Exopolysaccharides from Marine and Marine Extremophilic Bacteria: Structures, Properties, Ecological Roles and Applications
Abstract
1. Introduction
2. Purification and Characterization Methodologies
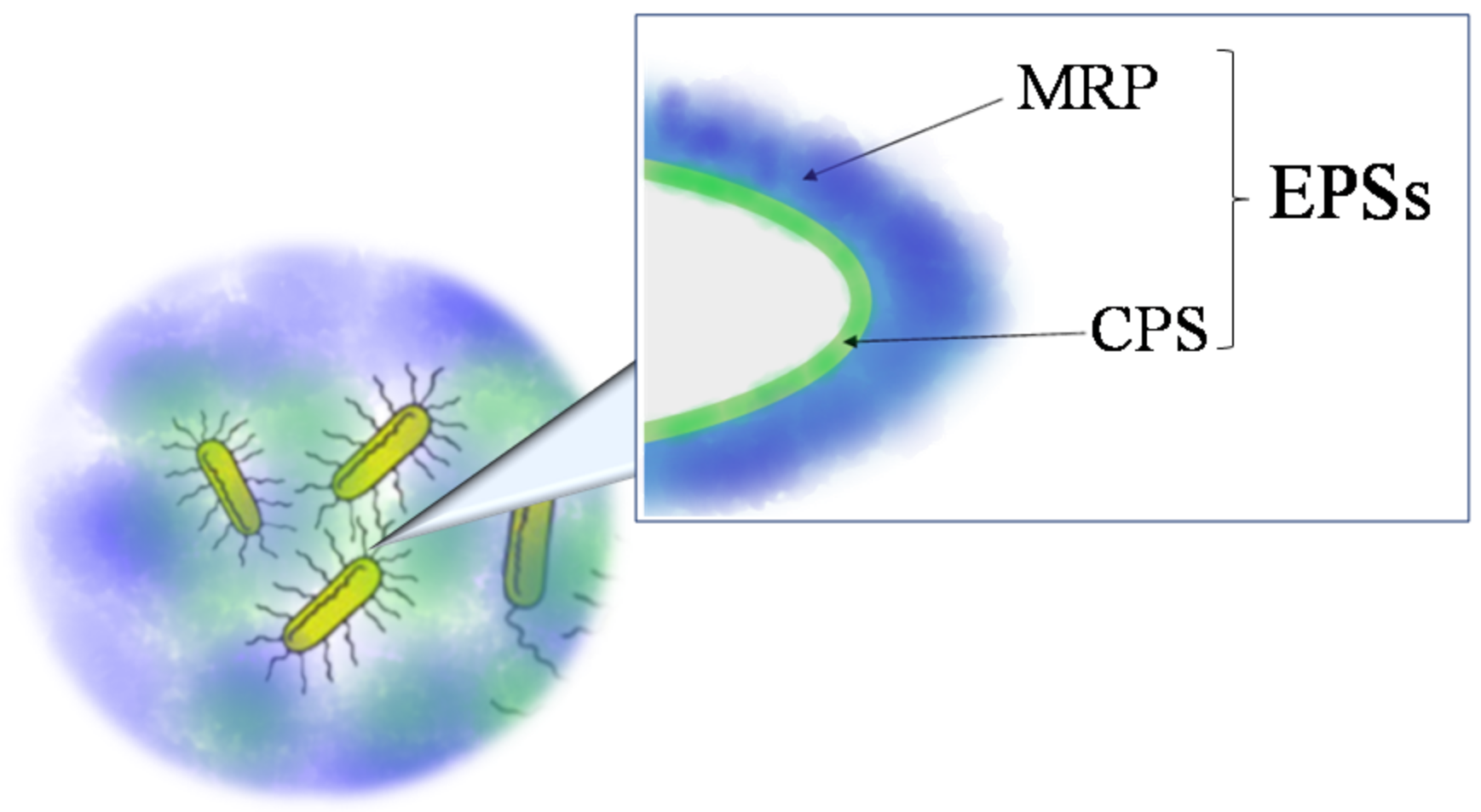
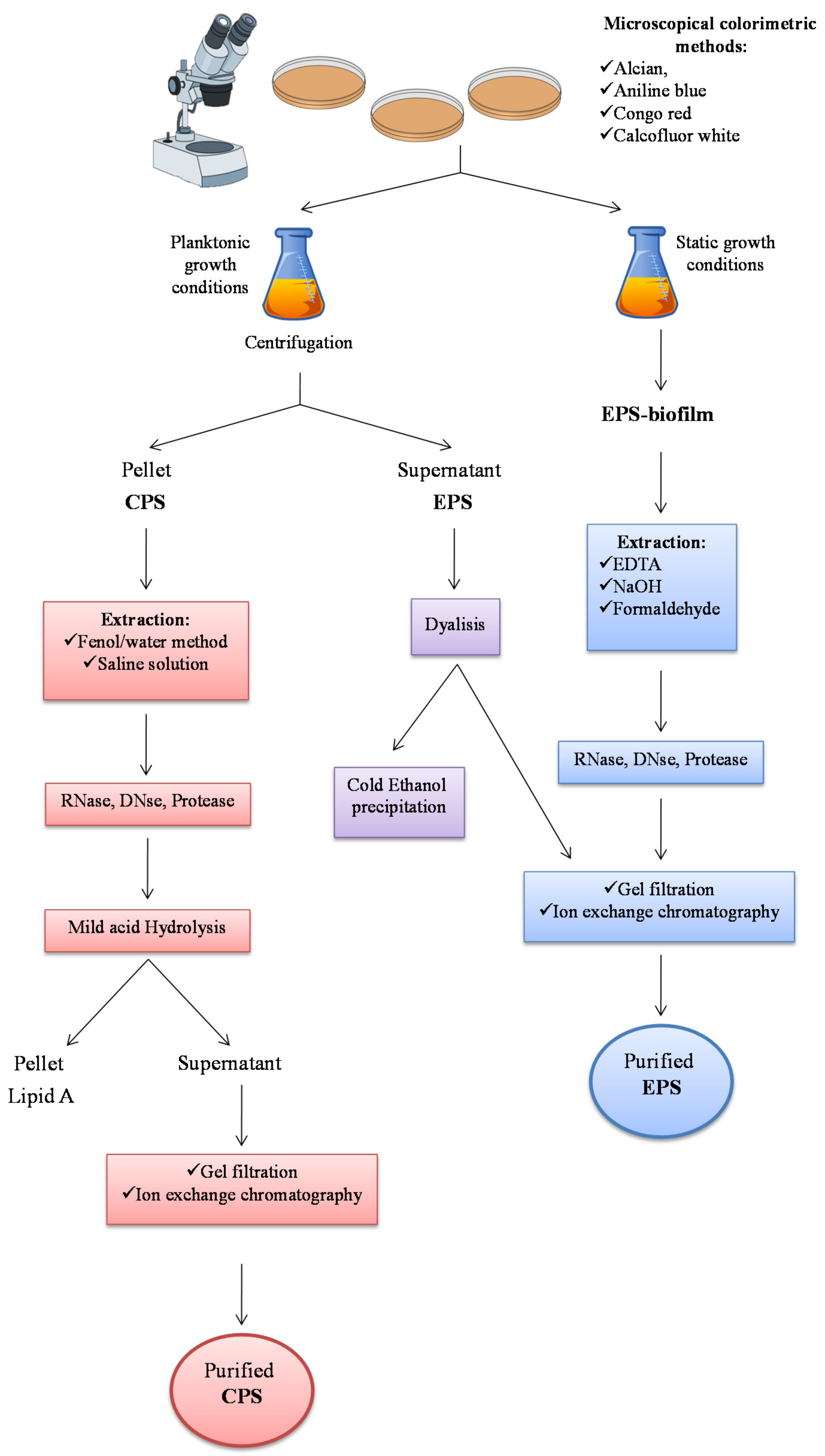
2.1. Extraction from Cell-bound Polysaccharides: Capsular Polysaccharides (CPSs)
2.2. Extraction from Growth Medium: Medium-released Polysaccharides (MRPs)
2.3. Extraction of EPS from Biofilm
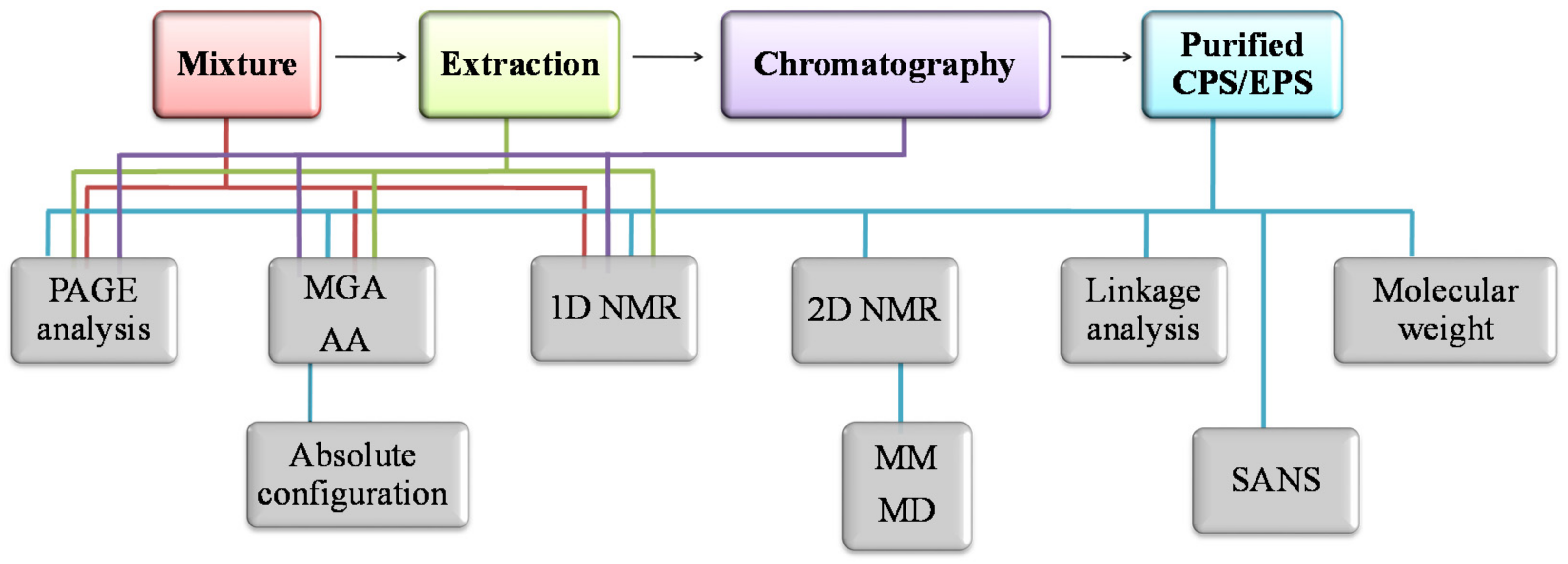
2.4. Methods for Structural Characterization
3. Structure of Exopolysaccharides (EPSs) from Marine Bacteria and Archaebacteria
3.1. Alteromonas
3.2. Bacillus and Geobacillus
3.3. Colwellia
3.4. Halomonas
3.5. Hyphomonas
3.6. Idiomarina
3.7. Pseudoalteromonas
3.8. Pseudomonas
3.9. Rhodococcus
3.10. Shewanella
3.11. Vibrio
3.12. Other EPS-Producing Bacteria
3.13. Marine Archaea
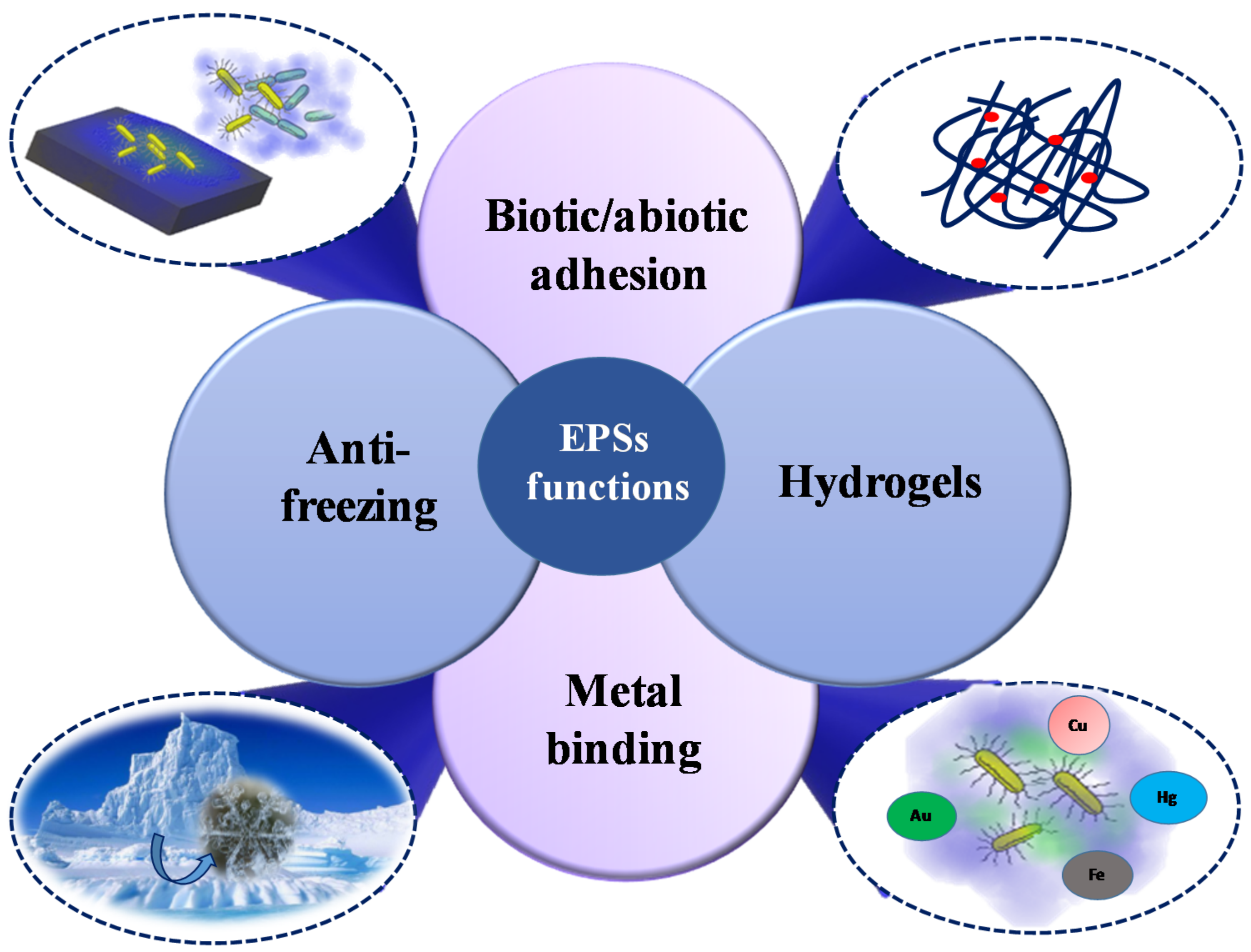
4. Structure-Function Relationships and Ecological Role
5. Biotechnological Applications of Marine EPS
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Berteau, O.; Mulloy, B. Sulfated fucans, fresh perspectives: Structures, functions and biological properties of sulphated fucans and an overview of enzymes active toward this class of polysaccharide. Glycobiology 2003, 13, 29R–40R. [Google Scholar] [CrossRef] [PubMed]
- Barsett, H.; Ebringerovà, A.; Harding, S.E.; Heinze, T.; Hromàdkovà, Z.; Muzzarelli, C.; Muzzarelli, R.A.A.; Paulsen, B.S.; El Seoud, S.A. Polysaccharides I. Structure, Characterisation and Use; Heinze, T., Ed.; Springer: Berlin, Germany, 2005. [Google Scholar]
- Abreu, N.A.; Taga, M.E. Decoding molecular interactions in microbial communities. FEMS Microbiol. Rev. 2016, 40, 648–663. [Google Scholar] [CrossRef] [PubMed]
