Adsorption of Fluoride onto Acid-Modified Low-Cost Pyrolusite Ore: Adsorption Characteristics and Efficiencies
Abstract
1. Introduction
2. Method and Methodology
2.1. Materials and Chemicals
2.2. Preparation of Pyrolusite Ore Adsorbent (PA)
2.3. Characterization of PAs
2.4. Adsorption Kinetics and Isotherm of the PAs
3. Results and Discussion
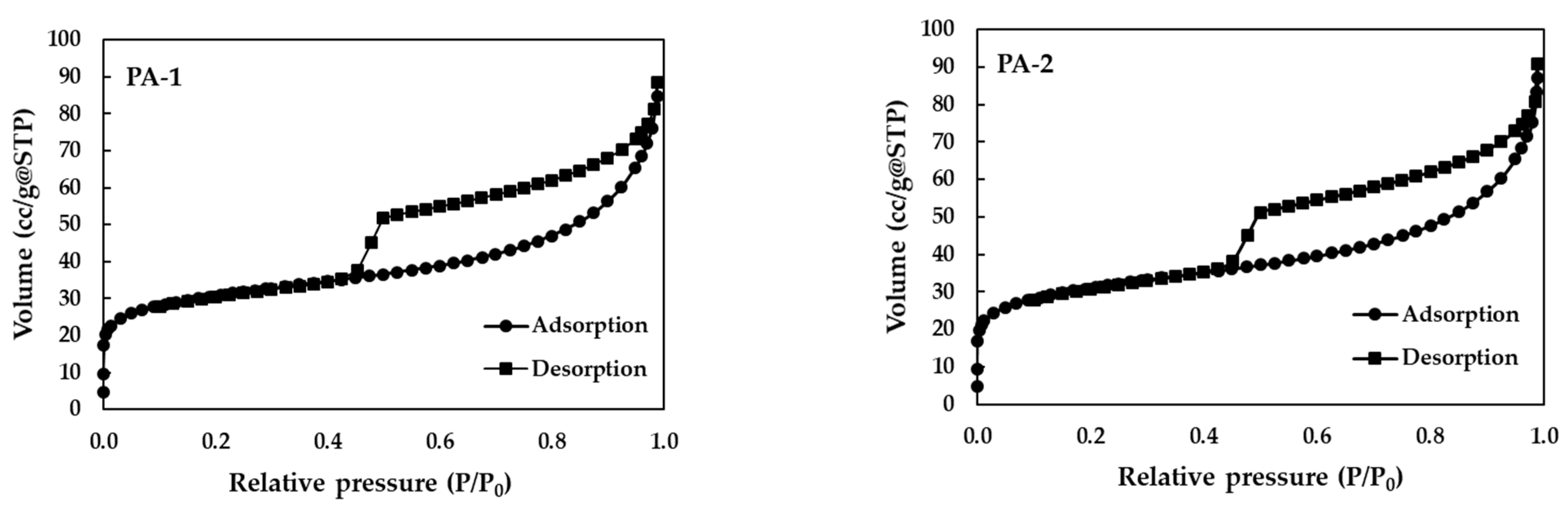
3.1. Physio-Chemical Properties of Materials
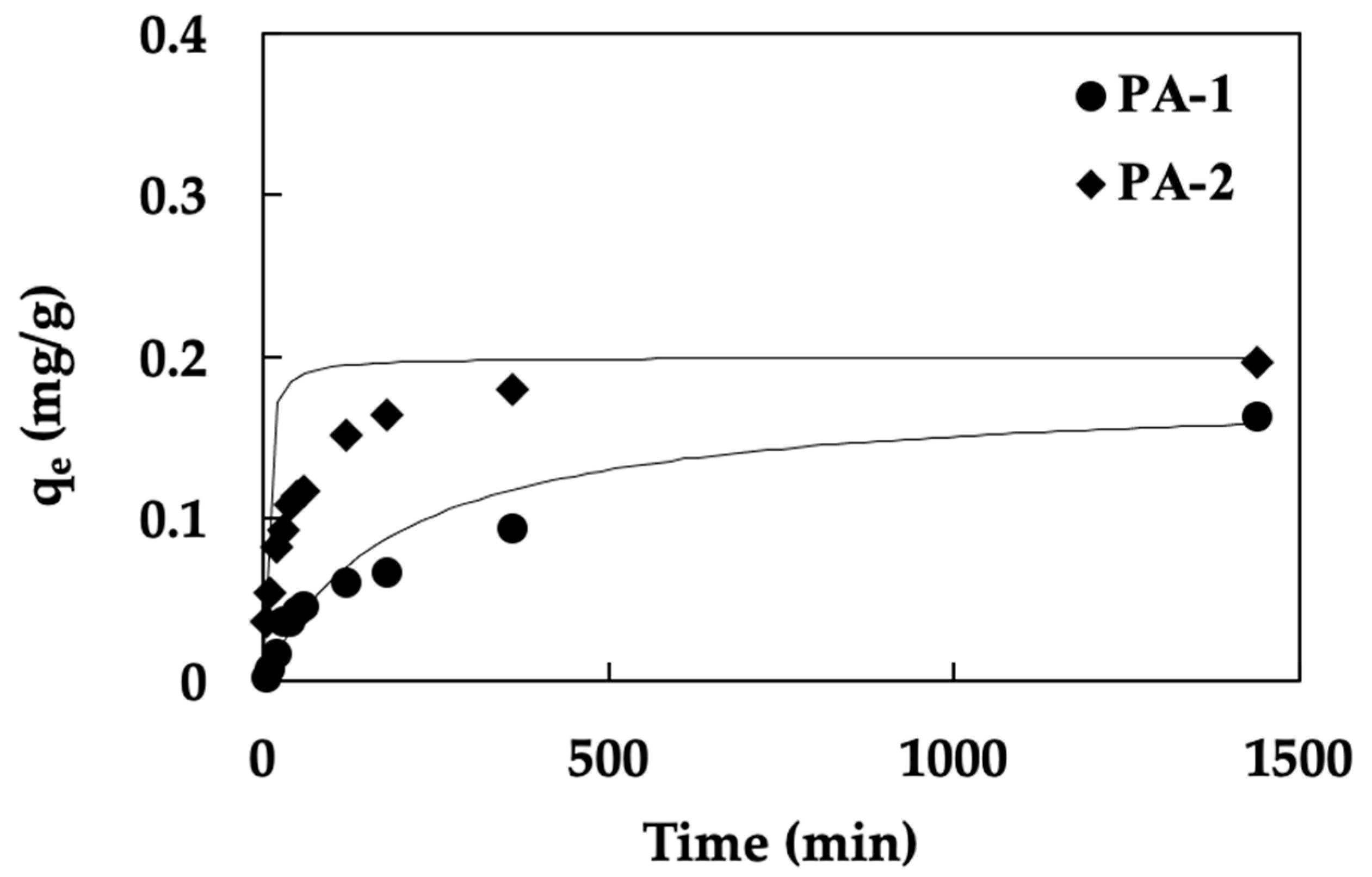
3.2. Adsorption Kinetics Study
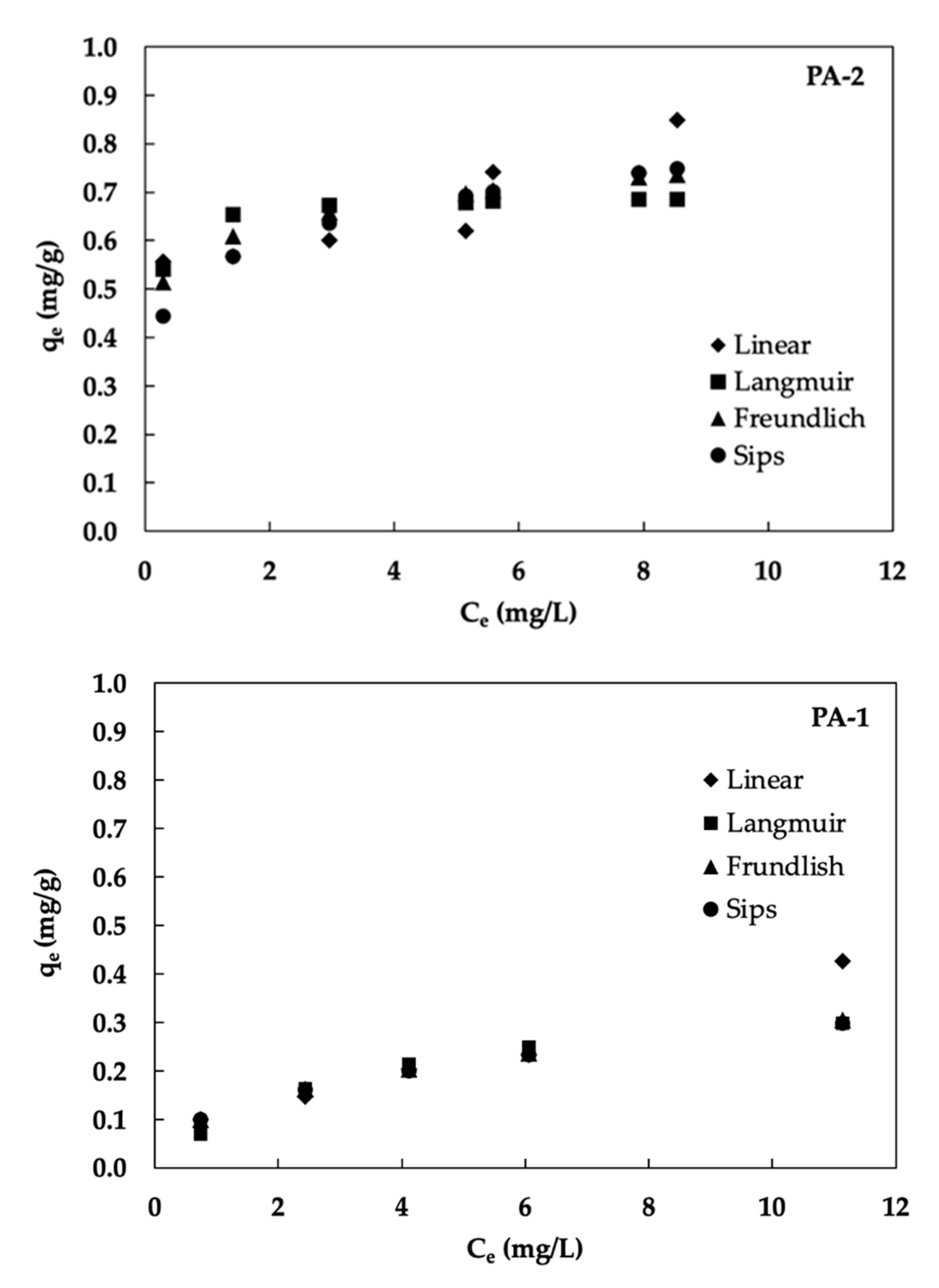
3.3. Adsorption Isotherms
3.4. The Stability of Adsorbents
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jia, H.; Qian, H.; Qu, W.; Zheng, L.; Feng, W.; Ren, W. Fluoride occurrence and human health risk in drinkingwaterwells from southern edge of Chinese loess plateau. Int. J. Environ. Res. Public Health 2019, 16, 1683. [Google Scholar] [CrossRef] [PubMed]
- Chuah, C.J.; Lye, H.R.; Ziegler, A.D.; Wood, S.H.; Kongpun, C.; Rajchagool, S. Fluoride: A naturally-occurring health hazard in drinking-water resources of Northern Thailand. Sci. Total Environ. 2016, 545–546, 266–279. [Google Scholar] [CrossRef] [PubMed]
- Theerawasttanasiri, N.; Taneepanichskul, S.; Pingchai, W.; Nimchareon, Y.; Sriwichai, S. Implementing a geographical information system to assess endemic fluoride areas in Lamphun, Thailand. Risk Manag. Healthc. Policy 2018, 11, 15–24. [Google Scholar] [CrossRef]
- Rojanaworarit, C.; Claudio, L.; Howteerakul, N.; Siramahamongkol, A.; Ngernthong, P.; Kongtip, P.; Woskie, S. Hydrogeogenic fluoride in groundwater and dental fluorosis in Thai agrarian communities: A prevalence survey and case–control study. BMC Oral Health 2021, 21, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Singh, K.; Lataye, D.H.; Wasewar, K.L.; Yoo, C. Removal of fluoride from aqueous solution: Status and techniques. Desalination Water Treat. 2013, 51, 3233–3247. [Google Scholar] [CrossRef]
- World Health Organization. Fluorides and Oral Health. Report of a WHO Expert Committee on Oral Health Status and Flouride Use; World Health Organization: Geneva, Switzerland, 1994; Volume 846, pp. 1–37. [Google Scholar]
