Extraction of Fatty Acids and Phenolics from Mastocarpus stellatus Using Pressurized Green Solvents
Abstract
:1. Introduction
2. Results
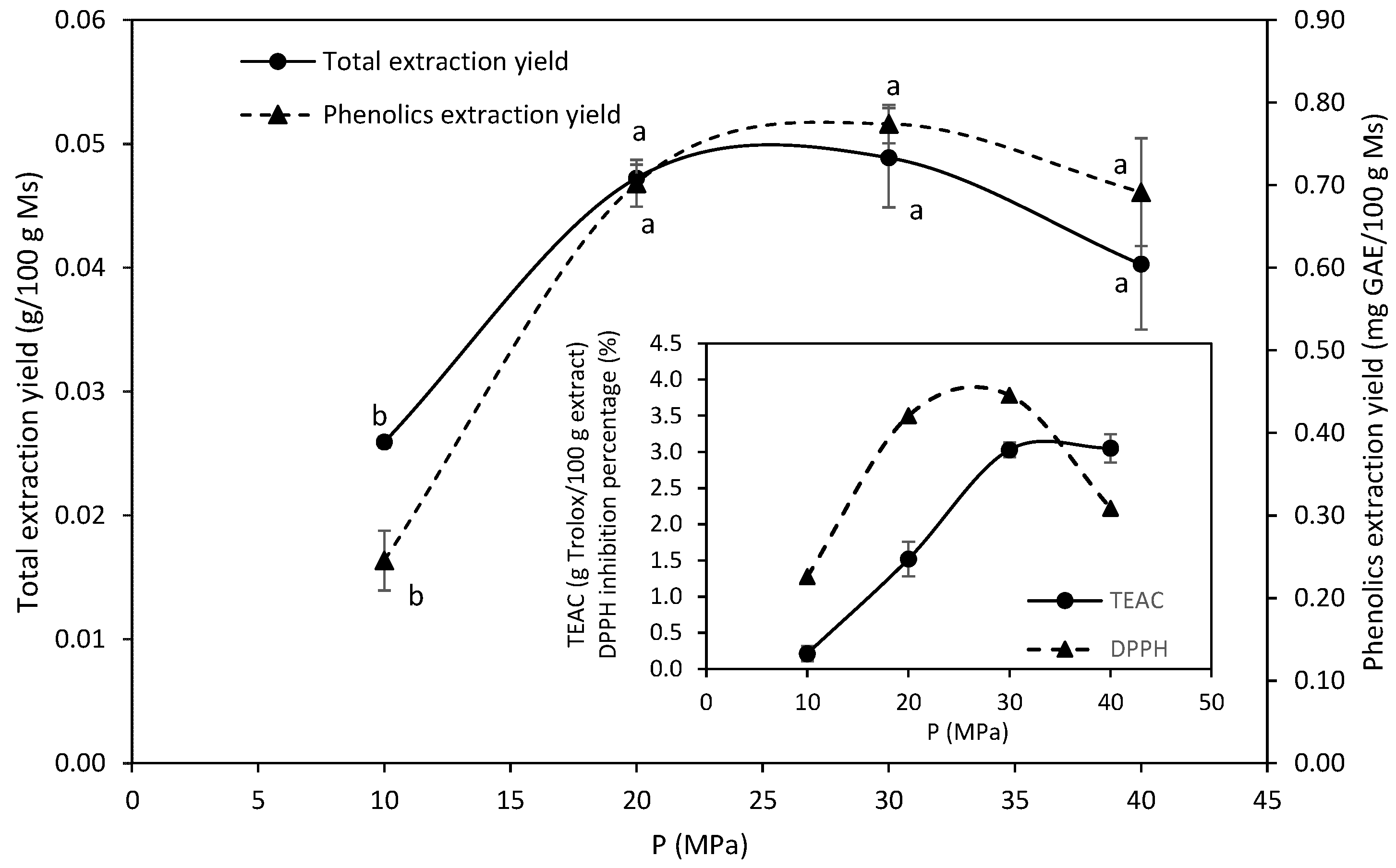
2.1. Supercritical Fluid Extraction
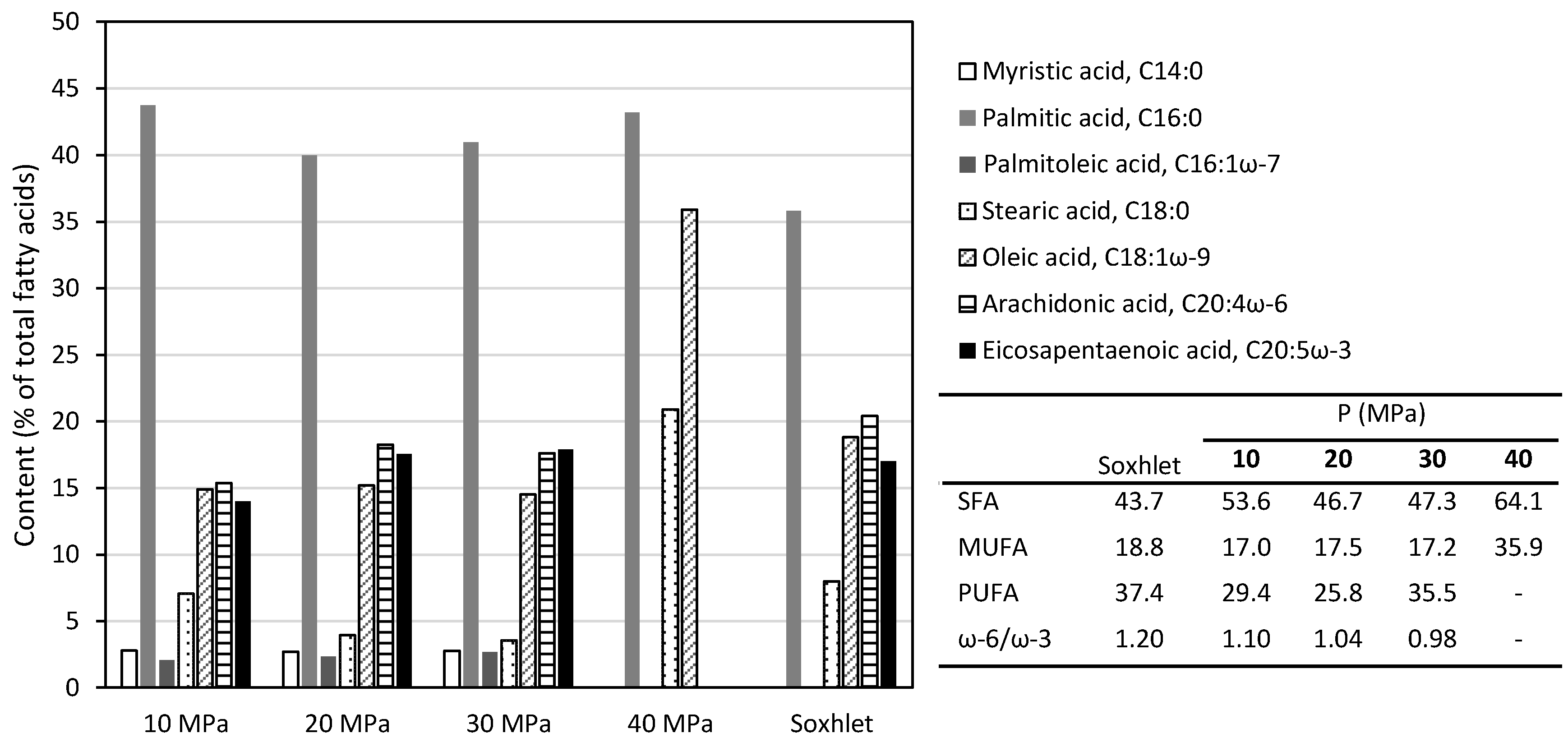
2.1.1. Pure Carbon Dioxide
2.1.2. Supercritical Extraction with Ethanol Modified CO2
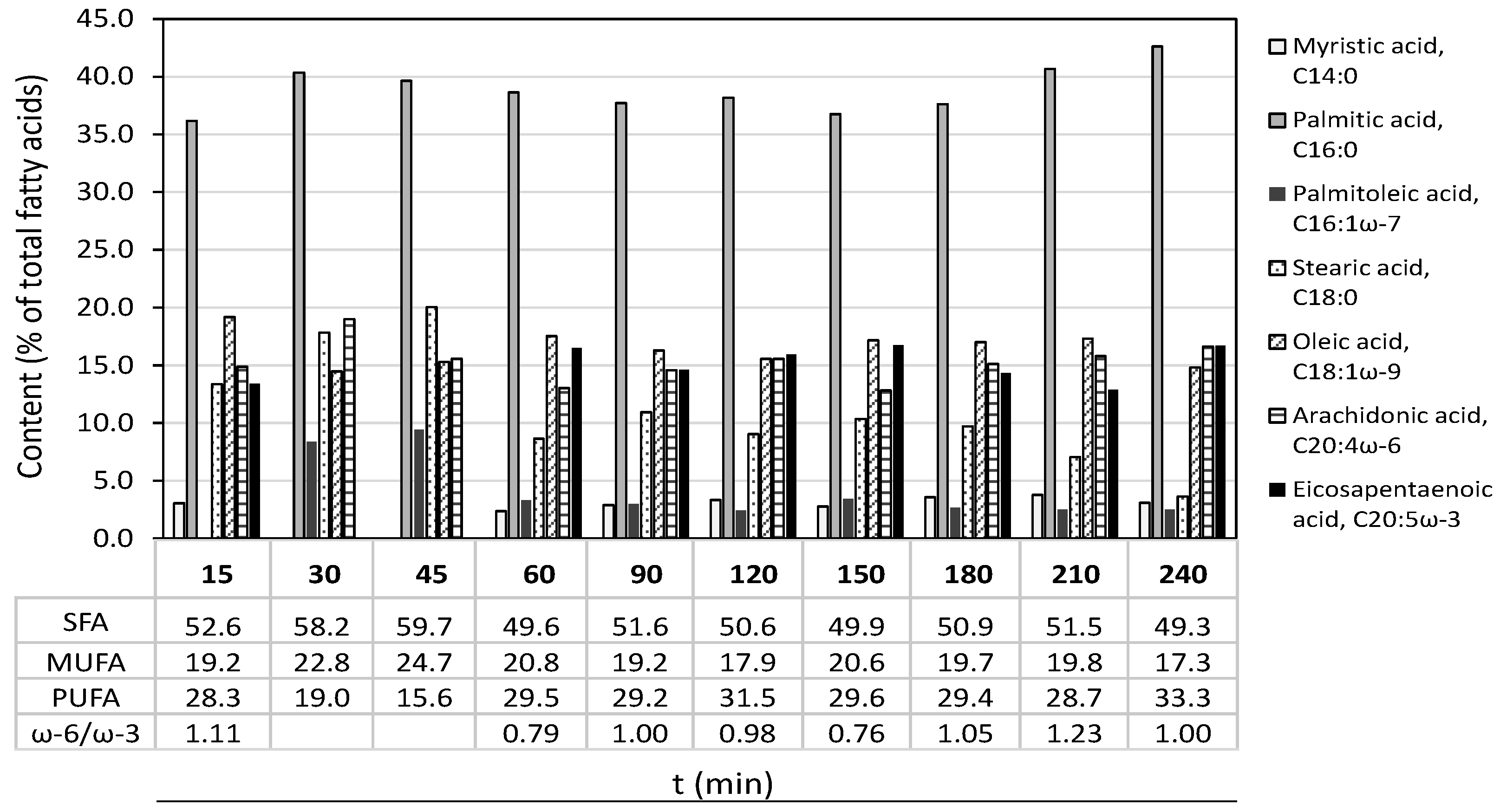
2.2. Microwave-Assisted Subcritical Water Extraction
3. Materials and Methods
3.1. Seaweed
3.2. Soxhlet Extraction
3.3. Supercritical CO2 Extraction
3.4. Microwave-Assisted Subcritical Water Extraction
3.5. Analytical Methods
3.6. Fatty Acid Composition
3.7. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ahmad, A.; Ahsan, H. Lipid-based formulations in cosmeceuticals and biopharmaceuticals. Biomed. Dermatol. 2020, 4, 12. [Google Scholar] [CrossRef]
- Yang, M.; Zhou, M.; Song, L. A review of fatty acids influencing skin condition. J. Cosmet. Dermatol. 2020, 19, 3199–3204. [Google Scholar] [CrossRef] [PubMed]
- Huang, T.-H.; Wang, P.W.; Yang, S.C.; Chou, W.L.; Fang, J.Y. Cosmetic and Therapeutic Applications of Fish Oil’s Fatty Acids on the Skin. Mar. Drugs 2018, 16, 256. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thomsen, B.J.; Chow, E.Y.; Sapijaszko, M.J. The potential uses of omega-3 fatty acids in dermatology: A review. J. Cutan. Med. Surg. 2020, 24, 481–494. [Google Scholar] [CrossRef]
- Afifi, L.; Danesh, M.J.; Lee, K.M.; Beroukhim, K.; Farahnik, B.; Ahn, R.S.; Yan, D.; Singh, R.K.; Nakamura, M.; Koo, J.; et al. Dietary Behaviors in Psoriasis: Patient-Reported Outcomes from a U.S. National Survey. Dermatol. Ther. 2017, 7, 227–242. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.Y.; Kwon, H.H.; Hong, J.S.; Yoon, J.Y.; Park, M.S.; Jang, M.Y.; Suh, D.H. Effect of dietary supplementation with omega-3 fatty acid and gamma-linolenic acid on acne vulgaris: A randomised, double-blind, controlled trial. Acta Derm. Venereol. 2014, 94, 521–525. [Google Scholar] [CrossRef] [Green Version]
