Childhood Obesity and its Influence on Sleep Disorders: Kids-Play Study
Abstract
1. Introduction
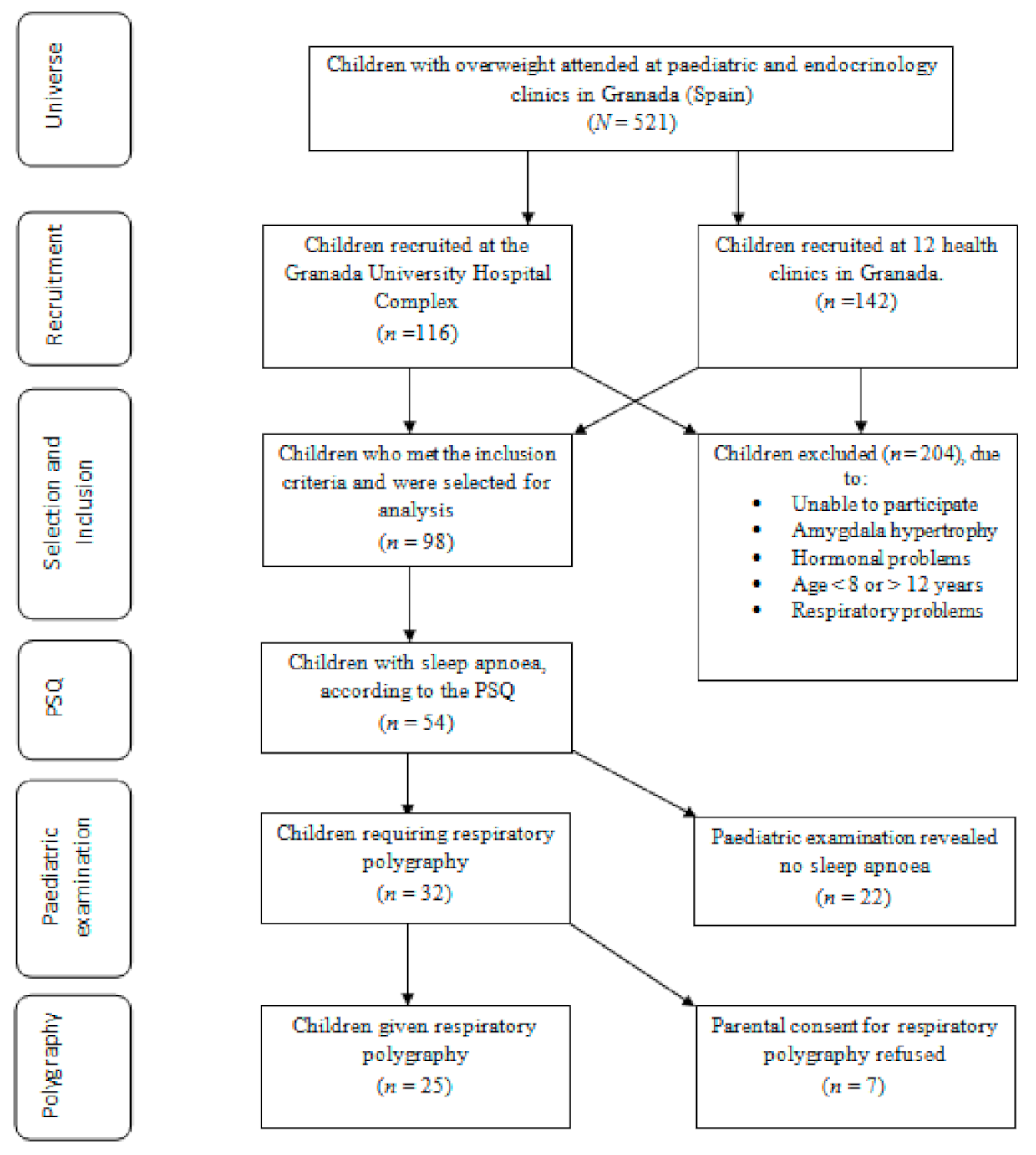
2. Materials and Methods
2.1. Design and Sample Selection
2.2. Body Composition
2.3. Paediatric Sleep Questionnaire: Short-Form Spanish Language Version
2.4. Respiratory Polygraphy
- Flow signal, recorded using a nasal pressure transducer.
