Using Mobile Applications to Increase Physical Activity: A Systematic Review
Abstract
1. Introduction
2. Materials and Methods
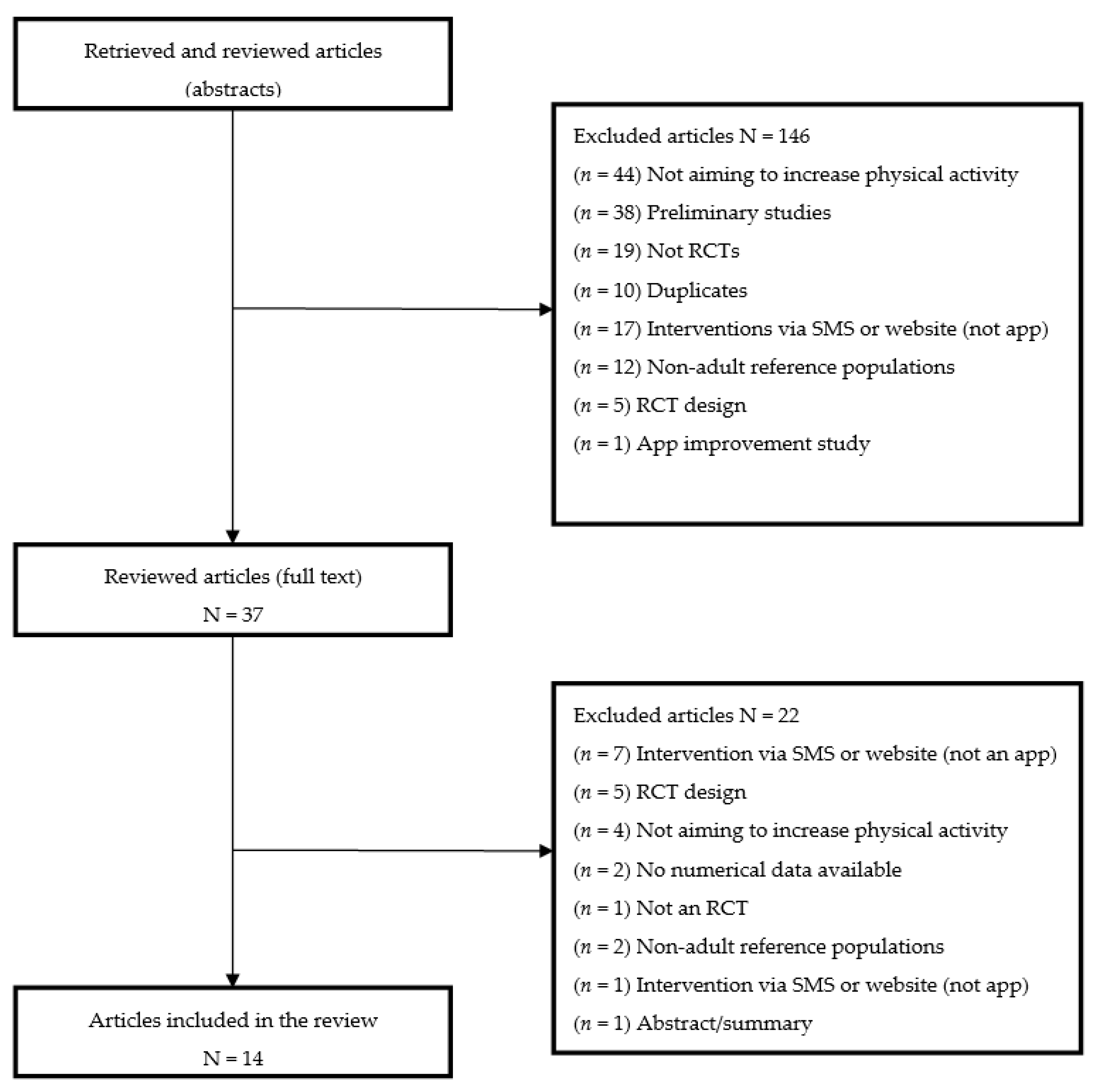
Study Selection
3. Results
3.1. Population and Sample
3.2. Interventions
3.3. Measurements and Results
3.4. Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- World Health Organization. Fact Sheets. Physical Activity. Available online: https://www.who.int/news-room/fact-sheets/detail/physical-activity (accessed on 4 December 2019).
- World Health Organization (Regional Office for Europe). Integrating Diet, Physical Activity and Weight Management Services into Primary Care. 2016. Available online: https://www.euro.who.int/__data/assets/pdf_file/0016/324304/Integrating-diet-physical-activity-weight-management-services-primary-care.pdf (accessed on 31 August 2020).
- Guthold, R.; Stevens, G.A.; Riley, L.M.; Bull, F.C. Worldwide trends in insufficient physical activity from 2001 to 2016: A pooled analysis of 358 population-based surveys with 1·9 million participants. Lancet Glob. Health 2018. [Google Scholar] [CrossRef]
- World Health Organization. Obesity. Data and Statistics. 2018. Available online: http://www.euro.who.int/en/health-topics/disease-prevention/physical-activity/data-and-statistics (accessed on 4 December 2019).
