Abundance of Pathogenic Escherichia coli Virulence-Associated Genes in Well and Borehole Water Used for Domestic Purposes in a Peri-Urban Community of South Africa
Abstract
:1. Introduction
2. Materials and Methods
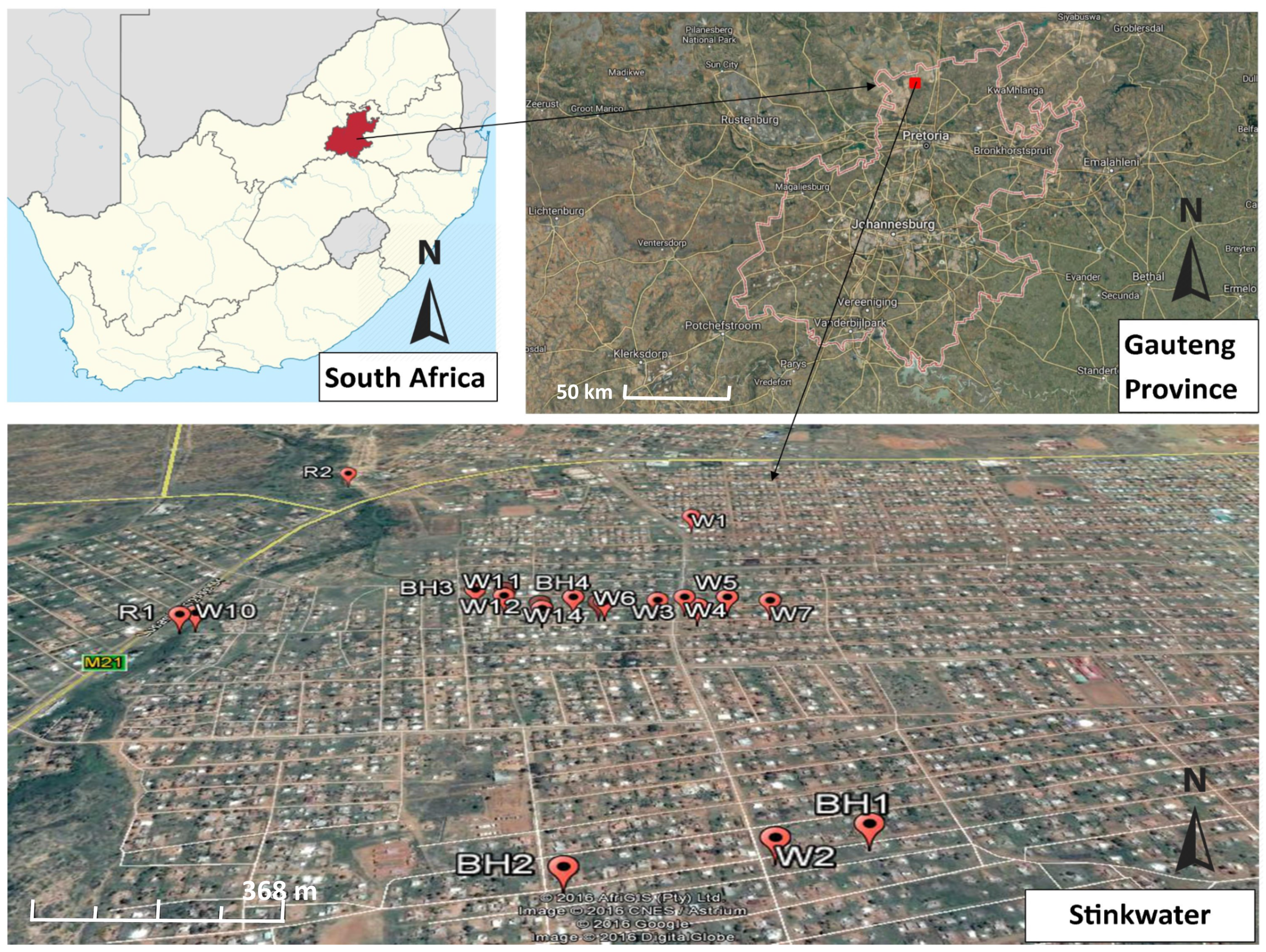
2.1. Description of Study Site
2.2. Sample Collection and Enumeration of E. coli
2.3. DNA Extraction and Real-Time PCR Identification of E. coli Virulence-Associated Genes
2.4. Data Analysis
3. Results and Discussion
3.1. Enumeration of E. coli
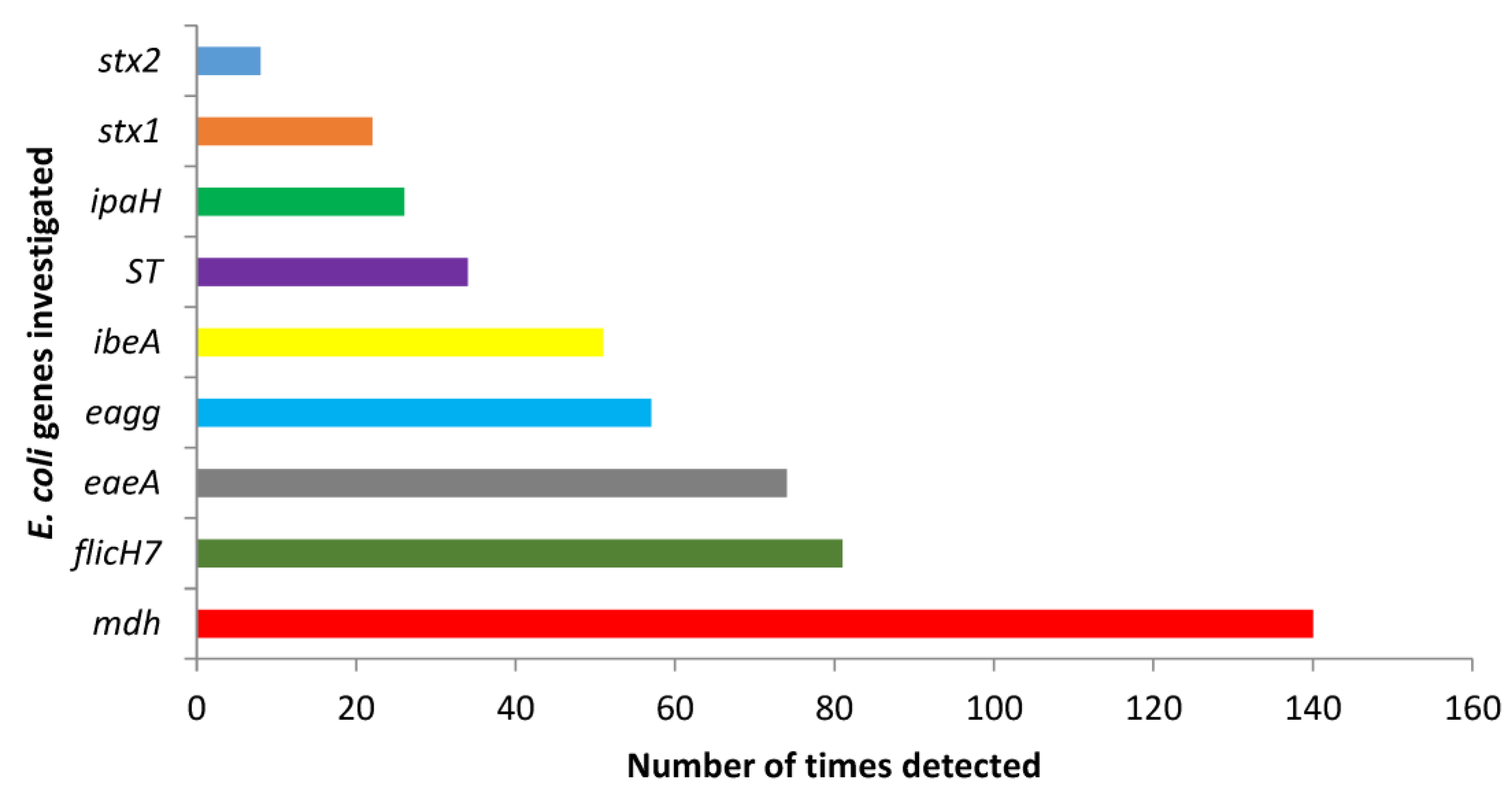
3.2. Identification of E. coli Virulence-Associated Genes
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- World Health Organization. Health in 2015: From MDGs, Millennium Development Goals to SDGs, Sustainable Development Goals; World Healh Organization Press: Geneva, Switzerland, 2015. [Google Scholar]
- Samie, A.; Mashao, M.B.; Bessong, P.O.; Nkgau, T.F.; Momba, M.N.B.; Obi, C.L. Diversity and antibiograms of bacterial organisms isolated from samples of household drinking-water consumed by HIV-positive individuals in rural settings, South Africa. J. Health Popul. Nutr. 2012, 30, 241–249. [Google Scholar] [CrossRef] [PubMed]
- Palamuleni, L.; Akoth, M. Physico-chemical and microbial analysis of selected borehole water in Mahikeng, South Africa. Int. J. Environ. Res. Public Health 2015, 12, 8619–8630. [Google Scholar] [CrossRef] [PubMed]
- Paulse, A.; Jackson, V.; Khan, S.; Khan, W. Isolation and identification of bacterial pollutants from the Berg and Plankenburg Rivers in the Western Cape, South Africa. Water SA 2012, 38, 819–824. [Google Scholar] [CrossRef]
