The Potential of Exerkines in Women’s COVID-19: A New Idea for a Better and More Accurate Understanding of the Mechanisms behind Physical Exercise
Abstract
Key points
- With the prevalence of COVID-19 in the world as a dangerous disease, many studies have given physical exercise interventions, but the understanding of the effective and basic mechanisms of physical exercise is still unclear.
- The ACE2 receptor that the SARS-CoV-2 spike protein binds to is more expressed in fat tissue than in other organs, which is very high in obese and overweight women. Therefore, behavioral changes of adipokines in COVID-19 lead to high inflammation and, through crosstalk with other organs, such as muscle (myokines) or liver, hepatokines lead to a decrease in physical function and an increase in inflammation.
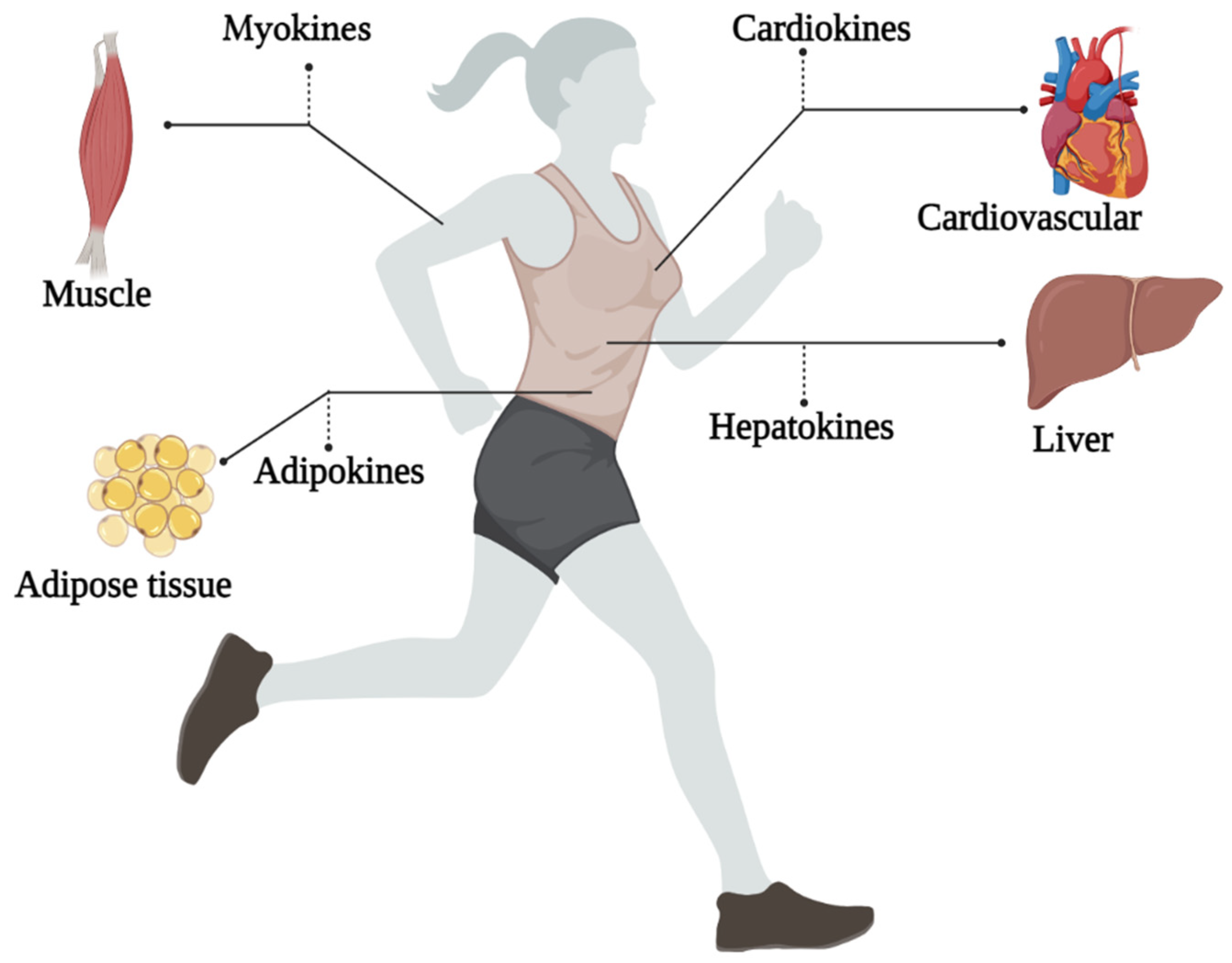
- Exerkines include myokines (muscle), cardiokines (heart), hepatokines (liver), neurokines (nervous system), and baptokines (brown adipose tissue) and adipokines (adipose tissue).
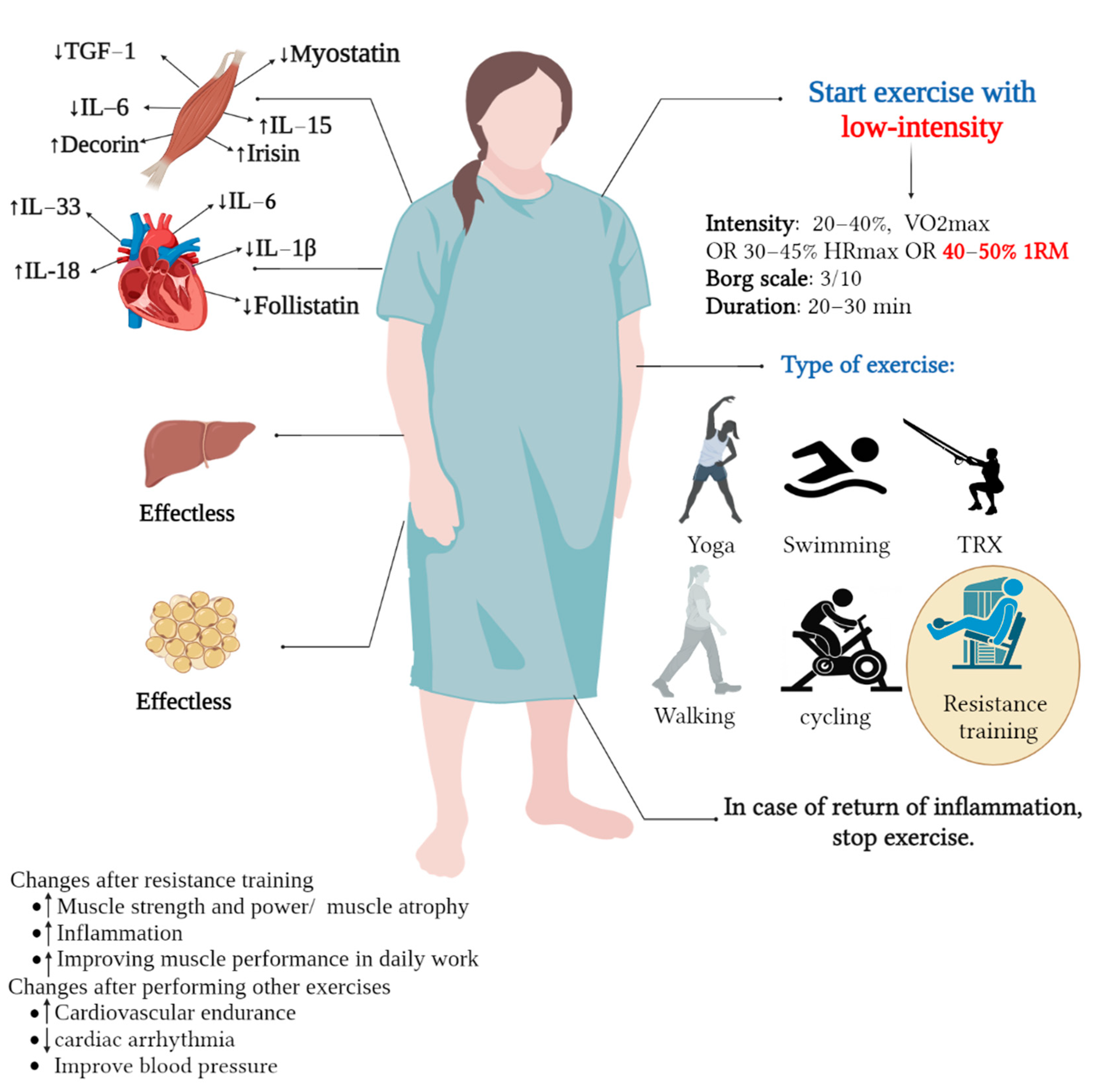
- Resistance training that includes weight training and exercise equipment can improve muscle strength and reduce inflammation by affecting myokines during COVID-19 disease. After discharge from the hospital, this physical exercise is suitable for returning to a quality of life. Moreover, aerobic exercise that includes moderate-intensity exercise (running on a treadmill or cycling) can have the greatest effect on the stimulation of exerkines.
- Exerkines have a high potential for treating diseases, but investigating their high potential for COVID-19 and the underlying mechanisms remained unanswered.
- The emerging research on exerkines opens up several promising avenues for future research where researchers can explore the potential importance of exerkines in elderly or coexisting COVID-19 patients.
- Future studies on exerkines can be very effective in designing accurate physical exercise and more practical recommendations for patients, especially COVID-19 patients, during illness or after discharge from the hospital.