- Research2Guidance. mNutrition. The Impact of Smartphone Apps on the Nutrition Industry. 2015. Available online: https://research2guidance.com/wp-content/uploads/2015/08/mNutrition-2014-Preview.pdf (accessed on 4 December 2019).
- Ditrendia (Digital Marketing Trends). Informe Mobile en España y en el Mundo 2019. Available online: https://mktefa.ditrendia.es/hubfs/Ditrendia-Informe%20Mobile%202019.pdf?utm_campaign=Informe%20Mobile%202019&utm_medium=email&_hsmi=77268109&_hsenc=p2ANqtz-8vyoNQYMsV6iNVrECCOxqAvVOVBKZl_t2nIyAL1D1zWimz3OB3pK5bggQ-jwFWEv9Jt_kwsAc-_7MbgXy2jY4Wb3Mxgw&utm_content=77268109&utm_source=hs_automation (accessed on 3 July 2020).
- Gill, D.P.; Blunt, W.; Bartol, C.; Pulford, R.W.; De Cruz, A.; Simmavong, P.K.; Gavarkovs, A.; Newhouse, I.; Pearson, E.; Ostenfeldt, B.; et al. HealtheStepsTM Study Protocol: A pragmatic randomized controlled trial promoting active living and healthy lifestyles in at-risk Canadian adults delivered in primary care and community-based clinics. BMC Public Health 2017. [Google Scholar] [CrossRef] [PubMed]
- Flores-Mateo, G.; Granado-Font, E.; Ferré-Grau, C.; Montaña-Carreras, X. Mobile Phone Apps to Promote Weight Loss and Increase Physical Activity: A Systematic Review and Meta-Analysis. J. Med. Internet Res. 2015, 17, e253. Available online:https://www.jmir.org/2015/11/e253 (accessed on 25 October 2020). [CrossRef] [PubMed]
- Puigdomènech, E.; Saigí, F.; Zamora, A.; Moharra, M.; Paluzié, G.; Balfegó, M.; Cuatrecasas, G.; Garcia-Lorda, P.; Carrion, C. Assessment of the Efficacy, Safety, and Effectiveness of Weight Control and Obesity Management Mobile Health Interventions: Systematic Review. JMIR Mhealth Uhealth 2019, 7, e12612. [Google Scholar] [CrossRef]
- Glynn, L.G.; Hayes, P.S.; Casey, M.; Glynn, F.; Alvarez-Iglesias, A.; Newell, J.; OLaighin, G.; Heaney, D.; O’Donell, M.; Murphy, A.W. Effectiveness of a smartphone application to promote physical activity in primary care: The SMART MOVE randomised controlled trial. Br. J. Gen. Pract. 2014. [Google Scholar] [CrossRef]
- Romeo, A.; Edney, S.; Plotnikoff, R.; Curtis, R.; Ryan, J.; Sanders, I.; Crozier, A.; Maher, C. Can Smartphone Apps Increase Physical Activity? Systematic Review and Meta-Analysis. J. Med. Internet Res. 2019, 21, e12053. Available online:https://www.jmir.org/2019/3/e12053 (accessed on 25 October 2020). [CrossRef] [PubMed]
- Greenhalgh, T.; Peacock, R. Effectiveness and efficiency of search methods in systematic reviews of complex evidence: Audit of primary sources. Br. Med. J. 2005, 331, 1064–1065. [Google Scholar] [CrossRef] [PubMed]
- Shea, B.J.; Grimshaw, J.M.; Wells, G.A.; Boers, M.; Andersson, N.; Hamel, C.; Porter, A.C.; Tugwell, P.; Moher, D.; Bouter, L.M. Development of AMSTAR: A measurement tool to assess the methodological quality of systematic reviews. BMC Med. Res. Methodol. 2007, 7, 10. [Google Scholar] [CrossRef]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gotzsche, P.C.; Ioannidis, J.P.A.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: Explanation and elaboration. BMJ 2009, 339, b2700. [Google Scholar] [CrossRef]
- Schulz, K.F.; Altman, D.G.; Moher, D. CONSORT 2010 Statement: Updated guidelines for reporting parallel group randomised trials. BMC Med. 2010, 8. [Google Scholar] [CrossRef] [PubMed]
- Ferreira González, I.; Urrútia, G.; Alonso-Coello, P. Revisiones sistemáticas y metaanálisis: Bases conceptuales e interpretación. Rev. Española Cardiol. 2011, 64, 688–696. [Google Scholar] [CrossRef]
- Allen, J.K.; Stephens, J.; Dennison Himmelfarb, C.R.; Stewart, K.J.; Hauck, S. Randomized controlled pilot study testing use of smartphone technology for obesity treatment. J. Obes. 2013. [Google Scholar] [CrossRef]
- Fanning, J.; Roberts, S.; Hillman, C.H.; Mullen, S.P.; Ritterband, L.; McAuley, E. A smartphone “app”-delivered randomized factorial trial targeting physical activity in adults. J. Behav. Med. 2017. [Google Scholar] [CrossRef]
- King, A.C.; Hekler, E.B.; Grieco, L.A.; Winter, S.J.; Sheats, J.L.; Buman, M.P.; Banerjee, B.; Robinson, T.N.; Cirimele, J. Effects of three motivationally targeted mobile device applications on initial physical activity and sedentary behavior change in midlife and older adults: A randomized trial. PLoS ONE 2016. [Google Scholar] [CrossRef]
- Martin, S.S.; Feldman, D.I.; Blumenthal, R.S.; Jones, S.R.; Post, W.S.; McKibben, R.A.; Michos, E.D.; Ndumele, C.E.; Ratchford, E.V.; Coresh, J.; et al. mActive: A randomized clinical trial of an automated mHealth intervention for physical activity promotion. J. Am. Heart Assoc. 2015. [Google Scholar] [CrossRef] [PubMed]
