Streptinone, a New Indanone Derivative from a Marine-Derived Streptomyces massiliensis, Inhibits Particulate Matter-Induced Inflammation
Abstract
1. Introduction
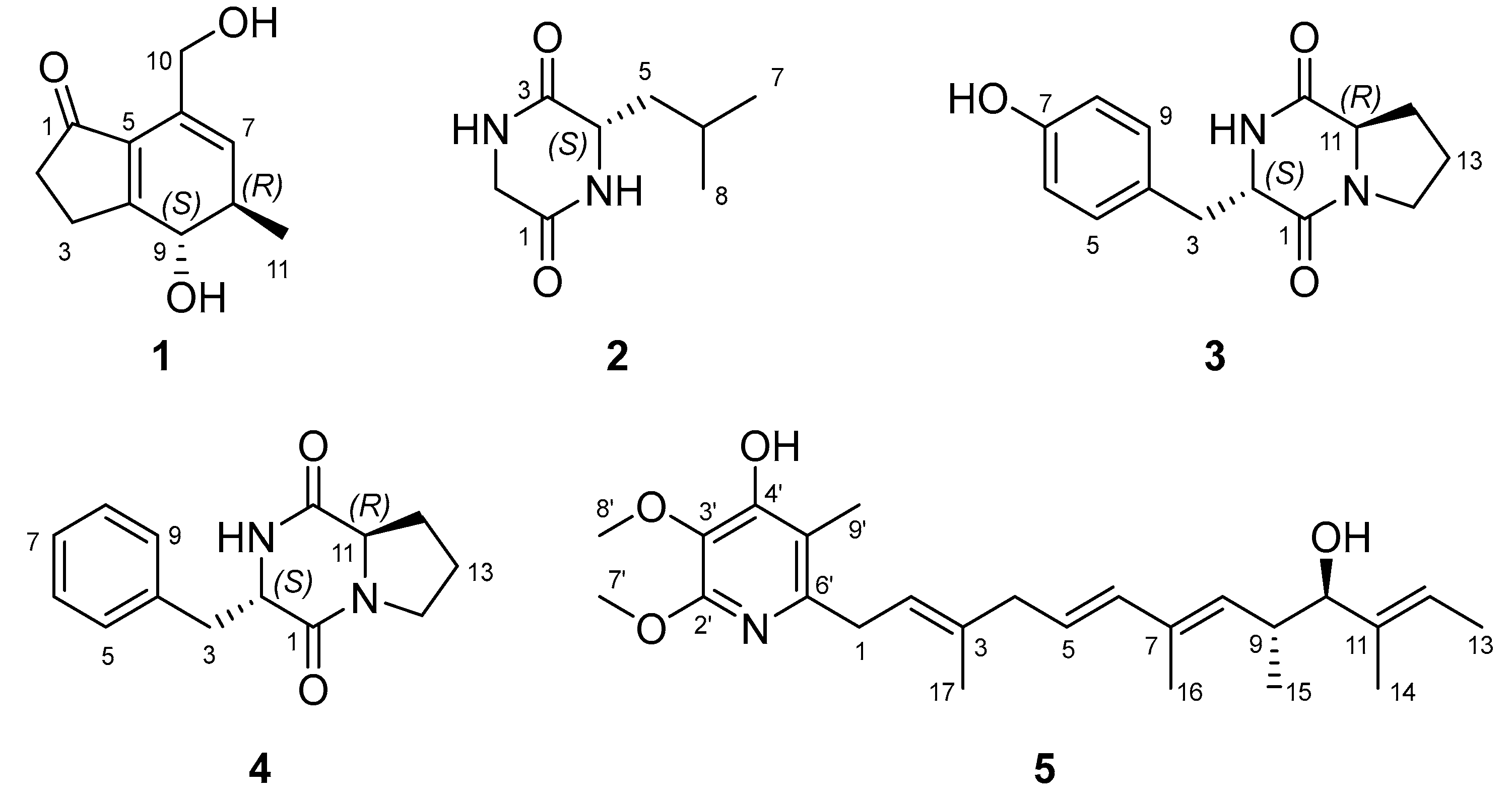
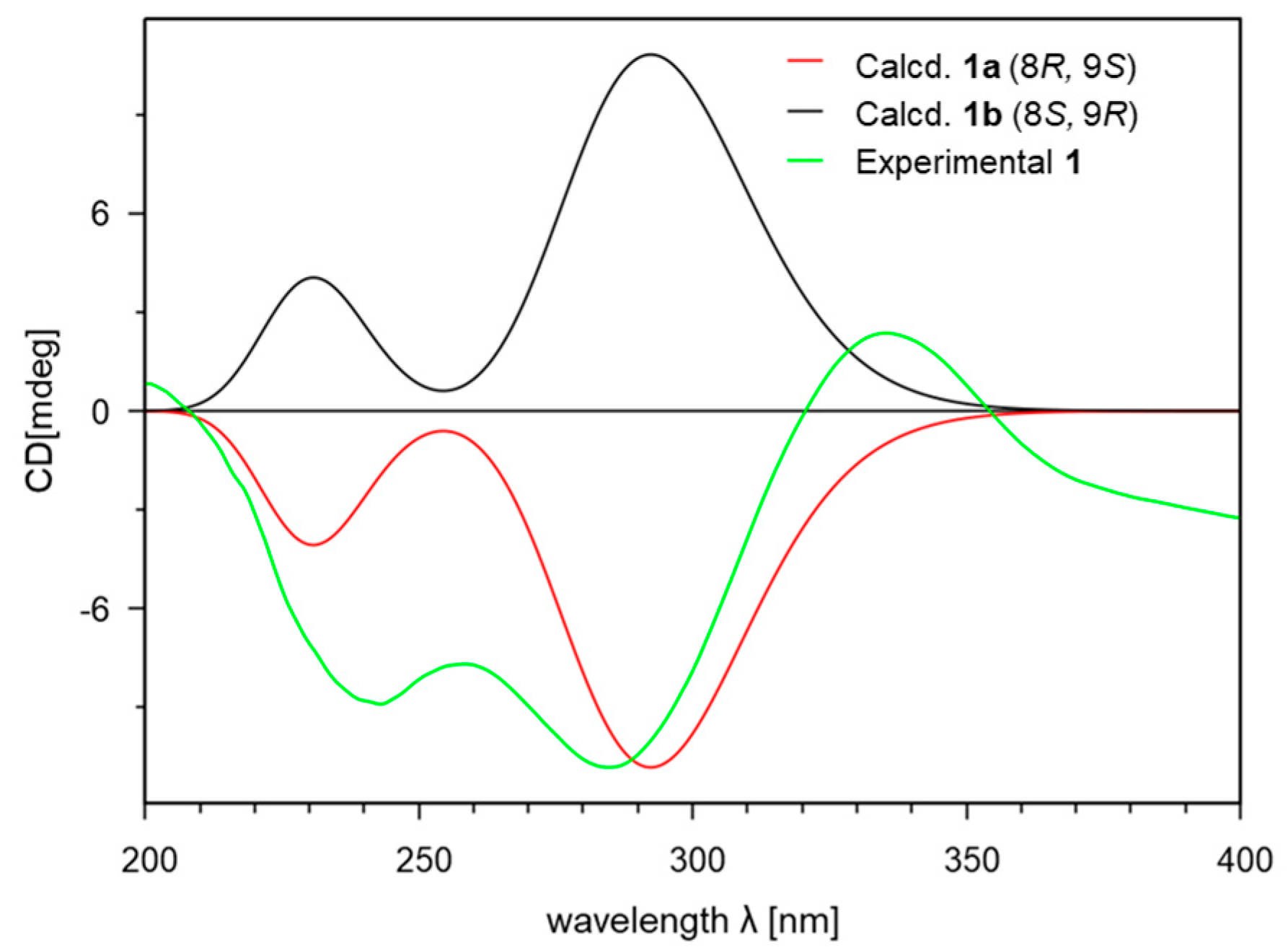
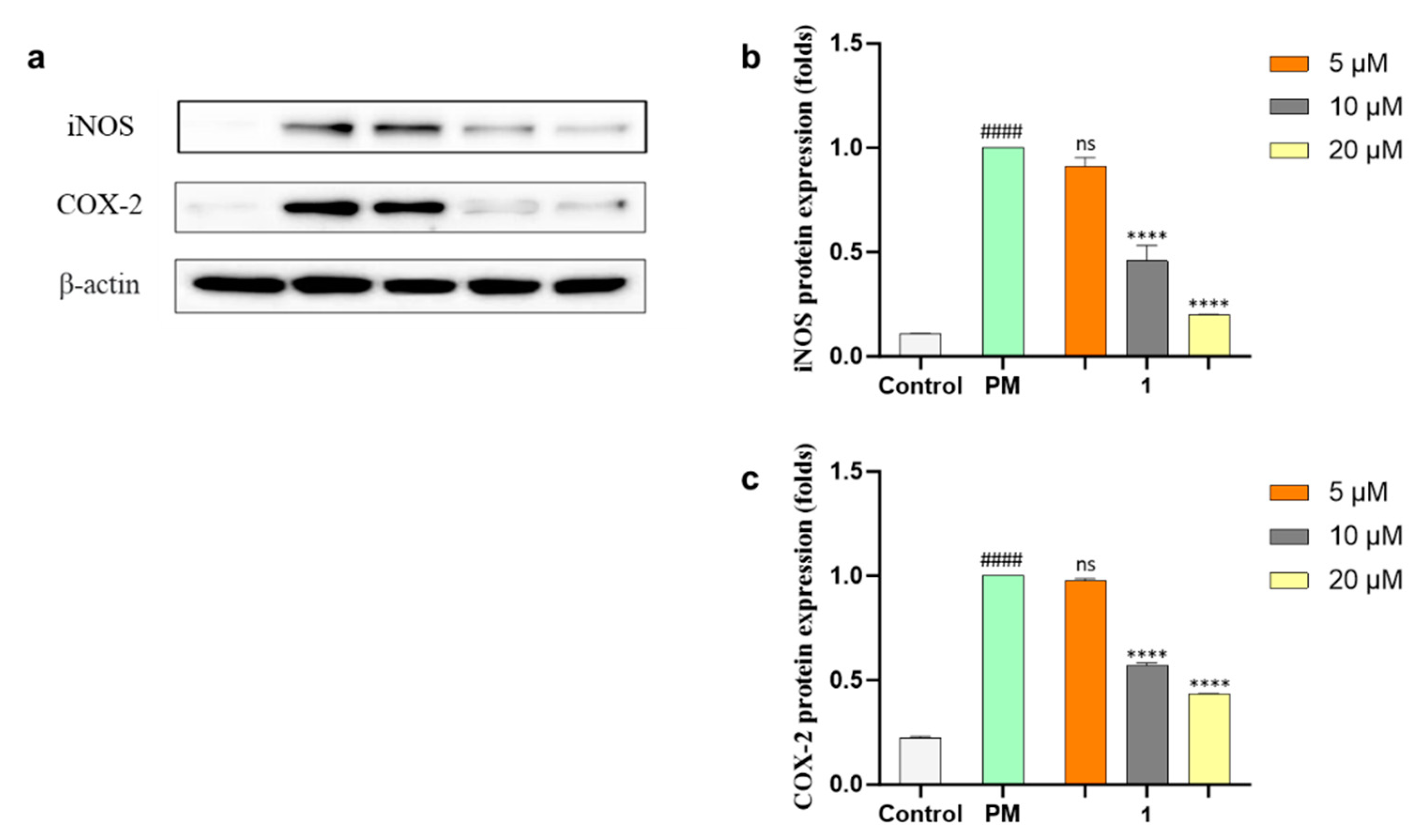
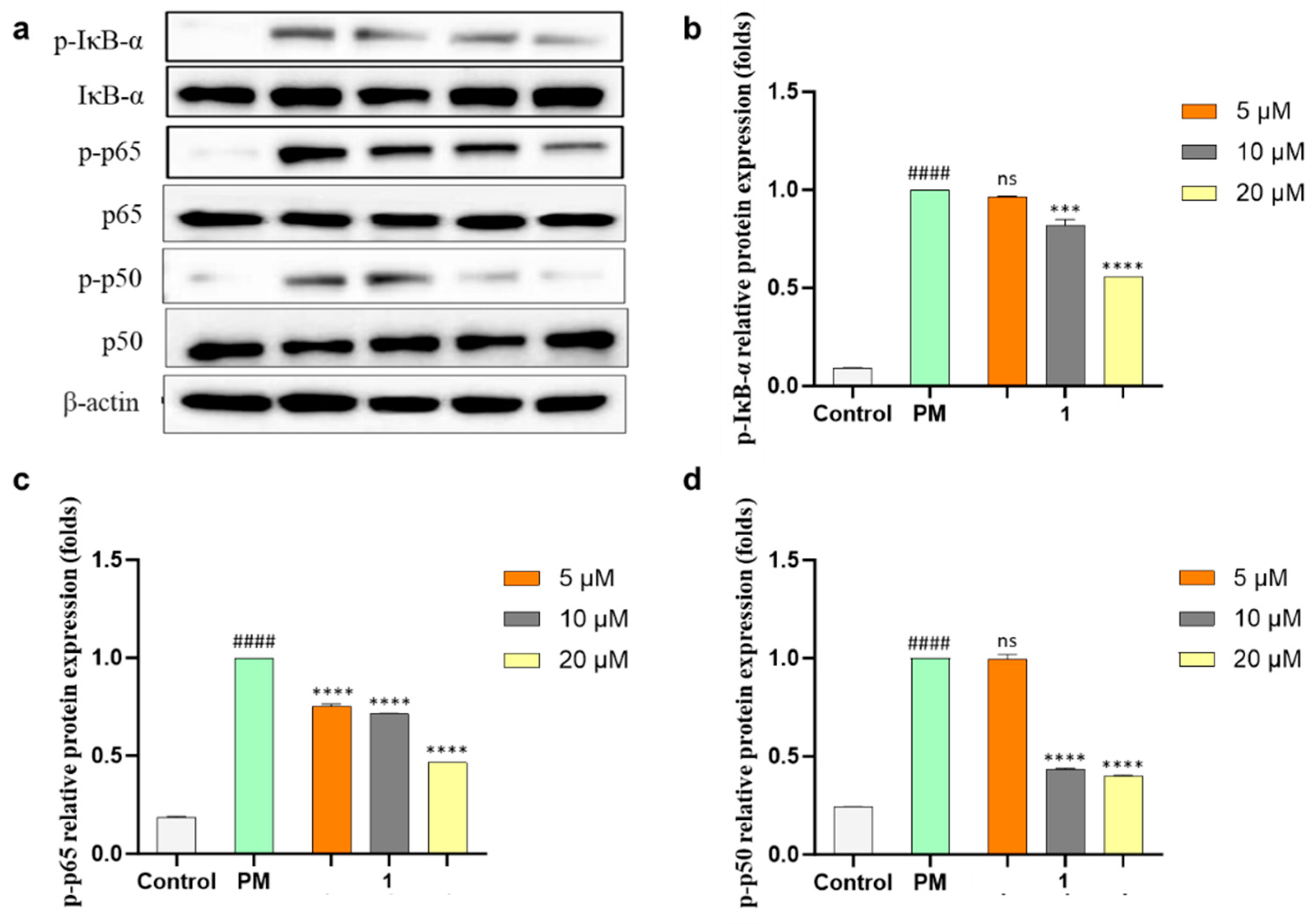
2. Results and Discussion
3. Materials and Methods
3.1. General Experimental Procedures and Reagents
3.2. Strain and Fermentation
3.3. Extraction and Isolation of Compounds
3.4. Computational Analysis
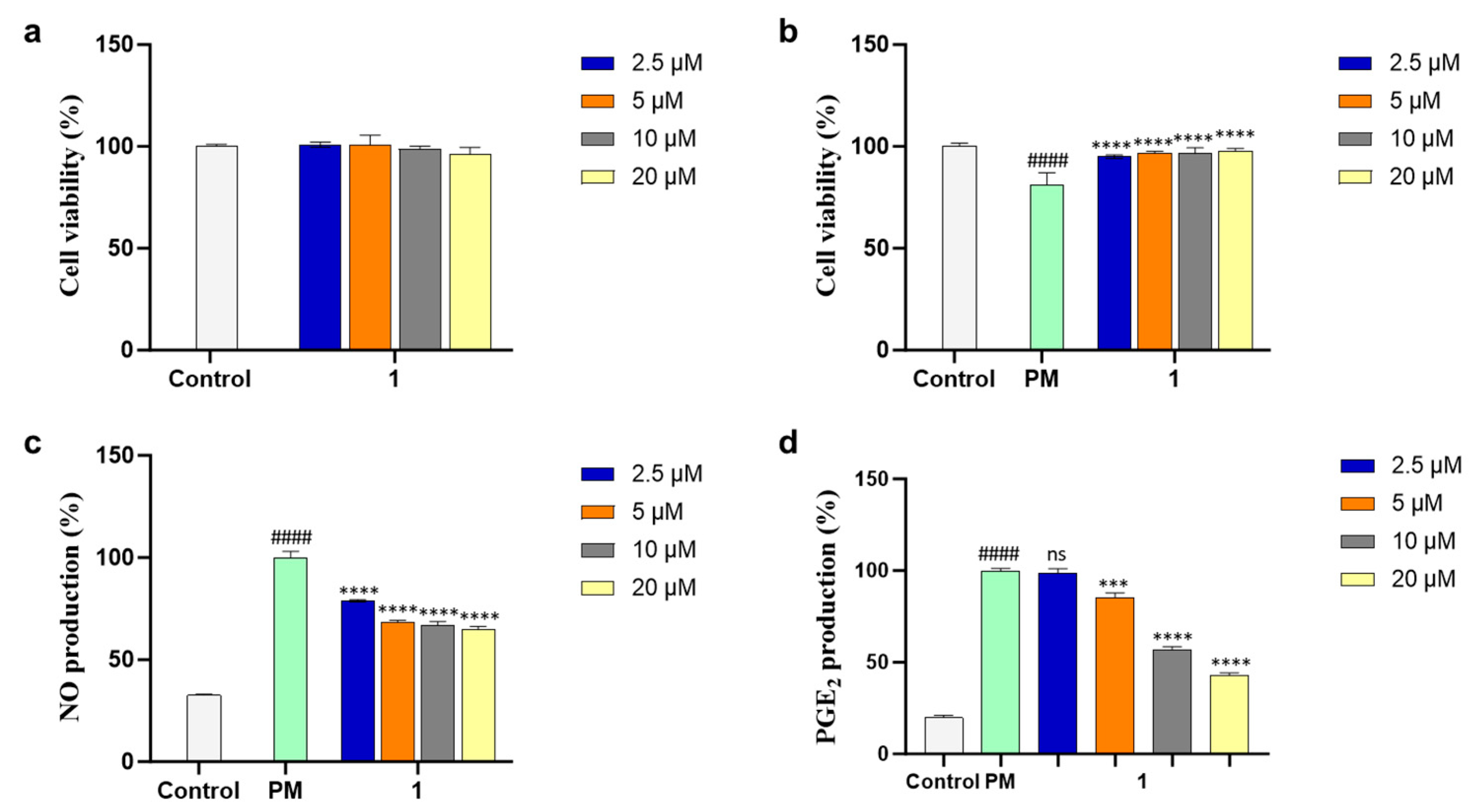
3.5. Cell Culture and Cell Viability Evaluation
3.6. Cytoprotectivity and NO-Inhibitory Activity Evaluation
3.7. Evaluation of PGE2 and Levels of Pro-Inflammatory Cytokines in PM-Stimulated RAW 264.7 Cells
3.8. Gene Expression Analysis
3.9. Protein Expression Evaluation
3.10. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bhatnagar, A. Cardiovascular Effects of Particulate Air Pollution. Annu. Rev. Med. 2022, 73, 393–406. [Google Scholar] [CrossRef]
