Widespread Existence of Quorum Sensing Inhibitors in Marine Bacteria: Potential Drugs to Combat Pathogens with Novel Strategies
Abstract
1. Introduction
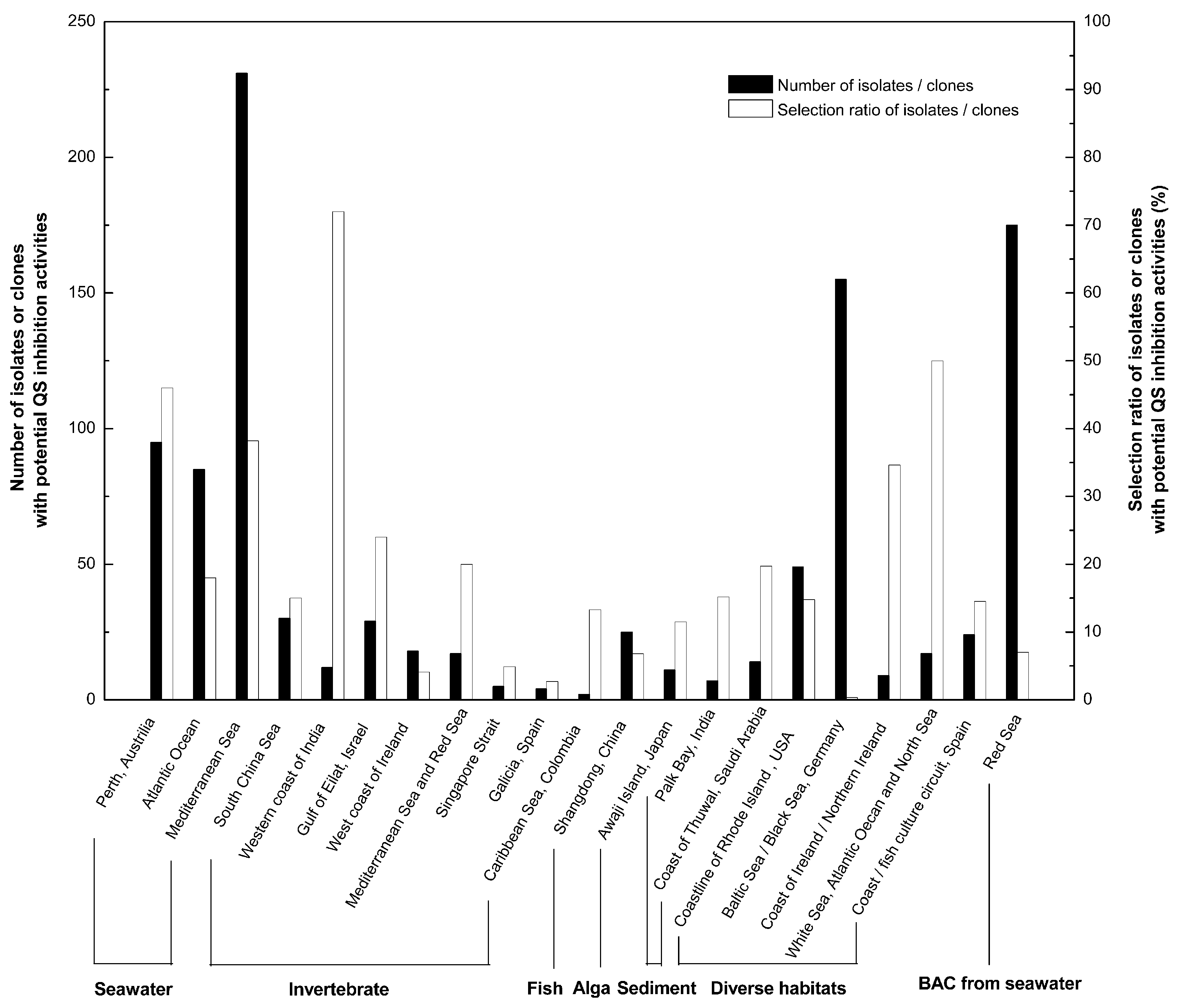
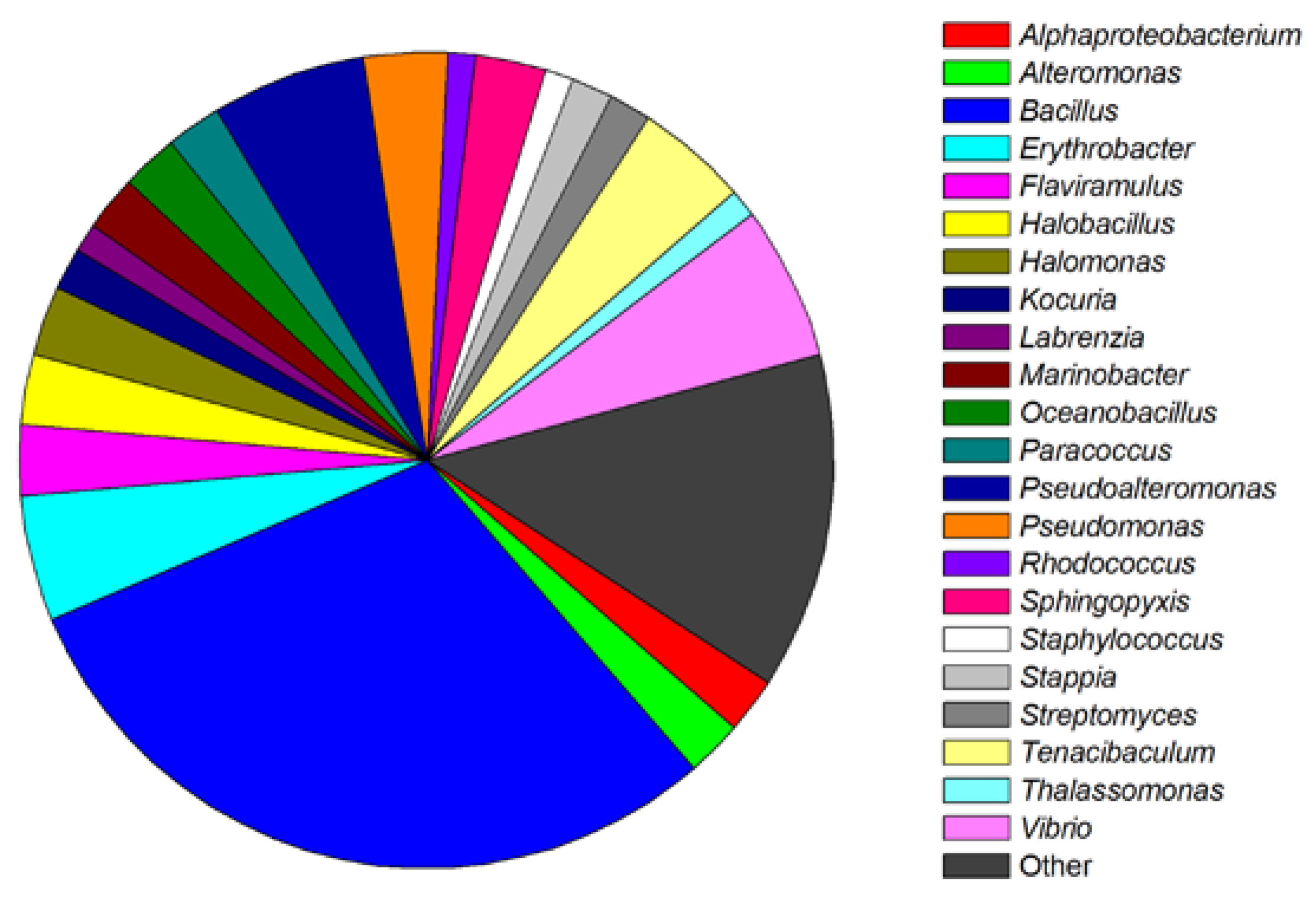
2. Screening and Classification of Marine Bacteria with QS Inhibition Capabilities
3. Small Molecule QSIs Derived from Marine Bacteria
3.1. Natural Small Molecule QSIs
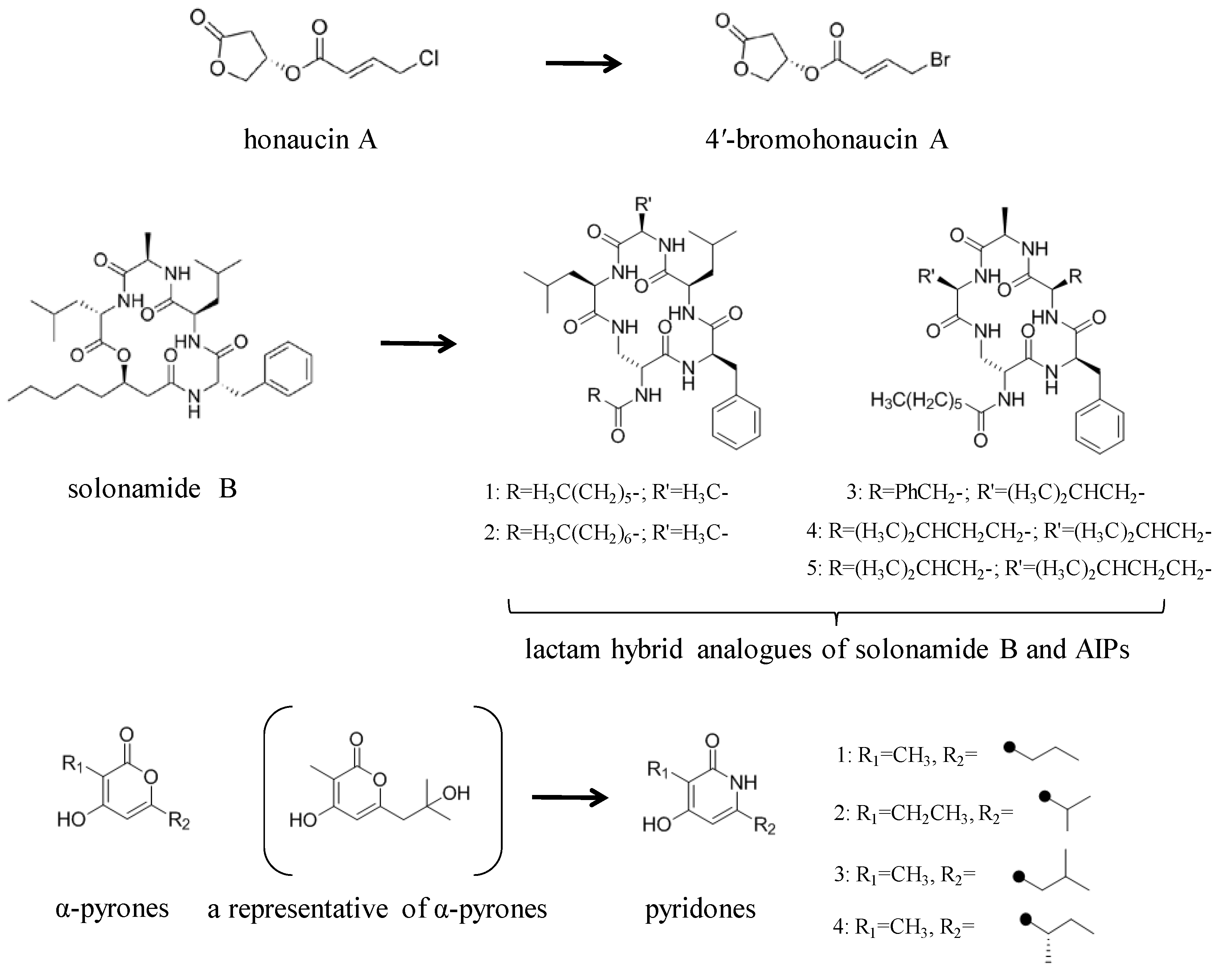
3.2. Structural Modification of Natural Small Molecule QSIs
4. Quorum Quenching Enzymes Derived from Marine Bacteria
5. Potential Application Perspectives of QSIs or QSI-Producing Bacteria
6. Concluding Remarks
Funding
Conflicts of Interest
References
- Miller, M.B.; Bassler, B.L. Quorum sensing in bacteria. Annu. Rev. Microbiol. 2001, 55, 165–199. [Google Scholar] [CrossRef]
- Jayaraman, A.; Wood, T.K. Bacterial quorum sensing: Signals, circuits, and implications for biofilms and disease. Annu. Rev. Biomed. Eng. 2008, 10, 145–167. [Google Scholar] [CrossRef]
- Boyer, M.; Wisniewski-Dye, F. Cell-cell signalling in bacteria: Not simply a matter of quorum. FEMS Microbiol. Ecol. 2009, 70, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Di Cagno, R.; De Angelis, M.; Calasso, M.; Gobbetti, M. Proteomics of the bacterial cross-talk by quorum sensing. J. Proteomics 2011, 74, 19–34. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Chen, M.; Quan, C.; Fan, S. Mechanisms of quorum sensing and strategies for quorum sensing disruption in aquaculture pathogens. J. Fish Dis. 2014, 38, 771–786. [Google Scholar] [CrossRef]
- Turkina, M.V.; Vikström, E. Bacteria-host crosstalk: Sensing of the quorum in the context of Pseudomonas aeruginosa Infections. J. Innate Immun. 2018, 11, 1–17. [Google Scholar] [CrossRef]
- Le, K.Y.; Otto, M. Quorum-sensing regulation in staphylococci-an overview. Front. Microbiol. 2015, 6, 1174. [Google Scholar] [CrossRef]
- Kalia, V.C.; Purohit, H.J. Quenching the quorum sensing system: Potential antibacterial drug targets. Crit. Rev. Microbiol. 2011, 37, 121–140. [Google Scholar] [CrossRef]
- Chen, F.; Gao, Y.; Chen, X.; Yu, Z.; Li, X. Quorum quenching enzymes and their application in degrading signal molecules to block quorum sensing-dependent infection. Int. J. Mol. Sci. 2013, 14, 17477–17500. [Google Scholar] [CrossRef]
- Defoirdt, T.; Boon, N.; Bossier, P.; Verstraete, W. Disruption of bacterial quorum sensing: An unexplored strategy to fight infections in aquaculture. Aquaculture 2004, 240, 69–88. [Google Scholar] [CrossRef]
- Lade, H.; Paul, D.; Kweon, J.H. Quorum quenching mediated approaches for control of membrane biofouling. Int. J. Biol. Sci. 2014, 10, 547–562. [Google Scholar] [CrossRef] [PubMed]
- Kalia, V.C. Quorum sensing inhibitors: An overview. Biotechnol. Adv. 2013, 31, 224–245. [Google Scholar] [CrossRef]
- Defoirdt, T.; Brackman, G.; Coenye, T. Quorum sensing inhibitors: How strong is the evidence? Trends Microbiol. 2013, 21, 619–624. [Google Scholar] [CrossRef]
- Dong, Y.H.; Zhang, L.H. Quorum sensing and quorum-quenching enzymes. J. Microbiol. 2005, 43, 101–109. [Google Scholar]
- Czajkowski, R.; Jafra, S. Quenching of acyl-homoserine lactone-dependent quorum sensing by enzymatic disruption of signal molecules. Acta Biochimica Polonica 2009, 56, 1–16. [Google Scholar] [CrossRef]
- Fetzner, S. Quorum quenching enzymes. J. Biotechnol. 2015, 201, 2–14. [Google Scholar] [CrossRef]
- Tang, K.; Zhang, X.H. Quorum quenching agents: resources for antivirulence therapy. Mar. Drugs 2014, 12, 3245–3282. [Google Scholar] [CrossRef] [PubMed]
- Delago, A.; Mandabi, A.; Meijler, M.M. Natural quorum sensing inhibitors-small molecules, big messages. Isr. J. Chem. 2015, 56, 310–320. [Google Scholar] [CrossRef]
- Ta, C.A.K.; Arnason, J.T. Mini review of phytochemicals and plant taxa with activity as microbial biofilm and quorum sensing Inhibitors. Molecules 2016, 21, 29. [Google Scholar] [CrossRef] [PubMed]
- Das, R.; Mehta, D.K. Microbial biofilm and quorum sensing inhibition: endowment of medicinal plants to combat multidrug-resistant bacteria. Curr. Drug Targets 2018, 19, 1916–1932. [Google Scholar] [CrossRef]
- Skindersoe, M.E.; Ettinger-Epstein, P.; Rasmussen, T.B.; Bjarnsholt, T.; de Nys, R.; Givskov, M. Quorum sensing antagonism from marine organisms. Mar. Biotechnol. 2007, 10, 56–63. [Google Scholar] [CrossRef]
- Natrah, F.M.I.; Defoirdt, T.; Sorgeloos, P.; Bossier, P. Disruption of bacterial cell-to-cell communication by marine organisms and its relevance to aquaculture. Mar. Biotechnol. 2011, 13, 109–126. [Google Scholar] [CrossRef]