- Mayer, D.K.; Landucci, G.; Awoyinka, L.; Atwood, A.K.; Carmack, C.L. Demark-Wahnefried, W. McTavish, F.; Gustafson, D. SurvivorCHESS to increase physical activity in colon cancer survivors: Can we get them moving? J. Cancer Surviv. 2018. [Google Scholar] [CrossRef] [PubMed]
- Spring, B.; Pellegrini, C.A.; Pfammatter, A.; Duncan, J.M.; Pictor, A.; McFadden, H.G.; Siddique, J.; Hedeker, D. Effects of an abbreviated obesity intervention supported by mobile technology: The ENGAGED randomized clinical trial. Obesity 2017. [Google Scholar] [CrossRef]
- Ashton, L.M.; Morgan, P.J.; Hutchesson, M.J.; Rollo, M.E.; Collins, C.E. Feasibility and preliminary efficacy of the “HEYMAN” healthy lifestyle program for young men: A pilot randomised controlled trial. Nutr. J. 2017. [Google Scholar] [CrossRef]
- Duncan, M.; Vandelanotte, C.; Kolt, G.S.; Rosenkranz, R.R.; Caperchione, C.M.; George, E.S.; Ding, H.; Hooker, C.; Karunanithi, M.; Maeder, A.J.; et al. Effectiveness of a web- and mobile phone-based intervention to promote physical activity and healthy eating in middle-Aged males: Randomized controlled trial of the manup study. J. Med. Internet Res. 2014, 21, b2700. [Google Scholar] [CrossRef]
- Harries, T.; Eslambolchilar, P.; Rettie, R.; Stride, C.; Walton, S.; Van Woerden, H.C. Effectiveness of a smartphone app in increasing physical activity amongst male adults: A randomised controlled trial. BMC Public Health 2016. [Google Scholar] [CrossRef]
- Johnston, N.; Bodegard, J.; Jerström, S.; Åkesson, J.; Brorsson, H.; Alfredsson, J.; Albertsson, P.A.; Karlsson, J.E.; Varenhorst, C. Effects of interactive patient smartphone support app on drug adherence and lifestyle changes in myocardial infarction patients: A randomized study. Am. Heart J. 2016. [Google Scholar] [CrossRef]
- Naimark, J.S.; Madar, Z.; Shahar, D.R. The impact of a Web-based app (eBalance) in promoting healthy lifestyles: Randomized controlled trial. J. Med. Internet Res. 2015. [Google Scholar] [CrossRef]
- Memon, A.R.; Masood, T.; Awan, W.A.; Waqas, A. The effectiveness of an incentivized physical activity programme (Active Student) among female medical students in Pakistan: A Randomized Controlled Trial. J. Pak. Med. Assoc. 2018, 68, 1438–1445. [Google Scholar]
- Valentiner, L.S.; Thorsen, I.K.; Kongstad, M.B.; Brinkløv, C.F.; Larsen, R.T.; Karstoft, K.; Nielsen, J.S.; Pedersen, B.K.; Langberg, H.; Ried-Larsen, M. Effect of ecological momentary assessment, goal-setting and personalized phone-calls on adherence to interval walking training using the InterWalk application among patients with type 2 diabetes-A pilot randomized controlled trial. PLoS ONE 2019, 14, e0208181. [Google Scholar] [CrossRef]
- Ginis, P.; Nieuwboer, A.; Dorfman, M.; Ferrari, A.; Gazit, E.; Canning, C.G.; Rocchi, L.; Chiari, L.; Hausdorff, J.M.; Mirelman, A. Feasibility and effects of home-based smartphone-delivered automated feedback training for gait in people with Parkinson’s disease: A pilot randomized controlled trial. Park Relat. Disord. 2016. [Google Scholar] [CrossRef]
- Reiner, M.; Niermann, C.; Jekauc, D.; Woll, A. Long-term health benefits of physical activity--a systematic review of longitudinal studies. BMC Public Health. 2013, 13, 813. [Google Scholar] [CrossRef]
- Aguilar-Martínez, A.; Solé-Sedeño, J.M.; Mancebo-Moreno, G.; Medina, F.X.; Saigí-Rubió, F. Use of mobile phones as a tool for weight loss: A systematic review. J. Telemed. Telecare 2014, 20, 339–349. [Google Scholar] [CrossRef]
- Stevens, W.; Hillsdon, M.; Thorogood, M.; McArdle, D. Cost-effectiveness of a primary care based physical activity intervention in 45–74 year old men and women: A randomised controlled trial. Br. J. Sports Med. 1998, 32, 236–241. [Google Scholar] [CrossRef]
- Robroek, S.J.; van Lenthe, F.J.; van Empelen, P.; Burdorf, A. Determinants of participation in worksite health promotion programmes: A systematic review. Int. J. Behav. Nutr. Phys. Act 2009, 6, 26. [Google Scholar] [CrossRef]
- Greaves, C.J.; Sheppard, K.E.; Abraham, C.; Greaves, C.J.; Sheppard, K.E.; Abraham, C.; Hardeman, W.; Roden, M.; Evans, P.H.; Schwarz, P.; et al. Systematic review of reviews of intervention components associated with increased effectiveness in dietary and physical activity interventions. BMC Public Health 2011, 11. [Google Scholar] [CrossRef]
| Inclusion Criteria |
|---|
| RCT: use of mobile apps that promote exercise and/or physical activity. Language: English or Spanish. Study population: adults (18 years and older). |