- Field, C.B.; Behrenfeld, M.J.; Randerson, J.T.; Falkowski, P. Primary production of the biosphere: Integrating terrestrial and oceanic components. Science 1998, 281, 237–240. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.X.; Chin, W.C.; Rodriguez, A.; Hung, C.C.; Santschi, P.H.; Verdugo, P. Amphiphilic exopolymers from Sagittula stellata induce DOM self-assembly and formation of marine microgels. Mar. Chem. 2008, 112, 11–19. [Google Scholar] [CrossRef]
- Verdugo, P. Marine microgels. Annu. Rev. Mar. Sci. 2012, 4, 375–400. [Google Scholar] [CrossRef] [PubMed]
- Pagliano, G.; Ventorino, V.; Panico, A.; Pepe, O. Integrated systems for biopolymers and bioenergy production from organic waste and by-products: A review of microbial processes. Biotechnol. Biofuels 2017, 10, 113–136. [Google Scholar] [CrossRef] [PubMed]
- Decho, A.W. Microbial exopolymer secretions in ocean environments: Their role(s) in food webs and marine processes. Oceanogr. Mar. Biol. Ann. Rev. 1990, 28, 73–153. [Google Scholar]
- Mancuso Nichols, C.A.; Guezennec, J.; Bowman, J.P. Bacterial exopolysaccharides from extreme marine environments with special consideration of the southern ocean, sea ice and deep-sea hydrothermal vents: A review. Mar. Biotechnol. 2005, 7, 253–271. [Google Scholar] [CrossRef] [PubMed]
- Zhenming, C.; Yan, F. Exopolysaccharides from Marine Bacteria. J. Ocean Univ. China 2005, 4, 67–74. [Google Scholar]
- Decho, A.W.; Gutierrez, T. Microbial Extracellular Polymeric Substances (EPSs) in Ocean Systems. Front. Microbiol. 2017, 8, 92. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.S.; Mody, K.; Jha, B. Bacterial exopolysaccharides—A perception. J. Basic Microbiol. 2007, 47, 103–117. [Google Scholar] [CrossRef] [PubMed]
