The Anticancer Drug Discovery Potential of Marine Invertebrates from Russian Pacific
Abstract
1. Introduction
2. Assessing the Biodiversity of Marine Invertebrates of the North-West Pacific
3. Natural Products from North-West Pacific Invertebrates
4. Marine-Derived Compounds from the North-West Pacific Invertebrates with an Anticancer Potential
4.1. Alkaloids
4.2. Glycosides (Saponins)
4.3. Triterpene Glycosides
4.4. Steroid Glycosides
4.5. Polyketides
4.6. Fatty Acids
5. The Drug Discovery Potential of Invertebrate Compounds of the North-West Pacific
6. Recommendations to Unlock the Drug Discovery Potential of the Russian Pacific
- Regular, multi-vessel marine expeditions, both local (at the seas/oceans of the Russian coastline) and global (far away from the Russian coasts), aiming at the massive collection of diverse marine organisms targeted at subsequent extraction of the natural products, accompanied by the proper microbiological, zoological, algal, etc. investigations. Accompanying molecular biology investigations, aiming at the possibility of medium-quality genomic sequencing, appear to be a useful additional angle to these studies. The ultimate goal of this block of the strategic initiative is the creation and constant enlargement of the massive bank of marine extracts, each entry in which is to be accompanied by extensive biological characterization, live specimen preservation where applicable (e.g., in the case of marine microbial collections), and genomic sequencing data.
- High-throughput multi-scale separation of these extracts, with characterization, maximally permitted for the high-throughput separation, of the resulting mixtures of natural compounds. The ultimate goal of this block of activities is the establishment of a huge collection of fractions and individual natural products in the high-throughput screening-ready format. The scale of this collection should be tens of millions of entries.
- Establishment of a versatile panel of primary screening assays, all of the high-throughput (HTS) format, aiming at identification of biologically active compounds relevant for the broadest range of medical needs: cancer in its various forms, the broad scope of cardiovascular conditions, the broad scope of neurological diseases, the broad scope of infection diseases, etc. Next, continuous and parallel application of these HTS assays to the screening of the primary marine extracts from block 1 and especially of the fraction/compounds collection of block 2. On the regular basis, new acquisitions to the collections are to be rescreened across the whole panel of the primary assays. The ultimate goals of this block of the national program are two-fold. First, establishment and enlargement of the primary screening assays for various health and disease conditions. Second, generation of the primary hits, originating from the marine natural products, as the starting points for the drug discovery programs (further blocks of the national program) against the respective disease conditions.
- The national drug discovery/drug development (DD/DD) program based on the marine natural products will start from these primary hits of the previous block. This DD/DD program will have all the features of the typical big pharma DD/DD programs, but will additionally have certain peculiarities, stemming from the specifics of the source of material. These peculiarities are the re-iterative nature of the whole national initiative “Marine Natural Products in Health and Disease”. Specifically, more marine expeditions may be needed to replenish the source of a particular marine organism for the continuous production of the necessary bioactives. These will be complemented, where applicable, with aquaculturing/microbial culturing of the organisms; with genetic production of the bioactive compounds in artificial hosts (which will require the genomic investigations of block 1 of the biosynthetic routes of the bioactives); and with extensive organic synthesis. Otherwise, the typical hit-to-lead optimization, medicinal chemistry, animal experimentation, and clinical studies will be part of this national marine-based DD/DD program. The ultimate goal of this program will be to continuously deliver to the clinical trials and the market of dozens to hundreds of first-in-class or novel drugs against various disease conditions.
- -
- Marine expeditions and sample collection, including deep-diving expertise.
- -
- Marine zoology, microbiology, algal biology, marine ecology; and other aspects of marine biology.
- -
- Marine molecular biology, sequencing, genomics, and metagenomics, including big data generation and operations.
- -
- Intensive separation and analytics techniques, including massive creation and storage of sample collections.
- -
- Massive establishment of HTS-ready primary screening assays for the broadest range of health and disease conditions.
- -
- Massive organic synthesis of marine compounds and derivatives.
- -
- Marine microbiology and synthetic biology (biotechnological-scale production of marine natural products in natural and engineered hosts).
- -
- HTS, drug discovery, and drug development centers of competence on the national scale, possessing capacities all the way from the HTS to clinical trials.
7. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Bernardini, S.; Tiezzi, A.; Laghezza Masci, V.; Ovidi, E. Natural products for human health: An historical overview of the drug discovery approaches. Nat. Prod. Res. 2018, 32, 1926–1950. [Google Scholar] [CrossRef] [PubMed]
- Harvey, A.L.; Edrada-Ebel, R.; Quinn, R.J. The re-emergence of natural products for drug discovery in the genomics era. Nat. Rev. Drug Discov. 2015, 14, 111–129. [Google Scholar] [CrossRef] [PubMed]
- Altmann, K.H. Drugs from the Oceans: Marine Natural Products as Leads for Drug Discovery. Chimia (Aarau) 2017, 71, 646–652. [Google Scholar] [CrossRef] [PubMed]
- Snelgrove, P.V. An Ocean of Discovery: Biodiversity Beyond the Census of Marine Life. Planta Med. 2016, 82, 790–799. [Google Scholar] [CrossRef] [PubMed]
- Adrianov, A.V. Current Problems in Marine Biodiversity Studies. Russ. J. Mar. Biol. 2004, 30, S1–S16. [Google Scholar] [CrossRef]
- Jimenez, P.C.; Wilke, D.V.; Costa-Lotufo, L.V. Marine drugs for cancer: Surfacing biotechnological innovations from the oceans. Clinics (Sao Paulo) 2018, 73 (Suppl. 1), e482s. [Google Scholar] [CrossRef] [PubMed]
- Newman, D.J.; Cragg, G.M. Marine-sourced anti-cancer and cancer pain control agents in clinical and late preclinical development. Mar. Drugs 2014, 12, 255–278. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Torres, V.; Encinar, J.A.; Herranz-Lopez, M.; Perez-Sanchez, A.; Galiano, V.; Barrajon-Catalan, E.; Micol, V. An Updated Review on Marine Anticancer Compounds: The Use of Virtual Screening for the Discovery of Small-Molecule Cancer Drugs. Molecules 2017, 22, 1037. [Google Scholar] [CrossRef] [PubMed]
- Song, X.; Xiong, Y.; Qi, X.; Tang, W.; Dai, J.; Gu, Q.; Li, J. Molecular Targets of Active Anticancer Compounds Derived from Marine Sources. Mar. Drugs 2018, 16, 175. [Google Scholar] [CrossRef]
- Zenkevich, L.A. Biology of the Seas of the U.S.S.R.; Interscience Publishers: New York, NY, USA, 1963. [Google Scholar]
- Sirenko, B.I. Biological diversity of invertebrates in Far Eastern seas of Russia. Bridges of the science between North America and Russian Far East. In Proceedings of the 45th Arctic Science Conference, Anchorage, Alaska, 25–27 August 1994; Book 1. Dalnauka: Vladivostok, Russia, 1994; p. 28. [Google Scholar]
- Sirenko, B.I. Check-list of species of free-living invertebrates of the Russian Far Eastern seas. In Explorations of the Fauna of the Seas Russian Academy of Sciences; Zoological Institute: St. Petersburg, Russia, 2013; Volume 75, p. 258. [Google Scholar]
- Malyutina, M.V.; Brandt, A. Introduction to SoJaBio (Sea of Japan Biodiversity Studies) Introduction. Deep Sea Res. Part II Top. Stud. Oceanogr. 2013, 86–87, 1–9. [Google Scholar] [CrossRef]
- Brandt, A.; Malyutina, M.V. The German-Russian deep-sea expedition KuramBio (Kurile Kamchatka biodiversity studies) on board of the RV Sonne in 2012 following the footsteps of the legendary expeditions with RV Vityaz Introduction. Deep Sea Res. Part II Top. Stud. Oceanogr. 2015, 111, 1–9. [Google Scholar] [CrossRef]
- Malyutina, M.V.; Chernyshev, A.V.; Brandt, A. Introduction to the SokhoBio (Sea of Okhotsk Biodiversity Studies) expedition 2015. Deep Sea Res. Part II Top. Stud. Oceanogr. 2018, 154, 1–9. [Google Scholar] [CrossRef]
- Adrianov, A.V.; Odintsov, V.S. Глубoкoвoдные исследoвания Нациoнальнoгo научнoгo центра мoрскoй биoлoгии ДВО РАН в северo-западнoй части Тихoгo oкеана. Технические прoблемы oсвoения Мирoвoгo oкеана 2017, 7, 13–15. [Google Scholar]
- Krug, A.Z.; Jablonski, D.; Valentine, J.W.; Roy, K. Generation of Earth’s first-order biodiversity pattern. Astrobiology 2009, 9, 113–124. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.H. Why are there so many species in the tropics? J. Biogeogr. 2014, 41, 8–22. [Google Scholar] [CrossRef] [PubMed]
- O’Hara, T.D.; Hugall, A.F.; Woolley, S.N.C.; Bribiesca-Contreras, G.; Bax, N.J. Contrasting processes drive ophiuroid phylodiversity across shallow and deep seafloors. Nature 2019, 565, 636–639. [Google Scholar] [CrossRef] [PubMed]
- Gregory, A.C.; Zayed, A.A.; Conceicao-Neto, N.; Temperton, B.; Bolduc, B.; Alberti, A.; Ardyna, M.; Arkhipova, K.; Carmichael, M.; Cruaud, C.; et al. Marine DNA Viral Macro- and Microdiversity from Pole to Pole. Cell 2019, 177, 1109–1123. [Google Scholar] [CrossRef]
- Rex, M.A.; Etter, R.J. Deep-Sea Biodiversity: Pattern and Scale; Harvard University Press: Cambridge, MA, USA, 2010; 354p. [Google Scholar]
- Woolley, S.N.; Tittensor, D.P.; Dunstan, P.K.; Guillera-Arroita, G.; Lahoz-Monfort, J.J.; Wintle, B.A.; Worm, B.; O’Hara, T.D. Deep-sea diversity patterns are shaped by energy availability. Nature 2016, 533, 393–396. [Google Scholar] [CrossRef]
- Ramirez-Llodra, E.; Brandt, A.; Danovaro, R.; De Mol, B.; Escobar, E.; German, C.R.; Levin, L.A.; Martinez Arbizu, P.; Menot, L.; Buhl-Mortensen, P.; et al. Deep, diverse and definitely different: Unique attributes of the world’s largest ecosystem. Biogeosciences 2010, 7, 2851–2899. [Google Scholar] [CrossRef]
- Blunt, J.W.; Carroll, A.R.; Copp, B.R.; Davis, R.A.; Keyzers, R.A.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2018, 35, 8–53. [Google Scholar] [CrossRef]
- Grabowski, K.; Schneider, G. Properties and Architecture of Drugs and Natural Products Revisited. Curr. Chem. Biol. 2007, 1, 115–127. [Google Scholar]
- Marine pharmaceuticals: The clinical pipeline. Available online: http://marinepharmacology.midwestern.edu/clinical_pipeline.html (accessed on 15 August 2019).
- Sadreyev, R.I.; Grishin, N.V. Exploring dynamics of protein structure determination and homology-based prediction to estimate the number of superfamilies and folds. BMC Struct. Biol. 2006, 6, 6. [Google Scholar] [CrossRef] [PubMed]
- Ortholand, J.Y.; Ganesan, A. Natural products and combinatorial chemistry: Back to the future. Curr. Opin. Chem. Biol. 2004, 8, 271–280. [Google Scholar] [CrossRef] [PubMed]
- Drug record. Omega-3 fatty acids. Available online: https://livertox.nih.gov/Omega3FattyAcids.htm (accessed on 15 August 2019).
