Postpartum Weight Retention and Its Determinants in Lebanon and Qatar: Results of the Mother and Infant Nutrition Assessment (MINA) Cohort
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Subjects
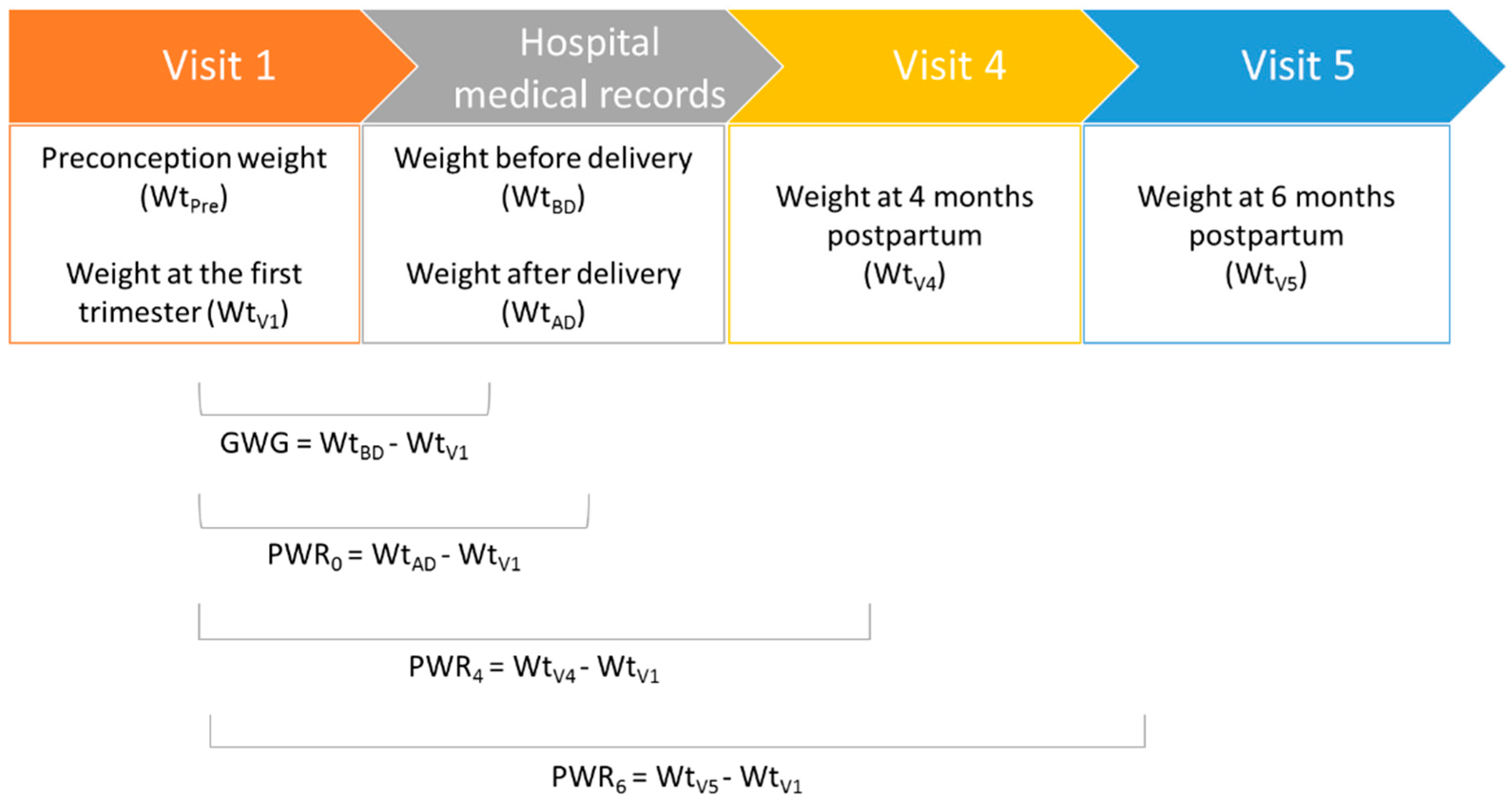
2.2. Data Collection
2.3. Statistical Analysis
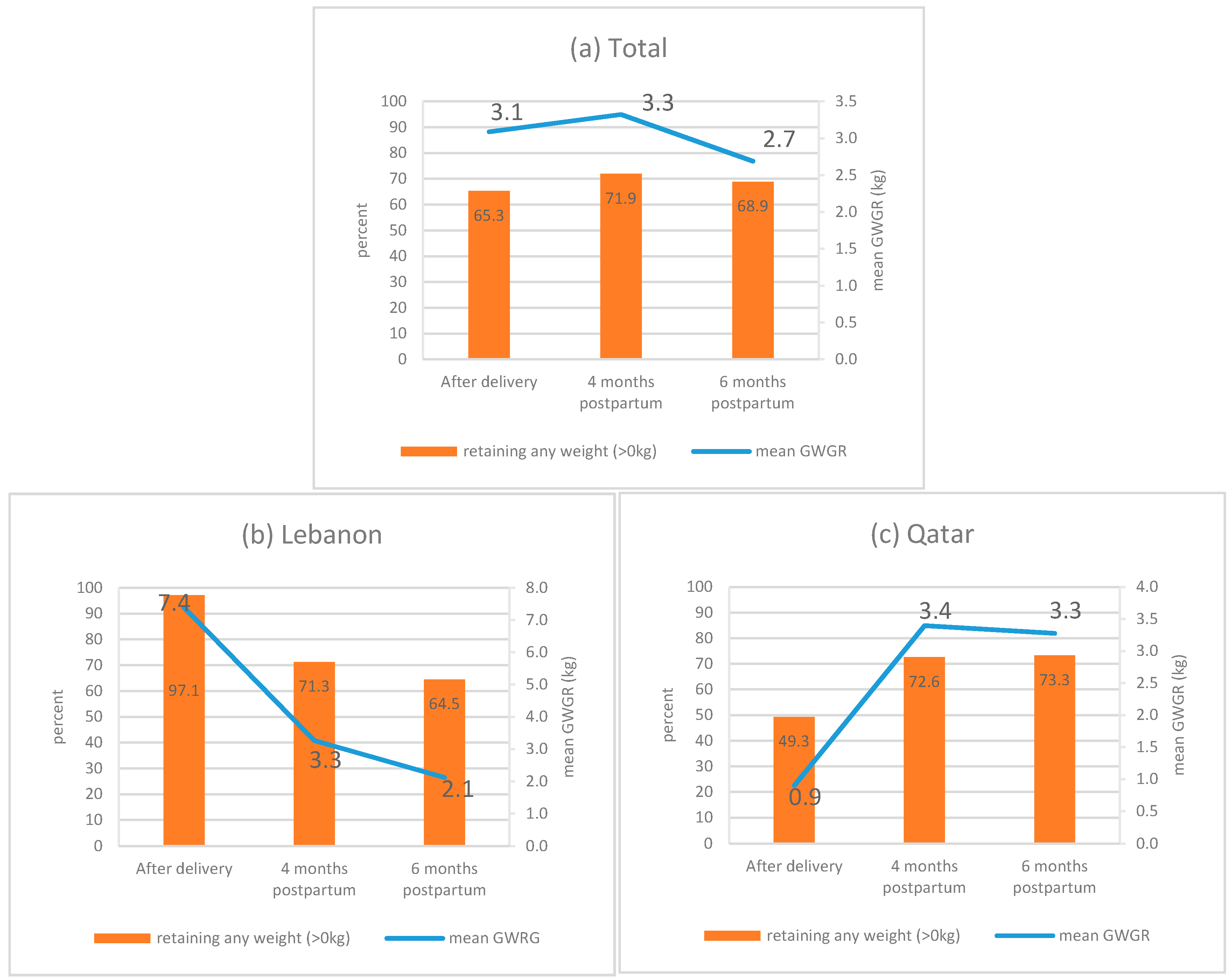
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gunderson, E.; Murtaugh, M.; Lewis, C.; Quesenberry, C.; West, D.S.; Sidney, S. Excess gains in weight and waist circumference associated with childbearing: The Coronary Artery Risk Development in Young Adults Study (CARDIA). Int. J. Obes. 2004, 28, 525–535. [Google Scholar] [CrossRef]
- National Institute for Health and Clinical Excellence. NICE Public Health Guidance 27: Weight Management Before, During and After Pregnancy; NICE: London, UK, 2010. [Google Scholar]
- Oken, E.; Taveras, E.M.; Popoola, F.A.; Rich-Edwards, J.W.; Gillman, M.W. Television, walking, and diet: Associations with postpartum weight retention. Am. J. Prev. Med. 2007, 32, 305–311. [Google Scholar] [CrossRef]
- Cheng, H.R.; Walker, L.; Tseng, Y.F.; Lin, P.C. Post-partum weight retention in women in Asia: A systematic review. Obes. Rev. 2011, 12, 770–780. [Google Scholar] [CrossRef]
- Lovelady, C. Balancing exercise and food intake with lactation to promote post-partum weight loss. Proc. Nutr. Soc. 2011, 70, 181–184. [Google Scholar] [CrossRef]
- Mannan, M.; Doi, S.A.; Mamun, A.A. Association between weight gain during pregnancy and postpartum weight retention and obesity: A bias-adjusted meta-analysis. Nutr. Rev. 2013, 71, 343–352. [Google Scholar] [CrossRef]
- Gore, S.A.; Brown, D.M.; West, D.S. The role of postpartum weight retention in obesity among women: A review of the evidence. Ann. Behav. Med. 2003, 26, 149–159. [Google Scholar] [CrossRef]
- Linné, Y.; Barkeling, B.; Rössner, S. Long-term weight development after pregnancy. Obes. Rev. 2002, 3, 75–83. [Google Scholar] [CrossRef]
- Harris, H.; Ellison, G.; Clement, S. Do the psychosocial and behavioral changes that accompany motherhood influence the impact of pregnancy on long-term weight gain’. J. Psychosom. Obstet. Gynaecol. 1999, 20, 65–79. [Google Scholar] [CrossRef]
- Williamson, D.F.; Madans, J.; Pamuk, E.; Flegal, K.M.; Kendrick, J.S.; Serdula, M.K. A prospective study of childbearing and 10-year weight gain in US white women 25 to 45 years of age. Int. J. Obes. Relat. Metab. Disord. 1994, 18, 561–569. [Google Scholar]
- Smith, D.E.; Lewis, C.E.; Caveny, J.L.; Perkins, L.L.; Burke, G.L.; Bild, D.E. Longitudinal changes in adiposity associated with pregnancy: The CARDIA study. JAMA 1994, 271, 1747–1751. [Google Scholar] [CrossRef]
- Jiang, M.; Gao, H.; Vinyes-Pares, G.; Yu, K.; Ma, D.; Qin, X.; Wang, P. Association between breastfeeding duration and postpartum weight retention of lactating mothers: A meta-analysis of cohort studies. Clin. Nutr. 2018, 37, 1224–1231. [Google Scholar] [CrossRef]