1. Introduction
2. The Potential Role of Exerkines Can Be Effective in COVID-19
3. Can Exerkines (Generated/Stimulated by Physical Activity/Physical Exercise) Help COVID-19 Patients?
Low-Intensity Physical Exercise
4. Can Exerkines (Generated/Stimulated by Physical Activity/Physical Exercise) Help Newly Discharged COVID-19 Patients?
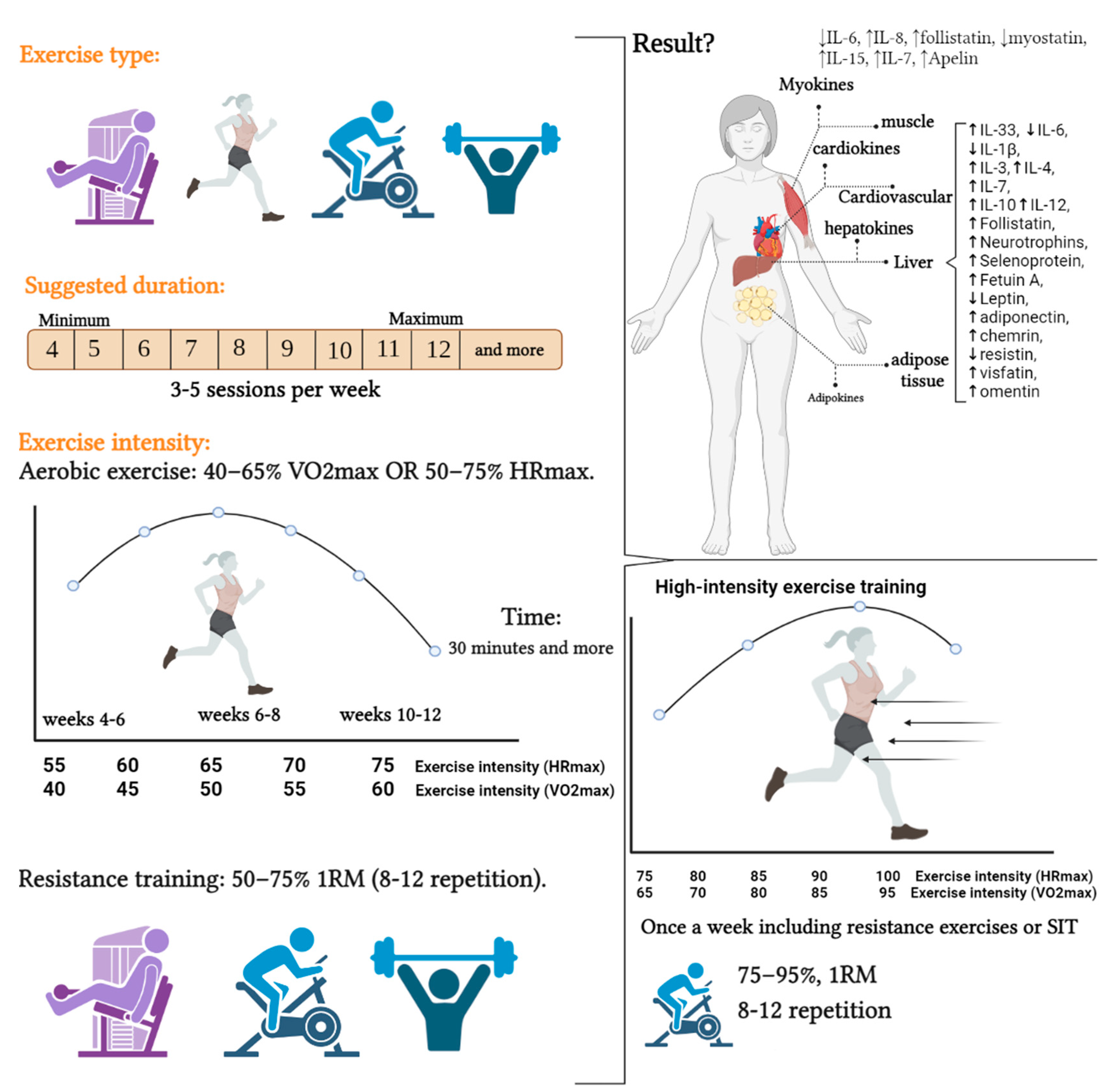
4.1. Moderate-Intensity Physical Exercise
4.2. High-Intensity Physical Exercise
5. Discussion
5.1. Findings on Exerkines and Women COVID-19 Patients
5.2. Findings on Exerkines and Women COVID-19 Patients (after Discharge from the Hospital)
5.3. Conclusions
5.4. Limitations and Prospects for Future Research
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Schett, G.; Manger, B.; Simon, D.; Caporali, R. COVID-19 revisiting inflammatory pathways of arthritis. Nat. Rev. Rheumatol. 2020, 16, 465–470. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Chua, B.Y.; Selva, K.J.; Kedzierski, L.; Ashhurst, T.M.; Haycroft, E.R.; Shoffner-Beck, S.K.; Hensen, L.; Boyd, D.F.; James, F.; et al. SARS-CoV-2 infection results in immune responses in the respiratory tract and peripheral blood that suggest mechanisms of disease severity. Nat. Commun. 2022, 13, 2774. [Google Scholar] [CrossRef] [PubMed]
- Yu, W.; Rohli, K.E.; Yang, S.; Jia, P. Impact of obesity on COVID-19 patients. J. Diabetes Complicat. 2021, 35, 107817. [Google Scholar] [CrossRef] [PubMed]
- Steenblock, C.; Hassanein, M.; Khan, E.G.; Yaman, M.; Kamel, M.; Barbir, M.; Lorke, D.E.; Everett, D.; Bejtullah, S.; Lohmann, T. Obesity and COVID-19: What are the consequences? Horm. Metab. Res. 2022, 54, 496–502. [Google Scholar] [CrossRef] [PubMed]
- Cai, Q.; Chen, F.; Wang, T.; Luo, F.; Liu, X.; Wu, Q.; He, Q.; Wang, Z.; Liu, Y.; Liu, L. Obesity and COVID-19 severity in a designated hospital in Shenzhen, China. Diabetes Care 2020, 43, 1392–1398. [Google Scholar] [CrossRef]
- Soares, M.N.; Eggelbusch, M.; Naddaf, E.; Gerrits, K.H.; van der Schaaf, M.; van den Borst, B.; Wiersinga, W.J.; van Vugt, M.; Weijs, P.J.; Murray, A.J. Skeletal muscle alterations in patients with acute Covid-19 and post-acute sequelae of Covid-19. J. Cachexia Sarcopenia Muscle 2022, 13, 11–22. [Google Scholar] [CrossRef]
- Moro, T.; Paoli, A. When COVID-19 affects muscle: Effects of quarantine in older adults. Eur. J. Transl. Myol. 2020, 30, 9069. [Google Scholar] [CrossRef]
- Di Stefano, V.; Battaglia, G.; Giustino, V.; Gagliardo, A.; D’Aleo, M.; Giannini, O.; Palma, A.; Brighina, F. Significant reduction of physical activity in patients with neuromuscular disease during COVID-19 pandemic: The long-term consequences of quarantine. J. Neurol. 2021, 268, 20–26. [Google Scholar] [CrossRef]
- Scully, E.P.; Haverfield, J.; Ursin, R.L.; Tannenbaum, C.; Klein, S.L. Considering how biological sex impacts immune responses and COVID-19 outcomes. Nat. Rev. Immunol. 2020, 20, 442–447. [Google Scholar] [CrossRef]
- Takahashi, T.; Ellingson, M.K.; Wong, P.; Israelow, B.; Lucas, C.; Klein, J.; Silva, J.; Mao, T.; Oh, J.E.; Tokuyama, M. Sex differences in immune responses that underlie COVID-19 disease outcomes. Nature 2020, 588, 315–320. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, T.; Iwasaki, A. Sex differences in immune responses. Science 2021, 371, 347–348. [Google Scholar] [CrossRef]
- Burki, T. The indirect impact of COVID-19 on women. Lancet Infect. Dis. 2020, 20, 904–905. [Google Scholar] [CrossRef] [PubMed]
- WHO. Gender and COVID-19: Advocacy Brief, 14 May 2020; World Health Organization: Geneva, Switzerland, 2020. [Google Scholar]
- Boniol, M.; McIsaac, M.; Xu, L.; Wuliji, T.; Diallo, K.; Campbell, J. Gender Equity in the Health Workforce: Analysis of 104 Countries; World Health Organization: Geneva, Switzerland, 2019. [Google Scholar]
- Connor, J.; Madhavan, S.; Mokashi, M.; Amanuel, H.; Johnson, N.R.; Pace, L.E.; Bartz, D. Health risks and outcomes that disproportionately affect women during the Covid-19 pandemic: A review. Soc. Sci. Med. 2020, 266, 113364. [Google Scholar] [CrossRef]
- Ahmed, S.B.; Dumanski, S.M. Sex, gender and COVID-19: A call to action. Can. J. Public Health 2020, 111, 980–983. [Google Scholar] [CrossRef]
- United Nations. Policy Brief: The Impact of COVID-19 on Women—9 April 2020. 2020. Available online: https://unsdg.un.org/resources/policy-brief-impact-covid-19-women (accessed on 3 June 2022).