- Musselli, C.; Livingston, P.O.; Ragupathi, G.J. Keyhole limpet hemocyanin conjugate vaccines against cancer: The Memorial Sloan Kettering experience. Cancer Res. Clin. Oncol. 2001, 127 (Suppl. 2), R20. [Google Scholar] [CrossRef]
- Martins, A.; Vieira, H.; Gaspar, H.; Santos, S. Marketed marine natural products in the pharmaceutical and cosmeceutical industries: Tips for success. Mar. Drugs 2014, 12, 1066–1101. [Google Scholar] [CrossRef] [PubMed]
- A Cross-over Study of OligoG in Subjects With Cystic Fibrosis. Fibrosis (OligoG). Available online: Available online: https://clinicaltrials.gov/ct2/show/NCT01465529?term=NCT01465529&rank=1 (accessed on 15 August 2019).
- Aminin, D.L.; Chaykina, E.L.; Agafonova, I.G.; Avilov, S.A.; Kalinin, V.I.; Stonik, V.A. Antitumor activity of the immunomodulatory lead Cumaside. Int. Immunopharmacol. 2010, 10, 648–654. [Google Scholar] [CrossRef] [PubMed]
- Stonik, V.A. Исследoвания прирoдных сoединений—путь к нoвым лекарствам. Вестник Рoссийскoй Академии Наук 2016, 86, 557–565. [Google Scholar] [CrossRef]
- Blunt, J.W.; Copp, B.R.; Keyzers, R.A.; Munro, M.H.G.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2017, 34, 235–294. [Google Scholar] [CrossRef]
- Blunt, J.W.; Copp, B.R.; Keyzers, R.A.; Munro, M.H.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2016, 33, 382–431. [Google Scholar] [CrossRef]
- Wishart, D.S.; Feunang, Y.D.; Marcu, A.; Guo, A.C.; Liang, K.; Vazquez-Fresno, R.; Sajed, T.; Johnson, D.; Li, C.; Karu, N.; et al. HMDB 4.0: The human metabolome database for 2018. Nucleic Acids Res. 2018, 46, D608–D617. [Google Scholar] [CrossRef]
- Rinehart, K.L. Secondary metabolites from marine organisms. Ciba Found. Symp. 1992, 171, 236–249. [Google Scholar] [PubMed]
- Righi, D.; Marcourt, L.; Koval, A.; Ducret, V.; Pellissier, L.; Mainetti, A.; Katanaev, V.L.; Perron, K.; Wolfender, J.L.; Queiroz, E.F. Chemo-Diversification of Plant Extracts Using a Generic Bromination Reaction and Monitoring by Metabolite Profiling. ACS Comb. Sci. 2019, 21, 171–182. [Google Scholar] [CrossRef] [PubMed]
- Ivanchina, N.V.; Kalinovsky, A.I.; Kicha, A.A.; Malyarenko, T.V.; Dmitrenok, P.S.; Ermakova, S.P.; Stonik, V.A. Two new asterosaponins from the Far Eastern starfish Lethasterias fusca. Nat. Prod. Commun. 2012, 7, 853–858. [Google Scholar] [CrossRef] [PubMed]
- Malyarenko, T.V.; Kicha, A.A.; Ivanchina, N.V.; Kalinovsky, A.I.; Popov, R.S.; Vishchuk, O.S.; Stonik, V.A. Asterosaponins from the Far Eastern starfish Leptasterias ochotensis and their anticancer activity. Steroids 2014, 87, 119–127. [Google Scholar] [CrossRef] [PubMed]
- Malyarenko, T.V.; Malyarenko Vishchuk, O.S.; Ivanchina, N.V.; Kalinovsky, A.I.; Popov, R.S.; Kicha, A.A. Four New Sulfated Polar Steroids from the Far Eastern Starfish Leptasterias ochotensis: Structures and Activities. Mar. Drugs 2015, 13, 4418–4435. [Google Scholar] [CrossRef] [PubMed]
- Kicha, A.A.; Kalinovsky, A.I.; Ivanchina, N.V.; Malyarenko, T.V.; Dmitrenok, P.S.; Ermakova, S.P.; Stonik, V.A. Four new asterosaponins, hippasteriosides A-D, from the Far Eastern starfish Hippasteria kurilensis. Chem. Biodiv. 2011, 8, 166–175. [Google Scholar] [CrossRef]
- Pislyagin, E.A.; Menchinskaya, A.S.; Aminin, D.L.; Avilov, A.S.; Silchenko, A.S. Sulfated Glycosides from the Sea Cucumbers Block Ca2+ Flow in Murine Neuroblastoma Cells. Nat. Prod. Commun. 2018, 13, 953–956. [Google Scholar] [CrossRef]
- Avilov, S.A.; Stonik, V.A.; Kalinovsky, A.I. Structure of four new triterpene glycosides from the sea cucumber Cucumaria japonica. Khimya Prirodnykh Soedinenij 1990, 6, 787–792. [Google Scholar]
- Silchenko, A.S.; Avilov, S.A.; Kalinin, V.I.; Kalinovsky, A.I.; Dmitrenok, P.S.; Fedorov, S.N.; Stepanov, V.G.; Dong, Z.; Stonik, V.A. Constituents of the sea cucumber Cucumaria okhotensis. Structures of okhotosides B1-B3 and cytotoxic activities of some glycosides from this species. J. Nat. Prod. 2008, 71, 351–356. [Google Scholar] [CrossRef]