- Respiratory effort, assessed from thoracoabdominal movements and detected by two plethysmographic bands. A third band, fastening the junction box, contained the body-position sensor.
- Arterial oxygen saturation and pulse, recorded by a pulse oximeter placed on a finger.
- severely reduced airflow (>50%);
- moderately reduced airflow (<50%) with >3% oxygen desaturation;
- moderately reduced airflow (<50%) with associated electroencephalographic evidence on awakening.
2.5. Ethical Questions
2.6. Statistical Analysis
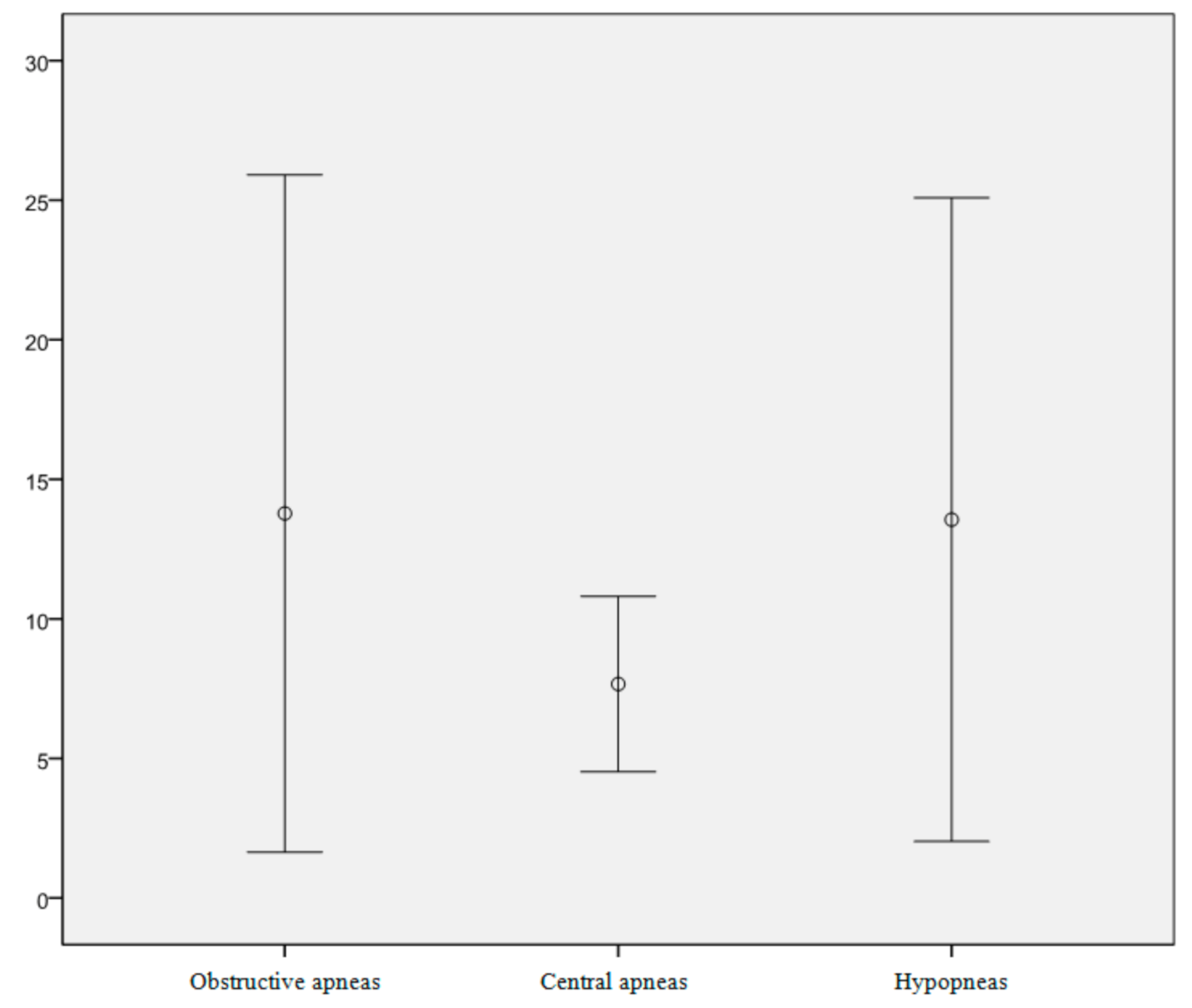
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Obesity and Overweight. WHO Fact Sheet No. 311. 2014. Available online: http://www.who.int/mediacentre/factsheets/fs311/en/# (accessed on 10 July 2020).