| Exclusion Criteria |
| Opinion articles, editorials, and commentary interpreting published results. Several articles published on the same study by the same authors: the latest study was selected, and all other studies were excluded. Informative material. Interventions based exclusively on websites or SMS use (not on app use). |
| Study | Population and Sample | Objective(s) | Type & Duration | Intervention | Measures | Results | Conclusions |
|---|---|---|---|---|---|---|---|
| Allen [17] | N = 68 Age: 21–65 y BMI: 28–42 USA Women: 78% Means: Age: 44.9 y Weight: 97.3 kg BMI: 34.3 | To evaluate feasibility, acceptability and preliminary efficacy of smartphone-based behavioural interventions | Pilot RCT 6 mo | App-based behavioural interventions plus professional guidance Goals: 5% weight loss and 150 min MVPA Groups:
-Smartphone intervention
-Smartphone intervention
| Start and 6 mo -Weight, height, BMI -PA (7D-PAR) -Estimated energy expenditure -Food intake over 3 d -Diet and PA record monitoring. -Intervention satisfaction (in-depth interviews) | Change at 6 mo PA (mean h/wk) -(IC): −1.4 -(IC+SP): −2.0 -(LIC+SP): −3.6 -(SP): 0.19 BMI -(IC): −0.8 -(IC+SP): 1.8 -(LIC+SP): −1.1 -(SP): −0.7 Weight loss ≥5%: 64% (IC+SP); 40% (LIC+SP) ≤5%: 25; (IC); 20% (SP) Other -Improved diet Retention: 59–69% | -No statistically significant differences between the 4 groups -Satisfaction and improvements: possibility of automatic PA and weight recording -No dropout differences by sex or ethnicity |
| Ashton [23] | N = 50 Age: 18–25 y Men Australia Means: Age: 22.1 y BMI: 25.5 Steps/d: 6994.4 | To evaluate programme (Heyman) viability and impact on PA levels (steps/d and MVPA), diet, subjective wellbeing and other measures | Pilot RCT 3 mo | Intervention based on behavioural theories on changing habits. Groups:
-Jawbone wearable PA tracker with associated app for goal setting and health behaviour self-monitoring -Group/individual face-to-face sessions -Private Facebook discussion group -Gymstick™ resistance band. -Portion planner disc™ | Start and 3 mo -Change in steps/d: pedometer -Diet quality (score): AES-FFQ -Changes in lifestyle, psychological, anthropometric and physiological measures | Change at 3 mo PA Pedometer (steps/d) -C: 575.4 -I: 1588.2 MVPA (min/wk) -C: 26.1 -I: 154.1 Diet quality (score) -C: 2.3 -I: 5.9 Wellbeing (total score) -C: 0.5 -I: 0.9 Other Improved diet, weight loss, other Use and acceptability Reasonable levels for most programme components Retention: 94% | -No significant between-group differences for steps/d, diet quality or wellbeing -Viability of programme demonstrated for a subsequent RCT -RCT not designed to detect between-group differences |
| Duncan [24] | N = 301 Men Age: 35–54 y Australia | To assess effectiveness of technology-based (IT) compared to paper-based (IP) interventions in improving PA, eating behaviours and health literacy | RCT 9 mo | Social cognitive theory/self-regulation theory. ManUP intervention based on challenges (6 PA+1 diet) adapted to starting level (light, moderate, intense) Groups:
-No information provided on other participants -Hardcopies not collected -> no information obtained on challenges or self-control
-Automatic feedback on progress and goals -Possibility of social (website) interaction with other participants | Online surveys at 0, 3, 9 mo PA AAS (total min. PA + number of PA sessions/wk) Diet Adapted Australian population survey (strong psychometric properties) Health literacy PA and diet surveys Satisfaction Likert scale | PA (min/wk) -IT vs IP = 1.03 3 mo vs 0 = 1.45; 9 mo vs 0 = 1.55 PA (sessions/wk) -IT vs IP = 0.97 3 mo vs 0 = 1.61; 9 mo vs 0 = 1.51 Diet (score) -IT vs IP = 1.02 -3 mo vs 0 = 1.07; 9 mo vs 0 = 1.10 Health literacy -Significantly more IT than IP participants considered MVPA of 20 min/d × 3 d/wk to be essential for health Retention: 49.2% (lower in IT group, 46.8%) | -ManUp effective in improving PA and diet, but no significant differences between interventions. |
| Fanning [18] | N = 116 Age: 30–54 y Sedentary participants (<30 min MVPA × 2/wk) USA Means: Age: 41.38 y Women 80% | To determine individual and combined impact of a self-monitoring app and 2 theoretical modules (goals and rewards) on moderate/intense PA, psychosocial outcomes and app use | RCT 12 wk | Interventions based on social cognitive theory plus S.M.A.R.T. goals Groups:
-Goals module -Rewards system (points)
-Goals module
-Rewards system (points)