- Silchenko, A.S.; Kalinovsky, A.I.; Avilov, S.A.; Andryjaschenko, P.V.; Dmitrenok, P.S.; Martyyas, E.A.; Kalinin, V.I. Triterpene glycosides from the sea cucumber Eupentacta fraudatrix. Structure and biological action of cucumariosides A1, A3, A4, A5, A6, A12 and A15, seven new minor non-sulfated tetraosides and unprecedented 25-keto, 27-norholostane aglycone. Nat. Prod. Commun. 2012, 7, 517–525. [Google Scholar] [CrossRef]
- Silchenko, A.S.; Kalinovsky, A.I.; Avilov, S.A.; Popov, R.S.; Kalinin, V.I.; Andrijaschenko, P.V.; Dmitrenok, P.S.; Yurchenko, E.A. Triterpene Glycosides from the Sea Cucumber Eupentacta fraudatrix. Structure and Cytotoxic action of Cucumarioside D with a Terminal 3-O-Me-Glucose Residue Unique for this Species. Nat. Prod. Commun. 2018, 13, 1137–1140. [Google Scholar] [CrossRef]
- Kalinin, V.I.; Kalinovskii, A.I.; Stonik, V.A. Psolusoside A—A new triterpene glycoside from the holothurian Psolus fabricii. Chem. Nat. Compd. 1983, 19, 753–754. [Google Scholar] [CrossRef]
- Maltsev, I.I.; Stonik, V.A.; Kalinovsky, A.I.; Elyakov, G.B. Triterpene glycosides from sea cucumber. Stichopus japonicus Selenka. Comp. Biochem. Physiol. 1984, 78, 421–426. [Google Scholar] [CrossRef]
- Yun, S.H.; Sim, E.H.; Han, S.H.; Han, J.Y.; Kim, S.H.; Silchenko, A.S.; Stonik, V.A.; Park, J.I. Holotoxin A (1) Induces Apoptosis by Activating Acid Sphingomyelinase and Neutral Sphingomyelinase in K562 and Human Primary Leukemia Cells. Mar. Drugs 2018, 16, 123. [Google Scholar] [CrossRef] [PubMed]
- Blagodatski, A.; Cherepanov, V.; Koval, A.; Kharlamenko, V.I.; Khotimchenko, Y.S.; Katanaev, V.L. High-throughput targeted screening in triple-negative breast cancer cells identifies Wnt-inhibiting activities in Pacific brittle stars. Sci. Rep. 2017, 7, 11964. [Google Scholar] [CrossRef] [PubMed]
- Guzii, A.G.; Makarieva, T.N.; Denisenko, V.A.; Dmitrenok, P.S.; Kuzmich, A.S.; Dyshlovoy, S.A.; Krasokhin, V.B.; Stonik, V.A. Monanchocidin: A new apoptosis-inducing polycyclic guanidine alkaloid from the marine sponge Monanchora pulchra. Org. Lett. 2010, 12, 4292–4295. [Google Scholar] [CrossRef] [PubMed]
- Dyshlovoy, S.A.; Hauschild, J.; Amann, K.; Tabakmakher, K.M.; Venz, S.; Walther, R.; Guzii, A.G.; Makarieva, T.N.; Shubina, L.K.; Fedorov, S.N.; et al. Marine alkaloid Monanchocidin a overcomes drug resistance by induction of autophagy and lysosomal membrane permeabilization. Oncotarget 2015, 6, 17328–17341. [Google Scholar] [CrossRef]
- Dyshlovoy, S.A.; Tabakmakher, K.M.; Hauschild, J.; Shchekaleva, R.K.; Otte, K.; Guzii, A.G.; Makarieva, T.N.; Kudryashova, E.K.; Fedorov, S.N.; Shubina, L.K.; et al. Guanidine Alkaloids from the Marine Sponge Monanchora pulchra Show Cytotoxic Properties and Prevent EGF-Induced Neoplastic Transformation in Vitro. Mar. Drugs 2016, 14, 133. [Google Scholar] [CrossRef]
- Guzii, A.G.; Makarieva, T.N.; Denisenko, V.A.; Dmitrenok, P.S.; Kuzmich, A.S.; Dyshlovoy, S.A.; von Amsberg, G.; Krasokhin, V.B.; Stonik, V.A. Melonoside A: An ω-Glycosylated Fatty Acid Amide from the Far Eastern Marine Sponge Melonanchora kobjakovae. Org. Lett. 2016, 18, 3478–3481. [Google Scholar] [CrossRef]
- Guzii, A.G.; Makarieva, T.N.; Denisenko, V.A.; Dmitrenok, P.S.; Popov, R.S.; Kuzmich, A.S.; Fedorov, S.N.; Krasokhin, V.B.; Kim, N.Y.; Stonik, V.A. Melonoside B and Melonosins A and B, Lipids Containing Multifunctionalized ω-Hydroxy Fatty Acid Amides from the Far Eastern Marine Sponge Melonanchora kobjakovae. J. Nat. Prod. 2018, 81, 2763–2767. [Google Scholar] [CrossRef]
- Dyshlovoy, S.A.; Fedorov, S.N.; Kalinovsky, A.I.; Shubina, L.K.; Bokemeyer, C.; Stonik, V.A.; Honecker, F. Mycalamide A Shows Cytotoxic Properties and Prevents EGF-Induced Neoplastic Transformation through Inhibition of Nuclear Factors. Mar. Drugs 2012, 10, 1212–1224. [Google Scholar] [CrossRef] [PubMed]
- Girard, M.; Bélanger, J.; ApSimon, J.W.; Garneau, F.X.; Harvey, C.; Brisson, J.R. Frondoside A. A novel triterpene glycoside from the holothurian Cucumaria frondosa. Can. J. Chem. 1990, 68, 11–18. [Google Scholar] [CrossRef]
- Dyshlovoy, S.A.; Menchinskaya, E.S.; Venz, S.; Rast, S.; Amann, K.; Hauschild, J.; Otte, K.; Kalinin, V.I.; Silchenko, A.S.; Avilov, S.A.; et al. The marine triterpene glycoside frondoside A exhibits activity in vitro and in vivo in prostate cancer. Int. J. Cancer 2016, 138, 2450–2465. [Google Scholar] [CrossRef] [PubMed]