- Graham, J.P.; Polizzotto, M.L. Pit latrines and their impacts on groundwater quality: A systematic review. Environ. Health Perspect. 2013, 121, 521–530. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Chang, B.; Xagoraraki, I. Human health risk assessment of pharmaceuticals in water: Issues and challenges ahead. Int. J. Environ. Res. Public Health 2010, 7, 3929–3953. [Google Scholar] [CrossRef] [PubMed]
- Mwanamoki, P.M.; Devarajan, N.; Thevenon, F.; Atibu, E.K.; Tshibanda, J.B.; Ngelinkoto, P.; Mpiana, P.T.; Prabakar, K.; Mubedi, J.I.; Kabele, C.G.; et al. Assessment of pathogenic bacteria in water and sediment from a water reservoir under tropical conditions (Lake Ma Vallée), Kinshasa Democratic Republic of Congo. Environ. Monit. Assess. 2014, 186, 6821–6830. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. WHO Guidelines for Drinking-Water Quality: Recommendations, 3rd ed.; World Health Organization Press: Geneva, Switzerland, 2008; Volume 1. [Google Scholar]
- Parveen, S.; Hodge, N.C.; Stall, R.E.; Farrah, S.R.; Tamplin, M.L. Phenotypic and genotypic characterization of human and nonhuman Escherichia coli. Water Res. 2001, 35, 379–386. [Google Scholar] [CrossRef]
- Reid, S.D.; Herbelin, C.J.; Bumbaugh, A.C.; Selander, R.K.; Whittam, T.S. Parallel evolution of virulence in pathogenic Escherichia coli. Nature 2000, 406, 64–67. [Google Scholar] [PubMed]
- Robins-Browne, R.M. Editorial commentary: The relentless evolution of pathogenic Escherichia coli. Clin. Infect. Dis. 2005, 41, 793–794. [Google Scholar] [CrossRef] [PubMed]
- Clarke, S.C. Diarrhoeagenic Escherichia coli—An emerging problem? Diagn. Microbiol. Infect. Dis. 2001, 41, 93–98. [Google Scholar] [CrossRef]