- Wietz, M.; Duncan, K.; Patin, N.V.; Jensen, P.R. Antagonistic interactions mediated by marine bacteria: The role of small molecules. J. Chem. Ecol. 2013, 39, 879–891. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Wang, M.; Zhu, X.; Yu, W.; Gong, Q. Equisetin as potential quorum sensing inhibitor of Pseudomonas aeruginosa. Biotechnol. Lett. 2018, 40, 865–870. [Google Scholar] [CrossRef] [PubMed]
- Reen, F.J.; Gutiérrez-Barranquero, J.A.; Dobson, A.D.; Adams, C.; O’Gara, F. Emerging concepts promising new horizons for marine biodiscovery and synthetic biology. Mar. Drugs 2015, 13, 2924–2954. [Google Scholar] [CrossRef]
- Costantino, V.; Della Sala, G.; Saurav, K.; Teta, R.; Bar-Shalom, R.; Mangoni, A.; Steindler, L. Plakofuranolactone as a quorum quenching agent from the Indonesian sponge Plakortis cf. lita. Mar. Drugs 2017, 15, 59. [Google Scholar] [CrossRef] [PubMed]
- Heo, Y.M.; Kim, K.; Ryu, S.M.; Kwon, S.L.; Park, M.Y.; Kang, J.E.; Hong, J.H.; Lim, Y.W.; Kim, C.; Kim, B.S.; et al. Diversity and ecology of marine algicolous Arthrinium species as a source of bioactive natural products. Mar. Drugs 2018, 16, 508. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Wang, B.; Lu, Y.; Guo, Y.; Sun, J.; Wei, B.; Zhang, H.; Wang, H. Quorum sensing inhibitors from marine microorganisms and their synthetic derivatives. Mar. Drugs 2019, 17, 80. [Google Scholar] [CrossRef]
- Romero, M.; Martin-Cuadrado, A.B.; Otero, A. Determination of whether quorum quenching is a common activity in marine bacteria by analysis of cultivable bacteria and metagenomic sequences. Appl. Environ. Microb. 2012, 78, 6345–6348. [Google Scholar] [CrossRef] [PubMed]
- Linthorne, J.S.; Chang, B.J.; Flematti, G.R.; Ghisalberti, E.L.; Sutton, D.C. A direct pre-screen for marine bacteria producing compounds inhibiting quorum sensing reveals diverse planktonic bacteria that are bioactive. Mar. Biotechnol. 2014, 17, 33–42. [Google Scholar] [CrossRef] [PubMed]
- Ma, Z.P.; Song, Y.; Cai, Z.H.; Lin, Z.J.; Lin, G.H.; Wang, Y.; Zhou, J. Anti-quorum sensing activities of selected coral symbiotic bacterial extracts from the South China Sea. Front. Cell Infect. Microbiol. 2018, 8, 144. [Google Scholar] [CrossRef] [PubMed]
- Naik, D.N.; Wahidullah, S.; Meena, R.M. Attenuation of Pseudomonas aeruginosa virulence by marine invertebrate-derived Streptomyces sp. Lett. Appl. Microbiol. 2013, 56, 197–207. [Google Scholar] [CrossRef]
- Tang, K.; Zhang, Y.; Yu, M.; Shi, X.; Coenye, T.; Bossier, P.; Zhang, X.H. Evaluation of a new high-throughput method for identifying quorum quenching bacteria. Sci. Rep. 2013, 3, 2935. [Google Scholar] [CrossRef]
- Golberg, K.; Pavlov, V.; Marks, R.S.; Kushmaro, A. Coral-associated bacteria, quorum sensing disrupters, and the regulation of biofouling. Biofouling 2013, 29, 669–682. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez-Barranquero, J.A.; Reen, F.J.; Parages, M.L.; McCarthy, R.; Dobson, A.D.W.; O’Gara, F. Disruption of N-acyl-homoserine lactone-specific signalling and virulence in clinical pathogens by marine sponge bacteria. Microb. Biotechnol. 2017. [Google Scholar] [CrossRef]
- Yaniv, K.; Golberg, K.; Kramarsky-Winter, E.; Marks, R.; Pushkarev, A.; Béjà, O.; Kushmaro, A. Functional marine metagenomic screening for anti-quorum sensing and anti-biofilm activity. Biofouling 2016, 33, 1–13. [Google Scholar] [CrossRef]
- Teasdale, M.E.; Donovan, K.A.; Forschner-Dancause, S.R.; Rowley, D.C. Gram-positive marine bacteria as a potential resource for the discovery of quorum sensing inhibitors. Mar. Biotechnol. 2011, 13, 722–732. [Google Scholar] [CrossRef]
- Muras, A.; López-Pérez, M.; Mayer, C.; Parga, A.; Amaro-Blanco, J.; Otero, A. High Prevalence of quorum-sensing and quorum-quenching activity among cultivable bacteria and metagenomic sequences in the Mediterranean Sea. Genes 2018, 9, 100. [Google Scholar] [CrossRef]
- Weiland-Bräuer, N.; Kisch, M.J.; Pinnow, N.; Liese, A.; Schmitz, R.A. Highly Effective inhibition of biofilm formation by the first metagenome-derived AI-2 quenching enzyme. Front. Microbiol. 2016, 7, 1098. [Google Scholar] [CrossRef]
- Saurav, K.; Bar-Shalom, R.; Haber, M.; Burgsdorf, I.; Oliviero, G.; Costantino, V.; Morgenstern, D.; Steindler, L. In search of alternative antibiotic drugs: Quorum-quenching activity in sponges and their bacterial isolates. Front. Microbiol. 2016, 7, 416. [Google Scholar] [CrossRef]
- Ong, J.F.M.; Goh, H.C.; Lim, S.C.; Pang, L.M.; Chin, J.S.F.; Tan, K.S.; Liang, Z.X.; Yang, L.; Glukhov, E.; Gerwick, W.H.; Tan, L.K. Integrated genomic and metabolomic approach to the discovery of potential anti-quorum sensing natural products from microbes associated with marine samples from Singapore. Mar. Drugs 2019, 17, 72. [Google Scholar] [CrossRef] [PubMed]