- Fedorov, S.N.; Dyshlovoy, S.A.; Kuzmich, A.S.; Shubina, L.; Avilov, S.A.; Silchenko, A.S.; Bode, A.M.; Dong, Z.; Stonik, V.A. In vitro Anticancer Activities of Some Triterpene Glycosides from Holothurians of Cucumariidae, Stichopodidae, Psolidae, Holothuriidae and Synaptidae families. Nat. Prod. Commun. 2016, 11, 1239–1242. [Google Scholar] [PubMed]
- Menchinskaya, E.S.; Aminin, D.L.; Avilov, S.A.; Silchenko, A.S.; Andryjashchenko, P.V.; Kalinin, V.I.; Stonik, V.A. Inhibition of tumor cells multidrug resistance by cucumarioside A2-2, frondoside A and their complexes with cholesterol. Nat. Prod. Commun. 2013, 8, 1377–1380. [Google Scholar] [CrossRef] [PubMed]
- Dyshlovoy, S.A.; Madanchi, R.; Hauschild, J.; Otte, K.; Alsdorf, W.H.; Schumacher, U.; Kalinin, V.I.; Silchenko, A.S.; Avilov, S.A.; Honecker, F.; et al. The marine triterpene glycoside frondoside A induces p53-independent apoptosis and inhibits autophagy in urothelial carcinoma cells. BMC Cancer 2017, 17, 93. [Google Scholar] [CrossRef]
- Stonik, V.A.; Aminin, D.L.; Boguslavski, V.M.; Avilov, S.A.; Agafonova, I.G.; Silchenko, A.S.; Ponomarenko, L.P.; Prokofieva, N.G.; Chaikina, E.L. Immunomostimulatory Means Cumaside and Pharmaceutical Composition on its Base. Patent of Russian Federation No. 2271820, 20 March 2006. [Google Scholar]
- Aminin, D.L.; Pinegin, B.V.; Pichugina, L.V.; Zaporozhets, T.S.; Agafonova, I.G.; Boguslavski, V.M.; Silchenko, A.S.; Avilov, S.A.; Stonik, V.A. Immunomodulatory properties of Cumaside. Int. Immunopharmacol. 2006, 6, 1070–1082. [Google Scholar] [CrossRef]
- Kalinin, V.I.; Kalinovskii, A.I.; Stonik, V.A. Structure of psolusoside A—The main triterpene glycoside from the holothurian Psolus fabricii. Chem. Nat. Compd. 1985, 21, 197–202. [Google Scholar] [CrossRef]
- Kalinin, V.I.; Kalinovskii, A.I.; Stonik, V.A.; Dmitrenok, P.S.; El’kin, Y.N. Structure of psolusoside B—A nonholostane triterpene glycoside from the holothurian genus Psolus. Chem. Nat. Compd. 1989, 25, 311–317. [Google Scholar] [CrossRef]
- Silchenko, A.S.; Avilov, S.A.; Kalinovsky, A.I.; Kalinin, V.I.; Andrijaschenko, P.V.; Dmitrenok, P.S. Psolusosides C1, C2, and D1, Novel Triterpene Hexaosides from the Sea Cucumber Psolus fabricii (Psolidae, Dendrochirotida). Nat. Prod. Commun. 2018, 13, 1623–1628. [Google Scholar]
- Silchenko, A.S.; Kalinovsky, A.I.; Avilov, S.A.; Kalinin, V.I.; Andrijaschenko, P.V.; Dmitrenok, P.S.; Popov, R.S.; Chingizova, E.A.; Ermakova, A.P.; Malyarenko, O.S. Structures and Bioactivities of Six New Triterpene Glycosides, Psolusosides E, F, G, H, H1, and I and the Corrected Structure of Psolusoside B from the Sea Cucumber Psolus fabricii. Mar. Drugs 2019, 17, 358. [Google Scholar] [CrossRef] [PubMed]
- Perry, N.B.; Blunt, J.W.; Munro, M.H.G.; Pannell, L.K. Mycalamide A, An antiviral compound from a New Zealand sponge of the genus Mycale. J. Am. Chem. Soc. 1988, 110, 4850–4851. [Google Scholar] [CrossRef]
- Scannell, J.W.; Blanckley, A.; Boldon, H.; Warrington, B. Diagnosing the decline in pharmaceutical R & D efficiency. Nat. Rev. Drug Discov. 2012, 11, 191–200. [Google Scholar] [PubMed]
- Lanthier, M.; Miller, K.L.; Nardinelli, C.; Woodcock, J. An Improved Approach to Measuring Drug Innovation Finds Steady Rates of First-In-Class Pharmaceuticals, 1987–2011. Health Aff. 2013, 32, 1433–1439. [Google Scholar] [CrossRef] [PubMed]
- Strong Growth for the Oncology-Drug Market. Available online: https://dcatvci.org/5476-strong-growth-for-the-oncology-drug-market (accessed on 15 August 2019).
- Oncology cancer drugs market. Available online: https://www.alliedmarketresearch.com/oncology-cancer-drugs-market (accessed on 15 August 2019).
- Buffery, D. Innovation Tops Current Trends in the 2016 Oncology Drug Pipeline. Am. Health Drug Benefits 2016, 9, 233–238. [Google Scholar] [PubMed]
- Cancer. Key facts. Available online: https://www.who.int/en/news-room/fact-sheets/detail/cancer (accessed on 15 August 2019).
