Determination of Selected Chemical Levels in Room Air and on Surfaces after the Use of Cartridge- and Tank-Based E-Vapor Products or Conventional Cigarettes
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design
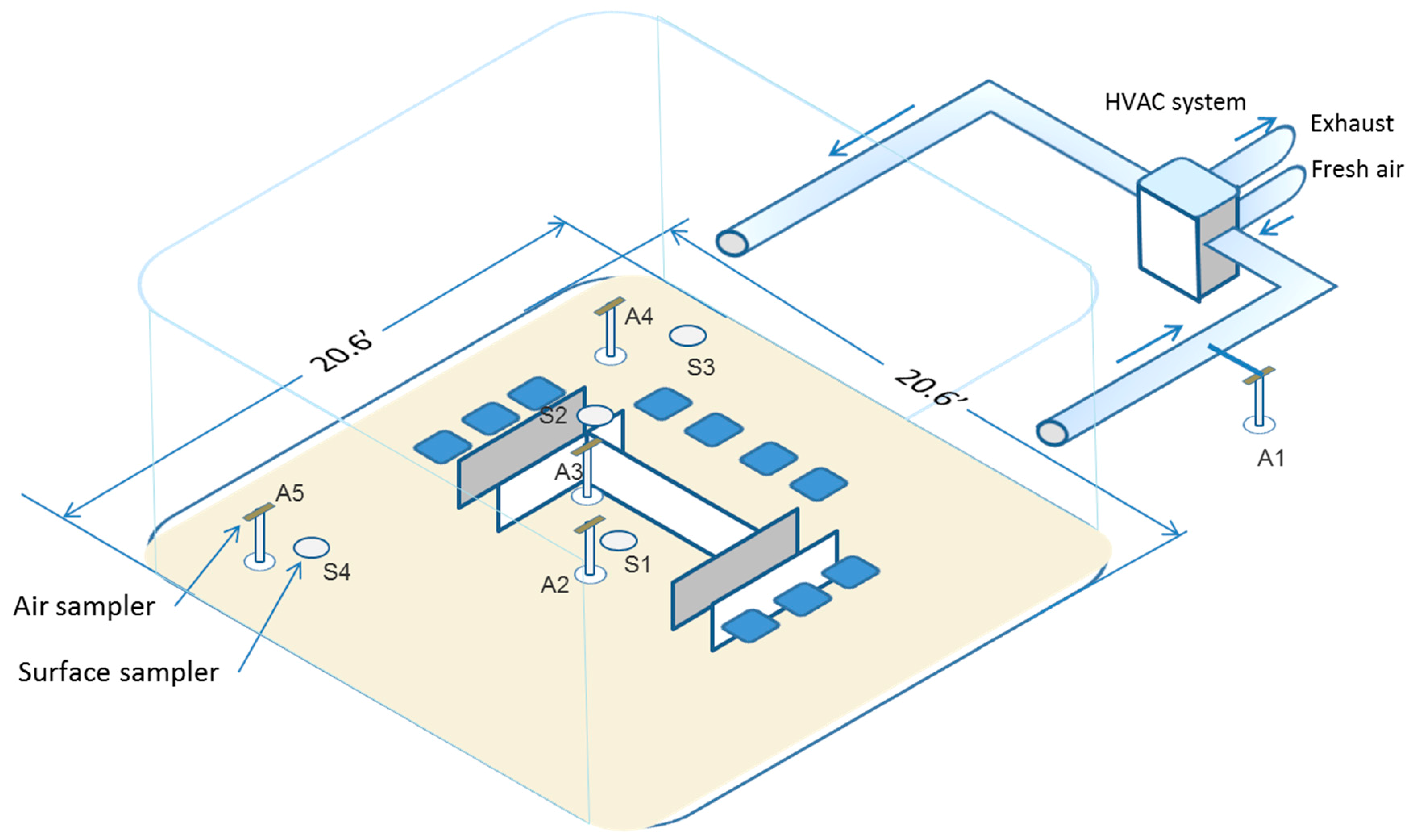
2.1.1. Overall Design and Schedule (Figure 1)
- (1)
- Background condition (all study days, at multiple times)—Measurements were conducted after a complete 2-h air wash of the EC, in the absence of any study participants. The background assessment was conducted primarily to account for potential off-gassing of carbonyls and VOCs from construction material and fixtures in the EC.
- (2)
- Baseline condition (all Groups)—Baseline measurements were made when study participants were sitting in the EC for four hours without using any tobacco or nicotine containing products.
- (3)
- Pre-specified product use condition (only Group I and II)—Room air and surface levels were measured when study participants used the product every 30 min for four hours (each product use was pre-specified as one 5-s puff every 30 s for a total of ten puffs).
- (4)
- Ad libitum product use condition (all Groups)—Room air and surface levels were measured when study participants used the product ad libitum during four hours. Groups I–III study participants were asked to take at least 40 puffs, without any puff duration or puff interval restrictions and Group IV study participants were asked to smoke at least one complete cigarette every hour. Study participants were provided a counter to track the number of puffs.