| PA -Actigraph accelerometer × 7 d (wk 1 and wk 12) -MVPA (>1952 counts/min) OTHER Self-efficacy -Modified BARSE -Modified EXSE -LSE Perceived barriers -Perceived barriers scale Expected results -MOEES Goals EGS Use and usability -Access recorded to apps -Open questions on acceptability -Ease/difficulty associated with use of each module (5-point Likert scale) | PA- from 34.88 to 46.77 min in MVPA - increase 11.90 min/d PA in conditions (d = 0.70) −5.94 extra min/d PA for rewards module Psychosocial variables -Less self-efficacy in overcoming barriers for no-points module (d = −0.39) PA self-efficacy 3-way time-points-goals interaction was significant (p = 0.01) Lifestyle self-efficacy Only time-point interaction was significant (p = 0.03). -Goal setting -06:59 more units in perceived ability to set goals for the intervention (d = 0.82). -Better perceived ability to set goals with the points system (d = 0.99) Expected results Slight-moderate increase for points system (d = 12:28), decrease for no-points system Self-assessment (p = 0.07) Slight-moderate increase for points system (d = 12:25), decrease for no-points system Retention: 88% | -Individuals in all conditions improved daily PA -The rewards module was effective in promoting PA change -Positive evaluation of motivational SMS and request for more such SMS |
| Ginis [30] | N = 40 Age: N/K Participants with Parkinson on stable medication, able to walk 10 min non-stop MoCA score ≥24 Belgium & Israel | To determine feasibility and effectiveness of real-time feedback on gait performance (CuPiD) compared to conventional gait training in the home setting | RCT6 wk + 4 wk follow-up | Groups:
-Gait training 3 × 30 min/wk -Phone with ABF-Gait app: positive/corrective comments on the fly
-Gait training 3 × 30 min/wk -Personalized on-the-fly advice -No CuPiD | Gait speed (primary results) -Walk 1 min on treadmill -Usual conditions: comfortable dual-task (DT) speed while reciting as many words as possible starting with a pre-specified letter Secondary gait and balance -2MWT -Mini-BESTest -FSST -FES-I -PASE FOG severity NFOG-Q and Ziegler protocol Cognitive evaluations CTT and VF when sitting and walking | Gait speed (M/s) Start Significant improvement at both speeds for (I) and (C): -(I) 9.0% (comfortable) and 13.5% (DT) -(C) 5.2% (comfortable) and 5.8% (DT) Stamina and physical capacity 2MWT Start Gait improvements were also noticeable for the 2MWT PASE (0–400) (I): balance significantly improved (Mini-BESTest) in post-test (from 24.8 to 26.1, SD = ~5) (I): QoL maintained (SF-36) at follow-up (C): QoL reduced (from 50.4 to 48.3, SD = ~16) at follow-up Other between-group differences were not significant | -CuPiD was feasible, well accepted and effective in promoting gait training, with participants improving in equal measure -The impact of (C) can be interpreted as small, while that of (I) can be considered clinically moderate and comparable to similar studies -Balance was improved more with(I) than with conventional training for Parkinson |
| Harries [25] | N = 152 Age: 22–40 y Men UK Means: Sedentary work 50%. Regular sports 59% Motorized transport 63% | To determine impact of feedback on number of steps | RCT 8 wk | Groups:
| Steps/d App measurement of steps/d Other Attitudes to PA and perceived barriers (start and end surveys) | PA Mean steps/d recorded: (C) = 2822 (II) = 3842 (IS) = 3984 Compared: (II) vs (C) +60% (IS) vs (C) +67% Other -Any form of feedback (II) and (IS) explained 7.7% of inter-subject variability in step count (F = 6.626, p < 0.0005) -Differences between the 2 intervention groups were not statistically significant Retention: 92% | -Apps to count steps can increase PA in young men -Feedback increased PA, but there were no significant differences between the 2 feedback groups |
| Johnston [26] | N = 166 Age: >18 y Patients with myocardial infarction receiving ticagrelor Sweden Means: Age: 58 y Men: 81% BMI: 29 Diabetes: 13% Smokers: 21% | To evaluate an app aimed at improving treatment adherence and lifestyle in patients with myocardial infarction | Multi-centre RCT 6 mo | Groups:
-Missed dose: SMS the next day + educational message -Prevention education modules (referenced medical information) and personalized message (status, progress) -Extensive treatment adherence module -Exercise -Weight -Smoking -Possibility of recording blood pressure, cholesterol and glucose