- Amor, Z.; Bariou, B.; Mameri, N.; Taky, M.; Nicolas, S.; Elmidaoui, A. Fluoride removal from brackish water by electrodialysis. Desalination 2001, 133, 215–223. [Google Scholar] [CrossRef]
- Menkouchi Sahli, M.A.; Annouar, S.; Tahaikt, M.; Mountadar, M.; Soufiane, A.; Elmidaoui, A. Fluoride removal for underground brackish water by adsorption on the natural chitosan and by electrodialysis. Desalination 2007, 212, 37–45. [Google Scholar] [CrossRef]
- Mumtaz, N.; Pandey, G.; Labhasetwar, P.K. Global fluoride occurrence, available technologies for fluoride removal, and electrolytic defluoridation: A review. Crit. Rev. Environ. Sci. Technol. 2015, 45, 2357–2389. [Google Scholar] [CrossRef]
- Sehn, P. Fluoride removal with extra low energy reverse osmosis membranes: Three years of large scale field experience in Finland. Desalination 2008, 223, 73–84. [Google Scholar] [CrossRef]
- Chantharawong, P.; Wongrueng, A.; Rakruam, P.; Wattanachira, S.; Takizawa, S. Effects of Activated Carbon and Cationic Exchange Resin Pretreatments on Groundwater Defluoridation by Reverse Osmosis Process. Eng. J. 2017, 21, 123–132. [Google Scholar] [CrossRef][Green Version]
- Ma, W.; Ya, F.; Wang, R.; Zhao, Y. Fluoride Removal from Drinking Water by Adsorption Using Bone Char as a Biosorbent. Int. J. Environ. Technol. Manag. 2008, 9, 59–69. [Google Scholar] [CrossRef]
- Saini, A.; Maheshwari, P.H.; Tripathy, S.S.; Waseem, S.; Gupta, A.; Dhakate, S.R. A novel alum impregnated CaO/carbon composite for de-fluoridation of water. Groundw. Sustain. Dev. 2021, 14, 100622. [Google Scholar] [CrossRef]
- Ravulapalli, S.; Ravindhranath, K. Defluoridation studies using active carbon derived from the barks of Ficus racemosa plant. J. Fluor. Chem. 2017, 193, 58–66. [Google Scholar] [CrossRef]
- Wongrueng, A.; Sookwong, B.; Rakruam, P.; Wattanachira, S. Kinetic adsorption of fluoride from an aqueous solution onto a dolomite sorbent. Eng. J. 2016, 20, 1–9. [Google Scholar] [CrossRef]
- Sawangjang, B.; Induvesa, P.; Wongrueng, A.; Pumas, C.; Wattanachira, S.; Rakruam, P.; Punyapalakul, P.; Takizawa, S.; Khan, E. Evaluation of fluoride adsorption mechanism and capacity of different types of bone char. Int. J. Environ. Res. Public Health 2021, 18, 6878. [Google Scholar] [CrossRef]