- Thangavel, P.; Park, D.; Lee, Y.C. Recent Insights into Particulate Matter (PM)-Mediated Toxicity in Humans: An Overview. Int. J. Environ. Res. Public Health 2022, 19, 7511. [Google Scholar] [CrossRef] [PubMed]
- Hantrakool, S.; Kumfu, S.; Chattipakorn, S.C.; Chattipakorn, N. Effects of Particulate Matter on Inflammation and Thrombosis: Past Evidence for Future Prevention. Int. J. Environ. Res. Public Health 2022, 19, 8771. [Google Scholar] [CrossRef]
- Newman, D.J.; Cragg, G.M. Natural Products as Sources of New Drugs over the Nearly Four Decades from 01/1981 to 09/2019. J. Nat. Prod. 2020, 83, 770–803. [Google Scholar] [CrossRef] [PubMed]
- Pfleiderer, A.; Lagier, J.C.; Armougom, F.; Robert, C.; Vialettes, B.; Raoult, D. Culturomics identified 11 new bacterial species from a single anorexia nervosa stool sample. Eur. J. Clin. Microbiol. Infect. Dis. 2013, 32, 1471–1481. [Google Scholar] [CrossRef]
- Fritz, S.; Rajaonison, A.; Chabrol, O.; Raoult, D.; Rolain, J.M.; Merhej, V. Full-length title: NRPPUR database search and in vitro analysis identify an NRPS-PKS biosynthetic gene cluster with a potential antibiotic effect. BMC Bioinform. 2018, 19, 463. [Google Scholar] [CrossRef] [PubMed]
- Nagle, D.G.; Zhou, Y.D.; Park, P.U.; Paul, V.J.; Rajbhandari, I.; Duncan, C.J.G.; Pasco, D.S. A new indanone from the marine cyanobacterium Lyngbya majuscula that inhibits hypoxia-induced activation of the VEGF promoter in Hep3B cells. J. Nat. Prod. 2000, 63, 1431–1433. [Google Scholar] [CrossRef] [PubMed]
- Yoshihira, K.; Fukuoka, M.; Kuroyanagi, M.; Natori, S. 1-Indanone Derivatives from Bracken, Pteridium aquilinum var. latiusculum. Chem. Pharm. Bull. 1971, 19, 1491–1495. [Google Scholar] [CrossRef][Green Version]
- Saraux, N.; Cretton, S.; Kilicaslan, O.S.; Occioni, C.; Ferro, A.; Quirós-Guerrero, L.; Karimou, S.; Christen, P.; Cuendet, M. Isolation and Structure Elucidation of Compounds from Sesamum alatum and Their Antiproliferative Activity against Multiple Myeloma Cells. J. Nat. Prod. 2022, 85, 2706–2713. [Google Scholar] [CrossRef]