- World Health Organization. Global Database on Body Mass Index; WHO: Geneva, Switzerland, 2009. [Google Scholar]
- Lifestyles Statistics Team, Health and Social Care Information Centre. Statistics on Obesity, Physical Activity and Diet: England. 2014. Available online: http://www.hscic.gov.uk/catalogue/PUB13648/Obes-phys-acti-diet-eng-2014-rep.pdf (accessed on 9 July 2020).
- Joint UNICEF/WHO/World Bank Child Malnutrition Database. Estimates for 2013 and Interactive Data Dashboards. Available online: http://www.who.int/nutgrowthdb/estimates2013/en/ (accessed on 20 October 2014).
- Arens, R.; Muzumdar, H. Childhood obesity and obstructive sleep apnea syndrome. J. Appl. Physiol. 2010, 108, 436–444. [Google Scholar] [CrossRef] [PubMed]
- Aguilar, M.C.; Sánchez, A.L.; Mur, N.V.; Sánchez, A.M.; Guisado, R.B. Influence of a program of physical activity in children and adolescents obese with sleep apnea; study protocol. Nutr. Hosp. 2013, 28, 701–704. [Google Scholar]
- Culebras, A. Sleep apnea syndrome: Short-term solutions and long-term cerebrovascular risk. Rev. Neurolol. 2006, 42, 34–41. [Google Scholar]
- American Academy of Sleep Medicine. International Classification of Sleep Disorders, 2 Edition: Diagnostic and Coding Manual; American Academy of Sleep Medicine: Westchester, IL, USA, 2005. [Google Scholar]
- Salvador, J.; Iriarte, J.; Silva, C. El síndrome de apneas obstructivas del sueño en la obesidad: Un conspirador en la sombra. Rev. Med. Univ. Navar. 2004, 48, 55–62. [Google Scholar]
- Llombart, M.; Chiner, E.; Gómez-Merino, E.; Andreu, A.; Pastor, E.; Senent, C.; Camarasa, A.; Signes-Costa, J. Sleep Apnea-Hypopnea Syndrome in a Pediatric Population: Differences between Children with Tonsillar Hypertrophy and Those with Concomitant Disease. Arch. Bronconeumol. 2007, 43, 655–661. [Google Scholar] [CrossRef]
- Caminiti, C.; Evangelista, P.; Leske, V.; Loto, Y.; Mazza, C. Obstructive sleep apneas in symptomatic obese children: Polisomnographic confirmation and its association with disturbances in carbohydrate metabolism. Arch. Argent. Pediatr. 2010, 108, 226–233. [Google Scholar]
- Bronconeumol, A. Consensus document on sleep apnea-hypopnea syndrome in children (full version). Arch. Bronconeumol. 2011, 47 (Suppl. 5), 2–18. [Google Scholar]
- Guimarães, K.C.; Drager, L.F.; Genta, P.R.; Marcondes, B.F.; Lorenzi-Filho, G. Effects of oropharyngeal exercises on patients with moderate obstructive sleep apnea syndrome. Am. J. Respir. Crit. Care Med. 2009, 179, 962–966. [Google Scholar] [CrossRef]
- Martinez Rivera, C.; Abad, J.; Fiz, J.A.; Rios, J.; Morera, J. Usefulness of truncal obesity indices as predictive factors for obstructive sleep apnea syndrome. Obesity 2012, 16, 113–118. [Google Scholar] [CrossRef]
- Maeder, M.T.; Ammann, P.; Schoch, O.D.; Rickli, H.; Korte, W.; Hürny, C.; Myers, J.; Münzer, T. Determinants of postexercise heart rate recovery in patients with the obstructive sleep apnea syndrome. CHEST J. 2010, 137, 310–317. [Google Scholar] [CrossRef]
- Nanas, S.; Sakellariou, D.; Kapsimalakou, S.; Dimopoulos, S.; Tassiou, A.; Tasoulis, A.; Anastasiou-Nana, M.; Vagiakis, E.; Rousso, C. Heart rate recovery and oxygen kinetics after exercise in obstructive sleep apnea syndrome. Clin. Cardiol. 2010, 33, 46–51. [Google Scholar] [CrossRef]
- Sánchez-López, A.M.; Menor-Rodríguez, M.J.; Sánchez-García, J.C.; Aguilar-Cordero, M.J. Play as a Method to Reduce Overweight and Obesity in Children: An RCT. Int. J. Environ. Res. Public Health 2020, 17, 346. [Google Scholar] [CrossRef]
- Bergel, M.L.; Cesani, M.F.; Cordero, M.L.; Navazo, B.; Olmedo, S.; Quintero, F.; Sardi, M.; Torres, M.F.; Aréchiga, J.; Méndez de Pérez, B.; et al. Nutritional valuation of schoolchildren from three Ibero-American countries: A comparative analysis of the references proposed by International Obesity Task Force (IOTF) and World Health Organization (WHO). Nutr. Clín. Diet. Hosp. 2014, 34, 8–15. [Google Scholar]
- Ogawa, H.; Fujitani, K.; Tsujinaka, T.; Imanishi, K.; Shirakata, H.; Kantani, A.; Hirao, M.; Kurokawa, Y.; Utsumi, S. InBody 720 as a new method of evaluating visceral obesity. Hepato Gastroenterol. 2011, 58, 42–44. [Google Scholar]
- Chervin, R.D.; Hedger, K.; Dilon, J.E.; Pituch, K.J. Pediatric Sleep Questionnaire (PSQ): Validity and reliability of scales-disordered breathing, snoring, sleepiness and behavioral problems. Sleep Med. 2000, 1, 21–32. [Google Scholar] [CrossRef]
- Vila, M.; Miralles Torres, T.A.; Beseler Soto, B. Spanish version of the pediatric sleep questionnaire (PSQ). A useful instrument in investigation of sleep disturbances in childhood. Reliability analysis. An. Pediatr. 2007, 66, 121–128. [Google Scholar]
- Franco, P.; Bourdin, H.; Braun, F.; Briffod, J.; Pin, I.; Challamel, M.J. [Overnight polysomnography versus respiratory polygraphy in the diagnosis of pediatric obstructive sleep apnea]. Arch. Pediatr. 2017, 24 (Suppl. 1), S16–S27. [Google Scholar] [CrossRef]
- Susanna, S.S.N.; Chan, T.; To, K.; Ngai, J.; Tung, A.; Ko, F.; Hui, D.S.C. Validation of Embletta portable diagnostic system for identifying patients with suspected obstructive sleep apnoea syndrome (OSAS). Respirology 2010, 15, 336–342. [Google Scholar]
- Villa Asensia, J.R.; Martínez Carrasco, C.; Pérez Pérez, G.; Cortell Aznard, I.; Gómez-Pastrana, D.; Álvarez Gil, D.; González Pérez-Yarza, E. Guidelines for the diagnosis and management of sleep apnea-hypoapnea syndrome in children. An. Pediatr. 2006, 65, 364–376. [Google Scholar]
- Chang, L.; Wu, J.; Cao, L. Combination of symptoms and oxygen desaturation index in predicting childhood obstructive sleep apnea. Int. J. Pediatr. Otorhinolaryngol. 2013, 77, 365–371. [Google Scholar] [CrossRef]
- Douglas, N.J.; Thomas, S.; Jan, M.A. Clinical value of polysomnography. Lancet 1992, 339, 347–350. [Google Scholar] [CrossRef]
- Wing, Y.K.; Hui, S.H.; Pak, W.M.; Ho, C.; Cheung, A.; Li, A.; Fok, T. A controlled study of sleep related disordered breathing in obese children. Arch. Dis. Child. 2003, 88, 1043–1047. [Google Scholar] [CrossRef]
- Xu, Z.; Jiaqing, A.; Yuchuan, L.; Shen, K. A case-control study of obstructive sleep apnea-hypopnea syndrome in obese and nonobese Chinese children. Chest 2008, 133, 684–689. [Google Scholar] [CrossRef] [PubMed]
- Kang, K.T.; Chou, C.H.; Weng, W.C.; Lee, P.L.; Hsu, W.C. Associations between adenotonsillar hypertrophy, age, and obesity in children with obstructive sleep apnea. PLoS ONE 2013, 8, e78666. [Google Scholar] [CrossRef]