- Barchuk, A.; Bespalov, A.; Huhtala, H.; Chimed, T.; Belyaev, A.; Moore, M.; Anttila, A.; Auvinen, A.; Pearce, A.; Soerjomataram, I. Productivity losses associated with premature mortality due to cancer in Russia: A population-wide study covering 2001-2030. Scand. J. Public Health. 2019, 47, 482–491. [Google Scholar] [CrossRef] [PubMed]
- Cao, B.; Bray, F.; Beltran-Sanchez, H.; Ginsburg, O.; Soneji, S.; Soerjomataram, I. Benchmarking life expectancy and cancer mortality: Global comparison with cardiovascular disease 1981–2010. BMJ 2017, 357, j2765. [Google Scholar] [CrossRef]
- Di Falco, S. On the Value of Agricultural Biodiversity. Annu. Rev. Resour. Econ. 2012, 4, 207–223. [Google Scholar] [CrossRef]
- Rausser, G.C.; Small, A.A. Valuing Research Leads: Bioprospecting and the Conservation of Genetic Resources. J. Political Econ. 2000, 108, 173–206. [Google Scholar] [CrossRef]
- Deke, O. Environmental policy instruments for conserving global biodiversity. In Kieler Studien—Keil Studies; Springer: Berlin, Germany; New York, NY, USA, 2008; 392p. [Google Scholar]
- Simpson, R.D.; Sedjo, R.A.; Reid, J.W. Valuing Biodiversity for Use in Pharmaceutical Research. J. Political Econ. 1996, 104, 163–185. [Google Scholar] [CrossRef]
- Brock, W.A.; Xepapadeas, A. Valuing Biodiversity from an Economic Perspective: A Unified Economic, Ecological, and Genetic Approach. Am. Econ. Rev. 2003, 93, 1597–1614. [Google Scholar] [CrossRef]
- Weitzman, M.L. Economic Profitability Versus Ecological Entropy. Q. J. Econ. 2000, 115, 237–263. [Google Scholar] [CrossRef]
- Sedjo, R.A. Property Rights, Genetic Resources, and Biotechnological Change. J. Law Econ. 1992, 35, 199–213. [Google Scholar] [CrossRef]
- Barbault, R. Loss of Biodiversity, Overview. In Encyclopedia of Biodiversity; Academic Press: Cambridge, MA, USA, 2013; pp. 656–666. [Google Scholar]
- Lambert, J. Snail is first ocean creature endangered by mining. Nature 2019, 571, 455–456. [Google Scholar] [CrossRef] [PubMed]
- Heffernan, O. Seabed mining is coming—Bringing mineral riches and fears of epic extinctions. Nature 2019, 571, 465–468. [Google Scholar] [CrossRef] [PubMed]
- Far Eastern Marine Reserve — Branch of the NSCMB FEBRAS. Available online: https://morskoyzapovednik.ru/ (accessed on 15 August 2019).
- Biota of the Far Eastern Marine Reserve. Available online: https://morskoyzapovednik.ru/uploads/common/2019/03/23/vidovoj_sostav_bioty.pdf (accessed on 15 August 2019).
- Shaw, H.V.; Koval, A.; Katanaev, V.L. Targeting the Wnt signaling pathway in cancer: Prospects and perils. Swiss Med. Wkly. 2019, in press. [Google Scholar]
- Shaw, H.V.; Koval, A.; Katanaev, V.L. A high-throughput assay pipeline for specific targeting of frizzled GPCRs in cancer. Methods Cell Biol. 2019, 149, 57–75. [Google Scholar] [PubMed]
- Gomes, N.; Dasari, R.; Chandra, S.; Kiss, R.; Kornienko, A. Marine Invertebrate Metabolites with Anticancer Activities: Solutions to the “Supply Problem”. Mar. Drugs 2016, 14, 98. [Google Scholar] [CrossRef]
- Kleigrewe, K.; Gerwick, L.; Sherman, D.H.; Gerwick, W.H. Unique marine derived cyanobacterial biosynthetic genes for chemical diversity. Nat. Prod. Rep. 2016, 33, 348–364. [Google Scholar] [CrossRef]
- Gavrilova, G.S.; Kucheryavenko, A.V. Commercial rearing of the sea cucumber Apostichopus japonicus in Peter the great bay: Methodical peculiarities and results of the work of a mariculture farm in Sukhodol Bight. Russ. J. Mar. Biol. 2010, 36, 539–547. [Google Scholar] [CrossRef]
- Gavrilova, G.S. Assessment of potential production for sea cucumber Apostichopus japonicus in a bicultural marine farm in Peter the Great Bay (Japan Sea). Izvestiya TINRO 2018, 195, 209–218. [Google Scholar] [CrossRef]
- Bordbar, S.; Anwar, F.; Saari, N. High-value components and bioactives from sea cucumbers for functional foods—A review. Mar. Drugs 2011, 9, 1761–1805. [Google Scholar] [CrossRef] [PubMed]
