Seasonal Variability of the Biochemical Composition and Antioxidant Properties of Fucus spiralis at Two Azorean Islands
Abstract
1. Introduction
2. Results and Discussion
2.1. Crude Protein Determination
2.2. Soluble Carbohydrate Content Determination
2.3. Total Lipid Determination
2.4. Fatty Acids (FA) Determination
2.5. Total, Soluble, and Insoluble Dietary Fiber (TDF, SDF and IDS) Determination
2.6. Ash Determination
2.7. Energy Value Determination
2.8. Total Phenolic Content (TPC) Determination in F. spiralis Extracts
2.9. Total Flavonoid Content (TFC) Determination in F. spiralis Extracts
2.10. Antioxidant Activity Assays in F. spiralis Extracts
2.10.1. 2,2-Diphenyl-1-Picrylhydrazyl (DPPH) Free Radical Scavenging Activity (FRSA) Assay
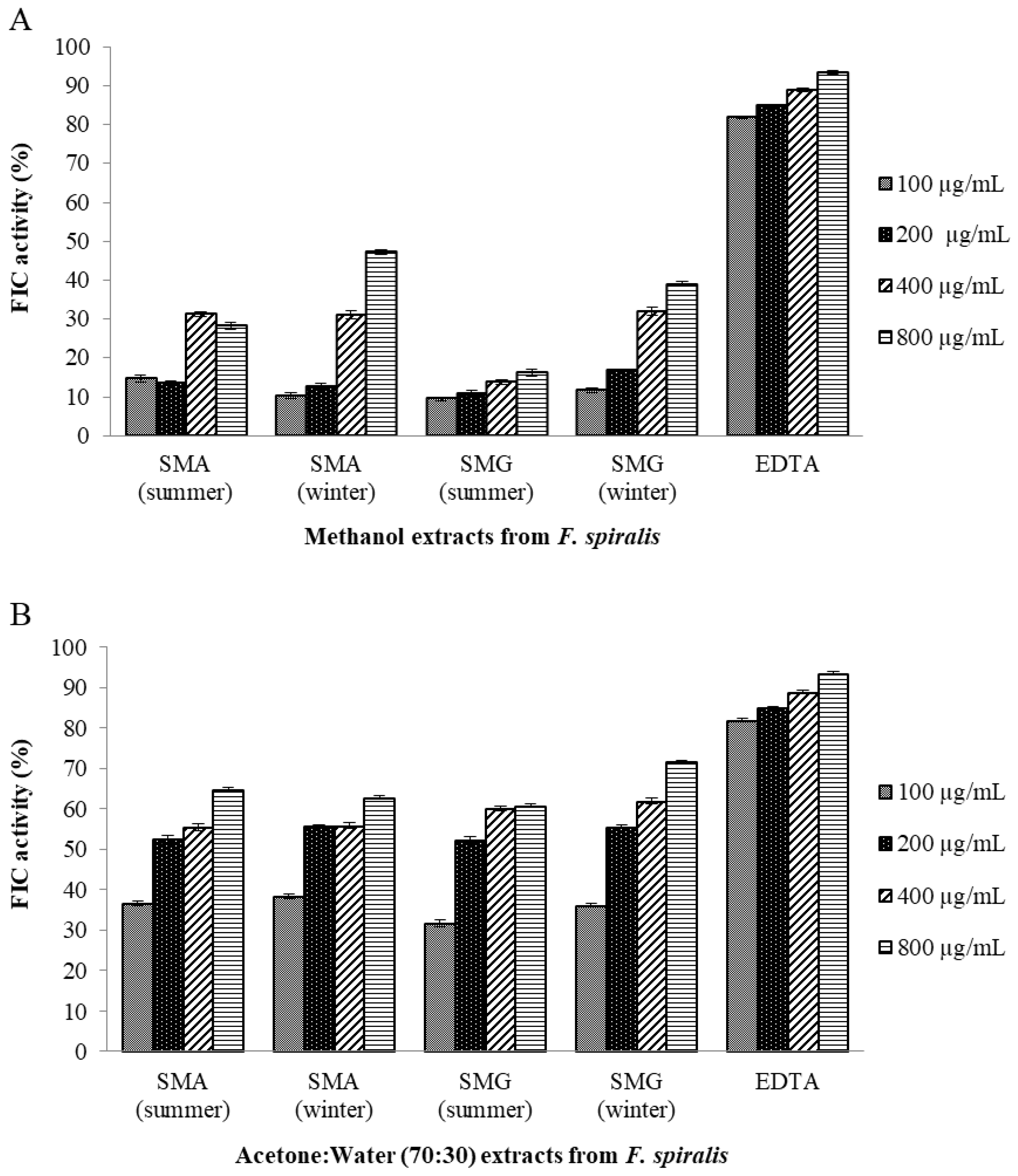
2.10.2. Ferrous Ion-Chelating (FIC) Activity Assay
2.10.3. Ferric Reducing Antioxidant Power (FRAP) Assay
2.10.4. Pearson Correlation between the Antioxidant Activity Parameters
3. Materials and Methods
3.1. Chemicals and Reagents
3.2. Study Site and Environmental Parameters of Season Collecting Samples
3.3. Collection and Preparation of F. spiralis Samples
3.4. Preparation of F. spiralis Extracts
3.5. Nutrient Analysis
3.5.1. Crude Protein Determination
3.5.2. Soluble Carbohydrate Content Determination
3.5.3. Total Lipid and Fatty Acids (FA) Determination
3.5.4. Total, Soluble, and Insoluble Dietary Fiber (TDF, SDF and IDS) Determination
3.5.5. Ash Determination
3.5.6. Energy Value Determination
3.6. Total Phenolic Content (TPC) Determination in F. spiralis Extracts
3.7. Total Flavonoid Content (TFC) Determination in F. spiralis Extracts
3.8. Antioxidant Activity Assays in F. spiralis Extracts
3.8.1. DPPH Free Radical Scavenging Activity (FRSA) Assay
3.8.2. Ferrous Ion-Chelating (FIC) Activity Assay
3.8.3. Ferric Reducing Antioxidant Power (FRAP) Assay
3.9. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Fleurence, J. Seaweed proteins: Biochemical, nutritional aspects and potential uses. Trends Food Sci. Technol. 1999, 10, 25–28. [Google Scholar] [CrossRef]
- Ortiz, J.; Romero, N.; Robert, P.; Araya, J.; Lopez-Hernández, J.; Bozzo, C.; Navarrete, E.; Osorio, A.; Rios, A. Dietary fiber, amino acid, fatty acid and tocopherol contents of the edible seaweeds Ulva lactuca and Durvillaea antarctica. Food Chem. 2006, 99, 98–104. [Google Scholar] [CrossRef]
- Bocanegra, A.; Bastida, S.; Benedí, J.; Ródenas, S.; Sánchez-Muniz, F.J. Characteristics and nutritional and cardiovascular-health properties of seaweeds. J. Med. Food 2009, 12, 236–258. [Google Scholar] [CrossRef] [PubMed]
- Lordan, S.; Ross, R.P.; Stanton, C. Marine bioactives as functional food ingredients: Potential to reduce the incidence of chronic diseases. Mar. Drugs 2011, 9, 1056–1100. [Google Scholar] [CrossRef] [PubMed]
- Holdt, S.L.; Kraan, S. Bioactive compounds in seaweed: Functional food applications and legislation. J. Appl. Phycol. 2011, 23, 543–597. [Google Scholar] [CrossRef]
- Stengel, D.B.; Connan, S.; Popper, Z.A. Algal chemodiversity and bioactivity: Sources of natural variability and implications for commercial application. Biotechnol. Adv. 2011, 29, 483–501. [Google Scholar] [CrossRef] [PubMed]
- Zavodnik, N. Seasonal variations in rate of photosynthetic activity and chemical composition of the littoral seaweeds common to the north Adriatic. Part I. Fucus virsoides (Don) J. AG. Bot. Mar. 1973, 16, 155–165. [Google Scholar]