- Ito, T.; Tanaka, T.; Iinuma, M.; Nakaya, K.; Takahashi, Y.; Sawa, R.; Murata, J.; Darnaedi, D. Three new resveratrol oligomers from the stem bark of Vatica pauciflora. J. Nat. Prod. 2004, 67, 932–937. [Google Scholar] [CrossRef]
- Ge, X.; Ye, G.; Li, P.; Tang, W.J.; Gao, J.L.; Zhao, W.M. Cytotoxic diterpenoids and sesquiterpenoids from Pteris multifida. J. Nat. Prod. 2008, 71, 227–231. [Google Scholar] [CrossRef]
- Teruya, T.; Nakagawa, S.; Koyama, T.; Suenaga, K.; Kita, M.; Uemura, D. Nakiterpiosin, a novel cytotoxic C-nor-D-homosteroid from the Okinawan sponge Terpios hoshinota. Tetrahedron Lett. 2003, 44, 5171–5173. [Google Scholar] [CrossRef]
- Nakashima, T.; Boonsnongcheep, P.; Kimura, T.; Iwatsuki, M.; Sato, N.; Nonaka, K.; Prathanturarug, S.; Takahashi, Y.; Omura, S. New compounds, nanaomycin F and G, discovered by physicochemical screening from a culture broth of Streptomyces rosa subsp. notoensis OS-3966. J. Biosci. Bioeng. 2015, 120, 596–600. [Google Scholar]