2.1.2. Study Participants
2.1.3. Study Outcome Measures
2.1.4. EC and Room Air Sampling
2.1.5. Surface Sampling
2.1.6. Products Used
2.2. Analytical Procedures
2.3. Statistical Analyses
3. Results
3.1. Study Participants Disposition
3.2. Product Use
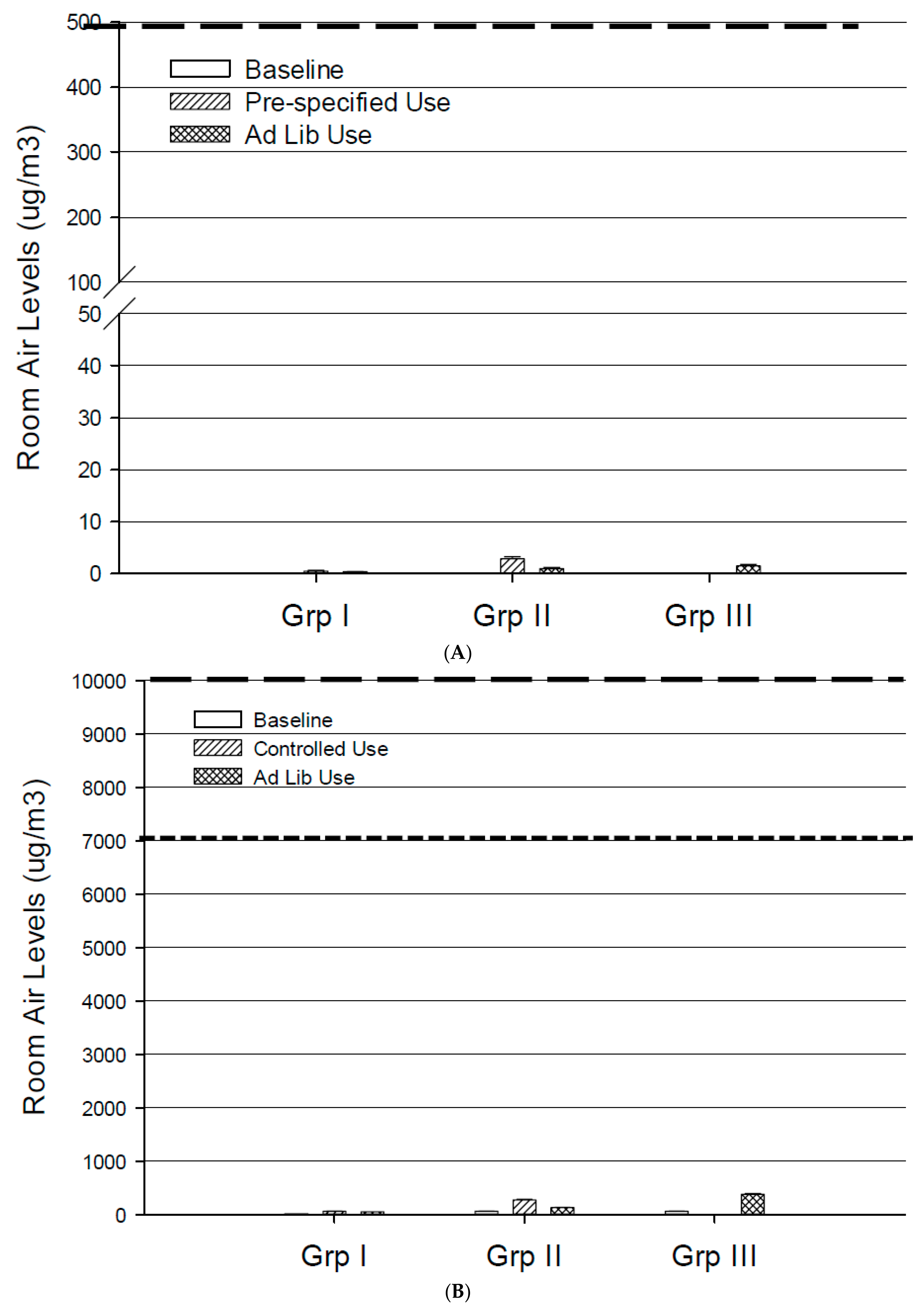
3.3. Levels of Selected Constituents in RAS
3.3.1. Nicotine, PG and Glycerol
3.3.2. Carbonyls
3.3.3. Volatile Organic Constituents
3.3.4. Selected Trace Elements
3.4. Nicotine, PG and Glycerol in Surface Samples
4. Discussion
4.1. Room Air Levels from Use of Different Tobacco Products
4.2. Levels of Different Groups of Chemicals
4.3. Potential Limitations
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| AE | adverse event |
| BLOQ | below the limit of quantification |
| BMI | body mass index |
| CC | conventional cigarette |
| CPD | cigarettes per day |
| EC | exposure chamber |
| EVP | electronic vapor product |
| GC | gas chromatograph |
| HPHC | harmful and potentially harmful constituent |
| LOQ | limit of quantification |
| PG | propylene glycol |
| RAS | room air sample |
| VOC | volatile organic compound |
References
- Etter, J.F.; Bullen, C. Electronic cigarette: Users profile, utilization, satisfaction and perceived efficacy. Addiction 2011, 106, 2017–2028. [Google Scholar] [CrossRef] [PubMed]
- Regan, A.K.; Promoff, G.; Dube, S.R.; Arrazola, R. Electronic nicotine delivery systems: Adult use and awareness of the ‘e-cigarette’ in the USA. Tob. Control 2013, 22, 19–23. [Google Scholar] [CrossRef] [PubMed]
- Zhu, S.H.; Gamst, A.; Lee, M.; Cummins, S.; Yin, L.; Zoref, L. The use and perception of electronic cigarettes and snus among the U.S. population. PLoS ONE 2013, 8, e79332. [Google Scholar] [CrossRef] [PubMed]
- Etter, J.F. Characteristics of users, and usage of different types of electronic cigarettes: Findings from an online survey. Addiction 2016, 111, 724–733. [Google Scholar] [CrossRef] [PubMed]
- Rodgman, A.; Smith, C.J.; Perfetti, T.A. The composition of cigarette smoke: A retrospective, with emphasis on polycyclic components. Hum. Exp. Toxicol. 2000, 19, 573–595. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, M.; Kapur, S.; Frost-Pineda, K.; Feng, S.; Wang, J.; Liang, Q.; Roethig, H. Evaluation of biomarkers of exposure to selected cigarette smoke constituents in adult smokers switched to carbon-filtered cigarettes in short-term and long-term clinical studies. Nicotine Tob. Res. 2008, 10, 1761–1772. [Google Scholar] [CrossRef] [PubMed]
- Flora, J.W.; Meruva, N.; Huang, C.B.; Wilkinson, C.T.; Ballentine, R.; Smith, D.C.; Werley, M.S.; McKinney, W.J. Characterization of potential impurities and degradation products in electronic cigarette formulations and aerosols. Regul. Toxicol. Pharmacol. 2016, 74, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Goniewicz, M.L.; Knysak, J.; Gawron, M.; Kosmider, L.; Sobczak, A.; Kurek, J.; Prokopowicz, A.; Jablonska-Czapla, M.; Rosik-Dulewska, C.; Havel, C.; et al. Levels of selected carcinogens and toxicants in vapour from electronic cigarettes. Tob. Control 2014, 23, 133–139. [Google Scholar] [CrossRef] [PubMed]
- Kosmider, L.; Sobczak, A.; Fik, M.; Knysak, J.; Zaciera, M.; Kurek, J.; Goniewicz, M.L. Carbonyl compounds in electronic cigarette vapors: Effects of nicotine solvent and battery output voltage. Nicotine Tob. Res. 2014, 16, 1319–1326. [Google Scholar] [CrossRef] [PubMed]
- Tayyarah, R.; Long, G.A. Comparison of select analytes in aerosol from e-cigarettes with smoke from conventional cigarettes and with ambient air. Regul. Toxicol. Pharmacol. 2014, 70, 704–710. [Google Scholar] [CrossRef] [PubMed]