- Nygård, C.A.; Dring, M.J. Influence of salinity, temperature, dissolved inorganic carbon and nutrient concentration on the photosynthesis and growth of Fucus vesiculosus from the Baltic and Irish seas. Eur. J. Phycol. 2008, 43, 253–262. [Google Scholar] [CrossRef]
- Gosch, B.J.; Paul, N.A.; de Nys, R.; Magnusson, M. Seasonal and within-plant variation in fatty acid content and composition in the brown seaweed Spatoglossum macrodontum (Dictyotales, Phaeophyceae). J. Appl. Phycol. 2015, 27, 387–398. [Google Scholar] [CrossRef]
- Rickert, E.; Wahl, M.; Link, H.; Richter, H.; Pohnert, G. Seasonal variations in surface metabolite composition of Fucus vesiculosus and Fucus serratus from the Baltic Sea. PLoS ONE 2016, 11, e0168196. [Google Scholar] [CrossRef] [PubMed]
- Schmid, M.; Guihéneuf, F.; Stengel, D.B. Ecological and commercial implications of temporal and spatial variability in the composition of pigments and fatty acids in five Irish macroalgae. Mar. Biol. 2017, 164, 158. [Google Scholar] [CrossRef]
- Neto, A.I.; Brotas, V.; Azevedo, J.M.N.; Patarra, R.F.; Álvaro, N.M.V.; Gameiro, C.; Prestes, A.C.L.; Nogueira, E.M. Qualidade de Águas Costeiras do Grupo Oriental do Arquipélago dos Açores e Proposta de Monitorização; Universidade dos Açores: Ponta Delgada, Portugal, 2009. [Google Scholar]
- Neto, A.I.; Tittley, I.; Raposeiro, P.M. Flora Marinha do Litoral dos Açores. Rocky Shore Marine Flora of the Azores; Secretaria Regional do Ambiente e do Mar: Horta, Portugal, 2005. [Google Scholar]
- Paiva, L.; Lima, E.; Patarra, R.F.; Neto, A.I.; Baptista, J. Edible Azorean macroalgae as source of rich nutrients with impact on human health. Food Chem. 2014, 164, 128–135. [Google Scholar] [CrossRef] [PubMed]
- Paiva, L.; Lima, E.; Neto, A.I.; Baptista, J. Angiotensin I-converting enzyme (ACE) inhibitory activity of Fucus spiralis macroalgae and influence of the extracts storage temperature: A short report. J. Pharm. Biomed. Anal. 2016, 131, 503–507. [Google Scholar] [CrossRef] [PubMed]
- Paiva, L.; Lima, E.; Neto, A.I.; Baptista, J. Angiotensin I-converting enzyme (ACE) inhibitory activity, antioxidant properties, phenolic content and amino acid profiles of Fucus spiralis L. protein hydrolysate fractions. Mar. Drugs 2017, 15, 311. [Google Scholar] [CrossRef] [PubMed]
- Catarino, M.D.; Silva, A.M.S.; Cardoso, S.M. Fucaceae: A source of bioactive phlorotannins. Int. J. Mol. Sci. 2017, 18, 1327. [Google Scholar] [CrossRef] [PubMed]
- Cerantola, S.; Breton, F.; Ar Gall, E.; Deslandes, E. Co-occurrence and antioxidant activities of fucol and fucophlorethol classes of polymeric phenols in Fucus spiralis. Bot. Mar. 2006, 49, 347–351. [Google Scholar] [CrossRef]
- Tierney, M.S.; Smyth, T.J.; Rai, D.K.; Soler-Vila, A.; Croft, A.K.; Brunton, N. Enrichment of polyphenol contents and antioxidant activities of Irish brown macroalgae using food-friendly techniques based on polarity and molecular size. Food Chem. 2013, 139, 753–761. [Google Scholar] [CrossRef] [PubMed]
- Pinteus, S.; Silva, J.; Alves, C.; Horta, A.; Fino, N.; Rodrigues, A.I.; Mendes, S.; Pedrosa, R. Cytoprotective effect of seaweeds with high antioxidant activity from the Peniche coast (Portugal). Food Chem. 2017, 218, 591–599. [Google Scholar] [CrossRef] [PubMed]
- Munda, I.M.; Kremer, B.P. Chemical composition and physiological properties of fucoids under conditions of reduced salinity. Mar. Biol. 1977, 42, 9–15. [Google Scholar] [CrossRef]