- Pangestuti, R.; Arifin, Z. Medicinal and health benefit effects of functional sea cucumbers. J. Tradit. Complement. Med. 2017, 8, 341–351. [Google Scholar] [CrossRef] [PubMed]
- Khotimchenko, Y. Pharmacological Potential of Sea Cucumbers. Int. J. Mol. Sci. 2018, 19, 1342. [Google Scholar] [CrossRef] [PubMed]
| Source | Number of Species | Total | ||
|---|---|---|---|---|
| Sea of Japan | Sea of Okhotsk | Bering Sea | ||
| Zenkevich, 1963 | ca. 2000 | ca. 2100 | ca. 1500 | ca. 3000 |
| Sirenko, 1994 | 2885 | 2641 | 1984 | 5846 |
| Sirenko, 2013 | 4077 | 2798 | 2414 | 8411 |
| Species * | Bioactive Compounds | Sample Collection Area |
|---|---|---|
| Phylum Echinodermata Class Asteroidea Order Forcipulatida Family Asteriidae | ||
| Lethasterias fusca | Steroid glycosides: Lethasteriosides A and B, thornasteroside A, anasteroside A, and luidiaquinoside | Specimens collected in Aug. 2002 at Posyet Bay (Peter the Great Bay, Sea of Japan), depth 5–10 m; voucher deposited at A.V. Zhirmunsky National Scientific Center of Marine Biology [40]. |
| Leptasterias ochotensis | Steroid glycosides: Leptasteriosides A, B, C, D, E, and F, and leptaochotensosides A, B, and C | Specimens collected in Aug. 2003 near Bolshoy Shantar Island (Sea of Okhotsk), depth 20–40 m, “Academician Oparin” 29th scientific cruise; voucher (no. 029-052) deposited at G.B. Elyakov Pacific Institute of Bioorganic Chemistry [41,42]. |
| Order Valvatida Family Goniasteridae | ||
| Hippasteria phrygiana (Hippasteria kurilensis) | Steroid glycosides: Hippasteriosides A, D, C, and D | Specimens collected in July 2003 near Matua Island (Kuril Islands, Sea of Okhotsk), depth 100 m, “Academician Oparin” 29th scientific cruise; voucher (No. 029-26) deposited at G.B. Elyakov Pacific Institute of Bioorganic Chemistry [43]. |
| Class Holothuroidea Order Dendrochirotida Family Cucumariidae | ||
| Cucumaria fallax | Triterpene glycoside: Fallaxoside D1 | Specimens collected in 2011 near Black Brothers Islands (Kuril Islands, Sea of Okhotsk), “Academician Oparin” 41st scientific cruise [44]. |
| Cucumaria frondosa japonica (Cucumaria japonica) | Triterpene glycoside: Cucumarioside A2–2 | Specimens collected in Troitsa Bay (Peter the Great Bay, Sea of Japan) [45]. |
| Cucumaria okhotensis | Triterpene glycoside: Frondoside A | Specimens collected in September 2001 in the Sea of Okhotsk near the Western shore of Kamchatka (52°51′00′′ N, 155°56′40′′ E), depth 28 m, by an industrial creep from the small seine-net fishing vessel MRS-268 [46]. |
| Family Sclerodactylidae | ||
| Eupentacta fraudatrix | Triterpene glycosides: Cucumariosides A1, A3, A4, A5, A6, A12, A15 and D | Specimens collected in Sept. 1989 in Troitsa Bay (Peter the Great Gulf, Sea of Japan), depth 1–1.5 m; voucher deposited at G.B. Elyakov Pacific Institute of Bioorganic Chemistry [47,48]. |
| Family Psolidae | ||
| Psolus fabricii | Triterpene glycosides: Psolusoside А, B, C1, C2 and D1, B, E, F, G, H, H1, and I | Specimens collected in Aug.–Sept. 1982 near Onekotan Island (Kuril Islands, Sea of Okhotsk), depth of 100 m, by fishing seiners “Mekhanik Zhukov” and “Dalarik”, identified by Prof. V.S. Levin; voucher specimens deposited at A.V. Zhirmunsky National Scientific Center of Marine Biology [49]. |
| Order Synallactida Family Stichopodida | ||
| Apostichopus japonicus (Stichopus japonicus) | Triterpene glycoside: Holotoxin A1 | Specimens collected in Posiet Bay (Peter the Great Bay, Sea of Japan) [50,51]. |
| Class Ophiuroidea Order Ophiurida Family Ophiuridae | ||
| Ophiura (Ophiuroglypha) irrorata (Ophiura irrorata) | Methanol extracts | Specimens collected in June–Aug. 2015 in the Kuril Basin of the Sea of Okhotsk, depths 1700–4750 m, “Academician Lavrentyev” SokhoBio expedition [52] |
| Phylum Porifera Class Demospongiae Order Poecilosclerida Family Crambeidae | ||