- Linne, Y.; Neovius, M. Identification of women at risk of adverse weight development following pregnancy. Int. J. Obes. 2006, 30, 1234–1239. [Google Scholar] [CrossRef] [PubMed]
- Farpour-Lambert, N.J.; Ells, L.J.; Martinez de Tejada, B.; Scott, C. Obesity and weight gain in pregnancy and postpartum: An evidence review of lifestyle interventions to inform maternal and child health policies. Front. Endocrinol. 2018, 9, 546. [Google Scholar] [CrossRef]
- Gillman, M.W. Interrupting intergenerational cycles of maternal obesity. In Prev Aspects Early Nutr; Karger Publishers: Berlin, Germany, 2016; Volume 85, pp. 59–69. [Google Scholar]
- Fadzil, F.; Shamsuddin, K.; Puteh, S.E.W.; Tamil, A.M.; Ahmad, S.; Hayi, N.S.A.; Samad, A.A.; Ismail, R.; Shauki, N.I.A. Predictors of postpartum weight retention among urban Malaysian mothers: A prospective cohort study. Obes. Res. Clin. Pract. 2018, 12, 493–499. [Google Scholar] [CrossRef]
- Nehring, I.; Schmoll, S.; Beyerlein, A.; Hauner, H.; von Kries, R. Gestational weight gain and long-term postpartum weight retention: A meta-analysis. Am. J. Clin. Nutr. 2011, 94, 1225–1231. [Google Scholar] [CrossRef]
- Hill, B.; McPhie, S.; Skouteris, H. The role of parity in gestational weight gain and postpartum weight retention. Women’s Health Issues 2016, 26, 123–129. [Google Scholar] [CrossRef]
- Maddah, M.; Nikooyeh, B. Weight retention from early pregnancy to three years postpartum: A study in Iranian women. Midwifery 2009, 25, 731–737. [Google Scholar] [CrossRef]
- Kac, G.; D’Aquino Benicio, M.H.; Valente, J.G.; Velásquez-Meléndez, G. Postpartum weight retention among women in Rio de Janeiro: A follow-up study. Cad. Saude Publica 2003, 19, S149–S161. [Google Scholar] [CrossRef][Green Version]
- Rong, K.; Yu, K.; Han, X.; Szeto, I.M.; Qin, X.; Wang, J.; Ning, Y.; Wang, P.; Ma, D. Pre-pregnancy BMI, gestational weight gain and postpartum weight retention: A meta-analysis of observational studies. Public Health Nutr. 2015, 18, 2172–2182. [Google Scholar] [CrossRef]
- Siega-Riz, A.M.; Herring, A.H.; Carrier, K.; Evenson, K.R.; Dole, N.; Deierlein, A. Sociodemographic, perinatal, behavioral, and psychosocial predictors of weight retention at 3 and 12 months postpartum. Obesity 2010, 18, 1996–2003. [Google Scholar] [CrossRef]
- Abebe, D.S.; Von Soest, T.; Von Holle, A.; Zerwas, S.C.; Torgersen, L.; Bulik, C.M. Developmental trajectories of postpartum weight 3 years after birth: Norwegian Mother and Child Cohort study. Matern Child. Health J. 2015, 19, 917–925. [Google Scholar] [CrossRef][Green Version]
- Zanotti, J.; Capp, E.; Wender, M.C.O. Factors associated with postpartum weight retention in a Brazilian cohort. Rev. Bras. Ginecol. Obstet. 2015, 37, 164–171. [Google Scholar] [CrossRef]
- Herring, S.J.; Rich-Edwards, J.W.; Oken, E.; Rifas-Shiman, S.L.; Kleinman, K.P.; Gillman, M.W. Association of postpartum depression with weight retention 1 year after childbirth. Obesity 2008, 16, 1296–1301. [Google Scholar] [CrossRef]