- Rubin, R. As their numbers grow, COVID-19 “long haulers” stump experts. JAMA 2020, 324, 1381–1383. [Google Scholar] [CrossRef] [PubMed]
- Cooper, A.J.; Gupta, S.R.; Moustafa, A.F.; Chao, A.M. Sex/Gender Differences in Obesity Prevalence, Comorbidities, and Treatment. Curr. Obes. Rep. 2021, 10, 458–466. [Google Scholar] [CrossRef] [PubMed]
- Leblanc, V.; Hudon, A.M.; Royer, M.M.; Corneau, L.; Dodin, S.; Bégin, C.; Lemieux, S. Differences between men and women in dietary intakes and metabolic profile in response to a 12-week nutritional intervention promoting the Mediterranean diet. J. Nutr. Sci. 2015, 4, e13. [Google Scholar] [CrossRef]
- Jacobsen, H.; Klein, S.L. Sex Differences in Immunity to Viral Infections. Front. Immunol. 2021, 12, 720952. [Google Scholar] [CrossRef]
- Stelzig, K.E.; Canepa-Escaro, F.; Schiliro, M.; Berdnikovs, S.; Prakash, Y.; Chiarella, S.E. Estrogen regulates the expression of SARS-CoV-2 receptor ACE2 in differentiated airway epithelial cells. Am. J. Physiol. Lung Cell. Mol. Physiol. 2020, 318, L1280–L1281. [Google Scholar] [CrossRef]
- Gupte, M.; Thatcher, S.E.; Boustany-Kari, C.M.; Shoemaker, R.; Yiannikouris, F.; Zhang, X.; Karounos, M.; Cassis, L.A. Angiotensin converting enzyme 2 contributes to sex differences in the development of obesity hypertension in C57BL/6 mice. Arterioscler. Thromb. Vasc. Biol. 2012, 32, 1392–1399. [Google Scholar] [CrossRef]
- Fishman, J.; Boyar, R.M.; Hellman, L. Influence of body weight on estradiol metabolism in young women. J. Clin. Endocrinol. Metab. 1975, 41, 989–991. [Google Scholar] [CrossRef]
- Khoramipour, K.; Basereh, A.; Hekmatikar, A.A.; Castell, L.; Ruhee, R.T.; Suzuki, K. Physical activity and nutrition guidelines to help with the fight against COVID-19. J. Sports Sci. 2021, 39, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Shariat, A.; Cleland, J.A.; Hakakzadeh, A. Home-based exercises during the COVID-19 quarantine situation for office workers: A commentary. Work 2020, 66, 381–382. [Google Scholar] [CrossRef]
- Chow, L.S.; Gerszten, R.E.; Taylor, J.M.; Pedersen, B.K.; van Praag, H.; Trappe, S.; Febbraio, M.A.; Galis, Z.S.; Gao, Y.; Haus, J.M.; et al. Exerkines in health, resilience and disease. Nat. Rev. Endocrinol. 2022, 18, 273–289. [Google Scholar] [CrossRef]
- Suzuki, K. Chronic inflammation as an immunological abnormality and effectiveness of exercise. Biomolecules 2019, 9, 223. [Google Scholar] [CrossRef] [PubMed]
- Piercy, K.L.; Troiano, R.P.; Ballard, R.M.; Carlson, S.A.; Fulton, J.E.; Galuska, D.A.; George, S.M.; Olson, R.D. The physical activity guidelines for Americans. JAMA 2018, 320, 2020–2028. [Google Scholar] [CrossRef]
- Caspersen, C.J.; Powell, K.E.; Christenson, G.M. Physical activity, exercise, and physical fitness: Definitions and distinctions for health-related research. Public Health Rep. 1985, 100, 126–131. [Google Scholar] [PubMed]
- Ahmadi Hekmatikar, A.H.; Ferreira Júnior, J.B.; Shahrbanian, S.; Suzuki, K. Functional and Psychological Changes after Exercise Training in Post-COVID-19 Patients Discharged from the Hospital: A PRISMA-Compliant Systematic Review. Int. J. Environ. Res. Public Health 2022, 19, 2290. [Google Scholar] [CrossRef]
- Khoramipour, K.; Hekmatikar, A.A.; Sotvan, H. An overview of Fatmax and MFO in exercise. Razi J. Med. Sci. 2020, 27, 49–59. [Google Scholar]
- Tayebi, S.M.; Hekmatikar, A.A.; Ghanbari-Niaki, A.; Fathi, R. Ghrelin behavior in exercise and training. J Med. Sci 2020, 27, 85–111. [Google Scholar]
- Ahmadi Hekmatikar, A.; Haghshenas, R.; Mohammad Sadeghipor, A. The effect of carbohydrate supplementation and pure water on interleukin 10, glucose and hematologicalindexes in male football players. Sport Physiol. Manag. Investig. 2019, 11, 135–145. [Google Scholar]
- Bull, F.C.; Al-Ansari, S.S.; Biddle, S.; Borodulin, K.; Buman, M.P.; Cardon, G.; Carty, C.; Chaput, J.-P.; Chastin, S.; Chou, R. World Health Organization 2020 guidelines on physical activity and sedentary behaviour. Br. J. Sports Med. 2020, 54, 1451–1462. [Google Scholar] [CrossRef]
- Safdar, A.; Saleem, A.; Tarnopolsky, M.A. The potential of endurance exercise-derived exosomes to treat metabolic diseases. Nat. Rev. Endocrinol. 2016, 12, 504–517. [Google Scholar] [CrossRef] [PubMed]
- Abodonya, A.M.; Abdelbasset, W.K.; Awad, E.A.; Elalfy, I.E.; Salem, H.A.; Elsayed, S.H. Inspiratory muscle training for recovered COVID-19 patients after weaning from mechanical ventilation: A pilot control clinical study. Medicine 2021, 100, e25339. [Google Scholar] [CrossRef] [PubMed]
- Al Chikhanie, Y.; Veale, D.; Schoeffler, M.; Pépin, J.L.; Verges, S.; Hérengt, F. Effectiveness of pulmonary rehabilitation in COVID-19 respiratory failure patients post-ICU. Respir. Physiol. Neurobiol. 2021, 287, 103639. [Google Scholar] [CrossRef] [PubMed]
- Capin, J.J.; Jolley, S.E.; Morrow, M.; Connors, M.; Hare, K.; MaWhinney, S.; Nordon-Craft, A.; Rauzi, M.; Flynn, S.; Stevens-Lapsley, J.E.; et al. Safety, feasibility and initial efficacy of an app-facilitated telerehabilitation (AFTER) programme for COVID-19 survivors: A pilot randomised study. BMJ Open 2022, 12, e061285. [Google Scholar] [CrossRef]
- Clavario, P.; De Marzo, V.; Lotti, R.; Barbara, C.; Porcile, A.; Russo, C.; Beccaria, F.; Bonavia, M.; Bottaro, L.C.; Caltabellotta, M. Cardiopulmonary exercise testing in COVID-19 patients at 3 months follow-up. Int. J. Cardiol. 2021, 340, 113–118. [Google Scholar] [CrossRef] [PubMed]
- Dalbosco-Salas, M.; Torres-Castro, R.; Rojas Leyton, A.; Morales Zapata, F.; Henríquez Salazar, E.; Espinoza Bastías, G.; Beltrán Díaz, M.E.; Tapia Allers, K.; Mornhinweg Fonseca, D.; Vilaró, J. Effectiveness of a Primary Care Telerehabilitation Program for Post-COVID-19 Patients: A Feasibility Study. J. Clin. Med. 2021, 10, 4428. [Google Scholar] [CrossRef]
- do Amaral, V.T.; Viana, A.A.; Heubel, A.D.; Linares, S.N.; Martinelli, B.; Witzler, P.H.C.; de Souza Zanini, G.; Mendes, R.; Ciolac, E. Cardiovascular, Respiratory, and Functional Effects of Home-based Exercise Training after COVID-19 Hospitalization. Med. Sci. Sports Exerc. 2022, 54, 1795–1803. [Google Scholar] [CrossRef]
- Dorelli, G.; Braggio, M.; Gabbiani, D.; Busti, F.; Caminati, M.; Senna, G.; Girelli, D.; Laveneziana, P.; Ferrari, M.; Sartori, G.; et al. Importance of Cardiopulmonary Exercise Testing amongst Subjects Recovering from COVID-19. Diagnostics 2021, 11, 507. [Google Scholar] [CrossRef]
- Everaerts, S.; Heyns, A.; Langer, D.; Beyens, H.; Hermans, G.; Troosters, T.; Gosselink, R.; Lorent, N.; Janssens, W. COVID-19 recovery: Benefits of multidisciplinary respiratory rehabilitation. BMJ Open Respir. Res. 2021, 8, e000837. [Google Scholar] [CrossRef] [PubMed]
- Gloeckl, R.; Leitl, D.; Jarosch, I.; Schneeberger, T.; Nell, C.; Stenzel, N.; Vogelmeier, C.F.; Kenn, K.; Koczulla, A.R. Benefits of pulmonary rehabilitation in COVID-19: A prospective observational cohort study. ERJ Open Res. 2021, 7, 00108–02021. [Google Scholar] [CrossRef] [PubMed]
- Hayden, M.C.; Limbach, M.; Schuler, M.; Merkl, S.; Schwarzl, G.; Jakab, K.; Nowak, D.; Schultz, K. Effectiveness of a three-week inpatient pulmonary rehabilitation program for patients after COVID-19: A prospective observational study. Int. J. Environ. Res. Public Health 2021, 18, 9001. [Google Scholar] [CrossRef] [PubMed]
- Kortianou, E.A.; Tsimouris, D.; Mavronasou, A.; Lekkas, S.; Kazatzis, N.; Apostolara, Z.E.; Isakoglou, M.; Dimakou, G.; Barmparessou, Z.; Tsikrika, S. Application of a home-based exercise program combined with tele-rehabilitation in previously hospitalized patients with COVID-19: A feasibility, single-cohort interventional study. Pneumon 2022, 35, 12. [Google Scholar] [CrossRef]
- Liu, K.; Zhang, W.; Yang, Y.; Zhang, J.; Li, Y.; Chen, Y. Respiratory rehabilitation in elderly patients with COVID-19: A randomized controlled study. Complement. Ther. Clin. Pract. 2020, 39, 101166. [Google Scholar] [CrossRef]
- Mapelli, M.; Vignati, C.; Gugliandolo, P.; Fumagalli, D.; Agostoni, P. Feasibility of remote home monitoring with a T-shirt wearable device in post-recovery COVID-19 patients. J. Cardiovasc. Med. 2021, 22, 860–863. [Google Scholar] [CrossRef]
- Paneroni, M.; Vitacca, M.; Bernocchi, P.; Bertacchini, L.; Scalvini, S. Feasibility of tele-rehabilitation in survivors of COVID-19 pneumonia. Pulmonology 2022, 28, 152. [Google Scholar] [CrossRef]