-Missed dose SMS the next day | Primary measure Adherence to ticagrelor Secondary measures -Changes in cardiovascular risk factors (BMI, PA, smoking) -QoL -Satisfaction Scales and surveys -EQ-5D VAS (visits 1, 2, 3) -PA surveys (visits 1, 2, 3) -MARS-5 (visits 2, 3) -SUS (visits 2, 3) Support app also evaluated for active group | PA PA sessions/wk (SD) (I) = +1.5 (C) = +1.0 PA m/wk (SD) (I) = +1.5 (C) = +65.0 PA > 150 m/wk (I) = +33.8% (C) = +21.1% Change in QoL (EQ-5D VAS) 14.7 vs 8.4 (p = 0.059) Positive trend for (I) with respect to (C) but not statistically significant Satisfaction Significantly higher in (I) vs (C): SUS 87.3 vs 78.1 (p = 0.001) | -PA increased in (I) compared to (C) -Users would recommend use of app to others |
| King [19] | N = 89 Age: >45 y No smartphone experiences MVPA <60 min/wk Seated >10 h/d USA Means: Age: 60 y Women: 75.3% BMI: 28.8 | To evaluate 3 personalized PA apps based on conceptually different motivational frameworks in comparison to a commercial control app | RCT 8 wk | Groups:
Custom apps sharing basic functions:
| Daily PA and sedentary behaviour -Smartphone accelerometer -Valid data (h) or no more than 60 consecutive 0 values (non-wear time) -Valid day: minimum 10 valid h/d -MVPA (>301 counts/min) -Sedentarism (<56 counts/min) Daily self-report measures -EMA -Brisk walking min/d -Sitting time, h | Moderate/intense PA Differences between groups p = 0.04-0.005 (I-2) vs (C): d = 01:05, CI = 0.44, 1.67 (I-2) vs (I-3): d = 0.89, CI = 0.27, 1.5 (I-2) vs (I-1): d = 0.89, CI = 0.27, 1.51 Sedentarism Differences between groups p = 0.02–0.001 (I-2) vs (C): d = 1.10, CI = 0.48, 1.72 (I-2) vs (I-3): d = 0.94, CI = 0.32, 1.56 (I-2) vs (I-1): d = 1.24, CI = 0.59, 1.89 Seated time Differences between groups p < 0.001 (I-2) vs (C): d = 1.59; CI = 0.92, 2.25 (I-2) vs (I-1): d = 1.89, CI = 1.17, 2.61 (I-3) vs (C): d = 1.19, CI = 0.56, 1.81 (I-3) vs (I-1): d = 1.41, CI = 0.74, 02.07 91.3% of social app users used the message board (total: 775 SMS). Retention: I-1 (95%), I-2 (100%), I-3 (92%), C (89%) | -Social app users significantly increased MVPA (weekly accelerometer) relative to the other 3 groups -Social app users overall had significantly greater accelerometer-derived sedentarism -Social and affective app users reported less time seated than users in the other 2 groups -Satisfaction was high among users -No significant between-group differences in app use |
| Martin [20] | N = 48 Age: 18–69 y Cardiology outpatients MVPA for ≥30 min/d for less 3 day/week USA Means: Age: 58 Sex: Men 54% BMI: 31 Diabetes: 23% Cardiopathy 29% Hypertension: 50% Steps/d: 9670 | To assess if a fully automated mHealth intervention with tracking and texting increased PA | Pilot RCT 5 wk | Fitbug Orb (app with accelerometer linked to intelligent SMS system) Goal 10,000 steps/d Groups/phases: PHASE I (wk 2–3)
-steps/d- activity time -aerobic activity time -previous data history
PHASE II (wk 4–5)
-Personalized SMS in doctor’s name (x 3 per d) -Positive reinforcement SMS -Reinforcement SMS
| Primary result -Mean change in accelerometer-counted step/d from baseline to phases I and II -Achievement of goal of 10,000 steps/d Secondary results -Change in PA/d -Change in aerobic time (>10 min continuous walking with no pause >1 min) -Satisfaction (end-of-trial online survey, with qualitative and quantitative elements). | PHASE I Change in steps/d No significant change/difference between groups Activity min/d No significant change/difference between groups Aerobics min/d Smaller significant decrease in time limit in (I) (8 min difference, 95% CI: 0–16, p = 0.05) PHASE II Change in steps/d 37% absolute increase/84% relative increase in (I-1) over other groups at 10,000 steps/d (p = 0.02) Activity min/d Increase in (I-1) by 21 min/d (+23%) Aerobics min/d Statistically very significant increase in (I-1) by 13 min/d (+160% relative to other groups) Satisfaction -PA tracking: mean 4.0 out of 5.0 -SMS: mean 3.8 out of 5.0 | −48% of participants achieved 10,000 steps/d at the study start. -PA trajectories were different for the 3 groups: For (C), but not for (I-1) or (I-2), there was a progressive downward trend over time, while for (I-1) the clear upward trend was due to SMS -PA increased with automatic intervention with but not without SMS -> increase depended on the SMS component |
| Mayer [21] | N = 284 Age: ≥21 y Colon cancer (stage I-III, treatment completed) PA level <150 min/wk Absence of other cancers (except skin cancer) USA Means: Age: 58 y Men: 48.5% Caucasian: 89% Obesity: 69.5% | To assess Survivor-CHESS app impact on PA in colon cancer survivors and explore Survivor-CHESS impact on QoL and anxiety | RCT 6 mo + 3 mo maintenance | Goal: PA 150 m/wk Groups:
-NCCS Cancer Survival Toolbox -Pedometer.