- Poli, A.; Anzelmo, G.; Nicolaus, B. Bacterial Exopolysaccharides from Extreme Marine Habitats: Production, Characterization and Biological Activities. Mar. Drugs 2010, 8, 1779–1802. [Google Scholar] [CrossRef] [PubMed]
- Satpute, S.K.; Banat, I.M.; Dhakephalkar, P.K.; Banpurkar, A.G.; Chopade, B.A. Biosurfactants, bioemulsifiers and exopolysaccharides from marine microorganisms. Biotechnol. Adv. 2010, 28, 436–450. [Google Scholar] [CrossRef] [PubMed]
- Laurienzo, P. Marine Polysaccharides in Pharmaceutical Applications: An Overview. Mar. Drugs 2010, 8, 2435–2465. [Google Scholar] [CrossRef] [PubMed]
- Delbarre-Ladrat, C.; Sinquin, C.; Lebellenger, L.; Zykwinskaand, A.; Colliec-Jouault, S. Exopolysaccharides produced by marine bacteria and their applications as glycosaminoglycan-like molecules. Front. Chem. 2014, 2, 85. [Google Scholar] [CrossRef] [PubMed]
- Biswas, J.; Paul, A.K. Diversity and Production of Extracellular Polysaccharide by Halophilic Microorganisms. Biodiversity Int. J. 2017, 1, 00006. [Google Scholar] [CrossRef]
- Elsakhawy, T.A.; Sherief, F.A.; Abd-EL-Kodoos, R.Y. Marine Microbial Polysaccharides: Environmental Role and Applications (An Overview). Env. Biodiv. Soil Security 2017, 1, 61–70. [Google Scholar] [CrossRef]
- Rühmann, B.; Schmid, J.; Sieber, V. Methods to identify the unexplored diversity of microbial exopolysaccharides. Front. Microbiol. 2015, 6, 565. [Google Scholar] [CrossRef] [PubMed]
- Deming, J.W.; Young, J.N. The role of exopolysaccharides in microbial adaptation to cold habitats. In Psychrophiles: from biodiversity to biotechnology, 2nd ed.; Margesin, R., Ed.; Springer: Cham, Switzerland, 2017; pp. 259–284. [Google Scholar]
- Ma, J.-J.; Yin, R.-C. Primary study on extracellular polysaccharide producing bacteria in different environments. Anhui Daxue Xuebao Ziran Kexueban 2011, 35, 94–100. [Google Scholar]
