Activity–Inactivity Patterns, Screen Time, and Physical Activity: The Association with Overweight, Central Obesity and Muscle Strength in Polish Teenagers. Report from the ABC of Healthy Eating Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethical Approval
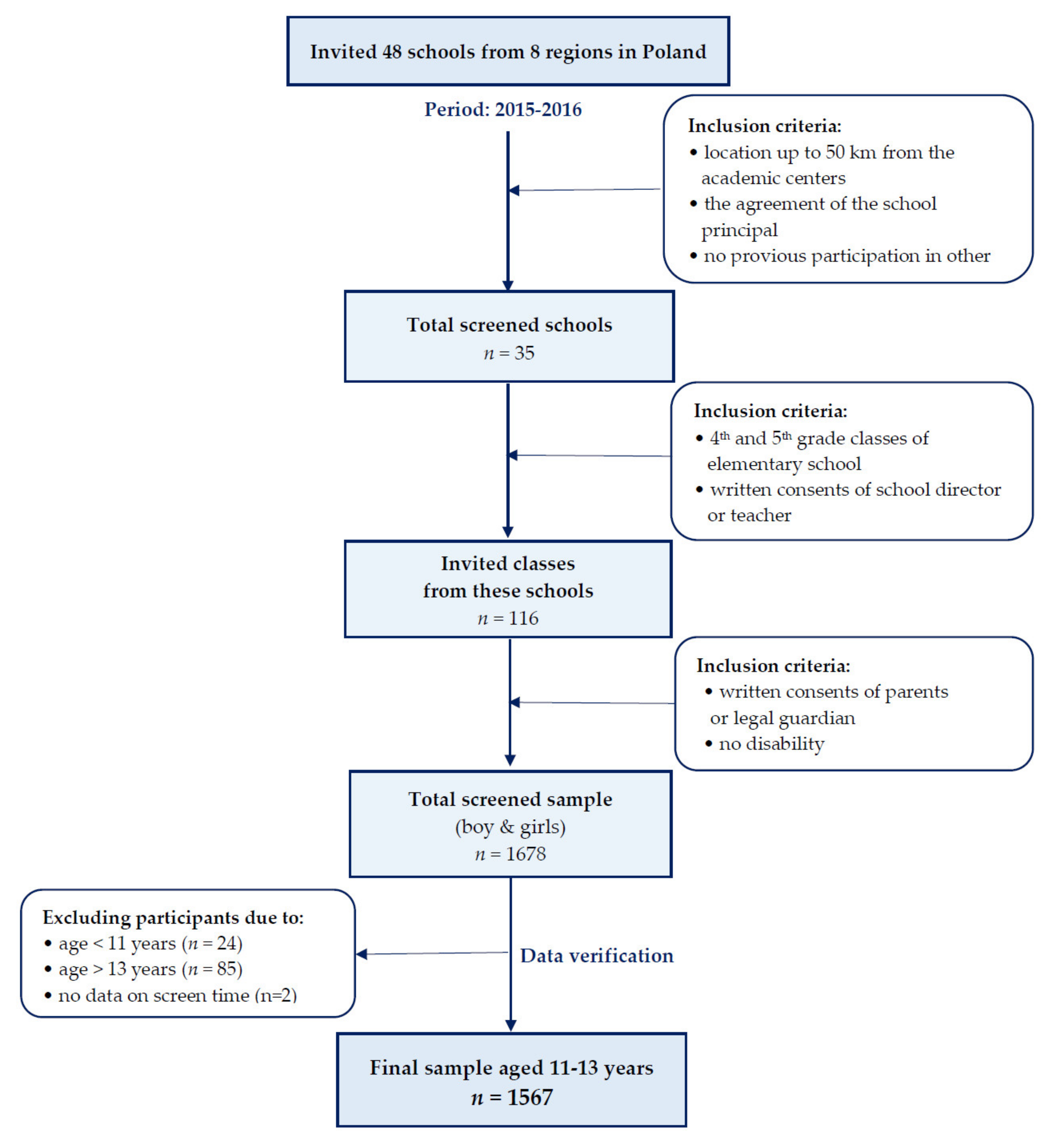
2.2. Study Design and Participants
2.3. Measures
2.3.1. Screen Time (ST)
2.3.2. Physical Activity (PA)
2.3.3. Activity–Inactivity Patterns
2.3.4. Anthropometric Data
2.3.5. Socioeconomic and Demographic Data
2.3.6. Nutrition Knowledge Score
2.4. Statistical Analysis
- The chance to fall in the category of central obesity or overweight or higher muscle strength. Activity (highST-highPA) and inactivity patterns (lowST-lowPA, highST-lowPA) were used as predictors, while the lowST-highPA pattern was used as a reference.