- Melnik, B.C. Linking diet to acne metabolomics, inflammation, and comedogenesis: An update. Clin. Cosmet. Investig. Dermatol. 2015, 8, 371–388. [Google Scholar] [CrossRef] [Green Version]
- Aslan, I.; Ozcan, F.; Karaarslan, T.; Kirac, E.; Aslan, M. Decreased eicosapentaenoic acid levels in acne vulgaris reveals the presence of a proinflammatory state. Prostaglandins Other Lipid Mediat. 2017, 128, 1–71. [Google Scholar] [CrossRef] [PubMed]
- Ames, F.Q.; Bracht, L.; Sato, F.; Vizioli, L.; Ambrósio, B.; Oliveira, L.A.D.; Parreira de Lima, E.; Kenji Nakamura Cuman, R.; Luciano Baesso, M.; Aparecida Bersani-Amado, C. Fish oil preparation inhibits leukocyte recruitment and bands that characterize inflamed tissue in a model of phenol-induced skin inflammation: Percutaneous penetration of a topically applied preparation demonstrated by photoacoustic spectroscopy. Nat. Prod. Res. 2020, 34, 2341–2345. [Google Scholar] [CrossRef]
- Upala, S.; Yong, W.C.; Theparee, T.; Sanguankeo, A. Effect of omega-3 fatty acids on disease severity in patients with psoriasis: A systematic review. Int. J. Rheum. Dis. 2017, 20, 442–450. [Google Scholar] [CrossRef]
- Jara, C.P.; Mendes, N.F.; Prado, T.P.D.; de Araújo, E.P. Bioactive Fatty Acids in the Resolution of Chronic Inflammation in Skin Wounds. Adv. Wound Care 2020, 9, 472–490. [Google Scholar] [CrossRef]
- Dorweiler, B.; Trinh, T.T.; Dünschede, F.; Vahl, C.F.; Debus, E.S.; Storck, M.; Diener, H. The marine Omega3 wound matrix for treatment of complicated wounds: A multicenter experience report. Gefasschirurgie 2018, 23, 46–55. [Google Scholar] [CrossRef] [Green Version]
- Kim, S.-K. Marine cosmeceuticals. J. Cosm. Dermat. 2014, 13, 56–67. [Google Scholar] [CrossRef]
- Naser, W. The cosmetic effects of various natural biofunctional ingredients against skin aging: A review. Int. J. Appl. Pharm. 2021, 13, 10–18. [Google Scholar] [CrossRef]