- Pitout, J.D.D. Extraintestinal pathogenic Escherichia coli: A combination of virulence with antibiotic resistance. Front. Microbiol. 2012, 3, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Kaper, J.B.; Nataro, J.P.; Mobley, H.L.T. Pathogenic Escherichia coli. Nat. Rev. Microbiol. 2004, 2, 123–140. [Google Scholar] [CrossRef] [PubMed]
- Bovolo, C.I.; Parkin, G.; Sophocleous, M. Groundwater resources, climate and vulnerability. Environ. Res. Lett. 2009, 4, 35001. [Google Scholar] [CrossRef]
- Luczaj, J. Groundwater quantity and quality. Resources 2016, 5, 10. [Google Scholar] [CrossRef]
- Mulamattathil, S.G.; Bezuidenhout, C.; Mbewe, M.; Ateba, C.N. Isolation of environmental bacteria from surface and drinking water in Mafikeng, South Africa, and characterization using their antibiotic resistance profiles. J. Pathog. 2014, 2014, 371208. [Google Scholar] [CrossRef] [PubMed]
- Gyedu-Ababio, T.K.; Van Wyk, F.; Ababio, T.K.G. Effects of human activities on the Waterval River, Vaal River catchment, South Africa. Afr. J. Aquat. Sci. 2004, 29, 75–81. [Google Scholar] [CrossRef]
- Chigor, V.N.; Sibanda, T.; Okoh, A.I. Studies on the bacteriological qualities of the Buffalo River and three source water dams along its course in the Eastern Cape Province of South Africa. Environ. Sci. Pollut. Res. 2013, 20, 4125–4136. [Google Scholar] [CrossRef] [PubMed]
- Luyt, C.D.; Tandlich, R.; Muller, W.J.; Wilhelmi, B.S. Microbial monitoring of surface water in South Africa: An overview. Int. J. Environ. Res. Public Health 2012, 9, 2669–2693. [Google Scholar] [CrossRef] [PubMed]
- Department of Water and Sanitation, Republic of South Africa (DWS). South African Department of Water and Sanitation. National Groundwater Strategy. Draft 4. April 2016; DWS: Cape Town, South Africa, 2016.