- Rashid, R.; Shafiq, I.; Akhter, P.; Iqbal, M.J.; Hussain, M. A state-of-the-art review on wastewater treatment techniques: The effectiveness of adsorption method. Environ. Sci. Pollut. Res. 2021, 28, 9050–9066. [Google Scholar] [CrossRef]
- Aigbe, U.O.; Onyancha, R.B.; Ukhurebor, K.E.; Obodo, K.O. Removal of fluoride ions using a polypyrrole magnetic nanocomposite influenced by a rotating magnetic field. RSC Adv. 2019, 10, 595–609. [Google Scholar] [CrossRef]
- Yang, B.; Sun, G.; Quan, B.; Tang, J.; Zhang, C.; Jia, C.; Tang, Y.; Wang, X.; Zhao, M.; Wang, W.; et al. An experimental study of fluoride removal from wastewater by mn-ti modified zeolite. Water 2021, 13, 3343. [Google Scholar] [CrossRef]
- Hu, J.; Wu, D.; Rao, R.; Liu, R.; Lai, W. Adsorption kinetics of fluoride on bone char and its regeneration. Environ. Prot. Eng. 2021, 43, 93–112. [Google Scholar] [CrossRef]
- Medellin-Castillo, N.A.; Leyva-Ramos, R.; Padilla-Ortega, E.; Perez, R.O.; Flores-Cano, J.V.; Berber-Mendoza, M.S. Adsorption capacity of bone char for removing fluoride from water solution. Role of hydroxyapatite content, adsorption mechanism and competing anions. J. Ind. Eng. Chem. 2014, 20, 4014–4021. [Google Scholar] [CrossRef]
- Nijhawan, A.; Butler, E.C.; Sabatini, D.A. Fluoride Adsorption on Porous Hydroxyapatite Ceramic Filters: A Study of Kinetics. Environ. Eng. Sci. 2020, 37, 409–416. [Google Scholar] [CrossRef]
- Chen, N.; Zhang, Z.; Feng, C.; Sugiura, N.; Li, M.; Chen, R. Fluoride Removal from Water by Granular Ceramic Adsorption. J. Colloid Interface Sci. 2010, 348, 579–584. [Google Scholar] [CrossRef] [PubMed]
- Kebede, B.; Beyene, A.; Fufa, F.; Megersa, M.; Behm, M. Experimental evaluation of sorptive removal of fluoride from drinking water using iron ore. Appl. Water Sci. 2016, 6, 57–65. [Google Scholar] [CrossRef]
- Huang, L.; Luo, Z.; Huang, X.; Wang, Y.; Yan, J.; Liu, W.; Guo, Y.; Babu Arulmani, S.R.; Shao, M.; Zhang, H. Applications of biomass-based materials to remove fluoride from wastewater: A review. Chemosphere 2022, 301, 134679. [Google Scholar] [CrossRef] [PubMed]