- Alonso-Álvarez, M.L.; Cordero-Guevara, J.A.; Terán-Santos, J.; Gonzalez-Martinez, M.; Jurado-Luque, M.J.; Corral-Peñafiel, J.; Duran-Cantolla, J.; Kheirandish-Gozal, L.; Gozal, D. Obstructive sleep apnea in obese community-dwelling children: The NANOS study. Sleep 2014, 37, 943–949. [Google Scholar] [PubMed]
- Matsumoto, E.; Tanaka, E.; Tabe, H.; Wakisaka, N.; Nakata, Y.; Ueda, H.; Hori, T.; Abe, T.; Tanne, K. Sleep architecture and the apnoea-hypopnoea index in children with obstructive-sleep apnoea syndrome. J. Oral Rehabil. 2007, 34, 112–120. [Google Scholar] [CrossRef]
- Miano, S.; Esposito, M.; Foderaro, G.; Ramelli, G.P.; Pezzoli, V.; Manconi, M. Sleep-related disorders in children with attention-deficit hyperactivity disorder: Preliminary results of a full sleep assessment study. CNS Neurosci. Ther. 2016, 22, 906–914. [Google Scholar] [CrossRef]
- Zaffanello, M.; Piacentini, G.; Gasperi, E.; Maffeis, C.; Fava, C.; Bonafini, S.; Tagetti, A.; Antoniazzi, F. Snoring in a cohort of obese children: Association with palate position and nocturnal desaturations. J. Pediatr. Neonat. Individual. Med. 2016, 5, e050134. [Google Scholar]
| Weight | Height | BMI | Body Fat % | |
|---|---|---|---|---|
| Mean (SD) | 66.05 (17.39) | 150.75 (11.08) | 28.60 (4.05) | 40.19 (5.74) |
| Minimum | 38.80 | 129.00 | 21.43 | 25.10 |
| Maximum | 106.20 | 180.00 | 38.50 | 50.70 |
| % SO2 | HRmean | HRmax | HRmin | AHI | Destotal | DesIndex | |
|---|---|---|---|---|---|---|---|
| Mean (SD) | 95.74 (1.02) | 73.57 (1.79) | 99.61 (10.42) | 60.68 (4.70) | 4.08 (3.93) | 21.33 (14.79) | 2.83 (1.87) |
| Minimum | 94.40 | 70.60 | 86.50 | 52.00 | 1.00 | 0.00 | 0.00 |
| Maximum | 97.10 | 76.60 | 124.00 | 66.80 | 10.80 | 51.00 | 5.40 |
| Desaturation Index % (n) | AHI % (n) | |
|---|---|---|
| Healthy | 12 (3) | 0 (0) |
| Mild SAHS | 32 (8) | 56 (14) |
| Severe SAHS | 56 (14) | 44 (11) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sánchez-López, A.M.; Noack-Segovia, J.P.; Núñez-Negrillo, A.M.; Latorre-García, J.; Aguilar-Cordero, M.J. Childhood Obesity and its Influence on Sleep Disorders: Kids-Play Study. Int. J. Environ. Res. Public Health 2020, 17, 7948. https://doi.org/10.3390/ijerph17217948
Sánchez-López AM, Noack-Segovia JP, Núñez-Negrillo AM, Latorre-García J, Aguilar-Cordero MJ. Childhood Obesity and its Influence on Sleep Disorders: Kids-Play Study. International Journal of Environmental Research and Public Health. 2020; 17(21):7948. https://doi.org/10.3390/ijerph17217948
Chicago/Turabian StyleSánchez-López, Antonio Manuel, Jessica Pamela Noack-Segovia, Ana María Núñez-Negrillo, Julio Latorre-García, and María José Aguilar-Cordero. 2020. "Childhood Obesity and its Influence on Sleep Disorders: Kids-Play Study" International Journal of Environmental Research and Public Health 17, no. 21: 7948. https://doi.org/10.3390/ijerph17217948
APA StyleSánchez-López, A. M., Noack-Segovia, J. P., Núñez-Negrillo, A. M., Latorre-García, J., & Aguilar-Cordero, M. J. (2020). Childhood Obesity and its Influence on Sleep Disorders: Kids-Play Study. International Journal of Environmental Research and Public Health, 17(21), 7948. https://doi.org/10.3390/ijerph17217948