- Zhao, Q.; Chen, G.D.; Feng, X.L.; Yu, Y.; He, R.R.; Li, X.X.; Huang, Y.; Zhou, W.X.; Guo, L.D.; Zheng, Y.Z.; et al. Nodulisporiviridins A-H, Bioactive Viridins from Nodulisporium sp. J. Nat. Prod. 2015, 78, 1221–1230. [Google Scholar] [CrossRef]
- Yang, L.; Qin, L.H.; Bligh, S.W.A.; Bashall, A.; Zhang, C.F.; Zhang, M.A.; Wang, Z.T.; Xu, L.S. A new phenanthrene with a spirolactone from Dendrobium chrysanthum and its anti-inflammatory activities. Bioorg. Med. Chem. 2006, 14, 3496–3501. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Zhao, Y.J.; Zhao, Y.L.; Liu, Y.P.; Li, X.N.; Zhang, H.B.; Luo, X.D. Exploring Aporphine as Anti-inflammatory and Analgesic Lead from Dactylicapnos scandens. Org. Lett. 2020, 22, 257–260. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.L.; Chen, M.Y.; Hou, Y.; Wang, C.H.; Sun, Z.C.; Yang, Y.; Liang, H.Q.; Ma, G.X.; Xu, X.D.; Wei, J.H. Cadinane-Type Sesquiterpenoids with Cytotoxic Activity from the Infected Stems of the Semi-mangrove Hibiscus tiliaceus. J. Nat. Prod. 2022, 85, 127–135. [Google Scholar] [CrossRef] [PubMed]
