Abstract
The concept of food and aging is of great concern to humans. So far, more than 300 theories of aging have been suggested, and approaches based on these principles have been investigated. It has been reported that antioxidants in foods might play a role in human aging. To clarify the current recognition and positioning of the relationship between these food antioxidants and aging, this review is presented in the following order: (1) aging theories, (2) food and aging, and (3) individual food antioxidants and aging. Clarifying the significance of food antioxidants in the field of aging will lead to the development of strategies to achieve healthy human aging.
Introduction
Aging is regarded as a biological phenomenon that results in decreasing biological function and increasing mortality over time, in terms of personal, organ, and tissue levels. The concept of aging in terms of individual aging has been recognized since ancient times, but the new view of aging as a “population scenario” that focuses on the overall population has only appeared in the last century [1]. There is also still a debate over the definition of the relationship between aging and disease [2,3]. Although research related to aging continues the trends of the past and present for many researchers, it seems to be difficult to define the concept of aging because it is an ambiguous and changing term that changes its view at different times. Numerous mechanisms related to the induction of aging have been reported, and there are currently more than 300 existing theories [4]. Among them, the recognition and positioning of the relationship between foods, food antioxidants, and the aging process remain vague, even though there has been interest in this topic in the past. Therefore, this review aims to organize the recognition of the relationship between food antioxidants and the aging process by considering: (1) aging theories, (2) food and aging, and (3) individual food antioxidants and aging, in that order, and to clarify the current status of knowledge and the problems in the field.
1. Aging Theories
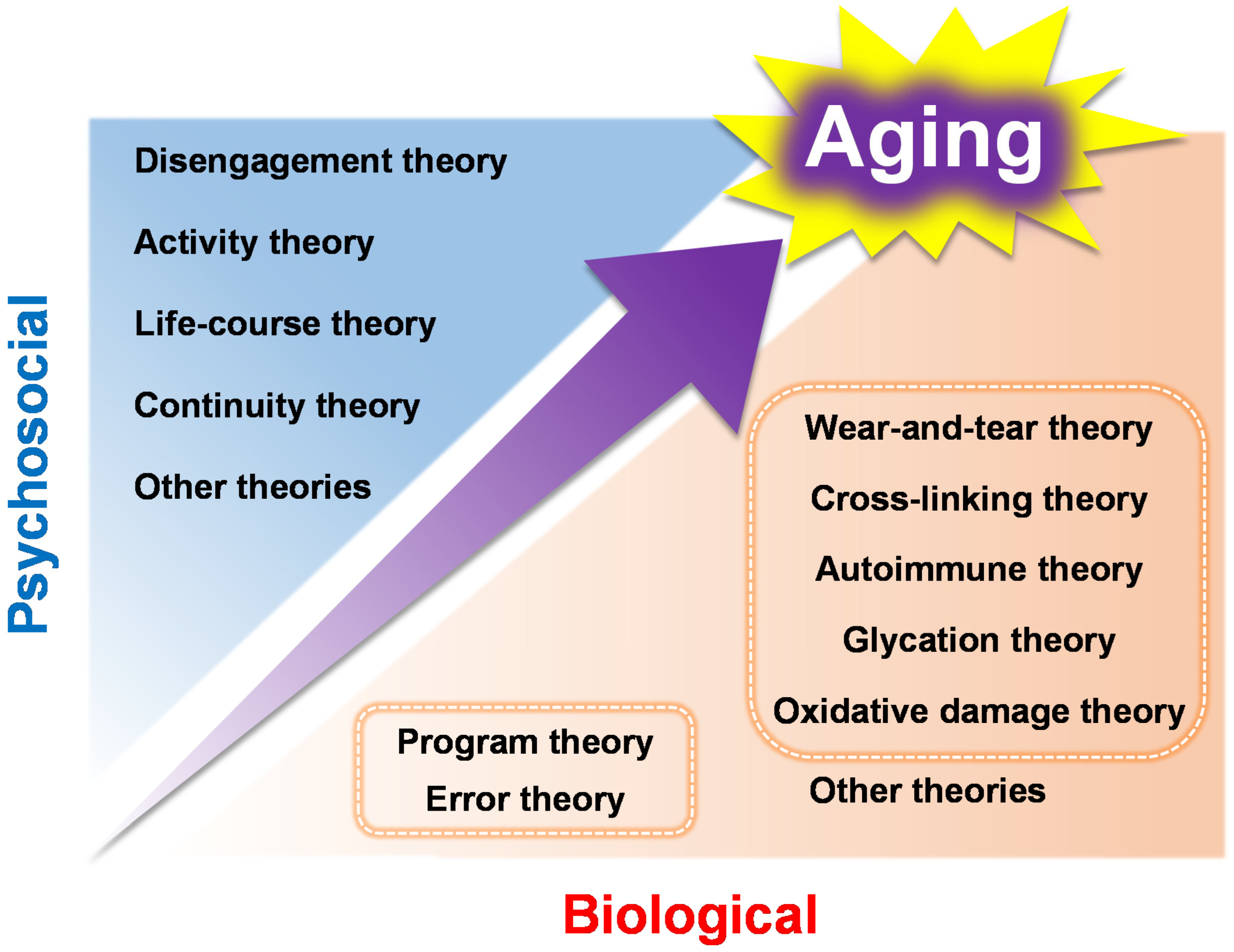
Since it is difficult to cover all these aging theories in this review, the most fundamental of those that have been proposed to date are outlined in Figure 1. Although it is difficult to clarify the classification of aging theories as they are complementary to other theories, this chapter will categorize them into two major groups: (1) genetic factors associated with aging, and (2) non-genetic factors associated with aging, along with the presentation of the relevant molecular reactions and examples of the aging characteristics associated with each theory.
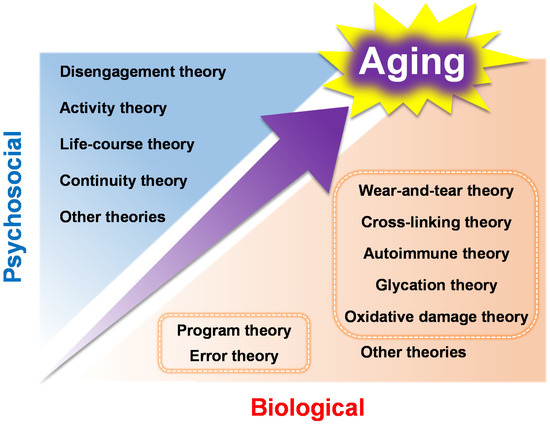
Figure 1.
The biological and psychosocial aging theories that are mentioned in this review.
1.1. Genetic Factors Associated with Aging
1.1.1. Program Theory
The program theory proposes that aging is not a random occurrence but is instead inherent in our genetic information. This theory is based on a discovery by Hayflick et al. in 1961 that there is a limitation on the number of divisions of and the proliferation of human and animal cells (the so-called Hayflick limit) [5]. Telomeres and telomerase are closely related to the program theory. DNA loses its replicative ability when its terminal bases are lost during replication. Mammals have a TTAGGG repeat structure called a telomere at the end of their DNA, which protects the genetic information and allows replication. Telomeres are elongated by telomerase. Therefore, telomere length is believed to be a determinant of the number of cell divisions and is thus a factor in aging and lifespan [6]. However, telomeres are shortened with age and affect the process of aging because they are regenerated with each replication, but not completely. More recently, telomerase-deficient mice have been shown to demonstrate impaired neuronal differentiation and neurogenesis [7]. The recovery of telomere function and length has also been reported to extend the lifespan of mammals and can delay physical aging in mouse experiments [8,9].
1.1.2. Error Theory
The error theory states that random errors in DNA transcription and translation lead to the accumulation of mutant proteins, which causes cellular dysfunction and aging [10]. To date, no direct evidence of the age-dependent dysfunction of protein synthesis has been reported [11]. Furthermore, another study using Escherichia coli showed that the induction of errors in the gene increased the frequency of error generation but did not induce bacterial death [12]. Therefore, the number of recent reports supporting this theory has relatively decreased.
1.2. Non-Genetic Factors Associated with Aging
1.2.1. Wear-and-Tear Theory
The wear-and-tear theory, which was proposed by Weismann in 1882, states that aging progresses when cells and tissues are worn down by risk factors over the years [13]. However, this theory was invalidated by several phenomena. For instance, hyperactive mice wore down their tissues but can live longer than normal mice [14]. Caterpillars that have lost the ability to express antioxidant enzymes can live longer [15]. Recently, there seems to be increasing evidence that the wearing down is not simply explainable as a physical inevitability in aging and needs to be considered in terms of natural selection from an evolutionary aspect.
1.2.2. Cross-Linking Theory
The cross-linking theory states that the accumulation of molecules with multiple reactive units due to the cross-linking of poorly degradable macromolecules impairs cellular function and promotes aging. Increased viscosity of the extracellular environment is induced by decreased solubility, elasticity, and permeability, due to the cross-linking of collagens and other molecules. As a result, the circulation of nutrients and wastes in the cells is delayed and, thus, aging progresses [16]. The cross-linking products of glucose and collagen (products of the Maillard reaction) are widely recognized as products that increase along with aging in the body [17]. Bjorksten et al. pointed out that the free radical theory is also a form of cross-linking theory because free radicals that are generated in the body induce cross-linking reactions in collagen and other molecules [18]. Despite qualitative and quantitative evidence of cross-linked molecules that has been provided to support this theory, it is still not clear whether the molecules are crucial in biological aging.
1.2.3. Autoimmune Theory
The autoimmune system has been regarded as being at least indirectly associated with aging. Higher vertebrates have two major immune mechanisms, innate and acquired immunity. The dysfunction of acquired immunity is particularly significant in terms of aging. Innate immunity captures antigens via pattern recognition receptors and presents them to T cells, while acquired immunity is characterized by the antigen-specific effector and memory responses of T cells and B cells (T cells: directives, antigen memory, antigen destruction; B cells: antibody production). Involution of the thymus, the primary lymphoid tissues for T-cell education and development, starts at a relatively early age in different species [19]. In an aged thymus, biological processes for the elimination of self-reactive T cells and the induction of regulatory T cells, namely, central tolerance, are seen to decline [20]. In addition, aging is associated with a decline in B cell production in the bone marrow, along with increased self-reactive B cell populations. Taken together, aged adaptive immunity promotes development that results in damage to the body’s own tissues and is one of the major causes of death in women under 65 years of age in the US [21,22].
1.2.4. Glycation Theory
In 1912, the aminocarbonyl reaction (Maillard reaction) was reported by Maillard; it is considered part of the phenomenon of biological body aging [23]. The glucose and lysine residues of proteins react at body temperature, to form advanced glycation end products (AGEs) through a condensation reaction. The formed AGEs are accumulated in various tissues, such as the blood vessel walls, and induce tissue inflexibility, leading to vasodilator dysfunction and hypertension, which is regarded as a trigger of aging [24].
1.2.5. Oxidative Damage Theory
Aerobic organisms consume oxygen for the purposes of energy metabolism, which produces reactive oxygen species (ROS) in the metabolic process. In 1956, Harman proposed that ROS leads to aging by causing damage to cells and tissues [25]. Older individuals have higher concentrations of oxidized products, such as proteins, DNA, and lipids, than younger individuals, whereas antioxidants have often been highlighted as molecules that reduce ROS generation and contribute toward extending the lifespan [26,27,28]. Among cellular organelles, especially in mitochondria, which are involved in aerobic energy metabolism, there is a higher level of ROS in aging individuals. Furthermore, in mammals, there is an inverse correlation between the concentration of ROS in mitochondria and the lifespan, suggesting that damage to mitochondrial DNA and membrane lipids is also a factor closely related to aging [29]. However, ROS also plays a role in the mammalian immune system for biological defense that eliminates intracellular pathogens [30]. Therefore, it is widely recognized that dysfunction of the redox balance induces inflammatory reactions. Related to this, the term “inflammaging” has developed to describe the concept of aging as it is related to innate immunity, which corresponds to (1) chronically, (2) no contact with bacteria, etc., and (3) weak inflammation [31]. Recently, based on the concept of “inflammaging,” it has been shown that chronic tissue inflammation caused by ROS has a significant impact on the regulatory systems of the nervous system and immune system, as well as the aging process; it can be induced by suppressing the regulation of the inflammatory factors of the immune system [32,33].
1.2.6. Other Biological Aging-Related Theories
The existence of “senescent cells,” which accelerate aging in the body, has been the topic of growing attention among gerontologists. In general, aged cells that have stopped dividing are removed from the body through cell death or the phagocytosis of immune cells. However, there are senescent cells that accumulate in the tissues despite the cell division having stopped. Recent studies have shown that these accumulated senescent cells release inflammatory substances that accelerate the senescence of neighboring cells, trigger excessive inflammation, and lead to tissue dysfunction [34]. Since the immune system in the body cannot eliminate all the senescent cells, some anti-aging approaches have begun to be developed that target these senescent cells [35]. Since there is still a lack of direct information on the relationship of these senescent cells with aging, further elucidation of the physiological phenomenon may be necessary.
In contrast to the theory that certain factor(s) accelerate the rate of aging, there is a theory stating that the rate of aging is unchanged by any factor. Colchero et al. reported strong linearity between life expectancy and lifespan equality in various primates, mainly connected to infant mortality or age-independent mortality improvement, with no impact on the aging rate [36]. This result suggests that only the biological limit determines longevity.