- Juneja, A.; Ceballos, R.M.; Murthy, G.S. Effects of environmental factors and nutrient availability on the biochemical composition of algae for biofuels production: A review. Energies 2013, 6, 4607–4638. [Google Scholar] [CrossRef]
- Marinho-Soriano, E.; Fonseca, P.C.; Carneiro, M.A.A.; Moreira, W.S.C. Seasonal variation in the chemical composition of two tropical seaweeds. Bioresour. Technol. 2006, 97, 2402–2406. [Google Scholar] [CrossRef] [PubMed]
- Rupérez, P.; Saura-Calixto, F. Dietary fibre and physicochemical properties of edible Spanish seaweeds. Eur. Food Res. Technol. 2001, 212, 349–354. [Google Scholar] [CrossRef]
- Rodrigues, J.M. Avaliação do valor Nutricional e de Propriedades Antioxidantes das Macroalgas Chaetomorpha linum, Ulva lactuca, Gracilaria vermiculophylla e Fucus vesiculosus. Master’s Thesis, Escola Superior Agrária, Instituto Politécnico de Coimbra, Coimbra, Portugal, 2013. [Google Scholar]
- Marrion, O.; Schwertz, A.; Fleurence, J.; Gueant, J.L.; Villaume, C. Improvement of the digestibility of the proteins of the red alga Palmaria palmata by physical processes and fermentation. Nahrung/Food 2003, 47, 339–344. [Google Scholar] [CrossRef] [PubMed]
- Wong, K.H.; Cheung, P.C.K. Nutritional evaluation of some subtropical red and green seaweeds: Part 1—Proximate composition, amino acid profiles and some physico-chemical properties. Food Chem. 2000, 71, 475–482. [Google Scholar] [CrossRef]
- Moskaug, J.Ø.; Carlsen, H.; Myhrstad, M.C.W.; Blomhoff, R. Polyphenols and glutathione synthesis regulation. Am. J. Clin. Nutr. 2005, 81, 277–283. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, S.; Santra, S.C. Biochemical composition of eight benthic algae collected from Sunderban. Indian J. Mar. Sci. 2008, 37, 329–332. [Google Scholar]
- Manivannan, K.; Thirumaran, G.; Devi, G.K.; Hemalatha, A.; Anantharaman, P. Biochemical composition of seaweeds from Mandapam coastal regions along Southeast Coast of India. Am. Eurasian J. Bot. 2008, 1, 32–37. [Google Scholar]
- Rosemberg, G.; Ramus, J. Ecological growth strategies in the seaweeds Gracilaria follifera (Rhodophyceae) and Ulva sp. (Chlorophyceae): Soluble nitrogen and reserve carbohydrates. Mar. Biol. 1982, 66, 251–259. [Google Scholar] [CrossRef]
- Yuan, Y.V. Marine algal constituens. In Marine Nutraceuticals and Functional Foods; Barrow, C., Shahidi, F., Eds.; CRC Press & Taylor and Francis Group: Boca Raton, FL, USA, 2008; pp. 259–296. [Google Scholar]
- Darcy-Vrillon, B. Nutritional aspects of the developing use of marine macroalgae for the human food industry. Int. J. Food Sci. Nutr. 1993, 44, 23–35. [Google Scholar]
- Lorenzo, J.M.; Agregán, R.; Munekata, P.E.S.; Franco, D.; Carballo, J.; Sahin, S.; Lacomba, R.; Barba, F.J. Proximate composition and nutritional value of three macroalgae: Ascophyllum nodosum, Fucus vesiculosus and Bifurcaria bifurcate. Mar. Drugs 2017, 15, 360. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.-K.; Dubacq, J.-P.; Thomas, J.-C.; Giraud, G. Seasonal variations of triacylglycerols and fatty acids in Fucus serratus. Phytochemistry 1996, 43, 49–55. [Google Scholar] [CrossRef]
- Schmid, M.; Guihéneuf, F.; Stengel, D.B. Fatty acid contents and profiles of 16 macroalgae collected from the Irish Coast at two seasons. J. Appl. Phycol. 2014, 26, 451–463. [Google Scholar] [CrossRef]
- Schmid, M.; Guihéneuf, F.; Stengel, D.B. Plasticity and remodelling of lipids support acclimation potential in two species of low-intertidal macroalgae, Fucus serratus (Phaeophyceae) and Palmaria palmata (Rhodophyta). Algal Res. 2017, 26, 104–114. [Google Scholar] [CrossRef]