- Philippot, A.; Moulin, P.; Charon, M.-H.; Balestra, C.; Dubois, V.; de Timary, P.; De Volder, A.; Bleyenheuft, Y.; Lambrechts, K. Feasibility of Online High-Intensity Interval Training (HIIT) on Psychological Symptoms in Students in Lockdown During the COVID-19 Pandemic: A Randomized Controlled Trial. Front. Psychiatry 2022, 13, 904283. [Google Scholar] [CrossRef]
- Piquet, V.; Luczak, C.; Seiler, F.; Monaury, J.; Martini, A.; Ward, A.B.; Gracies, J.M.; Motavasseli, D. Do Patients With COVID-19 Benefit from Rehabilitation? Functional Outcomes of the First 100 Patients in a COVID-19 Rehabilitation Unit. Arch. Phys. Med. Rehabil. 2021, 102, 1067–1074. [Google Scholar] [CrossRef]
- Rodriguez-Blanco, C.; Gonzalez-Gerez, J.J.; Bernal-Utrera, C.; Anarte-Lazo, E.; Perez-Ale, M.; Saavedra-Hernandez, M. Short-Term Effects of a Conditioning Telerehabilitation Program in Confined Patients Affected by COVID-19 in the Acute Phase. A Pilot Randomized Controlled Trial. Medicina 2021, 57, 684. [Google Scholar] [CrossRef]
- Spruit, M.A.; Holland, A.E.; Singh, S.J.; Tonia, T.; Wilson, K.C.; Troosters, T. COVID-19: Interim guidance on rehabilitation in the hospital and post-hospital phase from a European Respiratory Society-and American Thoracic Society-coordinated international task force. Eur. Respir. J. 2020, 56, 2002197. [Google Scholar] [CrossRef]
- Watanabe, R.; Kojima, M.; Yasuoka, M.; Kimura, C.; Kamiji, K.; Otani, T.; Tsujimura, S.; Fujita, H.; Nogimura, A.; Ozeki, S. Home-Based Frailty Prevention Program for Older Women Participants of Kayoi-No-Ba during the COVID-19 Pandemic: A Feasibility Study. Int. J. Environ. Res. Public Health 2022, 19, 6609. [Google Scholar] [CrossRef] [PubMed]
- Zampogna, E.; Paneroni, M.; Belli, S.; Aliani, M.; Gandolfo, A.; Visca, D.; Bellanti, M.T.; Ambrosino, N.; Vitacca, M. Pulmonary rehabilitation in patients recovering from COVID-19. Respiration 2021, 100, 416–422. [Google Scholar] [CrossRef] [PubMed]
- Peiris, S.; Mesa, H.; Aysola, A.; Manivel, J.; Toledo, J.; Borges-Sa, M.; Aldighieri, S.; Reveiz, L. Pathological findings in organs and tissues of patients with COVID-19: A systematic review. PLoS ONE 2021, 16, e0250708. [Google Scholar] [CrossRef] [PubMed]
- Bhalla, V.; Blish, C.A.; South, A.M. A historical perspective on ACE2 in the COVID-19 era. J. Hum. Hypertens. 2021, 35, 935–939. [Google Scholar] [CrossRef] [PubMed]
- Zarei, M.; Bose, D.; Nouri-Vaskeh, M.; Tajiknia, V.; Zand, R.; Ghasemi, M. Long-term side effects and lingering symptoms post COVID-19 recovery. Rev. Med. Virol. 2022, 32, e2289. [Google Scholar] [CrossRef]
- Barisic, A.; Leatherdale, S.T.; Kreiger, N. Importance of frequency, intensity, time and type (FITT) in physical activity assessment for epidemiological research. Can. J. Public Health 2011, 102, 174–175. [Google Scholar] [CrossRef]
- Leuchtmann, A.B.; Adak, V.; Dilbaz, S.; Handschin, C. The role of the skeletal muscle secretome in mediating endurance and resistance training adaptations. Front. Physiol. 2021, 12, 1296. [Google Scholar] [CrossRef]
- Wahren, J.; Felig, P.; Ahlborg, G.; Jorfeldt, L. Glucose metabolism during leg exercise in man. J. Clin. Investig. 1971, 50, 2715–2725. [Google Scholar] [CrossRef]
- Magliulo, L.; Bondi, D.; Pini, N.; Marramiero, L.; Di Filippo, E.S. The wonder exerkines—Novel insights: A critical state-of-the-art review. Mol. Cell. Biochem. 2021, 447, 105–113. [Google Scholar] [CrossRef]
- Zinman, B.; Ruderman, N.; Campaigne, B.N.; Devlin, J.T.; Schneider, S.H.; American Diabetes Association. Physical activity/exercise and diabetes mellitus. Diabetes Care 2003, 26, s73–s77. [Google Scholar]
- Ataeinosrat, A.; Saeidi, A.; Abednatanzi, H.; Rahmani, H.; Daloii, A.A.; Pashaei, Z.; Hojati, V.; Basati, G.; Mossayebi, A.; Laher, I. Intensity Dependent Effects of Interval Resistance Training on Myokines and Cardiovascular Risk Factors in Males with Obesity. Front. Endocrinol. 2022, 13, 895512. [Google Scholar] [CrossRef] [PubMed]
- Belardinelli, R.; Georgiou, D.; Scocco, V.; Barstow, T.J.; Purcaro, A. Low intensity exercise training in patients with chronic heart failure. J. Am. Coll. Cardiol. 1995, 26, 975–982. [Google Scholar] [CrossRef] [PubMed]
- Yaman, B.; Akpınar, O.; Kemal, H.S.; Cerit, L.; Sezenöz, B.; Açıkgöz, E.; Duygu, H. The beneficial effect of low-intensity exercise on cardiac performance assessed by two-dimensional speckle tracking echocardiography. Echocardiography 2020, 37, 1989–1999. [Google Scholar] [CrossRef]
- Hanani, M.; Gaeini, A.; Nuri, R.; Hemati Nafar, M. The effect of intensity of interval training on expression of cordial myostatin and follistatin proteins in rats with myocardial infarction. Razi J. Med. Sci. 2020, 27, 239–252. [Google Scholar]
- Kon, M.; Ebi, Y.; Nakagaki, K. Effects of acute sprint interval exercise on follistatin-like 1 and apelin secretions. Arch. Physiol. Biochem. 2021, 127, 223–227. [Google Scholar] [CrossRef]
- Willis, S.A.; Sargeant, J.A.; Thackray, A.E.; Yates, T.; Stensel, D.J.; Aithal, G.P.; King, J.A. Effect of exercise intensity on circulating hepatokine concentrations in healthy men. Appl. Physiol. Nutr. Metab. 2019, 44, 1065–1072. [Google Scholar] [CrossRef]
- Görgens, S.W.; Eckardt, K.; Jensen, J.; Drevon, C.A.; Eckel, J. Exercise and Regulation of Adipokine and Myokine Production. Prog. Mol. Biol. Transl. Sci. 2015, 135, 313–336. [Google Scholar] [CrossRef]
- Ingerslev, B.; Hansen, J.S.; Hoffmann, C.; Clemmesen, J.O.; Secher, N.H.; Scheler, M.; Hrabĕ de Angelis, M.; Häring, H.U.; Pedersen, B.K.; Weigert, C.; et al. Angiopoietin-like protein 4 is an exercise-induced hepatokine in humans, regulated by glucagon and cAMP. Mol. Metab. 2017, 6, 1286–1295. [Google Scholar] [CrossRef]
- Kim, K.H.; Kim, S.H.; Min, Y.-K.; Yang, H.-M.; Lee, J.-B.; Lee, M.-S. Acute exercise induces FGF21 expression in mice and in healthy humans. PLoS ONE 2013, 8, e63517. [Google Scholar] [CrossRef]
- Sargeant, J.A.; Aithal, G.P.; Takamura, T.; Misu, H.; Takayama, H.; Douglas, J.A.; Turner, M.C.; Stensel, D.J.; Nimmo, M.A.; Webb, D.R.; et al. The influence of adiposity and acute exercise on circulating hepatokines in normal-weight and overweight/obese men. Appl. Physiol. Nutr. Metab. 2018, 43, 482–490. [Google Scholar] [CrossRef]
- Misu, H.; Takayama, H.; Saito, Y.; Mita, Y.; Kikuchi, A.; Ishii, K.A.; Chikamoto, K.; Kanamori, T.; Tajima, N.; Lan, F.; et al. Deficiency of the hepatokine selenoprotein P increases responsiveness to exercise in mice through upregulation of reactive oxygen species and AMP-activated protein kinase in muscle. Nat. Med. 2017, 23, 508–516. [Google Scholar] [CrossRef]
- Golbidi, S.; Laher, I. Exercise induced adipokine changes and the metabolic syndrome. J. Diabetes Res. 2014, 2014, 726861. [Google Scholar] [CrossRef] [PubMed]
- Park, K.M.; Park, S.C.; Kang, S. Effects of resistance exercise on adipokine factors and body composition in pre- and postmenopausal women. J. Exerc. Rehabil. 2019, 15, 676–682. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Gil, A.M.; Elizondo-Montemayor, L. The role of exercise in the interplay between myokines, hepatokines, osteokines, adipokines, and modulation of inflammation for energy substrate redistribution and fat mass loss: A review. Nutrients 2020, 12, 1899. [Google Scholar] [CrossRef]
- Méry, G.; Epaulard, O.; Borel, A.-L.; Toussaint, B.; Le Gouellec, A. COVID-19: Underlying adipokine storm and angiotensin 1-7 umbrella. Front. Immunol. 2020, 11, 1714. [Google Scholar] [CrossRef] [PubMed]
- Mosaffa, N.; Abedi, B. The Effect of Eight Week Interval training on The Serum VCAM-1 and PAI-1 in Obesity Women. J. Sport Biosci. 2018, 10, 193–206. [Google Scholar]
- Voss, S.; Nikolovski, Z.; Bourdon, P.; Alsayrafi, M.; Schumacher, Y. The effect of cumulative endurance exercise on leptin and adiponectin and their role as markers to monitor training load. Biol. Sport 2016, 33, 23–28. [Google Scholar] [PubMed]
- Jamurtas, A.Z.; Theocharis, V.; Koukoulis, G.; Stakias, N.; Fatouros, I.; Kouretas, D.; Koutedakis, Y. The effects of acute exercise on serum adiponectin and resistin levels and their relation to insulin sensitivity in overweight males. Eur. J. Appl. Physiol. 2006, 97, 122–126. [Google Scholar] [CrossRef]
- Kraemer, R.R.; Aboudehen, K.S.; Carruth, A.K.; Durand, R.J.; Acevedo, E.O.; Hebert, E.P.; Johnson, L.G.; Castracane, V.D. Adiponectin responses to continuous and progressively intense intermittent exercise. Med. Sci. Sport. Exerc. 2003, 35, 1320–1325. [Google Scholar] [CrossRef] [PubMed]