- Olson, C.; Strawderman, M.; Hinton, P.; Pearson, T. Gestational weight gain and postpartum behaviors associated with weight change from early pregnancy to 1 y postpartum. Int. J. Obes. 2003, 27, 117–127. [Google Scholar] [CrossRef]
- Schauberger, C.W.; Rooney, B.L.; Brimer, L.M. Factors that influence weight loss in the puerperium. Obstet. Gynecol. 1992, 79, 424–429. [Google Scholar] [CrossRef] [PubMed]
- Knudsen, V.K.; Heitmann, B.L.; Halldorsson, T.I.; Sørensen, T.I.; Olsen, S.F. Maternal dietary glycaemic load during pregnancy and gestational weight gain, birth weight and postpartum weight retention: A study within the Danish National Birth Cohort. Br. J. Nutr. 2013, 109, 1471–1478. [Google Scholar] [CrossRef] [PubMed]
- Hollis, J.L.; Crozier, S.R.; Inskip, H.M.; Cooper, C.; Godfrey, K.M.; Harvey, N.C.; Collins, C.E.; Robinson, S.M. Modifiable risk factors of maternal postpartum weight retention: An analysis of their combined impact and potential opportunities for prevention. Int. J. Obes. 2017, 41, 1091–1098. [Google Scholar] [CrossRef] [PubMed]
- Lyu, L.-C.; Lo, C.-C.; Chen, H.-F.; Wang, C.-Y.; Liu, D.-M. A prospective study of dietary intakes and influential factors from pregnancy to postpartum on maternal weight retention in Taipei, Taiwan. Br. J. Nutr. 2009, 102, 1828–1837. [Google Scholar] [CrossRef]
- Sha, T.; Cheng, G.; Li, C.; Gao, X.; Li, L.; Chen, C.; Yan, Y. Patterns of Women’s Postpartum Weight Retention and Its Associations with Maternal Obesity-Related Factors and Parity. Int. J. Environ. Res. Public Health 2019, 16, 4510. [Google Scholar] [CrossRef]
- Shao, H.-H.; Hwang, L.-C.; Huang, J.-P.; Hsu, H.-Y. Postpartum weight retention risk factors in a Taiwanese Cohort Study. Obes. Facts 2018, 11, 37–45. [Google Scholar] [CrossRef]
- Huang, T.-T.; Wang, H.-S.; Dai, F.-T. Effect of pre-pregnancy body size on postpartum weight retention. Midwifery 2010, 26, 222–231. [Google Scholar] [CrossRef] [PubMed]
- Krause, K.M.; Lovelady, C.A.; Peterson, B.L.; Chowdhury, N.; Østbye, T. Effect of breast-feeding on weight retention at 3 and 6 months postpartum: Data from the North Carolina WIC Programme. Public Health Nutr. 2010, 13, 2019–2026. [Google Scholar] [CrossRef]
- Sibai, A.M.; Nasreddine, L.; Mokdad, A.H.; Adra, N.; Tabet, M.; Hwalla, N. Nutrition transition and cardiovascular disease risk factors in Middle East and North Africa countries: Reviewing the evidence. Ann. Nutr. Metab. 2010, 57, 193–203. [Google Scholar] [CrossRef] [PubMed]
- Nasreddine, L.; Naja, F.; Chamieh, M.C.; Adra, N.; Sibai, A.-M.; Hwalla, N. Trends in overweight and obesity in Lebanon: Evidence from two national cross-sectional surveys (1997 and 2009). BMC Public Health 2012, 12, 798. [Google Scholar] [CrossRef] [PubMed]
- Haj Bakri, A.; Al-Thani, A. Chronic Disease Risk Factor Surveillance: Qatar STEPS Report 2012; The Supreme Council of Health: Doha, Qatar, 2013. [Google Scholar]
- Kingdom of Bahrain Ministry of Health and UNDP. National Non-Communicable Diseases Risk Factors Survey 2007. In Report of the National Non-Communicable Diseases STEPwise Survey; 2009; Available online: https://www.who.int/ncds/surveillance/steps/2007_STEPS_Survey_Bahrain.pdf (accessed on 25 September 2020).
- Sarwer, D.B.; Allison, K.C.; Gibbons, L.M.; Markowitz, J.T.; Nelson, D.B. Pregnancy and obesity: A review and agenda for future research. J. Women’s Health 2006, 15, 720–733. [Google Scholar] [CrossRef]
- Naja, F.; Nasreddine, L.; Yunis, K.; Clinton, M.; Nassar, A.; Jarrar, S.F.; Moghames, P.; Ghazeeri, G.; Rahman, S.; Al-Chetachi, W. Study protocol: Mother and infant nutritional assessment (MINA) cohort study in Qatar and Lebanon. BMC Pregnancy Childbirth 2016, 16, 98. [Google Scholar] [CrossRef]
- Abdulmalik, M.A.; Ayoub, J.J.; Mahmoud, A.; Collaborators, M.; Nasreddine, L.; Naja, F. Pre-pregnancy BMI, gestational weight gain and birth outcomes in Lebanon and Qatar: Results of the MINA cohort. PLoS ONE 2019, 14, e0219248. [Google Scholar] [CrossRef]
- World Health Organization. Body Mass Index-BMI. Available online: http://www.euro.who.int/en/health-topics/disease-prevention/nutrition/a-healthy-lifestyle/body-mass-index-bmi (accessed on 19 April 2019).
- Posner, B.M.; Smigelski, C.; Duggal, A.; Morgan, J.; Cobb, J.; Cupples, L. Validation of two-dimensional models for estimation of portion size in nutrition research. J. Am. Diet. Assoc. 1992, 92, 738–741. [Google Scholar]
- Institute of Medicine and National Research Council. Weight Gain During Pregnancy: Reexamining the Guidelines; National Academies Press: Washington, DC, USA, 2009. [Google Scholar]
- Willet, W. Nutritional Epidemiology, 2nd ed.; Oxford University Press: New York, NY, USA, 1998. [Google Scholar]
- Rooney, B.L.; Schauberger, C.W. Excess pregnancy weight gain and long-term obesity: One decade later. Obstet. Gynecol. 2002, 100, 245–252. [Google Scholar] [CrossRef]
- Gunderson, E.P.; Lewis, C.E.; Murtaugh, M.A.; Quesenberry, C.P.; Smith West, D.; Sidney, S. Long-term plasma lipid changes associated with a first birth: The Coronary Artery Risk Development in Young Adults study. Am. J. Epidemiol. 2004, 159, 1028–1039. [Google Scholar] [CrossRef]
- Reaven, G. Banting lecture 1988. Role of insulin resistance in human disease. Diabetes 1988, 37, 1595–1607. [Google Scholar] [CrossRef] [PubMed]
- Soria-Contreras, D.C.; Rifas-Shiman, S.L.; Aris, I.M.; Perng, W.; Switkowski, K.M.; Téllez-Rojo, M.M.; Trejo-Valdivia, B.; López-Ridaura, R.; Oken, E. Weight Trajectories After Delivery are Associated with Adiposity and Cardiometabolic Markers at 3 Years Postpartum Among Women in Project Viva. J. Nutr. 2020. [Google Scholar] [CrossRef]
- STEPS. Palestine STEPS Survey 2010-2011 Fact Sheet. WHO STEPS Chronic Disease Risk Factor Surveillance (6-3C-1). Available online: http://www.who.int/chp/steps/Palestine_FactSheet_2010-11.pdf?ua=1 (accessed on 25 April 2019).
- ALNohair, S. Obesity in gulf countries. Int. J. Health Sci. 2014, 8, 79. [Google Scholar] [CrossRef] [PubMed]
- Ng, S.W.; Zaghloul, S.; Ali, H.; Harrison, G.; Popkin, B.M. The prevalence and trends of overweight, obesity and nutrition-related non-communicable diseases in the Arabian Gulf States. Obes. Rev. 2011, 12, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Ministry of Health. Non-Communicable Diseases and Their Risk Factors. Syrian Arab Republic. Directorate of Environmental and Chronic Diseases. Available online: http://www.who.int/chp/steps/SyrianArabRepublicSTEPSReport.PDF?ua=1 (accessed on 25 April 2019).
- Department of Statistics [Jordan] and ICF International. Jordan Population and Family Health Survey 2012. Available online: https://dhsprogram.com/pubs/pdf/FR282/FR282.pdf (accessed on 26 April 2019).
- World Health Organization. Nutrition. Available online: http://www.emro.who.int/health-topics/nutrition/index.html (accessed on 20 April 2019).
- Koh, H.; Ee, T.X.; Malhotra, R.; Allen, J.C.; Tan, T.C.; Østbye, T. Predictors and adverse outcomes of inadequate or excessive gestational weight gain in an Asian population. J. Obstet. Gynaecol. Res. 2013, 39, 905–913. [Google Scholar] [CrossRef]
- Scott, C.; Andersen, C.T.; Valdez, N.; Mardones, F.; Nohr, E.A.; Poston, L.; Loetscher, K.C.Q.; Abrams, B. No global consensus: A cross-sectional survey of maternal weight policies. BMC Pregnancy Childbirth 2014, 14, 167. [Google Scholar] [CrossRef]
- Heslehurst, N.; Newham, J.; Maniatopoulos, G.; Fleetwood, C.; Robalino, S.; Rankin, J. Implementation of pregnancy weight management and obesity guidelines: A meta-synthesis of healthcare professionals’ barriers and facilitators using the T heoretical D omains F ramework. Obes. Rev. 2014, 15, 462–486. [Google Scholar] [CrossRef]
- Zhu, Y.; Bo, Y.; Liu, Y. Dietary total fat, fatty acids intake, and risk of cardiovascular disease: A dose-response meta-analysis of cohort studies. Lipids Health Dis. 2019, 18, 91. [Google Scholar] [CrossRef]
- Islam, M.A.; Amin, M.N.; Siddiqui, S.A.; Hossain, M.P.; Sultana, F.; Kabir, M.R. Trans fatty acids and lipid profile: A serious risk factor to cardiovascular disease, cancer and diabetes. Diabetes Metab. Syndr. Clin. Res. Rev. 2019, 13, 1643–1647. [Google Scholar] [CrossRef]
- Koh-Banerjee, P.; Chu, N.-F.; Spiegelman, D.; Rosner, B.; Colditz, G.; Willett, W.; Rimm, E. Prospective study of the association of changes in dietary intake, physical activity, alcohol consumption, and smoking with 9-y gain in waist circumference among 16 587 US men. Am. J. Clin. Nutr. 2003, 78, 719–727. [Google Scholar] [CrossRef]
- Memisoglu, A.; Hu, F.B.; Hankinson, S.E.; Manson, J.E.; De Vivo, I.; Willett, W.C.; Hunter, D.J. Interaction between a peroxisome proliferator-activated receptor γ gene polymorphism and dietary fat intake in relation to body mass. Hum. Mol. Genet. 2003, 12, 2923–2929. [Google Scholar] [CrossRef] [PubMed]
- Baer, D.J.; Judd, J.T.; Clevidence, B.A.; Tracy, R.P. Dietary fatty acids affect plasma markers of inflammation in healthy men fed controlled diets: A randomized crossover study. Am. J. Clin. Nutr. 2004, 79, 969–973. [Google Scholar] [CrossRef] [PubMed]
- Mozaffarian, D.; Pischon, T.; Hankinson, S.E.; Rifai, N.; Joshipura, K.; Willett, W.C.; Rimm, E.B. Dietary intake of trans fatty acids and systemic inflammation in women. Am. J. Clin. Nutr. 2004, 79, 606–612. [Google Scholar] [CrossRef] [PubMed]
- Engström, G.; Hedblad, B.; Stavenow, L.; Lind, P.; Janzon, L.; Lindgärde, F. Inflammation-sensitive plasma proteins are associated with future weight gain. Diabetes 2003, 52, 2097–2101. [Google Scholar] [CrossRef]