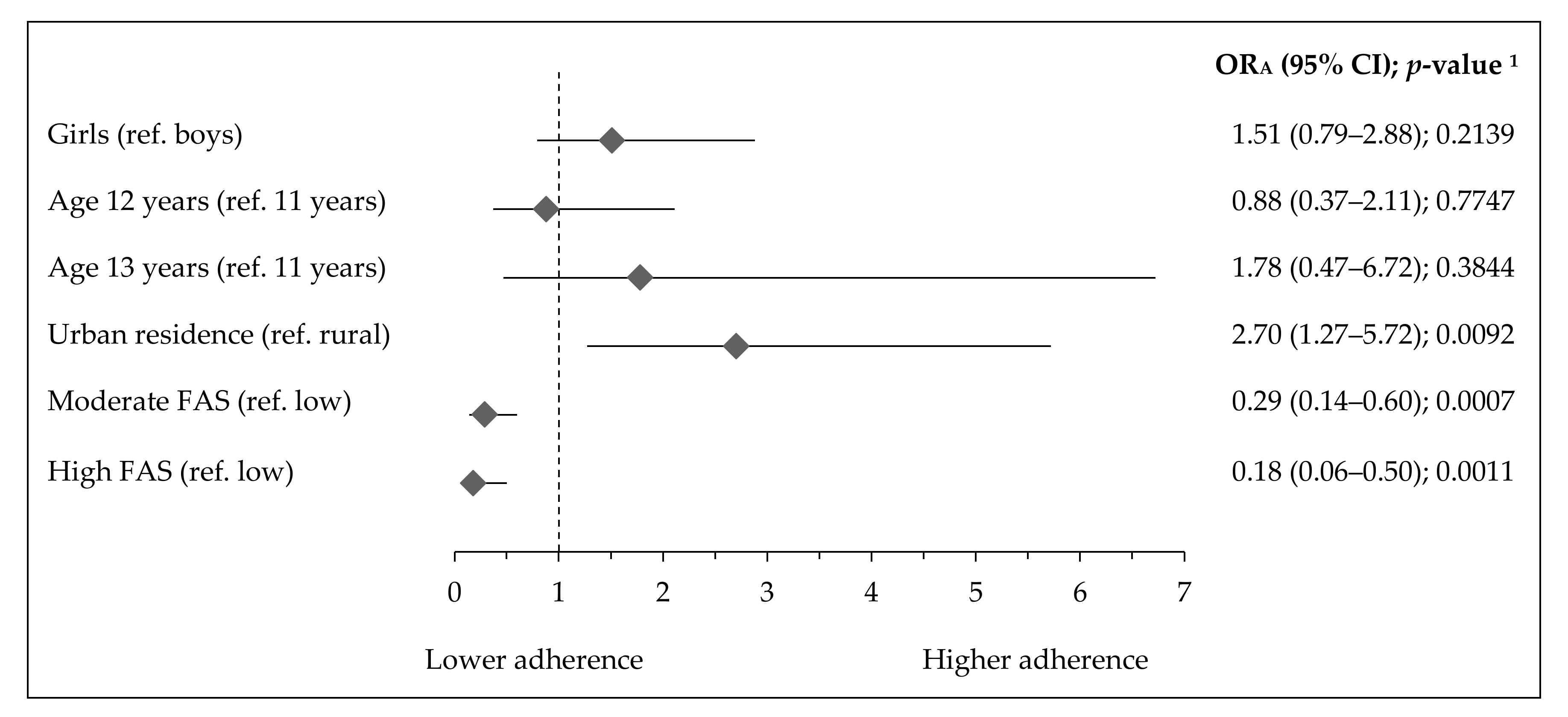
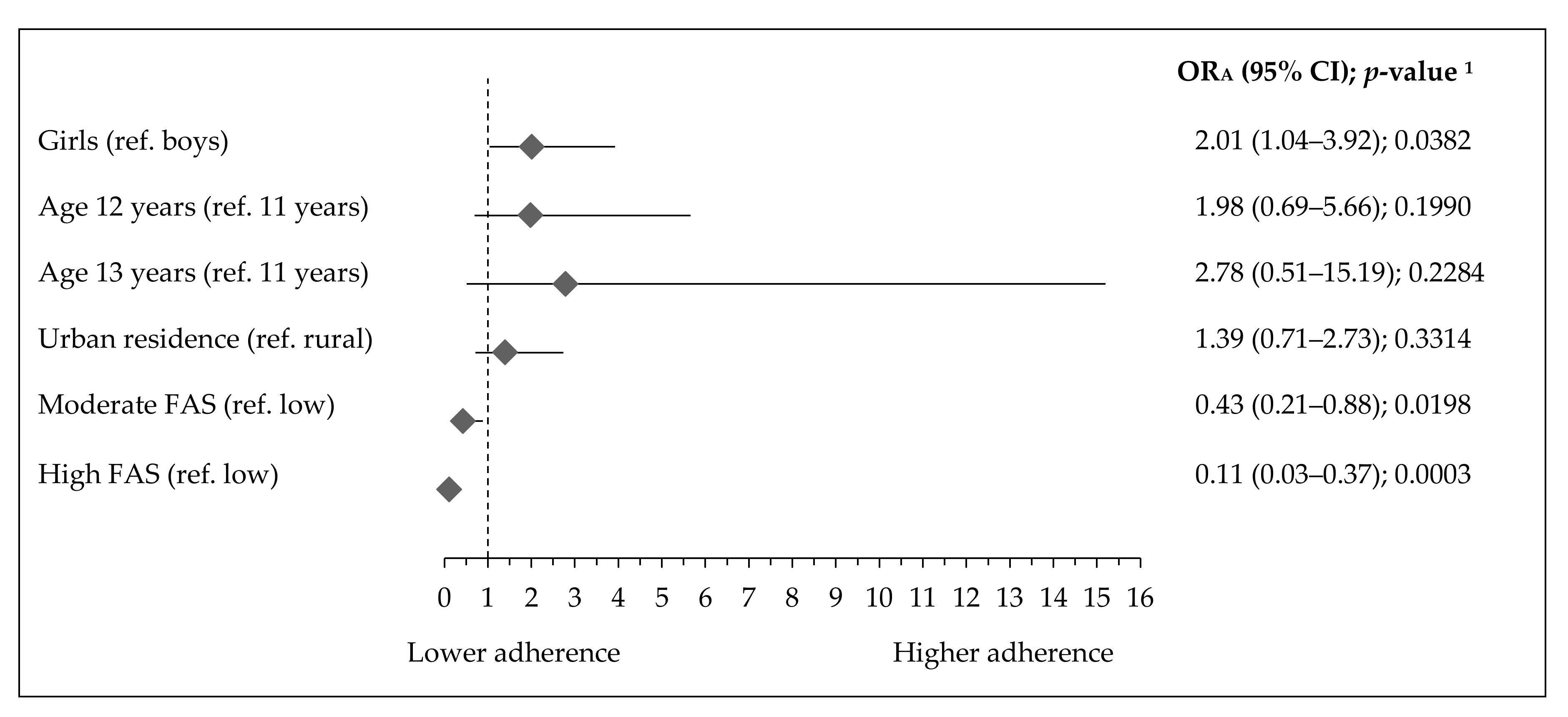
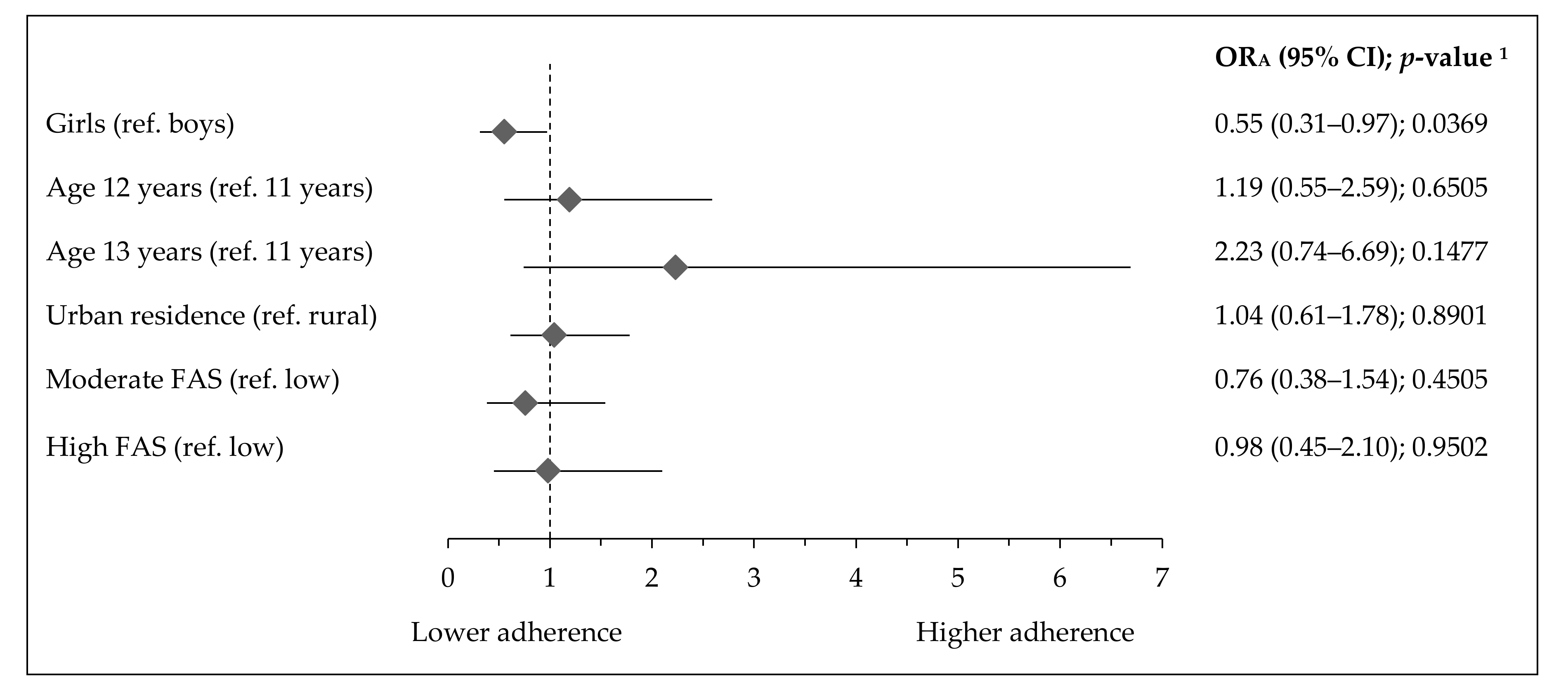
- The adherence to activity–inactivity patterns by socioeconomic and demographic factors, in respect to a referent lowST-highPA pattern, the following categorical variables were used as predictors (independent variables): gender (girls, reference: boys), age (12 or 13 years, reference: 11 years), residence (urban, reference: rural), and Family Affluence Scale (moderate or high, reference: low).
3. Results
4. Discussion
Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| 95% CI | Confidence Intervals |
| BMI | Body Mass Index |
| FAS | Family Affluence Scale |
| HGS | Hand Grip Strength |
| ORs | Odds Ratios |
| PA | Physical Activity |
| SD | Standard Deviation |
| ST | Screen Time |
| WC | Waist Circumference |
| WHtR | Waist-to-Height Ratio |
References
- Abarca-Gómez, L.; Abdeen, Z.A.; Hamid, Z.A.; Abu-Rmeileh, N.M.; Acosta-Cazares, B.; Acuin, C.; Adams, R.J.; Aekplakorn, W.; Afsana, K.; Aguilar-Salinas, C.A.; et al. Worldwide trends in body-mass index, underweight, overweight, and obesity from 1975 to 2016: A pooled analysis of 2416 population-based measurement studies in 128.9 million children, adolescents, and adults. Lancet 2017, 390, 2627–2642. [Google Scholar] [CrossRef]
- OECD Obesity Update 2017. Available online: http://www.oecd.org/health/obesity-update.htm (accessed on 5 February 2020).
- Suder, A.; Gomula, A.; Koziel, S. Central overweight and obesity in Polish schoolchildren aged 7–18 years: Secular changes of waist circumference between 1966 and 2012. Eur. J. Pediatr. 2017, 176, 909–916. [Google Scholar] [CrossRef] [PubMed]
- Schwarzfischer, P.; Gruszfeld, D.; Socha, P.; Luque, V.; Closa-Monasterolo, R.; Rousseaux, D.; Moretti, M.; Mariani, B.; Verduci, E.; Koletzko, B.; et al. Longitudinal analysis of physical activity, sedentary behaviour and anthropometric measures from ages 6 to 11 years. Int. J. Behav. Nutr. Phys. Act. 2018, 15, 126. [Google Scholar] [CrossRef]
- Serrano-Sanchez, J.A.; Martí-Trujillo, S.; Lera-Navarro, A.; Dorado-García, C.; González-Henríquez, J.J.; Sanchís-Moysi, J. Associations between Screen Time and Physical Activity among Spanish Adolescents. PLoS ONE 2011, 6, e24453. [Google Scholar] [CrossRef] [PubMed]
