Characterization of Low Molecular Weight Sulfate Ulva Polysaccharide and its Protective Effect against IBD in Mice
Abstract
1. Introduction
2. Results and Discussion
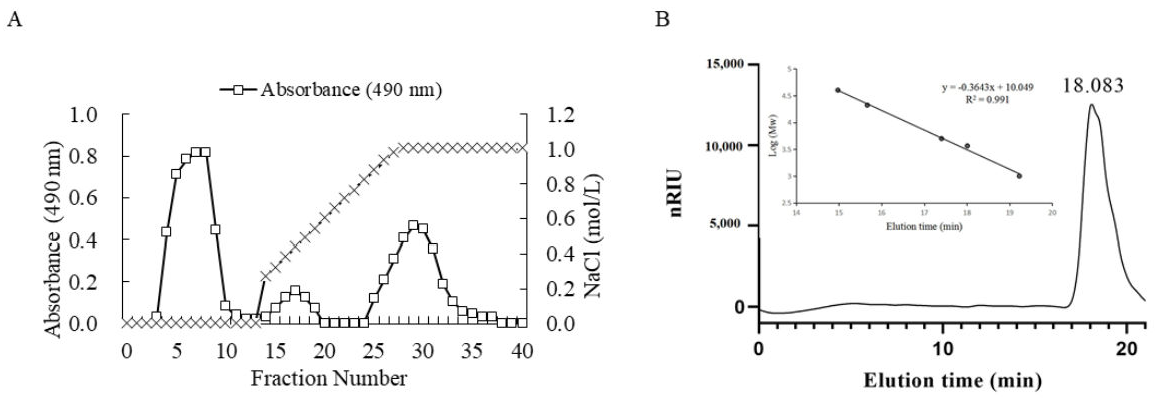
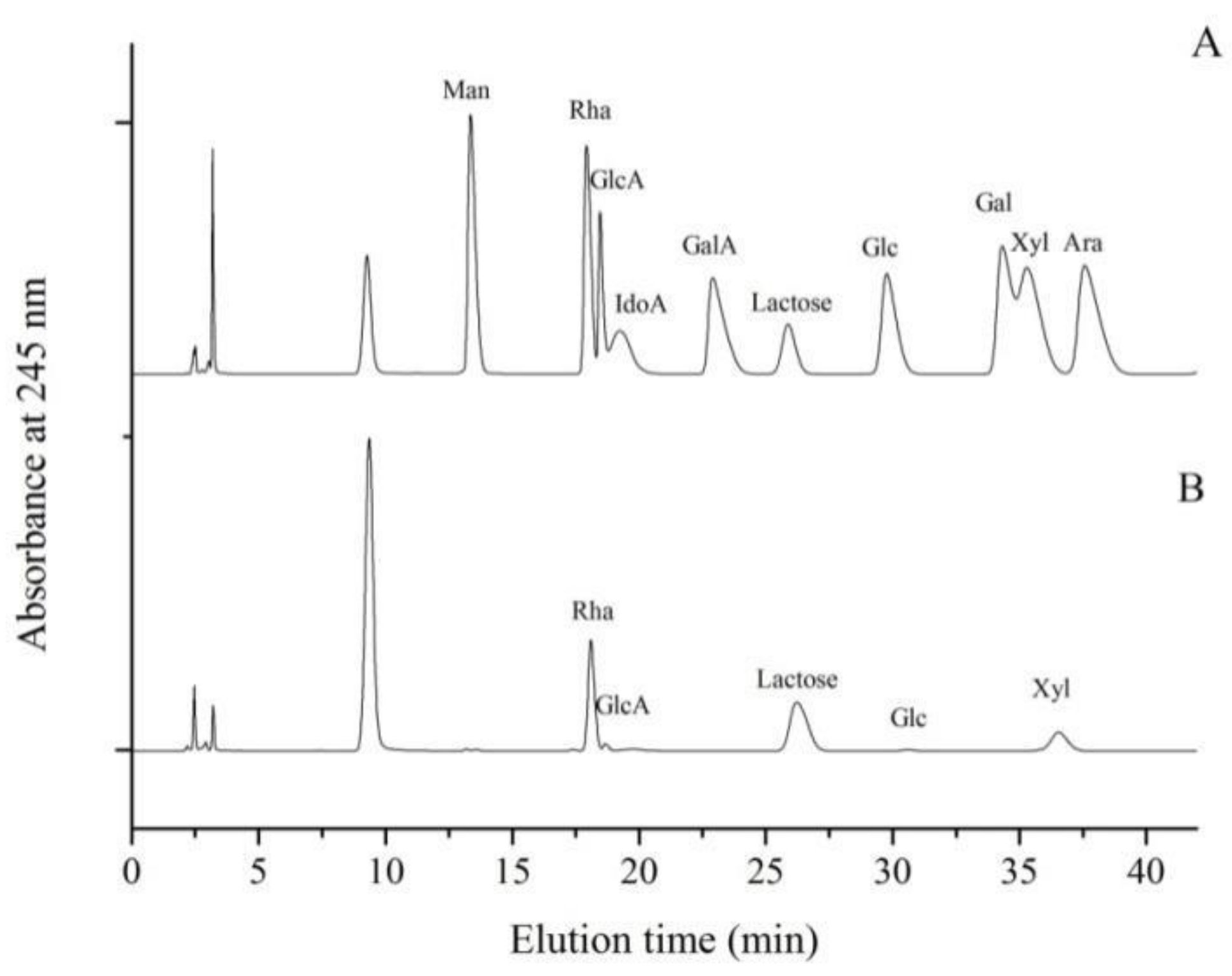
2.1. Characterization of LMW-Ulvan
2.1.1. Determination of Molecular Weight of LMW-Ulvan
2.1.2. Monosaccharide Composition Analysis of LMW-Ulvan
2.1.3. Methylation and Gas Chromatography-Mass Spectrometer (GC-MS) Analysis of LMW-Ulvan
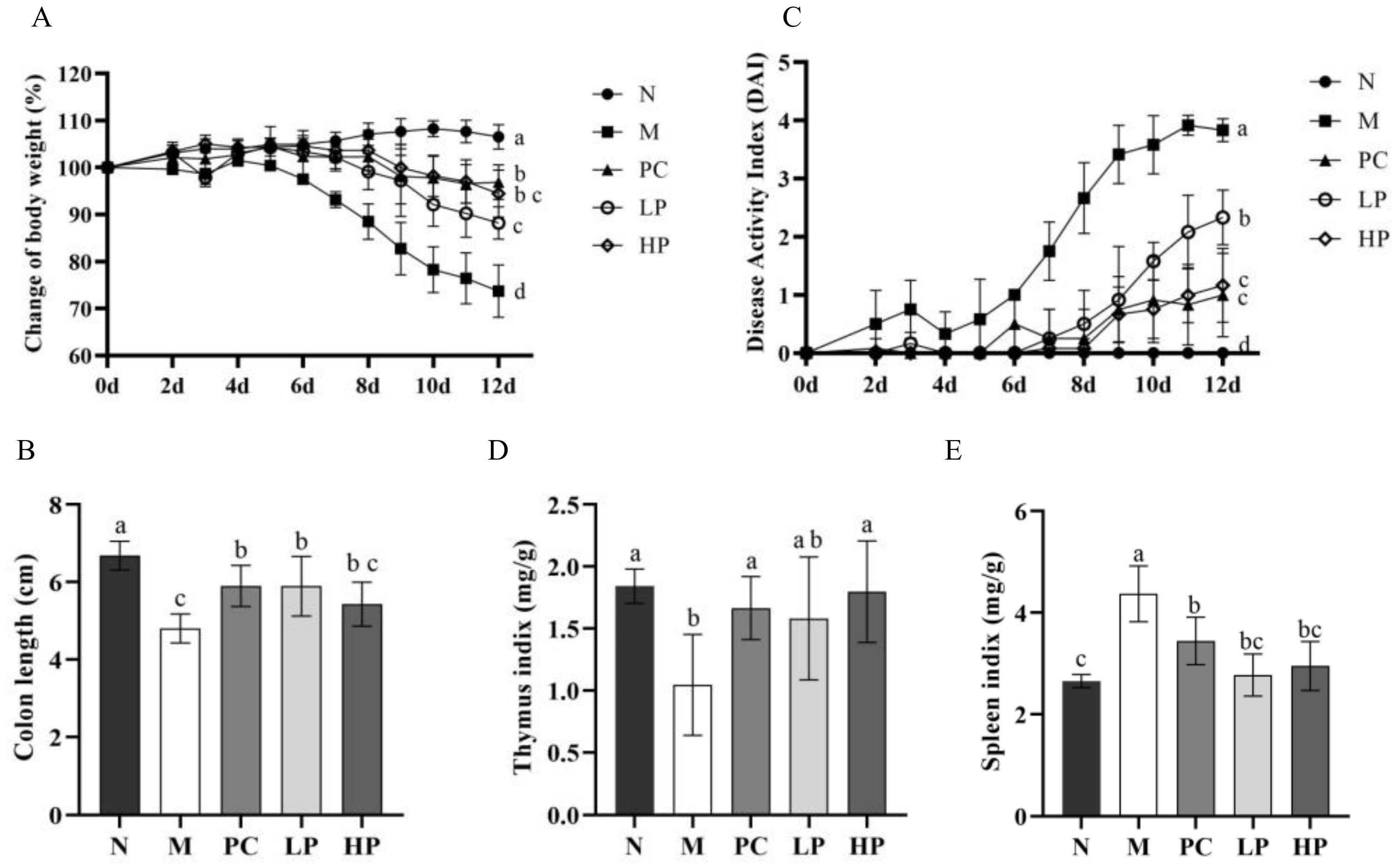
2.2. LMW-Ulvan Relieves the Manifestations of IBD Induced by DSS
2.3. LMW-Ulvan Reverses the Histological Injury of Colonic Epithelium Caused by DSS
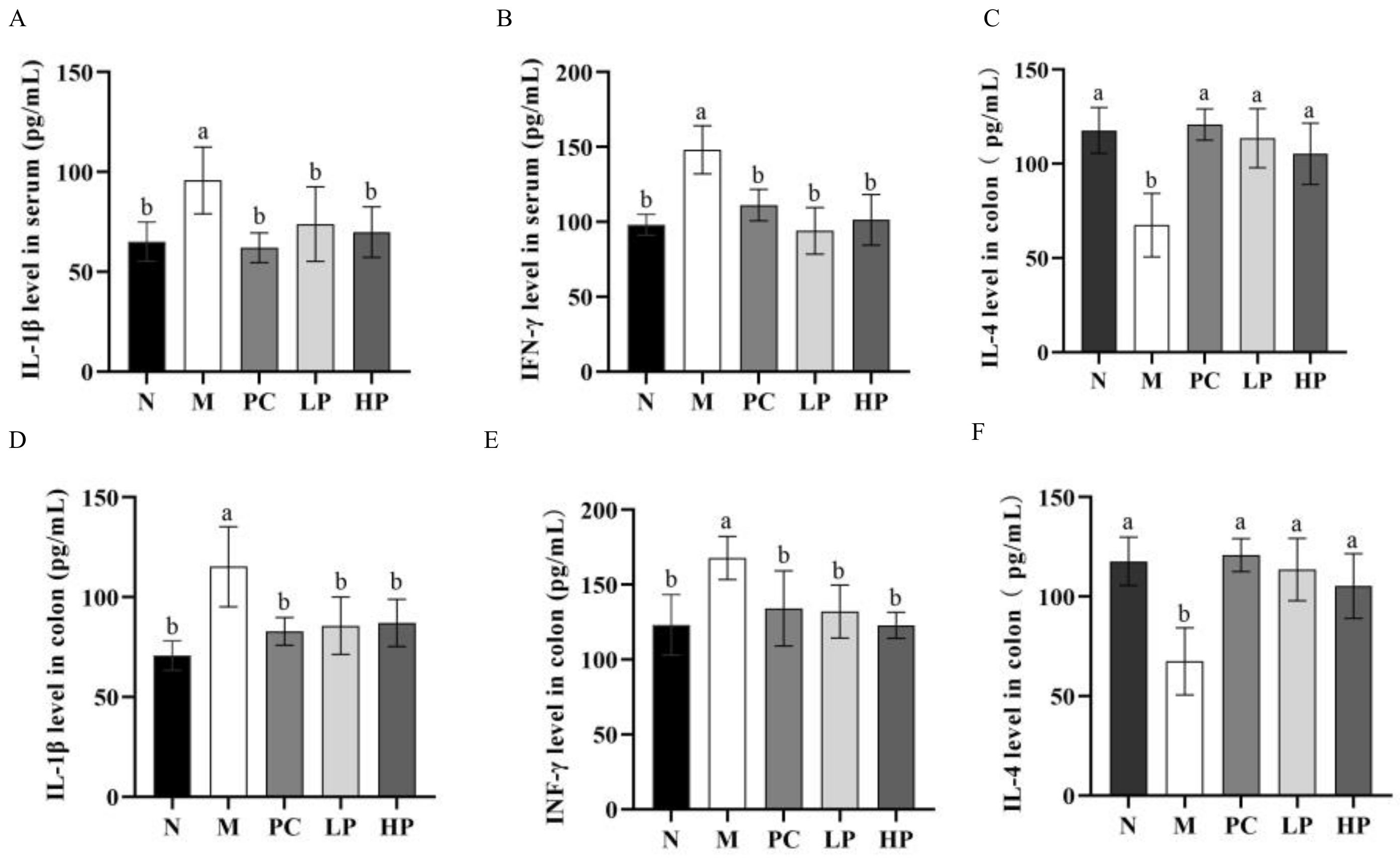
2.4. Effects of LMW-Ulvan on Cytokines in Colon Tissue and Serum
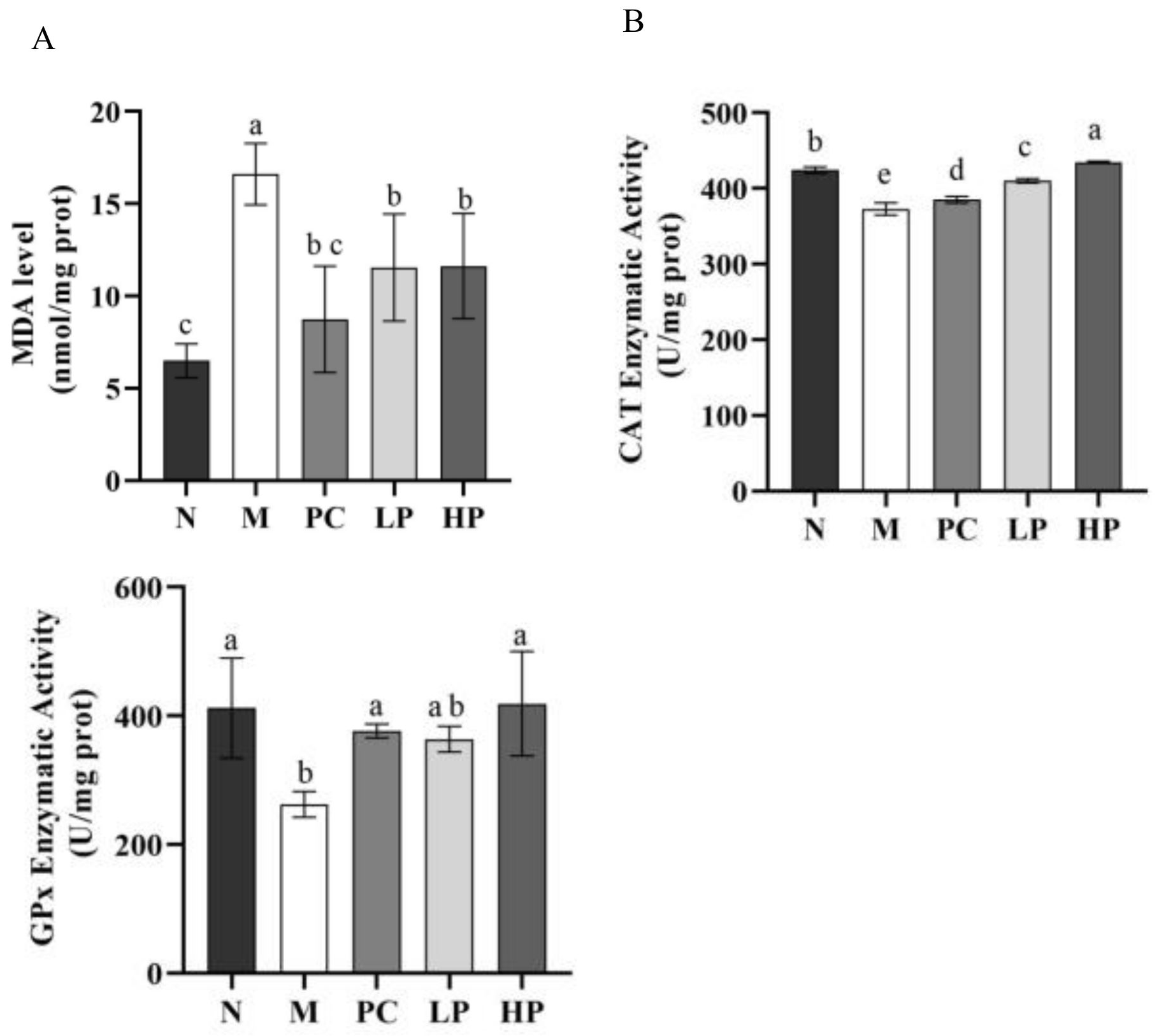
2.5. Effects of LMW-Ulvan on Malondialdehyde (MDA), GPx, and Catalase (CAT) in Colonic Tissue
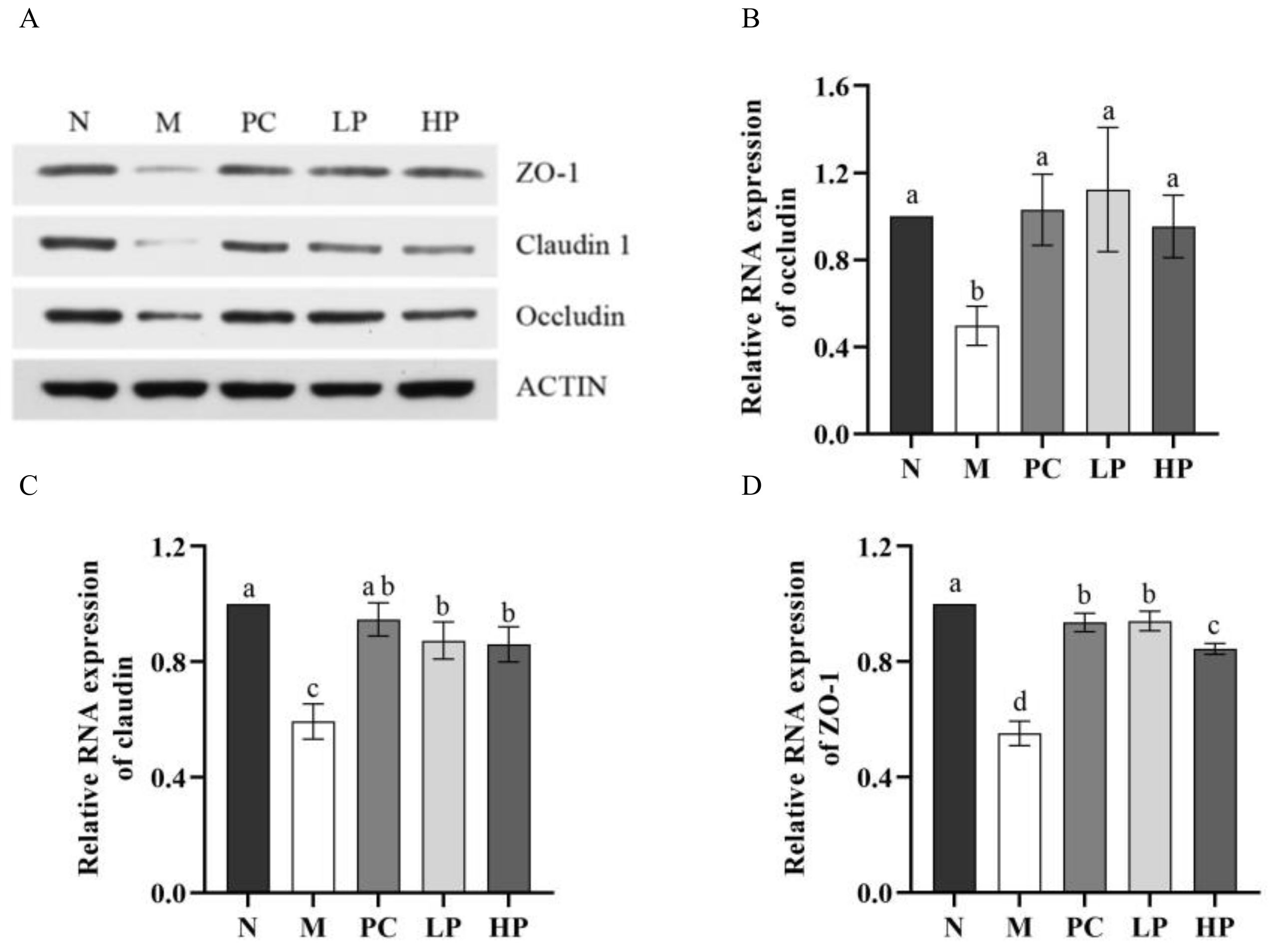
2.6. Effect of LMW-Ulvan on mRNA Expression of Colonic Epithelial Tight Junction Protein
3. Materials and Methods
3.1. Materials and Reagents
3.2. Enzymatic Preparation and Purification of LMW-Ulvan
3.3. Characterization of LMW-Ulvan
3.4. Methylation Analysis of LMW-Ulvan
3.5. Animals
3.6. The DSS-Induced Colitis Model
3.7. Assessment of Severity of Colitis
3.8. Cytokines and Activities of Antioxidant Enzyme Assay
3.9. Quantitative Real-Time Polymerase Chain Reaction (qRT-PCR) Analysis
3.10. Western Blot (WB) Assay
3.11. Statistics Analysis
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Shao, S.; Wang, D.D.; Zheng, W.; Li, X.Y.; Zhang, H.; Zhao, D.Q.; Wang, M.X. A unique polysaccharide from Hericiumerinaceus mycelium ameliorates acetic acid-induced ulcerative colitis rats by modulating the composition of the gut microbiota, short chain fatty acids levels and GPR41/43 respectors. Int. Immunopharmacol. 2019, 71, 411–422. [Google Scholar] [CrossRef] [PubMed]
