Radiation-Induced Recurrent Vesicovaginal Fistula—Treatment with Adjuvant Platelet-Rich Plasma Injection and Martius Flap Placement—Case Report and Review of Literature
Abstract
1. Introduction
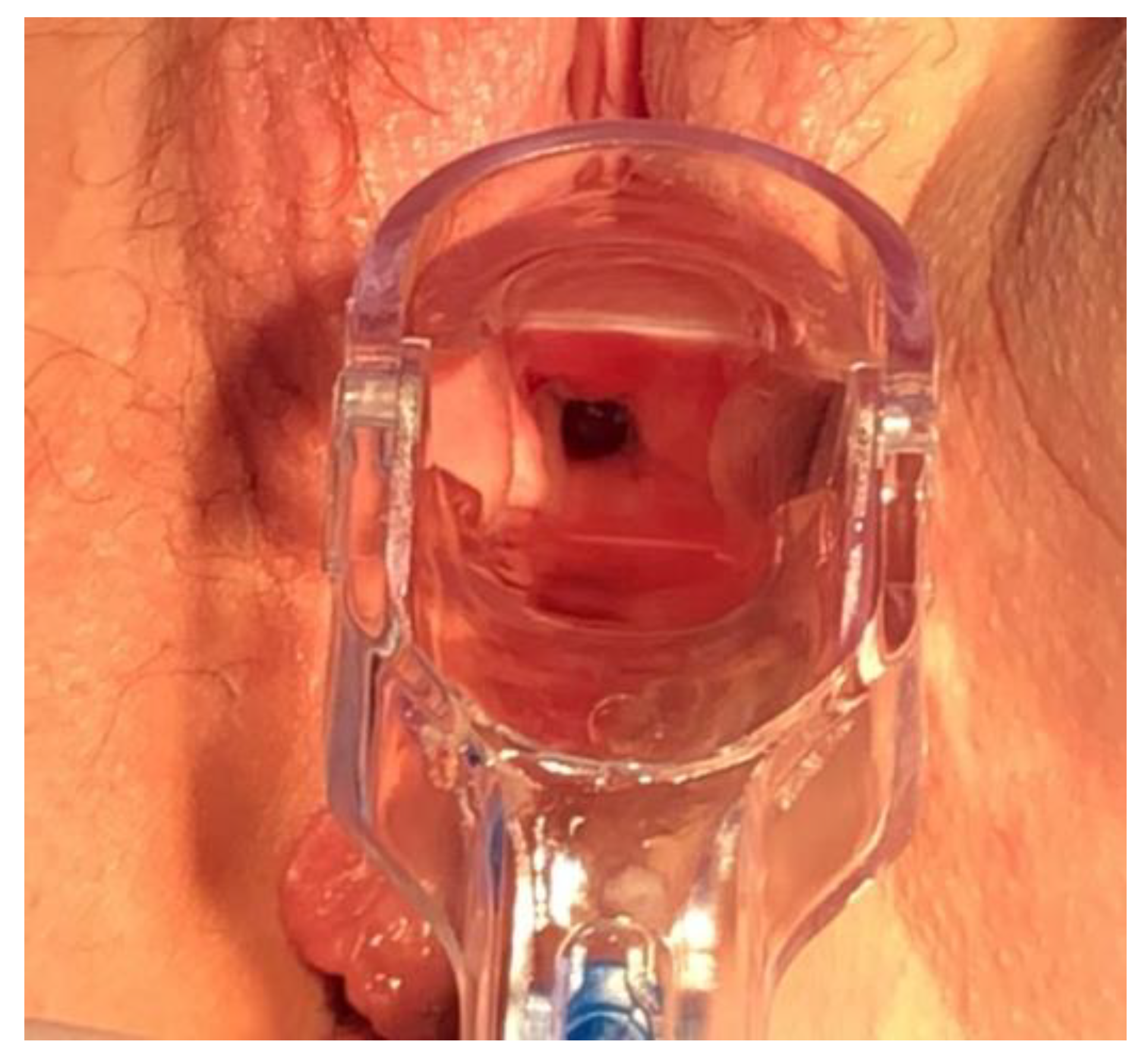
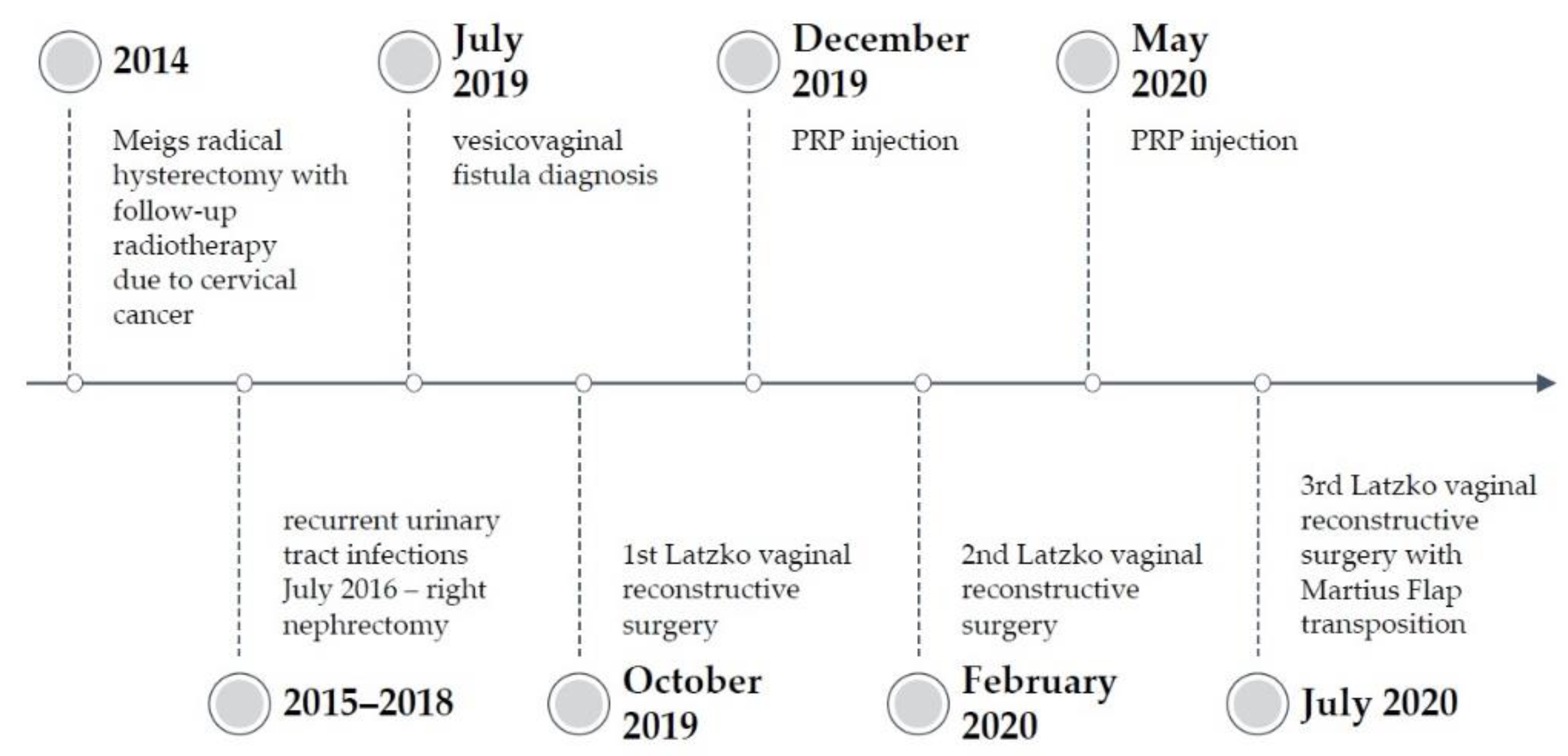
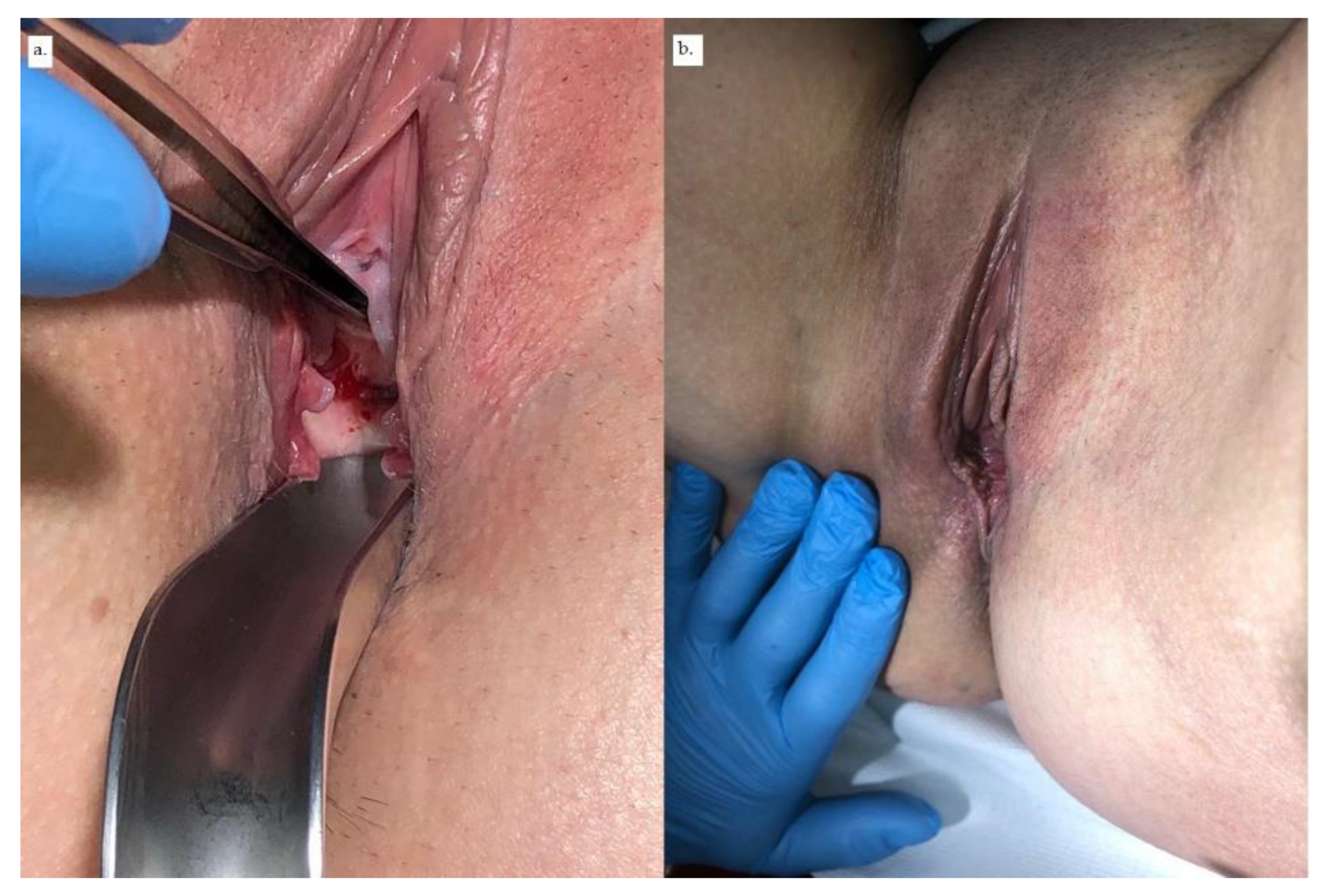
2. Case Report
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Abrams, P.; Cardozo, L.; Khoury, S.; Wein, A. Incontinence, 5th ed.; International Consultation on Urological Diseases: Bristol, UK, 2013.