- Jürimäe, J.; Hofmann, P.; Jürimäe, T.; Mäestu, J.; Purge, P.; Wonisch, M.; Pokan, R.; Von Duvillard, S. Plasma adiponectin response to sculling exercise at individual anaerobic threshold in college level male rowers. Int. J. Sports Med. 2006, 27, 272–277. [Google Scholar] [CrossRef]
- Racil, G.; Ben Ounis, O.; Hammouda, O.; Kallel, A.; Zouhal, H.; Chamari, K.; Amri, M. Effects of high vs. moderate exercise intensity during interval training on lipids and adiponectin levels in obese young females. Eur. J. Appl. Physiol. 2013, 113, 2531–2540. [Google Scholar] [CrossRef]
- Ferguson, M.A.; White, L.J.; McCoy, S.; Kim, H.-W.; Petty, T.; Wilsey, J. Plasma adiponectin response to acute exercise in healthy subjects. Eur. J. Appl. Physiol. 2004, 91, 324–329. [Google Scholar] [CrossRef] [PubMed]
- Vardar, S.A.; Karaca, A.; Güldiken, S.; Palabıyık, O.; Süt, N.; Demir, A.M. High-intensity interval training acutely alters plasma adipokine levels in young overweight/obese women. Arch. Physiol. Biochem. 2018, 124, 149–155. [Google Scholar] [CrossRef] [PubMed]
- De Souza, D.C.; Matos, V.A.; Dos Santos, V.O.; Medeiros, I.F.; Marinho, C.S.; Nascimento, P.R.; Dorneles, G.P.; Peres, A.; Müller, C.H.; Krause, M.; et al. Effects of high-intensity interval and moderate-intensity continuous exercise on inflammatory, leptin, IgA, and lipid peroxidation responses in obese males. Front. Physiol. 2018, 9, 567. [Google Scholar] [CrossRef] [PubMed]
- Nagendra, H. Yoga for COVID-19. Int. J. Yoga 2020, 13, 87. [Google Scholar] [CrossRef]
- Yaacoub, S.; Khabsa, J.; El-Khoury, R.; El-Harakeh, A.; Lotfi, T.; Saad, Z.; Itani, Z.; Khamis, A.M.; El Mikati, I.; Cuello-Garcia, C.A.; et al. COVID-19 transmission during swimming-related activities: A rapid systematic review. BMC Infect. Dis. 2021, 21, 1112. [Google Scholar] [CrossRef]
- Vitale, J.A.; Bonato, M.; Borghi, S.; Messina, C.; Albano, D.; Corbetta, S.; Sconfienza, L.M.; Banfi, G. Home-based resistance training for older subjects during the COVID-19 outbreak in Italy: Preliminary results of a six-months RCT. Int. J. Environ. Res. Public Health 2020, 17, 9533. [Google Scholar] [CrossRef] [PubMed]
- Budi, D.R.; Widyaningsih, R.; Nur, L.; Agustan, B.; Dwi, D.; Qohhar, W.; Asnaldi, A. Cycling during COVID-19 pandemic: Sports or lifestyle. Int. J. Hum. Mov. Sports Sci. 2021, 9, 765–771. [Google Scholar] [CrossRef]
- Ahn, N.; Kim, K. Effects of Aerobic and Resistance Exercise on Myokines in High Fat Diet-Induced Middle-Aged Obese Rats. Int. J. Environ. Res. Public Health 2020, 17, 2685. [Google Scholar] [CrossRef]
- Urzi, F.; Marusic, U.; Ličen, S.; Buzan, E. Effects of elastic resistance training on functional performance and Myokines in older women—A randomized controlled trial. J. Am. Med. Dir. Assoc. 2019, 20, 830–834.e2. [Google Scholar] [CrossRef]
- Zunner, B.E.; Wachsmuth, N.B.; Eckstein, M.L.; Scherl, L.; Schierbauer, J.R.; Haupt, S.; Stumpf, C.; Reusch, L.; Moser, O. Myokines and Resistance Training: A Narrative Review. Int. J. Mol. Sci. 2022, 23, 3501. [Google Scholar] [CrossRef] [PubMed]
- Eckardt, K.; Görgens, S.W.; Raschke, S.; Eckel, J. Myokines in insulin resistance and type 2 diabetes. Diabetologia 2014, 57, 1087–1099. [Google Scholar] [CrossRef]
- Cornish, S.M.; Bugera, E.M.; Duhamel, T.A.; Peeler, J.D.; Anderson, J.E. A focused review of myokines as a potential contributor to muscle hypertrophy from resistance-based exercise. Eur. J. Appl. Physiol. 2020, 120, 941–959. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.; Park, I.; Lim, S.-T. Changing Levels of Myokines after Aerobic Training and Resistance Training in Post-Menopausal Obese Females: A Randomized Controlled Trial. Sustainability 2020, 12, 8413. [Google Scholar] [CrossRef]
- Catoire, M.; Mensink, M.; Kalkhoven, E.; Schrauwen, P.; Kersten, S. Identification of human exercise-induced myokines using secretome analysis. Physiol. Genom. 2014, 46, 256–267. [Google Scholar] [CrossRef]
- Hosseini, M.; Bambaeichi, E.; Sarir, H.; Kargarfard, M. Effect of training with or without Ziziphus jujuba extract on cardiokines in heart tissue of myocardial infarcted rats. Int. J. Prev. Med. 2019, 10, 103. [Google Scholar]
- de Piano, A.; de Mello, M.T.; Sanches, P.d.L.; da Silva, P.L.; Campos, R.M.; Carnier, J.; Corgosinho, F.; Foschini, D.; Masquio, D.L.; Tock, L.; et al. Long-term effects of aerobic plus resistance training on the adipokines and neuropeptides in nonalcoholic fatty liver disease obese adolescents. Eur. J. Gastroenterol. Hepatol. 2012, 24, 1313–1324. [Google Scholar] [CrossRef] [PubMed]
- Ward, L.J.; Nilsson, S.; Hammar, M.; Lindh-Åstrand, L.; Berin, E.; Lindblom, H.; Spetz Holm, A.-C.; Rubér, M.; Li, W. Resistance training decreases plasma levels of adipokines in postmenopausal women. Sci. Rep. 2020, 10, 19837. [Google Scholar] [CrossRef] [PubMed]
- Negaresh, R.; Motl, R.W.; Mokhtarzade, M.; Dalgas, U.; Patel, D.; Shamsi, M.M.; Majdinasab, N.; Ranjbar, R.; Zimmer, P.; Baker, J.S. Effects of exercise training on cytokines and adipokines in multiple sclerosis: A systematic review. Mult. Scler. Relat. Disord. 2018, 24, 91–100. [Google Scholar] [CrossRef]
- Akbarpour, M.; Ozgoli, G.; Aryamanesh, Z.; Mojab, F.; Majd, H.A.; Karami, M.; Amirmozaffari, N.; Dody, M.; Bazzazi, N.; Akbarzadeh, S. The Effect of Resistance Training on Serum Levels of Adipokine and Inflammatory Markers of Cardiovascular Disease in Obese Men. Qom Univ. Med. Sci. J. 2013, 7, 1–10. [Google Scholar]
- Banitalebi, E.; Mardanpour Shahrekordi, Z.; Kazemi, A.R.; Bagheri, L.; Amani Shalamzari, S.; Faramarzi, M. Comparing the effects of eight weeks of combined training (Endurance and Resistance) in different orders on inflammatory factors and adipokines among elderly females. Women’s Health Bull. 2016, 3, 1–10. [Google Scholar] [CrossRef]
- Jensen-Cody, S.O.; Potthoff, M.J. Hepatokines and metabolism: Deciphering communication from the liver. Mol. Metab. 2021, 44, 101138. [Google Scholar] [CrossRef]
- Keihanian, A.; Arazi, H.; Kargarfard, M. Effects of aerobic versus resistance training on serum fetuin-A, fetuin-B, and fibroblast growth factor-21 levels in male diabetic patients. Physiol. Int. 2019, 106, 70–80. [Google Scholar] [CrossRef] [PubMed]
- Hashemi Jokar, S.E.; Peeri, M.; Azarbayjani, M.A. The effect of high, low and moderate intensity circuit resistance training on the levels of hepatocyte-derived fibrinogen-related protein 1 (HFREP1) and lipid profile in obese postmenopausal women. J. Basic Res. Med. Sci. 2022, 9, 61–70. [Google Scholar]
- Keihaniyan, A.; Arazi, H.; Kargarfard, M. The Effect of Eight-Week Resistance and Aerobic Training on Lipid Profile and Serum Levels of Hepatokine HFREP1 in Obese Men with Type 2 Diabetes. Sport Physiol. 2018, 10, 85–98. [Google Scholar]
- Ghorbanian, B.; Saberi, Y. The Response of Some Hepatokines and Insulin Resistance to High Intensity Interval Training in Women with Non-Alcoholic Fatty Liver. J. Appl. Exerc. Physiol. 2020, 16, 57–71. [Google Scholar]
- Manabe, Y.; Takagi, M.; Nakamura-Yamada, M.; Goto-Inoue, N.; Taoka, M.; Isobe, T.; Fujii, N.L. Redox proteins are constitutively secreted by skeletal muscle. J. Physiol. Sci. 2014, 64, 401–409. [Google Scholar] [CrossRef]
- Rahimi, M.; Nazarali, P.; Alizadeh, R. Pilates and TRX training methods can improve insulin resistance in overweight women by increasing an exercise-hormone, Irisin. J. Diabetes Metab. Disord. 2021, 20, 1455–1460. [Google Scholar] [CrossRef] [PubMed]
- Timmons, J.A.; Baar, K.; Davidsen, P.K.; Atherton, P.J. Is irisin a human exercise gene? Nature 2012, 488, E9–E10. [Google Scholar] [CrossRef] [PubMed]
- Olarescu, N.C.; Ueland, T.; Godang, K.; Lindberg-Larsen, R.; Jørgensen, J.; Bollerslev, J. Inflammatory adipokines contribute to insulin resistance in active acromegaly and respond differently to different treatment modalities. Eur. J. Endocrinol. 2014, 170, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Rahmatollahi, M.; Valizade-Orang, A.; Bahram, M.E.; Jafarnezhad, A. Effect of 12 Weeks TRX Training on Irisin and Chemerin Profile in Male Older Adults. Sci. J. Rehabil. Med. 2021. [Google Scholar] [CrossRef]
- Chaliki, K.; Delgado, D.; Dong, D.; Moreno, M.; Lee, J.; McCulloch, P.; Harris, J.; Lambert, B. Acute Effects of a Single Session of Hot Yoga on Caloric Expenditure, Range of Motion, and Metabolism. Proc. Int. J. Exerc. Sci. Conf. Proc. 2019, 2, 61. [Google Scholar]