- Williams, M.; Villarreal, A.; Bozhilov, K.; Lin, S.; Talbot, P. Metal and silicate particles including nanoparticles are present in electronic cigarette cartomizer fluid and aerosol. PLoS ONE 2013, 8, e57987. [Google Scholar] [CrossRef] [PubMed]
- Bam, T.S.; Bellew, W.; Berezhnova, I.; Jackson-Morris, A.; Jones, A.; Latif, E.; Molinari, M.A.; Quan, G.; Singh, R.J.; Wisotzky, M.; et al. Position statement on electronic cigarettes or electronic nicotine delivery systems. Int. J. Tuberc. Lung Dis. 2014, 18, 5–7. [Google Scholar] [CrossRef] [PubMed]
- Czogala, J.; Goniewicz, M.L.; Fidelus, B.; Zielinska-Danch, W.; Travers, M.J.; Sobczak, A. Secondhand exposure to vapors from electronic cigarettes. Nicotine Tob. Res. 2014, 16, 655–662. [Google Scholar] [CrossRef]
- Geiss, O.; Bianchi, I.; Barahona, F.; Barrero-Moreno, J. Characterisation of mainstream and passive vapours emitted by selected electronic cigarettes. Int. J. Hyg. Environ. Health 2015, 218, 169–180. [Google Scholar] [CrossRef] [PubMed]
- McAuley, T.R.; Hopke, P.K.; Zhao, J.; Babaian, S. Comparison of the effects of e-cigarette vapor and cigarette smoke on indoor air quality. Inhal. Toxicol. 2012, 24, 850–857. [Google Scholar] [CrossRef] [PubMed]
- Schripp, T.; Markewitz, D.; Uhde, E.; Salthammer, T. Does e-cigarette consumption cause passive vaping? Indoor Air 2013, 23, 25–31. [Google Scholar] [CrossRef] [PubMed]
- Hess, I.M.R.; Lachireddy, K.; Capon, A. A systematic review of the health risks from passive exposure to electronic cigarette vapour. Public Health Res. Pract. 2016, 26, 1–9. [Google Scholar] [CrossRef] [PubMed]
- St Helen, G.; Havel, C.; Dempsey, D.A.; Jacob, P., 3rd; Benowitz, N.L. Nicotine delivery, retention and pharmacokinetics from various electronic cigarettes. Addiction 2016, 111, 535–544. [Google Scholar] [CrossRef] [PubMed]
- Jensen, R.P.; Luo, W.; Pankow, J.F.; Strongin, R.M.; Peyton, D.H. Hidden formaldehyde in e-cigarette aerosols. N. Engl. J. Med. 2015, 372, 392–394. [Google Scholar] [CrossRef]
- Nitzkin, J.L.; Farsalinos, K.; Siegel, M. More on hidden formaldehyde in e-cigarette aerosols. N. Engl. J. Med. 2015, 372, 1575. [Google Scholar] [PubMed]
- Bates, C.D.; Farsalinos, K.E. E-cigarettes need to be tested for safety under realistic conditions. Addiction 2015, 110, 1688–1689. [Google Scholar] [CrossRef] [PubMed]
- Bekki, K.; Uchiyama, S.; Ohta, K.; Inaba, Y.; Nakagome, H.; Kunugita, N. Carbonyl compounds generated from Electronic Cigarettes. Int. J. Environ. Res. Public Health 2014, 11, 11192–11200. [Google Scholar] [CrossRef] [PubMed]
- Geiss, O.; Bianchi, I.; Barrero-Moreno, J. Correlation of volatile carbonyl yields emitted by e-cigarettes with the temperature of the heating coil and the perceived sensorial quality of the generated vapours. Int. J. Hyg. Environ. Health 2016, 219, 268–277. [Google Scholar] [CrossRef] [PubMed]
- Sleiman, M.; Logue, J.M.; Montesinos, V.N.; Russell, M.L.; Litter, M.I.; Gundel, L.A.; Destaillats, H. Emissions from electronic cigarettes: Key parameters affecting the release of harmful chemicals. Environ. Sci. Technol. 2016, 50, 9644–9651. [Google Scholar] [CrossRef] [PubMed]
- Khlystov, A.; Samburova, V. Flavoring compounds dominate toxic aldehyde production during e-cigarette vaping. Environ. Sci. Technol. 2016, 50, 13080–13085. [Google Scholar] [CrossRef] [PubMed]