- Rubio-Rodríguez, N.; Beltrán, S.; Jaime, I.; de Diego, S.M.; Sanz, M.T.; Carballido, J.R. Production of omega-3 polyunsaturated fatty acid concentrates: A review. Innov. Food Sci. Emerg. Technol. 2010, 11, 1–12. [Google Scholar] [CrossRef]
- Pereira, H.; Barreira, L.; Figueiredo, F.; Custódio, L.; Vizetto-Duarte, C.; Polo, C.; Rešek, E.; Engelen, A.; Varela, J. Polyunsaturated fatty acids of marine macroalgae: Potential for nutritional and pharmaceutical applications. Mar. Drugs 2012, 10, 1920–1935. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Conde, E.; Moure, A.; Domínguez, H. Supercritical CO2 extraction of fatty acids, phenolics and fucoxanthin from freeze-dried Sargassum muticum. J. Appl. Phycol. 2015, 27, 957–964. [Google Scholar] [CrossRef]
- Balboa, E.M.; Conde, E.; Moure, A.; Falqué, E.; Domínguez, H. In vitro antioxidant properties of crude extracts and compounds from brown algae. Food Chem. 2013, 138, 1764–1785. [Google Scholar] [CrossRef] [PubMed]
- Machmudah, S.; Wahyu, D.; Kanda, H.; Goto, M. Supercritical fluids extraction of valuable compounds from algae: Future perspectives and challenges. Eng. J. 2018, 22, 13–30. [Google Scholar] [CrossRef]
- Crampon, C.; Boutin, O.; Badens, E. Supercritical carbon dioxide extraction of molecules of interest from microalgae and seaweeds. Ind. Eng. Chem. Res. 2011, 50, 8941–8953. [Google Scholar] [CrossRef]
- Cruz, P.N.; Fetzer, D.L.; do Amaral, W.; de Andrade, E.F.; Corazza, M.L.; Masson, M.L. Antioxidant activity and fatty acid profile of yacon leaves extracts obtained by supercritical CO2 + ethanol solvent. J. Supercrit. Fluids 2019, 146, 55–64. [Google Scholar] [CrossRef]
- Cheung, P.C.K.; Leung, A.Y.H.; Ang, P.O., Jr. Comparison of supercritical carbon dioxide and soxhlet extraction of lipids from a brown seaweed, Sargassum hemiphyllum (Turn) C. Ag. J. Agric. Food Chem. 1998, 46, 4228–4232. [Google Scholar] [CrossRef]
