Identifying Prenatal and Postnatal Determinants of Infant Growth: A Structural Equation Modelling Based Cohort Analysis
Abstract
1. Introduction
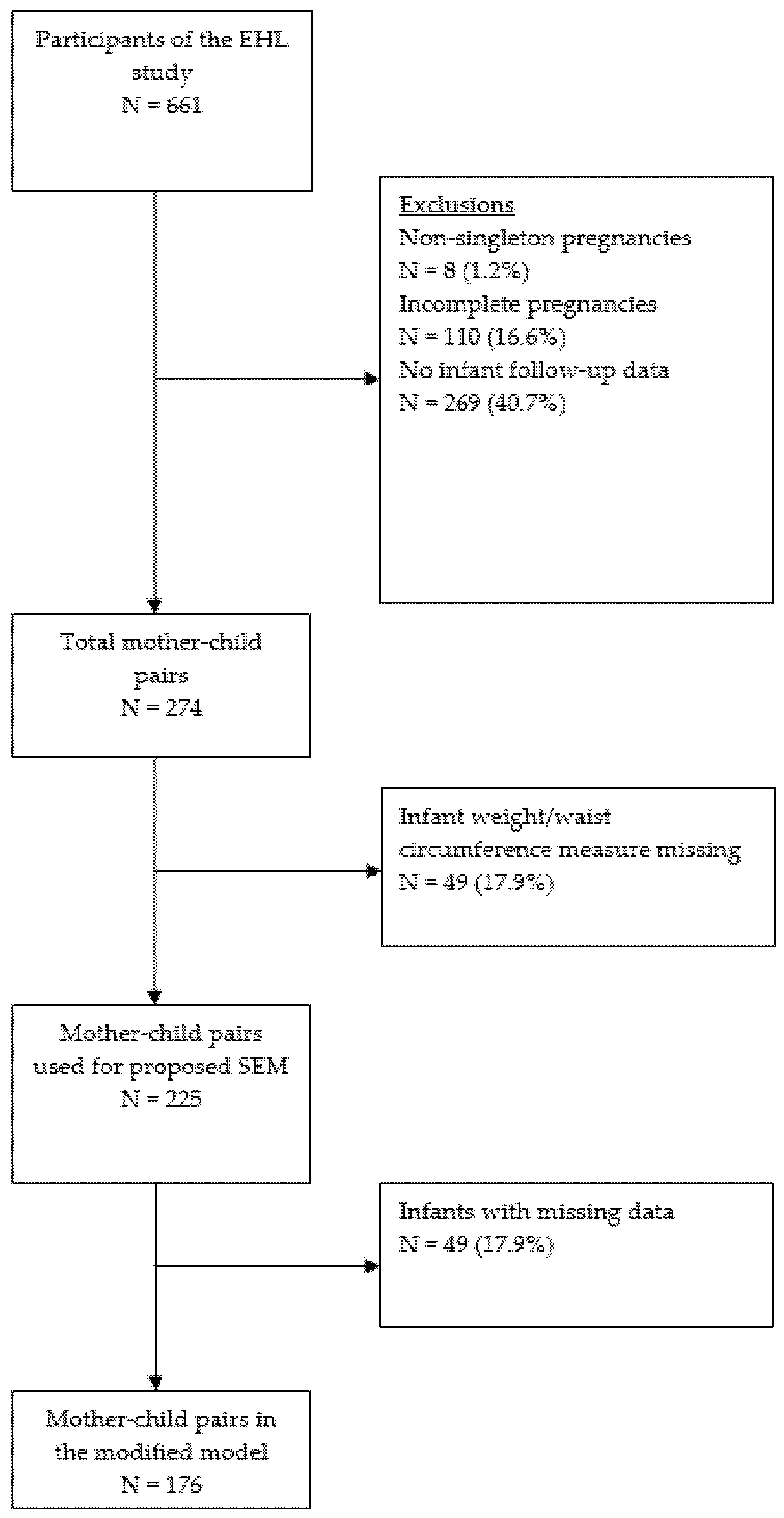
2. Materials and Methods
2.1. Study Population
2.2. Studied Variables
2.3. Statistical Analyses
3. Results
3.1. Description of the Data
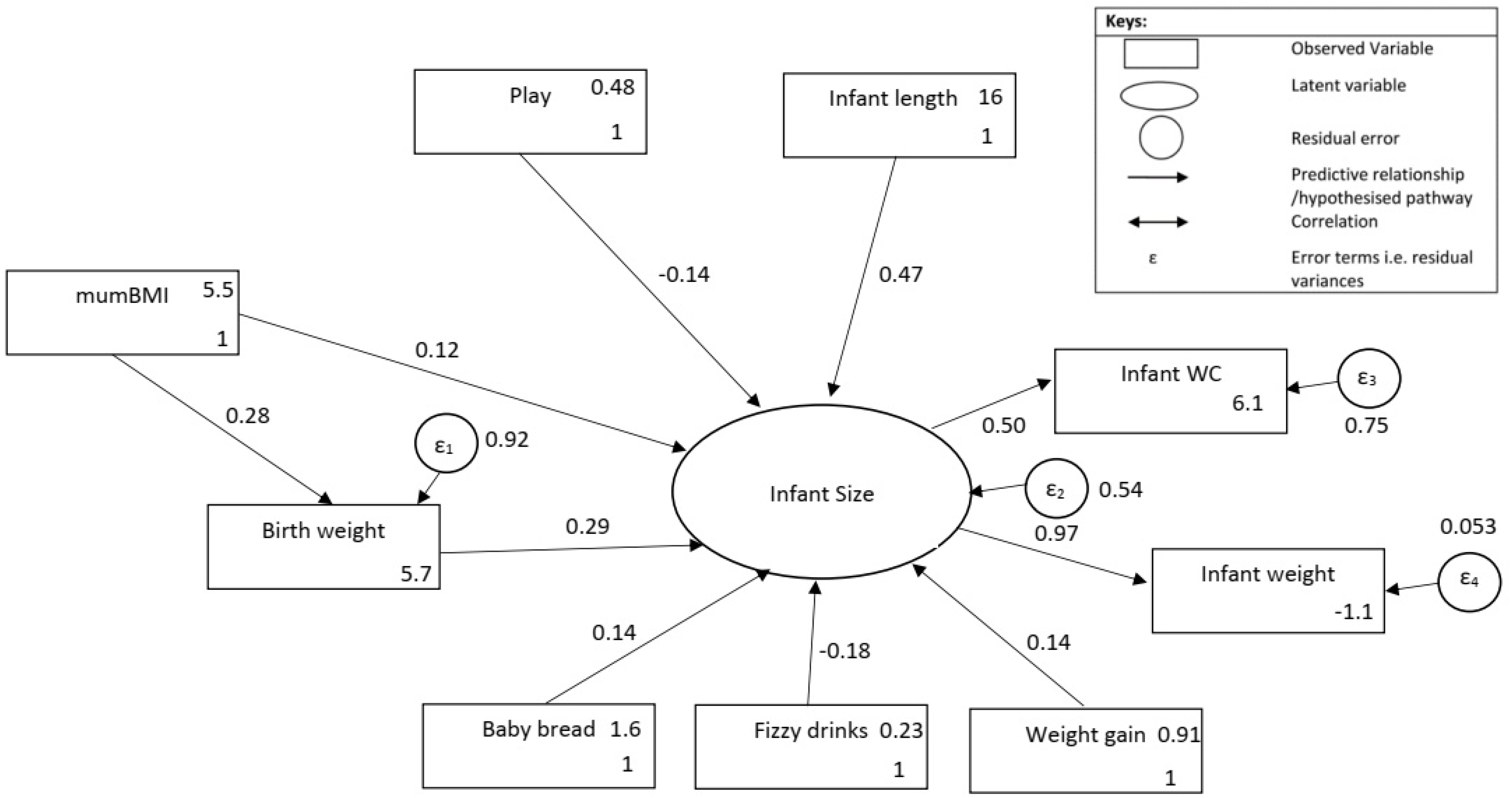
3.2. Direct, Indirect and Total Effects from the SEM
3.3. Modelling by Gender and Imputing Data
4. Discussion
4.1. Prenatal Exposures
4.2. Postnatal Growth
4.3. Postnatal Exposures
4.4. Strengths and Limitations
4.5. Implications
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Fuemmeler, B.F.; Wang, L.; Iversen, E.S.; Maguire, R.; Murphy, S.K.; Hoyo, C. Association between Prepregnancy Body Mass Index and Gestational Weight Gain with Size, Tempo, and Velocity of Infant Growth: Analysis of the Newborn Epigenetic Study Cohort. Child Obes. 2016. [Google Scholar] [CrossRef]
- Martorell, R. Improved nutrition in the first 1000 days and adult human capital and health. Am. J. Hum. Biol. 2017. [Google Scholar] [CrossRef] [PubMed]
- Walson, J.L.; Berkley, J.A. The impact of malnutrition on childhood infections. Curr. Opin. Infect. Dis. 2018. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, M.K.; Zambruni, M.; Melby, C.L.; Melby, P.C. Impact of childhood malnutrition on host defense and infection. Clin Microbiol. Rev. 2017. [Google Scholar] [CrossRef] [PubMed]