- Ballbe, M.; Martinez-Sanchez, J.M.; Sureda, X.; Fu, M.; Perez-Ortuno, R.; Pascual, J.A.; Salto, E.; Fernandez, E. Cigarettes vs. e-cigarettes: Passive exposure at home measured by means of airborne marker and biomarkers. Environ. Res. 2014, 135, 76–80. [Google Scholar] [CrossRef] [PubMed]
- Chorti, M.S.; Poulianiti, K.P.; Jamurtas, A.Z.; Kostikas, K.; Tzatzarakis, M.N.; Vynias, D.; Koutedakis, Y.; Flouris, A.D.; Tsatsakis, A.M. Effects of active and passive electronic and tobacco cigarette smoking on lung function. Toxicol. Lett. 2012, 211, S64. [Google Scholar] [CrossRef]
- Flouris, A.D.; Chorti, M.S.; Poulianiti, K.P.; Jamurtas, A.Z.; Kostikas, K.; Tzatzarakis, M.N.; Wallace Hayes, A.; Tsatsakis, A.M.; Koutedakis, Y. Acute impact of active and passive electronic cigarette smoking on serum cotinine and lung function. Inhal. Toxicol. 2013, 25, 91–101. [Google Scholar] [CrossRef] [PubMed]
- Fromme, H.; Schober, W. Waterpipes and e-cigarettes: Impact of alternative smoking techniques on indoor air quality and health. Atmos. Environ. 2015, 106, 429–441. [Google Scholar] [CrossRef]
- King, A.C.; Smith, L.J.; McNamara, P.J.; Matthews, A.K.; Fridberg, D.J. Passive exposure to electronic cigarette (e-cigarette) use increases desire for combustible and e-cigarettes in young adult smokers. Tob. Control 2015, 24, 501–504. [Google Scholar] [CrossRef] [PubMed]
- Maloney, J.C.; Thompson, M.K.; Oldham, M.J.; Stiff, C.L.; Lilly, P.D.; Patskan, G.J.; Shafer, K.H.; Sarkar, M.A. Insights from Two Industrial Hygiene Pilot E-Cigarette Passive Vaping Studies. J. Occup. Environ. Hyg. 2016, 13, 275–283. [Google Scholar] [CrossRef] [PubMed]
- O’Connell, G.; Colard, S.; Cahours, X.; Pritchard, J.D. An Assessment of Indoor Air Quality before, during and after Unrestricted Use of E-Cigarettes in a Small Room. Int. J. Environ. Res. Public Health 2015, 12, 4889–4907. [Google Scholar] [CrossRef] [PubMed]
- Romagna, G.; Zabarini, L.; Barbiero, L.; Bocchietto, E.; Todeschi, S.; Caravati, E.; Voster, D.; Farsalinos, K. Characterization of chemicals released to the environment by electronic cigarettes use (ClearStream-AIR project): Is passive vaping a reality? Presented at the 14th Annual Meeting of the Society for Research on Nicotine and Tobacco, Helsinki, Finland, 31 August 2012. [Google Scholar]
- Saffari, A.; Daher, N.; Ruprecht, A.; De Marco, C.; Pozzi, P.; Boffi, R.; Hamad, S.H.; Shafer, M.M.; Schauer, J.J.; Westerdahl, D.; et al. Particulate metals and organic compounds from electronic and tobacco-containing cigarettes: Comparison of emission rates and secondhand exposure. Environ. Sci. Process. Impacts 2014, 16, 2259–2267. [Google Scholar] [CrossRef] [PubMed]
- Schober, W.; Szendrei, K.; Matzen, W.; Osiander-Fuchs, H.; Heitmann, D.; Schettgen, T.; Jorres, R.A.; Fromme, H. Use of electronic cigarettes (e-cigarettes) impairs indoor air quality and increases FeNO levels of e-cigarette consumers. Int. J. Hyg. Environ. Health 2014, 217, 628–637. [Google Scholar] [CrossRef] [PubMed]
- Flouris, A.D.; Poulianiti, K.P.; Chorti, M.S.; Jamurtas, A.Z.; Kouretas, D.; Owolabi, E.O.; Tzatzarakis, M.N.; Tsatsakis, A.M.; Koutedakis, Y. Acute effects of electronic and tobacco cigarette smoking on complete blood count. Food Chem. Toxicol. 2012, 50, 3600–3603. [Google Scholar] [CrossRef] [PubMed]
- Kuschner, W.G.; Reddy, S.; Mehrotra, N.; Paintal, H.S. Electronic cigarettes and thirdhand tobacco smoke: Two emerging health care challenges for the primary care provider. Int. J. Gen. Med. 2011, 4, 115–120. [Google Scholar] [CrossRef] [PubMed]