- Das, A.P.; Sukla, L.B.; Pradhan, N.; Nayak, S. Manganese biomining: A review. Bioresour. Technol. 2011, 102, 7381–7387. [Google Scholar] [CrossRef]
- Demirkıran, N.; Turhan Özdemir, G.; Saraç, M.; Dardağan, M. Adsorption of methylene blue from aqueous solutions by pyrolusite ore. Mong. J. Chem. 2018, 18, 5. [Google Scholar] [CrossRef]
- Thi Hue, N.; Hoang Tung, N. Study about Doping Ion La3+ onto Surface of Pyrolusite Ore for Removing Simultaneously Both Fluoride and Phosphate from Wastewater. J. Chem. 2017, 2017, 1–11. [Google Scholar] [CrossRef]
- Wan, S.; Lin, J.; Tao, W.; Yang, Y.; Li, Y.; He, F. Enhanced Fluoride Removal from Water by Nanoporous Biochar-Supported Magnesium Oxide. Ind. Eng. Chem. Res. 2019, 58, 9988–9996. [Google Scholar] [CrossRef]
- Lee, S.G.; Ha, J.W.; Sohn, E.H.; Park, I.J.; Lee, S.B. Synthesis of pillar and microsphere-like magnesium oxide particles and their fluoride adsorption performance in aqueous solutions. Korean J. Chem. Eng. 2017, 34, 2738–2747. [Google Scholar] [CrossRef]
- Lee, J.-H.; Ham, J.-Y. Synthesis of manganese oxide particles in supercritical water. Korean J. Chem. Eng. 2006, 23, 714–719. [Google Scholar] [CrossRef]
- Safari, G.H.; Zarrabi, M.; Hoseini, M.; Kamani, H.; Jaafari, J.; Mahvi, A.H. Trends of natural and acid-engineered pumice onto phosphorus ions in aquatic environment: Adsorbent preparation, characterization, and kinetic and equilibrium modeling. Desalination Water Treat. 2014, 54, 1–13. [Google Scholar] [CrossRef]
- Gatabi, M.P.; Moghaddam, H.M.; Ghorbani, M. Point of zero charge of maghemite decorated multiwalled carbon nanotubes fabricated by chemical precipitation method. J. Mol. Liquids 2016, 216, 117–125. [Google Scholar] [CrossRef]
- Abegunde, S.M.; Idowu, K.S.; Adejuwon, O.M.; Adeyemi-Adejolu, T. A review on the influence of chemical modification on the performance of adsorbents. Resour. Environ. Sustain. 2020, 1, 1–9. [Google Scholar] [CrossRef]
- Rehman, A.; Park, M.; Park, S.J. Current progress on the surface chemical modification of carbonaceous materials. Coatings 2019, 9, 103. [Google Scholar] [CrossRef]
- Thorat, G.M.; Jadhav, H.S.; Roy, A.; Chung, W.J.; Seo, J.G. Dual Role of Deep Eutectic Solvent as a Solvent and Template for the Synthesis of Octahedral Cobalt Vanadate for an Oxygen Evolution Reaction. ACS Sustain. Chem. Eng. 2018, 6, 16255–16266. [Google Scholar] [CrossRef]
- Coates, J. Interpretation of Infrared Spectra, A Practical Approach. In Encyclopedia of Analytical Chemistry; John Wiley & Sons, Ltd.: Chichester, UK, 2006. [Google Scholar] [CrossRef]
- Ninwiwek, N.; Hongsawat, P.; Punyapalakul, P.; Prarat, P. Removal of the antibiotic sulfamethoxazole from environmental water by mesoporous silica-magnetic graphene oxide nanocomposite technology: Adsorption characteristics, coadsorption and uptake mechanism. Colloids Surf. A Physicochem. Eng. Asp. 2019, 580, 123716. [Google Scholar] [CrossRef]
- Prarat, P.; Hongsawat, P.; Punyapalakul, P. Amino-functionalized mesoporous silica-magnetic graphene oxide nanocomposites as water-dispersible adsorbents for the removal of the oxytetracycline antibiotic from aqueous solutions: Adsorption performance, effects of coexisting ions, and natural organic matter. Environ. Sci. Pollut. Res. 2020, 27, 6560–6576. [Google Scholar] [CrossRef]
- Ryan, S.K.; Radford, R.K. Fluoride removal from water using crushed limestone. Indian J. Chem. Technol. 1987, 17, 120–125. [Google Scholar]
- Induvesa, P.; Ratanatawanate, C.; Wongrueng, A.; Punyapalakul, P. Selective adsorption mechanisms of iodinated trihalomethanes onto thiol-functionalized HKUST-1s in a mixed solute. J. Environ. Manag. 2022, 315, 115099. [Google Scholar] [CrossRef]
- Ihsanullah, I.; Al-Khaldi, F.A.; Abu-Sharkh, B.; Abulkibash, A.M.; Qureshi, M.I.; Laoui, T.; Atieh, M.A. Effect of acid modification on adsorption of hexavalent chromium (Cr(VI)) from aqueous solution by activated carbon and carbon nanotubes. Desalination Water Treat. 2016, 57, 7232–7244. [Google Scholar] [CrossRef]
- Liu, X.; Zhou, F.; Chi, R.; Feng, J.; Ding, Y.; Liu, Q. Preparation of Modified Montmorillonite and Its Application to Rare Earth Adsorption. Minerals 2019, 9, 747. [Google Scholar] [CrossRef]