1.3. Sociology of Aging
The concept of aging exists not only from a biological viewpoint but also from a sociological one. Social animals, such as humans, are thought to be influenced by their surroundings in their biological development. Theories of aging in social gerontology can be broadly classified into four categories: (1) disengagement theory, (2) activity theory, (3) life-course theory, and (4) continuity theory.
1.3.1. Disengagement Theory
The disengagement theory posits that aging is a process of disengagement from being a part of society. The ultimate stage of disengagement is incurable illness or death. This is the concept that elderly people who contribute less to society could leave and give up their roles to young people who will contribute more to society, leading to the sustainability of the social system to which they belong [37,38].
1.3.2. Activity Theory
Activity theory is the concept that maintaining activities and attitudes in middle age for as long as possible is the key to healthy aging. This concept has existed previously but was given this name as a contrast to the disengagement theory proposed earlier. The difference between this theory and the disengagement theory is that the elderly may or may not be considered to be at a developmental stage. Disengagement theory describes old age as a natural developmental process, while activity theory describes the period after middle age as a completed stage [39].
1.3.3. Life-Course Theory
Life-course theory is the theory that human aging is part of the growth (development) process, with earlier experiences being important for later life adaptability [40]. More recently, based on this concept, an epidemiologic assessment of the relationship between aging, physical activity, and cognitive function has been conducted [41,42].
1.3.4. Continuity Theory
Continuity theory arose from discussions of disengagement theory and activity theory. Humans tend to keep their personalities for the long term, but they become more introspective with age, focusing their attention and interest inward to their individual selves. This theory proposes that healthy aging is the state of keeping a mature and integrated personality during aging [43].
1.4. Aging and Senescence
Living biological organisms age with time. With regard to this concept of aging, some researchers use the term “senescence” as a subgroup to distinguish between harmless changes and changes associated with mortality risk [1]. Dodig et al. defined senescence as an “irreversible form of long-term cell-cycle arrest, caused by excessive intracellular or extracellular stress or damage,” which is regarded as influencing various biological events related to aging, such as metabolism, immune function, autophagy changes, and chromatin development [44]. These investigations with the keyword “cellular senescence” and “biological senescence” are one of the most significant issues in the field of molecular biology and gerontology.
2. Food and Aging
2.1. Potential Foods for Anti-Aging
To make the relationship between food and lifespan clearer, there has been an ongoing discussion for some years, based on the theories of aging as described in the previous chapter [45,46]. Foods contain numerous bioactive compounds that are essential for maintaining human health. Therefore, dietary consumption is the most common and routine way for humans to supply nutrients to the body. Although there is still no proven relationship with the theory of aging, it has traditionally been suggested that certain foods themselves or certain compounds in foods have preventive and curative functions against diseases. Although no specific foods or nutrients in foods related to longevity have yet been identified, it is worth pointing out that a number of reports have suggested that the consumption of foods relatively rich in antioxidants has the ability to reduce mortality. Against this background, this section describes previous reports made in cohort studies on the relationship between diet and aging as it contributes to lifespan (Table 1).

Table 1.
Cohort studies examining diet and lifespan.
2.2. Mediterranean Diet
Among the diets associated with health and longevity, the traditional Mediterranean diet is widely recognized as one of the most popular. Mediterranean diets are rich in vegetables, legumes, fruits, nuts, grains, seafood, and olive oil; they are also low in saturated fats, dairy products, and meat. Drinking wine and other alcoholic beverages is also popular. Attention regarding the dietary functions of the Mediterranean diet was greatly increased in the early 1990s, with (1) growing concerns that large doses of simple carbohydrates might not be beneficial to health, and (2) growing interest in the use of the Mediterranean diet score to quantify the health benefits of the food [81]. In a cohort study of Greek adults, mortality related to cardiovascular (coronary heart) disease and cancer was examined using the Mediterranean diet score as a criterion and reported that Mediterranean diet ingestion was correlated with lower overall mortality [47]. This may support the report by Crous-Bou et al. that higher dietary Mediterranean diet scores tend to result in longer telomeres in healthy women [82]. Other research institutes also suggested that the Mediterranean diet is beneficial in reducing mortality among heart failure patients as well as healthy subjects [48].
2.3. Other Traditional Foods
The Nordic diet is characterized by low levels of processed foods and includes yogurt, berries, whole grain bread, oatmeal, apples/pears, root vegetables, cabbage, fish/shellfish, etc., It has also been reported that consumption of the Nordic diet lowers mortality in humans. A cohort study of Danes showed that consumption of the Nordic diet was associated with lower mortality, especially among middle-aged men [49]. Another cohort study of Swedish women indicated that all-cause mortality was significantly lower when on a diet with higher Nordic diet scores [50].
Japan is one of the leading countries of longevity in the world. With the “Japanese diet” being registered as a World Intangible Heritage in 2013, attention has been attracted by the relationship between the Japanese diet and aging. In a cohort study of Japanese subjects, the consumption of Japanese food (including rice, miso soup, seaweed, cucumber, green and yellow vegetables, fish, green tea, beef, pork, and coffee) was associated with prolonged survival [51]. Other cohort studies of Japanese people have also reported that consumption of the Japanese diet reduces mortality from all causes, especially among women [52]. On the other hand, the definition of Japanese food in cohort studies is still unclear and varies. Furthermore, the diet of Japanese people has been changing in recent years. Therefore, there is a consideration that the longevity of Japanese people in recent years might be related to other factors besides the Japanese diet [83].
2.4. Individual Foodstuffs
2.4.1. Fruits and Vegetables
Individual fruits and vegetables that are abundant in the Mediterranean diet have also been found to be related to aging. A cohort study of US adults showed that consumption of fruits and vegetables may reduce the risk of cardiovascular disease and all-cause mortality [53]. Cohort studies of adults in ten European countries have also indicated that an increased intake of fruits and vegetables tends to reduce mortality [54]. In a cohort study of participants from the Czech Republic, Poland, and Russia, an increased intake of fruits and vegetables was associated with lower mortality, especially among smokers and hypertensive patients [55]. Among vegetables, cruciferous vegetables are widely consumed in the general diet. A cohort study of Chinese adults indicated that fruit and vegetable intake was inversely associated with the risk of total mortality in both women and men, with a dose-response pattern that was particularly evident in the intake of cruciferous vegetables [56]. A multinational cohort study of adults in eleven countries who were being treated with hemodialysis has also indicated that a higher vegetable intake is associated with lower all-cause and non-cardiovascular mortality [57]. As mentioned in this section, vegetable consumption seems to be associated with longevity; however, the consumption of only vegetables may not result in longevity. For instance, a large-scale cohort study of adults in the United Kingdom observed that all-cause mortality rates for vegetarians and non-vegetarians were almost the same [58].
2.4.2. Nuts
Nuts, which are abundant in the Mediterranean diet, are consumed worldwide. A cohort study of U.S. women indicates that the consumption of nuts led to changes in plasma lipids, inflammation, and glucose metabolism, resulting in an association with a reduced risk of death from cardiovascular disease [59]. In another cohort study of US men, it was observed that subjects who consumed nuts five or more times per week had a 34% lower all-cause mortality rate than those who consumed nuts less than once per month [60]. Several types of nuts (walnuts, hazelnuts, almonds, and peanuts) were also reported to show a significant reduction in cancer death [61]. One Japanese cohort study showed that total nut intake (peanuts and chestnuts) was inversely associated with all-cause mortality in men [62]. Interestingly, the Golestan Cohort Study conducted in Iran evidenced that reduced mortality by nut consumption (peanuts, tree nuts, and overall nuts) is independent of a healthy lifestyle [63]. Among nuts, there are relatively numerous studies on walnuts, examining their relationship with longevity. Several cohort studies have found that walnut consumption may reduce mortality [64,65]. On the other hand, a cohort study in China reported that nut consumption dose-dependently induced better survival in long-term breast cancer survivors, regardless of the types of nuts [66]. Thus, further investigation and scientific evidence on the components of each nut type is warranted to clarify the essential factor in reduced mortality that is induced by various types of nuts.
2.4.3. Beverages
It is considered that daily beverage intake may also have an impact on aging. Among Japanese diets, green tea is one of the most widely studied beverages for aging. Several cohort studies of Japanese have reported that the consumption of green tea has the potential to reduce the risk of mortality from heart disease and cardiovascular disease [67,68]. Hao et al. compared the effects of minerals in food and drinking water on life expectancy, using the database of demographics from eighteen counties in the Chinese census [69]. As a result, the amount of Cu, Se, and Zn ingested from the diet and drinking water was positively correlated with longevity, while Pb was negatively correlated in Hainan Province, which is known for its longevity. In a meta-analysis of 757,304 individuals from 12 independent cohorts, Wang et al. reported that low to moderate doses of Ca were associated with reduced mortality, but not at higher doses, and concluded that each individual should consume the appropriate amount [70]. Navarro et al. reported an inverse linear association between total coffee consumption and the risk of death from all causes in Spaniards, which was particularly pronounced in those over 54 years old [71].
2.5. Age and Food Intake
The nutritional function that can be expected from the intake of food seems to differ significantly, depending on age at the time of intake. A cohort study of elderly women (65 to 81 years old) in the US tried to determine the association between coffee and tea consumption and survival to age 90 [72]. The results showed that coffee and tea consumption may not prolong survival until later life in women. Concerning protein intake, a cohort study of Chinese people aged 40 to 74 reported that in healthy people, red meat (a rich source of protein) intake increases the risk of death [73]. In contrast, a cohort study of Chinese people over 80 indicated that the consumption of protein-rich foods reduced the risk of death [74]. Such changes in dietary effects are considered to occur due to the various dysfunctions associated with aging, such as alteration of the gut microbiota, the decreased absorption of nutrients in the gastrointestinal tract, decreased chewing strength, decreased exercise, etc. The relationship between caloric restriction and lifespan has become a major topic in the field of gerontology, with reports indicating that caloric restriction may prevent aging in various species [84,85]. Caloric restriction is thought to have a significant impact on human aging. However, the effect of caloric restriction might be challenging to verify, as it would require very long-term, large-scale human lifestyle intervention studies.
2.6. Potential Foods That May Accelerate Aging
While food intake has been associated with longer life expectancy, others have also reported that it may contribute to accelerated aging and increased mortality. For example, there are cases in which traditional diets increase mortality. A cohort study of people in Russia, Poland, and the Czech Republic showed that the high mortality from cardiovascular disease in Eastern Europe is related to the traditional dietary habits in target regions, with lard consumption most likely to be responsible for increased mortality [75]. With the development of food processing technology in modern society, the concept of “ultra-processed food (UPF)” has increased in importance. It gradually became clear that the consumption of UPF might increase the risk of death [86]. UPF is defined as “formulations of ingredients, mostly of exclusive industrial use, typically created by a series of industrial techniques and processes” in the NOVA classification and is considered a different group from unprocessed and minimally processed foods, processed culinary ingredients, and processed foods [87]. UPF refers to ready-to-eat industrially formulated products that contain high levels of additives such as sugar, salt, hardened oil, flavorings, emulsifiers, and preservatives [88]. A large prospective cohort study in France (French NutriNet-Santé cohort), of subjects with a median age of 42.8 years, from 2009 to 2017 reported that the consumption of a diet classified as UPF according to the NOVA classification was associated with increased overall cancer risk [89]. A cohort study of French people indicated that the increased consumption of UPF was associated with an increased risk of total mortality in this adult population [76]. A cohort study of Italians also showed that UPFs, particularly those rich in sugar, were associated with an increased risk of death [77]. In addition, a cohort study of Spaniards, conducted by other research institutions, showed that replacing UPF with non- or low-processed foods with the same calories may reduce mortality [78]. Fried foods are highly palatable and popular but have been known to increase the risk of diseases that lead to death, such as type 2 diabetes and cardiovascular disease. A cohort study of postmenopausal women in the US indicated that the consumption of fried foods, especially fried chicken and fried seafood, was associated with mortality risk among US women, including from cardiovascular disease [79]. Furthermore, a cohort study of elderly people in the US also observed changes in mortality due to the consumption of fried and non-fried potatoes and indicated that the consumption of fried potatoes more than twice a week increased the risk of death [80]. As this section has shown, an evaluation of food and aging suggests that it is necessary to consider not only information on foodstuffs but also changes in certain foods’ properties due to processing in factories.
3. Individual Food Antioxidants and Aging
Based on the results of the numerous cohort studies described in the previous chapter that have examined the impact of diet on the risk of aging and death, it seems almost certain that the content of the human diet has a significant effect on aging. However, there is still no clear evidence on which molecular, cellular, and physiological changes are the most important factors of aging in individual organisms and on how they affect each other [90]. With current scientific technology, it seems hard to quantify the effect of “whole foods,” which are composed of many molecules, on aging. However, numerous studies have suggested that a variety of individual antioxidants in foods are related to aging: for example, the autoimmune theory [91]; cross-linking theory [92]; glycation theory [93]; and KEAP1-NRF2 theory [94].