- South African Department of Human Settlement. Ministerial Sanitation Task Team Report: Review, Investigation and Evaluation of the National Sanitation Programme—Towards Continuous Improvement; South African Department of Human Settlement: Pretoria, South Africa, 2012.
- Venter, A. Prioritization of River Basins in the Tshwane Area with Reference to Faecal Coliform Bacteria for the Purpose of the Identification of Candidate Wetlands for Rehabilitation. Master’s Thesis, University of the Witwatersrand, Johannesburg, South Africa, 2007. [Google Scholar]
- De Beer, S. Urban South Africa: An opportunity for liberating theological education. Missionalia 2012, 40, 251–277. [Google Scholar] [CrossRef]
- Water Research Commission. Quality of Domestic Water Supplies Volume 2: Sampling Guide; Report No. TT 117; Water Research Commission, Department of Water Affairs and Forestry, Department of Health: Pretoria, South Africa, 2000.
- Buckalew, D.W.; Hartman, L.J.; Grimsley, G.A.; Martin, A.E.; Register, K.M. A long-term study comparing membrane filtration with Colilert defined substrates in detecting fecal coliforms and Escherichia coli in natural waters. J. Environ. Manag. 2006, 80, 191–197. [Google Scholar] [CrossRef] [PubMed]
- Abia, A.L.K.; Ubomba-Jaswa, E.; Momba, M.N.B. Occurrence of diarrhoeagenic Escherichia coli virulence genes in water and bed sediments of a river used by communities in Gauteng, South Africa. Environ. Sci. Pollut. Res. 2016, 23, 15665–15674. [Google Scholar] [CrossRef] [PubMed]
- Omar, K.B.; Barnard, T.G. Detection of diarrhoeagenic Escherichia coli in clinical and environmental water sources in South Africa using single-step 11-gene m-PCR. World J. Microbiol. Biotechnol. 2014, 30, 2663–2671. [Google Scholar] [CrossRef] [PubMed]
- Caine, L.; Nwodo, U.; Okoh, A.; Ndip, R.; Green, E. Occurrence of virulence genes associated with diarrheagenic Escherichia coli isolated from raw cow’s milk from two commercial dairy farms in the Eastern Cape Province, South Africa. Int. J. Environ. Res. Public Health 2014, 11, 11950–11963. [Google Scholar] [CrossRef] [PubMed]
- Omar, K.B.; Potgieter, N.; Barnard, T.G. Development of a rapid screening method for the detection of pathogenic Escherichia coli using a combination of Colilert® Quanti-Trays/2000 and PCR. Water Sci. Technol. Water Supply 2010, 10, 7–13. [Google Scholar] [CrossRef]
- Titilawo, Y.; Obi, L.; Okoh, A. Occurrence of virulence gene signatures associated with diarrhoeagenic and non-diarrhoeagenic pathovars of Escherichia coli isolates from some selected rivers in South-Western Nigeria. BMC Microbiol. 2015, 15, 204. [Google Scholar] [CrossRef] [PubMed]
- Department of Water Affairs and Forestry (DWAF). South African Water Quality Guidelines: Domestic Use, 2nd ed.DWAF: Pretoria, South Africa, 1996; Volume 1.