- Al-Ghouti, M.A.; Da’ana, D.A. Guidelines for the use and interpretation of adsorption isotherm models: A review. J. Hazard. Mater. 2020, 393, 122383. [Google Scholar] [CrossRef] [PubMed]
- Shi, S.; Zhao, K.; Zhao, Q.; Luo, A.; Xie, S.; Feng, J. Fluoride Adsorption Comparison from Aqueous Solutions Using Al-and La-Modified Adsorbent Prepared from Polygonum orientale Linn. Water 2022, 14, 592. [Google Scholar] [CrossRef]

| Types of Modification | Modifier/Solution | Time | Temperature (°C) | |
|---|---|---|---|---|
| Original-PA | PA-1 | Deionized water (DI) | Soaked and washed with DI for 5 day | Room temperature |
| Acid-PA | PA-2 | 0.1 N HCl | Soaked with 0.1 N HCl for 24 h and wash with DI | Room temperature |
| Materials | Elements (% wt) | Total | ||||||
|---|---|---|---|---|---|---|---|---|
| C | N | O | Al | Si | Mn | Fe | ||
| PA-1 | 18.09 | 4.47 | 19.01 | 7.87 | 1.87 | 28.20 | 20.49 | 100 |
| PA-2 | 15.76 | n.d. * | 20.03 | 4.18 | n.d. | 23.77 | 36.26 | 100 |
| Materials | SBET a (m2/g) | Pore Diameter b (nm) | Pore Volume c (cc/g) | pHpzc |
|---|---|---|---|---|
| PA-1 | 111.48 | 3.65 | 0.112 | 4.00 |
| PA-2 | 108.93 | 4.62 | 0.107 | 4.00 |
| Materials | qe,exp (mg/g) | Pseudo-First Order | Pseudo-Second Order | ||||||
|---|---|---|---|---|---|---|---|---|---|
| K1 (1/min) | qe,cal (mg/g) | R2 | k2 (g/mg min) | qe,cal (mg/g) | h (mg/g min) | t 1/2 (min) | R2 | ||
| PA-1 | 0.17 | −0.0015 | 0.44 | 0.7951 | 0.03 | 0.18 | 0.0009 | 185.26 | 0.9575 |
| PA-2 | 0.20 | −0.0088 | 0.42 | 0.9860 | 0.15 | 0.20 | 0.0062 | 32.04 | 0.9995 |
| Materials | Linear Isotherm | Langmuir Isotherm | Freundlich Isotherm | Sip Isotherm | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Kp (L/g) | R2 | qm (mg/g) | KL (L/mg) | R2 | KF (mg/g) | R2 | Ks (L/mg) | R2 | ||||
| PA-1 | 0.03 | 0.8939 | 0.38 | 0.62 | 0.8765 | 0.11 | 0.51 | 0.9488 | 50.07 | 0.002 | 2.47 | 0.9358 |
| PA-2 | 0.03 | 0.8459 | 0.70 | 12.31 | 0.3605 | 0.58 | 0.56 | 0.9899 | 87.08 | 0.006 | 6.44 | 0.8821 |
| Materials | Adsorption Capacity (mg/g) | Operating Condition | References |
|---|---|---|---|
| Polygonum orientale Linn. | 0.39 (at 20 °C) | CInitial = 20 mg/L (pH = 7) Absorbance dose = 2 g/100 mL | [45] |