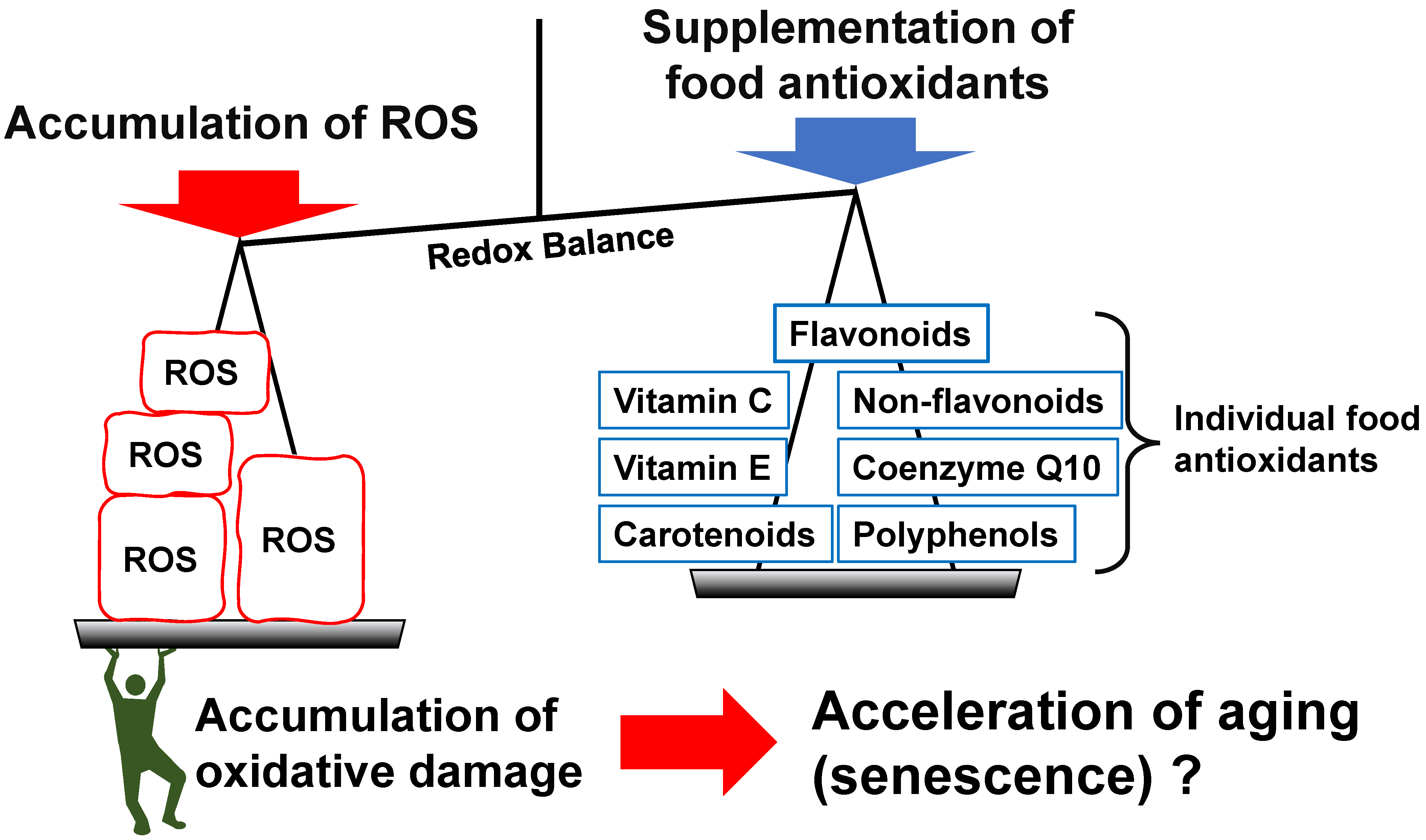
Most of these previous studies are based on the concept of “oxidative damage theory,” as described in the first half of this review. Among the various theories of aging, the oxidative damage theory has been one of the most popular theories in aging research, with much quantitative experimental evidence being reported from the past to the present [4]. There is growing evidence that ROS may act as signaling molecules that not only induce oxidative stress but also ultimately extend the lifespan [95]. Such trends led to the concept of “mitohormesis”, which states that ROS promotes aging but, in the appropriate concentrations, can enhance the biological defense system [96,97]. The regulation of redox balance in the body is a crucial factor in aging, as ATP production in mitochondria with ROS generation is an essential factor for energy acquisition in aerobic organisms. It is also expected that individual antioxidants in foods also contribute to the regulation of the redox balance (Figure 2). Cell signaling pathways involved with this balance, such as MAPKs, NF-κB, and Nrf2 seem important in the anti-aging effects of antioxidants [98,99,100,101,102]. However, a description of the detailed mechanism for this process will not be discussed here because that goes beyond the remit of this review. Please refer to Maleki et al. [103] and Luo et al. [104] for a review of this topic.
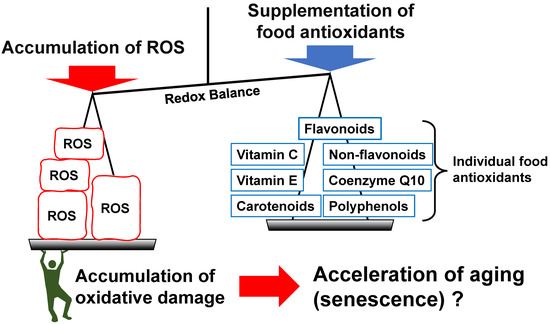
Figure 2.
A typical illustration showing food antioxidants in the redox balance of the body, as mentioned in this review. Reactive oxygen species (ROS) are produced by mitochondria, drugs, oxidized foods, etc., and are considered to induce the redox balance toward an oxidized state, resulting in accelerated aging that is caused by the accumulation of oxidative damage. It is considered that food antioxidants regulate the redox balance and aging.
However, with the current state of scientific technology, it is difficult to identify the effects of individual antioxidants on “aging” and “mortality,” which involve complex factors, and there are few direct reports. On the other hand, there have been several reports on the effects of these individual antioxidants on “various diseases related to aging and mortality” [28,105,106]. This chapter focuses on the relationship between individual antioxidants in foods, which have been reported and relatively well investigated, and diseases related to aging and mortality.
3.1. Vitamins
Vitamins are substances that are essential to animals and humans; they act as the cofactors and precursors of enzymes in the regulation of metabolic processes, do not provide energy, and are essential to the human body [107,108]. The body needs a supply of vitamins through the diet, among which vitamins A, C, and E are called the “antioxidant vitamins”, and their relationship with aging is of great interest [109].
3.1.1. Vitamin C
Vitamin C, also known as L-ascorbic acid, is a lipophobic compound that is involved in collagen synthesis in the body. Vitamin C exists in the human body at the highest concentration among all vitamins and is considered to regulate redox status in the body [110,111,112]. In a cohort study of 17,304 middle-aged and elderly Europeans aged 42 to 82 years old, Lewis et al. reported that an adequate intake of Vitamin C is necessary to reduce the progression of frailty and sarcopenia caused by increased oxidative stress due to aging [113]. Qu et al. reported that Vitamin C inhibited prelamin A expression and the secretion of inflammatory mediators that induce cellular aging in subchondral bone mesenchymal stem cells [114]. Laboratory mice lifespans were increased with an amount of Vitamin C in drinking water, in a study using gluconolactone oxidase-deficient mice [115]. This report suggested that Vitamin C modulates the stress response in the endoplasmic reticulum and the lifespan of mice. Oxidative damage to the brain due to aging is regarded as one of the factors that cause brain dysfunction. Experiments in mouse models of Alzheimer’s disease indicate that Vitamin C deficiency in the brain may affect the redox balance and accelerate the generation of amyloid-beta, an initiator of oxidative stress in Alzheimer’s disease [116]. On the other hand, a review by Kaźmierczak-Barańska et al. summarized that vitamin C is important as a pro-oxidant related to the upregulation of DNA repair and other biological functions [117]. They noted that the function of vitamin C varies in different cell lines and conditions, which makes research difficult. The “two faces” of vitamin C, consisting of its functions as both an anti- and pro-oxidant, may make it difficult to elucidate the detailed functions of such antioxidants [117]. With the continued advancement of science and technology in general, it is necessary to fully understand the effects of these antioxidants and pro-oxidants, and more progress should be achieved in the near future.
3.1.2. Vitamin E
Vitamin E, also known as α-tocopherol, is a lipophilic compound. There are other analogs, such as other tocopherols (β-, γ-, δ-) and tocotrienols (α-, β-, γ-, δ-), but only α-tocopherol can be called “vitamin E” [118]. Vitamin E is localized in cell membranes and is thought to play a role in protecting cells from oxidative damage [96]. The α-tocopherol, β-carotene cancer prevention (ATBC) study, a famous large-scale US trial that examined cancer prevention effects, found that an intake of α-tocopherol and β-carotene had no effect on mortality from liver cancer or chronic liver disease [119]. However, in 2019, Huang et al. reported on a 30-year cohort study of subjects in the ATBC study, which showed that plasma vitamin E levels were associated with a reduced risk of all-cause mortality and death from all major causes [120]. Vitamin E deficiency is thought to cause the increased fragility of red blood cells and the degeneration of neurons, especially peripheral axons, and dorsal horn neurons. Mangialasche et al. quantified vitamin E concentration in the serum of patients with Alzheimer’s disease, mild cognitive impairment, and normal cognitive function, and reported that the concentration was significantly lower in patients with Alzheimer’s disease and dementia than in subjects with normal cognitive function [121]. It has also been reported that the long-term intake of vitamin E suppressed the shortening of telomeres in the peripheral blood mononuclear cells of Alzheimer’s patients [122]. It should also be noted that each vitamin E analog has different biological activities. Tucker compared serum leukocyte telomere length with vitamin E and gamma-tocopherol concentrations in 5768 US adults and explained that high levels of gamma-tocopherol in the blood accelerated telomere length loss (vitamin E was not significant) and may accelerate cellular senescence [123]. A cohort study of 580 American people by Hanson et al. showed that there was a positive correlation between dietary vitamin E intake and lung function, but an inverse correlation between serum γ-tocopherol levels and lung function [124]. The ratio of vitamin E and its analogs in foods, or the coexistence of various other compounds, may have an effect on aging.
3.1.3. Carotenoids
Carotenoids, also known as provitamin A, are lipophilic pigments that are classified into two groups, carotenes and xanthophylls, based on their polarity. The blood concentrations of carotenoids were found to be decreased in the elderly and in Alzheimer’s disease patients [125]. Huang et al. confirmed the relationship between serum parameters and mortality in a cohort study of 29,103 men in the ATBC study and reported that higher serum β-carotene concentration was associated with lower mortality from cardiovascular disease, heart disease, stroke, cancer, and all causes of death [126]. Min et al. analyzed the plasma of 3660 people in the US and showed that elevated blood β-carotene levels were correlated positively with the length of leukocyte telomeres [127]. Experimental models using Caenorhabditis elegans show that the continuous intake of astaxanthin from a young age leads to the increased expression of genes encoding superoxide dismutase (SOD) and catalase and protects the mitochondria and nuclear organelle through the nuclear transfer of DAF-16 protein, resulting in lifespan extension [128]. Wu et al. reported that astaxanthin intake was associated with anti-aging in the D-galactose-induced rat brain aging model by the maintenance of antioxidant enzyme activity, the suppression of oxidative enzyme expression, and an increase in brain-derived neurotrophic factor (BDNF) [129].
3.2. Polyphenols
“Polyphenols” is a general term for compounds with multiple phenolic hydroxy groups in their molecules and are mainly present in plants. Polyphenols can be further classified, depending on their chemical structure, but most of the polyphenols in foods are flavonoids. Flavonoids are a group of phenyl compounds with a structure consisting of two benzene rings connected by three carbon atoms (diphenylpropane), and various reports have been made in recent years on their functional health properties. Although more than 5000 polyphenols have been reported, we will broadly classify them into flavonoids and non-flavonoids in this section and introduce the relationship between each typical compound and aging.
3.2.1. Flavonoids
Quercetin is one of the most recognized flavonoids and is reported to have antioxidant and anti-inflammatory effects. El-Far et al. investigated the effect of quercetin intake on an aged rat model induced by D-galactose [130]. As a result, they reported that quercetin intake suppressed apoptosis and the elevation of inflammatory markers by inducing the expression of anti-apoptotic markers related to aging in the pancreas and kidneys of rats. In a study using mouse oocytes, quercetin also contributed to oocyte maturation and embryo development by reducing age-related increases in mitochondrial oxidative stress and by regulating mitochondrial dysfunction [131]. Geng et al. used a Werner’s syndrome model, based on human mesenchymal stem cells to screen for potential natural compounds with anti-aging effects, and identified quercetin. They also reported that quercetin may reduce cellular aging by improving cell proliferation and the repair of the heterochromatin structure [132].
Anthocyanins are polyphenols that are well known due to the presence of the French paradox. Using aged rat models, Li et al. found that anthocyanin intake improves total antioxidant capacity and may ameliorate aging caused by oxidative stress through the induction of autophagy [133]. As is the case with polyphenols in general, it is also important to consider the relationship between their absorption, metabolism, and aging. For example, when anthocyanins are administered orally, the majority of molecules are not transferred into the bloodstream via antioxidant activity, which is likely to be because of glucuronidation and/or sulfate conjugation in the liver and small intestine [134,135].