- Devi, V.; Khanam, S. Optimization of the ratio of ω-6 linoleic and ω-3 α-linolenic fatty acids of hemp seed oil with Jackknife and Bootstrap resampling. Chem. Prod. Process Model. 2019, 15, 20190028. [Google Scholar] [CrossRef] [Green Version]
- Fernandes, S.S.; Tonato, D.; Mazutti, M.A.; de Abreu, B.R.; da Costa Cabrera, D.; D’Oca, C.D.R.M.; Prentice-Hernández, C.; Salas-Mellado, M.D.L.M. Yield and quality of chia oil extracted via different methods. J. Food Eng. 2019, 262, 200–208. [Google Scholar] [CrossRef]
- Sabeena Farvin, K.H.; Jacobsen, C. New natural antioxidants for protecting omega-3 rich products. Lipid Technol. 2012, 24, 59–62. [Google Scholar] [CrossRef]
- Dellarosa, N.; Laghi, L.; Martinsdóttir, E.; Jónsdóttir, R.; Sveinsdóttir, K. Enrichment of convenience seafood with omega-3 and seaweed extracts: Effect on lipid oxidation. LWT-Food Sci. Technol. 2015, 62, 746–752. [Google Scholar] [CrossRef]
- Rudke, A.R.; de Andrade, C.J.; Ferreira, S.R.S. Kappaphycus alvarezii macroalgae: An unexplored and valuable biomass for green biorefinery conversion. Trends Food Sci. Technol. 2020, 103, 214–224. [Google Scholar] [CrossRef]
- Álvarez-Viñas, M.; Rodríguez-Seoane, P.; Flórez-Fernández, N.; Torres, M.D.; Díaz-Reinoso, B.; Moure, A.; Domínguez, H. Subcritical water for the extraction and hydrolysis of protein and other fractions in biorefineries from agro-food wastes and algae: A review. Food Bioproc. Tech. 2020, 14, 373–387. [Google Scholar] [CrossRef]
- Ponthier, E.; Domínguez, H.; Torres, M.D. The microwave assisted extraction sway on the features of antioxidant compounds and gelling biopolymers from Mastocarpus stellatus. Algal Res. 2020, 51, 102081. [Google Scholar] [CrossRef]
- Cheung, P.C.K. Temperature and pressure effects on supercritical carbon dioxide extraction of n-3 fatty acids from red seaweed. Food Chem. 1999, 65, 399–403. [Google Scholar] [CrossRef]