- Harkki-Siren, P.; Sjoberg, J.; Tiitinen, A. Urinary Tract Injuries after Hysterectomy. Obstet. Gynecol. Surv. 1998, 92, 113–118. [Google Scholar] [CrossRef]
- Stamatakos, M.; Sargedi, C.; Stasinou, T.; Kontzoglou, K. Vesicovaginal Fistula: Diagnosis and Management. Indian J. Surg. 2014, 76, 131–136. [Google Scholar] [CrossRef]
- Streit–Ciećkiewicz, D.; Nowakowski, Ł.; Grzybowska, M.E.; Futyma, K. Predictive Value of Classification Systems and Single Fistula-Related Factors in Surgical Management of Vesicovaginal Fistula. Neurourol. Urodyn. 2021, 40, 529–537. [Google Scholar] [CrossRef]
- Maier, U.; Ehrenböck, P.M.; Hofbauer, J. Late Urological Complications and Malignancies after Curative Radiotherapy for Gynecological Carcinomas: A Retrospective Analysis of 10,709 Patients. J. Urol. 1997, 158, 814–817. [Google Scholar] [CrossRef]
- Bhatla, N.; Berek, J.S.; Cuello Fredes, M.; Denny, L.A.; Grenman, S.; Karunaratne, K.; Kehoe, S.T.; Konishi, I.; Olawaiye, A.B.; Prat, J.; et al. Revised FIGO Staging for Carcinoma of the Cervix Uteri. Int. J. Gynecol. Obstet. 2019, 145, 129–135. [Google Scholar] [CrossRef]
- Streit-Ciećkiewicz, D.; Futyma, K.; Miotła, P.; Grzybowska, M.E.; Rechberger, T. Platelet-Rich Plasma as Adjuvant Therapy for Recurrent Vesicovaginal Fistula: A Prospective Case Series. J. Clin. Med. 2019, 8, 2122. [Google Scholar] [CrossRef] [PubMed]
- Moses, R.A.; Ann Gormley, E. State of the Art for Treatment of Vesicovaginal Fistula. Curr. Urol. Rep. 2017, 18, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Kopp, D.M.; Tang, J.H.; Bengtson, A.M.; Chi, B.H.; Moyo, M.; Wilkinson, J. Continence, Quality of Life, and Depression Following Surgical Repair of Obstetric Vesicovaginal Fistula: A Cohort Study. BJOG 2020, 126, 926–934. [Google Scholar] [CrossRef]
- Singh, V.; Mehrotra, S.; Bansal, A.; Akhtar, A.; Sinha, R.J. Prospective Randomized Comparison of Repairing Vesicovaginal Fistula with or without the Interposition Flap: Result from a Tertiary Care Institute in Northern India. Turkish J. Urol. 2019, 45, 377–383. [Google Scholar] [CrossRef] [PubMed]
- Rangnekar, N.P.; Imdad Ali, N.; Kaul, S.A.; Pathak, H.R. Role of the Martins Procedure in the Management of Urinary-Vaginal Fistulas. J. Am. Coll. Surg. 2000, 191, 259–263. [Google Scholar] [CrossRef]
- Ghosh, B.; Wats, V.; Pal, D.K. Comparative Analysis of Outcome between Laparoscopic versus Open Surgical Repair for Vesico-Vaginal Fistula. Obstet. Gynecol. Sci. 2016, 59, 525. [Google Scholar] [CrossRef]
- Lobo, N.; Kulkarni, M.; Hughes, S.; Nair, R.; Khan, M.S.; Thurairaja, R. Urologic Complications Following Pelvic Radiotherapy. Urology 2018, 122, 1–9. [Google Scholar] [CrossRef]
- Pushkar, D.Y.; Dyakov, V.V.; Kasyan, G.R. Management of Radiation-Induced Vesicovaginal Fistula. Eur. Urol. 2009, 55, 131–138. [Google Scholar] [CrossRef]
- Barone, M.A.; Frajzyngier, V.; Ruminjo, J.; Asiimwe, F.; Barry, T.H.; Bello, A.; Danladi, D.; Ganda, S.O.; Idris, S.A.; Inoussa, M.; et al. Determinants of Postoperative Outcomes of Female Genital Fistula Repair Surgery. Obs. Gynecol. 2012, 120, 524–531. [Google Scholar] [CrossRef] [PubMed]