Isoflavones have been reported to exhibit estrogen-like effects due to their structural similarities, and to contribute to the regulation of hormones in women. Studies on age-related disorders have also reported that genistein prevents the acquisition of insulin resistance in aged rats, but the hormone replacement effect of genistein is only effective in early menopause, not in older age [136].
3.2.2. Non-Flavonoids
Chlorogenic acid is a type of monophenol recognized for its presence in beverages such as tea and coffee. Li et al. reported that the oral administration of a chlorogenic acid-phospholipid complex to senescence-accelerated mice (SAMP8) for two weeks suppressed the post-myocardial infarction response of the aged heart [137].
Resveratrol, a polyphenol that is prolific in fruit peels, is known for having a strong antioxidant capacity in in vitro studies. Gines et al. reported that the oral administration of resveratrol to senescence-accelerated mice (SAMP8) reduced inflammatory factor expression, inhibited apoptosis and oxidative stress in the mouse pancreas, and had a protective effect against age-related pancreatic damage [138]. Resveratrol switches the SIRT-1 gene, which is known as the longevity gene. Several cognitive function studies have reported that the long-term administration of resveratrol has a protective effect against hippocampal and neuronal damage [139,140]. Caldeira et al. reported that resveratrol has antioxidant and anti-inflammatory effects, but its mechanism of action varies with cell age [141]. The antioxidant effect of resveratrol was found to be mediated by the SIRT1/AMPK pathway in middle-aged mononuclear cells, but not in aged cells. Therefore, the biological activity of resveratrol may change with age. Semba et al. conducted a cohort study of 783 Italian seniors aged 65 years or older and noted that in the elderly, resveratrol metabolites were not associated with inflammatory markers, heart disease, cancer, or mortality and that the resveratrol levels reached in the Western diet had virtually no effect on health status or longevity [142]. Furthermore, recent findings have raised concerns that the intake of antioxidants such as resveratrol may rather inhibit the beneficial effect of exercise on improving vascular function in humans [143].
Curcumin is reported in terms of its functions of anti-aging via an anti-inflammatory effect [144,145]. The intake of curcumin for 6 months (200 mg/kg) suppressed the aging process by affecting anti-aging markers (decreased C-reactive protein levels, increased malondialdehyde levels, and nitric oxide levels) in aged albino rats. [146]. In vitro studies have shown that curcumin preserved endothelial cells (HUVEC) from H2O2-induced premature senescence via endothelial nitric oxide synthase phosphorylation and silent information regulator (SIRT)-1 expression [147]. Curcumin intake over several weeks (100 mg/kg/day) is reported to delay the aging process of oocytes in the mouse model through anti-aging-related genes (SIRT1 and SIRT3) [148]. Furthermore, 10–40 times higher absorbable and brain-accessible curcumin nanomicelles prevented the mitochondrial dysfunction involved in brain aging and neurodegeneration [149]. As a result of investigating curcumin intake on an obesity-related cognitive dysfunction mouse model, dietary curcumin and caloric restriction worked positively on the frontal cortical functions, regardless of effects on adiposity [150]. A combination of in vivo experiments and computer simulations revealed that curcumin upregulates antioxidant enzymes and improves memory by binding to β-secretase 1 and amine oxidase A [151]. In middle-aged monkeys, daily curcumin treatment for 14–18 months improved only the spatial memory, suggesting the anti-aging effect of curcumin might be dependent upon the stage of aging [152].
3.3. Coenzyme Q10
Endogenous antioxidants exist in the body to maintain the redox balance. One of the most widely recognized endogenous antioxidants is coenzyme Q10, which is also available as a supplement. Gutierrez-Mariscal et al. observed the effects on oxidative stress in 20 subjects who consumed three random, same-calorie diets (the Mediterranean diet, the Mediterranean diet plus coenzyme Q10, and a diet rich in saturated fatty acids) for four weeks and found that the DNA-protective effects of the Mediterranean diet were enhanced by coenzyme Q10 [153]. The supplementation of aged mice with coenzyme Q10 was found to delay the decay of ovarian reserves and restore mitochondrial gene expression in oocytes, with associated functional improvements in the body [154]. Zhang et al. reported that coenzyme Q10 inhibits aging through the Akt/mTOR signaling pathway in D-galactose-treated mesenchymal stem cells [155]. Aging models in which hydrogen peroxide was added to human vascular endothelial cells have also reported that coenzyme Q10 has an aging delay effect by suppressing the expression of genes related to the secretory phenotype associated with aging, inhibiting intracellular ROS production, increasing nitric oxide (NO) production by increasing endothelial nitric oxide synthase (eNOS) expression, and promoting mitochondrial function [156].
4. Limitations of the Current Investigations
In the first chapter of this review, the various theories of aging that have been reported were presented. From the past to the present, many theories have tried to explain the aging process, but no single theory can fully explain it because the process is essentially a complex scenario characterized by changes occurring at different levels of the biological system. However, it is noteworthy that today’s research in the field of aging has developed based on these theories of aging. Existing theories of aging would help to inspire new approaches to further the current understanding of the relationship between food and the aging process. In the future, theories of aging based on new discoveries and perspectives may well be proposed to elucidate the reality of diet and food components in aging.
In the second chapter, research conducted to investigate the impact of foodstuffs on life expectancy and mortality was introduced. Diet plays an important role in aging because the human body is composed of compounds found in the foods that are consumed every day. Considering the research reports presented in the second chapter, it seems that dietary patterns may have a significant impact on mortality. On the other hand, the evaluation of the physiological effects of foods composed of multiple molecules, unlike single molecules, seems to be difficult to explain with the current technology. Therefore, it is difficult to connect and discuss the findings of the cohort studies presented in this chapter with the individual theories of aging presented in the first chapter. However, in very recent years, with the development of AI technology, new methods of dietary assessment and physiological effects have begun to be established [157,158,159]. In addition, with the development of nanotechnology, the concept of “smart food,” in which food is processed at the nanoscale to enhance its bioavailability and shelf life, will also lead to significant progress in aging research [160]. Considering these concepts, it will be even more important to elucidate the detailed role of food-derived antioxidants in the regulation of aging. Aging is affected not only by the kinds of food human ingests but also by the surrounding environment; for example, the natural environment, air, climate, soil, artificial environment, social environment, urban green spaces, economic background, social cohesion, socioeconomic status, and physical activity [161]. These complex factors and their relationship to food and aging are extensive and are beyond the scope of this review, but they need to be considered and evaluated.
In the third chapter, individual antioxidants in the human dietary resources and their effects on aging and related diseases (including lifespan extension) were summarized. Most of the previous research on food-derived antioxidants and lifespan has been based on the theory of oxidative damage, as introduced in the first chapter, and many reports have suggested that antioxidative activity is a major factor in promoting longevity. The various foods presented in the second section that may extend the lifespan seem to be rich in the individual antioxidants discussed in this chapter. The concentrations of food-derived antioxidants in the body are found to decrease with aging (Figure 3). Therefore, it may be a possibility that individual antioxidants might contribute to lifespan extension, but a clear cause-and-effect relationship has not yet been elucidated. Current methods for elucidating the relationship between individual antioxidants and aging, such as those described in this chapter, may yet have some details to be elucidated. Although the mechanisms of the effects of individual antioxidants on individual diseases, including Alzheimer’s disease and lifestyle-related diseases, are becoming clearer, it seems that experimental models and evaluation methods to explain “aging” are currently insufficient. For example, the quantification of DNA repair capacity in humans regarding age-related genomic instability has not yet been established [162]. Proteostasis defects have been identified as a novel mechanism of aging but, to date, there is no effective technology for this purpose [163]. Stem cell depletion may also be an important marker of aging, but methods for its evaluation have not yet been established [164]. Telomeres, which are an important indicator of aging, have not yet been put to practical use in clinical practice because the current evaluation methods have problems with the accuracy of analysis, such as heterogeneity among cells and individuals [1]. As the technology for such evaluation methods develops, the concept of food-derived antioxidants and aging will become more visible. It will also be necessary to try to bring the two separate interpretations of “food” as a whole and “individual nutrients” closer together.
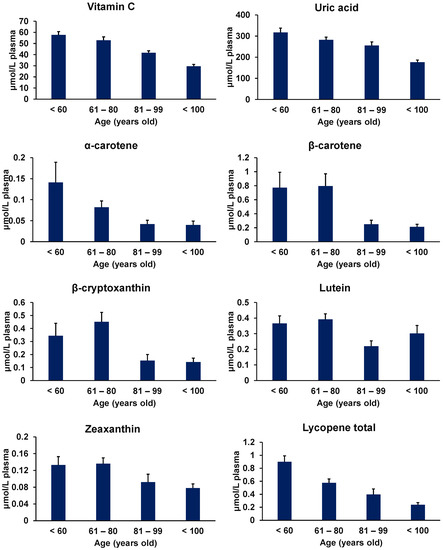
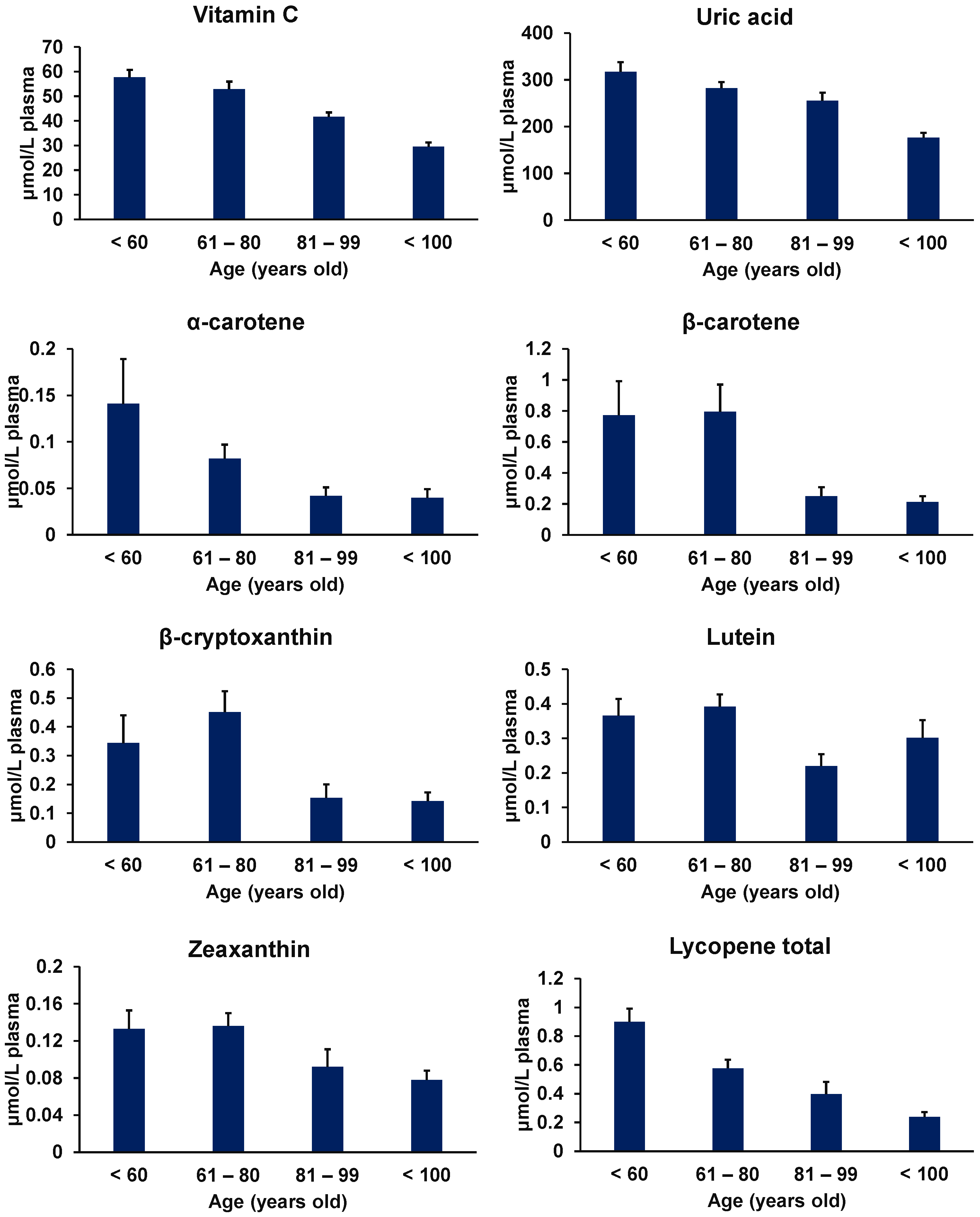
Figure 3.
Age-related decline in the plasma concentrations of food antioxidants (vitamin C, uric acid, α-carotene, β-carotene, β-cryptoxanthin, lutein, zeaxanthin, and lycopene in total). (Figure modified from Miyazawa et al. [160]).