- Jin, M.Y.; Wang, Y.X.; Yang, X.B.; Yin, H.; Nie, S.P.; Wu, X.Y. Structure characterization of a polysaccharide extracted from noni (Morindacitrifolia, L.) and its protective effect against DSS-induced bowel disease in mice. Food Hydrocolloid. 2019, 90, 189–197. [Google Scholar] [CrossRef]
- Minerwilliams, W.; Moughan, P.J. Intestinal barrier dysfunction: Implications for chronic inflammatory conditions of the bowel. Nutr. Res. Rev. 2016, 29, 40–59. [Google Scholar] [CrossRef] [PubMed]
- Hou, J.K.; Lee, D.; Lewis, J.D. Diet and inflammatory bowel disease: Review of patient-targeted recommendations. Clin. Gastroenterol. H. 2014, 12, 1592–1600. [Google Scholar] [CrossRef]
- Rogler, G.; Vavricka, S.R. Exposome in IBD: Recent insights in environmental factors that influence the onset and course of IBD. Inflamm. Bowel Dis. 2015, 21, 400–408. [Google Scholar] [CrossRef]
- Kanwal, S.; Joseph, T.P.; Aliya, S.; Song, S.Y.; Zubair, M.; Saleem, Z.; Nisar, M.A.; Wang, Y.; Meyiah, A.; Ma, Y.F.; et al. Attenuation of DSS induced colitis by Dictyophoraindusiata polysaccharide (DIP) via modulation of gut microbiota and inflammatory related signaling pathways. J. Funct. Foods 2020, 64, 103641. [Google Scholar] [CrossRef]
- Hu, T.; Lin, Q.L.; Guo, T.; Yang, T.; Zhou, W.H.; Deng, X.F.; Yan, J.K.; Luo, Y.; Ju, M.M.; Luo, F.J. Polysaccharide isolated from Phellinus linteus mycelia exerts anti-inflammatory effects via MAPK and PPAR signaling pathways. Carbohyd. Polym. 2018, 200, 487–497. [Google Scholar] [CrossRef]
- Wang, Y.L.; Ji, X.M.; Yan, M.L.; Chen, X.; Kang, M.Z.; Teng, L.P.; Wu, X.T.; Chen, J.H.; Deng, C. Protective effect and mechanism of polysaccharide from Dictyophoraindusiata on dextran sodium sulfate-induced colitis in C57BL/6 mice. Int. J. Biol. Macromol. 2019, 140, 973–984. [Google Scholar] [CrossRef]