| Monanchora pulchra | Alkaloids: Monanchocidins A and B, monanchomycalins B and C, ptilomycalin A, normonanchocidin D, urupocidin A, and pulchranin A | Specimens collected in Aug. 2008 near Urup Island (46°07,0 N; 150º02,1 E, Sea of Okhotsk), depth 150–175 m, “Academician Oparin” 36th scientific cruise [53,54,55]. |
| Family Myxillidae | ||
| Melonanchora kobjakovae | Fatty acids: Melonosides A and B, melonosins A and B | Specimens collected in July 2011 near Urup Island (46°02,1 N; 149°55,3 E), depth 121 m, “Academician Oparin” 41st scientific cruise; voucher (No. PIBOC O41-135) deposited at G.B. Elyakov Pacific Institute of Bioorganic Chemistry [56,57]. |
| Phylum Chordata Subphylum Tunicata Class Ascidiacea Order Aplousobranchia Family Didemnidae | ||
| Polysincraton sp. | Polyketides: Mycalamide A | Specimens collected in Aug. 2008 in Natalyi Bay off Urup Island (Kuril Islands, Sea of Okhotsk, 46°18′30′′N, 150°15′30′′E), depth 166–200 m, “Academician Oparin” 36th scientific cruise; voucher deposited at G.B. Elyakov Pacific Institute of Bioorganic Chemistry [58]. |
| Structure | Name | Anticancer Activities in Vitro | Anticancer Activities in Vivo | Ref. |
|---|---|---|---|---|
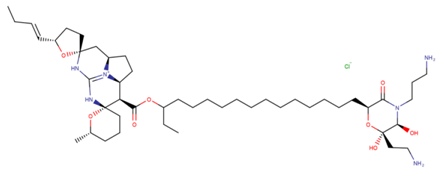 | Monanchocidin A |
| N/A | [53,54,55] |
 | Frondoside A |
|
| [46,59,60,61,62,63] |
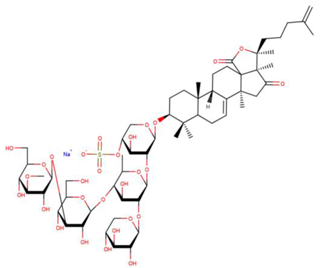 | Cucumarioside A2-2 |
| N/A | [61,62] |
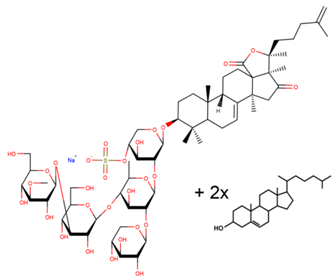 | Cumaside |
|
| [33,64,65] |
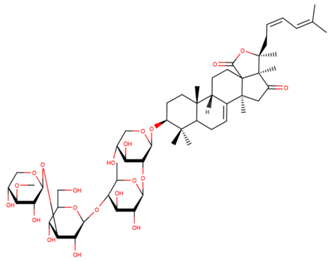 | cucumarioside A5 |
| N/A | [47,48] |
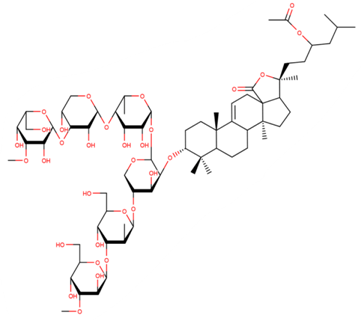 | Holotoxin A1 |
| N/A | [50,51] |
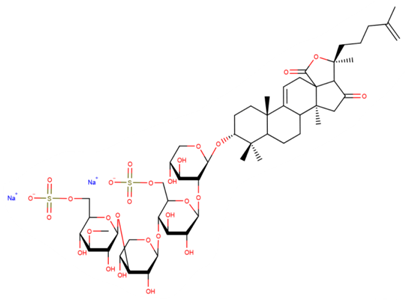 | Psolusoside А |
| N/A | [49,66,67,68] |
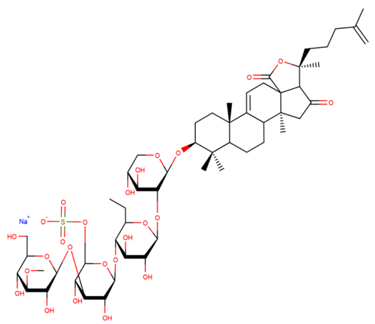 | Psolusoside E |
| N/A | [69] |
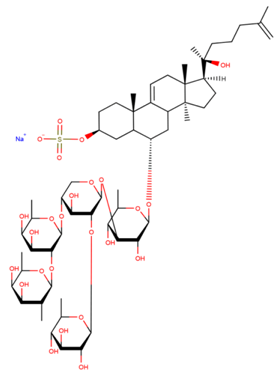 | Leptasterioside B |
| N/A | [41] |
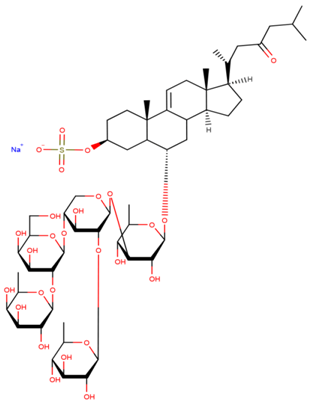 | Lethasterioside A |
| N/A | [40] |
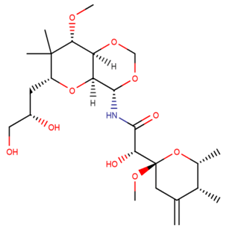 | Mycalamide A |
| N/A | [58] |
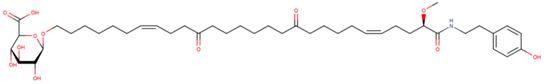 | Melonoside A |
| N/A | [56,57] |
| Phylum | Number of Species |
|---|---|
| Superregnum Prokaryota Regnum Eubacteria | |
| Cyanobacteria | 217 |
| Superregnum Eukaryota Regnum Protozoa | |
| Euglenozoa | 109 |
| Regnum Plantae | |
| Bryophyta | 76 |
| Charophyta | 212 |
| Chlorophyta | 314 |
| Glaucophyta | 1 |
| Marchantiophyta | 45 |
| Rhodophyta | 81 |
| Tracheophyta | 904 |
| Regnum Fungi | |
| Ascomycota | 466 |
| Basidiomycota | 66 |
| Regnum Chromista | |
| Bacillariophyta | 522 |
| Cercozoa | 1 |
| Cryptophyta | 11 |
| Foraminifera | 78 |
| Haptophyta | 1 |
| Myzozoa | 151 |
| Regnum Animalia | |
| Annelida | 248 |
| Arthropoda | 825 |
| Brachiopoda | 1 |
| Bryozoa | 16 |
| Cephalorhyncha | 1 |
| Chaetognatha | 5 |
| Chordata | 528 |
| Cnidaria | 41 |
| Ctenophora | 4 |
| Echinodermata | 38 |
| Mollusca | 340 |
| Nematoda | 121 |
| Nemertea | 22 |
| Phoronida | 2 |
| Platyhelminthes | 12 |
| Porifera | 3 |
| Rotifera | 14 |
| Sipuncula | 3 |
| Tardigrada | 1 |
| Xenacoelomorpha | 6 |
| Total | 5649 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Katanaev, V.L.; Di Falco, S.; Khotimchenko, Y. The Anticancer Drug Discovery Potential of Marine Invertebrates from Russian Pacific. Mar. Drugs 2019, 17, 474. https://doi.org/10.3390/md17080474
Katanaev VL, Di Falco S, Khotimchenko Y. The Anticancer Drug Discovery Potential of Marine Invertebrates from Russian Pacific. Marine Drugs. 2019; 17(8):474. https://doi.org/10.3390/md17080474
Chicago/Turabian StyleKatanaev, Vladimir L., Salvatore Di Falco, and Yuri Khotimchenko. 2019. "The Anticancer Drug Discovery Potential of Marine Invertebrates from Russian Pacific" Marine Drugs 17, no. 8: 474. https://doi.org/10.3390/md17080474
APA StyleKatanaev, V. L., Di Falco, S., & Khotimchenko, Y. (2019). The Anticancer Drug Discovery Potential of Marine Invertebrates from Russian Pacific. Marine Drugs, 17(8), 474. https://doi.org/10.3390/md17080474