What is the biological meaning of oxidative stress in aging? Although various reports have shown that techniques for measuring age-related ROS are available, their relationship with aging is still unclear [165]. As mentioned in the concept of mitohormesis introduced at the beginning of the third chapter, moderate oxidative stress in the body may have the potential to improve the lifespan. Based on the concept of the oxidative damage theory of aging, it seems that higher amounts of antioxidants in food would benefit longevity, but this may not necessarily be the case. Gladyshev et al. stated that excess antioxidant supplementation disrupts the redox balance of the organism and there is a risk of accelerated aging [166]. Pérez et al. reported that transgenic mice that produced more antioxidant enzymes in their bodies did not live as long as normal mice [167]. Furthermore, when using catalase-deficient mice, Pérez-Estrada et al. reported that catalase contributes to lifespan extension by regulating the lipid metabolism in the liver without contributing to oxidative stress [168]. Van Raamsdonk et al. found that C. elegans (sod-12345) without SOD activity had a normal lifespan, although it was more sensitive to all stresses [169]. Furthermore, whether superoxide extends or shortens the lifespan depends on the genotype of the strain and the initial concentration of superoxide, which raises questions about the oxidative damage theory. In addition to the oxidative damage theory, it will be important to consider food antioxidants and aging on the basis of other aging theories. Throughout this review, we can report that there is definitely a promising future for diets that control aging, although more research needs to be performed to clarify the relationship between food antioxidants and aging.
5. Conclusions and Outlook
Research on food antioxidants and aging has been widely based on the “oxidative damage theory” among the aging theories. Most of the previously reported cohort studies suggest that food antioxidant components may affect the lifespan; however, there is no direct evidence and the effect remains unclear. New technologies are expected to be developed in the future that can show that individual food antioxidants have the potential to extend the lifespan; moreover, the technology can evaluate the relationship between aging and the food as a whole, which is composed of various food antioxidants.
Author Contributions
Conceptualization, T.M. and C.A.; writing, review, and editing, T.M., C.A., G.C.B., A.M. and M.T.; supervision, T.M., A.M. and M.T. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Colloca, G.; Di Capua, B.; Bellieni, A.; Fusco, D.; Ciciarello, F.; Tagliaferri, L.; Valentini, V.; Balducci, L. Biological and functional biomarkers of aging: Definition, characteristics, and how they can impact everyday cancer treatment. Curr. Oncol. Rep. 2020, 22, 115. [Google Scholar] [CrossRef]
- Gladyshev, T.V.; Gladyshev, V.N. A disease or not a disease? Aging as a pathology. Trends. Mol. Med. 2016, 22, 995–996. [Google Scholar] [CrossRef] [PubMed]
- López-Otín, C.; Blasco, M.A.; Partridge, L.; Serrano, M.; Kroemer, G. The hallmarks of aging. Cell 2013, 153, 1194–1217. [Google Scholar] [CrossRef]
- Vina, J.; Borras, C.; Miquel, J. Theories of ageing. IUBMB Life 2007, 59, 249–254. [Google Scholar] [CrossRef] [PubMed]
- Hayflick, L.; Moorhead, P.S. The serial cultivation of human diploid cell strains. Exp. Cell Res. 1961, 25, 585–621. [Google Scholar] [CrossRef]
- Vidacek, N.S.; Nanic, L.; Ravlic, S.; Sopta, M.; Geric, M.; Gajski, G.; Garaj-Vrhovac, V.; Rubelj, I. Telomeres, Nutrition, and Longevity: Can We Really Navigate Our Aging? J. Gerontol. A Biol. Sci. Med. Sci. 2017, 73, 39–47. [Google Scholar] [CrossRef] [PubMed]
- Ferron, S.R.; Marques-Torrejon, M.A.; Mira, H.; Flores, I.; Taylor, K.; Blasco, M.A.; Farinas, I. Telomere shortening in neural stem cells disrupts neuronal differentiation and neuritogenesis. J. Neurosci. 2009, 29, 14394–14407. [Google Scholar] [CrossRef]
- Derevyanko, A.; Whittemore, K.; Schneider, R.P.; Jiménez, V.; Bosch, F.; Blasco, M.A. Gene therapy with the TRF1 telomere gene rescues decreased TRF1 levels with aging and prolongs mouse health span. Aging Cell 2017, 16, 1353–1368. [Google Scholar] [CrossRef] [PubMed]
- de Jesus, B.B.; Vera, E.; Schneeberger, K.; Tejera, A.M.; Ayuso, E.; Bosch, F.; Blasco, M.A. Telomerase gene therapy in adult and old mice delays aging and increases longevity without increasing cancer. EMBO Mol. Med. 2012, 4, 691–704. [Google Scholar] [CrossRef] [PubMed]
- Orgel, L.E. The maintenance of the accuracy of protein synthesis and its relevance to ageing. Proc. Natl. Acad. Sci. USA 1963, 49, 517–521. [Google Scholar] [CrossRef] [PubMed]
- Troen, B.R. The biology of aging. Mt. Sinai J. Med. 2003, 70, 3–22. [Google Scholar] [PubMed]
- Edelmann, P.; Gallant, J. On the translational error theory of aging. Proc. Natl. Acad. Sci. USA 1977, 74, 3396–3398. [Google Scholar] [CrossRef] [PubMed]
- Weismann, A. Essays Upon Heredity and Kindred Biological Problems; Poulton, E.B., Selmar, S., Arthur, S.E., Eds.; Clarendon Press: Oxford, UK, 1889. [Google Scholar]
- Hanson, R.W.; Hakimi, P. Born to run; the story of the PEPCK-Cmus mouse. Biochimie 2008, 90, 838–842. [Google Scholar] [CrossRef][Green Version]
- Van Raamsdonk, J.M.; Hekimi, S. Deletion of the mitochondrial superoxide dismutase sod-2 extends lifespan in Caenorhabditis elegans. PLoS Genet. 2009, 5, e1000361. [Google Scholar] [CrossRef] [PubMed]
- Bjorksten, J. The Crosslinkage Theory of Aging. Finska Kemists. Medd. 1971, 2, 23–38. [Google Scholar] [CrossRef]
- Monnier, V.M.; Mustata, G.T.; Biemel, K.L.; Reihl, O.; Lederer, M.O.; Dai, Y.; Sell, D.R. Cross-linking of the extracellular matrix by the maillard reaction in aging and diabetes: An update on “a puzzle nearing resolution”. Ann. N. Y. Acad. Sci. 2005, 1043, 533–544. [Google Scholar] [CrossRef] [PubMed]
- Bjorksten, J. Theoretical Aspects of Aging; Rockstein, M., Ed.; Academic Press: New York, NY, USA, 1974; p. 43. [Google Scholar]
- Watad, A.; Bragazzi, N.L.; Adawi, M.; Amital, H.; Toubi, E.; Porat, B.-S.; Shoenfeld, Y. Autoimmunity in the elderly: Insights from basic science and clinics-a mini-review. Gerontology 2017, 63, 515–523. [Google Scholar] [CrossRef] [PubMed]
- Ma, S.; Wang, C.; Mao, X.; Hao, Y. B cell dysfunction associated with aging and autoimmune diseases. Front. Immunol. 2019, 10, 318. [Google Scholar] [CrossRef]
- Shanley, D.P.; Aw, D.; Manley, N.R.; Palmer, D.B. An evolutionary perspective on the mechanisms of immunosenescence. Trends Immunol. 2009, 30, 374–381. [Google Scholar] [CrossRef] [PubMed]
- Cooper, G.S.; Stroehla, B.C. The epidemiology of autoimmune diseases. Autoimmun. Rev. 2003, 2, 119–125. [Google Scholar] [CrossRef]
- Tessier, F.J. The Maillard reaction in the human body. The main discoveries and factors that affect glycationLa réaction de Maillard dans le corps humain. Découvertes majeures et facteurs qui affectent la glycation. Pathol. Biol. 2010, 58, 214–219. [Google Scholar] [CrossRef] [PubMed]
- Simm, A. Protein glycation during aging and in cardiovascular disease. J. Proteom. 2013, 92, 248–259. [Google Scholar] [CrossRef] [PubMed]
- Harman, D. Aging: A theory based on free radical and radiation chemistry. J. Gerontol. 1956, 11, 298–300. [Google Scholar] [CrossRef] [PubMed]
- Stadtman, E.R. Protein oxidation and aging. Science 1992, 257, 1220–1224. [Google Scholar] [CrossRef]
- Agarwal, S.; Sohal, R.S. DNA oxidative damage and life expectancy in houseflies. Proc. Natl. Acad. Sci. USA 1994, 91, 12332–12335. [Google Scholar] [CrossRef]
- Miyazawa, T. Lipid hydroperoxides in nutrition, health, and diseases. Proc. Jpn. Acad. Ser. B Phys. Biol. Sci. 2021, 97, 161–196. [Google Scholar] [CrossRef]
- Cadenas, E.; Davies, K.J. Mitochondrial free radical generation, oxidative stress, and aging. Free Radic. Biol. Med. 2000, 29, 222–230. [Google Scholar] [CrossRef]
- Kulinsky, V.I. Biochemical aspects of inflammation. Biochemistry 2007, 72, 595–607. [Google Scholar] [CrossRef] [PubMed]
- Franceschi, C.; Garagnani, P.; Parini, P.; Giuliani, C.; Santoro, A. Inflammaging: A new immune-metabolic viewpoint for age-related diseases. Nat. Rev. Endocrinol. 2018, 14, 576–590. [Google Scholar] [CrossRef]
- Fulop, T.; Larbi, A.; Witkowski, J.M. Human Inflammaging. Gerontology 2019, 65, 495–504. [Google Scholar] [CrossRef] [PubMed]
- Santoro, A.; Martucci, M.; Conte, M.; Capri, M.; Franceschi, C.; Salvioli, S. Inflammaging, hormesis and the rationale for anti-aging strategies. Ageing Res. Rev. 2020, 64, 101142. [Google Scholar] [CrossRef] [PubMed]
- Laberge, R.M.; Awad, P.; Campisi, J.; Desprez, P.Y. Epithelial-mesenchymal transition induced by senescent fibroblasts. Cancer Microenviron. 2012, 5, 39–44. [Google Scholar] [CrossRef] [PubMed]
- Gasek, N.S.; Kuchel, G.A.; Kirkland, J.L.; Xu, M. Strategies for targeting senescent cells in human disease. Nat. Aging 2021, 1, 870–879. [Google Scholar] [CrossRef]
- Colchero, F.; Aburto, J.M.; Archie, E.A.; Boesch, C.; Breuer, T.; Campos, F.A.; Collins, A.; Conde, D.A.; Cords, M.; Crockford, C.; et al. The long lives of primates and the ‘invariant rate of ageing’ hypothesis. Nat. Commun. 2021, 12, 3666. [Google Scholar] [CrossRef] [PubMed]
- Robert, J.H. Successful Aging. Gerontologist 1961, 1, 8–13. [Google Scholar]
- Cumming, E.; Henry, W. Growing Old: The Process of Disengagement. Basic Books, New York, 1961. (Reprint: Arno, New York, 1979, ISBN 0405 118147). Ageing Soc. 1991, 11, 217–220. [Google Scholar]
- Atchley, R.C. Activity theory. In The Encyclopedia of Aging, 2nd ed.; Maddox, G.L., Ed.; Springer: Berlin/Heidelberg, Germany, 1995; pp. 9–12. [Google Scholar]
- Elder, G.H., Jr. Life Course and Human Development. In Handbook of Child Psychology; Damon, W., Ed.; Wiley: New York, NY, USA, 1998; pp. 939–991. [Google Scholar]
- Cheval, B.; Sieber, S.; Guessous, I.; Orsholits, D.; Courvoisier, D.S.; Kliegel, M.; Stringhini, S.; Swinnen, S.P.; Burton-Jeangros, C.; Cullati, S.; et al. Effect of early-and adult-life socioeconomic circumstances on physical inactivity. Med. Sci. Sports Exerc. 2018, 50, 476–485. [Google Scholar] [CrossRef]
- Selvamani, Y.; Arokiasamy, P. Association of life course socioeconomic status and adult height with cognitive functioning of older adults in India and China. BMC Geriatr. 2021, 21, 354. [Google Scholar] [CrossRef]
- Atchley, R.C. Continuity Theory. In The Encyclopedia of Aging, 2nd ed.; Maddox, G.L., Ed.; Springer: Berlin/Heidelberg, Germany, 1995; pp. 227–230. [Google Scholar]