- Nithya, C.; Begum, M.F.; Pandian, S.K. Marine bacterial isolates inhibit biofilm formation and disrupt mature biofilms of Pseudomonas aeruginosa PAO1. Appl. Microbiol. Biotechnol. 2010, 88, 341–358. [Google Scholar] [CrossRef]
- Busetti, A.; Shaw, G.; Megaw, J.; Gorman, S.P.; Maggs, C.A.; Gilmore, B.F. Marine-derived quorum-sensing inhibitory activities enhance the antibacterial efficacy of tobramycin against Pseudomonas aeruginosa. Mar. Drugs 2015, 13, 1–28. [Google Scholar] [CrossRef] [PubMed]
- Torres, M.; Romero, M.; Prado, S.; Dubert, J.; Tahrioui, A.; Otero, A.; Llamas, I. N-acylhomoserine lactone-degrading bacteria isolated from hatchery bivalve larval cultures. Microbiol. Res. 2013, 168, 547–554. [Google Scholar] [CrossRef] [PubMed]
- Weiland-Bräuer, N.; Pinnow, N.; Schmitz, R.A. Novel reporter for identification of interference with acyl homoserine lactone and autoinducer-2 quorum sensing. Appl. Environ. Microb. 2015, 81, 1477–1489. [Google Scholar] [CrossRef]
- Kanagasabhapathy, M.; Yamazaki, G.; Ishida, A.; Sasaki, H.; Nagata, S. Presence of quorum-sensing inhibitor-like compounds from bacteria isolated from the brown alga Colpomenia sinuosa. Lett. Appl. Microbiol. 2009, 49, 573–579. [Google Scholar] [CrossRef] [PubMed]
- Romero, M.; Martin-Cuadrado, A.B.; Roca-Rivada, A.; Cabello, A.M.; Otero, A. Quorum quenching in cultivable bacteria from dense marine coastal microbial communities. FEMS Microbiol. Ecol. 2010, 75, 205–217. [Google Scholar] [CrossRef]
- Rehman, Z.U.; Leiknes, T. Quorum-quenching bacteria isolated from Red Sea sediments reduce biofilm formation by Pseudomonas aeruginosa. Front. Microbiol. 2018, 9, 1354. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Matamoros, D.; Laiton Fonseca, M.; Duque, C.; RAMOS, F.A.; Castellanos, L. Screening of marine bacterial strains as source of quorum sensing inhibitors (QSI): First chemical study of Oceanobacillus profundus (RKHC-62B). VITAE 2016, 23, 30–47. [Google Scholar] [CrossRef]
- Thenmozhi, R.; Nithyanand, P.; Rathna, J.; Pandian, S.K. Antibiofilm activity of coral-associated bacteria against different clinical M serotypes of Streptococcus pyogenes. FEMS Immunol. Med. Microbiol. 2009, 57, 284–294. [Google Scholar] [CrossRef]
- Musthafa, K.S.; Saroja, V.; Pandian, S.K.; Ravi, A.V. Antipathogenic potential of marine Bacillus sp. SS4 on N-acyl-homoserine-lactone-mediatedvirulence factors production in Pseudomonas aeruginosa (PAO1). J. Biosci. 2011, 36, 55–67. [Google Scholar] [CrossRef]
- You, J.; Xue, X.; Cao, L.; Lu, X.; Wang, J.; Zhang, L.; Zhou, S. Inhibition of Vibrio biofilm formation by a marine actinomycete strain A66. Appl. Microbiol. Biotechnol. 2007, 76, 1137–1144. [Google Scholar] [CrossRef] [PubMed]
- Abed, R.M.M.; Dobretsov, S.; Al-Fori, M.; Gunasekera, S.P.; Sudesh, K.; Paul, V.J. Quorum-sensing inhibitory compounds from extremophilic microorganisms isolated from a hypersaline cyanobacterial mat. J. Ind. Microbiol. Biotechnol. 2013, 40, 759–772. [Google Scholar] [CrossRef]
- Younis, K.M.; Usup, G.; Ahmad, A. Secondary metabolites produced by marine streptomyces as antibiofilm and quorum-sensing inhibitor of uropathogen Proteus mirabilis. Environ. Sci. Pollut. Res. 2015, 23, 4756–4767. [Google Scholar] [CrossRef]
- Chang, H.; Zhou, J.; Zhu, X.; Yu, S.; Chen, L.; Jin, H.; Cai, Z. Strain identification and quorum sensing inhibition characterization of marine-derived Rhizobium sp. NAO1. Roy. Soc. Open Sci. 2017, 4, 170025. [Google Scholar] [CrossRef]
- Tinh, N.T.N.; Asanka Gunasekara, R.A.; Boon, N.; Dierckens, K.; Sorgeloos, P.; Bossier, P. N-acyl homoserine lactone-degrading microbial enrichment cultures isolated from Penaeus vannamei shrimp gut and their probiotic properties in Brachionus plicatilis cultures. FEMS Microbiol. Ecol. 2007, 62, 45–53. [Google Scholar] [CrossRef] [PubMed]
- Tinh, N.T.N.; Yen, V.H.N.; Dierckens, K.; Sorgeloos, P.; Bossier, P. An acyl homoserine lactone-degrading microbial community improves the survival of first-feeding turbot larvae (Scophthalmus maximus L.). Aquaculture 2008, 285, 56–62. [Google Scholar] [CrossRef]
- Cam, D.T.V.; Nhan, D.T.; Ceuppens, S.; Hao, N.V.; Dierckens, K.; Wille, M.; Sorgeloos, P.; Bossier, P. Effect of N-acyl homoserine lactone-degrading enrichment cultures on Macrobrachium rosenbergii larviculture. Aquaculture 2009, 294, 5–13. [Google Scholar] [CrossRef]
- Sun, S.; Dai, X.; Sun, J.; Bu, X.; Weng, C.; Li, H.; Zhu, H. A diketopiperazine factor from Rheinheimera aquimaris QSI02 exhibits anti-quorum sensing activity. Sci. Rep. 2016, 6, 39637. [Google Scholar] [CrossRef] [PubMed]
- Gowrishankar, S.; Poornima, B.; Pandian, S.K. Inhibitory efficacy of cyclo (l-leucyl-l-prolyl) from mangrove rhizosphere bacterium-Bacillus amyloliquefaciens (MMS-50) toward cariogenic properties of Streptococcus mutans. Res. Microbiol. 2014, 165, 278–289. [Google Scholar] [CrossRef] [PubMed]
- Gowrishankar, S.; Pandian, S.K.; Balasubramaniam, B.; Balamurugan, K. Quorum quelling efficacy of marine cyclic dipeptide -cyclo(l-leucyl-l-prolyl) against the uropathogen Serratia marcescens. Food Chem. Toxicol. 2019, 123, 326–336. [Google Scholar] [CrossRef]
- Kjaerulff, L.; Nielsen, A.; Mansson, M.; Gram, L.; Larsen, T.O.; Ingmer, H.; Gotfredsen, C.H. Identification of four new agr quorum sensing-interfering cyclodepsipeptides from a marine photobacterium. Mar. Drugs 2013, 11, 5051–5062. [Google Scholar] [CrossRef]
- Mansson, M.; Nielsen, A.; Kjærulff, L.; Gotfredsen, C.H.; Wietz, M.; Ingmer, H.; Gram, L.; Larsen, T.O. Inhibition of virulence gene expression in Staphylococcus aureus by novel depsipeptides from a marine photobacterium. Mar. Drugs 2011, 9, 2537–2552. [Google Scholar] [CrossRef]
- Nielsen, A.; Månsson, M.; Bojer, M.S.; Gram, L.; Larsen, T.O.; Novick, R.; Frees, D.; Frøkiær, H.; Ingmer, H. Solonamide B inhibits quorum sensing and reduces Staphylococcus aureus mediated killing of human neutrophils. PLoS ONE 2014, 9, e84992. [Google Scholar] [CrossRef]
- Chen, X.; Chen, J.; Yan, Y.; Chen, S.; Xu, X.; Zhang, H.; Wang, H. Quorum sensing inhibitors from marine bacteria Oceanobacillus sp. XC22919. Nat. Prod. Res. 2018, 12, 1–5. [Google Scholar] [CrossRef]
- Teasdale, M.E.; Liu, J.; Wallace, J.; Akhlaghi, F.; Rowley, D.C. Secondary metabolites produced by the marine bacterium Halobacillus salinus that inhibit quorum sensing-controlled phenotypes in gram-negative bacteria. Appl. Environmen. Microb. 2009, 75, 567–572. [Google Scholar] [CrossRef]
- Kwan, J.C.; Teplitski, M.; Gunasekera, S.P.; Paul, V.J.; Luesch, H. Isolation and biological evaluation of 8-epi-malyngamide C from the Floridian marine cyanobacterium Lyngbya majuscula. J. Nat. Prod. 2010, 73, 463–466. [Google Scholar] [CrossRef]
- Kiran, G.S.; Priyadharshini, S.; Dobson, A.D.W.; Gnanamani, E.; Selvin, J. Degradation intermediates of polyhydroxy butyrate inhibits phenotypic expression of virulence factors and biofilm formation in luminescent Vibrio sp. PUGSK8. NPJ Biofilms Microb. 2016, 2, 16002. [Google Scholar] [CrossRef]
- Kwan, J.C.; Meickle, T.; Ladwa, D.; Teplitski, M.; Paul, V.; Luesch, H. Lyngbyoic acid, a “tagged” fatty acid from a marine cyanobacterium, disrupts quorum sensing in Pseudomonas aeruginosa. Mol. BioSyst. 2011, 7, 1205–1216. [Google Scholar] [CrossRef]
- Meyer, J.L.; Gunasekera, S.P.; Scott, R.M.; Paul, V.J.; Teplitski, M. Microbiome shifts and the inhibition of quorum sensing by Black Band Disease cyanobacteria. ISME J. 2015, 10, 1204–1216. [Google Scholar] [CrossRef]
- Montaser, R.; Paul, V.J.; Luesch, H. Modular strategies for structure and function employed by marine cyanobacteria: Characterization and synthesis of pitinoic acids. Org. Lett. 2013, 15, 4050–4053. [Google Scholar] [CrossRef] [PubMed]
- Padmavathi, A.R.; Abinaya, B.; Pandian, S.K. henol, 2,4-bis(1,1-dimethylethyl) of marine bacterial origin inhibits quorum sensing mediated biofilm formation in the uropathogen Serratia marcescens. Biofouling 2014, 30, 1111–1122. [Google Scholar] [CrossRef]
- Zhao, W.; Yuan, T.; Piva, C.; Spinard, E.J.; Schuttert, C.; Rowley, D.C.; Nelson, D.R. The probiotic bacterium Phaeobacter inhibens downregulates virulence factor transcription in the shellfish pathogen, Vibrio coralliilyticus, by N-acyl homoserine lactone production. Appl. Environ. Microb. 2019, 85, e01545-18. [Google Scholar] [CrossRef]
- Choi, H.; Mascuch, S.J.; Villa, F.A.; Byrum, T.; Teasdale, M.E.; Smith, J.E.; Preskitt, L.B.; Rowley, D.C.; Gerwick, L.; Gerwick, W.H. Honaucins A–C, potent inhibitors of inflammation and bacterial quorum sensing: synthetic derivatives and structure-activity relationships. Chem. Biol. 2012, 19, 589–598. [Google Scholar] [CrossRef]
- Dobretsov, S.; Teplitski, M.; Alagely, A.; Gunasekera, S.P.; Paul, V.J. Malyngolide from the cyanobacterium Lyngbya majuscula interferes with quorum sensing circuitry. Env. Microbiol. Rep. 2010, 2, 739–744. [Google Scholar] [CrossRef]
- Kamarudheen, N.; Rao, K.V.B. Fatty acyl compounds from marine Streptomyces griseoincarnatus strain HK12 against two major biofilm forming nosocomial pathogens; an in vitro and in silico approach. Microb. Pathogenesis 2019, 127, 121–130. [Google Scholar] [CrossRef] [PubMed]