- Moschonis, G.; Kaliora, A.C.; Karatzi, K.; Michaletos, A.; Lambrinou, C.-P.; Karachaliou, A.K.; Chrousos, G.P.; Lionis, C.; Manios, Y. Perinatal, sociodemographic and lifestyle correlates of increased total and visceral fat mass levels in schoolchildren in Greece: The Healthy Growth Study. Public Health Nutr. 2017, 20, 660–670. [Google Scholar] [CrossRef]
- Lajous, M.; Chavarro, J.; Peterson, K.E.; Hernández-Prado, B.; Cruz-Valdéz, A.; Hernández-Ávila, M.; Lazcano-Ponce, E. Screen time and adiposity in adolescents in Mexico. Public Health Nutr. 2009, 12, 1938–1945. [Google Scholar] [CrossRef]
- Epstein, L.H.; Roemmich, J.N.; Robinson, J.L.; Paluch, R.A.; Winiewicz, D.D.; Fuerch, J.H.; Robinson, T.N. A Randomized Trial of the Effects of Reducing Television Viewing and Computer Use on Body Mass Index in Young Children. Arch. Pediatr. Adolesc. Med. 2008, 162, 239. [Google Scholar] [CrossRef]
- Fletcher, E.A.; McNaughton, S.A.; Crawford, D.; Cleland, V.; Della Gatta, J.; Hatt, J.; Dollman, J.; Timperio, A. Associations between sedentary behaviours and dietary intakes among adolescents. Public Health Nutr. 2018, 21, 1115–1122. [Google Scholar] [CrossRef]
- Pearson, N.; Griffiths, P.; Biddle, S.J.; Johnston, J.P.; McGeorge, S.; Haycraft, E. Clustering and correlates of screen-time and eating behaviours among young adolescents. BMC Public Health 2017, 17, 533. [Google Scholar] [CrossRef]
- Abbade, E.B.; Dewes, H. Behavioral and societal drivers of an obesogenic environment worldwide. Nutr. Food Sci. 2015, 45, 229–241. [Google Scholar] [CrossRef]
- Katzmarzyk, P.T.; Church, T.S.; Craig, C.L.; Bouchard, C. Sitting Time and Mortality from All Causes, Cardiovascular Disease, and Cancer. Med. Sci. Sports Exerc. 2009, 41, 998–1005. [Google Scholar] [CrossRef]
- Hamilton, M.T.; Healy, G.N.; Dunstan, D.W.; Zderic, T.W.; Owen, N. Too little exercise and too much sitting: Inactivity physiology and the need for new recommendations on sedentary behavior. Curr. Cardio Risk. Rep. 2008, 2, 292–298. [Google Scholar] [CrossRef] [PubMed]
- McManus, A.M.; Mellecker, R.R. Physical activity and obese children. J. Sport Health Sci. 2012, 1, 141–148. [Google Scholar] [CrossRef]