- Rodriguezcanales, M.; Jimenezrivas, R.; Canalesmartinez, M.M.; Garcialopez, A.J.; Riverayanez, N.; Nietoyanez, O.; Ledesmasoto, Y.; Sancheztorres, L.E.; Rodriguezsosa, M.; Terrazas, L.I.; et al. Protective effect of amphipterygiumadstringens extract on dextran sulphate sodium-induced ulcerative colitis in mice. Mediat. Inflamm. 2016, 2016, 8543561. [Google Scholar]
- Shen, Y.M.; Zou, J.F.; Chen, M.J.; Zhang, Z.M.; Liu, C.; Jiang, S.; Qian, D.; Duan, J.A. Protective effects of Lizhong decoction on ulcerative colitis in mice by suppressing inflammation and ameliorating gut barrier. J. Ethnopharmacol. 2020, 259, 112919. [Google Scholar] [CrossRef]
- Perrier, C.; Rutgeerts, P. Cytokine blockade in inflammatory bowel diseases. Immunotherapy 2011, 3, 1341–1352. [Google Scholar] [CrossRef] [PubMed]
- Ji, Y.; Dai, Z.L.; Sun, S.Q.; Ma, X.S.; Yang, Y.; Tso, P.; Wu, G.Y.; Wu, Z.L. Hydroxyproline attenuates dextran sulfate sodium induced colitis in mice: Involvment of the NF-κB signaling and oxidative stress. Mol. Nutr. Food Res. 2018, 62, 1800494. [Google Scholar] [CrossRef]
- Tziveleka, L.; Ioannou, E.; Roussis, V. Ulvan, a bioactive marine sulphated polysaccharide as a key constituent of hybrid biomaterials: A review. Carbohyd. Polym. 2019, 218, 355–370. [Google Scholar] [CrossRef] [PubMed]
- Shi, Q.; Wang, A.; Lu, Z.; Qin, C.; Hu, J.; Yin, J. Overview on the antiviral activities and mechanisms of marine polysaccharides from seaweeds. Carbohyd. Res. 2017, 453, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Wijesekara, I.; Pangestuti, R.; Kim, S. Biological activities and potential health benefits of sulfated polysaccharides derived from marine algae. Carbohyd. Polym. 2011, 84, 14–21. [Google Scholar] [CrossRef]
- Qi, H.; Zhao, T.; Zhang, Q.; Li; Z.; Zhao, Z.; Xing, R. Antioxidant activity of different molecular weight sulfated polysaccharides from Ulva pertusaKjellm (Chlorophyta). J. Appl. Phycol. 2005, 17, 527–534. [Google Scholar] [CrossRef]
- Li, W.; Jiang, N.; Li, B.; Wang, M.; Chang, X.; Liu, H.; Zhang, L.; Yin, S.; Qi, H.; Liu, S. Antioxidant activity of purified ulvan in hyperlipidemic mice. Int. J. Biol. Macromol. 2018, 113, 971–975. [Google Scholar] [CrossRef]
- De Araujo, I.W.; Rodrigues, J.A.; Quindere, A.L.; Silva, J.F.; Maciel, G.F.; Ribeiro, N.A.; de Sousa Oliveira Vanderlei, E.; Ribeiro, K.A.; Chaves, H.V.; Pereira, K.M.; et al. Analgesic and anti-inflammatory actions on bradykinin route of a polysulfated fraction from alga Ulva lactuca. Int. J. Biol. Macromol. 2016, 92, 820–830. [Google Scholar] [CrossRef]
- Berri, M.; Oliver, M.; Holbert, S.; Dupont, J.; Demais, H.; Goff, M.L.; Collen, P.N. Ulvan from Ulva armoricana (Chlorophyta) activates the PI3K/Akt signaling pathway via TLR4 to induce intestinal cytokine production. Algal Res. 2017, 28, 39–47. [Google Scholar] [CrossRef]
- Chi, Y.Z.; Zhang, M.F.; Wang, X.; Fu, X.J.; Guan, H.S.; Wang, P. Ulvan lyase assisted structural characterization of ulvan from Ulva pertusa and its antiviral activity against vesicular stomatitis virus. Int. J. Biol. Macromol. 2020, 157, 75–82. [Google Scholar] [CrossRef]
- Ulaganathan, T.; Boniecki, M.T.; Foran, E.; Buravenkov, V.; Mizrachi, N.; Banin, E.; Helbert, W.; Cygler, M. New Ulvan-degrading polysaccharide lyase Family: Structure and catalytic mechanism suggests convergent evolution of active site architecture. ACS Chem. Biol. 2017, 12, 1269–1280. [Google Scholar] [CrossRef]
- Yu, Y.; Li, Y.P.; Du, C.Y.; Mou, H.J.; Wang, P. Compositional and structural characteristics of sulfated polysaccharide from Enteromorpha prolifera. Carbohyd. Polym. 2017, 165, 221–228. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.P.; Wang, X.P.; Jiang, Y.C.; Wang, J.F.; Hwang, H.M.; Yang, X.H.; Wang, P. Structure characterization of low molecular weight sulfate Ulva polysaccharide and the effect of its derivative on iron deficiency anemia. Int. J. Biol. Macromol. 2019, 126, 747–754. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Li, X.; Chen, J.; Chen, T.J.; Shi, Z.M.; Lei, M.N.; Zhang, Y.J.; Bai, P.F.; Li, Y.F.; Fei, X. The pentacyclic triterpene Lupeol switches M1 macrophages to M2 and ameliorates experimental inflammatory bowel disease. Int. Immunopharmacol. 2016, 30, 74–84. [Google Scholar] [CrossRef] [PubMed]
- Qiu, X.Y.; Li, X.; Wu, Z.; Zhang, F.; Wang, N.; Wu, N.; Yang, X.; Liu, Y.L. Fungal-bacterial interactions in mice with dextran sulfate sodium (DSS)-induced acute and chronic colitis. RSC Adv. 2016, 6, 65995–66006. [Google Scholar] [CrossRef]
- Kim, D.; Lee, M.; Yoo, J.; Park, K.; Ma, J. Fermented herbal formula KIOM-MA-128 protects against acute colitis induced by dextran sodium sulfate in mice. BMC Complem. Altern. M. 2017, 17, 354. [Google Scholar] [CrossRef]
- Zong, S.; Ye, Z.Y.; Zhang, X.M.; Chen, H.; Ye, M. Protective effect of Lachnum polysaccharide on dextran sulfate sodium-induced colitis in mice. Food Funct. 2020, 11, 846–859. [Google Scholar] [CrossRef]
- Abderrazak, A.; Hadri, K.E.; Bosc, E.; Blondeau, B.; Slimane, M.N.; Büchele, B.; Simmet, T.; Couchie, D.; Rouis, M. Inhibition of the inflammasome NLRP3 by arglabin attenuates inflammation, protects pancreatic beta-cells from apoptosis, and prevents Type 2 diabetes mellitus development in ApoE2Ki mice on a chronic high-fat diet. J. Pharmacol. Exp. Ther. 2016, 357, 487–494. [Google Scholar] [CrossRef]
- Zhao, Y.L.; Wang, J.B.; Shan, L.M.; Jin, C.; Xiao, X.H. Effect of Radix isatidis polysaccharides on immunological function and expression of immune related cytokines in mice. Chin. J. Integr. Med. 2008, 14, 207–211. [Google Scholar] [CrossRef]
- Gao, X.; Qu, H.; Gao, Z.L.; Zeng, D.Y.; Wang, J.P.; Baranenko, D.; Li, Y.Z.; Lu, W.H. Protective effects of Ulva pertusa polysaccharide and polysaccharide iron (III) complex on cyclophosphamide induced immunosuppression in mice. Int. J. Biol. Macromol. 2019, 133, 911–919. [Google Scholar] [CrossRef]
- Oestreich, K.J.; Weinmann, A.S. Transcriptional mechanisms that regulate T helper 1cell differentiation. Curr. Opin. Immunol. 2012, 24, 191–195. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.C.; Li, S.; Cao, H.Y.; Shen, P.; Liu, J.X.; Fu, Y.H.; Gao, Y.G.; Zhang, N.S. The protective role of phloretin against dextran sulfate sodium-induced ulcerative colitis in mice. Food Funct. 2019, 10, 422–431. [Google Scholar] [CrossRef] [PubMed]
- Gong, J.; Wang, X.Z.; Wang, T.; Chen, J.J.; Xie, X.Y.; Hu, H.; Yu, F.; Liu, H.L.; Jiang, X.Y.; Fan, H.D. Molecular signal networks and regulating mechanisms of the unfolded protein response. J. Zhejiang Univ. SC. B. 2017, 18, 1–14. [Google Scholar]
- Wang, Y.; Ouyang, Q.; Xia, B.; Liu, L.N.; Gu, F.; Zhou, K.F.; Mei, Q.; Shi, R.H.; Ran, Z.H.; Wang, X.D.; et al. Multicenter case-control study of the risk factors for ulcerative colitis in China. World J. Gastroentero. 2013, 19, 1827–1833. [Google Scholar] [CrossRef]
- Li, R.Y.; Chen, Y.Y.; Shi, M.J.; Xu, X.X.; Zhao, Y.X.; Wu, X.J.; Zhang, Y.B. GegenQinlian decoction alleviates experimental colitis via suppressing TLR4/NF-κB signaling and enhancing antioxidant effect. Phytomedicine 2016, 23, 1012–1020. [Google Scholar] [CrossRef]
- Kjellev, S. The trefoil factor family-small peptides with multiple functionalities. Cell. Mol. Life Sci. 2009, 66, 1350–1369. [Google Scholar] [CrossRef]
- Qiao, L.K.; Yang, X.K.; Xie, R.Z.; Du, C.; Chi, Y.; Zhang, J.; Wang, P. Efficient production of ulvan lyase from Ulva prolifera by Catenovulum sp. LP based on stage-controlled fermentation strategy. Algal Res. 2020, 46, 101812. [Google Scholar]