- Myers, L.; Kane, J. The impact of summer cattle grazing on surface water quality in high elevation mountain meadows. Water Qual. Expo. Heal. 2011, 3, 51–62. [Google Scholar] [CrossRef]
- Habing, G.G.; Lombard, J.E.; Kopral, C.A.; Dargatz, D.A.; Kaneene, J.B. Farm-Level Associations with the Shedding of Salmonella and Antimicrobial-Resistant Salmonella in U.S. Dairy Cattle. Foodborne Pathog. Dis. 2012, 9, 815–821. [Google Scholar] [CrossRef] [PubMed]
- Abia, A.L.K.; Ubomba-Jaswa, E.; Momba, M.N.B. Impact of seasonal variation on Escherichia coli concentrations in the riverbed sediments in the Apies River, South Africa. Sci. Total Environ. 2015, 537, 462–469. [Google Scholar] [CrossRef] [PubMed]
- Zingoni, E.; Love, D.; Magadza, C.; Moyce, W.; Musiwa, K. Effects of a semi-formal urban settlement on groundwater quality Epworth (Zimbabwe): Case study and groundwater quality zoning. Phys. Chem. Earth 2005, 30, 680–688. [Google Scholar] [CrossRef]
- Paulse, A.N.; Jackson, V.A.; Khan, W. Comparison of microbial contamination at various sites along the Plankenburg- and Diep Rivers, Western Cape, South Africa. Water SA 2009, 35, 469–478. [Google Scholar] [CrossRef]
- United Nations Environment Programme (UNEP). Clearing the Waters over Hygiene Debate; UNEP: Nairobi, Kenya, 2010. [Google Scholar]
- Traoré, A.; Mulaudzi, K.; Chari, G.; Foord, S.; Mudau, L.; Barnard, T.; Potgieter, N. The impact of human activities on microbial quality of rivers in the Vhembe District, South Africa. Int. J. Environ. Res. Public Health 2016, 13, 817. [Google Scholar] [CrossRef] [PubMed]
- Tamrakar, A.K.; Jain, M.; Goel, A.K.; Kamboj, D.V.; Singh, L. Characterization of Vibrio cholerae from deep ground water in a cholera endemic area in Central India. Indian J. Microbiol. 2009, 49, 271–275. [Google Scholar] [CrossRef] [PubMed]
- Shelton, D.R.; Karns, J.S.; Coppock, C.; Patel, J.; Sharma, M.; Pachepsky, Y.A. Relationship between eae and stx virulence genes and Escherichia coli in an agricultural watershed: Implications for irrigation water standards and leafy green commodities. J. Food Prot. 2011, 74, 18–23. [Google Scholar] [CrossRef] [PubMed]
- Sidhu, J.P.S.; Ahmed, W.; Hodgers, L.; Toze, S. Occurrence of virulence genes associated with diarrheagenic pathotypes in Escherichia coli isolates from surface water. Appl. Environ. Microbiol. 2013, 79, 328–335. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, Y.; Sperandio, V. Enterohemorrhagic E. coli (EHEC) pathogenesis. Front. Cell. Infect. Microbiol. 2012, 2, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.V.; Le Van, P.; Le Huy, C.; Gia, K.N.; Weintraub, A. Detection and characterization of diarrheagenic Escherichia coli from young children in Hanoi, Vietnam. J. Clin. Microbiol. 2005, 43, 755–760. [Google Scholar] [CrossRef] [PubMed]
- Nontongana, N.; Sibanda, T.; Ngwenya, E.; Okoh, A. Prevalence and antibiogram profiling of Escherichia coli pathotypes isolated from the Kat River and the Fort Beaufort abstraction water. Int. J. Environ. Res. Public Health 2014, 11, 8213–8227. [Google Scholar] [CrossRef] [PubMed]
- Blount, Z.D. The unexhausted potential of E coli. Elife 2015, 4, 1–12. [Google Scholar]