- Głąbska, D.; Guzek, D.; Mellová, B.; Zadka, K.; Żywczyk, K.; Gutkowska, K. The National After-School Athletics Program Participation as a Tool to Reduce the Risk of Obesity in Adolescents after One Year of Intervention: A Nationwide Study. Int. J. Environ. Res. Public Health 2019, 16, 405. [Google Scholar] [CrossRef] [PubMed]
- Iannotti, R.J.; Kogan, M.D.; Janssen, I.; Boyce, W.F. Patterns of Adolescent Physical Activity, Screen-Based Media Use, and Positive and Negative Health Indicators in the U.S. and Canada. J. Adolesc. Health 2009, 44, 493–499. [Google Scholar] [CrossRef] [PubMed]
- Hallal, P.C.; Andersen, L.B.; Bull, F.C.; Guthold, R.; Haskell, W.; Ekelund, U. Global physical activity levels: Surveillance progress, pitfalls, and prospects. Lancet 2012, 380, 247–257. [Google Scholar] [CrossRef]
- Henriksson, H.; Henriksson, P.; Tynelius, P.; Ekstedt, M.; Berglind, D.; Labayen, I.; Ruiz, J.R.; Lavie, C.J.; Ortega, F.B. Cardiorespiratory fitness, muscular strength, and obesity in adolescence and later chronic disability due to cardiovascular disease: A cohort study of 1 million men. Eur. Heart J. 2020, 41, 1503–1510. [Google Scholar] [CrossRef]
- Ng, A.K.; Hairi, N.N.; Jalaludin, M.Y.; Majid, H.A. Dietary intake, physical activity and muscle strength among adolescents: The Malaysian Health and Adolescents Longitudinal Research Team (MyHeART) study. BMJ Open 2019, 9, e026275. [Google Scholar] [CrossRef]
- Martínez-Gómez, D.; Welk, G.J.; Puertollano, M.A.; del-Campo, J.; Moya, J.M.; Marcos, A.; Veiga, O.L.; AFINOS Study Group. Associations of physical activity with muscular fitness in adolescents: Physical activity and muscular fitness in adolescents. Scand. J. Med. Sci. Sports 2011, 21, 310–317. [Google Scholar] [CrossRef]
- Jiménez-Pavón, D.; Fernández-Vázquez, A.; Alexy, U.; Pedrero, R.; Cuenca-García, M.; Polito, A.; Vanhelst, J.; Manios, Y.; Kafatos, A.; Molnar, D.; et al. Association of objectively measured physical activity with body components in European adolescents. BMC Public Health 2013, 13, 667. [Google Scholar] [CrossRef]
- WHO. Global Recommendations on Physical Activity for Health; World Health Organization: Genève, Switzerland, 2010; ISBN 978-92-4-159997-9. [Google Scholar]
- Pyramid of Healthy Nutrition and Physical Activity for Children. Available online: http://www.izz.waw.pl/zasady-prawidowego-ywienia (accessed on 18 April 2020). (In Polish).
