.; Meterissian, S.; et al. Analysis of incidence, mortality trends, and geographic distribution of breast cancer patients in Canada. Breast Cancer Res. Treat. 2019, 178, 683–691. [Google Scholar] [CrossRef]
- Ghazawi, F.M.; Ramanakumar, A.V.; Alakel, A.; Lagacé, F.; Chen, A.; Le, M.; Nedjar, H.; Sasseville, D.; Rahme, E.; Litvinov, I.V. Incidence of acute myeloid leukemia: A regional analysis of Canada. Cancer 2019, 126, 1356–1361. [Google Scholar] [CrossRef]
- Ghazawi, F.M.; Netchiporouk, E.; Rahme, E.; Tsang, M.; Moreau, L.; Glassman, S.; Provost, N.; Gilbert, M.; Jean, S.-E.; Roshdy, O.; et al. Distribution and Clustering of Cutaneous T-Cell Lymphoma (CTCL) Cases in Canada During 1992 to 2010. J. Cutan. Med. Surg. 2017, 22, 154–165. [Google Scholar] [CrossRef]
- Ghazawi, F.M.; Netchiporouk, E.; Rahme, E.; Tsang, M.; Moreau, L.; Glassman, S.; Provost, N.; Gilbert, M.; Jean, S.-E.; Pehr, K.; et al. Comprehensive analysis of cutaneous T-cell lymphoma (CTCL) incidence and mortality in Canada reveals changing trends and geographic clustering for this malignancy. Cancer 2017, 123, 3550–3567. [Google Scholar] [CrossRef]
- Ghazawi, F.M.; Lu, J.; Savin, E.; Zubarev, A.; Chauvin, P.; Sasseville, D.; Zeitouni, A.; Litvinov, I.V. Epidemiology and Patient Distribution of Oral Cavity and Oropharyngeal SCC in Canada. J. Cutan. Med. Surg. 2020, 24, 340–349. [Google Scholar] [CrossRef]
- Ghazawi, F.M.; Le, M.; Lagacé, F.; Cyr, J.; AlGhazawi, N.; Zubarev, A.; Roy, S.F.; Rahme, E.; Netchiporouk, E.; Roshdy, O.; et al. Incidence, Mortality, and Spatiotemporal Distribution of Cutaneous Malignant Melanoma Cases Across Canada. J. Cutan. Med. Surg. 2019, 23, 394–412. [Google Scholar] [CrossRef]
- Ghazawi, F.M.; Le Bsc, M.; Cyr, J.; Netchiporouk, E.; Rahme, E.; Alakel, A.; Zubarev, A.; Powell, M.; Moreau, L.; Roshdy, O.; et al. Analysis of acute myeloid leukemia incidence and geographic distribution in Canada from 1992 to 2010 reveals disease clusters in Sarnia and other industrial US border cities in Ontario. Cancer 2019, 125, 1886–1897. [Google Scholar] [CrossRef]
- Ghazawi, F.M.; Le, M.; Alghazawi, N.; Rahme, E.; Moreau, L.; Netchiporouk, E.; Zubarev, A.; Roshdy, O.; Glassman, S.J.; Sasseville, D.; et al. Trends in incidence of cutaneous malignant melanoma in Canada: 1992–2010 versus 2011–2015. J. Am. Acad. Dermatol. 2019, 80, 1157–1159. [Google Scholar] [CrossRef]
- Ghazawi, F.M.; Darwich, R.; Le, M.; Rahme, E.; Zubarev, A.; Moreau, L.; Burnier, J.V.; Sasseville, D.; Burnier, M.N.; Litvinov, I.V. Uveal melanoma incidence trends in Canada: A national comprehensive population-based study. Br. J. Ophthalmol. 2019, 103, 1872–1876. [Google Scholar] [CrossRef]
- Ghazawi, F.M.; Cyr, J.; Darwich, R.; Le, M.; Rahme, E.; Moreau, L.; Netchiporouk, E.; Zubarev, A.; Roshdy, O.; Glassman, S.J.; et al. Cutaneous malignant melanoma incidence and mortality trends in Canada: A comprehensive population-based study. J. Am. Acad. Dermatol. 2019, 80, 448–459. [Google Scholar] [CrossRef]
- Darwich, R.; Ghazawi, F.M.; Rahme, E.; Alghazawi, N.; Zubarev, A.; Moreau, L.; Sasseville, D.; Jr, M.N.B.; Litvinov, I.V. Epidemiology of ophthalmic lymphoma in Canada during 1992–2010. Br. J. Ophthalmol. 2019, 104, 1176–1180. [Google Scholar] [CrossRef]
- Cattelan, L.; Ghazawi, F.M.; Le, M.; Lagacé, F.; Savin, E.; Zubarev, A.; Gantchev, J.; Tomaszewski, M.; Sasseville, D.; Waschke, K.; et al. Epidemiologic trends and geographic distribution of esophageal cancer in Canada: A national population-based study. Cancer Med. 2020, 9, 401–417. [Google Scholar] [CrossRef]
- Lagacé, F.; Ghazawi, F.; Le, M.; Savin, E.; Zubarev, A.; Powell, M.; Moreau, L.; Sasseville, D.; Popa, I.; Litvinov, I. Incidence and Mortality of Prostate Cancer in Canada during 1992–2010. Curr. Oncol. 2021, 28, 96. [Google Scholar] [CrossRef]
- Xiao, Y.; Cattelan, L.; Lagacé, F.; Ghazawi, F.M.; Alakel, A.; Grose, E.; Le, M.; Nechaev, V.; Sasseville, D.; Waschke, K.; et al. Epidemiologic trends and geographic distribution of patients with gallbladder and extrahepatic biliary tract cancers in Canada. HPB 2021. [Google Scholar] [CrossRef] [PubMed]
- 2006 Census of Population; Statistics Canada: Ottawa, ON, Canada, 2006.