- Matt, G.E.; Quintana, P.J.; Destaillats, H.; Gundel, L.A.; Sleiman, M.; Singer, B.C.; Jacob, P.; Benowitz, N.; Winickoff, J.P.; Rehan, V.; et al. Thirdhand tobacco smoke: Emerging evidence and arguments for a multidisciplinary research agenda. Environ. Health Perspect. 2011, 119, 1218–1226. [Google Scholar] [CrossRef]
- Bush, D.; Goniewicz, M.L. A pilot study on nicotine residues in houses of electronic cigarette users, tobacco smokers, and non-users of nicotine-containing products. Int. J. Drug Policy 2015, 26, 609–611. [Google Scholar] [CrossRef]
- Goniewicz, M.L.; Lee, L. Electronic cigarettes are a source of thirdhand exposure to nicotine. Nicotine Tob. Res. 2015, 17, 256–258. [Google Scholar] [CrossRef] [PubMed]
- WHO: World Health Organization. The Conference of the Parties to the WHO Framework Convention on Tobacco Control, Sixth Session, Moscow, Russian, 13–18 October 2014; Provisional Agenda Item 4.4.2; WHO: Geneva, Switzerland, 2014. [Google Scholar]
- Chang, H. Research gaps related to the environmental impacts of electronic cigarettes. Tob. Control 2014, 23 (Suppl. 2), ii54–ii58. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Liang, Q.; Oldham, M.J.; Rostami, A.; Wagner, K.A.; Gillman, G.; Patel, P.; Savioz, R.; Sarkar, M. Levels of selected constituents in exhaled breath from the use of cartridge, tank-based e-vapor products or conventional cigarettes by adult tobacco product users. 2016; in preparation. [Google Scholar]
- ICH: International Conference on Harmonisation. Guidance for Industry E6 Good Clinical Practice: Consolidated Guidance. Available online: https://www.fda.gov/downloads/drugs/guidances/ucm073122.pdf (accessed on 16 August 2017).
- Murdoch, R.D.; Bareille, P.; Ignar, D.; Mark, S.; Miller, S.R.; Gupta, A.; Salapatek, A.M.; Patel, P. Once-daily dosing of levocabastine has comparable efficacy to twice-daily dosing in the treatment of allergic rhinitis assessed in an allergen challenge chamber. Int. J. Clin. Pharmacol. Ther. 2015, 53, 811–818. [Google Scholar] [CrossRef] [PubMed]
- ASHRAE: American Society of Heating, Refrigerating and Air-Conditioning Engineers, Inc. Addendum n to ANSI/ASHRAE Standard 62–2001. Ventilation for Acceptable Indoor Air Quality; ASHRAE: Atlanta, GA, USA, 2003; ISSN 1041-2336. [Google Scholar]
- Oldham, M.J.; Wagner, K.A.; Gillman, G.; Beach, J.; Liu, J.; Rostami, A.; Sarkar, M. Development/Verification of Methods for Measurement of Exhaled Breath and Environmental E-Cigarette Vapor. Regul. Toxicol. Pharmacol. 2017, 85, 55–63. [Google Scholar] [CrossRef] [PubMed]
- ISO: International Organization for Standardization. 16200-1. Workplace Air Quality—Sampling and Analysis of Volatile Organic Compounds by Solvent Desorption/Gas Chromatography—Part 1: Pumped Sampling Method; ISO: Geneva, Switzerland, 2001. [Google Scholar]
- ISO: International Organization for Standardization. 16000-6. Indoor Air—Part 6: Determination of volatile Organic Compounds in Indoor and Test Chamber Air by Active Sampling on Tenax TA Sorbent, Thermal Desorption and Gas Chromatography Using MS or MS-FID; ISO: Geneva, Switzerland, 2011. [Google Scholar]
- ISO: International Organization for Standardization. 16000-3. Indoor Air—Part 3: Determination of Formaldehyde and Other Carbonyl Compounds in Indoor Air and Test Chamber Air—Active Sampling Method; ISO: Geneva, Switzerland, 2011. [Google Scholar]
- EPA: US Environmental Protection Agency. Compendium Method IO-2.1. Sampling of Ambient Air for Total Suspended Particulate Matter (Spm) and PM10 Using High Volume (Hv) Sampler; EPA: Washington, DC, USA, 1999.
- EPA: US Environmental Protection Agency. Compendium Method IO-3.5. Determination of Metals in Ambient Particulate Matter Using Inductively Coupled Plasma/ Mass Spectrometry (ICP/MS); EPA: Washington, DC, USA, 1999.
- AIHA: American Industrial Hygiene Association. ERPG/WEEL Handbook; AIHA: Falls Church, VA, USA, 2014; ISBN 10 1935082442. [Google Scholar]
- OSHA: United States Department of Labor. Occupational Safety and Health Administration. Toxic and Hazardous Substances. Standards—29 CFR 1910.1000 Table Z-1 Limits for Air Contaminants; OSHA: Washington, DC, USA. Available online: https://www.dli.mn.gov/OSHA/PDF/pels.pdf (accessed on 28 August 2017).
- OSHA: United States Department of Labor. Occupational Safety and Health Administration. Toxic and Hazardous Substances. Standards—29 CFR 1910.1048 Formaldehyde; OSHA: Washington, DC, USA. Available online: https://www.osha.gov/pls/oshaweb/owadisp.show_document?p_id=10075&p_table=STANDARDS (accessed on 28 August 2017).
- Hubbs, A.F.; Cummings, K.J.; McKernan, L.T.; Dankovic, D.A.; Park, R.M.; Kreiss, K. Comment on Farsalinos et al. Evaluation of Electronic Cigarette Liquids and Aerosol for the Presence of Selected Inhalation Toxins. Nicotine Tob. Res. 2015, 17, 1288–1289. [Google Scholar] [CrossRef] [PubMed]
- DFG: Deutsche Forschungsgemeinschaft. List of MAK and BAT Values 2015: Maximum Concentrations and Biological Tolerance Values at the Workplace. Chapter II List of Substances; Wiley-VCH: Weinheim, Geramny, 2015; pp. 19–162. ISBN 9783527339914. [Google Scholar]
- OEHHA: Office of Environmental Health Hazard Assessment. California Environmental Protection Agency, State of California, Acute, 8-Hour and Chronic Reference Exposure Levels (chRELs) as of June 2016; OEHHA: Sacramento, CA, USA, 2016. [Google Scholar]
- The WHO European Centre for Environment and Health. WHO: World Health Organization Guidelines for Indoor Air Quality: Selected Pollutants; The WHO European Centre for Environment and Health, Bonn Office: WHO Regional Office for Europe: Bonn, Germany, 2009. [Google Scholar]
- Salthammer, T.; Mentese, S.; Marutzky, R. Formaldehyde in the indoor environment. Chem. Rev. 2010, 110, 2536–2572. [Google Scholar] [CrossRef] [PubMed]
- Salthammer, T. The formaldehyde dilemma. Int. J. Hyg. Environ. Health 2015, 218, 433–436. [Google Scholar] [CrossRef] [PubMed]
- Fromme, H.; Nitschke, L.; Boehmer, S.; Kiranoglu, M.; Goen, T. Hbmnet: Exposure of German residents to ethylene and propylene glycol ethers in general and after cleaning scenarios. Chemosphere 2013, 90, 2714–2721. [Google Scholar] [CrossRef] [PubMed]
- Johanson, G. Aspects of biological monitoring of exposure to glycol ethers. Toxicol. Lett. 1988, 43, 5–21. [Google Scholar] [CrossRef]
- Carmines, E.L.; Gaworski, C.L. Toxicological evaluation of glycerin as a cigarette ingredient. Food Chem. Toxicol. 2005, 43, 1521–1539. [Google Scholar] [CrossRef] [PubMed]
- National Research Council (NRC). National Research Council Review of the Formaldehyde Assessment in the National Toxicology Program 12th Report on Carcinogens; National Academides Press (US): Washington, DC, USA, 2014; ISBN 978-0-309-31227-1. [Google Scholar]
- Spanel, P.; Smith, D. Quantification of trace levels of the potential cancer biomarkers formaldehyde, acetaldehyde and propanol in breath by SIFT-MS. J. Breath Res. 2008, 2, 046003. [Google Scholar] [CrossRef] [PubMed]