- American Academy of Pediatrics. Committee on Public Education. American Academy of Pediatrics: Children, adolescents, and television. Pediatrics 2001, 107, 423–426. [Google Scholar] [CrossRef] [PubMed]
- Pearson, N.; Braithwaite, R.E.; Biddle, S.J.H.; van Sluijs, E.M.F.; Atkin, A.J. Associations between sedentary behaviour and physical activity in children and adolescents: A meta-analysis: Active and sedentary behaviours in youth. Obes. Rev. 2014, 15, 666–675. [Google Scholar] [CrossRef] [PubMed]
- Hamulka, J.; Wadolowska, L.; Hoffmann, M.; Kowalkowska, J.; Gutkowska, K. Effect of an Education Program on Nutrition Knowledge, Attitudes toward Nutrition, Diet Quality, Lifestyle, and Body Composition in Polish Teenagers. The ABC of Healthy Eating Project: Design, Protocol, and Methodology. Nutrients 2018, 10, 1439. [Google Scholar] [CrossRef] [PubMed]
- Wadolowska, L.; Hamulka, J.; Kowalkowska, J.; Ulewicz, N.; Gornicka, M.; Jeruszka-Bielak, M.; Kostecka, M.; Wawrzyniak, A. Skipping Breakfast and a Meal at School: Its Correlates in Adiposity Context. Report from the ABC of Healthy Eating Study of Polish Teenagers. Nutrients 2019, 11, 1563. [Google Scholar] [CrossRef]
- Wadolowska, L.; Hamulka, J.; Kowalkowska, J.; Kostecka, M.; Wadolowska, K.; Biezanowska-Kopec, R.; Czarniecka-Skubina, E.; Kozirok, W.; Piotrowska, A. Prudent-Active and Fast-Food-Sedentary Dietary-Lifestyle Patterns: The Association with Adiposity, Nutrition Knowledge and Sociodemographic Factors in Polish Teenagers—The ABC of Healthy Eating Project. Nutrients 2018, 10, 1988. [Google Scholar] [CrossRef]
- Kowalkowska, J.; Wadolowska, L.; Hamulka, J.; Wojtas, N.; Czlapka-Matyasik, M.; Kozirok, W.; Bronkowska, M.; Sadowska, J.; Naliwajko, S.; Dziaduch, I.; et al. Reproducibility of a Short-Form, Multicomponent Dietary Questionnaire to Assess Food Frequency Consumption, Nutrition Knowledge, and Lifestyle (SF-FFQ4PolishChildren) in Polish Children and Adolescents. Nutrients 2019, 11, 2929. [Google Scholar] [CrossRef]
- Council on Communications and Media Media Use in School-Aged Children and Adolescents. Pediatrics 2016, 138, e20162592. [CrossRef]
- Guram, S.; Heinz, P. Media use in children: American Academy of Pediatrics recommendations 2016. Arch. Dis. Child. Edu. Pract. 2018, 103, 99–101. [Google Scholar] [CrossRef]
- Ortega, F.B.; Cadenas-Sanchez, C.; Lee, D.; Ruiz, J.R.; Blair, S.N.; Sui, X. Fitness and Fatness as Health Markers through the Lifespan: An Overview of Current Knowledge. Prog. Prev. Med. 2018, 3, e0013. [Google Scholar] [CrossRef]
- Australian 24-Hour Movement Guidelines for Children and Young People (5–17 Years)—An Integration of Physical Activity, Sedentary Behaviour and Sleep. Available online: https://www1.health.gov.au/internet/main/publishing.nsf/Content/health-24-hours-phys-act-guidelines (accessed on 10 December 2019).
- International Society for the Advancement of Kinanthropometry (ISAK). International Standards for Anthropometric Assessment; International Society for the Advancement of Kinanthropometry: Potchefstroom, South Africa, 2001. [Google Scholar]
- Cole, T.J.; Lobstein, T. Extended international (IOTF) body mass index cut-offs for thinness, overweight and obesity: Extended international BMI cut-offs. Pediatric Obes. 2012, 7, 284–294. [Google Scholar] [CrossRef]
- Ashwell, M.; Gunn, P.; Gibson, S. Waist-to-height ratio is a better screening tool than waist circumference and BMI for adult cardiometabolic risk factors: Systematic review and meta-analysis: Waist-to-height ratio as a screening tool. Obes. Rev. 2012, 13, 275–286. [Google Scholar] [CrossRef] [PubMed]
- Mazur, J. Family Affluence Scale—Validation study and suggested modification. Hygeia Public Health 2013, 48, 211–217. [Google Scholar]
- Whati, L.H.; Senekal, M.; Steyn, N.P.; Nel, J.H.; Lombard, C.; Norris, S. Development of a reliable and valid nutritional knowledge questionnaire for urban South African adolescents. Nutrition 2005, 21, 76–85. [Google Scholar] [CrossRef] [PubMed]
- Heshmat, R.; Qorbani, M.; Shahr Babaki, A.E.; Djalalinia, S.; Ataei-Jafari, A.; Motlagh, M.E.; Ardalan, G.; Arefirad, T.; Rezaei, F.; Asayesh, H.; et al. Joint Association of Screen Time and Physical Activity with Cardiometabolic Risk Factors in a National Sample of Iranian Adolescents: The CASPIANIII Study. PLoS ONE 2016, 11, e0154502. [Google Scholar] [CrossRef] [PubMed]
- Cristi-Montero, C.; Chillón, P.; Labayen, I.; Casajus, J.A.; Gonzalez-Gross, M.; Vanhelst, J.; Manios, Y.; Moreno, L.A.; Ortega, F.B.; Ruiz, J.R. Cardiometabolic risk through an integrative classification combining physical activity and sedentary behavior in European adolescents: HELENA study. J. Sport Health Sci. 2019, 8, 55–62. [Google Scholar] [CrossRef]
- Throuvala, M.A.; Griffiths, M.D.; Rennoldson, M.; Kuss, D.J. The Role of Recreational Online Activities in School-Based Screen Time Sedentary Behaviour Interventions for Adolescents: A Systematic and Critical Literature Review. Int. J. Ment. Health Addict. 2020. [Google Scholar] [CrossRef]
- Biddle, S.J.H.; García Bengoechea, E.; Wiesner, G. Sedentary behaviour and adiposity in youth: A systematic review of reviews and analysis of causality. Int. J. Behav. Nutr. Phys. Act. 2017, 14, 43. [Google Scholar] [CrossRef] [PubMed]
- Yan, H.; Zhang, R.; Oniffrey, T.; Chen, G.; Wang, Y.; Wu, Y.; Zhang, X.; Wang, Q.; Ma, L.; Li, R.; et al. Associations among Screen Time and Unhealthy Behaviors, Academic Performance, and Well-Being in Chinese Adolescents. Int. J. Environ. Res. Public Health 2017, 14, 596. [Google Scholar] [CrossRef]
- Xu, F.; Adams, S.K.; Cohen, S.A.; Earp, J.E.; Greaney, M.L. Relationship between Physical Activity, Screen Time, and Sleep Quantity and Quality in US Adolescents Aged 16–19. Int. J. Environ. Res. Public Health 2019, 16, 1524. [Google Scholar] [CrossRef]
- Brambilla, P.; Bedogni, G.; Heo, M.; Pietrobelli, A. Waist circumference-to-height ratio predicts adiposity better than body mass index in children and adolescents. Int. J. Obes. 2013, 37, 943–946. [Google Scholar] [CrossRef]
- Lo, K.; Wong, M.; Khalechelvam, P.; Tam, W. Waist-to-height ratio, body mass index and waist circumference for screening paediatric cardio-metabolic risk factors: A meta-analysis: Screening cardio-metabolic risk factors. Obes. Rev. 2016, 17, 1258–1275. [Google Scholar] [CrossRef] [PubMed]
- Savva, S.; Tornaritis, M.; Savva, M.; Kourides, Y.; Panagi, A.; Silikiotou, N.; Georgiou, C.; Kafatos, A. Waist circumference and waist-to-height ratio are better predictors of cardiovascular disease risk factors in children than body mass index. Int. J. Obes. 2000, 24, 1453–1458. [Google Scholar] [CrossRef] [PubMed]
- Engberg, E.; Figueiredo, R.A.O.; Rounge, T.B.; Weiderpass, E.; Viljakainen, H. Heavy screen users are the heaviest among 10,000 children. Sci. Rep. 2019, 9, 11158. [Google Scholar] [CrossRef] [PubMed]
- Milliken, L.A.; Faigenbaum, A.D.; Loud, R.L.; Westcott, W.L. Correlates of Upper and Lower Body Muscular Strength in Children. J. Strength Cond. Res. 2008, 22, 1339–1346. [Google Scholar] [CrossRef] [PubMed]
- Chan, D.C.C.; Lee, W.T.K.; Lo, D.H.S.; Leung, J.C.S.; Kwok, A.W.L.; Leung, P.C. Relationship between grip strength and bone mineral density in healthy Hong Kong adolescents. Osteoporos Int. 2008, 19, 1485–1495. [Google Scholar] [CrossRef] [PubMed]
- Edelson, L.R.; Mathias, K.C.; Fulgoni, V.L.; Karagounis, L.G. Screen-based sedentary behavior and associations with functional strength in 6–15 year-old children in the United States. BMC Public Health 2015, 16, 116. [Google Scholar] [CrossRef] [PubMed]
- Kardefelt-Winther, D. How Does the Time Children Spend Using Digital Technology Impact Their Mental Well-Being, Social Relationships and Physical Activity? An Evidence-Focused Literature Review. Available online: https://www.unicefirc.org/publications/pdf/Children-digital-technology-wellbeing.pdf (accessed on 23 April 2020).
- Hale, D.R.; Fitzgerald-Yau, N.; Viner, R.M. A Systematic Review of Effective Interventions for Reducing Multiple Health Risk Behaviors in Adolescence. Am. J. Public Health 2014, 104, e19–e41. [Google Scholar] [CrossRef]
- van Grieken, A.; Ezendam, N.P.; Paulis, W.D.; van der Wouden, J.C.; Raat, H. Primary prevention of overweight in children and adolescents: A meta-analysis of the effectiveness of interventions aiming to decrease sedentary behaviour. Int. J. Behav Nutr. Phys. Act. 2012, 9, 61. [Google Scholar] [CrossRef]
- Pujadas Botey, A.; Bayrampour, H.; Carson, V.; Vinturache, A.; Tough, S. Adherence to Canadian physical activity and sedentary behaviour guidelines among children 2 to 13 years of age. Prev. Med. Rep. 2016, 3, 14–20. [Google Scholar] [CrossRef][Green Version]
- Garcia-Continente, X.; Perez-Gimenez, A.; Espelt, A.; Nebot Adell, M. Factors associated with media use among adolescents: A multilevel approach. Eur. J. Public Health 2013, 24. [Google Scholar] [CrossRef]
- Wethington, H.; Sherry, B.; Park, S.; Blanck, H.M.; Fulton, J.E. Active Screen Time Among, U.S. Youth Aged 9–18 Years, 2009. Games Health J. 2013, 2, 362–368. [Google Scholar] [CrossRef]
- Sirard, J.R.; Bruening, M.; Wall, M.M.; Eisenberg, M.E.; Kim, S.K.; Neumark-Sztainer, D. Physical Activity and Screen Time in Adolescents and Their Friends. Am. J. Prev Med. 2013, 44, 48–55. [Google Scholar] [CrossRef]
- Finne, E.; Bucksch, J.; Lampert, T.; Kolip, P. Physical activity and screen-based media use: Cross-sectional associations with health-related quality of life and the role of body satisfaction in a representative sample of German adolescents. Health Psychol. Behav. Med. 2013, 1, 15–30. [Google Scholar] [CrossRef] [PubMed]
- Brophy, S.; Crowley, A.; Mistry, R.; Hill, R.; Choudhury, S.; Thomas, N.E.; Rapport, F. Recommendations to improve physical activity among teenagers—A qualitative study with ethnic minority and European teenagers. BMC Public Health 2011, 11, 412. [Google Scholar] [CrossRef]
- Marques, A.; da Costa, F.C. Levels of Physical Activity of Urban Adolescents According to Age and Gender. Int. J. Sports Sci. 2013, 3, 23–27. [Google Scholar] [CrossRef]