- Darwish, S.F.; Asfour, H.A.E. Investigation of biofilm forming ability in Staphylococci causing bovine mastitis using phenotypic and genotypic assays. Sci. World J. 2013, 99, 8607–8613. [Google Scholar] [CrossRef]
- Leigh, J.A.; Signer, E.R.; Walker, G.C. Exopolysaccharide-deficient mutants of Rhizobium meliloti that form in effective nodules. Proc. Natl. Acad. Sci. USA 1985, 82, 6231–6235. [Google Scholar] [CrossRef] [PubMed]
- Laemmli, U.K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 1970, 227, 680–685. [Google Scholar] [CrossRef] [PubMed]
- Tsai, C.M.; Frasch, C.E. A sensitive silver stain for detecting lipopolysaccharides in polyacrylamide gels. Anal. Biochem. 1982, 119, 115–119. [Google Scholar] [CrossRef]
- Krueger, R.C.; Schwartz, N.B. An improved method of sequential Alcian Blue and ammoniacal Silver staining of chondroitin sulfate proteoglycan in Polyacrylamide Gels. Anal. Biochem. 1987, 167, 295–300. [Google Scholar] [CrossRef]
- Thornton, D.C.O.; Fejes, E.M.; Di Marco, S.; Clancy, K.M. Measurement of acid polysaccharides in marine and freshwater samples using alcian blue. Limnol. Oceanogr. Methods 2007, 5, 73–87. [Google Scholar] [CrossRef]
- Tallgren, A.H.; Airaksinen, U.; VonWeissenberg, R.; Ojamo, H.; Kuusisto, J.; Leisola, M. Exopolysaccharide-producing bacteria from sugar beets. Appl. Environ. Microbiol. 1999, 65, 862–864. [Google Scholar] [PubMed]
- Ortega-Morales, B.O.; Santiago-García, J.L.; Chan-Bacab, M.J.; Moppert, X.; Miranda-Tello, E.; Fardeau, M.L.; Carrero, J.C.; Bartolo-Pèrez, P.; Valadèz-Gonzàlez, A.; Guezennec, J. Characterization of extracellular polymers synthesized by tropical intertidal biofilm bacteria. J. Appl. Microbiol. 2007, 102, 254–264. [Google Scholar] [CrossRef] [PubMed]