- Dodig, S.; Cepelak, I.; Pavic, I. Hallmarks of senescence and aging. Biochem. Med. 2019, 29, 030501. [Google Scholar] [CrossRef] [PubMed]
- Cheng, W.-H.; Bohr, V.A.; de Cabo, R. Nutrition and aging. Mech. Ageing Dev. 2010, 131, 223–224. [Google Scholar] [CrossRef] [PubMed]
- Flatt, T.; Partridge, L. Horizons in the evolution of aging. BMC Biol. 2018, 16, 93. [Google Scholar] [CrossRef] [PubMed]
- Trichopoulou, A.; Costacou, T.; Bamia, C.; Trichopoulos, D. Adherence to a Mediterranean diet and survival in a Greek population. N. Engl. J. Med. 2003, 348, 2599–2608. [Google Scholar] [PubMed]
- Levitan, E.B.; Lewis, C.E.; Tinker, L.F.; Eaton, C.B.; Ahmed, A.; Manson, J.E.; Snetselaar, L.G.; Martin, L.W.; Trevisan, M.; Howard, B.V.; et al. Mediterranean and DASH diet scores and mortality in women with heart failure: The Women’s Health Initiative. Circ. Heart Fail. 2013, 6, 1116–1123. [Google Scholar] [CrossRef] [PubMed]
- Olsen, A.; Egeberg, R.; Halkjær, J.; Christensen, J.; Overvad, K.; Tjønneland, A. Healthy aspects of the Nordic diet are related to lower total mortality. J. Nutr. 2011, 141, 639–644. [Google Scholar] [CrossRef]
- Roswall, N.; Sandin, S.; Lof, M.; Skeie, G.; Olsen, A.; Adami, H.O.; Weiderpass, E. Adherence to the Healthy Nordic Food Index and total and cause-specific mortality among Swedish women. Eur. J. Epidemiol. 2015, 30, 509–517. [Google Scholar] [CrossRef]
- Abe, S.; Zhang, S.; Tomata, Y.; Tsuduki, T.; Sugawara, Y.; Tsuji, I. Japanese diet and survival time: The Ohsaki Cohort 1994 study. Clin. Nutr. 2020, 39, 298–303. [Google Scholar] [CrossRef] [PubMed]
- Okada, E.; Nakamura, K.; Ukawa, S.; Wakai, K.; Date, C.; Iso, H.; Tamakoshi, A. The Japanese food score and risk of all-cause, CVD and cancer mortality: The Japan Collaborative Cohort Study. Br. J. Nutr. 2018, 120, 464–471. [Google Scholar] [CrossRef]
- Bazzano, L.A.; He, J.; Ogden, L.G.; Loria, C.M.; Vupputuri, S.; Myers, L.; Whelton, P.K. Fruit and vegetable intake and risk of cardiovascular disease in US adults: The first National Health and Nutrition Examination Survey Epidemiologic Follow-up Study. Am. J. Clin. Nutr. 2002, 76, 93–99. [Google Scholar] [CrossRef]
- Leenders, M.; Sluijs, I.; Ros, M.M.; Boshuizen, H.C.; Siersema, P.D.; Ferrari, P.; Weikert, C.; Tjønneland, A.; Olsen, A.; Boutron-Ruault, M.C.; et al. Fruit and vegetable consumption and mortality: European prospective investigation into cancer and nutrition. Am. J. Epidemiol. 2013, 178, 590–602. [Google Scholar] [CrossRef] [PubMed]
- Stefler, D.; Pikhart, H.; Kubinova, R.; Pajak, A.; Stepaniak, U.; Malyutina, S.; Simonova, G.; Peasey, A.; Marmot, M.G.; Bobak, M. Fruit and vegetable consumption and mortality in Eastern Europe: Longitudinal results from the Health, Alcohol and Psychosocial Factors in Eastern Europe study. Eur. J. Prev. Cardiol. 2016, 23, 493–501. [Google Scholar] [CrossRef]
- Zhang, X.; Shu, X.O.; Xiang, Y.B.; Yang, G.; Li, H.; Gao, J.; Cai, H.; Gao, Y.T.; Zheng, W. Cruciferous vegetable consumption is associated with a reduced risk of total and cardiovascular disease mortality. Am. J. Clin. Nutr. 2011, 94, 240–246. [Google Scholar] [CrossRef] [PubMed]
- Saglimbene, V.M.; Wong, G.; Ruospo, P.; Palmer, S.C.; Garcia-Larsen, V.; Natale, P.; Teixeira-Pinto, A.; Campbell, K.L.; Carrero, J.-J.; Stenvinkel, P.; et al. Fruit and vegetable intake and mortality in adults undergoing maintenance hemodialysis. Clin. J. Am. Soc. Nephrol. 2019, 14, 250–260. [Google Scholar] [CrossRef] [PubMed]
- Appleby, P.N.; Crowe, F.L.; Bradbury, K.E.; Travis, R.C.; Key, T.J. Mortality in vegetarians and comparable nonvegetarians in the United Kingdom. Am. J. Clin. Nutr. 2016, 103, 218–230. [Google Scholar] [CrossRef] [PubMed]
- Imran, T.F.; Kim, E.; Buring, J.E.; Lee, I.M.; Gaziano, J.M.; Djousse, L. Nut consumption, risk of cardiovascular mortality, and potential mediating mechanisms: The Women’s Health Study. J. Clin. Lipidol. 2021, 15, 266–274. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Yang, M.; Kenfield, S.A.; Hu, F.B.; Stampfer, M.J.; Willett, W.C.; Fuchs, C.S.; Giovannucci, E.L.; Bao, Y. Nut consumption and prostate cancer risk and mortality. Br. J. Cancer 2016, 115, 371–374. [Google Scholar] [CrossRef] [PubMed]
- Bonaccio, M.; Castelnuovo, A.D.; Curtis, D.A.; Costanzo, S.; Bracone, F.; Persichillo, M.; Donati, M.B.; Gaetano, G.; Iacoviello, L. Nut consumption is inversely associated with both cancer and total mortality in a Mediterranean population: Prospective results from the Moli-sani study. Br. J. Nutr. 2015, 114, 804–811. [Google Scholar] [CrossRef] [PubMed]
- Yamakawa, M.; Wada, K.; Koda, S.; Uji, T.; Nakashima, Y.; Onuma, S.; Oba, S.; Nagata, C. Associations of total nut and peanut intakes with all-cause and cause-specific mortality in a Japanese community: The Takayama study. Br. J. Nutr. 2022, 127, 1378–1385. [Google Scholar] [CrossRef] [PubMed]
- Eslamparast, T.; Sharafkhah, M.; Poustchi, H.; Hashemian, M.; Dawsey, S.M.; Freedman, N.D.; Boffetta, P.; Abnet, C.C.; Etemadi, A.; Pourshams, A.; et al. Nut consumption and total and cause-specific mortality: Results from the Golestan Cohort Study. Int. J. Epidemiol. 2017, 46, 75–85. [Google Scholar] [CrossRef] [PubMed]
- Luo, C.; Zhang, Y.; Ding, Y.; Shan, Z.; Chen, S.; Yu, M.; Hu, F.B.; Liu, L. Nut consumption and risk of type 2 diabetes, cardiovascular disease, and all-cause mortality: A systematic review and meta-analysis. Am. J. Clin. Nutr. 2014, 100, 256–269. [Google Scholar] [CrossRef] [PubMed]
- Guasch-Ferre, M.; Bullo, M.; Martinez-Gonzalez, M.A.; Ros, E.; Corella, D.; Estruch, R.; Fito, M.; Aros, F.; Warnberg, J.; Fiol, M.; et al. Frequency of nut consumption and mortality risk in the PREDIMED nutrition intervention trial. BMC Med. 2013, 11, 164. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Gu, K.; Wang, K.; Cai, H.; Zheng, W.; Bao, P.; Shu, X.-O. Nut consumption in association with overall mortality and recurrence/disease-specific mortality among long-term breast cancer survivors. Int. J. Cancer 2022, 150, 572–579. [Google Scholar] [CrossRef]
- Abe, S.K.; Saito, E.; Sawada, N.; Tsugane, S.; Ito, H.; Lin, Y.; Tamakoshi, A.; Sado, J.; Kitamura, Y.; Sugawara, Y.; et al. Green tea consumption and mortality in Japanese men and women: A pooled analysis of eight population-based cohort studies in Japan. Eur. J. Epidemiol. 2019, 34, 917–926. [Google Scholar] [CrossRef] [PubMed]
- Unno, K.; Nakamura, Y. Green tea suppresses brain aging. Molecules 2021, 26, 4897. [Google Scholar] [CrossRef] [PubMed]
- Hao, Z.; Liu, Y.; Li, Y.; Song, W.; Yu, J.; Li, H.; Wang, W. Association between longevity and element levels in food and drinking water of typical Chinese longevity area. J. Nutr. Health Aging 2016, 20, 897–903. [Google Scholar] [CrossRef]
- Wang, X.; Chen, H.; Ouyang, Y.; Liu, J.; Zhao, G.; Bao, W.; Yan, M. Dietary calcium intake and mortality risk from cardiovascular disease and all causes: A meta-analysis of prospective cohort studies. BMC Med. 2014, 12, 158. [Google Scholar] [CrossRef] [PubMed]
- Navarro, A.M.; Martinez-Gonzalez, M.A.; Gea, A.; Grosso, G.; Martín-Moreno, J.M.; Lopez-Garcia, E.; Martin-Calvo, N.; Toledo, E. Coffee consumption and total mortality in a Mediterranean prospective cohort. Am. J. Clin. Nutr. 2018, 108, 1113–1120. [Google Scholar] [CrossRef]
- Shadyab, A.H.; Manson, J.E.; Luo, J.; Haring, B.; Saquib, N.; Snetselaar, L.G.; Chen, J.C.; Groessl, E.J.; Wassertheil-Smoller, S.; Sun, Y.; et al. Associations of coffee and tea consumption with survival to age 90 years among older women. J. Am. Geriatr. Soc. 2020, 68, 1970–1978. [Google Scholar] [CrossRef] [PubMed]
- Takata, Y.; Shu, X.O.; Gao, Y.T.; Li, H.; Zhang, X.; Gao, J.; Cai, H.; Yang, G.; Xiang, Y.B.; Zheng, W. Red meat and poultry intakes and risk of total and cause-specific mortality: Results from cohort studies of Chinese adults in Shanghai. PLoS ONE 2013, 8, e56963. [Google Scholar]
- Lv, Y.; Kraus, V.B.; Gao, X.; Yin, Z.; Zhou, J.; Mao, C.; Duan, J.; Zeng, Y.; Brasher, M.S.; Shi, W.; et al. Higher dietary diversity scores and protein-rich food consumption were associated with lower risk of all-cause mortality in the oldest old. Clin. Nutr. 2020, 39, 2246–2254. [Google Scholar] [CrossRef]
- Stefler, D.; Brett, D.; Sarkadi-Nagy, E.; Kopczynska, E.; Detchev, S.; Bati, A.; Scrob, M.; Koenker, D.; Aleksov, B.; Douarin, E.; et al. Traditional Eastern European diet and mortality: Prospective evidence from the HAPIEE study. Eur. J. Nutr. 2021, 60, 1091–1100. [Google Scholar] [CrossRef]
- Schnabel, L.; Kesse-Guyot, E.; Alles, B.; Touvier, M.; Srour, B.; Hercberg, S.; Buscail, C.; Julia, C. Association between ultraprocessed food consumption and risk of mortality among middle-aged adults in France. JAMA Intern. Med. 2019, 179, 490–498. [Google Scholar] [CrossRef]
- Bonaccio, M.; Di Castelnuovo, A.; Costanzo, S.; De Curtis, A.; Persichillo, M.; Sofi, F.; Cerletti, C.; Donati, M.B.; de Gaetano, G.; Iacoviello, L. Ultra-processed food consumption is associated with increased risk of all-cause and cardiovascular mortality in the Moli-sani Study. Am. J. Clin. Nutr. 2021, 113, 446–455. [Google Scholar] [CrossRef] [PubMed]
- Blanco-Rojo, R.; Sandoval-Insausti, H.; Lopez-Garcia, E.; Graciani, A.; Ordovas, J.M.; Banegas, J.R.; Rodriguez-Artalejo, F.; Guallar-Castillon, P. Consumption of ultra-processed foods and mortality: A national prospective cohort in Spain. Mayo Clin. Proc. 2019, 94, 2178–2188. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Liu, B.; Snetselaar, L.G.; Robinson, J.G.; Wallace, R.B.; Peterson, L.L.; Bao, W. Association of fried food consumption with all cause, cardiovascular, and cancer mortality: Prospective cohort study. BMJ 2019, 364, k5420. [Google Scholar] [CrossRef] [PubMed]
- Veronese, N.; Stubbs, B.; Noale, M.; Solmi, M.; Vaona, A.; Demurtas, J.; Nicetto, D.; Crepaldi, G.; Schofield, P.; Koyanagi, A.; et al. Fried potato consumption is associated with elevated mortality: An 8-y longitudinal cohort study. Am. J. Clin. Nutr. 2017, 106, 162–167. [Google Scholar] [CrossRef]
- Trichopoulou, A.; Martínez-González, M.A.; Tong, T.Y.N.; Forouhi, N.G.; Khandelwal, S.; Prabhakaran, D.; Mozaffarian, D.; de Lorgeril, M. Definitions and potential health benefits of the Mediterranean diet: Views from experts around the world. BMC Med. 2014, 12, 112. [Google Scholar] [CrossRef]
- Crous-Bou, M.; Fung, T.T.; Prescott, J.; Julin, B.; Du, M.; Sun, Q.; Rexrode, K.M.; Hu, F.B.; De Vivo, I. Mediterranean diet and telomere length in Nurses’ Health Study: Population based cohort study. BMJ 2014, 349, g6674. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, S. What is the scientific definition of the Japanese diet from the viewpoint of nutrition and health? Nutr. Rev. 2020, 78, 18–26. [Google Scholar] [CrossRef]
- Colman, R.J.; Anderson, R.M.; Johnson, S.C.; Kastman, E.K.; Kosmatka, K.J.; Beasley, T.M.; Allison, D.B.; Cruzen, C.; Simmons, H.A.; Kemnitz, J.W.; et al. Caloric restriction delays disease onset and mortality in rhesus monkeys. Science 2009, 325, 201–204. [Google Scholar] [CrossRef] [PubMed]
- Fontana, L.; Partridge, L. Promoting health and longevity through diet: From model organisms to humans. Cell 2015, 161, 106–118. [Google Scholar] [CrossRef] [PubMed]
- Gourd, E. Ultra-processed foods might increase cancer risk. Lancet Oncol. 2018, 19, e186. [Google Scholar] [CrossRef]
- Monteiro, C.A.; Cannon, G.; Lawrence, M.; Costa Louzada, M.D.; Pereira Machado, P. Ultra-Processed Foods, Diet Quality, and Health Using the NOVA Classification System; FAO: Rome, Italy, 2019. [Google Scholar]
- Monteiro, C.A.; Cannon, G.; Levy, R.; Moubarac, J.-C.; Jaime, P.; Martins, A.P.; Canella, D.; Louzada, M.; Parra, D. NOVA. The star shines bright. World Nutr. 2016, 7, 28–38. [Google Scholar]
- Fiolet, T.; Srour, B.; Sellem, L.; Kesse-Guyot, E.; Alles, B.; Mejean, C.; Deschasaux, M.; Fassier, P.; Latino-Martel, P.; Beslay, M.; et al. Consumption of ultra-processed foods and cancer risk: Results from NutriNet-Sante prospective cohort. BMJ 2018, 360, k322. [Google Scholar] [CrossRef]
- da Costa, J.P.; Vitorino, R.; Silva, G.M.; Vogel, C.; Duarte, A.C.; Rocha-Santos, T. A synopsis on aging-Theories, mechanisms and future prospects. Ageing Res. Rev. 2016, 29, 90–112. [Google Scholar] [CrossRef]
- Jalel, A.; Soumaya, G.S.; Hamdaoui, M.H. Vitiligo treatment with vitamins, minerals and polyphenol supplementation. Indian J. Dermatol. 2009, 54, 357–360. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Jeong, I.H.; Kim, C.S.; Lee, Y.M.; Kim, J.M.; Kim, J.S. Chlorogenic acid inhibits the formation of advanced glycation end products and associated protein cross-linking. Arch. Pharm. Res. 2011, 34, 495–500. [Google Scholar] [CrossRef]
- Najjar, F.M.; Taghavi, F.; Ghadari, R.; Sheibani, N.; Moosavi-Movahedi, A.A. Destructive effect of non-enzymatic glycation on catalase and remediation via curcumin. Arch. Biochem. Biophys. 2017, 630, 81–90. [Google Scholar] [CrossRef]
- Matsumaru, D.; Motohashi, H. The KEAP1-NRF2 system in healthy aging and longevity. Antioxidants 2021, 10, 1929. [Google Scholar] [CrossRef] [PubMed]
- Tapia, P.C. Sublethal mitochondrial stress with an attendant stoichiometric augmentation of reactive oxygen species may precipitate many of the beneficial alterations in cellular physiology produced by caloric restriction, intermittent fasting, exercise and dietary phytonutrients: “Mitohormesis” for health and vitality. Med. Hypotheses 2006, 66, 832–843. [Google Scholar] [PubMed]
- Miyazawa, T.; Burdeos, G.C.; Itaya, M.; Nakagawa, K.; Miyazawa, T. Vitamin E: Regulatory redox interactions. IUBMB Life 2019, 71, 430–441. [Google Scholar] [CrossRef] [PubMed]
- Ristow, M.; Schmeisser, K. Mitohormesis: Promoting health and lifespan by increased levels of reactive oxygen species (ROS). Dose Response 2014, 12, 288–341. [Google Scholar] [CrossRef] [PubMed]
- Santos, M.A.; Franco, F.N.; Caldeira, C.A.; de Araujo, G.R.; Vieira, A.; Chaves, M.M.; Lara, R.C. Antioxidant effect of resveratrol: Change in MAPK cell signaling pathway during the aging process. Arch. Gerontol. Geriatr. 2021, 92, 104266. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Dong, L.; Chen, X.; Ding, C.; Hao, M.; Peng, X.; Zhang, Y.; Zhu, H.; Liu, W. Anti-aging effect of phlorizin on D-galactose–induced aging in mice through antioxidant and anti-inflammatory activity, prevention of apoptosis, and regulation of the gut microbiota. Exp. Gerontol. 2022, 163, 111769. [Google Scholar] [CrossRef] [PubMed]
- Tian, Y.; Wen, Z.; Lei, L.; Li, F.; Zhao, J.; Zhi, Q.; Li, F.; Yin, R.; Ming, J. Coreopsis tinctoria flowers extract ameliorates D-galactose induced aging in mice via regulation of Sirt1-Nrf2 signaling pathway. J. Funct. Foods. 2019, 60, 103464. [Google Scholar] [CrossRef]
- Wang, L.; Lee, W.; Cui, Y.R.; Ahn, G.; Jeon, Y.-J. Protective effect of green tea catechin against urban fine dust particle-induced skin aging by regulation of NF-κB, AP-1, and MAPKs signaling pathways. Environ. Pollut. 2019, 252, 1318–1324. [Google Scholar] [CrossRef]
- Wang, Y.; Xiong, Y.; Zhang, A.; Zhao, N.; Zhang, J.; Zhao, D.; Yu, Z.; Xu, N.; Yin, Y.; Luan, X.; et al. Oligosaccharide attenuates aging-related liver dysfunction by activating Nrf2 antioxidant signaling. Food Sci. Nutr. 2020, 8, 3872–3881. [Google Scholar] [CrossRef]
- Maleki, M.; Khelghati, N.; Alemi, F.; Bazdar, M.; Asemi, Z.; Majidinia, M.; Sadeghpoor, A.; Mahmoodpoor, A.; Jadidi-Niaragh, F.; Targhazeh, N.; et al. Stabilization of telomere by the antioxidant property of polyphenols: Anti-aging potential. Life Sci. 2020, 259, 118341. [Google Scholar] [CrossRef] [PubMed]
- Luo, J.; Si, H.; Jia, Z.; Liu, D. Dietary anti-aging polyphenols and potential mechanisms. Antioxidants 2021, 10, 283. [Google Scholar] [CrossRef]
- Ozawa, H.; Miyazawa, T.; Miyazawa, T. Effects of dietary food components on cognitive functions in older adults. Nutrients 2021, 13, 2804. [Google Scholar] [CrossRef] [PubMed]
- Lorenzon dos Santos, J.; Schaan de Quadros, A.; Weschenfelder, C.; Bueno Garofallo, S.; Marcadenti, A. Oxidative stress biomarkers, nut-related antioxidants, and cardiovascular disease. Nutrients 2020, 12, 682. [Google Scholar] [CrossRef] [PubMed]
- The Dictionary by Merriam-Webster. Available online: https://www.merriam-webster.com/dictionary/vitamin (accessed on 27 January 2022).
- Cambridge Dictionary. Available online: https://dictionary.cambridge.org/dictionary/english/vitamin (accessed on 27 January 2022).
- Meyers, D.G.; Maloley, P.A.; Weeks, D. Safety of Antioxidant Vitamins. Arch. Intern. Med. 1996, 156, 925–935. [Google Scholar] [CrossRef] [PubMed]
- Abe, C.; Higuchi, O.; Matsumoto, A.; Miyazawa, T. Determination of intracellular ascorbic acid using tandem mass spectrometry. Analyst 2022, 147, 2640–2643. [Google Scholar] [CrossRef] [PubMed]
- Miyazawa, T.; Matsumoto, A.; Miyahara, Y. Determination of cellular vitamin C dynamics by HPLC-DAD. Analyst 2019, 144, 3483–3487. [Google Scholar] [CrossRef] [PubMed]
- Lykkesfeldt, J.; Tveden-Nyborg, P. The pharmacokinetics of vitamin C. Nutrients 2019, 11, 2412. [Google Scholar] [CrossRef]
- Lewis, L.N.; Hayhoe, R.P.G.; Mulligan, A.A.; Luben, R.N.; Khaw, K.T.; Welch, A.A. Lower dietary and circulating vitamin C in middle-and older-aged men and women are associated with lower estimated skeletal muscle mass. J. Nutr. 2020, 150, 2789–2798. [Google Scholar] [CrossRef]
- Qu, Y.N.; Zhang, L.; Wang, T.; Zhang, H.Y.; Yang, Z.J.; Yuan, F.F.; Wang, Y.; Li, S.W.; Jiang, X.X.; Xie, X.H. Vitamin C Treatment rescues prelamin A-induced premature senescence of subchondral bone mesenchymal stem cells. Stem Cells Int. 2020, 2020, 3150716. [Google Scholar] [CrossRef]
- Aumailley, L.; Warren, A.; Garand, C.; Dubois, M.J.; Paquet, E.R.; Le Couteur, D.G.; Marette, A.; Cogger, V.C.; Lebel, M. Vitamin C modulates the metabolic and cytokine profiles, alleviates hepatic endoplasmic reticulum stress, and increases the life span of Gulo−/− mice. Aging 2016, 8, 458–483. [Google Scholar] [CrossRef]
- Dixit, S.; Bernardo, A.; Walker, J.M.; Kennard, J.A.; Kim, G.Y.; Kessler, E.S.; Harrison, F.E. Vitamin C deficiency in the brain impairs cognition, increases amyloid accumulation and deposition, and oxidative stress in APP/PSEN1 and normally-aging mice. ACS Chem. Neurosci. 2015, 6, 570–581. [Google Scholar] [CrossRef]
- Kaźmierczak-Barańska, J.; Boguszewska, K.; Adamus-Grabicka, A.; Karwowski, B.T. Two faces of vitamin C—Antioxidative and pro-oxidative agent. Nutrients 2020, 12, 1501. [Google Scholar] [CrossRef]
- Azzi, A. Tocopherols, tocotrienols and tocomonoenols: Many similar molecules but only one vitamin E. Redox. Biol. 2019, 26, 101259. [Google Scholar] [CrossRef]
- Lai, G.Y.; Weinstein, S.J.; Taylor, P.R.; McGlynn, K.A.; Virtamo, J.; Gail, M.H.; Albanes, D.; Freedman, N.D. Effects of alpha-tocopherol and beta-carotene supplementation on liver cancer incidence and chronic liver disease mortality in the ATBC study. Br. J. Cancer 2014, 111, 2220–2223. [Google Scholar] [CrossRef]
- Huang, J.; Weinstein, S.J.; Yu, K.; Mannisto, S.; Albanes, D. Relationship between serum alpha-tocopherol and overall and cause-specific mortality: A 30-year prospective cohort analysis. Circ. Res. 2019, 125, 29–40. [Google Scholar] [CrossRef] [PubMed]
- Mangialasche, F.; Xu, W.; Kivipelto, M.; Costanzi, E.; Ercolani, S.; Pigliautile, M.; Cecchetti, R.; Baglioni, M.; Simmons, A.; Soininen, H.; et al. Tocopherols and tocotrienols plasma levels are associated with cognitive impairment. Neurobiol. Aging 2012, 33, 2282–2290. [Google Scholar] [CrossRef]
- Guan, J.Z.; Guan, W.P.; Maeda, T.; Makino, N. Effect of vitamin E administration on the elevated oxygen stress and the telomeric and subtelomeric status in Alzheimer’s disease. Gerontology 2012, 58, 62–69. [Google Scholar] [CrossRef] [PubMed]
- Tucker, L.A. Alpha-and gamma-tocopherol and telomere length in 5768 US men and women: A NHANES study. Nutrients 2017, 9, 601. [Google Scholar] [CrossRef]
- Hanson, C.; Lyden, E.; Furtado, J.; Campos, H.; Sparrow, D.; Vokonas, P.; Litonjua, A.A. Serum tocopherol levels and vitamin E intake are associated with lung function in the normative aging study. Clin. Nutr. 2016, 35, 169–174. [Google Scholar] [CrossRef]
- Boccardi, V.; Arosio, B.; Cari, L.; Bastiani, P.; Scamosci, M.; Casati, M.; Ferri, E.; Bertagnoli, L.; Ciccone, S.; Rossi, P.D.; et al. Beta-carotene, telomerase activity and Alzheimer’s disease in old age subjects. Eur. J. Nutr. 2020, 59, 119–126. [Google Scholar] [CrossRef]
- Huang, J.; Weinstein, S.J.; Yu, K.; Mannisto, S.; Albanes, D. Serum beta carotene and overall and cause-specific mortality: A prospective cohort study. Circ. Res. 2018, 123, 1339–1349. [Google Scholar] [CrossRef]
- Min, K.B.; Min, J.Y. Association between leukocyte telomere length and serum carotenoid in US adults. Eur. J. Nutr. 2017, 56, 1045–1052. [Google Scholar] [CrossRef]
- Yazaki, K.; Yoshikoshi, C.; Oshiro, S.; Yanase, S. Supplemental cellular protection by a carotenoid extends lifespan via Ins/IGF-1 signaling in Caenorhabditis elegans. Oxid. Med. Cell Longev. 2011, 2011, 596240. [Google Scholar] [CrossRef]
- Wu, W.; Wang, X.; Xiang, Q.; Meng, X.; Peng, Y.; Du, N.; Liu, Z.; Sun, Q.; Wang, C.; Liu, X. Astaxanthin alleviates brain aging in rats by attenuating oxidative stress and increasing BDNF levels. Food Funct. 2014, 5, 158–166. [Google Scholar] [CrossRef] [PubMed]
- El-Far, A.H.; Lebda, M.A.; Noreldin, A.E.; Atta, M.S.; Elewa, Y.H.A.; Elfeky, M.; Mousa, S.A. Quercetin attenuates pancreatic and renal D-galactose-induced aging-related oxidative alterations in rats. Int. J. Mol. Sci. 2020, 21, 4348. [Google Scholar] [CrossRef]
- Wang, H.; Jo, Y.-J.; Oh, J.S.; Kim, N.-H. Quercetin delays postovulatory aging of mouse oocytes by regulating SIRT expression and MPF activity. Oncotarget 2017, 8, 38631–38641. [Google Scholar] [CrossRef]
- Geng, L.; Liu, Z.; Zhang, W.; Li, W.; Wu, Z.; Wang, W.; Ren, R.; Su, Y.; Wang, P.; Sun, L.; et al. Chemical screen identifies a geroprotective role of quercetin in premature aging. Protein Cell 2019, 10, 417–435. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Zhao, R.; Zhao, H.; Chen, G.; Jiang, Y.; Lyu, X.; Wu, T. Reduction of aging-induced oxidative stress and activation of autophagy by bilberry anthocyanin supplementation via the AMPK-mTOR signaling pathway in aged female rats. J. Agric. Food Chem. 2019, 67, 7832–7843. [Google Scholar] [CrossRef]
- Miyazawa, T.; Nakagawa, K.; Kudo, M.; Muraishi, K.; Someya, K. Direct intestinal absorption of red fruit anthocyanins, cyanidin-3-glucoside and cyanidin-3,5-diglucoside, into rats and humans. J. Agric. Food Chem. 1999, 47, 1083–1091. [Google Scholar] [CrossRef]
- de Ferrars, R.M.; Czank, C.; Zhang, Q.; Botting, N.P.; Kroon, P.A.; Cassidy, A.; Kay, C.D. The pharmacokinetics of anthocyanins and their metabolites in humans. Br. J. Pharmacol. 2014, 171, 3268–3282. [Google Scholar] [CrossRef]
- Alonso, A.; Gonzalez-Pardo, H.; Garrido, P.; Conejo, N.M.; Llaneza, P.; Diaz, F.; Rey, C.G.D.; Gonzalez, C. Acute effects of 17 β-estradiol and genistein on insulin sensitivity and spatial memory in aged ovariectomized female rats. Age 2010, 32, 421–434. [Google Scholar] [CrossRef]
- Li, Y.; Ren, X.; Lio, C.; Sun, W.; Lai, K.; Liu, Y.; Zhang, Z.; Liang, J.; Zhou, H.; Liu, L.; et al. A chlorogenic acid-phospholipid complex ameliorates post-myocardial infarction inflammatory response mediated by mitochondrial reactive oxygen species in SAMP8 mice. Pharmacol. Res. 2018, 130, 110–122. [Google Scholar] [CrossRef]
- Gines, C.; Cuesta, S.; Kireev, R.; Garcia, C.; Rancan, L.; Paredes, S.D.; Vara, E.; Tresguerres, J.A.F. Protective effect of resveratrol against inflammation, oxidative stress and apoptosis in pancreas of aged SAMP8 mice. Exp. Gerontol. 2017, 90, 61–70. [Google Scholar] [CrossRef]
- Porquet, D.; Casadesus, G.; Bayod, S.; Vicente, A.; Canudas, A.M.; Vilaplana, J.; Pelegri, C.; Sanfeliu, C.; Camins, A.; Pallas, M.; et al. Dietary resveratrol prevents Alzheimer’s markers and increases life span in SAMP8. Age 2013, 35, 1851–1865. [Google Scholar] [CrossRef]
- Navarro-Cruz, A.R.; Ramirez, Y.; Ayala, R.; Ochoa-Velasco, C.; Brambila, E.; Avila-Sosa, R.; Perez-Fernandez, S.; Morales-Medina, J.C.; Aguilar-Alonso, P. Effect of chronic administration of resveratrol on cognitive performance during aging process in rats. Oxid. Med. Cell Longev. 2017, 2017, 8510761. [Google Scholar] [CrossRef] [PubMed]
- Caldeira, C.A.; Santos, M.A.; Araujo, G.R.; Lara, R.C.; Franco, F.N.; Chaves, M.M. Resveratrol: Change of SIRT 1 and AMPK signaling pattern during the aging process. Exp. Gerontol. 2021, 146, 111226. [Google Scholar] [CrossRef]
- Semba, R.D.; Ferrucci, L.; Bartali, B.; Urpi-Sarda, M.; Zamora-Ros, R.; Sun, K.; Cherubini, A.; Bandinelli, S.; Andres-Lacueva, C. Resveratrol levels and all-cause mortality in older community-dwelling adults. JAMA Intern. Med. 2014, 174, 1077–1084. [Google Scholar] [CrossRef] [PubMed]
- Gliemann, L.; Nyberg, M.; Hellsten, Y. Effects of exercise training and resveratrol on vascular health in aging. Free Radic. Biol. Med. 2016, 98, 165–176. [Google Scholar] [CrossRef]
- Shen, L.R.; Xiao, F.; Yuan, P.; Chen, Y.; Gao, Q.K.; Parnell, L.D.; Meydani, M.; Ordovas, J.M.; Li, D.; Lai, C.Q. Curcumin-supplemented diets increase superoxide dismutase activity and mean lifespan in Drosophila. Age 2013, 35, 1133–1142. [Google Scholar] [CrossRef]
- Miyazawa, T.; Nakagawa, K.; Kim, S.H.; Thomas, M.J.; Paul, L.; Zingg, J.-M.; Dolnikowski, G.G.; Roberts, S.B.; Kimura, F.; Miyazawa, T.; et al. Curcumin and piperine supplementation of obese mice under caloric restriction modulates body fat and interleukin-1β. Nutr. Metab. 2018, 15, 12. [Google Scholar] [CrossRef]
- Shailaja, M.; Gowda, K.M.D.; Vishakh, K.; Kumari, N.S. Anti-aging role of curcumin by modulating the inflammatory markers in albino wistar rats. J. Natl. Med. Assoc. 2017, 109, 9–13. [Google Scholar] [CrossRef]
- Sun, Y.; Hu, X.; Hu, G.; Xu, C.; Jiang, H. Curcumin attenuates hydrogen peroxide-induced premature senescence via the activation of SIRT1 in human umbilical vein endothelial cells. Biol. Pharm. Bull. 2015, 38, 1134–1141. [Google Scholar] [CrossRef]
- Hasani, A.S.; Hamid, N.; Amin, A.M.; Fatemeh, E.; Allahbakhshian, F.M.; Ghaffari, N.M. The antioxidant curcumin postpones ovarian aging in young and middle-aged mice. Reprod. Fertil. Dev. 2020, 32, 292–303. [Google Scholar]
- Hagl, S.; Kocher, A.; Schiborr, C.; Kolesova, N.; Frank, J.; Eckert, G.P. Curcumin micelles improve mitochondrial function in neuronal PC12 cells and brains of NMRI mice—Impact on bioavailability. Neurochem. Int. 2015, 89, 234–242. [Google Scholar] [CrossRef] [PubMed]
- Sarker, M.R.; Franks, S.; Sumien, N.; Thangthaeng, N.; Filipetto, F.; Forster, M. Curcumin mimics the neurocognitive and anti-inflammatory effects of caloric restriction in a mouse model of midlife obesity. PLoS ONE 2015, 10, e0140431. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.A.; Shuvo, A.A.; Bepari, A.K.; Hasan, A.M.; Shill, M.C.; Hossain, M.; Uddin, M.; Islam, M.R.; Bakshi, M.K.; Hasan, J.; et al. Curcumin improves D-galactose and normal-aging associated memory impairment in mice: In vivo and in silico-based studies. PLoS ONE 2022, 17, e0270123. [Google Scholar] [CrossRef] [PubMed]
- Moore, T.L.; Bowley, B.; Shultz, P.; Calderazzo, S.; Shobin, E.; Killiany, R.J.; Rosene, D.L.; Moss, M.B. Chronic curcumin treatment improves spatial working memory but not recognition memory in middle-aged rhesus monkeys. Geroscience 2017, 39, 571–584. [Google Scholar] [CrossRef]
- Gutierrez-Mariscal, F.M.; Perez-Martinez, P.; Delgado-Lista, J.; Yubero-Serrano, E.M.; Camargo, A.; Delgado-Casado, N.; Cruz-Teno, C.; Santos-Gonzalez, M.; Rodriguez-Cantalejo, F.; Castano, J.P.; et al. Mediterranean diet supplemented with coenzyme Q10 induces postprandial changes in p53 in response to oxidative DNA damage in elderly subjects. Age 2012, 34, 389–403. [Google Scholar] [CrossRef]
- Ben-Meir, A.; Burstein, E.; Borrego-Alvarez, A.; Chong, J.; Wong, E.; Yavorska, T.; Naranian, T.; Chi, M.; Wang, Y.; Bentov, Y.; et al. Coenzyme Q10 restores oocyte mitochondrial function and fertility during reproductive aging. Aging Cell 2015, 14, 887–895. [Google Scholar] [CrossRef]
- Zhang, D.; Yan, B.; Yu, S.; Zhang, C.; Wang, B.; Wang, Y.; Wang, J.; Yuan, Z.; Zhang, L.; Pan, J. Coenzyme Q10 inhibits the aging of mesenchymal stem cells induced by D-galactose through Akt/mTOR signaling. Oxid. Med. Cell Longev. 2015, 2015, 867293. [Google Scholar] [CrossRef]
- Huo, J.; Xu, Z.; Hosoe, K.; Kubo, H.; Miyahara, H.; Dai, J.; Mori, M.; Sawashita, J.; Higuchi, K. Coenzyme Q10 prevents senescence and dysfunction caused by oxidative stress in vascular endothelial cells. Oxid. Med. Cell Longev. 2018, 2018, 3181759. [Google Scholar] [CrossRef]
- Miyazawa, T.; Hiratsuka, Y.; Toda, M.; Hatakeyama, N.; Ozawa, H.; Abe, C.; Cheng, T.Y.; Matsushima, Y.; Miyawaki, Y.; Ashida, K.; et al. Artificial intelligence in food science and nutrition: A narrative review. Nutr. Rev. 2022, nuac033, epub ahead of print. [Google Scholar] [CrossRef]
- Mezgec, S.; Seljak, B.K. NutriNet: A deep learning food and drink image recognition system for dietary assessment. Nutrients 2017, 9, 657. [Google Scholar] [CrossRef] [PubMed]
- Lemay, D.G.; Baldiviez, L.M.; Chin, E.L.; Spearman, S.S.; Cervantes, E.; Woodhouse, L.R.; Keim, N.L.; Stephensen, C.B.; Laugero, K.D. Technician-scored stool consistency spans the full range of the bristol scale in a healthy US population and differs by diet and chronic stress load. J. Nutr. 2021, 151, 1443–1452. [Google Scholar] [CrossRef] [PubMed]
- Miyazawa, T.; Itaya, M.; Burdeos, G.C.; Nakagawa, K.; Miyazawa, T. A critical review of the use of surfactant-coated nanoparticles in nanomedicine and food nanotechnology. Int. J. Nanomed. 2021, 16, 3937–3999. [Google Scholar] [CrossRef] [PubMed]
- Plagg, B.; Zerbe, S. How does the environment affect human ageing? An interdisciplinary review. J. Gerontol. Geriatr. 2020, 61, 53–67. [Google Scholar] [CrossRef]
- Trzeciak, A.R.; Barnes, J.; Ejiogu, N.; Foster, K.; Brant, L.J.; Zonderman, A.B.; Evans, M.K. Age, sex, and race influence single-strand break repair capacity in a human population. Free Radic. Biol. Med. 2008, 45, 1631–1641. [Google Scholar] [CrossRef]
- Cuervo, A.M.; Macian, F. Autophagy and the immune function in aging. Curr. Opin. Immunol. 2014, 29, 97–104. [Google Scholar] [CrossRef]
- Rando, T.A.; Chang, H.Y. Aging, rejuvenation, and epigenetic reprogramming: Resetting the aging clock. Cell 2012, 148, 46–57. [Google Scholar] [CrossRef] [PubMed]
- Dikalov, S.I.; Harrison, D.G. Methods for detection of mitochondrial and cellular reactive oxygen species. Antioxid. Redox Signal. 2014, 20, 372–382. [Google Scholar] [CrossRef]
- Gladyshev, V.N. The free radical theory of aging is dead. Long live the damage theory! Antioxid. Redox Signal. 2014, 20, 727–731. [Google Scholar] [CrossRef]
- Perez, V.I.; Van Remmen, H.; Bokov, A.; Epstein, C.J.; Vijg, J.; Richardson, A. The overexpression of major antioxidant enzymes does not extend the lifespan of mice. Aging Cell 2009, 8, 73–75. [Google Scholar] [CrossRef]
- Perez-Estrada, J.R.; Hernandez-Garcia, D.; Leyva-Castro, F.; Ramos-Leon, J.; Cuevas-Benítez, O.; Diaz-Munoz, M.; Castro-Obregon, S.; Ramirez-Solis, R.; Garcia, C.; Covarrubias, L. Reduced lifespan of mice lacking catalase correlates with altered lipid metabolism without oxidative damage or premature aging. Free Radic. Biol. Med. 2019, 135, 102–115. [Google Scholar] [CrossRef]
- Van Raamsdonk, J.M.; Hekimi, S. Superoxide dismutase is dispensable for normal animal lifespan. Proc. Natl. Acad. Sci. USA 2012, 109, 5785–5790. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).