- Wright, C.M.; Marryat, L.; McColl, J.; Harjunmaa, U.; Cole, T.J. Pathways into and out of overweight and obesity from infancy to mid-childhood. Pediatr. Obes. 2018. [Google Scholar] [CrossRef] [PubMed]
- Campbell, M.K. Biological, environmental, and social influences on childhood obesity. Pediatr. Res. 2016. [Google Scholar] [CrossRef]
- Holdsworth, E.A.; Schell, L.M. Maternal-infant interaction as an influence on infant adiposity. Am. J. Hum. Biol. 2017. [Google Scholar] [CrossRef]
- Hill, R.A.; Brophy, S.; Brunt, H.; Storey, M.; Thomas, N.E.; Thornton, C.A.; Palmer, S.; Dunstan, F.; Paranjothy, S.; McClure, R.; et al. Protocol of the baseline assessment for the Environments for Healthy Living (EHL) Wales cohort study. BMC Public Health 2010, 10, 150. [Google Scholar] [CrossRef]
- Hernandez, L.; Blazer, D. Committee on Assessing Interactions among Social Behavioral and Genetic Factors in Health; Hernandez, L., Blazer, D., Eds.; The National Academies Press: Washington, DC, USA, 2006. [Google Scholar]
- Kline, R.B. Principles and Practice of Structural Equation Modeling, 2nd ed.; The Guilford Press: New York, NY, USA, 2015. [Google Scholar]