- Kawamoto, Y.; Noguchi, N.; Kobayashi, T.; Ito, H. Total Synthesis of Lucidumone through Convenient One-pot Preparation of the Tetracyclic Skeleton by Claisen Rearrangement and Subsequent Intramolecular Aldol Reaction. Angew. Chem. Int. Edit. 2023, 62, e202304132. [Google Scholar] [CrossRef] [PubMed]
- Lan, X.; Guo, S.; Zhao, Y.; Zhang, M.; Zhang, D.; Leng, A.; Ying, X. A novel skeleton alkaloid from Portulaca oleracea L. and its bioactivities. Fitoterapia 2023, 169, 105608. [Google Scholar] [CrossRef]
- Hayakawa, Y.; Kobayashi, T.; Izawa, M. Indanostatin, a new neuroprotective compound from Streptomyces sp. J. Antibiot. 2013, 66, 731–733. [Google Scholar] [CrossRef]
- Liang, W.Q.; Xu, G.J.; Weng, D.; Gao, B.; Zheng, X.F.; Qian, Y. Anti-Osteoporotic Components of Rubus chingii. Chem. Nat. Compd. 2015, 51, 47–49. [Google Scholar] [CrossRef]
- Amatov, T.; Jangra, H.; Pohl, R.; Cisarova, I.; Zipse, H.; Jahn, U. Unique Stereoselective Homolytic C-O Bond Activation in Diketopiperazine-Derived Alkoxyamines by Adjacent Amide Pyramidalization. Chem. Eur. J. 2018, 24, 15336–15345. [Google Scholar] [CrossRef] [PubMed]