- Naja, F.; Nasreddine, L.; Itani, L.; Chamieh, M.C.; Adra, N.; Sibai, A.M.; Hwalla, N. Dietary patterns and their association with obesity and sociodemographic factors in a national sample of Lebanese adults. Public Health Nutr. 2011, 14, 1570–1578. [Google Scholar] [CrossRef]
- Egg, S.; Erler, J.; Perktold, B.; Hasenegger, V.; Rust, P.; Ramoner, R.; König, J.; Purtscher, A.E. Traditional v. modern dietary patterns among a population in western Austria: Associations with body composition and nutrient profile. Public Health Nutr. 2019, 22, 455–465. [Google Scholar] [CrossRef]
- Al Thani, M.; Al Thani, A.A.; Al-Chetachi, W.; Al Malki, B.; Khalifa, S.A.; Haj Bakri, A.; Hwalla, N.; Nasreddine, L.; Naja, F. A ‘high Risk’ Lifestyle pattern is associated with metabolic syndrome among Qatari women of reproductive age: A cross-sectional national study. Int. J. Mol. Sci. 2016, 17, 698. [Google Scholar] [CrossRef]
- Endres, L.K.; Straub, H.; McKinney, C.; Plunkett, B.; Minkovitz, C.S.; Schetter, C.D.; Ramey, S.; Wang, C.; Hobel, C.; Raju, T. Postpartum weight retention risk factors and relationship to obesity at one year. Obstet. Gynecol. 2015, 125, 144. [Google Scholar] [CrossRef]
- Huang, T.-T.; Dai, F.-T. Weight retention predictors for Taiwanese women at six-month postpartum. J. Nurs. Res. 2007, 15, 11–20. [Google Scholar] [CrossRef]
- Harris, H.; Ellison, G.; Holliday, M.; Lucassen, E. The impact of pregnancy on the long-term weight gain of primiparous women in England. Int. J. Obes. 1997, 21, 747–755. [Google Scholar] [CrossRef]
- Gunderson, E.; Abrams, B.; Selvin, S. Does the pattern of postpartum weight change differ according to pregravid body size? Int. J. Obes. 2001, 25, 853–862. [Google Scholar] [CrossRef] [PubMed]
- Rothberg, B.E.G.; Magriples, U.; Kershaw, T.S.; Rising, S.S.; Ickovics, J.R. Gestational weight gain and subsequent postpartum weight loss among young, low-income, ethnic minority women. Am. J. Obstet. Gynecol. 2011, 204, e51–e52. [Google Scholar] [CrossRef] [PubMed]
- Neville, C.; McKinley, M.; Holmes, V.; Spence, D.; Woodside, J. The relationship between breastfeeding and postpartum weight change—A systematic review and critical evaluation. Int. J. Obes. 2014, 38, 577–590. [Google Scholar] [CrossRef] [PubMed]
- Yekta, Z.; Ayatollahi, H.; Porali, R.; Farzin, A. The effect of pre-pregnancy body mass index and gestational weight gain on pregnancy outcomes in urban care settings in Urmia-Iran. BMC Pregnancy Childbirth 2006, 6, 15. [Google Scholar] [CrossRef] [PubMed]
- Ng, S.W.; Zaghloul, S.; Ali, H.; Harrison, G.; Yeatts, K.; El Sadig, M.; Popkin, B.M. Nutrition transition in the United Arab Emirates. Eur. J. Clin. Nutr. 2011, 65, 1328. [Google Scholar] [CrossRef] [PubMed]
- Institute of Medicine. Nutrition During Pregnancy: Part I, Weight Gain: Part II, Nutrient Supplements; National Academy Press: Washington, DC, USA, 1990. [Google Scholar]
- Okamoto, K.; Ohsuka, K.; Shiraishi, T.; Hukazawa, E.; Wakasugi, S.; Furuta, K. Comparability of epidemiological information between self-and interviewer-administered questionnaires. J. Clin. Epidemiol. 2002, 55, 505–511. [Google Scholar] [CrossRef]
| Participants’ Characteristics | Total (n = 183) | PWR6 below Median (≤2.4 kg) (n = 92) | PWR6 above Median (>2.4 kg) (n = 91) | p-Value |
|---|---|---|---|---|
| Maternal age (years) | 0.247 | |||
| 18–24.9 | 46 (25.7) | 22 (24.7) | 24 (25.7) | |
| 25–29.9 | 66 (36.3) | 28 (31.5) | 37 (41.1) | |
| ≥30 | 68 (38.0) | 39 (43.8) | 29 (32.2) | |
| Country of residence | 0.015 | |||
| Lebanon | 93 (50.8) | 55 (59.8) | 38 (41.8) | |
| Qatar | 90 (49.2) | 37 (40.2) | 53 (58.2) | |
| Employment status | 0.936 | |||
| Housewife | 94 (53.1) | 47 (53.4) | 47 (52.8) | |
| Employed | 83 (46.9) | 41 (46.6) | 42 (47.2) | |
| Education | 0.191 | |||
| Up to high school * | 26 (15.0) | 16 (18.6) | 10 (11.5) | |