- Dubois, M.; Gilles, K.A.; Hamilton, J.K.; Rebers, P.A.; Smith, F. Colorimetric method for determination of sugars and related substances. Anal. Chem. 1956, 28, 350–356. [Google Scholar] [CrossRef]
- Blumenkrantz, N.; Asboe-Hansen, G. New method for quantitative determination of uronic acids. Anal. Biochem. 1973, 54, 484–489. [Google Scholar] [CrossRef]
- Bitter, T.; Muir, H.M. A modified uronic acid carbazole reaction. Anal. Biochem. 1962, 4, 330–334. [Google Scholar] [CrossRef]
- Sheng, G.-P.; Yu, H.-Q.; Li, X.-Y. Extracellular polymeric substances (EPS) of microbial aggregates in biological wastewater treatment systems: A review. Biotechnol. Adv. 2010, 28, 882–894. [Google Scholar] [CrossRef] [PubMed]
- Stukalov, O.; Korenevsky, A.; Beveridge, T.J.; Dutcher, J.R. Use of atomic force microscopy and transmission electron microscopy for correlative studies of bacterial capsules. Appl. Environ. Microbiol. 2008, 74, 5457–5465. [Google Scholar] [CrossRef] [PubMed]
- Beveridge, T.J.; Lawrence, J.R.; Murray, R.G.E. Sampling and Staining for Light Microscopy. In Methods for General and Molecular Microbiology, 3rd ed.; Reddy, C.A., Beveridge, T.J., Breznak, J.A., Marzluf, G.A., Schmidt, T.M., Snyder, L.R., Eds.; ASM Press: Washington, DC, USA, 2007. [Google Scholar] [CrossRef]
- Reddy, C.P.; Hayat, U.; Abeygunawardana, C.; Fox, C.; Wright, A.C.; Maneval, D.R.; Bush, A.C.; Morris, J.C. Purification and determination of the structure of capsular polysaccharide of Vibrio vulnificus M06–24. J. Bacteriol. 1992, 174, 2620–2630. [Google Scholar] [CrossRef] [PubMed]
- Carillo, S.; Casillo, A.; Pieretti, G.; Parrilli, E.; Sannino, F.; Bayer-Giraldi, M.; Cosconati, S.; Novellino, E.; Ewert, M.; Deming, J.W.; et al. A unique capsular polysaccharide structure from the psychrophilic marine bacterium Colwellia psychrerythraea 34H that mimics antifreeze (Glyco)proteins. J. Am. Chem. Soc. 2015, 137, 179–189. [Google Scholar] [CrossRef] [PubMed]