- Bruns, H.; Ziesche, L.; Taniwal, N.K.; Wolter, L.; Brinkhoff, T.; Herrmann, J.; Müller, R.; Schulz, S. N-acylated amino acid methyl esters from marine Roseobacter group bacteria. Beilstein J. Org. Chem. 2018, 14, 2964–2973. [Google Scholar] [CrossRef]
- Casillo, A.; Papa, R.; Ricciardelli, A.; Sannino, F.; Ziaco, M.; Tilotta, M.; Selan, L.; Marino, G.; Corsaro, M.M.; Tutino, M.L.; et al. Anti-biofilm activity of a long-chain fatty aldehyde from Antarctic Pseudoalteromonas haloplanktis TAC125 against Staphylococcus epidermidis Biofilm. Front. Cell. Infect. Mi. 2017, 7, 46. [Google Scholar] [CrossRef]
- Durai, S.; Vigneshwari, L.; Balamurugan, K. Caenorhabditis elegans-based in vivo screening of bioactives from marine sponge-associated bacteria against Vibrio alginolyticus. J. Appl. Microbiol. 2013, 115, 1329–1342. [Google Scholar] [CrossRef] [PubMed]
- Clark, B.R.; Engene, N.; Teasdale, M.E.; Rowley, D.C.; Matainaho, T.; Valeriote, F.A.; Gerwick, W.H. Natural Products Chemistry and Taxonomy of the Marine Cyanobacterium Blennothrix cantharidosmum. J. Nat. Prod. 2008, 71, 1530–1537. [Google Scholar] [CrossRef]
- Ibacache-Quiroga, C.; Ojeda, J.; Espinoza-Vergara, G.; Olivero, P.; Cuellar, M.; Dinamarca, M.A. The hydrocarbon-degrading marine bacterium Cobetia sp. strain MM1IDA2H-1 produces a biosurfactant that interferes with quorum sensing of fish pathogens by signal hijacking. Microb. Biotechnol. 2013, 6, 394–405. [Google Scholar] [CrossRef]
- Hentzer, M.; Wu, H.; Andersen, J.B.; Riedel, K.; Rasmussen, T.B.; Bagge, N.; Kumar, N.; Schembri, M.A.; Song, Z.; Kristoffersen, P.; et al. Attenuation of Pseudomonas aeruginosa virulence by quorum sensing inhibitors. EMBO J. 2003, 22, 3803–3815. [Google Scholar] [CrossRef] [PubMed]
- Goh, W.K.; Gardner, C.R.; Chandra Sekhar, K.V.G.; Biswas, N.N.; Nizalapur, S.; Rice, S.A.; Wilcox, M.; Black, D.S.; Kumar, N. Synthesis, quorum sensing inhibition and docking studies of 1,5-dihydropyrrol-2-ones. Bioorg. Med. Chem. 2015, 23, 7366–7377. [Google Scholar] [CrossRef] [PubMed]
- Lillicrap, A.; Macken, A.; Wennberg, A.C.; Grung, M.; Rundberget, J.T.; Fredriksen, L.; Scheie, A.A.; Benneche, T.; d’Auriac, M.A. Environmental fate and effects of novel quorum sensing inhibitors that can control biofilm formation. Chemosphere 2016, 164, 52–58. [Google Scholar] [CrossRef] [PubMed]
- Hansen, A.M.; Peng, P.; Baldry, M.; Perez-Gassol, I.; Christensen, S.B.; Vinther, J.M.O.; Ingmer, H.; Franzyk, H. Lactam hybrid analogues of solonamide B and autoinducing peptides as potent S. aureus AgrC antagonists. Eur. J. Med. Chem. 2018, 152, 370–376. [Google Scholar] [CrossRef]
- Du, Y.; Sun, J.; Gong, Q.; Wang, Y.; Fu, P.; Zhu, W. New α-Pyridones with quorum-sensing inhibitory activity from diversity-enhanced extracts of a Streptomyces sp. derived from marine algae. J. Agric. Food Chem. 2018, 66, 1807–1812. [Google Scholar] [CrossRef] [PubMed]
- Romero, M.; Avendaño-Herrera, R.; Magariños, B.; Cámara, M.; Otero, A. Acylhomoserine lactone production and degradation by the fish pathogen Tenacibaculum maritimum, a member of the Cytophaga-Flavobacterium- Bacteroides (CFB) group. FEMS Microbiol. Lett. 2010, 304, 131–139. [Google Scholar] [CrossRef] [PubMed]
- Nithya, C.; Aravindraja, C.; Pandian, S.K. Bacillus pumilus of Palk Bay origin inhibits quorum-sensing-mediated virulence factors in Gram-negative bacteria. Res. Microbiol. 2010, 161, 293–304. [Google Scholar] [CrossRef] [PubMed]
- Ghani, N.A.; Norizan, S.N.M.; Chan, X.Y.; Yin, W.F.; Chan, K.G. Labrenzia sp. BM1: A quorum quenching bacterium that degrades N-acyl homoserine lactones via lactonase activity. Sensors 2014, 14, 11760–11769. [Google Scholar] [CrossRef]
- Phelan, R.W.; O’Halloran, J.A.; Kennedy, J.; Morrissey, J.P.; Dobson, A.D.W.; O’Gara, F.; Barbosa, T.M. Diversity and bioactive potential of endospore-forming bacteria cultured from the marine sponge Haliclona simulans. J. Appl. Microbiol. 2012, 112, 65–78. [Google Scholar] [CrossRef] [PubMed]
- Kem, M.P.; Naka, H.; Iinishi, A.; Haygood, M.G.; Butler, A. Fatty acid hydrolysis of acyl marinobactin siderophores by Marinobacter Acylases. Biochemistry 2015, 54, 744–752. [Google Scholar] [CrossRef]
- Gutiérrez-Barranquero, J.A.; Parages, M.L.; Dobson, A.D.W.; Reen, F.J.; O’Gara, F. Genome sequence of Paracoccus sp. JM45, a bacterial strain isolated from a marine sponge with a dual quorum sensing inhibition activity. Microbiol. Resour. Announc. 2019, 8, e01496-18. [Google Scholar] [CrossRef]
- Kalia, V.C.; Raju, S.C.; Purohit, H.J. Genomic analysis reveals versatile organisms for quorum quenching enzymes: acyl-homoserine lactone-acylase and -lactonase. Open Microbiol. J. 2011, 5, 1–13. [Google Scholar] [CrossRef]
- Wong, C.S.; Yin, W.F.; Choo, Y.M.; Sam, C.K.; Koh, CL.; Chan, K.G. Coexistence of quorum-quenching and quorum-sensing in tropical marine Pseudomonas aeruginosa strain MW3A. World J. Microbiol. Biotechnol. 2012, 28, 453–461. [Google Scholar] [CrossRef] [PubMed]
- Chan, K.G.; Wong, C.S.; Yin, W.F.; Chan, X.Y. Draft genome sequence of quorum-sensing and quorum-quenching Pseudomonas aeruginosa strain MW3a. Genome Announc. 2014, 2, e00258-14. [Google Scholar] [CrossRef] [PubMed]
- Cai, X.; Yu, M.; Shan, H.; Tian, X.; Zheng, Y.; Xue, C.; Zhang, X.H. Characterization of a novel N-Acylhomoserine Lactonase RmmL from Ruegeria mobilis YJ3. Mar. Drugs 2018, 16, 370. [Google Scholar] [CrossRef]
- Mayer, C.; Romero, M.; Muras, A.; Otero, A. Aii20J, a wide-spectrum thermostable N-acylhomoserine lactonase from the marine bacterium Tenacibaculum sp. 20J, can quench AHL-mediated acid resistance in Escherichia coli. Appl. Microbiol. Biotechnol. 2015, 99, 9523–9539. [Google Scholar] [CrossRef]
- Lu, X.; Yuan, Y.; Xue, X.L.; Zhang, G.P.; Zhou, S.N. Identification of the critical role of Tyr-194 in the catalytic activity of a novel N-acyl-homoserine lactonase from marine Bacillus cereus strain Y2. Curr. Microbiol. 2006, 53, 346–350. [Google Scholar] [CrossRef]
- Tang, K.; Su, Y.; Brackman, G.; Cui, F.; Zhang, Y.; Shi, X.; Coenye, T.; Zhang, X.H. MomL, a novel marine-derived N-acyl homoserine lactonase from Muricauda olearia. Appl. Environ. Microbiol. 2015, 81, 774–782. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Lin, Y.; Yi, S.; Liu, P.; Shen, J.; Shao, Z.; Liu, Z. QsdH, a novel AHL lactonase in the RND-type inner membrane of marine Pseudoalteromonas byunsanensis strain 1A01261. PLoS ONE 2012, 7, e46587. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, J.; Tang, K.; Yu, M.; Coenye, T.; Zhang, X.-H. Genome analysis of Flaviramulus ichthyoenteri Th78T in the family Flavobacteriaceae: Insights into its quorum quenching property and potential roles in fish intestine. BMC Genom. 2015, 16, 38. [Google Scholar] [CrossRef] [PubMed]
- Liu, N.; Yu, M.; Zhao, Y.; Cheng, J.; An, K.; Zhang, X.H. PfmA, a novel quorum-quenching N-acylhomoserine lactone acylase from Pseudoalteromonas flavipulchra. Microbiology 2017, 163, 1389–1398. [Google Scholar] [CrossRef]
- Glinel, K.; Thebault, P.; Humblot, V.; Pradier, C.M.; Jouenne, T. Antibacterial surfaces developed from bio-inspired approaches. Acta Biomater. 2012, 8, 1670–1684. [Google Scholar] [CrossRef] [PubMed]
| Structures of QS Inhibitors | Marine Bacterial Origins | Sources of the Bacteria | QS-Inhibitory Concentration | Anti-QS Working Mechanisms | Effects | References |
|---|---|---|---|---|---|---|
| Cyclic peptides and a linear peptide | ||||||
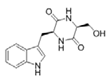 Cyclo (Trp-Ser) | Rheinheimera aquimaris | Marine sediment surrounding the Yellow Sea in Qingdao, China | sub-MIC: 0.2 mg/mL | Probably interfere in the stability of LasR receptor | Suppress biofilm formation and QS-regulated pyocyanin and elastase activity in Pseudomonas aeruginosa | [59] |
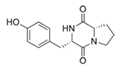 Cyclo-l-proline-l-tyrosine | Bacillus cereus | Marine sediment along the Rhode Island coastline | - | Possibly inhibit Vibrio sp. LuxR | - | [37] |
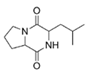 Cyclo (l-leucyl-l-prolyl) | Bacillus amyloliquefaciens | Mangrove rhizosphere of Palk Strait, Bay of Bengal, India | sub-MIC: 100 μg/mL | - | Inhibit QS-controlled biofilm and virulence factors (prodigiosin, extracellular polymeric substance, protease, and lipase) production in uropathogen Serratia marcescens | [60,61] |
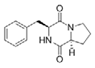 Cyclo (l-Pro-l-Phe) | Marinobacter sp. | A hypersaline cyanobacterial mat from wadi Muqshin in Oman, off the Arabian Sea coast | sub-MIC: μM grade | Possibly compete with signal molecules AHLs and inhibit QS | - | [53] |
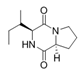 Cyclo (l-Pro-l-isoLeu) | ||||||
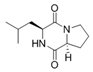 Cyclo (l-Pro-lLeu) | ||||||
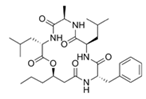
 Ngercheumicin F (R = C11H21), Ngercheumicin G (R = C11H23), Ngercheumicin H (R = C13H25), Ngercheumicin I (R = C13H27) (Cyclodepsipeptides) | Photobacterium halotolerans | Mussel surface in the tropical Pacific | 20 μg/mL | Interfere with agr QS | Reduce expression of virulence genes of hla (hemolysin) and rnaIII (an effector molecule) of agr QS in Staphylococcus aureus | [62] |