- Cahn, B.R.; Goodman, M.S.; Peterson, C.T.; Maturi, R.; Mills, P.J. Yoga, meditation and mind-body health: Increased BDNF, cortisol awakening response, and altered inflammatory marker expression after a 3-month yoga and meditation retreat. Front. Hum. Neurosci. 2017, 11, 315. [Google Scholar] [CrossRef]
- Yadav, R.; Yadav, R.K.; Khadgawat, R.; Pandey, R.M. Comparative efficacy of a 12 week yoga-based lifestyle intervention and dietary intervention on adipokines, inflammation, and oxidative stress in adults with metabolic syndrome: A randomized controlled trial. Transl. Behav. Med. 2019, 9, 594–604. [Google Scholar] [CrossRef]
- Supriya, R.; Yu, A.P.; Lee, P.H.; Lai, C.W.; Cheng, K.K.; Yau, S.Y.; Chan, L.W.; Yung, B.Y.; Siu, P.M. Yoga training modulates adipokines in adults with high-normal blood pressure and metabolic syndrome. Scand. J. Med. Sci. Sports 2018, 28, 1130–1138. [Google Scholar] [CrossRef]
- Yadav, R.; Yadav, R.K.; Khadgawat, R.; Mehta, N. OS 28-06 beneficial effects of a 12-week yoga-based lifestyle intervention on cardio-metabolic risk factors and adipokines in subjects with pre-hypertension or hypertension. J. Hypertens. 2016, 34, e252. [Google Scholar] [CrossRef]
- Kapilevich, L.V.; Kironenko, T.; Zakharova, A.; Kabachkova, A.; Orlov, S. Level of interleukins IL-6 and IL-15 in blood plasma of mice after forced swimming test. Bull. Exp. Biol. Med. 2017, 163, 10–13. [Google Scholar] [CrossRef]
- Peng, C.-C.; Chen, K.-C.; Hsieh, C.-L.; Peng, R.Y. Swimming exercise prevents fibrogenesis in chronic kidney disease by inhibiting the myofibroblast transdifferentiation. PLoS ONE 2012, 7, e37388. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, M.; Sadoshima, J. Mechanisms of physiological and pathological cardiac hypertrophy. Nat. Rev. Cardiol. 2018, 15, 387–407. [Google Scholar] [CrossRef]
- Mohr, M.; Nordsborg, N.B.; Lindenskov, A.; Steinholm, H.; Nielsen, H.P.; Mortensen, J.; Weihe, P.; Krustrup, P. High-intensity intermittent swimming improves cardiovascular health status for women with mild hypertension. Biomed. Res. Int. 2014, 2014, 728289. [Google Scholar] [CrossRef]
- Zahraei, H.; Mogharnasi, M.; Afzalpour, M.E.; Fanaei, H. The effect of 8 weeks of continuous and high intensity interval swimming on chemerin levels in liver and visceral fat tissues and insulin resistance in male rats with metabolic syndrome. J. Sport Exerc. Physiol. 2022, 15, 33–44. [Google Scholar] [CrossRef]
- Cabrera, A.; Pineda, W.; del Pilar Correa Valencia, N.; Gutierrez, M. Body mass conversion and improved insulin response in Colombian Paso horses subjected to a swimming training program. Comp. Exerc. Physiol. 2022, 18, 211–218. [Google Scholar] [CrossRef]
- Hansen, J.; Brandt, C.; Nielsen, A.R.; Hojman, P.; Whitham, M.; Febbraio, M.A.; Pedersen, B.K.; Plomgaard, P. Exercise induces a marked increase in plasma follistatin: Evidence that follistatin is a contraction-induced hepatokine. Endocrinology 2011, 152, 164–171. [Google Scholar] [CrossRef] [PubMed]
- Dobashi, S.; Nakamura, A.; Saito, K.; Ando, D.; Koyama, K. Acute swimming exercise, but not exposure to moderate hypoxic conditions reduces circulating selenoprotein P levels in short-term, high-fat diet-fed rats. J. Phys. Fit. Sports Med. 2019, 8, 181–184. [Google Scholar] [CrossRef]
- Nazari, M.; Kordi, M.; Minasian, V.; Saffar Kohneh Quchan, A.H. Ameliorating effect of 6-week swimming exercise on mice with experimental autoimmune encephalomyelitis (EAE) by reducing fetuin-A and increasing AMPK & NAD⁺ levels in liver tissue. Iran. J. Basic Med. Sci. 2022, 25, 1016–1020. [Google Scholar] [PubMed]
- Silva, R.; Bueno, P.; Avó, L.; Nonaka, K.; Selistre-Araújo, H.; Leal, A. Effect of physical training on liver expression of activin A and follistatin in a nonalcoholic fatty liver disease model in rats. Braz. J. Med. Biol. Res. 2014, 47, 746–752. [Google Scholar] [CrossRef]
- Arabzadeh, E.; Samadian, Z.; Tofighi, A.; Tolouei Azar, J. Alteration of follistatin-like 1, neuron-derived neurotrophic factor, and vascular endothelial growth factor in diabetic cardiac muscle after moderate-intensity aerobic exercise with insulin. Sport Sci. Health 2020, 16, 491–499. [Google Scholar] [CrossRef]
- Hutchinson, K.A.; Mohammad, S.; Garneau, L.; McInnis, K.; Aguer, C.; Adamo, K.B. Examination of the myokine response in pregnant and non-pregnant women following an acute bout of moderate-intensity walking. Front. Physiol. 2019, 10, 1188. [Google Scholar] [CrossRef]
- Da Silva Lage, V.K.; de Paula, F.A.; Lima, L.P.; Santos, J.N.V.; Dos Santos, J.M.; Viegas, Â.A.; da Silva, G.P.; de Almeida, H.C.; Teixeira, A.L.d.S.N.; Leopoldino, A.A.O. Plasma levels of myokines and inflammatory markers are related with functional and respiratory performance in older adults with COPD and sarcopenia. Exp. Gerontol. 2022, 164, 111834. [Google Scholar] [CrossRef]
- Gmiat, A.; Mieszkowski, J.; Prusik, K.; Kortas, J.; Kochanowicz, A.; Radulska, A.; Lipiński, M.; Tomczyk, M.; Jaworska, J.; Antosiewicz, J.; et al. Changes in pro-inflammatory markers and leucine concentrations in response to Nordic Walking training combined with vitamin D supplementation in elderly women. Biogerontology 2017, 18, 535–548. [Google Scholar] [CrossRef] [PubMed]
- Wahl, P.; Hein, M.; Achtzehn, S.; Bloch, W.; Mester, J. Acute effects of superimposed electromyostimulation during cycling on myokines and markers of muscle damage. J. Musculoskelet. Neuronal. Interact. 2015, 15, 53. [Google Scholar]
- Llanos, A.A.; Krok, J.L.; Peng, J.; Pennell, M.L.; Vitolins, M.Z.; Degraffinreid, C.R.; Paskett, E.D. Effects of a walking intervention using mobile technology and interactive voice response on serum adipokines among postmenopausal women at increased breast cancer risk. Horm. Cancer 2014, 5, 98–103. [Google Scholar] [CrossRef] [PubMed]
- Neumayr, G.; Engler, C.; Lunger, L.; Lechleitner, P. Effects of a One-week Vacation with Various Activity Programs on Metabolism and Adipokines. Int. J. Sports Med. 2021, 42, 703–707. [Google Scholar] [CrossRef]
- Tota, Ł.; Pilch, W.; Piotrowska, A.; Pałka, T.; Pilch, P. The effect of 12-week-long nordic walking exercise on body composition, changes in lipid and carbohydrate metabolism indices, concentration of selected adipokines and calcidiol in healthy middle-aged women. Cent. Eur. J. Sport Sci. Med. 2017, 20, 69–80. [Google Scholar] [CrossRef]
- Seo, D.-I.; So, W.-Y.; Sung, D.J. Changes in insulin resistance and adipokines in obese women following a 12-week programme of combined exercise training. S. Afr. J. Res. Sport Phys. Educ. Recreat. 2016, 38, 39–147. [Google Scholar]
- Stefanyk, L.E.; Dyck, D.J. The interaction between adipokines, diet and exercise on muscle insulin sensitivity. Curr. Opin. Clin. Nutr. Metab. Care 2010, 13, 255–259. [Google Scholar] [CrossRef] [PubMed]
- Babaei, P.; Hosseini, R. Exercise training modulates adipokines dysregulations in metabolic syndrome. Sports Med. Health Sci. 2022, 4, 18–28. [Google Scholar] [CrossRef]
- Sargeant, J.A. Exercise and Insulin Sensitivity: Interaction with Intrahepatic Triglyceride and Hepatokines. Ph.D. Thesis, Loughborough University, Loughborough, UK, 2018. [Google Scholar]
- Garneau, L.; Parsons, S.A.; Smith, S.R.; Mulvihill, E.E.; Sparks, L.M.; Aguer, C. Plasma myokine concentrations after acute exercise in non-obese and obese sedentary women. Front. Physiol. 2020, 11, 18. [Google Scholar] [CrossRef]
- Chikamoto, K.; Misu, H.; Takayama, H.; Kikuchi, A.; Ishii, K.-A.; Lan, F.; Takata, N.; Tajima-Shirasaki, N.; Takeshita, Y.; Tsugane, H.; et al. Rapid response of the steatosis-sensing hepatokine LECT2 during diet-induced weight cycling in mice. Biochem. Biophys. Res. Commun. 2016, 478, 1310–1316. [Google Scholar] [CrossRef] [PubMed]
- Takata, N.; Ishii, K.-A.; Takayama, H.; Nagashimada, M.; Kamoshita, K.; Tanaka, T.; Kikuchi, A.; Takeshita, Y.; Matsumoto, Y.; Ota, T.; et al. LECT2 as a hepatokine links liver steatosis to inflammation via activating tissue macrophages in NASH. Sci. Rep. 2021, 11, 555. [Google Scholar] [CrossRef] [PubMed]
- Vancini, R.L.; Andrade, M.S.; Viana, R.B.; Nikolaidis, P.T.; Knechtle, B.; Campanharo, C.R.; de Almeida, A.A.; Gentil, P.; de Lira, C.A. Physical exercise and COVID-19 pandemic in PubMed: Two months of dynamics and one year of original scientific production. Sport. Med. Health Sci. 2021, 3, 80–92. [Google Scholar] [CrossRef]