- Rozo, G.; Rozo, C.; Puyana, M.; Ramos, F.A.; Almonacid, C.; Castro, H. Two compounds of the Colombian algae Hypnea musciformis prevent oxidative damage in human low density lipoproteins LDLs. J. Func. Foods 2019, 60, 103399. [Google Scholar] [CrossRef]
- Kumar, L.R.G.; Treesa Paul, P.; Anas, K.K.; Tejpal, C.S.; Chatterjee, N.S.; Anupama, T.K.; Geethalakshmi, V.; Anandan, R.; Jayarani, R.; Mathew, S. Screening of effective solvents for obtaining antioxidant-rich seaweed extracts using principal component analysis. J. Food Process. Preserv. 2020, 44, e14716. [Google Scholar] [CrossRef]
- Barral-Martínez, L.; Flórez-Fernández, N.; Domínguez, H.; Torres, M.D. Tailoring hybrid carrageenans from Mastocarpus stellatus red seaweed using microwave hydrodiffusion and gravity. Carbohydr. Polym. 2020, 248, 116830. [Google Scholar] [CrossRef] [PubMed]
- Ospina, M.; Castro-Vargas, H.I.; Parada-Alfonso, F. Antioxidant capacity of Colombian seaweeds: 1. Extracts obtained from Gracilaria mammillaris by means of supercritical fluid extraction. J. Supercrit. Fluids 2017, 128, 314–322. [Google Scholar] [CrossRef]
- Shipeng, Y.; Woo, H.C.; Choi, J.H.; Park, Y.B.; Chun, B.S. Measurement of antioxidant activities and phenolic and flavonoid contents of the brown seaweed Sargassum horneri: Comparison of supercritical CO2 and various solvent extractions. Fish. Aquat. Sci. 2015, 18, 123–1301. [Google Scholar]
- Siahaan, E.A.; Pangestuti, R.; Chun, B.S. Antioxidant activity of two edible Korean seaweed oil obtained from SC-CO2 and solvent extraction. E3S Web Conf. 2020, 147, 03014. [Google Scholar] [CrossRef] [Green Version]
- Meireles, M.A.A. Extraction of Bioactive Compounds from Latin American Plants. In Supercritical Fluid Extraction of Nutraceuticals and Bioactive Compounds; Martinez, J.L., Ed.; CRC Press: Boca Raton, FL, USA, 2007; pp. 243–274. [Google Scholar]