| University or higher | 147 (85.0) | 70 (81.4) | 77 (88.5) | |
| Income | 0.466 | |||
| Low, <$1000 | 11 (10.89) | 7 (12.73) | 4 (8.7) | |
| Medium, $1000–$2000 | 24 (23.76) | 15 (27.27) | 9 (19.57) | |
| High, >$2000 | 66 (65.35) | 33 (60) | 33 (71.74) | |
| Number of children | 0.081 | |||
| 0 | 51 (30.4) | 20 (24.1) | 31 (36.5) | |
| 1 or more | 117 (69.6) | 63 (75.9) | 54 (63.5) | |
| Pre-pregnancy BMI ¶ | 0.458 | |||
| Underweight and normal (<25 kg/m2) | 98 (55.7) | 46 (52.9) | 52 (58.4) | |
| Overweight and obese (≥25 kg/m2) | 78 (44.3) | 41 (47.1) | 37 (41.6) | |
| First trimester BMI ¶ | 0.413 | |||
| Underweight and normal (<25 kg/m2) | 94 (52.5) | 44 (49.4) | 50 (55.6) | |
| Overweight and obese (≥25 kg/m2) | 85 (47.5) | 45 (50.6) | 40 (44.4) | |
| GWG (kg) | <0.001 | |||
| Insufficient | 64 (36.6) | 46 (52.9) | 18 (20.5) | |
| Adequate | 56 (32.0) | 25 (28.7) | 31 (35.2) | |
| Excessive | 55 (31.4) | 16 (18.4) | 39 (44.3) | |
| Type of delivery | 0.871 | |||
| Caesarean | 55 (30.9) | 27 (30.3) | 28 (31.5) | |
| Normal/vaginal | 123 (69.1) | 62 (69.7) | 61 (68.5) | |
| Occurrence of delivery complication | 0.113 | |||
| No | 92 (55.8) | 53 (61.6) | 39 (49.4) | |
| Yes | 73 (44.2) | 33 (38.4) | 40 (50.6) | |
| Preterm/full term delivery | 0.435 | |||
| Full term | 165 (92.7) | 83 (91.2) | 82 (94.3) | |
| Preterm | 13 (7.3) | 8 (8.8) | 5 (5.7) | |
| Exclusive Breastfeeding for 6 months | 0.302 | |||
| No | 30 (20.7) | 13 (17.3) | 17 (24.3) | |
| Yes | 115 (79.3) | 62 (82.7) | 53 (75.7) | |
| Pre-pregnancy smoking status | 0.113 | |||
| Non-smoker | 144 (78.7) | 68 (73.9) | 76 (83.5) | |
| Smoker | 39 (21.3) | 24 (26.1) | 15 (16.5) | |
| Pre-pregnancy breakfast consumption | 0.622 | |||
| Regular ** | 138 (77.1) | 68 (75.6) | 70 (78.7) | |
| Not regular | 41 (22.9) | 22 (24.4) | 19 (21.3) | |
| Physical activity | 0.364 | |||
| Low | 42 (33.1) | 18 (32.1) | 24 (33.8) | |
| Moderate | 43 (33.9) | 16 (28.6) | 27 (38.0) | |
| High | 42 (33.1) | 22 (39.3) | 20 (28.2) |
| Participants’ Characteristics | Model 1 (Crude) | Model 2 (Adjusted) | ||||
|---|---|---|---|---|---|---|
| OR | 95% CI | p-Value | OR | 95% CI | p-Value | |
| Maternal age | 0.97 | (0.92–1.03) | 0.390 | 1.04 | (0.94–1.14) | 0.493 |
| Country of residence (reference: Lebanon) | ||||||
| Qatar | 2.17 | (1.2–3.92) | 0.010 | 3.02 | (1.22–7.52) | 0.017 |
| Number of children (reference: 0) | ||||||
| 1 or more | 0.57 | (0.29–1.12) | 0.102 | 0.48 | (0.18–1.24) | 0.130 |
| GWG (reference: Adequate) | ||||||
| Insufficient | 0.32 | (0.15–0.67) | 0.003 | 0.27 | (0.1–0.69) | 0.007 |
| Excessive | 2.15 | (0.97–4.76) | 0.059 | 3.5 | (1.24–9.85) | 0.018 |
| Smoking (reference: non-smoker) | ||||||
| Smoker | 0.54 | (0.26–1.12) | 0.099 | 1.36 | (0.45–4.12) | 0.588 |
| Educational status (reference: Up to High school) | ||||||
| University or higher | 1.81 | (0.77–4.25) | 0.174 | 2.39 | (0.7–8.14) | 0.165 |
| Occurrence of Delivery Complications (reference: No) | ||||||
| Yes | 1.74 | (0.94–3.24) | 0.08 | 2.2 | (0.95–5.09) | 0.066 |
| Exclusive breastfeeding for 6 months (reference: No) | ||||||
| Yes | 1.48 | (0.66–3.32) | 0.345 | |||
| Preterm/full term delivery (reference: full term) | ||||||
| Preterm | 0.62 | (0.19–1.97) | 0.415 | |||
| Delivery type (reference: Normal/Vaginal)) | ||||||
| Caesarean section | 1.13 | (0.6–2.14) | 0.871 | |||
| Employment status (reference: Housewife) | ||||||
| Employed | 0.98 | (0.54–1.77) | 0.951 | - | - | |
| Income (Low, <$1000) | ||||||
| Medium, $1000–$2000 | 1.05 | (0.24–4.62) | 0.949 | - | - | |
| High, >$2000 | 1.75 | (0.47–6.62) | 0.406 | |||
| Pre-pregnancy BMI (reference: Underweight and normal) | ||||||
| Overweight and obese | 0.80 | (0.44–1.45) | 0.459 | - | - | |