- DuBois, M.; Gilles, K.A.; Hamilton, J.K.; Rebers, P.A.; Smith, F. Colorimetric method for determination of sugars and related substances. Anal. Chem. 1956, 28, 350–356. [Google Scholar]
- Tuulikki, T.T.; Hartiala, K. Method for determination of the sulfate content of glycosaminoglycans. Anal. Chem. 1971, 41, 471–476. [Google Scholar]
- Ye, H.; Shen, Z.P.; Cui, J.F.; Zhu, Y.Z.; Li, Y.Y.; Chi, Y.Z.; Wang, J.F.; Wang, P. Hypoglycemic activity and mechanism of the sulfated rhamnose polysaccharides chromium(III) complex in type 2 diabetic mice. Bioorg. Chem. 2019, 88, 102942. [Google Scholar] [CrossRef]
- Cui, L.; Wang, W.; Luo, L.; Ning, Q.; Xia, Z.; Chen, J.; Feng, L.; Wang, H.; Song, J.; Tan, X.B.; et al. Polysaccharide from Scutellariabaicalensis Georgi ameliorates colitis via suppressing NF-κB signaling and NLRP3 inflammasome activation. Int. J. Biol. Macromol. 2019, 132, 393–405. [Google Scholar] [CrossRef] [PubMed]
- Falshaw, R.; Furneaux, R.H. Structural analysis of carrageenans from the tetrasporic stages of the red algae, Gigartina lanceata and Gigartina chapmanii (Gigartinaceae, Rhodophyta). Carbohyd. Res. 1998, 307, 325–331. [Google Scholar] [CrossRef]
- Lin, L.Y.; Wang, P.P.; Du, Z.Y.; Wang, W.C.; Cong, Q.F.; Zheng, C.P.; Jin, C.; Ding, K.; Shao, C.H. Structural elucidation of a pectin from flowers of Lonicera japonica and its antipancreatic cancer activity. Int. J. Biol. Macromol. 2016, 88, 130–137. [Google Scholar] [CrossRef]
- Jeon, Y.; Lee, J.; Lee, Y.; Kin, D. Puerarin inhibits inflammation and oxidative stress in dextran sulfate sodium-induced colitis mice model. Biomed. Pharmacother. 2020, 124, 109847. [Google Scholar] [CrossRef]
- Tian, M.; Ma, P.; Zhang, Y.; Mi, Y.; Fan, D.D. Ginsenoside Rk3 alleviated DSS-induced ulcerative colitis by protectingcolon barrier and inhibiting NLRP3 inflammasome pathway. Int. Immunopharmacol. 2020, 85, 106645. [Google Scholar] [CrossRef] [PubMed]