- Wattana-Amorn, P.; Charoenwongsa, W.; Williams, C.; Crump, M.P.; Apichaisataienchote, B. Antibacterial activity of cyclo(L-Pro-L-Tyr) and cyclo(D-Pro-L-Tyr) from Streptomyces sp. strain 22-4 against phytopathogenic bacteria. Nat. Prod. Res. 2016, 30, 1980–1983. [Google Scholar] [CrossRef] [PubMed]
- Xiang, W.X.; Liu, Q.; Li, X.M.; Lu, C.H.; Shen, Y.M. Four pairs of proline-containing cyclic dipeptides from Nocardiopsis sp. HT88, an endophytic bacterium of Mallotus nudiflorus L. Nat. Prod. Res. 2020, 34, 2219–2224. [Google Scholar] [CrossRef]
- Phongsopitanun, W.; Kanchanasin, P.; Khanboon, A.; Pittayakhajonwut, P.; Suwanborirux, K.; Tanasupawat, S. Marine Streptomyces chumphonensis KK1-2T produces piericidin A1 as the major secondary metabolite. Sci. Asia 2021, 47, 271–276. [Google Scholar] [CrossRef]
- Bruhn, T.; Schaumlöffel, A.; Hemberger, Y.; Pescitelli, G. SpecDis Version 1.71, Berlin, Germany. 2017. Available online: http://specdis-software.jimdo.com (accessed on 29 November 2023).