- Venerito, M.; Vasapolli, R.; Rokkas, T.; Malfertheiner, P. Gastric cancer: Epidemiology, prevention, and therapy. Helicobacter 2018, 23 (Suppl. S1), e12518. [Google Scholar] [CrossRef]
- Pasechnikov, V.; Chukov, S.; Fedorov, E.; Kikuste, I.; Leja, M. Gastric cancer: Prevention, screening and early diagnosis. World J. Gastroenterol. 2014, 20, 13842–13862. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Graham, D.Y.; Khan, A.; Balakrishnan, M.; Abrams, H.R.; El-Serag, H.B.; Thrift, A.P. Incidence of gastric cancer in the USA during 1999 to 2013: A 50-state analysis. Int. J. Epidemiol. 2018, 47, 966–975. [Google Scholar] [CrossRef]
- D’Elia, L.; Rossi, G.; Ippolito, R.; Cappuccio, F.P.; Strazzullo, P. Habitual salt intake and risk of gastric cancer: A meta-analysis of prospective studies. Clin. Nutr. 2012, 31, 489–498. [Google Scholar] [CrossRef]
- Tramacere, I.; Negri, E.; Pelucchi, C.; Bagnardi, V.; Rota, M.; Scotti, L.; Islami, F.; Corrao, G.; La Vecchia, C.; Boffetta, P. A meta-analysis on alcohol drinking and gastric cancer risk. Ann. Oncol. 2012, 23, 28–36. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Liu, L.; Wang, X.; Wang, J.; Yan, Z.; Cheng, J.; Gong, G.; Li, G. Body Mass Index and Risk of Gastric Cancer: A Meta-analysis of a Population with More Than Ten Million from 24 Prospective Studies. Cancer Epidemiol. Biomark. Prev. 2013, 22, 1395–1408. [Google Scholar] [CrossRef]
- Canada, S. Fruit and Vegetable Consumption; Statistics Canada: Ottawa, ON, Canada, 2008.
- Tarasuk, V.; Mitchell, A.; Dachner, N. Research to identify policy options to reduce food insecurity. Household Food Insecurity in Canada 2011; Toronto, ON, Canada, 2013. Available online: https://proof.utoronto.ca/ (accessed on 1 May 2021).
- Canada PHAo. Obesity in Canada: A Joint Report from the Public Health Agency of Canada. And the Canadian Institute for Health Information; Canadian Institute for Health Information: Ottawa, ON, Canada, 2011. [Google Scholar]
- Food First NL Summary and Discussion Guide; Newfoundland and Labrador Public Health Association: St. John’s, NL, Canada, 2015.
- NYC Full Service Grocery Store Analysis; NYC Department of Health and Mental Hygiene: New York, NY, USA, 2010.
- Grocery Stores in Canadian Urban Centres; University of Alberta School of Retailing: Edmonton, AB, Canada, 2015.
- Quebec First Nations. 500 Nations; QC, Canada, 2020. Available online: https://www.500nations.com/Quebec_Tribes.asp (accessed on 1 May 2021).
- We’koqma’q First Nation; Nova Scotia Office of Aboriginal Affairs: Halifax, NS, Canada, 2020.