- Sanina, N.M.; Goncharova, S.N.; Kostetsky, E.Y. Seasonal changes of fatty acid composition and thermotropic behavior of polar lipids from marine macrophytes. Phytochemistry 2008, 69, 1517–1527. [Google Scholar] [CrossRef] [PubMed]
- Gerasimenko, N.; Logvinov, S. Seasonal composition of lipids, fatty acids pigments in the brown alga Sargassum pallidum: The potential for health. Open J. Mar. Sci. 2016, 6, 498–523. [Google Scholar] [CrossRef]
- Matanjun, P.; Mohamed, S.; Mustapha, N.M.; Muhammad, K. Nutrient content of tropical edible seaweeds, Eucheuma cottonii, Caulerpa lentillifera and Sargassum polycystum. J. Appl. Phycol. 2009, 21, 75–80. [Google Scholar] [CrossRef]
- Nelson, M.M.; Phleger, C.F.; Nichols, P.D. Seasonal lipid composition in macroalgae of the Northeastern Pacific Ocean. Bot. Mar. 2002, 45, 58–65. [Google Scholar] [CrossRef]
- Testi, S.; Bonaldo, A.; Gatta, P.P.; Badiani, A. Nutritional traits of dorsal and ventral fillets from three farmed fish species. Food Chem. 2006, 98, 104–111. [Google Scholar] [CrossRef]
- Tonial, I.B.; Oliveira, D.F.; Coelho, A.R.; Matsushita, M.; Coró, F.A.G.; Souza, N.E.; Visentainer, J.V. Quantification of essential fatty acids and assessment of the nutritional quality indexes of lipids in tilapia alevins and juvenile tilapia fish (Oreochromis niloticus). J. Food Res. 2014, 3, 105–114. [Google Scholar] [CrossRef]
- Simopoulos, A.P. An increase in the omega-6/omega-3 fatty acid ratio increases the risk for obesity. Nutrients 2016, 8, 128. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, D.; Freitas, A.C.; Pereira, L.; Rocha-Santos, T.A.P.; Vasconcelos, M.W.; Roriz, M.; Rodriguez-Alcalá, L.M.; Gomes, A.M.P.; Duarte, A.C. Chemical composition of red, brown and green macroalgae from Buarcos bay in Central West Coast of Portugal. Food Chem. 2015, 183, 197–207. [Google Scholar] [CrossRef] [PubMed]
- Ginsberg, H.N.; Barr, S.L.; Gilbert, A.; Karmalli, W.; Deckelbaum, R.; Kaplan, K.; Ramakrishnan, R.; Holleran, S.; Dell, R.B. Reduction of plasma cholesterol levels in normal men on an American Heart Association step 1 diet or a step 1 diet with added monounsaturated fat. N. Engl. J. Med. 1990, 322, 574–579. [Google Scholar] [CrossRef] [PubMed]
- Jiménez-Escrig, A.; Sánchez-Muniz, F.J. Dietary fibre from edible seaweeds: Chemical structure, physiochemical properties and effects on cholesterol metabolism. Nutr. Res. 2000, 20, 585–598. [Google Scholar] [CrossRef]
- Ortiz, J.; Uquiche, E.; Robert, P.; Romero, N.; Quitral, V.; Llantén, C. Functional and nutritional value of the Chilean seaweeds Codium fragile, Gracilaria chilensis and Macrocystis pyrifera. Eur. J. Lipid Sci. Technol. 2009, 111, 320–327. [Google Scholar] [CrossRef]
- McDermid, K.J.; Stuercke, B.; Haleakala, O.J. Total dietary fiber content in Hawaiian marine algae. Bot. Mar. 2005, 48, 437–440. [Google Scholar] [CrossRef]
- Díaz-Rubio, M.E.; Pérez-Jiménez, J.; Saura-Calixto, F. Dietary fiber and antioxidant capacity in Fucus vesiculosus products. Int. J. Food Sci. Nutr. 2009, 60, 23–34. [Google Scholar] [CrossRef] [PubMed]
- Southgate, D.A.T. Dietary fiber and health. In Dietary Fiber: Chemical and Biological Aspects; Southgate, D.A.T., Waldron, K., Johnson, I.T., Fenwick, G.R., Eds.; The Royal Society of Chemistry: Cambridge, UK, 1990; pp. 10–19. [Google Scholar]