- Nagahawatta, D.P.; Liyanage, N.M.; Jayawardhana, H.; Lee, H.G.; Jayawardena, T.U.; Jeon, Y.J. Anti-Fine Dust Effect of Fucoidan Extracted from Ecklonia maxima Leaves in Macrophages via Inhibiting Inflammatory Signaling Pathways. Mar. Drugs 2022, 20, 413. [Google Scholar] [CrossRef]
- Nagahawatta, D.P.; Lee, H.G.; Liyanage, N.M.; Jayawardhana, H.H.A.C.K.; Wang, L.; Kim, H.S.; Jeon, Y.J. Alginic acid, a functional dietary ingredient derived from Ecklonia maxima stipe, attenuates the pro-inflammatory responses on particulate matter-induced lung macrophages. J. Funct. Foods 2023, 104, 105539. [Google Scholar] [CrossRef]
- Ko, E.Y.; Cho, S.H.; Kwon, S.H.; Eom, C.Y.; Jeong, M.S.; Lee, W.; Kim, S.Y.; Heo, S.J.; Ahn, G.; Lee, K.P.; et al. The roles of NF-κB and ROS in regulation of pro-inflammatory mediators of inflammation induction in LPS-stimulated zebrafish embryos. Fish Shellfish Immunol. 2017, 68, 525–529. [Google Scholar] [CrossRef]


| No | δC, Type | δH, Mult. (J in Hz) |
|---|---|---|
| 1 | 209.7, C | |
| 2 | 36.9, CH2 | 2.52 t (overlapped) |
| 3 | 27.6, CH2 | 2.93 m 2.66 m |
| 4 | 180.0, C | |
| 5 | 136.2, C | |
| 6 | 132.5, C | |
| 7 | 131.2, CH | 5.70 br s |
| 8 | 39.3, CH | 2.54 m (overlapped) |
| 9 | 75.0, CH | 4.26 d (12.2) |
| 10 | 62.9, CH2 | 4.33 d (13.6) 4.29 d (13.6) |
| 11 | 18.4, CH3 | 1.20 d (7.2) |
| No | Primer Name | Sequence |
|---|---|---|
| 1 | GAPDH forward | 5′-AAGGGTCATCATCTCTGCCC-3′ |
| 2 | GAPDH reverse | 5′-GTGATGGCATGGACTGTGGT-3′ |
| 3 | TNF-α forward | 5′-TTGACCTCAGCGCTGAGTTG-3′ |
| 4 | TNF-α reverse | 5′-CCTGTAGCCCACGTCGTAGC-3′ |
| 5 | IL-6 forward | 5′-GTACTCCAGAAGACCAGAGG-3′ |
| 6 | IL-6 reverse | 5′-TGCTGGTGACAACCACGGCC-3′ |
| 7 | IL-1β forward | 5′-CAGGATGAGGACATGAGCACC-3′ |
| 8 | IL-1β reverse | 5′-CTCTGCAGACTCAAACTCCAC-3′ |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, H.-S.; Nagahawatta, D.P.; Jeon, Y.-J.; Lee, M.A.; Heo, C.-S.; Park, S.J.; Shin, H.J. Streptinone, a New Indanone Derivative from a Marine-Derived Streptomyces massiliensis, Inhibits Particulate Matter-Induced Inflammation. Mar. Drugs 2023, 21, 640. https://doi.org/10.3390/md21120640
Lee H-S, Nagahawatta DP, Jeon Y-J, Lee MA, Heo C-S, Park SJ, Shin HJ. Streptinone, a New Indanone Derivative from a Marine-Derived Streptomyces massiliensis, Inhibits Particulate Matter-Induced Inflammation. Marine Drugs. 2023; 21(12):640. https://doi.org/10.3390/md21120640
Chicago/Turabian StyleLee, Hwa-Sun, Dineth Pramuditha Nagahawatta, You-Jin Jeon, Min Ah Lee, Chang-Su Heo, Sun Joo Park, and Hee Jae Shin. 2023. "Streptinone, a New Indanone Derivative from a Marine-Derived Streptomyces massiliensis, Inhibits Particulate Matter-Induced Inflammation" Marine Drugs 21, no. 12: 640. https://doi.org/10.3390/md21120640
APA StyleLee, H.-S., Nagahawatta, D. P., Jeon, Y.-J., Lee, M. A., Heo, C.-S., Park, S. J., & Shin, H. J. (2023). Streptinone, a New Indanone Derivative from a Marine-Derived Streptomyces massiliensis, Inhibits Particulate Matter-Induced Inflammation. Marine Drugs, 21(12), 640. https://doi.org/10.3390/md21120640