- Amann, A.; Costello Bde, L.; Miekisch, W.; Schubert, J.; Buszewski, B.; Pleil, J.; Ratcliffe, N.; Risby, T. The human volatilome: Volatile organic compounds (VOCs) in exhaled breath, skin emanations, urine, feces and saliva. J. Breath Res. 2014, 8, 034001. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Pysanenko, A.; Dryahina, K.; Spanel, P.; Smith, D. Analysis of breath, exhaled via the mouth and nose, and the air in the oral cavity. J. Breath Res. 2008, 2, 037013. [Google Scholar] [CrossRef] [PubMed]
- Farsalinos, K.E.; Voudris, V.; Poulas, K. Are metals emitted from electronic cigarettes a reason for health concern? A risk-assessment analysis of currently available literature. Int. J. Environ. Res. Public Health 2015, 12, 5215–5232. [Google Scholar] [CrossRef] [PubMed]
- Bruinen de Bruin, Y.; Koistinen, K.; Kephalopoulos, S.; Geiss, O.; Tirendi, S.; Kotzias, D. Characterisation of urban inhalation exposures to benzene, formaldehyde and acetaldehyde in the European Union: Comparison of measured and modelled exposure data. Environ. Sci. Pollut. Res. Int. 2008, 15, 417–430. [Google Scholar] [CrossRef] [PubMed]
- Fromme, H.; Heitmann, D.; Dietrich, S.; Schierl, R.; Korner, W.; Kiranoglu, M.; Zapf, A.; Twardella, D. [Air quality in schools—Classroom levels of carbon dioxide (CO2), volatile organic compounds (VOC), aldehydes, endotoxins and cat allergen]. Gesundheitswesen 2008, 70, 88–97. [Google Scholar] [CrossRef]
- Wolkoff, P.; Schneider, T.; Kildeso, J.; Degerth, R.; Jaroszewski, M.; Schunk, H. Risk in cleaning: Chemical and physical exposure. Sci. Total Environ. 1998, 215, 135–156. [Google Scholar] [CrossRef]
- Talih, S.; Balhas, Z.; Salman, R.; Karaoghlanian, N.; Shihadeh, A. “Direct Dripping”: A High-Temperature, High-Formaldehyde Emission Electronic Cigarette Use Method. Nicotine Tob. Res. 2015, 18, 453–459. [Google Scholar] [CrossRef] [PubMed]
- Rostami, A.A.; Pithawalla, Y.B.; Liu, J.; Oldham, M.J.; Wagner, K.A.; Frost-Pineda, K.; Sarkar, M.A. A Well-Mixed Computational Model for Estimating Room Air Levels of Selected Constituents from E-Vapor Product Use. Int. J. Environ. Res. Public Health 2016, 13, 828. [Google Scholar] [CrossRef] [PubMed]


| RAS Constituent | LOQ * (μg/m3) | Group I (n = 9) | Group II (n = 9) | Group III (n = 9) | Group IV (n = 10) | ||
|---|---|---|---|---|---|---|---|
| Pre-Specified | Ad libitum | Pre-Specified | Ad libitum | Ad libitum | Ad libitum | ||
| Nicotine | 0.25 | 0.48 ± 0.16 | 0.38 ± 0.07 | 2.83 ± 0.44 | 0.96 ± 0.22 | 1.47 ± 0.32 | 40.65 ± 6.40 |
| Propylene glycol | 3.63 | 44.86 ± 3.84 | 33.06 ± 1.97 | 211.51 ± 14.23 | 68.51 ± 4.58 | 317.06 ± 12.45 | 56.21 ± 4.22 |
| Glycerol | 4.11 | 67.89 ± 16.81 | 98.90 ± 28.14 | 126.75 ± 12.71 | 78.65 ± 8.75 | 242.00 ± 7.62 | NC |
| Arsenic | 0.12 | n/a | n/a | n/a | n/a | n/a | n/a |
| Cadmium | 0.12 | n/a | n/a | n/a | n/a | n/a | n/a |
| Chromium | 0.12 | 0.01 ± 0.15 | 0.012 ± 0.11 | −0.14 ± 0.01 | −0.14 ± 0.01 | −0.05 ± 0.08 | 0.04 ± 0.06 |
| Nickel | 0.12 | 0.07 ± 0.08 | −0.04 ± 0.07 | −0.17 ± 0.13 | −0.20 ± 0.14 | NC | NC |
| Formaldehyde ‡ | 0.62 | −0.83 ± 0.69 | −0.78 ± 0.64 | −3.96 ± 0.45 | −4.53 ± 0.45 | −0.55 ± 0.82 | 49.74 ± 4.18 |
| Crotonaldehyde | 0.62 | NC | NC | NC | NC | NC | 1.09 ± 0.78 |
| o-Tolualdehyde | 0.62 | n/a | n/a | n/a | n/a | n/a | n/a |
| Acetaldehyde ‡ | 0.62 | −0.36 ± 0.57 | −0.54 ± 0.47 | −1.98 ± 0.87 | 0.12 ± 3.94 | 1.10 ± 0.51 | 105.16 ± 5.24 |
| Butyraldehyde | 0.70 | NC | NC | NC | NC | NC | 6.12 ± 0.86 |
| m- and p-Tolualdehyde | 1.24 | n/a | n/a | n/a | n/a | n/a | n/a |
| Acetone ‡ | 0.62 | −3.72 ± 9.27 | 1.91 ± 12.22 | −0.96 ± 8.09 | −8.45 ± 3.54 | n/a | 45.70 ± 7.03 |
| Benzaldehyde | 0.62 | NC | NC | NC | NC | NC | 0.53 ± 1.06 |