| First trimester BMI (reference: Underweight and normal) | ||||||
| Overweight and obese | 0.82 | (0.46–1.48) | 0.508 | - | - | |
| Pre-pregnancy breakfast consumption (reference: Regular) | ||||||
| Not regular | 0.82 | (0.41–1.64) | 0.566 | - | - | |
| Physical activity (reference: Low) | ||||||
| Moderate | 1.20 | (0.5–2.87) | 0.690 | - | - | |
| High | 0.68 | (0.29–1.6) | 0.374 | - | - | |
| Participants’ Characteristics | Total | PWR6 | Country | |||||
|---|---|---|---|---|---|---|---|---|
| PWR6 ≤ 2.4 kg | PWR6 > 2.4 g | p-Value i | Lebanon | Qatar | p-Value ii | p-Value iii | ||
| Energy (Kcal) | 2853.35 ± 166.99 | 2530.86 ± 147.23 | 3172.29 ± 295.74 | 0.054 | 2374.97 ± 141.6 | 3358.9 ± 300.91 | 0.004 | 0.0241 |
| Protein (% Kcal) | 15.59 ± 0.31 | 15.2 ± 0.42 | 15.98 ± 0.45 | 0.026 | 14.19 ± 0.32 | 17.08 ± 0.5 | <0.001 | 0.607 |
| Carbohydrate (% Kcal) | 45.11 ± 0.62 | 45.15 ± 0.98 | 45.07 ± 0.77 | 0.256 | 45.31 ± 0.88 | 44.89 ± 0.89 | 0.085 | 0.141 |
| Fat (% Kcal) | 39.41 ± 0.62 | 40.12 ± 1.06 | 38.7 ± 0.63 | 0.037 | 41.54 ± 0.91 | 37.16 ± 0.76 | <0.001 | 0.349 |
| Saturated fat (% Kcal) | 11.6 ± 0.21 | 11.62 ± 0.32 | 11.57 ± 0.28 | 0.405 | 12.33 ± 0.28 | 10.82 ± 0.31 | 0.001 | 0.219 |
| Monounsaturated fat (% Kcal) | 13.7 ± 0.27 | 13.99 ± 0.46 | 13.42 ± 0.29 | 0.024 | 14.81 ± 0.4 | 12.52 ± 0.32 | <0.001 | 0.530 |
| Polyunsaturated fat (% Kcal) | 10.56 ± 0.32 | 10.89 ± 0.56 | 10.23 ± 0.31 | 0.039 | 10.58 ± 0.5 | 10.53 ± 0.41 | 0.260 | 0.457 |
| Trans fatty acid (% Kcal) | 0.24 ± 0.01 | 0.22 ± 0.02 | 0.27 ± 0.02 | 0.025 | 0.26 ± 0.02 | 0.22 ± 0.02 | 0.101 | 0.050 |
| Sugar (% Kcal) | 15.54 ± 0.47 | 15.47 ± 0.7 | 15.61 ± 0.63 | 0.242 | 16.74 ± 0.65 | 14.27 ± 0.66 | 0.761 | 0.969 |
| Cholesterol (mg/1000 Kcal) | 128.05 ± 4.08 | 125 ± 6.15 | 131.07 ± 5.4 | 0.049 | 113.09 ± 5.01 | 143.87 ± 6.11 | <0.001 | 0.279 |
| Sodium (mg/1000 Kcal) | 999.51 ± 17.82 | 980.12 ± 25.26 | 1018.69 ± 25.11 | 0.028 | 1033.63 ± 24.76 | 963.45 ± 25.24 | 0.744 | 0.765 |
| Calcium (mg/1000 Kcal) | 410.62 ± 10.49 | 403.75 ± 14.02 | 417.41 ± 15.63 | 0.11 | 445.41 ± 15.51 | 373.85 ± 13 | 0.176 | 0.188 |
| Iron (mg/1000 Kcal) | 5.43 ± 0.13 | 5.54 ± 0.22 | 5.33 ± 0.13 | 0.547 | 5.84 ± 0.22 | 5 ± 0.11 | 0.205 | 0.448 |
| Dietary fiber (g/1000 Kcal) | 8.7 ± 0.22 | 9 ± 0.33 | 8.41 ± 0.3 | 0.878 | 9.49 ± 0.32 | 7.87 ± 0.28 | 0.272 | 0.962 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nasreddine, L.; Ayoub, J.; Abbas, N.; Abdul Malik, M.; Naja, F. Postpartum Weight Retention and Its Determinants in Lebanon and Qatar: Results of the Mother and Infant Nutrition Assessment (MINA) Cohort. Int. J. Environ. Res. Public Health 2020, 17, 7851. https://doi.org/10.3390/ijerph17217851
Nasreddine L, Ayoub J, Abbas N, Abdul Malik M, Naja F. Postpartum Weight Retention and Its Determinants in Lebanon and Qatar: Results of the Mother and Infant Nutrition Assessment (MINA) Cohort. International Journal of Environmental Research and Public Health. 2020; 17(21):7851. https://doi.org/10.3390/ijerph17217851
Chicago/Turabian StyleNasreddine, Lara, Jennifer Ayoub, Nada Abbas, Mariam Abdul Malik, and Farah Naja. 2020. "Postpartum Weight Retention and Its Determinants in Lebanon and Qatar: Results of the Mother and Infant Nutrition Assessment (MINA) Cohort" International Journal of Environmental Research and Public Health 17, no. 21: 7851. https://doi.org/10.3390/ijerph17217851
APA StyleNasreddine, L., Ayoub, J., Abbas, N., Abdul Malik, M., & Naja, F. (2020). Postpartum Weight Retention and Its Determinants in Lebanon and Qatar: Results of the Mother and Infant Nutrition Assessment (MINA) Cohort. International Journal of Environmental Research and Public Health, 17(21), 7851. https://doi.org/10.3390/ijerph17217851