- Pereira, C.G.; Meireles, M.A.A. Supercritical fluid extraction of bioactive compounds: Fundamentals, applications and economic perspectives. Food Bioprocess Technol. 2010, 3, 340–372. [Google Scholar] [CrossRef]
- Mittal, A.; Sara, U.V.S.; Ali, A.; Aqil, M. Status of fatty acids as skin penetration enhancers-A review. Curr. Drug Deliv. 2009, 6, 274–279. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.; Liu, Z.; Li, J.; Nie, S.; Pan, W. Effects of isopropyl palmitate on the skin permeation of drugs. Biol. Pharm. Bull. 2006, 29, 2324–2326. [Google Scholar] [CrossRef] [Green Version]
- Viljoen, J.M.; Cowley, A.; Du Preez, J.; Gerber, M.; du Plessis, J. Penetration enhancing effects of selected natural oils utilized in topical dosage forms. Drug Dev. Ind. Pharm. 2015, 41, 2045–2054. [Google Scholar] [CrossRef] [PubMed]
- Storey, A.; McArdle, F.; Friedmann, P.S.; Jackson, M.J.; Rhodes, L.E. Eicosapentaenoic acid and docosahexaenoic acid reduce UVB- and TNFalpha- induced IL-8 secretion in keratinocytes and UVB-induced IL-8 in fibroblasts. J. Investig. Dermatol. 2005, 124, 248–255. [Google Scholar] [CrossRef]
- Arantes, E.L.; Dragano, N.; Ramalho, A.; Vitorino, D.; de-Souza, G.F.; Lima, M.H.M.; Velloso, L.C.; Araújo, E.P. Topical docosahexaenoic acid (DHA) accelerates skin wound healing in rats and activates GPR120. Biol. Res. Nurs. 2016, 18, 411–419. [Google Scholar] [CrossRef]
- Álvarez-Viñas, M.; Flórez-Fernández, N.; Torres, M.D.; Domínguez, H. Successful approaches for a red seaweed biorefinery. Mar. Drugs 2019, 17, 620. [Google Scholar] [CrossRef] [Green Version]
- Peñuela, A.; Robledo, D.; Bourgougnon, N.; Bedoux, G.; Hernández-Núñez, E.; Freile-Pelegrín, Y. Environmentally friendly valorization of Solieria filiformis (Gigartinales, Rhodophyta) from IMTA using a biorefinery concept. Mar. Drugs 2018, 16, 487. [Google Scholar] [CrossRef] [Green Version]