| Propionaldehyde | 0.80 | n/a | n/a | n/a | n/a | n/a | n/a |
| Isovaleraldehyde | 0.62 | NC | NC | NC | NC | NC | 2.10 ± 0.57 |
| Hexaldehyde | 1.20 | NC | 2.07 ± 0.52 | NC | NC | NC | 3.27 ± 0.37 |
| Valeraldehyde | 0.62 | NC | NC | NC | NC | NC | 6.10 ± 0.89 |
| 2,5-Dimethylbenzaldehyde | 0.88 | NC | NC | NC | NC | NC | 4.54 ± 0.84 |
| Methyl ethyl ketone ‡ | 1.45 | 0.69 ± 1.20 | 0.02 ± 0.05 | −1.71 ± 1.69 | −0.88 ± 2.34 | 0.10 ± 0.20 | 16.97 ± 1.84 |
| Acrolein | 0.62 | n/a | n/a | n/a | n/a | n/a | n/a |
| 1,3-Butadiene | 0.19 | NC | NC | NC | NC | NC | 10.56 ± 0.57 |
| Benzene | 0.28 | 0.25 ± 0.02 | 0.04 ± 0.03 | −0.75 ± 0.18 | −0.73 ± 0.14 | 0.15 ± 0.05 | 13.24 ± 0.46 |
| Isoprene | 0.24 | 1.72 ± 0.31 | −0.41 ± 1.70 | 0.55 ± 0.15 | −0.18 ± 1.77 | 1.84 ± 0.17 | 167.81 ± 7.81 |
| Toluene | 0.33 | 1.90 ± 0.38 | 0.43 ± 0.23 | −2.02 ± 0.25 | −1.53 ± 0.31 | 0.13 ± 0.46 | 30.83 ± 1.20 |
| Furan | 0.18 | NC | NC | NC | NC | NC | 12.08 ± 0.50 |
| Ethylene oxide | 0.63 | n/a | n/a | n/a | n/a | n/a | n/a |
| Vinyl chloride | 0.22 | NC | NC | NC | NC | NC | 0.11 ± 0.23 |
| Propylene oxide | 0.83 | n/a | n/a | n/a | n/a | n/a | n/a |
| Nitromethane | 4.33 | n/a | n/a | n/a | n/a | n/a | n/a |
| 2-Nitropropane | 6.33 | n/a | n/a | n/a | n/a | n/a | n/a |
| Vinyl acetate | 6.11 | n/a | n/a | n/a | n/a | n/a | n/a |
| Ethylbenzene | 0.38 | NC | NC | NC | NC | NC | 3.52 ± 0.15 |
| Condition | LOQ (μg/cm2) | Group I | Group II | Group III | Group IV |
|---|---|---|---|---|---|
| (n = 9) | (n = 9) | (n = 9) | (n = 10) | ||
| Nicotine | 0.001 | ||||
| Pre-specified | 0.001 ± 0.002 | NC | n/a | n/a | |
| Ad libitum | NC | NC | NC | 0.001 ± 0.002 | |
| Propylene glycol | 0.019 | ||||
| Pre-specified | 0.02 ± 0.01 | 0.005 ± 0.010 | n/a | n/a | |
| Ad libitum | NC | 0.01 ± 0.01 | NC | NC | |
| Glycerol | 0.02 | ||||
| Pre-specified | NC | 0.07 ± 0.02 | n/a | n/a | |
| Ad libitum | 0.02 ± 0.01 | NC | 0.35 ± 0.12 | 0.00 ± 0.00 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, J.; Liang, Q.; Oldham, M.J.; Rostami, A.A.; Wagner, K.A.; Gillman, I.G.; Patel, P.; Savioz, R.; Sarkar, M. Determination of Selected Chemical Levels in Room Air and on Surfaces after the Use of Cartridge- and Tank-Based E-Vapor Products or Conventional Cigarettes. Int. J. Environ. Res. Public Health 2017, 14, 969. https://doi.org/10.3390/ijerph14090969
Liu J, Liang Q, Oldham MJ, Rostami AA, Wagner KA, Gillman IG, Patel P, Savioz R, Sarkar M. Determination of Selected Chemical Levels in Room Air and on Surfaces after the Use of Cartridge- and Tank-Based E-Vapor Products or Conventional Cigarettes. International Journal of Environmental Research and Public Health. 2017; 14(9):969. https://doi.org/10.3390/ijerph14090969
Chicago/Turabian StyleLiu, Jianmin, Qiwei Liang, Michael J. Oldham, Ali A. Rostami, Karl A. Wagner, I. Gene Gillman, Piyush Patel, Rebecca Savioz, and Mohamadi Sarkar. 2017. "Determination of Selected Chemical Levels in Room Air and on Surfaces after the Use of Cartridge- and Tank-Based E-Vapor Products or Conventional Cigarettes" International Journal of Environmental Research and Public Health 14, no. 9: 969. https://doi.org/10.3390/ijerph14090969
APA StyleLiu, J., Liang, Q., Oldham, M. J., Rostami, A. A., Wagner, K. A., Gillman, I. G., Patel, P., Savioz, R., & Sarkar, M. (2017). Determination of Selected Chemical Levels in Room Air and on Surfaces after the Use of Cartridge- and Tank-Based E-Vapor Products or Conventional Cigarettes. International Journal of Environmental Research and Public Health, 14(9), 969. https://doi.org/10.3390/ijerph14090969