- Kaakinen, M.; Sovio, U.; Hartikainen, A.-L.; Pouta, A.; Savolainen, M.J.; Herzig, K.-H.; Elliott, P.; De Stavola, B.L.; Läärä, E.; Jarvelin, M.-R. Life course structural equation model of the effects of prenatal and postnatal growth on adult blood pressure. J. Epidemiol. Community Health 2014, 68, 1161–1167. [Google Scholar] [CrossRef]
- Barker, D.J. Fetal origins of coronary heart disease. BMJ 1995, 311, 171–174. [Google Scholar] [CrossRef]
- Raza, H.; Zhou, S.; Todd, C.; Christian, D.; Marchant, E.; Morgan, K.; Khanom, A.; Hill, R.; Lyons, R.A.; Brophy, S. Predictors of objectively measured physical activity in 12-month-old infants: A study of linked birth cohort data with electronic health records. Pediatr. Obes. 2019, e12512. [Google Scholar] [CrossRef]
- Lyons, R.A.; Jones, K.H.; John, G.; Brooks, C.J.; Verplancke, J.-P.; Ford, D.V.; Brown, G.; Leake, K. The SAIL databank: Linking multiple health and social care datasets. BMC Med. Inform. Dec. Mak. 2009, 9, 3. [Google Scholar] [CrossRef]
- Damm, P.; Houshmand-Oeregaard, A.; Kelstrup, L.; Lauenborg, J.; Mathiesen, E.R.; Clausen, T.D. Gestational diabetes mellitus and long-term consequences for mother and offspring: A view from Denmark. Diabetologia 2016. [Google Scholar] [CrossRef] [PubMed]
- Activinsights. Activinsights Ltd: GENEActiv Instructions. Available online: https://www.activinsights.com/wp-content/uploads/2014/03/geneactiv_instruction_manual_v1.2.pdf (accessed on 20 August 2021).