- Godard, M.; Décordé, K.; Ventura, E.; Soteras, G.; Baccou, J.-C.; Cristol, J.-P.; Rouanet, J.M. Polysaccharides from the green alga Ulva rigida improve the antioxidant status and prevent fatty streak lesions in the high cholesterol fed hamster, an animal model of nutritionally-induced atherosclerosis. Food Chem. 2009, 115, 176–180. [Google Scholar] [CrossRef]
- Rosell, K.-G.; Srivastava, L.M. Seasonal variation in the chemical constituents of the brown algae Macrocystis integrifolia and Nereocystis luetkeana. Can. J. Bot. 1984, 62, 2229–2236. [Google Scholar] [CrossRef]
- Mabeau, S.; Fleurence, J. Seaweed in food products: Biochemical and nutritional aspects. Trends Food Sci. Technol. 1993, 4, 103–107. [Google Scholar] [CrossRef]
- Kaehler, S.; Kennish, R. Summer and winter comparisons in the nutritional value of 447 marine macroalgae from Hong Kong. Bot. Mar. 1996, 39, 11–17. [Google Scholar] [CrossRef]
- Ortega-Calvo, J.J.; Mazuelos, C.; Hermosín, B.; Sáiz-Jiménez, C. Chemical composition of Spirulina and Eukaryotic algae food-products marketed in Spain. J. Appl. Phycol. 1993, 5, 425–435. [Google Scholar] [CrossRef]
- Rupérez, P. Mineral content of edible marine seaweeds. Food Chem. 2002, 79, 23–26. [Google Scholar] [CrossRef]
- Renaud, S.M.; Luong-Van, J.T. Seasonal variation in the chemical composition of tropical Australian marine macroalgae. J. Appl. Phycol. 2006, 18, 381–387. [Google Scholar] [CrossRef]
- Tierney, M.S.; Soler-vila, A.; Croft, A.K.; Hayes, M. Antioxidant activity of the brown macroalgae Fucus spiralis Linnaeus harvested from the west coast of Ireland. Curr. Res. J. Biol. Sci. 2013, 5, 81–90. [Google Scholar]
- Wang, T.; Jónsdóttir, R.; Liu, H.; Gu, L.; Kristinsson, H.G.; Raghavan, S.; Ólafsdóttir, G. Antioxidant capacities of phlorotannins extracted from the brown algae Fucus vesiculosus. J. Agric. Food Chem. 2012, 60, 5874–5883. [Google Scholar] [CrossRef] [PubMed]
- Steevensz, A.J.; MacKinnon, S.L.; Hankinson, R.; Craft, C.; Connan, S.; Stengel, D.B.; Melanson, J.E. Profiling phlorotannins in brown macroalgae by liquid chromatography-high resolution mass spectrometry. Phytochem. Anal. 2012, 23, 547–553. [Google Scholar] [CrossRef] [PubMed]
- Cox, S.; Abu-Ghannam, N.; Gupta, S. An assessment of the antioxidant and antimicrobial activity of six species of edible Irish seaweeds. Int. Food Res. J. 2010, 17, 205–220. [Google Scholar]
- Dang, T.T.; Bowyer, M.C.; Van Altena, I.A.; Scarlett, C.J. Comparison of chemical profile and antioxidant properties of the brown algae. Int. J. Food Sci. Technol. 2018, 53, 174–181. [Google Scholar] [CrossRef]
- Zubia, M.; Fabre, M.S.; Kerjean, V.; Lann, K.L.; Stiger-Pouvreau, V.; Fauchon, M.; Deslandes, E. Antioxidant and antitumoural activities of some Phaeophyta from Brittany coasts. Food Chem. 2009, 116, 693–701. [Google Scholar] [CrossRef]
- Lobo, V.; Patil, A.; Phatak, A.; Chandra, N. Free radicals, antioxidants and functional foods: Impact on human health. Pharmacogn. Rev. 2010, 4, 118–126. [Google Scholar] [CrossRef] [PubMed]
- Heffernan, N.; Smyth, T.J.; Soler-Villa, A.; Fitzgerald, R.J.; Brunton, N.P. Phenolic content and antioxidant activity of fractions obtained from selected Irish macroalgae species (Laminaria digitata, Fucus serratus, Gracilaria gracilis and Codium fragile). J. Appl. Phycol. 2015, 27, 519–530. [Google Scholar] [CrossRef]