- Abongo, B.O.; Momba, M.N.B. Prevalence and potential link between E. coli O157:H7 isolated from drinking water, meat and vegetables and stools of diarrhoeic confirmed and non-confirmed HIV/AIDS patients in the Amathole District South Africa. J. Appl. Microbiol. 2008, 105, 424–431. [Google Scholar] [CrossRef] [PubMed]
- Lim, J.Y.; Yoon, J.; Hovde, C.J. A brief overview of Escherichia coli O157:H7 and its plasmid O157. J. Microbiol. Biotechnol. 2010, 20, 5–14. [Google Scholar] [PubMed]
- Chen, D.H.; Frankel, G. Enteropathogenic Escherichia coli: Unravelling pathogenesis. FEMS Microbiol. Rev. 2005, 29, 83–98. [Google Scholar] [CrossRef] [PubMed]
- Croxen, M.A.; Law, R.J.; Scholz, R.; Keeney, K.M.; Wlodarska, M.; Finlay, B.B. Recent advances in understanding enteric pathogenic Escherichia coli. Clin. Microbiol. Rev. 2013, 26, 822–880. [Google Scholar] [CrossRef] [PubMed]
- Ranjbar, R.; Hosseini, S.; Zahraei-Salehi, T.; Kheiri, R.; Khamesipour, F. Investigation on prevalence of Escherichia coli strains carrying virulence genes ipaH, estA, eaeA and bfpA isolated from different water sources. Asian Pac. J. Trop. Dis. 2016, 6, 278–283. [Google Scholar] [CrossRef]
- Ud-Din, A.; Wahid, S. Relationship among Shigella spp. and enteroinvasive Escherichia coli (EIEC) and their differentiation. Braz. J. Microbiol. 2014, 45, 1131–1138. [Google Scholar] [CrossRef]
- Sansonnetti, P.J.; Kopecko, D.J.; Formal, S.B. Involvement of a plasmid in the invasive ability of Shigella flexneri. Infect. Immun. 1982, 35, 852–860. [Google Scholar]
- Wijetunge, D.S.; Gongati, S.; DebRoy, C.; Kim, K.S.; Couraud, P.O.; Romero, I.A.; Weksler, B.; Kariyawasam, S. Characterizing the pathotype of neonatal meningitis causing Escherichia coli (NMEC). BMC Microbiol. 2015, 15, 211. [Google Scholar] [CrossRef] [PubMed]
- Kuhnert, P.; Boerlin, P.; Frey, J. Target genes for virulence assessment of Escherichia coli isolates from water, food and the environment. FEMS Microbiol. Rev. 2000, 24, 107–117. [Google Scholar] [CrossRef] [PubMed]
- Winter, S.E.; Keestra, A.M.; Tsolis, R.M.; Bäumler, A.J. The blessings and curses of intestinal inflammation. Cell Host Microbe 2010, 8, 36–43. [Google Scholar] [CrossRef] [PubMed]
| PCR Set | E. coli Pathotype | Gene Targeted | Primer Sequence (5′→ 3′) | Reference |
|---|---|---|---|---|
| 1 | E. coli | mdh | F: GGTATGGATCGTTCCGACCT | [28] |
| R: GGCAGAATGGTAACACCAGAGT | ||||
| 2 | EPEC/EHEC | eaeA | F: ATGCTTAGTGCTGGTTTAGG | [27] |
| R: GCCTTCATCATTTCGCTTTC | ||||
| EAEC | eagg | F: AGACTCTGGCGAAAGACTGTATC | ||
| R: ATGGCTGTCTAATAGATGAGAAC | ||||
| EIEC | ipaH | F: GTTCCTTGACCGCCTTTCCGATACCGTC | ||
| R: GCCGGTCAGCCACCCTCTGAGAGTAC | ||||
| 3 | EHEC | stx1 | F: CTGGATTTAATGTCGCATAGTG | |
| R: AGAACGCCCACTGAGATCATC | ||||
| flicH7 | F: TACCATCGCAAAAGCAAC TCC | [29] | ||
| R: GTCGGCAACGTTAGTGATACC | ||||
| 4 | EHEC | stx2 | F: CCATGACAACGGACAGCAGTTCCT | [30] |
| R: GTCAACTGAGCACTTTG | ||||
| 5 | ETEC/NMEC | ST | F: TTTCCCCTCTTTTAGTCAGTCAACTG | |
| R: GGCAGGATTACAACAAAGTTCACA | ||||
| ibeA | F: TGGAACCCCGCT CGTAATATAC | [31] | ||
| R: CTGCCTGTTCAAGCATTGCA |