 Solonamide A (Fatty acid-l-Phe- d-Leu-d-Ala-l-Leu) | Photobacterium halotolerans | Mussel surface in the tropical Pacific | μg/mL–mg/mL | Possibly interfere with the agr QS system by competing with AIP for binding to sensor histidine kinase AgrC | Inhibit virulence gene expression of hla (hemolysin), rnaIII (an effector molecule of agr) and psmα (phenol soluble modulins) in Staphylococcus aureus, and reduce toxicity of S. aureus toward human neutrophils | [63,64] |
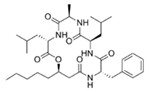 Solonamide B (Fatty acid-l-Phe- d-Leu-d-Ala-l-Leu) | ||||||
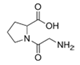 Linear dipeptide proline-glycine | Streptomyces sp. | Marine invertebrates from the western coast of India | sub-MIC: 0.1 mg/mL | - | Inhibit QS-mediated virulence factors (swarming, pyocyanin pigmentation, biofilm formation, rhamnolipid production, and Las A) in Pseudomonas aeruginosa | [32] |
| Amides | ||||||
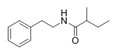 2-Methyl-N-(2′-phenylethyl) butyramide | Oceanobacillus sp. | Marine environment | sub-MIC: μg/mL grade | - | Inhibit pyocyanin production, elastase activity and biofilm formation of Pseudomonas aeruginosa | [65] |
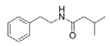 3-Methyl-N-(2′-phenylethyl)-butyramide | ||||||
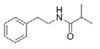 N-(2′-Phenylethyl)-isobutyramide | Halobacillus salinus | Sea grass sample from Point Judith Salt Pond, Rhode Island | sub-MIC: μg/mL grade | Possibly compete with AHLs for receptor binding | - | [66] |
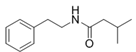 3-Methyl-N-(2′-phenylethyl)-butyramide | ||||||
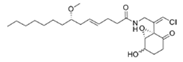 8-epi-Malyngamide C (8R) and malyngamide C (8S) | cyanobacterium Lyngbya majuscula | Off Bush Key, Florida | μM grade | - | - | [67] |
| Fatty Acids and phenol derivatives | ||||||
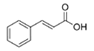 cinnamic acid | Streptomyces sp. | Marine invertebrates from the western coast of India | sub-MIC: 0.1 mg/mL | - | Inhibit QS-mediated virulence factors (swarming, biofilm formation, LasA, pyocyanin and rhamnolipid production) in Pseudomonas aeruginosa | [32] |
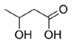 β-Hydroxy butyric acid (degradation product of PHB) | Brevibacterium casei (sources of PHB) | Marine sponge Dendrilla nigra | 50 μg/mL (PHB) | Possibly by AHL degradation | Control biofilm formation, colonization capacity, motility and hemolysin activity of Vibrio sp. | [68] |
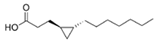 Lyngbyoic acid | cyanobacterium Lyngbya cf. majuscula | Indian River Lagoon near Fort Pierce, Florida | μM–mM grade | Possibly inhibit lasR signaling by competing with AHL for binding LasR | Reduce pyocyanin and elastase (LasB) in Pseudomonas aeruginosa | [69] |
 Lyngbic acid | cyanobacterium | Corals from the Florida Keys and Belize | nM–μM grade | Compete with CAI-1 for binding to QS signal receptor CqsS | - | [70] |
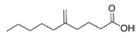 Pitinoic acid A | cyanobacterium similar to Lyngbya sp. | A channel at the north end of Piti Bay at Guam | sub-MIC: μM–mM grade | - | Inhibit expression of QS-related virulence factor LasB (elastase) and the pyocyanin in Pseudomonas aeruginosa | [71] |
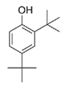 Phenol, 2,4-bis (1,1-dimethylethyl) | Vibrio alginolyticus | Red seaweed Gracilaria gracilis from the Karankadu coastal region of Palk Bay, India | sub-MIC: μg/mL grade | - | Inhibit QS-mediated biofilm formation and virulence factor production (protease, hemolysin, lipase, prodigiosin, and extracellular polysaccharide) in the uropathogen Serratia marcescens | [72] |
 Tyrosol/tyrosol acetate (R = H or Ac) | Oceanobacillus profundus | Caribbean soft coral Antillogorgia elisabethae | - | - | - | [49] |
| AHL analogs | ||||||
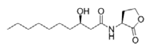 N-(3-Hydroxydecanoyl)-l-homoserine lactone | Phaeobacter inhibens | Inner surface of an oyster shell | μM grade | Possibly by competitive inhibition of AHL-mediated QS | Inhibit virulence factor metalloprotease in Vbrio coralliilyticus | [73] |
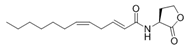 N-(Dodecanoyl-2,5-diene)-l-homoserine lactone | ||||||
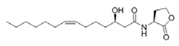 O-(3-Hydroxytetradecanoyl-7-ene)-l-homoserine lactone | ||||||
 d,l-Homocysteine thiolactone | Staphylococcus hominis | coral species (Pocillopora damicornis) in Xishan Islands, South China Sea | above 0.0625 μg/mL | Possibly compete with AHL in occupying the AHL receptor | Suppress biofilm formation and elastase production in Pseudomonas aeruginosa | [31] |
| Others | ||||||
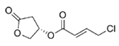 Honaucin A | cyanobacterium Leptolyngbya crossbyana | Corals on the Hawaiian coast | μM grade | Possibly compete with AHL in occupying the AHL receptor | - | [74] |
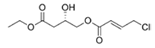 Honaucin B | - | |||||