| Polygonum orientale Linn.—modification with Al2(SO4)3 | 0.77 (at 20 °C) | CInitial = 20 mg/L (pH = 7) Absorbance dose = 2 g/100 mL | [45] |
| Iron ore | 1.45 (at 22 °C) | CInitial = 10 mg/L (pH = 6) Absorbance dose = 0.5 g/100 mL | [24] |
| Dolomite | 0.011 (at room temp) | CInitial = 10 mg/L (pH = 7) Absorbance dose = 10 g/100 mL | [15] |
| Pyrolusite (PA-1) | 0.11 (at room temp) | CInitial = 10 mg/L (pH = 7) Absorbance dose = 1.5 g/100 mL | This study |
| Acid-modified pyrolusite (PA-2) | 0.58 (at room temp) | CInitial = 10 mg/L (pH = 7) Absorbance dose = 1.5 g/100 mL | This study |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Induvesa, P.; Rattanakom, R.; Sriboonnak, S.; Pumas, C.; Duangjan, K.; Rakruam, P.; Nitayavardhana, S.; Sittisom, P.; Wongrueng, A. Adsorption of Fluoride onto Acid-Modified Low-Cost Pyrolusite Ore: Adsorption Characteristics and Efficiencies. Int. J. Environ. Res. Public Health 2022, 19, 17103. https://doi.org/10.3390/ijerph192417103
Induvesa P, Rattanakom R, Sriboonnak S, Pumas C, Duangjan K, Rakruam P, Nitayavardhana S, Sittisom P, Wongrueng A. Adsorption of Fluoride onto Acid-Modified Low-Cost Pyrolusite Ore: Adsorption Characteristics and Efficiencies. International Journal of Environmental Research and Public Health. 2022; 19(24):17103. https://doi.org/10.3390/ijerph192417103
Chicago/Turabian StyleInduvesa, Phacharapol, Radamanee Rattanakom, Sornsiri Sriboonnak, Chayakorn Pumas, Kritsana Duangjan, Pharkphum Rakruam, Saoharit Nitayavardhana, Prattakorn Sittisom, and Aunnop Wongrueng. 2022. "Adsorption of Fluoride onto Acid-Modified Low-Cost Pyrolusite Ore: Adsorption Characteristics and Efficiencies" International Journal of Environmental Research and Public Health 19, no. 24: 17103. https://doi.org/10.3390/ijerph192417103
APA StyleInduvesa, P., Rattanakom, R., Sriboonnak, S., Pumas, C., Duangjan, K., Rakruam, P., Nitayavardhana, S., Sittisom, P., & Wongrueng, A. (2022). Adsorption of Fluoride onto Acid-Modified Low-Cost Pyrolusite Ore: Adsorption Characteristics and Efficiencies. International Journal of Environmental Research and Public Health, 19(24), 17103. https://doi.org/10.3390/ijerph192417103