- Vinogradov, E.; Nossova, L.; Korenevsky, A.; Beveridge, T.J. The structure of the capsular polysaccharide of Shewanella oneidensis strain MR-4. Carbohydr. Res. 2005, 340, 1750–1753. [Google Scholar] [CrossRef] [PubMed]
- Kwon, K.J.; Park, K.J.; Kim, J.D.; Kong, J.Y.; Kong, J.Y. Isolation of two different polysaccharides from halophilic Zoogloea sp. Biotechnol. Lett. 1994, 16, 783–788. [Google Scholar] [CrossRef]
- Kim, S.J.; Kim, B.G.; Parka, H.U.; Yim, J.H. Cryoprotective properties and preliminary characterization of exopolysaccharide (P-Arcpo 15) produced by the Arctic bacterium Pseudoalteromonas elyakovii Arcpo 15. Prep. Biochem. Biotechnol. 2016, 46, 261–266. [Google Scholar] [CrossRef]
- Lembre, P.; Lorentz, C.; Di Martino, P. Exopolysaccharides of the Biofilm Matrix: A Complex Biophysical World. In The Complex World of Polysaccharides; Karunaratne, D.N., Ed.; InTech: London, UK, 2012; pp. 371–392. [Google Scholar]
- Oliveira, R.; Marques, F.; Azeredo, J. Purification of polysaccharides from a biofilm matrix by selective precipitation of proteins. Biotechnol. Tech. 1999, 13, 391–393. [Google Scholar] [CrossRef]
- Boualam, M.; Quiles, F.; Mathieu, L.; Block, J.C. Monitoring the effect of organic matter on biofilm growth in low nutritive waters by ATR-FT-IR spectroscopy. Biofouling 2002, 18, 73–81. [Google Scholar] [CrossRef]
- Liu, H.; Fang, H.H.P. Extraction of extracellular polymeric substances (EPS) of sludges. J. Biotechnol. 2002, 95, 249–256. [Google Scholar] [CrossRef]