- Zhou, S.-M.; A Hill, R.; Morgan, K.; Stratton, G.; Gravenor, M.; Bijlsma, G.; Brophy, S. Classification of accelerometer wear and non-wear events in seconds for monitoring free-living physical activity. BMJ Open 2015, 5. [Google Scholar] [CrossRef] [PubMed]
- Hager, E.R.; Gormley, C.E.; Latta, L.W.; Treuth, M.S.; Caulfield, L.E.; Black, M.M. Toddler physical activity study: Laboratory and community studies to evaluate accelerometer validity and correlates. BMC Public Health 2016, 16. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, M.D.; Dwyer, T.; Magnussen, C.G.; Venn, A.J. Predictive associations between alternative measures of childhood adiposity and adult cardio-metabolic health. Int. J. Obes. 2011. [Google Scholar] [CrossRef] [PubMed]
- Shastry, C.; Bhat, B. Anthropometric measurements of newborns. Int. J. Contemp. Pediatr. 2015. [Google Scholar] [CrossRef]
- Hu, L.; Bentler, P. Cutoff criteria for fit indexes in covariance structure analysis: Conventional criteria versus new alternatives. Struct. Equ. Model. A Multidiscip. J. 1999, 6, 1–55. [Google Scholar] [CrossRef]
- Davis, C.; Levitan, R.D.; Smith, M.; Tweed, S.; Curtis, C. Associations among overeating, overweight, and attention deficit/hyperactivity disorder: A structural equation modelling approach. Eat. Behav. 2006, 7, 266–274. [Google Scholar] [CrossRef]
- Linabery, A.; Nahhas, R.W.; Johnson, W.; Choh, A.C.; Towne, B.; Odegaard, A.O.; Czerwinski, S.A.; Demerath, E.W. Stronger influence of maternal than paternal obesity on infant and early childhood body mass index: The Fels Longitudinal Study. Pediatr. Obes. 2013. [Google Scholar] [CrossRef]
- Birch, L.L.; Fisher, J.O. Mothers’ child-feeding practices influence daughters’ eating and weight. Am. J. Clin. Nutr. 2000, 71, 1054–1061. [Google Scholar] [CrossRef] [PubMed]
- Sallis, J.F.; Cutter, C.L.; Lou, D.; Spoon, C.; Wilson, A.L.; Ding, D.; Ponkshe, P.; Cervero, R.; Patrick, K.; Schmid, T.L.; et al. Active Living Research: Creating and using evidence to support childhood obesity prevention. Am. J. Prev. Med. 2014, 46, 195–207. [Google Scholar] [CrossRef]
- Robinson, S.; Marriott, L.; Poole, J.; Crozier, S.; Borland, S.; Lawrence, W.; Law, C.; Godfrey, K.; Cooper, C.; Inskip, H.; et al. Dietary patterns in infancy: The importance of maternal and family influences on feeding practice. Br. J. Nutr. 2007, 98, 1029–1037. [Google Scholar] [CrossRef] [PubMed]
- Cooke, L.J.; Wardle, J.; Gibson, E.L.; Sapochnik, M.; Sheiham, A.; Lawson, M. Demographic, familial and trait predictors of fruit and vegetable consumption by pre-school children. Public Health Nutr. 2004, 7, 295–302. [Google Scholar] [CrossRef] [PubMed]
- Johnson, L.; Llewellyn, C.; Van Jaarsveld, C.H.; Cole, T.; Wardle, J. Genetic and Environmental Influences on Infant Growth: Prospective Analysis of the Gemini Twin Birth Cohort. PLoS ONE 2011, 6, e19918. [Google Scholar] [CrossRef]
- Ong, K.K.; Loos, R.J. Rapid infancy weight gain and subsequent obesity: Systematic reviews and hopeful suggestions. Acta Paediatr. 2006, 95, 904–908. [Google Scholar] [CrossRef]
- Durmus, B.; Ay, L.; Duijts, L.; Moll, H.A.; Hokken-Koelega, A.C.S.; Raat, H.; Hofman, A.; Steegers, E.A.P.; Jaddoe, V.W.V. Infant diet and subcutaneous fat mass in early childhood. Eur. J. Clin. Nutr. 2012, 66, 253–260. [Google Scholar] [CrossRef]