-Smartphone with SurvivorCHESS app (voice/data service) with: -skills development (My Tracker/Be Mobile). -support services (My Friends). -information services and tools (My Cancer Care) Note: After 6 mo, certified trainer available to users through app | Start -Demographic and medical data on cancer -BMI -Comorbidity conditions (OARS) App (group (I) only) -Number of session started -Pages viewed -SMS content -Internet use convenience (5-point Likert scale: 0–4, min-max). PA -GPAQ: during 1 wk, mean for exercise type (intense, moderate, light) >15 min -Total min: weekly frequency of MVPA | PA (I) 19.4–60.0 min (MPA) (C) 15.5–40.3 min (MPA) (*) Non-significant intervention effect (F(1, 221) = 2.404, p = 0.122) 9 mo -Intervention effect for the same outcome at 9 mo controlling for outcome at 6 mo was not statistically significant (F(1, 202) = 0.722, p = 0.396) -No significant between-group differences for intervention effect sustainability at 9 mo Dropout (6 mo): (C) 28.1% (I) 18.2% | -Greater increase in MVPA in (I) relative to (C) -PA increased over time in both groups with no significant between-group differences -Patients with higher BMI and more comorbidities were less likely to increase PA |
| Memon [28] | N = 56 Age: 18–25 y Women BMI > 25 Pakistan | To evaluate PA increase and weight loss in university students using financial incentives and a smartphone app | RCT 5 wk | Groups:
-ProtoGeoO app
-ProtoGeoO app | PA -Steps/d measured by app Demographics -Various questionnaires -BMI Secondary variables -Body image perceptions -Anxiety -Weight control strategies Measurements based on various questionnaires | PA (C) = 47314.36 steps vs (I) = 57799.61 steps, p > 0.05 Weight loss (C) 68.67 kg (start) to 67.96 kg (end), p = 0.004 (I) 72.13 (start) to 70.97 kg (end), p < 0.001 Retention: 100% | -Notable weight loss in both groups after 5 wk. -No significant difference between the 2 groups in weight loss and steps increase -Significant drop in app use over time |
| Naimark [27] | N = 85 Age: >18 y Technical experience Healthy living interest Israel Means: Age: 47.9 y Women: 64% BMI: 25.8 | To compare adherence to a healthy lifestyle between an app group receiving educational information and a group receiving only educational information | RCT 14 wk | Groups:
-Access to eBalance app without face-to-face support -Diet and PA control tools that also educate on health
-Life as normal | Start and wk 14 -Weight -Waist circumference -Evaluation (online surveys) on nutritional knowledge, diet quality and PA (28 items on type, frequency, duration/wk) Wk 14 -App usability (frequency and convenience) -Satisfaction questionnaire | PA Low activity <150 min -Start: (C) 45% (I) 28% -Wk 14: (C) 55% (I) 17.3% Recommended 150–300 min-Start: (C) 30% (I) 36.5% -Wk 14: (C) 25% (I) 32.7% High activity >300 min -Start: (C) 25% (I) 34.7% -Wk 14: (C) 20% (I) 50% Mean PA change (I) 63 min (SD 20.8) vs (C) −30 min (SD 27.5), Mean weight and BMI change (I) weight −1.44 kg (SD 0.4), BMI −0.48(C) weight −0.128 kg (SD 0.36), BMI −0.03 Retention: 86% | -App motivates users to significantly increase PA time/wk. More users increased PA to >150 min/wk. -Greater weight loss for (I) than (C) -Significant increase in nutritional knowledge in (I) -Frequent app use was significantly associated with greater success (p < 0.001) -Most users stated that the app helped them/they would recommend it to others |
| Spring [22] | N = 96 Age: 18–60 y BMI: 30–40 No weight loss >11.3 kg in previous 6 mo USA Means: Age: 39.3 y Women: 84.4% Weight: 94.8 kg BMI: 34.6 | To determine the impact of 3 weight loss interventions with/without training and mobile technology | RCT 6 mo + 12 mo follow-up | ENGAGED intervention: technical and social weight control measures Groups:
-Hardcopy self-monitoring diaries (6 mo): diet, PA, weight