- Scartoni, F.R.; Sant’Ana, L.d.O.; Murillo-Rodriguez, E.; Yamamoto, T.; Imperatori, C.; Budde, H.; Vianna, J.M.; Machado, S. Physical exercise and immune system in the elderly: Implications and importance in COVID-19 pandemic period. Front. Psychol. 2020, 11, 593903. [Google Scholar] [CrossRef]
- Dixit, S. Can moderate intensity aerobic exercise be an effective and valuable therapy in preventing and controlling the pandemic of COVID-19? Med. Hypotheses 2020, 143, 109854. [Google Scholar] [CrossRef]
- Lin, J.; Guo, T.; Becker, B.; Yu, Q.; Chen, S.-T.; Brendon, S.; Hossain, M.M.; Cunha, P.M.; Soares, F.C.; Veronese, N.; et al. Depression is associated with moderate-intensity physical activity among college students during the COVID-19 pandemic: Differs by activity level, gender and gender role. Psychol. Res. Behav. Manag. 2020, 13, 1123. [Google Scholar] [CrossRef] [PubMed]
- Besse-Patin, A.; Montastier, E.; Vinel, C.; Castan-Laurell, I.; Louche, K.; Dray, C.; Daviaud, D.; Mir, L.; Marques, M.; Thalamas, C.; et al. Effect of endurance training on skeletal muscle myokine expression in obese men: Identification of apelin as a novel myokine. Int. J. Obes. 2014, 38, 707–713. [Google Scholar] [CrossRef] [PubMed]
- Kwak, S.E.; Cho, S.C.; Bae, J.H.; Lee, J.; Shin, H.E.; Di Zhang, D.; Lee, Y.-I.; Song, W. Effects of exercise-induced apelin on muscle function and cognitive function in aged mice. Exp. Gerontol. 2019, 127, 110710. [Google Scholar] [CrossRef]
- Vinel, C.; Lukjanenko, L.; Batut, A.; Deleruyelle, S.; Pradere, J.-P.; Le Gonidec, S.; Dortignac, A.; Geoffre, N.; Pereira, O.; Karaz, S.; et al. The exerkine apelin reverses age-associated sarcopenia. Nat. Med. 2018, 24, 1360–1371. [Google Scholar] [CrossRef] [PubMed]
- Chapman, M.A.; Arif, M.; Emanuelsson, E.B.; Reitzner, S.M.; Lindholm, M.E.; Mardinoglu, A.; Sundberg, C.J. Skeletal muscle transcriptomic comparison between long-term trained and untrained men and women. Cell Rep. 2020, 31, 107808. [Google Scholar] [CrossRef] [PubMed]
- McPherron, A.C.; Lawler, A.M.; Lee, S.-J. Regulation of skeletal muscle mass in mice by a new TGF-p superfamily member. Nature 1997, 387, 83–90. [Google Scholar] [CrossRef]
- Domin, R.; Dadej, D.; Pytka, M.; Zybek-Kocik, A.; Ruchała, M.; Guzik, P. Effect of various exercise regimens on selected exercise-induced cytokines in healthy people. Int. J. Environ. Res. Public Health 2021, 18, 1261. [Google Scholar] [CrossRef]
- Yeo, N.H.; Woo, J.; Shin, K.O.; Park, J.Y.; Kang, S. The effects of different exercise intensity on myokine and angiogenesis factors. J. Sports Med. Phys. Fit. 2012, 52, 448–454. [Google Scholar]
- Banitalebi, E.; Kazemi, A.; Faramarzi, M.; Nasiri, S.; Haghighi, M.M. Effects of sprint interval or combined aerobic and resistance training on myokines in overweight women with type 2 diabetes: A randomized controlled trial. Life Sci. 2019, 217, 101–109. [Google Scholar] [CrossRef] [PubMed]
- Bugera, E.M.; Duhamel, T.A.; Peeler, J.D.; Cornish, S.M. The systemic myokine response of decorin, interleukin-6 (IL-6) and interleukin-15 (IL-15) to an acute bout of blood flow restricted exercise. Eur. J. Appl. Physiol. 2018, 118, 2679–2686. [Google Scholar] [CrossRef] [PubMed]
- Vints, W.A.; Levin, O.; Fujiyama, H.; Verbunt, J.; Masiulis, N. Exerkines and long-term synaptic potentiation: Mechanisms of exercise-induced neuroplasticity. Front. Neuroendocrinol. 2022, 66, 100993. [Google Scholar] [PubMed]
- Türkel, İ.; Özerkliğ, B.; Atakan, M.M.; Aktitiz, S.; Koşar, Ş.N.; Yazgan, B. Exercise and Metabolic Health: The Emerging Roles of Novel Exerkines. Curr. Protein Pept. Sci. 2022, 23, 437–455. [Google Scholar] [PubMed]
- Correia, J.C.; Ruas, J.L. Exercised cytokines promote endurance. Science 2020, 368, 470–471. [Google Scholar] [CrossRef]
- Shokri, E.; Heidarianpour, A.; Razavi, Z. Positive effect of combined exercise on adipokines levels and pubertal signs in overweight and obese girls with central precocious puberty. Lipids Health Dis. 2021, 20, 152. [Google Scholar] [CrossRef] [PubMed]
- You, T.; Nicklas, B.J. Effects of exercise on adipokines and the metabolic syndrome. Curr. Diabetes Rep. 2008, 8, 7–11. [Google Scholar] [CrossRef]
- Fyfe, J.J.; Bishop, D.J.; Stepto, N.K. Interference between concurrent resistance and endurance exercise: Molecular bases and the role of individual training variables. Sports Med. 2014, 44, 743–762. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, K.; Totsuka, M.; Nakaji, S.; Yamada, M.; Kudoh, S.; Liu, Q.; Sugawara, K.; Yamaya, K.; Sato, K. Endurance exercise causes interaction among stress hormones, cytokines, neutrophil dynamics, and muscle damage. J. Appl. Physiol. 1999, 87, 1360–1367. [Google Scholar] [CrossRef]
- Jiménez-Pavón, D.; Carbonell-Baeza, A.; Lavie, C.J. Physical exercise as therapy to fight against the mental and physical consequences of COVID-19 quarantine: Special focus in older people. Prog. Cardiovasc. Dis. 2020, 63, 386. [Google Scholar] [CrossRef] [PubMed]
- Kakanis, M.; Peake, J.; Hooper, S.; Gray, B.; Marshall-Gradisnik, S. The open window of susceptibility to infection after acute exercise in healthy young male elite athletes. J. Sci. Med. Sport 2010, 13, e85–e86. [Google Scholar] [CrossRef]
- Campbell, J.P.; Turner, J.E. Debunking the myth of exercise-induced immune suppression: Redefining the impact of exercise on immunological health across the lifespan. Front. Immunol. 2018, 9, 648. [Google Scholar] [CrossRef] [PubMed]
- Molanouri Shamsi, M.; Hassan, Z.M.; Quinn, L.S.; Gharakhanlou, R.; Baghersad, L.; Mahdavi, M. Time course of IL-15 expression after acute resistance exercise in trained rats: Effect of diabetes and skeletal muscle phenotype. Endocrine 2015, 49, 396–403. [Google Scholar] [CrossRef] [PubMed]
- He, Z.; Tian, Y.; Valenzuela, P.L.; Huang, C.; Zhao, J.; Hong, P.; He, Z.; Yin, S.; Lucia, A. Myokine Response to High-Intensity Interval vs. Resistance Exercise: An Individual Approach. Front. Physiol. 2018, 9, 1735. [Google Scholar] [CrossRef]
- Farhani, F.; Shahrbanian, S.; Auais, M.; Hekmatikar, A.H.A.; Suzuki, K. Effects of Aerobic Training on Brain Plasticity in Patients with Mild Cognitive Impairment: A Systematic Review of Randomized Controlled Trials. Brain Sci. 2022, 12, 732. [Google Scholar] [CrossRef]
- Dun, Y.; Smith, J.R.; Liu, S.; Olson, T.P. High-Intensity Interval Training in Cardiac Rehabilitation. Clin. Geriatr. Med. 2019, 35, 469–487. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Xing, J.; Zhao, B.; Wang, Y.; Zhang, L.; Wang, Y.; Zheng, M.; Liu, G. The Effects of High-Intensity Interval Training on Exercise Capacity and Prognosis in Heart Failure and Coronary Artery Disease: A Systematic Review and Meta-Analysis. Cardiovasc. Ther. 2022, 2022, 4273809. [Google Scholar] [CrossRef] [PubMed]
- Seo, D.Y.; Park, S.H.; Marquez, J.; Kwak, H.B.; Kim, T.N.; Bae, J.H.; Koh, J.H.; Han, J. Hepatokines as a Molecular Transducer of Exercise. J. Clin. Med. 2021, 10, 385. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, K. Recent Progress in Applicability of Exercise Immunology and Inflammation Research to Sports Nutrition. Nutrients 2021, 13, 4299. [Google Scholar] [CrossRef] [PubMed]

| Physical Exercise Type (L/M/H) | Exerkines | |||||
|---|---|---|---|---|---|---|
| Myokines | Cardiokines | Adipokines | Hepatokines | Outcome | Reference | |
| Resistance training (Resistance weight training exercise with a resistance machine) | (L/M) | (L/M) | (L/M) | (L/M) | Resistance training with low to moderate intensity can reduce inflammation and increase muscle strength by changing myokines. This can be effective in COVID-19 patients who suffer from decreased muscle strength and high inflammation during the illness. Moreover, resistance training with these intensities can benefit patients discharged from the hospital. However, high-intensity resistance training cannot positively affect myokines, and the results of articles have reported an increase in inflammation and a decrease in muscle strength. The effect of resistance training on cardiokines with low to moderate intensity can be suitable for improving high blood pressure and reducing inflammation. However, high-intensity resistance training may not be good for COVID-19 patients or those discharged from the hospital. Although studies on cardiokines are very limited, they can increase inflammation and possibly increase blood pressure. Low to moderate resistance training has a good effect on adipokines and hepatokines and can reduce inflammation, improve immune system function, improve insulin sensitivity, and increase fat tissue metabolism. In connection with high-intensity resistance training, the increase in inflammation is seen because of the behavioral changes in fat tissue and the decrease in the body function’s immune system. | |