- Botton, J.; Heude, B.; Maccario, J.; Ducimetière, P.; Charles, M.A.; FLVS Study Group. Postnatal weight and height growth velocities at different ages between birth and 5 y and body composition in adolescent boys and girls. Am. J. Clin. Nutr. 2008, 87, 1760–1768. [Google Scholar] [CrossRef]
- Aalinkeel, R.; Srinivasan, M.; Kalhan, S.C.; Laychock, S.G.; Patel, M.S. A dietary intervention (high carbohydrate) during the neonatal period causes islet dysfunction in rats. Am. J. Physiol. Endocrinol. Metab. 1999, 277, E1061–E1069. [Google Scholar] [CrossRef]
- Skinner, J.D.; Ziegler, P.; Ponza, M. Transitions in infants’ and toddlers’ beverage patterns. J. Am. Diet. Assoc. 2004, 104 (Suppl. 1), s45–s50. [Google Scholar] [CrossRef]
- Fulgoni, V.; Quann, E. National trends in beverage consumption in children from birth to 5 years: Analysis of NHANES across three decades. Nutr. J. 2012, 11, 92. [Google Scholar] [CrossRef]
- Newby, P.; Peterson, K.E.; Berkey, C.S.; Leppert, J.; Willett, W.C.; Colditz, G.A. Beverage consumption is not associated with changes in weight and body mass index among low-income preschool children in North Dakota. J. Am. Diet. Assoc. 2004, 104, 1086–1094. [Google Scholar] [CrossRef]
- Welsh, J.A.; Cogswell, M.E.; Rogers, S.; Rockett, H.; Mei, Z.; Grummer-Strawn, L.M. Overweight among low-income preschool children associated with the consumption of sweet drinks: Missouri. Pediatrics 2005, 115, e223–e229. [Google Scholar] [CrossRef]
- Macintyre, A.K.; Marryat, L.; Chambers, S. Exposure to liquid sweetness in early childhood: Artificially-sweetened and sugar-sweetened beverage consumption at 4–5 years and risk of overweight and obesity at 7–8 years. Pediatr. Obes. 2018. [Google Scholar] [CrossRef]
- Fox, M.K.; Pac, S.; Devaney, B.; Jankowski, L. Feeding infants and toddlers study: What foods are infants and toddlers eating? J. Am. Diet. Assoc. 2004, 104, 22–30. [Google Scholar] [CrossRef]
- Slemenda, C.; Miller, J.; Hui, S.; Teresa, K.R.; Conrad, C.J., Jr. Role of physical activity in the development of skeletal mass in children. J. Bone Min. Res. 1991, 6, 1227–1233. [Google Scholar] [CrossRef] [PubMed]
- Davies, P.S.; Gregory, J.; White, A. Physical activity and body fatness in pre-school children. Int. J. Obes. Relat. Metab. Disord. 1995, 19, 6–10. [Google Scholar] [PubMed]
- Rodríguez-Rodríguez, I.; Rodríguez, J.V.; Woo, W.L.; Wei, B.; Pardo-Quiles, D.J. A comparison of feature selection and forecasting machine learning algorithms for predicting glycaemia in type 1 diabetes mellitus. Appl. Sci. 2021, 11, 1742. [Google Scholar] [CrossRef]
- Koh, B.H.D.; Lim, C.L.P.; Rahimi, H.; Woo, W.L.; Gao, B. Deep temporal convolution network for time series classification. Sensors 2021, 21, 603. [Google Scholar] [CrossRef]
| Samples Available | Mean (SD) or % | |
|---|---|---|
| N | ||
| Maternal characteristics | ||
| Socio-demographics during pregnancy | ||
| ge at delivery (years) | 264 | 30.9 (5.6) |
| White ethnicity | 270 | 89.5% |
| First or higher degree attained | 201 | 50.2% |
| Household income <£15,000 | 238 | 21.0% |
| Full time employment | 267 | 43.1% |
| Smoked during pregnancy | 259 | 15.8% |
| Reproductive factors | ||
| Pre-pregnancy BMI (kg/m2) | 274 | 24.4 (4.7) |
| Healthy weight | 149 | 54.4% |
| Overweight | 89 | 32.5% |
| Obese | 36 | 13.1% |
| Number of children <1 | 274 | 83.10% |
| Socio-demographics at 12 months | ||
| Household income <£15,000 | 226 | 20.8% |
| Full time employment | 223 | 25.6% |