- Rocha, C.P.; Pacheco, D.; Cotas, J.; Marques, J.C.; Pereira, L.; Ana, M.M. Gonçalves. Seaweeds as Valuable Sources of Essential Fatty Acids for Human Nutrition. Int. J. Environ. Res. Public Health 2021, 18, 4968. [Google Scholar] [CrossRef]
- Singleton, V.L.; Rossi, J.A. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye Binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- von Gadow, A.; Jounert, E.; Hansmann, C.F. Comparison of the antioxidant activitiy aspalathin with that of other plant phenols of Rooibos tea (Aspalathus linearis), α-Tocopherol, BHT and BHA. J. Agric. Food Chem. 1997, 45, 632–638. [Google Scholar] [CrossRef]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]


| Adjusted Parameters | R2 | |
|---|---|---|
| tCER (min) | 70.01 | 0.994 |
| MCER (g/min) | 5.39 × 10−4 | |
| YCER (g extract/g CO2) | 2.16 × 10−5 | |
| xCER (%) | 0.038 | |
| S/FCER | 17.5 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cid, U.; Rodríguez-Seoane, P.; Díaz-Reinoso, B.; Domínguez, H. Extraction of Fatty Acids and Phenolics from Mastocarpus stellatus Using Pressurized Green Solvents. Mar. Drugs 2021, 19, 453. https://doi.org/10.3390/md19080453
Cid U, Rodríguez-Seoane P, Díaz-Reinoso B, Domínguez H. Extraction of Fatty Acids and Phenolics from Mastocarpus stellatus Using Pressurized Green Solvents. Marine Drugs. 2021; 19(8):453. https://doi.org/10.3390/md19080453
Chicago/Turabian StyleCid, Uxía, Paula Rodríguez-Seoane, Beatriz Díaz-Reinoso, and Herminia Domínguez. 2021. "Extraction of Fatty Acids and Phenolics from Mastocarpus stellatus Using Pressurized Green Solvents" Marine Drugs 19, no. 8: 453. https://doi.org/10.3390/md19080453
APA StyleCid, U., Rodríguez-Seoane, P., Díaz-Reinoso, B., & Domínguez, H. (2021). Extraction of Fatty Acids and Phenolics from Mastocarpus stellatus Using Pressurized Green Solvents. Marine Drugs, 19(8), 453. https://doi.org/10.3390/md19080453