| Site | n | Mean | Min | Max |
|---|---|---|---|---|
| W1 | 12 | 443.1 | <1 | >2419.6 |
| W2 | 12 | 51.6 | <1 | 307.6 |
| W3 | 12 | 415.0 | <1 | 1986.3 |
| W4 | 12 | 41.7 | <1 | 325.5 |
| W5 | 12 | 250.1 | <1 | >2419.6 |
| W6 | 12 | 174.7 | <1 | 1553.1 |
| W7 | 11 | 149.7 | 12.1 | 365.4 |
| W8 | 12 | 214.2 | <1 | >2419.6 |
| W9 | 1 | 44.8 * | 44.8 | 44.8 |
| W10 | 4 | 24.23 | 3 | 59.8 |
| W11 | 2 | 1.5 | 1 | 2 |
| W12 | 5 | 2.36 | <1 | 9.8 |
| W13 | 2 | 26.45 | 13 | 39.9 |
| W14 | 4 | 7.875 | <1 | 27.5 |
| BH1 | 1 | 0 * | <1 | <1 |
| BH2 | 1 | 40.4 * | 40.4 | 40.4 |
| BH3 | 1 | 16.6 * | 16.6 | 16.6 |
| BH4 | 5 | 3.2 | <1 | 12 |
| R1 | 11 | 1119.9 | 43.9 | >2419.6 |
| R2 | 12 | 1147.9 | 178.5 | >2419.6 |
| Site | n | eaeA | eagg | ipaH | ST | ibeA | stx1 | stx2 | flicH7 |
|---|---|---|---|---|---|---|---|---|---|
| W1 | 12 | 7 | 3 | 2 | 1 | 6 | 1 | 0 | 8 |
| W2 | 12 | 2 | 5 | 3 | 1 | 1 | 1 | 0 | 5 |
| W3 | 12 | 6 | 6 | 2 | 3 | 5 | 1 | 0 | 8 |
| W4 | 12 | 7 | 2 | 2 | 3 | 4 | 1 | 0 | 6 |
| W5 | 12 | 5 | 4 | 0 | 1 | 3 | 1 | 1 | 4 |
| W6 | 12 | 7 | 6 | 3 | 2 | 3 | 2 | 0 | 6 |
| W7 | 11 | 6 | 4 | 0 | 2 | 4 | 0 | 0 | 8 |
| W8 | 12 | 4 | 2 | 2 | 6 | 1 | 1 | 0 | 4 |
| W9 | 1 | 1 | 1 | 0 | 0 | 0 | 0 | 0 | 1 |
| W10 | 4 | 2 | 1 | 1 | 0 | 2 | 1 | 0 | 3 |
| W11 | 2 | 1 | 0 | 0 | 0 | 1 | 0 | 0 | 0 |
| W12 | 5 | 1 | 1 | 1 | 0 | 0 | 0 | 0 | 1 |
| W13 | 2 | 2 | 1 | 2 | 0 | 0 | 0 | 0 | 2 |
| W14 | 4 | 0 | 0 | 3 | 0 | 2 | 0 | 0 | 1 |
| BH1 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| BH2 | 1 | 1 | 0 | 0 | 0 | 1 | 1 | 0 | 1 |
| BH4 | 1 | 0 | 1 | 1 | 0 | 0 | 0 | 0 | 0 |
| R1 | 11 | 11 | 10 | 1 | 7 | 8 | 5 | 3 | 11 |
| R2 | 12 | 11 | 10 | 3 | 8 | 10 | 7 | 4 | 12 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abia, A.L.K.; Schaefer, L.; Ubomba-Jaswa, E.; Le Roux, W. Abundance of Pathogenic Escherichia coli Virulence-Associated Genes in Well and Borehole Water Used for Domestic Purposes in a Peri-Urban Community of South Africa. Int. J. Environ. Res. Public Health 2017, 14, 320. https://doi.org/10.3390/ijerph14030320
Abia ALK, Schaefer L, Ubomba-Jaswa E, Le Roux W. Abundance of Pathogenic Escherichia coli Virulence-Associated Genes in Well and Borehole Water Used for Domestic Purposes in a Peri-Urban Community of South Africa. International Journal of Environmental Research and Public Health. 2017; 14(3):320. https://doi.org/10.3390/ijerph14030320
Chicago/Turabian StyleAbia, Akebe Luther King, Lisa Schaefer, Eunice Ubomba-Jaswa, and Wouter Le Roux. 2017. "Abundance of Pathogenic Escherichia coli Virulence-Associated Genes in Well and Borehole Water Used for Domestic Purposes in a Peri-Urban Community of South Africa" International Journal of Environmental Research and Public Health 14, no. 3: 320. https://doi.org/10.3390/ijerph14030320
APA StyleAbia, A. L. K., Schaefer, L., Ubomba-Jaswa, E., & Le Roux, W. (2017). Abundance of Pathogenic Escherichia coli Virulence-Associated Genes in Well and Borehole Water Used for Domestic Purposes in a Peri-Urban Community of South Africa. International Journal of Environmental Research and Public Health, 14(3), 320. https://doi.org/10.3390/ijerph14030320