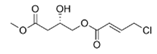 Honaucin C | - | |||||
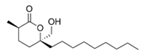 Malyngolide | Cyanobacterium Lyngbya majuscula | Indian River Lagoon, USA | sub-MIC: 3.57–57 μM | Possibly inhibit QS by reducing or partially blocking the expression of lasR | Inhibit Las QS-dependent production of elastase by Pseudomonas aeruginosa, and possibly help the cyanobacterium to control growth of heterotrophic bacteria | [75] |
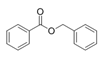 Benzyl benzoate | Oceanobacillus sp. | Marine environment | sub-MIC: μg/mL grade | - | Inhibit pyocyanin production, elastase activity and biofilm formation of Pseudomonas aeruginosa | [65] |
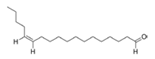 13Z-Octadecenal | Streptomyces griseoincarnatus | Marine sponge Callyspongia sp. from Gulf of Mannar, India | - | Bind to AHL synthase LasI of Pseudomonas aeruginosa | Possibly inhibit Pseudomonas aeruginosa biofilm | [76] |
| Quorum Quenching Enzymes | Protein Sequence Accession Number | Protein Superfamily/Family | Bacteria | Marine Origin of the Bacteria | Substrate Spectrum | Environmental Tolerance | Applications | References |
|---|---|---|---|---|---|---|---|---|
| AHL lactonase Aii20J | AKN24544 | Metallo-β-lactamase | Tenacibaculum sp. | Sediment of fish culture tank, Spain | AHLs (C4-HSL, C6-HSL, C8-HSL, C10-HSL, C12-HSL, C14-HSL, OC6-HSL, OC10-HSL, OC12-HSL, OC13-HSL, OC14-HSL, OHC10-HSL, OHC12-HSL) | With heat resistance in cell extracts, tolerance to protease and wide pH range 3–9 | Quench AHL-mediated acid resistance in Escherichia coli (gastrointestinal pathogens) | [97] |
| AHL lactonase AiiA | CAJ84442 | Metallo-β-lactamase | Bacillus cereus | Seawater samples of South China Sea | OC8-HSL | - | - | [98] |
| AHL lactonase MomL | AIY30473 | Metallo-β-lactamase | Muricauda olearia | Skin mucus of flounders from marine fish farms in China | AHLs (C4-HSL, C6-HSL, C8-HSL, OC6-HSL, OC8-HSL, OC10-HSL) | No heat resistance, tolerance to pH range 7–11 | Attenuate the virulence (extracellular protease activity and pyocyanin production) of Pseudomonas aeruginosa, increase the survival of Caenorhabditis elegans | [99] |
| AHL lactonase QsdH | (Included in) AFV15299 | GDSL hydrolase | Pseudoalteromonas byunsanensis | Marine Culture Collection of China | AHL (C4HSL, C6HSL, C8HSL, C10HSL, C12HSL, C14HSL, OC6-HSL, OC8-HSL) | No heat resistance | Attenuate the plant pathogenicity of Erwinia carotovora | [100] |
| AHL lactonase RmmL | AYM45058 | Metallo-β-lactamase | Ruegeria mobilis | Healthy shrimp larvae | AHL (C6-HSL, C8-HSL, C10-HSL, C12-HSL, OC6-HSL, OC8-HSL, OC10-HSL, OC12-HSL, OC14-HSL) | No heat resistance, tolerance to pH range 2–9 | Reduce the production of virulent factor pyocyanin by Pseudomonas aeruginosa | [96] |
| AHL lactonase FiaL | - | Metallo-β-lactamase | Flaviramulus ichthyoenteri | Intestine of cultured healthy flounder in China | AHL (C6-HSL, C8-HSL, C10-HSL, C12-HSL, C14-HSL, OC6-HSL, OC8-HSL, OC10-HSL, OC12-HSL, OC14-HSL) | - | - | [101] |
| AHL acylase MhtA | ENO13542 | Ntn-hydrolases | Marinobacter nanhaiticus | Sediment of the South China Sea | AHL (C12-HSL) | - | - | [91] |
| AHL acylase PfmA | ASS36259 | Ntn hydrolase | Pseudoalteromonas flavipulchra | Water used to rear healthy turbot in China | AHL (C10-HSL, C12-HSL, C14-HSL, OC12-HSL, OC14-HSL, OHC14-HSL) | No heat resistance, tolerance to pH range 5–11 | Reduce virulence factor protease production in Vibrio anguillarum, reduce protease and pyocyanin in Pseudomonas aeruginosa, increase survival of infected Artemia | [102] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, J.; Li, X.; Hou, X.; Quan, C.; Chen, M. Widespread Existence of Quorum Sensing Inhibitors in Marine Bacteria: Potential Drugs to Combat Pathogens with Novel Strategies. Mar. Drugs 2019, 17, 275. https://doi.org/10.3390/md17050275
Zhao J, Li X, Hou X, Quan C, Chen M. Widespread Existence of Quorum Sensing Inhibitors in Marine Bacteria: Potential Drugs to Combat Pathogens with Novel Strategies. Marine Drugs. 2019; 17(5):275. https://doi.org/10.3390/md17050275
Chicago/Turabian StyleZhao, Jing, Xinyun Li, Xiyan Hou, Chunshan Quan, and Ming Chen. 2019. "Widespread Existence of Quorum Sensing Inhibitors in Marine Bacteria: Potential Drugs to Combat Pathogens with Novel Strategies" Marine Drugs 17, no. 5: 275. https://doi.org/10.3390/md17050275
APA StyleZhao, J., Li, X., Hou, X., Quan, C., & Chen, M. (2019). Widespread Existence of Quorum Sensing Inhibitors in Marine Bacteria: Potential Drugs to Combat Pathogens with Novel Strategies. Marine Drugs, 17(5), 275. https://doi.org/10.3390/md17050275