- Wang, T.; Jónsdóttir, R.; Ólafsdóttir, G. Total phenolic compounds, radical scavenging and metal chelation of extracts from Icelandic seaweeds. Food Chem. 2009, 116, 240–248. [Google Scholar] [CrossRef]
- Senevirathne, M.; Kim, S.-H.; Siriwardhana, N.; Ha, J.-H.; Lee, K.-W.; Jeon, Y.-J. Antioxidant potential of Ecklonia cava on reactive oxygen species scavenging, metal chelating, reducing power and lipid peroxidation inhibition. Food Sci. Technol. Int. 2006, 12, 27–38. [Google Scholar] [CrossRef]
- Santoso, J.; Yoshie-Stark, Y.; Suzuki, T. Anti-oxidant activity of methanol extracts from Indonesian seaweeds in an oil emulsion model. Fish. Sci. 2004, 70, 183–188. [Google Scholar] [CrossRef]
- Meir, S.; Kanner, J.; Akiri, B.; Philosoph-Hadas, S. Determination and involvement of aqueous reducing compounds in oxidative defense systems of various senescing leaves. J. Agric. Food Chem. 1995, 43, 1813–1819. [Google Scholar] [CrossRef]
- Ganesan, P.; Kumar, C.S.; Bhaskar, N. Antioxidant properties of methanol extract and its solvent fractions obtained from selected Indian red seaweeds. Bioresour. Technol. 2008, 99, 2717–2723. [Google Scholar] [CrossRef] [PubMed]
- Cruz, J.V.; Freire, P.; Costa, A. Mineral waters characterization in the Azores archipelago (Portugal). J. Volcanol. Geotherm. Res. 2010, 190, 353–364. [Google Scholar] [CrossRef]
- Quartau, R.M. The Insular Shelf of Faial: Morphological and Sedimentary Evolution. Ph.D. Thesis, Universidade de Aveiro, Aveiro, Portugal, 2007. [Google Scholar]
- Instituto Português do Mar e da Atmosfera. Available online: www.ipma.pt (accessed on 1 June 2018).
- Association of Official Analytical Chemists (AOAC). Official Methods of Analysis, 15th ed.; Association of Official Analytical Chemists: Washington, DC, USA, 1990. [Google Scholar]
- Dubois, M.; Gilles, K.A.; Hamilton, J.K.; Rebers, P.A.; Smith, F. Colorimetric method for determination of sugars and related substances. Anal. Chem. 1956, 28, 350–356. [Google Scholar] [CrossRef]
- Association of Official Analytical Chemists (AOAC). Official Methods of Analysis, 16th ed.; Association of Official Analytical Chemists: Washington, DC, USA, 1995. [Google Scholar]
- Folch, J.; Lees, M.; Solam-Stanley, G.H. A simple method for the isolation and purification of total lipid from animal tissue. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar] [PubMed]
- Association of Official Analytical Chemists (AOAC). Official Methods of Analysis, 18th ed.; Association of Official Analytical Chemists: Washington, DC, USA, 2006. [Google Scholar]
- Paiva, L.; Lima, E.; Neto, A.I.; Massimo, M.; Baptista, J. Health-promoting ingredients from four selected Azorean macroalgae. Food Res. Int. 2016, 89, 432–438. [Google Scholar] [CrossRef] [PubMed]
- Brett, J.R.; Groves, T.D. Physiological energetics. In Fish Physiology; Hoar, W.S., Randall, D.J., Eds.; Academic Press: London, UK, 1979; Volume VIII, pp. 279–351. [Google Scholar]
- Waterhouse, A.L. Determination of total phenolics. In Current Protocols in Food Analytical Chemistry; Wrolstad, R.E., Ed.; John Wiley & Sons: New York, NY, USA, 2002. [Google Scholar]
- Chang, C.-C.; Yang, M.-H.; Wen, H.-M.; Chern, J.-C. Estimation of total flavonoid content in propolis by two complementary colorimetric methods. J. Food Drug Anal. 2002, 10, 178–182. [Google Scholar]
- Molyneux, P. The use of the stable free radical diphenylpicrylhydrazyl (DPPH) for estimating antioxidant activity. Songklanakarin J. Sci. Technol. 2004, 26, 211–219. [Google Scholar]