- Michelin, O. Reserves in Newfoundland and Labrador. In The Canadian Encyclopedia; Historica Canada: Toronto, ON, Canada, 2019; Available online: https://www.thecanadianencyclopedia.ca/en/article/reserves-in-newfoundland-and-labrador (accessed on 1 May 2021).
- Colquhoun, A.; Hannah, H.; Corriveau, A.; Hanley, B.; Yuan, Y.; Goodman, K.J.; The CANHelp Working Group. Gastric Cancer in Northern Canadian Populations: A Focus on Cardia and Non-Cardia Subsites. Cancers 2019, 11, 534. [Google Scholar] [CrossRef] [PubMed]
- Aboriginal Food Security in Northern Canada: An Assessment of the State of Knowledge; Council of Canadian Academics: Ottawa, ON, Canada, 2014.
- Food Insecurity among Inuit living in Inuit Nunangat; Statistics Canada: Ottawa, ON, Canada, 2017.
- Egeland, G.M.; Johnson-Down, L.; Cao, Z.R.; Sheikh, N.; Weiler, H. Food Insecurity and Nutrition Transition Combine to Affect Nutrient Intakes in Canadian Arctic Communities. J. Nutr. 2011, 141, 1746–1753. [Google Scholar] [CrossRef] [PubMed]
- Kirkpatrick, S.I.; Tarasuk, V. Food Insecurity Is Associated with Nutrient Inadequacies among Canadian Adults and Adolescents. J. Nutr. 2008, 138, 604–612. [Google Scholar] [CrossRef] [PubMed]
- To, Q.G.; Frongillo, E.A.; Gallegos, D.; Moore, J.B. Household food insecurity is associated with less physical activity among children and adults in the U.S. population. J. Nutr. 2014, 144, 1797–1802. [Google Scholar] [CrossRef] [PubMed]
- Bhawra, J.; Cooke, M.J.; Guo, Y.; Wilk, P. The association of household food security, household characteristics and school environment with obesity status among off-reserve First Nations and Metis children and youth in Canada: Results from the 2012 Aboriginal Peoples Survey. Health Promot. Chronic Dis. Prev. Can. 2017, 37, 77–86. [Google Scholar] [CrossRef]
- Nutrition North Canada: Results from 2011–2016; Government of Canada: Ottawa, ON, Canada, 2016.
- Paszat, L.; Sutradhar, R.; Liu, Y.; Baxter, N.N.; Tinmouth, J.; Rabeneck, L. Risk of colorectal cancer among immigrants to Ontario, Canada. BMC Gastroenterol. 2017, 17, 85. [Google Scholar] [CrossRef] [PubMed]
- Lindsay, C. The Latin American Community in Canada; Social and Aboriginal Statistics Division; Statistics Canada: Ottawa, ON, Canada, 2001.
- Chang, E.T.; Gomez, S.L.; Fish, K.; Schupp, C.W.; Parsonnet, J.; DeRouen, M.C.; Keegan, T.H.M.; Clarke, C.A.; Glaser, S.L. Gastric Cancer Incidence among Hispanics in California: Patterns by Time, Nativity, and Neighborhood Characteristics. Cancer Epidemiol. Biomark. Prev. 2012, 21, 709–719. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Chen, V.W.; Ruiz, B.; Andrews, P.; Su, L.J.; Correa, P. Incidence of esophageal and gastric carcinomas among American Asians/Pacific Islanders, whites, and blacks: Subsite and histology differences. Cancer 2006, 106, 683–692. [Google Scholar] [CrossRef]
- 2016 Census of Population; Statistics Canada: Ottawa, ON, Canada, 2016.
- Sarkar, S.; Dauer, M.J.; In, H. Socioeconomic Disparities in Gastric Cancer and Identification of a Single SES Variable for Predicting Risk. J. Gastrointest. Cancer 2021, 1–9. [Google Scholar] [CrossRef]
- Mancini, S.; Ravaioli, A.; Giuliani, O.; Giorgetti, S.; Falcini, F.; Colamartini, A.; Bucchi, L. Gastric cancer incidence in the Romagna Region of Italy: A spatial and temporal analysis. Dig. Liver Dis. 2015, 47, 1076–1081. [Google Scholar] [CrossRef]
- Crew, K.D.; Neugut, A.I. Epidemiology of gastric cancer. World J. Gastroenterol. 2006, 12, 354–362. [Google Scholar] [CrossRef]
- Webb, P.M.; Knight, T.; Greaves, S.; Wilson, A.; Newell, D.G.; Elder, J.; Forman, D. Relation between infection with Helicobacter pylori and living conditions in childhood: Evidence for person to person transmission in early life. BMJ 1994, 308, 750–753. [Google Scholar] [CrossRef]
- Buckley, M.J.M.; O’shea, J.; Grace, A.; English, L.; Keane, C.; Hourihan, D.; O’morain, C.A. A community-based study of the epidemiology of Helicobacter pylori infection and associated asymptomatic gastroduodenal pathology. Eur. J. Gastroenterol. Hepatol. 1998, 10, 375–380. [Google Scholar] [CrossRef]
- Kurosawa, M.; Kikuchi, S.; Inaba, Y.; Ishibashi, T.; Kobayashi, F. Helicobacter pylori infection among Japanese children. J. Gastroenterol. Hepatol. 2000, 15, 1382–1385. [Google Scholar] [CrossRef]
- Krieger, N.; Williams, D.R.; Moss, N.E. Measuring Social Class in US Public Health Research: Concepts, Methodologies, and Guidelines. Annu. Rev. Public Health 1997, 18, 341–378. [Google Scholar] [CrossRef] [PubMed]
- Engel, L.S.; Vaughan, T.L.; Gammon, M.D.; Chow, W.-H.; Risch, H.A.; Dubrow, R.; Mayne, S.T.; Rotterdam, H.; Schoenberg, J.B.; Stanford, J.L.; et al. Occupation and risk of esophageal and gastric cardia adenocarcinoma. Am. J. Ind. Med. 2002, 42, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Hemminki, K.; Zhang, H.; Czene, K. Socioeconomic factors in cancer in Sweden. Int. J. Cancer 2003, 105, 692–700. [Google Scholar] [CrossRef] [PubMed]
- Lagergren, J.; Andersson, G.; Talbäck, M.; Drefahl, S.; Bihagen, E.; Härkönen, J.; Feychting, M.; Ljung, R. Marital status, education, and income in relation to the risk of esophageal and gastric cancer by histological type and site. Cancer 2015, 122, 207–212. [Google Scholar] [CrossRef]
- Nagel, G.; Linseisen, J.; Boshuizen, H.C.; Pera, G.; Del Giudice, G.; Westert, G.P.; Bueno-De-Mesquita, H.B.; Allen, N.E.; Key, T.J.; Numans, M.E.; et al. Socioeconomic position and the risk of gastric and oesophageal cancer in the European Prospective Investigation into Cancer and Nutrition (EPIC-EURGAST). Int. J. Epidemiol. 2007, 36, 66–76. [Google Scholar] [CrossRef] [PubMed]
- Zali, H.; Rezaei-Tavirani, M.; Azodi, M.Z. Gastric cancer: Prevention, risk factors and treatment. Gastroenterol. Hepatol. Bed Bench 2011, 4, 175–185. [Google Scholar] [PubMed]
- Banks, M.; Graham, D.; Jansen, M.; Gotoda, T.; Coda, S.; Di Pietro, M.; Uedo, N.; Bhandari, P.; Pritchard, D.M.; Kuipers, E.J.; et al. British Society of Gastroenterology guidelines on the diagnosis and management of patients at risk of gastric adenocarcinoma. Gut 2019, 68, 1545–1575. [Google Scholar] [CrossRef]
- Ohata, H.; Oka, M.; Yanaoka, K.; Shimizu, Y.; Mukoubayashi, C.; Mugitani, K.; Iwane, M.; Nakamura, H.; Tamai, H.; Arii, K.; et al. Gastric cancer screening of a high-risk population in Japan using serum pepsinogen and barium digital radiography. Cancer Sci. 2005, 96, 713–720. [Google Scholar] [CrossRef] [PubMed]
- Jun, J.K.; Choi, K.S.; Lee, H.-Y.; Suh, M.; Park, B.; Song, S.H.; Jung, K.W.; Lee, C.W.; Choi, I.J.; Park, E.-C.; et al. Effectiveness of the Korean National Cancer Screening Program in Reducing Gastric Cancer Mortality. Gastroenterology 2017, 152, 1319–1328.e7. [Google Scholar] [CrossRef] [PubMed]
- Choi, K.S.; Jun, J.K.; Lee, H.-Y.; Park, S.; Jung, K.W.; Han, M.A.; Choi, I.J.; Park, E.-C. Performance of gastric cancer screening by endoscopy testing through the National Cancer Screening Program of Korea. Cancer Sci. 2011, 102, 1559–1564. [Google Scholar] [CrossRef] [PubMed]
| Gastric Adenocarcinoma Subtypes | ICD-O-3 Code | No. of Patients | Percentage of Total |
|---|---|---|---|
| Adenocarcinoma | 8140 | 29,135 | 66.3 |
| Intestinal adenocarcinoma | 8144 | 4525 | 10.3 |
| Diffuse adenocarcinoma | 8145 | 1535 | 3.6 |
| Tubular adenocarcinoma | 8211 | 430 | 1.0 |
| Papillary adenocarcinoma | 8260 | 220 | 0.5 |
| Mucinous adenocarcinoma | 8480 | 825 | 1.9 |
| Signet ring cell carcinoma | 8490 | 6955 | 15.8 |
| Medullary carcinoma | 8512 | 5 | 0.01 |
| Adenosquamous carcinoma | 8560 | 165 | 0.3 |
| Undifferentiated carcinoma | 8020 | 160 | 0.3 |
| Overall Gastric Adenocarcinoma | - | 43,955 | 100 |
| Patient Demographics | Incidence |
|---|---|
| Total number of patients | 43,955 |
| By sex | |
| Number of males (%) | 28,830 (66%) |
| Number of females (%) | 15,125 (34%) |
| By age group | |
| <10 | 0 (0%) |
| 10–19 | 5 (0.01%) |
| 20–29 | 175 (4.0%) |
| 30–39 | 990 (2.3%) |
| 40–49 | 3155 (7.2%) |
| 50–59 | 6490 (15%) |
| 60–69 | 11,005 (25%) |
| 70–79 | 13,800 (31%) |
| 80–89 | 7490 (17%) |
| 90+ | 825 (1.9%) |
| Average age | 68.36 ± 0.51 |
| Average ASIR in Males in number of cases per 100,000 individuals per year (95% CI) | 6.74 (6.64–6.83) |
| Average ASIR in Females in number of cases per 100,000 individuals per year (95% CI) | 3.41 (3.34–3.47) |
| A: Socioeconomic Status | |||
| Quintile | Median Income ($) | Incidence Rate (per 100,000) | Incidence Rate Ratio (95% CI) |
| Q1 | <20,000 | 9.17 | - |
| Q2 | 20,000–25,000 | 7.35 | 0.80 (0.78–0.82) |
| Q3 | 25,000–30,000 | 5.86 | 0.64 (0.62–0.66) |
| Q4 | 30,000–35,000 | 4.43 | 0.48 (0.46–0.51) |
| Q5 | >35,000 | 3.53 | 0.39 (0.35–0.42) |
| B: Ethnicity | |||
| Quintile | Percentage of Hispanic Individuals (%) | Incidence Rate (per 100,000) | Incidence Rate Ratio (95% CI) |
| Q1 | 0.00 | 7.69 | - |
| Q2 | 0.01–1.99 | 7.17 | 0.93 (0.89–0.98) |
| Q3 | 2.00–3.99 | 7.52 | 0.98 (0.92–1.03) |
| Q4 | 4.00–5.99 | 9.61 | 1.25 (1.15–1.35) |
| Q5 | >6.00 | 11.32 | 1.47 (1.35–1.60) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cattelan, L.; Ghazawi, F.M.; Le, M.; Lagacé, F.; Rahme, E.; Zubarev, A.; Sasseville, D.; Litvinov, I.V.; Waschke, K.A.; Netchiporouk, E. Geographic and Socioeconomic Disparity of Gastric Cancer Patients in Canada. Curr. Oncol. 2021, 28, 2052-2064. https://doi.org/10.3390/curroncol28030190
Cattelan L, Ghazawi FM, Le M, Lagacé F, Rahme E, Zubarev A, Sasseville D, Litvinov IV, Waschke KA, Netchiporouk E. Geographic and Socioeconomic Disparity of Gastric Cancer Patients in Canada. Current Oncology. 2021; 28(3):2052-2064. https://doi.org/10.3390/curroncol28030190
Chicago/Turabian StyleCattelan, Leila, Feras M. Ghazawi, Michelle Le, François Lagacé, Elham Rahme, Andrei Zubarev, Denis Sasseville, Ivan V. Litvinov, Kevin A. Waschke, and Elena Netchiporouk. 2021. "Geographic and Socioeconomic Disparity of Gastric Cancer Patients in Canada" Current Oncology 28, no. 3: 2052-2064. https://doi.org/10.3390/curroncol28030190
APA StyleCattelan, L., Ghazawi, F. M., Le, M., Lagacé, F., Rahme, E., Zubarev, A., Sasseville, D., Litvinov, I. V., Waschke, K. A., & Netchiporouk, E. (2021). Geographic and Socioeconomic Disparity of Gastric Cancer Patients in Canada. Current Oncology, 28(3), 2052-2064. https://doi.org/10.3390/curroncol28030190