-8 group sessions -Coaching phone calls (1/wk first 8 wk, then 1/mo)
-App -Shimmer accelerometer for 6 mo -8 group sessions -Coaching phone calls -SMS and social network At 3 and 6 mo, competition between groups with financial incentives | Primary results -Weight (start, 3 mo, 6 mo, 12 mo) -Significant weight loss (≥5%) -Goal for all groups: 7% weight loss (approx. 0.5–1 kg/wk) Behavioural adherence (mo 1-6) -Diet self-monitoring: % days with intake ≥1000 cal/d -PA self-monitoring: % days PA reported or detected -Goals (45–175 min/wk) Fidelization -Phone call/monitoring checklist: 2 × mo for mo 1–2, then 1 × mo from mo 3 (to intervention end) | Weight loss 6 mo -Higher in TECH and STND than in SELF (25.7 kg [95%CI: 27.2–24.1] vs 22.7 kg [95%CI: 25.1–20.3], p < 0.05) 12 mo -Loss ≥5% for 47% STND, 28% TECH and 25% SELF (non-significant differences) Self-control adherence (6 mo) -Self-control of diet, PA and weight (% day adherence) greater for TECH and STND than for SELF (p < 0.001) -Higher PA in TECH 56.8 (4.8) than in STND 30.5 (4.4) or SELF 9.8 (2.4) Treatment fidelity -Training time (1–6 min): greater for TECH (285.71 min [SD = 83.9]) than for STND (202.8 min [SD 89.4]): F(1, 61) = 14.39, p < 0.001 Dropout (12 mo) -Higher for SELF (25.0%) than for STND (12.5%) or TECH (3.1%) | -Self-control adherence to PA higher for TECH than for SELF and especially so than for STND -Weight loss was not significantly different in any group at 12 mo |
| Valentiner [29] | N = 37 Age: 30–80 y DM-II Denmark Means: Age: 66 y Women: 65% BMI: 28.5 Body fat: 37.9% | To investigate feasibility of IWT adherence using EMA and InterWalk in patients with DM-II | RCT 12 wk | Groups:
-Individual goals via interview -Automated survey each wk -Phone call on IWT barriers | PA -Adherence to IWT (total accumulated time during the intervention in InterWalk data) Other secondary measures (exploratory) -Usability of SMS -Self-reported PA -Satisfaction with trial participation -Quality of life (Short-Form Health) Survey (SF-12) -Anthropometric measurements | PA -I: 434 min overall more than C -I: 36 min/wk more than C -Goal achievement: 47% I and 11% C Usability -Women more participatory in the experimental group Satisfaction and perceptions −68% very satisfied −78% intended to continue using app after intervention end Retention: 100% | -The I combination is suitable for achieving IWT adherence -Men respond less to SMS than women |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pradal-Cano, L.; Lozano-Ruiz, C.; Pereyra-Rodríguez, J.J.; Saigí-Rubió, F.; Bach-Faig, A.; Esquius, L.; Medina, F.X.; Aguilar-Martínez, A. Using Mobile Applications to Increase Physical Activity: A Systematic Review. Int. J. Environ. Res. Public Health 2020, 17, 8238. https://doi.org/10.3390/ijerph17218238
Pradal-Cano L, Lozano-Ruiz C, Pereyra-Rodríguez JJ, Saigí-Rubió F, Bach-Faig A, Esquius L, Medina FX, Aguilar-Martínez A. Using Mobile Applications to Increase Physical Activity: A Systematic Review. International Journal of Environmental Research and Public Health. 2020; 17(21):8238. https://doi.org/10.3390/ijerph17218238
Chicago/Turabian StylePradal-Cano, Laura, Carolina Lozano-Ruiz, José Juan Pereyra-Rodríguez, Francesc Saigí-Rubió, Anna Bach-Faig, Laura Esquius, F. Xavier Medina, and Alicia Aguilar-Martínez. 2020. "Using Mobile Applications to Increase Physical Activity: A Systematic Review" International Journal of Environmental Research and Public Health 17, no. 21: 8238. https://doi.org/10.3390/ijerph17218238
APA StylePradal-Cano, L., Lozano-Ruiz, C., Pereyra-Rodríguez, J. J., Saigí-Rubió, F., Bach-Faig, A., Esquius, L., Medina, F. X., & Aguilar-Martínez, A. (2020). Using Mobile Applications to Increase Physical Activity: A Systematic Review. International Journal of Environmental Research and Public Health, 17(21), 8238. https://doi.org/10.3390/ijerph17218238