| ↓myostatin ↓TGF-β1 ↓IL-6 ↑IL-15 ↑decorin ↑irisin ↑BDNF ↑IL-15 ↑FGF ↔SPARC | ↓ANP ↔BNP ↑IL-33 ↓IL-6 ↑IL-18 ↓IL-1β ↓Follistatin ↑FGF ↔Sfrp ↑Neurotrophins ↓TNF-α ↓TGF-β | ↓Leptin ↑Adiponectin ↑Chemerin ↓Visfatin ↓Omentin ↑Vaspin ↔Progranulin ↔CTRP-4 | ↑Activin-E ↔ANGPTL3 ↔ANGPTL4 ↑ANGPTL6 ↑Fetuin-A ↑FGF21 ↓Follistatin ↑GDF15 ↓Hepassocin ↑IGF1 ↓LECT2 ↑Lipocalin 13 ↑Selenoprotein-P ↑Tsukushi | [78,93,94,95,96,97,98,99,100,101,102,103,104,105,106,107,108,109,110] | ||
| H | H | H | H | |||
| ↓myostatin ↓TGF-β1 ↑IL-6 ↑IL-15 ↑decorin ↑irisin ↑BDNF ↑IL-15 ↑FGF ↑SPARC | ↔ANP ↔BNP ↔IL-33 ↔IL-6 ↑IL-18 ↓IL-1β ↓Follistatin ↑FGF ↔Sfrp ↑Neurotrophins ↓TNF-α ↓TGF-β | ↔Leptin ↑↔Adiponectin ↑↓Chemerin ↓↔Visfatin ↓↑Omentin ↑↔Vaspin ↔↑Progranulin ↔↑CTRP-4 | ↓Activin-E ↑ANGPTL3 ↑ANGPTL4 ↓ANGPTL6 ↓Fetuin-A ↔FGF21 ↔Follistatin ↔GDF15 ↑Hepassocin ↔IGF1 ↔LECT2 ↓Lipocalin 13 ↔Selenoprotein-P ↔Tsukushi | |||
| TRX | (L/M) | (L/M) | (L/M) | (L/M) | Low- to moderate-intensity TRX training can positively change some myokines, resulting in improved muscle strength and reduced inflammation. However, no studies were found in connection with high-intensity resistance training. Moreover, TRX training with low to moderate intensity can probably lead to a decrease in inflammation or an increase in metabolism and an improvement in the functioning of the immune system, with positive changes in adipokines; however, the results are limited and no study was found concerning high intensity. | [111,112,113,114,115] |
| ↓myostatin ↔TGF-β1 ↔IL-6 ↔IL-15 ↔decorin ↑irisin ↔BDNF ↔IL-15 ↑FGF ↔SPARC | N/A | ↓Leptin ↑Adiponectin ↑Chemerin ↓Visfatin ↓Omentin ↑Vaspin ↔Progranulin ↔CTRP-4 | N/A | |||
| H | H | H | H | |||
| N/A | N/A | Leptin? Adiponectin? Chemerin? Visfatin? Omentin? Vaspin? Progranulin? CTRP-4? | N/A | |||
| Yoga | (L/M) | (L/M) | (L/M) | (L/M) | No study focusing on exerkines was found. However, studies showed that practicing yoga can lead to a decrease in inflammatory cytokines and an increase in anti-inflammatory cytokines. However, it is impossible to conclude whether yoga focusing on exerkines is suitable. | [116,117,118,119,120] |
| N/A | N/A | N/A | N/A | |||
| H | H | H | H | |||
| N/A | N/A | N/A | N/A | |||
| Swimming | (L) | (L) | (L/M) | (L) | Low-intensity swimming training seems not to affect myokines, but moderate to intense training shows positive changes that can reduce inflammation and increase muscle strength. Concerning adipokines, low- to moderate-intensity swimming training can cause positive changes in adipokines and reduce inflammation and improve immune system function. Nevertheless, it seems like this concerns low intensity. No study was found in connection with other exerkines, but some studies have reported improved cardiovascular function and improved metabolism. | [121,122,123,124,125,126,127,128,129,130] |
| ↔myostatin ↔TGF-β1 ↓IL-6 ↓IL-15 ↔decorin ↔irisin ↔BDNF ↔IL-15 ↔FGF ↔SPARC | N/A | ↓Leptin ↑Adiponectin ↑Chemerin ↓Visfatin ↓Omentin ↑Vaspin ↔Progranulin ↔CTRP-4 | N/A | |||
| (H/M) | (H/M) | (H) | (H/M) | |||
| ↑myostatin ↓TGF-β1 ↓IL-6 ↓IL-15 ↑decorin ↑irisin ↑BDNF ↑IL-15 ↑FGF ↔SPARC | N/A | Leptin↑ Adiponectin↓Chemerin↑ Visfatin↑ Omentin↑ Vaspin↑ Progranulin↑ CTRP-4↑ | N/A | |||
| Walking OR running | (L/M) | (L/M) | (L/M) | (L/M) | Walking with low to moderate intensity (running) positively affects some myokines, reducing inflammation and, sometimes, increasing muscle hypertrophy. However, in intense running, negative changes in myokines can lead to increased inflammation. The association between cardiokines and low- to moderate-intensity walking suggests that changes in some cardiokines can lead to lower blood pressure and, in some cases, reduced inflammation. It also improves cardiovascular endurance. However, with high intensity, there are few results and, in some cases, an increase in inflammation is seen. In connection with walking with low to moderate intensity (running) and adipokines and hepatokines, it seems that the best results occur and significant changes are seen in adipokines and hepatokines, the results of which are reducing inflammation, improving the function of the immune system, increasing metabolism, and increasing insulin sensitivity. | [78,131,132,133,134,135,136,137,138,139,140,141,142] |
| ↑myostatin ↓TGF-β1 ↓IL-6 ↓IL-15 ↔decorin ↔irisin ↑BDNF ↑IL-15 ↔FGF ↔SPARC | ↓ANP ↓BNP ↑IL-33 ↓IL-6 ↑IL-18 ↓IL-1β ↓Follistatin ↑FGF ↓Sfrp ↑Neurotrophins ↓TNF-α ↓TGF-β | ↓Leptin ↑Adiponectin ↑Chemerin ↓Visfatin ↓Omentin ↑Vaspin ↔Progranulin ↔CTRP-4 | ↑Activin-E ↔ANGPTL3 ↔ANGPTL4 ↑ANGPTL6 ↑Fetuin-A ↑FGF21 ↓Follistatin ↑GDF15 ↓Hepassocin ↑IGF1 ↓LECT2 ↑Lipocalin 13 ↑Selenoprotein-P ↑Tsukushi | |||
| H | H | H | H | |||
| ↓myostatin ↑TGF-β1 ↑IL-6 ↑IL-15 ↓decorin ↓irisin ↔BDNF ↓IL-15 ↓FGF ↔SPARC | ↔? ANP ↔? BNP ↓IL-33 ↑IL-6 ↓IL-18 ↔↑IL-1β ↔Follistatin ↔FGF ↓Sfrp ↑Neurotrophins ↑TNF-α ↑TGF-β | Leptin↑ Adiponectin↓ Chemerin↑ Visfatin↑ Omentin↑ Vaspin↑ Progranulin↑ CTRP-4↑ | ↓Activin-E ↑ANGPTL3 ↑ANGPTL4 ↓ANGPTL6 ↓Fetuin-A ↓FGF21 ↑Follistatin ↔GDF15 ↑Hepassocin ↓IGF1 ↓LECT2 ↓Lipocalin 13 ↔Selenoprotein-P ↔Tsukushi | |||
| Cycling | (L/M) | (L/M) | (L/M) | (L/M) | Cycling with low to moderate intensity can increase the strength of lower body muscles and reduce inflammation by changing myokines, which cannot be seen at high intensity. Moreover, low- to moderate-intensity cycling can lead to increased muscular endurance, cardiovascular endurance, blood pressure, and cardiac output. These results were not seen at high intensity and increased inflammation occurs at high intensity. Low- to moderate-intensity cycling focusing on adipokines can reduce inflammation and improve immune function. No study was found in connection with high intensity and hepatokines changes. | [99,135,143,144,145] |
| ↓myostatin ↔TGF-β1 ↓IL-6 ↓IL-15 ↑decorin ↑irisin ↑BDNF ↑IL-15 ↑FGF ↑SPARC | ↓ANP ↓BNP ↑IL-33 ↓IL-6 ↑IL-18 ↓IL-1β ↓Follistatin ↑FGF ↓Sfrp ↑Neurotrophins ↓TNF-α ↓TGF-β | ↓Leptin ↑Adiponectin ↑Chemerin ↓Visfatin ↓Omentin ↑Vaspin ↔Progranulin ↔CTRP-4 | N/A | |||
| H | H | H | H | |||
| ↓myostatin ↑TGF-β1 ↑IL-6 ↑IL-15 ↓decorin ↓irisin ↔BDNF ↓IL-15 ↓FGF ↔SPARC | ↔? ANP ↔? BNP ↓IL-33 ↑IL-6 ↓IL-18 ↔↑IL-1β ↔Follistatin ↔FGF ↓Sfrp ↑Neurotrophins ↑TNF-α ↑TGF-β | N/A | N/A | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Suzuki, K.; Hekmatikar, A.H.A.; Jalalian, S.; Abbasi, S.; Ahmadi, E.; Kazemi, A.; Ruhee, R.T.; Khoramipour, K. The Potential of Exerkines in Women’s COVID-19: A New Idea for a Better and More Accurate Understanding of the Mechanisms behind Physical Exercise. Int. J. Environ. Res. Public Health 2022, 19, 15645. https://doi.org/10.3390/ijerph192315645
Suzuki K, Hekmatikar AHA, Jalalian S, Abbasi S, Ahmadi E, Kazemi A, Ruhee RT, Khoramipour K. The Potential of Exerkines in Women’s COVID-19: A New Idea for a Better and More Accurate Understanding of the Mechanisms behind Physical Exercise. International Journal of Environmental Research and Public Health. 2022; 19(23):15645. https://doi.org/10.3390/ijerph192315645
Chicago/Turabian StyleSuzuki, Katsuhiko, Amir Hossein Ahmadi Hekmatikar, Shadi Jalalian, Shaghayegh Abbasi, Elmira Ahmadi, Abdolreza Kazemi, Ruheea Taskin Ruhee, and Kayvan Khoramipour. 2022. "The Potential of Exerkines in Women’s COVID-19: A New Idea for a Better and More Accurate Understanding of the Mechanisms behind Physical Exercise" International Journal of Environmental Research and Public Health 19, no. 23: 15645. https://doi.org/10.3390/ijerph192315645
APA StyleSuzuki, K., Hekmatikar, A. H. A., Jalalian, S., Abbasi, S., Ahmadi, E., Kazemi, A., Ruhee, R. T., & Khoramipour, K. (2022). The Potential of Exerkines in Women’s COVID-19: A New Idea for a Better and More Accurate Understanding of the Mechanisms behind Physical Exercise. International Journal of Environmental Research and Public Health, 19(23), 15645. https://doi.org/10.3390/ijerph192315645