- Larson, L.R.; Szczytko, R.; Bowers, E.P.; Stephens, L.E.; Stevenson, K.T.; Floyd, M.F. Outdoor Time, Screen Time, and Connection to Nature: Troubling Trends Among Rural Youth? Environ. Behav. 2018, 51, 966–991. [Google Scholar] [CrossRef]
- Arundell, L.; Salmon, J.; Koorts, H.; Contardo Ayala, A.M.; Timperio, A. Exploring when and how adolescents sit: Cross-sectional analysis of activPAL-measured patterns of daily sitting time, bouts and breaks. BMC Public Health 2019, 19, 653. [Google Scholar] [CrossRef] [PubMed]
- Haidar, A.; Ranjit, N.; Archer, N.; Hoelscher, D.M. Parental and peer social support is associated with healthier physical activity behaviours in adolescents: A cross-sectional analysis of Texas School Physical Activity and Nutrition (TX SPAN) data. BMC Public Health 2019, 19, 640. [Google Scholar] [CrossRef]
- De Lucena, J.M.S.; Cheng, L.A.; Cavalcante, T.L.M.; da Silva, V.A.; de Farias Júnior, J.C. Prevalência de tempo excessivo de tela e fatores associados em adolescentes. Rev. Paul. De Pediatr. 2015, 33, 407–414. [Google Scholar] [CrossRef] [PubMed]
- Melkevik, O.; Torsheim, T.; Iannotti, R.J.; Wold, B. Is spending time in screen-based sedentary behaviours associated with less physical activity: A cross national investigation. Int. J. Behav. Nutr. Phys. Act. 2010, 7, 46. [Google Scholar] [CrossRef] [PubMed]
- Syed, N.; Schneider, M. Determining If Differences in Socioeconomic Status Are Associated With Screen Time Behavior and Physical Activity Levels. J. Nutr. Educ. Behav. 2016, 48, S58–S59. [Google Scholar] [CrossRef]
- Hankonen, N.; Heino, M.T.J.; Kujala, E.; Hynynen, S.-T.; Absetz, P.; Araújo-Soares, V.; Borodulin, K.; Haukkala, A. What explains the socioeconomic status gap in activity? Educational differences in determinants of physical activity and screentime. BMC Public Health 2017, 17, 144. [Google Scholar] [CrossRef]
- Drenowatz, C.; Eisenmann, J.C.; Pfeiffer, K.A.; Welk, G.; Heelan, K.; Gentile, D.; Walsh, D. Influence of socio-economic status on habitual physical activity and sedentary behavior in 8- to 11-year old children. BMC Public Health 2010, 10, 214. [Google Scholar] [CrossRef] [PubMed]
- Lisinskiene, A.; Juskeliene, V. Links between Adolescents’ Engagement in Physical Activity and Their Attachment to Mothers, Fathers, and Peers. Int. J. Environ. Res. Public Health 2019, 16, 866. [Google Scholar] [CrossRef] [PubMed]
- Yi, X.; Fu, Y.; Burns, R.D.; Bai, Y.; Zhang, P. Body mass index and physical fitness among Chinese adolescents from Shandong Province: A cross-sectional study. BMC Public Health 2019, 19, 81. [Google Scholar] [CrossRef] [PubMed]
- Regis, M.F.; Oliveira, L.M.F.T.d.; Santos, A.R.M.d.; Leonidio, A.d.C.R.; Diniz, P.R.B.; Freitas, C.M.S.M.d. Urban versus rural lifestyle in adolescents: Associations between environment, physical activity levels and sedentary behavior. Einstein (São Paulo) 2016, 14, 461–467. [Google Scholar] [CrossRef]
- Neto, A.F.; Eto, F.N.; Silva Pereira, T.S.; Carletti, L.; Bisi Molina, M.d.C. Active and sedentary behaviours in children aged 7 to 10 years old: The urban and rural contexts, Brazil. BMC Public Health 2014, 14, 1174. [Google Scholar] [CrossRef]
- Machado-Rodrigues, A.M.; Coelho-E.-Silva, M.J.; Mota, J.; Padez, C.; Martins, R.A.; Cumming, S.P.; Riddoch, C.; Malina, R.M. Urban-rural contrasts in fitness, physical activity, and sedentary behaviour in adolescents. Health Promot. Inter. 2014, 29, 118–129. [Google Scholar] [CrossRef]
- Weinberg, D.; Stevens, G.W.J.M.; Bucksch, J.; Inchley, J.; de Looze, M. Do country-level environmental factors explain cross-national variation in adolescent physical activity? A multilevel study in 29 European countries. BMC Public Health 2019, 19, 680. [Google Scholar] [CrossRef]
- Fijałkowska, A. Current Evaluation of the Physical Activity Level Children and Teenagers Aged 3–19 Years in Poland; Ministry of Sport and Tourism: Warsaw, Poland, 2018.
- Del Boca, F.K.; Noll, J.A. Truth or consequences: The validity of self-report data in health services research on addictions. Addiction 2000, 95, 347–360. [Google Scholar] [CrossRef]



| Physical Activity | Screen Time | Patterns * | ||
|---|---|---|---|---|
| Low | Moderate | High | ||
| low | lowST-lowPA | other | highST-lowPA THE MOST INACTIVE PATTERN | inactive |
| moderate | other | other | other | --- |
| high | lowST-highPA THE MOST ACTIVE PATTERN | other | highST-highPA | active |
| Variables | Total Sample | Screen Time (h/day) | p-Value 2 | Physical Activity | p-Value | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| N | % | <2 | 2 to <4 | ≥4 | Low | Moderate | High | |||
| Sample size 1 | 1567 | 726 | 539 | 302 | 154 | 923 | 490 | |||
| Sample percentage | 100.0 | 46.3 | 34.4 | 19.3 | 9.8 | 58.9 | 31.3 | |||
| Gender | ||||||||||
| boys | 758 | 48.4 | 42.1 | 35.2 | 22.7 | 0.0006 | 10.0 | 53.3 | 36.7 | <0.0001 |
| girls | 809 | 51.6 | 50.3 | 33.6 | 16.1 | 9.5 | 64.2 | 26.3 | ||
| Age (years) | ||||||||||
| 11 | 260 | 16.6 | 47.3 | 37.3 | 15.4 | 0.4753 | 8.4 | 58.2 | 33.3 | 0.3168 |