- Lin, M.H.; Yang, Y.L.; Chen, Y.P.; Hua, K.F.; Lu, C.P.; Sheu, F.; Lin, G.H.; Tsay, S.S.; Liang, S.M.; Wu, S.H. A novel exopolysaccharide from the biofilm of Thermus aquaticus YT-1 induces the Immune response through toll-like receptor 2. J. Biol. Chem. 2011, 286, 36–45. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Lu, H.; Sprinkle, A.; Parsek, M.R.; Wozniak, D.J. Pseudomonas aeruginosa Psl Is a Galactose- and Mannose-Rich Exopolysaccharide. J. Bacteriol. 2007, 189, 8353–8356. [Google Scholar] [CrossRef] [PubMed]
- Abazia, C.; Ferrara, R.; Corsaro, M.M.; Barone, G.; Coccoli, P.; Parrilli, G. Simultaneous Gas-chromatographic Evaluation of Rhamnose, Lactulose and Sucrose for testing Gastrointestinal Permeability. Clin. Chim. Acta 2003, 338, 25–32. [Google Scholar] [CrossRef] [PubMed]
- Fresno, S.; Jiménez, N.; Canals, R.; Merino, S.; Corsaro, M.M.; Lanzetta, R.; Parrilli, M.; Pieretti, G.; Regué, M.; Tomàs, J.M. A Second Galacturonic Acid Transferase is Required for Core Lipopolysaccharide Biosynthesis and Complete Capsule Association to the Cell Surface in Klebsiella pneumoniae. J. Bacteriol. 2007, 189, 1128–1137. [Google Scholar] [CrossRef] [PubMed]
- Lönngren, J.; Svensson, S. Mass spectrometry in structural analysis of natiral carbohydrates. Adv. Carbohydr. Chem. Biochem. 1974, 29, 41–106. [Google Scholar]
- Leontein, K.; Lindberg, B.; Lönngren, J. Assignment of absolute configuration of sugars by g.l.c. of their acetylated glycosides from chiral alcohols. Carbohydr. Res. 1978, 62, 359–362. [Google Scholar] [CrossRef]
- Ciucanu, I.; Kerek, F. A simple and rapid method for the permethylation of carbohydrates. Carbohydr. Res. 1984, 131, 209–217. [Google Scholar] [CrossRef]
- Perlin, A.S.; Casu, B. Spectroscopic methods. In Polysaccharides; Aspinall, G.O., Ed.; Academic Press: London, UK, 1982; pp. 133–193. [Google Scholar]
- Agrawal, P.K. NMR Spectroscopy in the structural elucidation of oligosaccharides and glycosides. Phytochemistry 1992, 31, 3307–3330. [Google Scholar] [CrossRef]
- