Assessment of Breast Cancer Surgery in Manitoba: A Descriptive Study
Abstract
1. Introduction
2. Experimental Section
2.1. Study Design and Data Sources
2.2. Study Population
2.3. Definition of Surgical Treatment
2.4. Outcomes
2.5. Statistical Analysis
3. Results
3.1. Surgical Treatment Patterns
3.2. Quality of Care
3.3. Post-Operative Outcomes
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Health Council of Canada. Which Way to Quality? Key Perspectives on Quality Improvement in Canadian Health Care Systems. Available online: https://healthcouncilcanada.ca/455/ (accessed on 3 July 2020).
- Health Council of Canada. Value for Money: Making Canadian Health Care Stronger; Health Council of Canada: Toronto, ON, Canada, 2009. [Google Scholar]
- ClinicalTrials.gov. Trends, Charts, and Maps. Available online: https://clinicaltrials.gov/ct2/resources/trends (accessed on 3 July 2020).
- Statistics Canada. Gross Domestic Expenditures on Research and Development, by Science Type and by Funder and Performer Sector. Available online: https://www150.statcan.gc.ca/t1/tbl1/en/tv.action?pid=2710027301 (accessed on 3 July 2020).
- Wennberg, J.; McPherson, K.; Goodman, D. Small Area Analysis and the Challenge of Practice Variation. Med. Pract. Var. 2015. [Google Scholar] [CrossRef]
- Institute of Medicine. Statement of Quality of Care; The National Academies Press: Washington, DC, USA, 1998. [Google Scholar]
- Greenberg, C.C.; Lipsitz, S.R.; Hughes, M.E.; Edge, S.B.; Theriault, R.; Wilson, J.L.; Carter, W.B.; Blayney, D.W.; Niland, J.; Weeks, J.C. Institutional Variation in the Surgical Treatment of Breast Cancer. Ann. Surg. 2011, 254, 339–345. [Google Scholar] [CrossRef] [PubMed]
- Rococo, E.; Mazouni, C.; Or, Z.; Mobillion, V.; Koon Sun Pat, M.; Bonastre, J. Variation in rates of breast cancer surgery: A national analysis based on French Hospital Episode Statistics. Eur. J. Surg. Oncol. 2016, 42, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Hawley, S.T.; Hofer, T.P.; Janz, N.K.; Fagerlin, A.; Schwartz, K.; Liu, L.; Deapen, D.; Morrow, M.; Katz, S.J. Correlates of between-surgeon variation in breast cancer treatments. Med. Care 2006, 44, 609–616. [Google Scholar] [CrossRef]
- Lovrics, P.J.; Gordon, M.; Cornacchi, S.D.; Farrokhyar, F.; Ramsaroop, A.; Hodgson, N.; Quan, M.L.; Wright, F.; Porter, G. Practice patterns and perceptions of margin status for breast conserving surgery for breast carcinoma: National Survey of Canadian General Surgeons. Breast 2012, 21, 730–734. [Google Scholar] [CrossRef]
- van Maaren, M.C.; Strobbe, L.J.A.; Koppert, L.B.; Poortmans, P.M.P.; Siesling, S. Nationwide population-based study of trends and regional variation in breast-conserving treatment for breast cancer. Br. J. Surg. 2018, 105, 1768–1777. [Google Scholar] [CrossRef]
- Jeevan, R.; Mennie, J.C.; Mohanna, P.N.; O’Donoghue, J.M.; Rainsbury, R.M.; Cromwell, D.A. National trends and regional variation in immediate breast reconstruction rates. Br. J. Surg. 2016, 103, 1147–1156. [Google Scholar] [CrossRef]
- Porter, G.; Wagar, B.; Bryant, H.; Hewitt, M.; Wai, E.; Dabbs, K.; McFarlane, A.; Rahal, R. Rates of breast cancer surgery in Canada from 2007/08 to 2009/10: Retrospective cohort study. CMAJ Open 2014, 2, E102–E108. [Google Scholar] [CrossRef]
- Quan, M.L.; Hodgson, N.; Przybysz, R.; Gunraj, N.; Schultz, S.; Baxter, N.; Urbach, D.; Simunovic, M. Surgery for Breast Cancer; Urbach, D.R., Simunovic, M., Schultz, S.E., Eds.; Institute for Clinical Evauative Sciences: Toronto, ON, Canada, 2008. [Google Scholar]
- McCahill, L.E. Quality Measures for Breast Cancer Surgery. Arch. Surg. 2009, 144, 455. [Google Scholar] [CrossRef]
- Birkmeyer, J.D.; Reames, B.N.; McCulloch, P.; Carr, A.J.; Campbell, W.B.; Wennberg, J.E. Understanding of regional variation in the use of surgery. Lancet 2013, 382, 1121–1129. [Google Scholar] [CrossRef]
- Glover, J.A. The Incidence of Tonsillectomy in School Children: (Section of Epidemiology and State Medicine). Proc. R. Soc. Med. 1938, 31, 1219–1236. [Google Scholar]
- Wennberg, J.; Gittelsohn, A. Small area variations in health care delivery. Science 1973, 182, 1102–1108. [Google Scholar] [CrossRef]
- Statistics Canada. Census Profile, 2016 Census. Available online: https://www12.statcan.gc.ca/census-recensement/2016/dp-pd/prof/details/page.cfm?Lang=E&Geo1=CSD&Code1=4611040&Geo2=PR&Code2=01&Data=Count&SearchText=4611040&SearchType=Begins&SearchPR=01&B1=All&Custom=&TABID=3 (accessed on 3 July 2020).
- MB Health Care Providers Network. Northern Health Region. Available online: http://www.mhpnetwork.ca/mb-ho-northern.html (accessed on 3 July 2020).
- Baker, G.R.; Norton, P.G.; Flintoft, V.; Blais, R.; Brown, A.; Cox, J.; Etchells, E.; Ghali, W.A.; Hebert, P.; Majumdar, S.R.; et al. The Canadian Adverse Events Study: The incidence of adverse events among hospital patients in Canada. CMAJ 2004, 170, 1678–1686. [Google Scholar] [CrossRef]
- McCarthy, C.M.; Hamill, J.B.; Kim, H.M.; Qi, J.; Wilkins, E.; Pusic, A.L. Impact of Bilateral Prophylactic Mastectomy and Immediate Reconstruction on Health-Related Quality of Life in Women at High Risk for Breast Carcinoma: Results of the Mastectomy Reconstruction Outcomes Consortium Study. Ann. Surg. Oncol. 2017, 24, 2502–2508. [Google Scholar] [CrossRef] [PubMed]
- Schain, W.S.; Wellisch, D.K.; Pasnau, R.O.; Landsverk, J. The sooner the better: A study of psychological factors in women undergoing immediate versus delayed breast reconstruction. Am. J. Psychiatry 1985, 142, 40–46. [Google Scholar] [CrossRef] [PubMed]
- Jahkola, T.; Asko-Seljavaara, S.; von Smitten, K. Immediate breast reconstruction. Scand. J. Surg. 2003, 92, 249–256. [Google Scholar] [CrossRef] [PubMed]
- Karunanayake, M.; Bortoluzzi, P.; Chollet, A.; Lin, J.C. Factors Influencing the Rate of Post-Mastectomy Breast Reconstruction in a Canadian Teaching Hospital. Plast. Surg. 2017, 25, 242–248. [Google Scholar] [CrossRef]
- Canadian Institute for Health Information; Canadian Partnership Against Cancer. Breast Cancer Surgery in Canada, 2007–2008 to 2009–2010; Canadian Institute for Health Information: Ottawa, ON, Canada, 2010. [Google Scholar]
- Canadian Partnership Against Cancer. Pan-Canadian Standards for Breast Cancer Surgery. 2019. Available online: https://www.partnershipagainstcancer.ca/topics/breast-cancer-surgical-standards/# (accessed on 3 July 2020).
- Murante, A.M.; Candelori, S.; Rucci, P.; Nuti, S.; Roncella, M.; Ghilli, M.; Mercatelli, A.; Fantini, M.P. Catching and monitoring clinical innovation through performance indicators. The case of the breast-conserving surgery indicator. BMC Res. Notes 2017, 10. [Google Scholar] [CrossRef]
- Schumacher, J.R.; Taylor, L.J.; Tucholka, J.L.; Poore, S.; Eggen, A.; Steiman, J.; Wilke, L.G.; Greenberg, C.C.; Neuman, H.B. Socioeconomic Factors Associated with Post-Mastectomy Immediate Reconstruction in a Contemporary Cohort of Breast Cancer Survivors. Ann. Surg. Oncol. 2017, 24, 3017–3023. [Google Scholar] [CrossRef]
- Restrepo, D.J.; Boczar, D.; Huayllani, M.T.; Sisti, A.; Gabriel, E.; McLaughlin, S.A.; Bagaria, S.; Spaulding, A.C.; Rinker, B.D.; Forte, A.J. Influence of Race, Income, Insurance, and Education on the Rate of Breast Reconstruction. Anticancer Res. 2019, 39, 2969–2973. [Google Scholar] [CrossRef]
- Hebbard, P. Personal Communication: Delayed Reconstruction; CancerCare Manitoba: Winnipeg, MB, Canada, 2020. [Google Scholar]
- Pilewskie, M.L.; Morrow, M. Management of the clinically node-negative axilla: What have we learned from the clinical trials? Oncology 2014, 28, 371–378. [Google Scholar] [PubMed]
- Lyman, G.H.; Somerfield, M.R.; Bosserman, L.D.; Perkins, C.L.; Weaver, D.L.; Giuliano, A.E. Sentinel Lymph Node Biopsy for Patients With Early-Stage Breast Cancer: American Society of Clinical Oncology Clinical Practice Guideline Update. J. Clin. Oncol. 2017, 35, 561–564. [Google Scholar] [CrossRef] [PubMed]
- Del Turco, M.R.; Ponti, A.; Bick, U.; Biganzoli, L.; Cserni, G.; Cutuli, B.; Decker, T.; Dietel, M.; Gentilini, O.; Kuehn, T.; et al. Quality indicators in breast cancer care. Eur. J. Cancer 2010, 46, 2344–2356. [Google Scholar] [CrossRef] [PubMed]
- Canadian Association of General Surgeons. Six Things Physicians and Patients Should Question; Choosing Wisely Canada: Toronto, ON, Canada, 2014. [Google Scholar]
- Yen, T.W.F.; Laud, P.W.; Pezzin, L.E.; McGinley, E.L.; Wozniak, E.; Sparapani, R.; Nattinger, A.B. Prevalence and Consequences of Axillary Lymph Node Dissection in the Era of Sentinel Lymph Node Biopsy for Breast Cancer. Med Care 2018, 56, 78–84. [Google Scholar] [CrossRef]
- Biganzoli, L.; Cardoso, F.; Beishon, M.; Cameron, D.; Cataliotti, L.; Coles, C.E.; Delgado Bolton, R.C.; Trill, M.D.; Erdem, S.; Fjell, M.; et al. The requirements of a specialist breast centre. Breast 2020, 51, 65–84. [Google Scholar] [CrossRef]
- Mustard, C.A.; Derksen, S.; Berthelot, J.M.; Wolfson, M. Assessing ecologic proxies for household income: A comparison of household and neighbourhood level income measures in the study of population health status. Health Place 1999, 5, 157–171. [Google Scholar] [CrossRef]
- Chateau, D.; Metge, C.; Prior, H.; Soodeen, R.A. Learning from the census: The Socio-economic Factor Index (SEFI) and health outcomes in Manitoba. Can. J. Public Health 2012, 103, S23–S27. [Google Scholar] [CrossRef]
| Characteristic | Total | Had Surgery n (%) | Did not Have Surgery n (%) |
|---|---|---|---|
| Manitoba | 3962 (100.0) | 3658 (92.3) | 304 (7.7) |
| Age Group | |||
| 20–49 | 664 (16.8) | 631 (17.2) | 33 (10.9) |
| 50–59 | 919 (23.2) | 880 (24.1) | 39 (12.8) |
| 60–69 | 1091 (27.5) | 1042 (28.5) | 49 (16.1) |
| 70–79 | 752 (19.0) | 705 (19.3) | 47 (15.5) |
| 80+ | 536 (13.5) | 400 (10.9) | 136 (44.7) |
| Income Quintile | |||
| Urban 1 (lowest) | 450 (11.7) | 401 (17.4) | 49 (26.9) |
| U2 | 491 (12.8) | 457 (19.9) | 34 (18.7) |
| U3 | 531 (13.8) | 497 (21.6) | 34 (18.7) |
| U4 | 511 (13.3) | 479 (20.8) | 32 (17.6) |
| U5 (highest) | 501 (13.0) | 468 (20.3) | 33 (18.1) |
| Rural 1 (lowest) | 238 (6.2) | 213 (16.9) | 25 (25.0) |
| R2 | 276 (7.2) | 254 (20.2) | 22 (22.0) |
| R3 | 275 (7.2) | 260 (20.7) | 15 (15.0) |
| R4 | 272 (7.1) | 252 (20.0) | 20 (20.0) |
| R5 (highest) | 296 (7.7) | 278 (22.1) | 18 (18.0) |
| RHA of Residence (at diagnosis) | |||
| Urban | * (60.6) | 2206 (60.3) | * (64.5) |
| Rural 1 | * (13.8) | 511 (14.0) | * (11.2) |
| Rural 2 | * (12.2) | 446 (12.2) | * (12.2) |
| Rural 3 | * (10.9) | 400 (10.9) | * (10.8) |
| Rural 4 | * (2.5) | 95 (2.6) | * (1.3) |
| Stage | |||
| Stage l | 1764 (44.9) | 1699 (46.6) | 65 (23.0) |
| Stage ll | 1447 (36.8) | 1374 (37.7) | 73 (25.9) |
| Stage lll | 545 (13.9) | 506 (13.9) | 39 (13.8) |
| Stage lV | 172 (4.4) | 67 (1.8) | 105 (37.2) |
| Characteristic | Breast-Conserving Surgery | Mastectomy with Immediate Reconstruction | Mastectomy without Immediate Reconstruction | |||
|---|---|---|---|---|---|---|
| n | % (95% CI) | n | % (95% CI) | n | % (95% CI) | |
| Total | 2439 | 66.7 | 383 | 10.5 | 836 | 22.8 |
| Age Group | ||||||
| 20–39 | 56 | 42.1 (33.7, 50.5) | 48 | 36.1 (27.9, 44.3) | 29 | 21.8 (14.8, 28.8) |
| 40–49 | 291 | 58.4 (54.1, 62.8) | 128 | 25.7 (21.9, 29.5) | 79 | 15.9 (12.7, 19.1) |
| 50–59 | 590 | 67.0 (63.9, 70.2) | 135 | 15.3 (13.0, 17.7) | 155 | 17.6 (15.1, 20.1) |
| 60–69 | 755 | 72.5 (69.7, 75.2) | 66 | 6.3 (4.9, 7.8) | 221 | 21.2 (18.7, 23.7) |
| 70–80+ | 747 | 67.6 (64.8, 70.4) | 6 | 0.5 (0.1, 1.0) | 352 | 31.9 (29.1, 34.6) |
| Income Quintile | ||||||
| Urban 1 (lowest) | 277 | 69.1 (64.6, 73.6) | 26 | 6.5 (4.1, 8.9) | 98 | 24.4 (20.2, 28.6) |
| U2 | 299 | 65.4 (61.1, 69.8) | 43 | 9.4 (6.7, 12.1) | 115 | 25.2 (21.2, 29.1) |
| U3 | 355 | 71.4 (67.5, 75.4) | 48 | 9.7 (7.1, 12.3) | 94 | 18.9 (15.5, 22.4) |
| U4 | 325 | 67.8 (63.7, 72.0) | 70 | 14.6 (11.5, 17.8) | 84 | 17.5 (14.1, 20.9) |
| U5 (highest) | 331 | 70.7 (66.6, 74.8) | 65 | 13.9 (10.8, 17.0) | 72 | 15.4 (12.1, 18.7) |
| Rural 1 (lowest) | 129 | 60.6 (54.0, 67.1) | 17 | 8.0 (4.3, 11.6) | 67 | 31.5 (25.2, 37.7) |
| R2 | 157 | 61.8 (55.8, 67.8) | 15 | 5.9 (3.0, 8.8) | 82 | 32.3 (26.5, 38.0) |
| R3 | 162 | 62.5 (56.7, 68.4) | 15 | 5.8 (2.9, 8.6) | 82 | 31.7 (26.0, 37.3) |
| R4 | 151 | 59.9 (53.9, 66.0) | 35 | 13.9 (9.6, 18.2) | 66 | 26.2 (20.8, 31.6) |
| R5 (highest) | 191 | 68.7 (63.3, 74.2) | 38 | 13.7 (9.6, 17.7) | 49 | 17.6 (13.1, 22.1) |
| RHA of Residence (at diagnosis) | ||||||
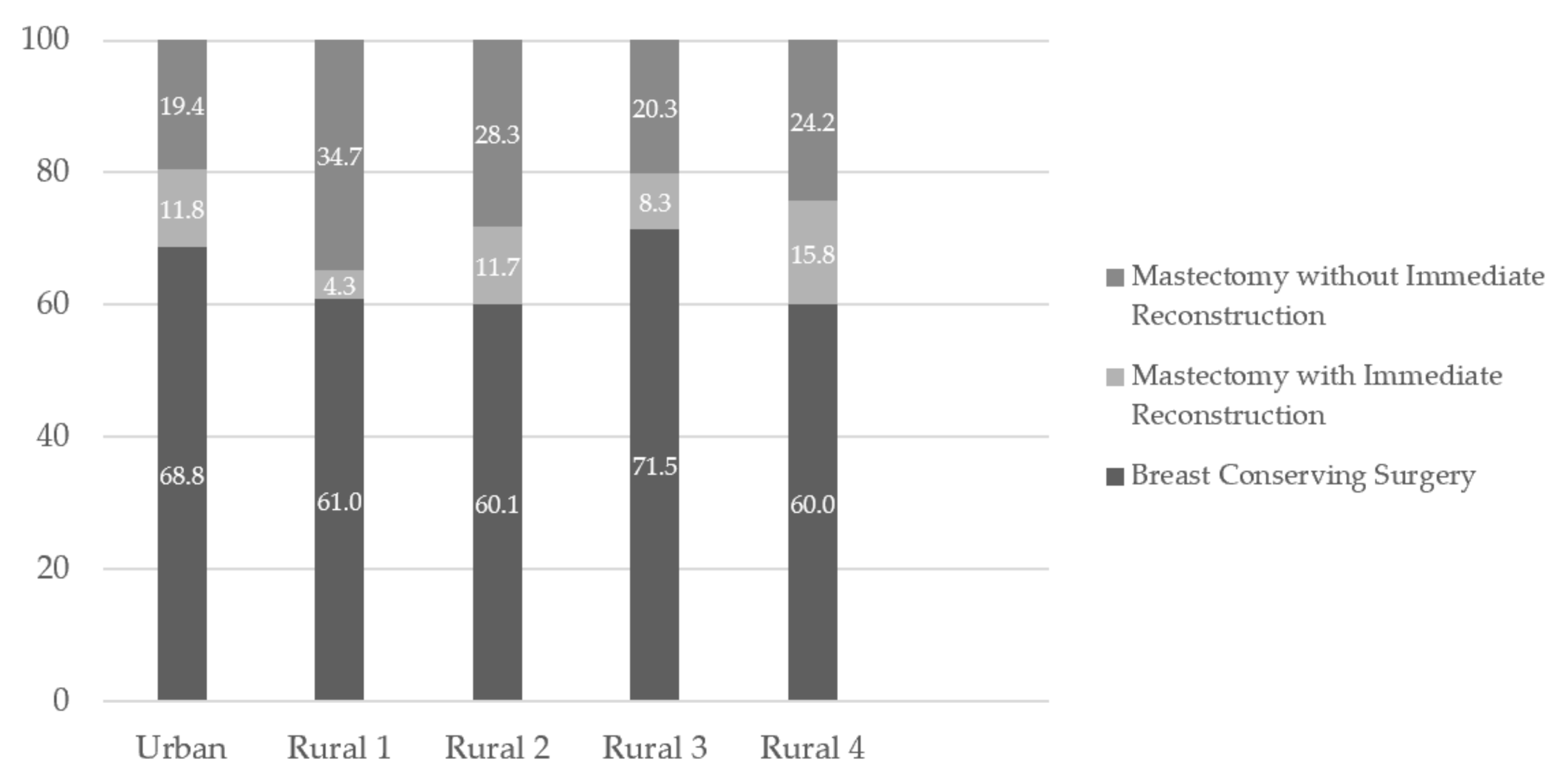
| Urban | 1517 | 68.8 (66.8, 70.7) | 261 | 11.8 (10.5, 13.2) | 428 | 19.4 (17.8, 21.1) |
| Rural 1 | 311 | 61.0 (56.7, 65.2) | 22 | 4.3 (2.6, 6.1) | 177 | 34.7 (30.6, 38.8) |
| Rural 2 | 268 | 60.1 (55.5, 64.6) | 52 | 11.7 (8.7, 14.6) | 126 | 28.3 (24.1, 32.4) |
| Rural 3 | 286 | 71.5 (67.1, 75.9) | 33 | 8.3 (5.6, 10.9) | 81 | 20.3 (16.3, 24.2) |
| Rural 4 | 57 | 60.0 (50.1, 69.9) | 15 | 15.8 (8.5, 23.1) | 23 | 24.2 (15.6, 32.8) |
| RHA of Surgery | ||||||
| Urban RHA | 2148 | 67.5 (65.9, 69.9) | 383 | 12.0 (10.9, 13.2) | 652 | 20.5 (19.1, 21.9) |
| Rural 1 | 248 | 64.9 (60.1, 69.7) | n/a | 134 | 35.1 (30.3, 29.9) | |
| Rural 2–4 | 32 | 43.8 (32.5, 55.2) | n/a | 41 | 56.2 (44.8, 67.5) | |
| Out of Province | 11 | 57.9 (35.7, 80.1) | n/a | 8 | 42.1 (19.9, 64.3) | |
| Stage | ||||||
| Stage l | 1342 | 79.0 (77.1, 80.9) | 113 | 6.7 (5.5, 7.8) | 244 | 14.4 (12.7, 16.0) |
| Stage ll | 889 | 64.7 (62.2, 67.3) | 146 | 10.6 (9.0, 12.3) | 338 | 24.6 (22.3, 26.9) |
| Stage lll | 171 | 33.8 (29.7, 37.9) | 111 | 21.9 (18.3, 25.5) | 224 | 44.3 (39.9, 48.6) |
| Stage lV | 28 | 41.8 (30.0, 53.6) | 12 | 17.9 (8.7, 27.1) | 27 | 40.3 (28.6, 52.0) |
| Characteristic | Axillary Lymph Node Dissection for Node Negative Disease (n = 2379) | ≤30 Days between First Surgical Consult and First Surgery (n = 2526) | Re-excision after Breast-Conserving Surgery (n = 2439) | |||
|---|---|---|---|---|---|---|
| n | % (95% CI) | n | % (95% CI) | n | % (95% CI) | |
| Manitoba | 137 | 5.8 | 1245 | 49.3 | 450 | 18.5 |
| Age Group | ||||||
| 20–39 | - | 1.9 (0.0, 5.7) | 27 | 32.9 (22.8, 43.1) | 20 | 35.7 (23.2, 48.3) |
| 40–49 | - | 6 (3.1, 9.0) | 176 | 49.4 (44.2, 54.6) | 70 | 24.1 (19.1, 29.0) |
| 50–59 | - | 3.8 (2.2, 5.4) | 299 | 46.7 (42.9, 50.6) | 118 | 20.0 (16.8, 23.2) |
| 60–69 | - | 5.4 (3.8, 7.0) | 409 | 54.0 (50.4, 57.5) | 133 | 17.6 (14.9, 20.3) |
| 70–79 | - | 7.3 (5.0, 9.6) | 227 | 48.6 (44.1, 53.1) | 76 | 15.7 (12.5, 19.0) |
| 80+ | - | 7.9 (4.9, 10.9) | 107 | 48.0 (41.4, 54.5) | 33 | 12.5 (8.5, 16.5) |
| RHA of Residence (at diagnosis) | ||||||
| Urban | - | 3.0 (2.1, 3.9) | 766 | 47.9 (45.5, 50.4) | 258 | 17.0 (15.1, 18.9) |
| Rural 1 | - | 15.9 (12.0, 19.8) | 160 | 60.2 (54.3, 66.0) | 73 | 23.5 (18.8, 28.2) |
| Rural 2 | - | 8.0 (4.9, 11.2) | 151 | 49.2 (43.6, 54.8) | 66 | 24.6 (19.5, 29.8) |
| Rural 3 | - | 4.8 (2.3, 7.4) | 150 | 49.8 (44.2, 55.5) | 42 | 14.7 (10.6, 18.8) |
| Rural 4 | - | 7.1 (0.4, 13.9) | 18 | 33.3 (20.8, 45.9) | 11 | 19.3 (9.1, 29.5) |
| RHA of Surgery | ||||||
| Urban 1 | 71 | 3.4 (2.6, 4.2) | n/a | 376 | 17.5 (15.9, 19.1) | |
| Rural 1 | 52 | 20.4 (15.4, 25.3) | n/a | 57 | 23.0 (17.7, 28.2) | |
| Rural 2 | 14 | 32.6 (18.6, 46.6) | ||||
| Rural 2–4 | n/a | n/a | n/a | n/a | 15 | 46.8 (29.6, 64.2) |
| Stage | ||||||
| Stage I | 80 | 4.9 (3.9, 6.0) | 609 | 49.6 (46.8, 52.4) | 208 | 15.5 (13.6, 17.4) |
| Stage II | 57 | 7.7 (5.8, 9.6) | 496 | 52.2 (49.0, 55.4) | 179 | 20.1 (17.5, 22.8) |
| Stage III | 0 | 0.0 (0.0, 0.0) | 140 | 40.1 (35.0, 45.3) | 56 | 32.7 (25.7, 39.8) |
| Length of Stay | Median (Days) | 90th Percentile (Days) |
|---|---|---|
| Breast-Conserving Surgery | 0 | 1 |
| Mastectomy without Immediate Reconstruction | 1 | 4 |
| Mastectomy with Immediate Reconstruction | 3 | 5 |
| In-Hospital Complication | n | % (95% CI) |
| Breast-Conserving Surgery | 43 | 1.5 (1.1, 1.9) |
| Mastectomy without Immediate Reconstruction | 42 | 4.6 (3.2, 5.9) |
| Mastectomy with Immediate Reconstruction | 43 | 9.9 (7.1, 12.7) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ratnayake, I.; Hebbard, P.; Feely, A.; Biswanger, N.; Decker, K. Assessment of Breast Cancer Surgery in Manitoba: A Descriptive Study. Curr. Oncol. 2021, 28, 581-592. https://doi.org/10.3390/curroncol28010058
Ratnayake I, Hebbard P, Feely A, Biswanger N, Decker K. Assessment of Breast Cancer Surgery in Manitoba: A Descriptive Study. Current Oncology. 2021; 28(1):581-592. https://doi.org/10.3390/curroncol28010058
Chicago/Turabian StyleRatnayake, Iresha, Pamela Hebbard, Allison Feely, Natalie Biswanger, and Kathleen Decker. 2021. "Assessment of Breast Cancer Surgery in Manitoba: A Descriptive Study" Current Oncology 28, no. 1: 581-592. https://doi.org/10.3390/curroncol28010058
APA StyleRatnayake, I., Hebbard, P., Feely, A., Biswanger, N., & Decker, K. (2021). Assessment of Breast Cancer Surgery in Manitoba: A Descriptive Study. Current Oncology, 28(1), 581-592. https://doi.org/10.3390/curroncol28010058