| Infant characteristics | ||
| Male | 122 | 52.6% |
| Mean birth weight (kg) | 274 | 3.4 (0.5) |
| % <2.5 kg | 5 | 1.5% |
| % >4.0 kg | 37 | 13.5% |
| Mean gestation (weeks) | 225 | 39.6 (1.5) |
| % born <37 weeks | 8 | 3.6 |
| % born ≥42 weeks | 12 | 5.3 |
| Mean weight gain 0–6 months (per week) | 187 | 0.14 (0.04) |
| % Breastfed at birth | 228 | 84.2% |
| % Breastfed at age 12 months | 227 | 20.4% |
| Mean Weight at 12 months (kg) | 270 | 9.8 (1.2) |
| Parameter Estimate | Modified Model | Full Model | Gender Specific | ||
|---|---|---|---|---|---|
| Unstandardised | Standardised | Males | Females | ||
| N | 176 | 176 | 225 | 97 | 79 |
| Structural Model | |||||
| Pre-pregnancy BMI ≥ birth weight | 0.03 (0.01) | 0.28 (0.07) *** | 0.26 (0.06) *** | 0.26 (0.1) * | 0.31 (0.1) * |
| Birth weight ≥ Size | 1.3 (0.34) | 0.29 (0.07) *** | 0.32 (0.05) *** | 0.36 (0.09) * | 0.22 (0.1) * |
| Infant length ≥ Size | 0.19 (0.05) | 0.47 (0.06) *** | 0.38 (0.06) *** | 0.52 (0.1) * | 0.4 (0.1) * |
| Weight gain ≥ Size | 0.73 (0.27) | 0.14 (0.07) ** | 0.22 (0.06) *** | 0.12 (0.1) * | 0.12 (0.1) |
| Play ≥ Size | −0.72 (0.36) | −0.14 (0.08) ** | -0.09 (0.06) | −0.14 (0.15) * | −0.15 (0.1) * |
| Infant carbohydrate consumption ≥ Size | 0.65 (0.31) | 0.14 (0.08) ** | 0.15 (0.06) * | 0.28 (0.01) ** | 0.01 (0.09) * |
| Infant fizzy drink consumption ≥ Size | −1.66(0.64) | −0.18 (0.08) *** | −0.16 (0.06) * | −0.17 (0.1) * | −0.25 (0.1) * |
| Pre-pregnancy BMI ≥ Size | 0.06 (0.03) | 0.12 (0.08) * | 0.18 (0.07) *** | 0.1 (0.1) | 0.15 (0.1) * |
| Measurement model | |||||
| Size ≥ Infant waist circumference | 1 | 0.5 (0.09) *** | 0.54 (1.10) *** | 0.49 (0.08) *** | 0.61 (0.09) *** |
| Size ≥ Infant weight | 0.52 (0.10) | 0.97 (0.12) *** | 0.97 (0.83) *** | 0.89 (0.11) *** | 0.90 (0.12) *** |
| Fit Indexes | Proposed Model | Modified Model | Good Fit Level |
|---|---|---|---|
| X2 (df) | 44.49 (15) ** | 21.5(11) * | Close to 0 (p < 0.05) |
| RMSEA | 0.11 | 0.07 | <0.07 |
| CFI | 0.84 | 0.94 | >0.90 |
| SRMR | 0.06 | 0.05 | <0.08 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Morgan, K.; Zhou, S.-M.; Hill, R.; Lyons, R.A.; Paranjothy, S.; Brophy, S.T. Identifying Prenatal and Postnatal Determinants of Infant Growth: A Structural Equation Modelling Based Cohort Analysis. Int. J. Environ. Res. Public Health 2021, 18, 10265. https://doi.org/10.3390/ijerph181910265
Morgan K, Zhou S-M, Hill R, Lyons RA, Paranjothy S, Brophy ST. Identifying Prenatal and Postnatal Determinants of Infant Growth: A Structural Equation Modelling Based Cohort Analysis. International Journal of Environmental Research and Public Health. 2021; 18(19):10265. https://doi.org/10.3390/ijerph181910265
Chicago/Turabian StyleMorgan, Kelly, Shang-Ming Zhou, Rebecca Hill, Ronan A. Lyons, Shantini Paranjothy, and Sinead T. Brophy. 2021. "Identifying Prenatal and Postnatal Determinants of Infant Growth: A Structural Equation Modelling Based Cohort Analysis" International Journal of Environmental Research and Public Health 18, no. 19: 10265. https://doi.org/10.3390/ijerph181910265
APA StyleMorgan, K., Zhou, S.-M., Hill, R., Lyons, R. A., Paranjothy, S., & Brophy, S. T. (2021). Identifying Prenatal and Postnatal Determinants of Infant Growth: A Structural Equation Modelling Based Cohort Analysis. International Journal of Environmental Research and Public Health, 18(19), 10265. https://doi.org/10.3390/ijerph181910265