- Corrêa, A.P.; Daroit, D.J.; Coelho, J.; Meira, S.M.; Lopes, F.C.; Risso, P.H.; Brandelli, A. Antioxidant, antihypertensive and antimicrobial properties of ovine milk caseinate hydrolyzed with a microbial protease. J. Sci. Food Agric. 2011, 91, 2247–2254. [Google Scholar] [CrossRef] [PubMed]
- Oyaizu, M. Studies on products of browning reactions: Antioxidative activities of products of browning reaction prepared from glucosamine. Jpn. J. Nutr. 1986, 44, 307–315. [Google Scholar] [CrossRef]
| Composition and Energy Value | F. spiralis Samples | |||
|---|---|---|---|---|
| SMA (Summer) | SMA (Winter) | SMG (Summer) | SMG (Winter) d | |
| Protein b | 4.14 ± 0.10 d | 6.85 ± 0.10 c | 8.25± 0.13 b | 9.71 ± 0.03 a |
| Lipids b | 11.54 ± 0.51 a | 4.40 ± 0.10 b | 5.33 ± 0.31 b | 5.23 ± 0.03 b |
| Carbohydrates b | 17.03 ± 0.59 a | 12.77 ± 0.65 b | 13.45 ± 0.79 b | 17.59 ± 0.27 a |
| Ash b | 22.43 ± 0.45 c | 29.57 ± 0.55 a | 25.40 ± 0.36 b | 22.67 ± 0.47 c |
| TDF b | 52.27 ± 1.53 a | 50.24 ± 1.40 a | 40.47 ± 1.33 b | 40.44 ± 0.61 b |
| SDF b | 24.77 ± 2.07 a | 24.71 ± 1.53 a | 17.77 ± 2.26 bc | 18.75 ± 0.97 b |
| IDF b | 27.50 ± 0.61 a | 25.53 ± 0.77 b | 22.70 ± 1.01 c | 21.69 ± 0.97 c |
| SDF/IDF ratio | 0.90 | 0.97 | 0.78 | 0.86 |
| Energy value c | 8.12 ± 0.25 a | 5.43 ± 0.14 d | 6.22 ± 0.04 c | 7.24 ± 0.05 b |
| Fatty Acids Profile and Fatty Acids Groups | RT of FAME (min) | F. spiralis Samples | |||
|---|---|---|---|---|---|
| SMA (Summer) | SMA (Winter) | SMG (Summer) | SMG (Winter) | ||
| Fatty Acids | |||||
| Myristic, C14:0 | 5.75 | 14.75 ± 1.26 | 13.59 ± 0.98 | 13.73 ± 0.92 | 11.35 ± 0.90 |
| Myristoleic, C14:1 c9 (n5) | 6.271 | tc | tc | 0.54 ± 0.09 | 1.25 ± 0.15 |
| Palmitic, C16:0 | 8.903 | 19.49 ± 1.21 | 23.62 ± 1.43 | 15.34 ± 1.02 | 20.15 ± 1.61 |
| Palmitoleic, C16:1 c9 (n7) | 9.309 | tc | tc | 1.21 ± 0.09 | 1.06 ± 0.08 |
| Heptadecenoic, C17:1 c10 (n7) | 11.087 | tc | 0 | 0.46 ± 0.05 | 1.39 ± 0.10 |
| Stearic, C18:0 | 12.487 | 1.20 ± 0.11 | 3.44 ± 0.39 | 1.57 ± 0.15 | 1.01 ± 0.04 |
| Oleic, C18:1 c9 (n9) | 12.845 | 29.72 ± 2.41 | 21.62 ± 1.11 | 38.04 ± 1.91 | 20.36 ± 1.43 |
| Cis-7-Octadecenoic, C18:1 c7 (n11) | 12.939 | tc | 0 | tc | tc |
| Linolelaidic, C18:2 t9,12 (n6) | 13.666 | 5.27 ± 0.74 | 4.35 ± 0.34 | 5.24 ± 0.39 | 6.15 ± 0.57 |
| Linoleic (LA), C18:2 c9,12 (n6) | 13.759 | tc | tc | 0.45 ± 0.08 | tc |
| Arachidic, C20:0 | 14.233 | tc | 0 | tc | tc |
| γ-Linolenic (GLA), C18:3 c6,9,12 (n6) | 14.868 | 3.20 ± 0.51 | 6.73 ± 0.98 | 3.46 ± 0.80 | 7.04 ± 0.81 |
| Eicosenoic, C20:1 c11 (n9) | 16,196 | tc | tc | 0.57 ± 0.14 | tc |
| α-Linolenic (ALA), C18:3 c9,12,15 (n3) | 16.516 | tc | tc | tc | tc |
| Heneicosanoic, C21:0 | 17.381 | tc | tc | 0.37 ± 0.06 | tc |
| Eicosadienoic, C20:2 c11,14 (n6) | 17.871 | tc | tc | tc | tc |
| Dihomo-γ-linolenic (DHGLA), C20:3 c8,11,14 (n6) | 18.289 | 11.96 ± 1.10 | 9.40 ± 1.00 | 11.87 ± 0.98 | 13.83 ± 1.10 |
| Eicosatrienoic, C20:3 c11,14,17 (n3) | 19.484 | 5.32 ± 0.41 | 7.80 ± 0.33 | 4.33 ± 0.39 | 11.31 ± 0.95 |
| Arachidonic (ARA), C20:4 c5,8,11,14 (n6) | 19.812 | tc | tc | tc | tc |
| Docosadienoic, C22:2 c13,16 (n6) | 20.991 | 1.90 ± 0.22 | 2.20 ± 0.20 | tc | tc |
| Lignoceric, C24:0 | 21.526 | tc | 0 | tc | 0 |
| Eicosapentaenoic (EPA), C20:5 c5,8,11,14,17 (n3) | 23.263 | 1.89 ± 0.29 | 3.44 ± 0.24 | 0.62 ± 0.09 | 1.01 ± 0.07 |
| Nervonic, C24:1 c15 (n9) | 23.579 | 3.82 ± 0.31 | tc | tc | 2.83 ± 0.20 |
| Docosahexaenoic (DHA), C22:6 c4,7,10,13,16,19 (n3) | 35.159 | 1.49 ± 0.23 | 3.80 ± 1.06 | 2.20 ± 0.76 | 1.25 ± 0.09 |