| O-Me-Alditol Acetate | Molar Percentage | Linkage Pattern | |
|---|---|---|---|
| LMW-Ulvan | dsLMW-Ulvan | ||
| 1,4,5-Tri-O-acetrl-2,3-di-O-metyl-L-Rha | 6.65% | 53.32% | →4)-Rha-(1→ |
| 1,3,4,5-Tetro-O-acetrl-2-O-metyl-L-Rha | 50.12% | 4.16% | →3,4)-Rha-(1→ |
| 1,4,5-Tri-O-acetrl-2,3-di-O-metyl-D-xyl | 29.34% | 28.45% | →4)-xyl-(1→ |
| Gene. | Forward | Reverse |
|---|---|---|
| ZO-1 | CGCGGAGAGAGACAAGATGT | AAACCCAGGAGCCCTGTGAA |
| claudin | CAACCACAATAGCGGCATCG | GCACAGACCTGCAAGGAGAT |
| occludin | GATCGTGTTTGCGGATGAGC | AGCCTCCTTAGCTCGTAGTCA |
| β-actin | CAAGGCATTGCTGACAGGATG | TGCTGATCCACATCTGCTGG |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, Y.; Ye, H.; Wang, T.; Wang, P.; Liu, R.; Li, Y.; Tian, Y.; Zhang, J. Characterization of Low Molecular Weight Sulfate Ulva Polysaccharide and its Protective Effect against IBD in Mice. Mar. Drugs 2020, 18, 499. https://doi.org/10.3390/md18100499
Li Y, Ye H, Wang T, Wang P, Liu R, Li Y, Tian Y, Zhang J. Characterization of Low Molecular Weight Sulfate Ulva Polysaccharide and its Protective Effect against IBD in Mice. Marine Drugs. 2020; 18(10):499. https://doi.org/10.3390/md18100499
Chicago/Turabian StyleLi, Yuanyuan, Han Ye, Ting Wang, Peng Wang, Ruizhi Liu, Yinping Li, Yingying Tian, and Jingliang Zhang. 2020. "Characterization of Low Molecular Weight Sulfate Ulva Polysaccharide and its Protective Effect against IBD in Mice" Marine Drugs 18, no. 10: 499. https://doi.org/10.3390/md18100499
APA StyleLi, Y., Ye, H., Wang, T., Wang, P., Liu, R., Li, Y., Tian, Y., & Zhang, J. (2020). Characterization of Low Molecular Weight Sulfate Ulva Polysaccharide and its Protective Effect against IBD in Mice. Marine Drugs, 18(10), 499. https://doi.org/10.3390/md18100499