- Bengtson, A.M.; Kopp, D.; Tang, J.H.; Chipungu, E.; Moyo, M.; Wilkinson, J. Identifying Patients with Vesicovaginal Fistula at High Risk of Urinary Incontinence After Surgery. Obs. Gynecol. 2016, 128, 945–953. [Google Scholar] [CrossRef]
- Reiss, R.F.; Oz, M.C. Autologous Fibrin Glue: Production and Clinical Use. Transfus. Med. Rev. 1996, 10, 85–92. [Google Scholar] [CrossRef]
- Sierra, D.H. Fibrin Sealant Adhesive Systems: A Review of Their Chemistry, Material Properties and Clinical Applications. J. Biomater. Appl. 1993, 7, 309–352. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.K.; Perry, K.T.; Turk, T.M.T. Endoscopic Injection of Fibrin Glue for the Treatment of Urinary-Tract Pathology. J. Endourol. 2005, 19, 419–423. [Google Scholar] [CrossRef] [PubMed]
- Lubkowska, A.; Dolegowska, B.; Banfi, G. Growth Factor Content in PRP and Their Applicability in Medicine. J. Biol. Regul. Homeost. Agents 2012, 26(2 Suppl. 1), 3S–22S. [Google Scholar]
- Dawood, A.S.; Salem, H.A. Current Clinical Applications of Platelet-Rich Plasma in Various Gynecological Disorders: An Appraisal of Theory and Practice. Clin. Exp. Reprod. Med. 2018, 45, 67–74. [Google Scholar] [CrossRef]
- Siemińska, L. Adipose tissue. Pathophysiology, distribution, sex differences and the role in inflammation and cancerogenesis. Endokrynol. Polska 2007, 58, 330–342. [Google Scholar]
- Leach, D.A.; Gebhart, J.B. Martius labial fat pad graft (use in rectovaginal fistula repair). Int. Urogynecol. J. 2020. [CrossRef] [PubMed]
- Kasyan, G.; Tupikina, N.; Pushkar, D. Use of Martius flap in the complex female urethral surgery. Cent. Eur. J. Urol. 2014, 67, 202–207. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kołodyńska, A.; Streit-Ciećkiewicz, D.; Kot, A.; Kuliniec, I.; Futyma, K. Radiation-Induced Recurrent Vesicovaginal Fistula—Treatment with Adjuvant Platelet-Rich Plasma Injection and Martius Flap Placement—Case Report and Review of Literature. Int. J. Environ. Res. Public Health 2021, 18, 4867. https://doi.org/10.3390/ijerph18094867
Kołodyńska A, Streit-Ciećkiewicz D, Kot A, Kuliniec I, Futyma K. Radiation-Induced Recurrent Vesicovaginal Fistula—Treatment with Adjuvant Platelet-Rich Plasma Injection and Martius Flap Placement—Case Report and Review of Literature. International Journal of Environmental Research and Public Health. 2021; 18(9):4867. https://doi.org/10.3390/ijerph18094867
Chicago/Turabian StyleKołodyńska, Aleksandra, Dominika Streit-Ciećkiewicz, Agata Kot, Iga Kuliniec, and Konrad Futyma. 2021. "Radiation-Induced Recurrent Vesicovaginal Fistula—Treatment with Adjuvant Platelet-Rich Plasma Injection and Martius Flap Placement—Case Report and Review of Literature" International Journal of Environmental Research and Public Health 18, no. 9: 4867. https://doi.org/10.3390/ijerph18094867
APA StyleKołodyńska, A., Streit-Ciećkiewicz, D., Kot, A., Kuliniec, I., & Futyma, K. (2021). Radiation-Induced Recurrent Vesicovaginal Fistula—Treatment with Adjuvant Platelet-Rich Plasma Injection and Martius Flap Placement—Case Report and Review of Literature. International Journal of Environmental Research and Public Health, 18(9), 4867. https://doi.org/10.3390/ijerph18094867