| 12 | 1153 | 73.6 | 46.3 | 33.8 | 19.9 | 10.0 | 58.2 | 31.8 | ||
| 13 | 154 | 9.8 | 44.8 | 33.8 | 21.4 | 10.5 | 65.4 | 24.2 | ||
| Residence | ||||||||||
| rural | 631 | 40.3 | 46.8 | 37.4 | 15.8 | 0.0099 | 7.9 | 58.2 | 33.9 | 0.0507 |
| urban | 936 | 59.7 | 46.0 | 32.4 | 21.6 | 11.0 | 59.4 | 29.6 | ||
| Family Affluence Scale (categories) | ||||||||||
| low | 381 | 24.3 | 40.9 | 36.5 | 22.6 | 0.0293 | 15.0 | 58.8 | 26.2 | <0.0001 |
| moderate | 781 | 49.9 | 49.7 | 31.6 | 18.7 | 9.5 | 59.2 | 31.3 | ||
| high | 403 | 25.8 | 45.2 | 37.5 | 17.4 | 5.4 | 58.2 | 36.4 | ||
| Family Affluence Scale (0–7 points) | ||||||||||
| Mean ± SD | 1565 | 99.9 | 5.4 ± 1.5 | 5.4 ± 1.5 | 5.2 ± 1.6 | 0.1208 | 4.7 ± 1.8 | 5.4 ± 1.5 | 5.5 ± 1.4 | <0.0001 |
| Nutrition Knowledge Score (0–18 points) | ||||||||||
| Mean ± SD | 1566 | 99.9 | 6.2 ± 2.8 | 6.2 ± 2.8 | 5.5 ± 2.8 | 0.0003 | 5.8 ± 2.9 | 6.0 ± 2.8 | 6.3 ± 2.9 | 0.1619 |
| Variables | n | Activity-Inactivity Patterns (N = 1567) | p-Value 1 | |||
|---|---|---|---|---|---|---|
| LowST-HighPA | HighST-HighPA | LowST-LowPA | HighST-LowPA | |||
| N | 438 | 261 | 76 | 50 | 51 | |
| % N | 28.0 | 16.7 | 4.8 | 3.2 | 3.3 | |
| Gender | ||||||
| boys | 241 | 18.6 | 7.1 | 2.8 | 3.3 | 0.0002 |
| girls | 197 | 14.9 | 2.7 | 3.6 | 3.2 | |
| Age (years) | ||||||
| 11 | 68 | 16.9 | 3.8 | 1.9 | 3.5 | 0.6596 |
| 12 | 332 | 17.2 | 4.9 | 3.6 | 3.1 | |
| 13 | 38 | 12.4 | 6.5 | 2.6 | 3.9 | |
| Residence | ||||||
| rural | 171 | 17.3 | 5.1 | 3.0 | 1.7 | 0.0914 |
| urban | 267 | 16.3 | 4.7 | 3.3 | 4.3 | |
| Family Affluence Scale (categories) | ||||||
| low | 104 | 11.8 | 4.5 | 5.0 | 6.3 | <0.0001 |
| moderate | 218 | 17.3 | 4.4 | 3.5 | 2.7 | |
| high | 116 | 20.1 | 6.2 | 1.0 | 1.5 | |
| Family Affluence Scale (0–7 points) | ||||||
| Mean ± SD | 438 | 5.6 ± 1.4 | 5.5 ± 1.5 | 4.4 ± 1.9 | 4.5 ± 1.7 | <0.0001 |
| Nutrition Knowledge Score (0–18 points) | ||||||
| Mean ± SD | 438 | 6.4 ± 2.8 | 5.4 ± 2.6 | 6.2 ± 3.0 | 5.3 ± 2.8 | 0.0016 |
| Variables | Gender-Age-Related-BMI 2 | p-Value 5 | Central Obesity 3 | p-Value | Muscle Strength 4 | p-Value | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| Underweight | Normal Weight | Overweight | Lower | Normal | Higher | |||||
| Sample size 1 | 145 | 980 | 368 | 182 | 147 | 892 | 148 | |||
| Sample percentage | 9.7 | 65.6 | 24.6 | 12.2 | 12.4 | 75.1 | 12.5 | |||
| Activity-inactivity patterns | ||||||||||
| lowST-highPA | 11.2 | 77.2 | 11.6 | <0.0001 | 4.4 | <0.0001 | 9.8 | 77.8 | 12.4 | 0.3367 |
| highST-highPA | 8.2 | 68.5 | 23.3 | 8.2 | 5.3 | 77.2 | 17.5 | |||
| lowST-lowPA | 6.4 | 59.6 | 34.0 | 23.4 | 22.2 | 69.4 | 8.3 | |||
| highST-lowPA | 6.3 | 54.2 | 39.6 | 20.8 | 16.7 | 72.2 | 11.1 | |||
| Screen time (hours/day) | ||||||||||
| <2 | 11.1 | 70.6 | 18.3 | <0.0001 | 9.2 | 0.0004 | 11.9 | 76.9 | 11.2 | 0.6425 |
| 2 to <4 | 9.2 | 61.7 | 29.1 | 12.9 | 13.2 | 74.2 | 12.7 | |||
| ≥4 | 7.3 | 60.5 | 32.2 | 18.2 | 12.2 | 73.0 | 14.9 | |||
| Physical activity | ||||||||||
| low | 5.6 | 52.1 | 42.4 | <0.0001 | 22.2 | <0.0001 | 16.5 | 75.7 | 7.8 | 0.0072 |
| moderate | 10.2 | 63.6 | 26.1 | 13.8 | 13.3 | 75.8 | 10.9 | |||
| high | 9.8 | 73.6 | 16.6 | 6.2 | 9.3 | 73.9 | 16.8 | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Górnicka, M.; Hamulka, J.; Wadolowska, L.; Kowalkowska, J.; Kostyra, E.; Tomaszewska, M.; Czeczelewski, J.; Bronkowska, M. Activity–Inactivity Patterns, Screen Time, and Physical Activity: The Association with Overweight, Central Obesity and Muscle Strength in Polish Teenagers. Report from the ABC of Healthy Eating Study. Int. J. Environ. Res. Public Health 2020, 17, 7842. https://doi.org/10.3390/ijerph17217842
Górnicka M, Hamulka J, Wadolowska L, Kowalkowska J, Kostyra E, Tomaszewska M, Czeczelewski J, Bronkowska M. Activity–Inactivity Patterns, Screen Time, and Physical Activity: The Association with Overweight, Central Obesity and Muscle Strength in Polish Teenagers. Report from the ABC of Healthy Eating Study. International Journal of Environmental Research and Public Health. 2020; 17(21):7842. https://doi.org/10.3390/ijerph17217842
Chicago/Turabian StyleGórnicka, Magdalena, Jadwiga Hamulka, Lidia Wadolowska, Joanna Kowalkowska, Eliza Kostyra, Marzena Tomaszewska, Jan Czeczelewski, and Monika Bronkowska. 2020. "Activity–Inactivity Patterns, Screen Time, and Physical Activity: The Association with Overweight, Central Obesity and Muscle Strength in Polish Teenagers. Report from the ABC of Healthy Eating Study" International Journal of Environmental Research and Public Health 17, no. 21: 7842. https://doi.org/10.3390/ijerph17217842
APA StyleGórnicka, M., Hamulka, J., Wadolowska, L., Kowalkowska, J., Kostyra, E., Tomaszewska, M., Czeczelewski, J., & Bronkowska, M. (2020). Activity–Inactivity Patterns, Screen Time, and Physical Activity: The Association with Overweight, Central Obesity and Muscle Strength in Polish Teenagers. Report from the ABC of Healthy Eating Study. International Journal of Environmental Research and Public Health, 17(21), 7842. https://doi.org/10.3390/ijerph17217842