| Fatty Acids Groups | |||||
| Total saturated fatty acids (SFA) | - | 35.44 ± 1.64 | 40.65 ± 1.49 | 31.01 ± 1.83 | 32.51 ± 1.64 |
| Total monounsaturated fatty acids (MUFA) | - | 33.54 ± 1.98 | 21.62 ± 1.11 | 40.82 ± 2.08 | 26.89 ± 1.46 |
| Total polyunsaturated fatty acids (PUFA) | - | 31.03 ± 1.34 | 37.72 ± 1.57 | 28.17 ± 1.08 | 40.59 ± 2.11 |
| Total trans fatty acids (TFA) | - | 5.27 ± 0.74 | 4.35 ± 0.34 | 5.24 ± 0.39 | 6.15 ± 0.57 |
| Total n3 fatty acids | - | 8.70 ± 0.41 | 15.04 ± 1.29 | 7.15 ± 0.70 | 13.57 ± 0.83 |
| Total n6 fatty acids | - | 22.33 ± 1.21 | 22.68 ± 1.12 | 21.02 ± 1.99 | 27.02 ± 1.50 |
| Total n9 fatty acids | - | 33.54 ± 1.98 | 21.62 ± 1.11 | 38.61 ± 1.94 | 23.19 ± 1.28 |
| Ratio n6/n3 | - | 2.57 | 1.51 | 2.94 | 1.99 |
| Ratio h/H | - | 1.89 | 1.59 | 2.37 | 2.14 |
| F. spiralis Samples | Yield (%) | TPC | TFC | ||
|---|---|---|---|---|---|
| mg PE/g DW | mg PE/g DE | mg RE/g DW | mg RE/g DE | ||
| Methanolic Extracts | |||||
| SMA (summer) | 11.90 | 19.21 ± 0.28 a | 172.00 ± 3.00 a | 4.21 ± 0.16 b | 34.50 ± 1.32 b |
| SMA (winter) | 10.30 | 20.42 ± 0.33 a | 187.00 ± 2.65 a | 4.10 ± 0.09 b | 32.00 ± 0.87 b |
| SMG (summer) | 9.70 | 10.95 ± 0.20 c | 113.33 ± 2.08 c | 6.02 ± 0.08 a | 62.00 ± 0.87 a |
| SMG (winter) | 9.90 | 15.18 ± 0.06 b | 153.33 ± 0.58 b | 6.75 ± 0.03 a | 68.17 ± 0.29 a |
| Acetone:Water Extracts (70:30) | |||||
| SMA (summer) | 19.00 | 46.60 ± 1.15 a | 245.67 ± 5.86 a | 3.04 ± 0.10 b | 16.00 ± 0.50 b |
| SMA (winter) | 14.30 | 34.77 ± 0.58 b | 243.33 ± 3.79 a | 4.09 ± 0.11 a | 30.67 ± 0.76 a |
| SMG (summer) | 13.00 | 29.78 ± 0.75 c | 229.33 ± 5.86 b | 1.84 ± 0.10 c | 14.20 ± 0.72 b |
| SMG (winter) | 14.90 | 25.27 ± 0.51 d | 170.67 ± 3.06 c | 2.28 ± 0.11 c | 15.33 ± 0.76 b |
| F. spiralis Samples and BHT | FRSA (EC50 b, mg/mL) | FRAP (EC50 c, mg/mL) |
|---|---|---|
| Methanolic Extracts | ||
| SMA (summer) | 0.076 ± 0.005 c | 0.022 ± 0.006 b |
| SMA (winter) | 0.045 ± 0.004 d | 0.016 ± 0.004 cd |
| SMG (summer) | 0.092 ± 0.005 b | 0.019 ± 0.004 bc |
| SMG (winter) | 0.123 ± 0.01 a | 0.033 ± 0.005 a |
| Acetone:Water Extracts (70:30) | ||
| SMA (summer) | 0.061 ± 0.004 cd | 0.017 ± 0.004 c |
| SMA (winter) | 0.059 ± 0.004 e | 0.020 ± 0.006 b |
| SMG (summer) | 0.064 ± 0.007 b | 0.024 ± 0.005 a |
| SMG (winter) | 0.067 ± 0.006 a | 0.024 ± 0.005 a |
| BHT | 0.062 ± 0.006 c | 0.006 ± 0.001 d |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Paiva, L.; Lima, E.; Neto, A.I.; Baptista, J. Seasonal Variability of the Biochemical Composition and Antioxidant Properties of Fucus spiralis at Two Azorean Islands. Mar. Drugs 2018, 16, 248. https://doi.org/10.3390/md16080248
Paiva L, Lima E, Neto AI, Baptista J. Seasonal Variability of the Biochemical Composition and Antioxidant Properties of Fucus spiralis at Two Azorean Islands. Marine Drugs. 2018; 16(8):248. https://doi.org/10.3390/md16080248
Chicago/Turabian StylePaiva, Lisete, Elisabete Lima, Ana Isabel Neto, and José Baptista. 2018. "Seasonal Variability of the Biochemical Composition and Antioxidant Properties of Fucus spiralis at Two Azorean Islands" Marine Drugs 16, no. 8: 248. https://doi.org/10.3390/md16080248
APA StylePaiva, L., Lima, E., Neto, A. I., & Baptista, J. (2018). Seasonal Variability of the Biochemical Composition and Antioxidant Properties of Fucus spiralis at Two Azorean Islands. Marine Drugs, 16(8), 248. https://doi.org/10.3390/md16080248






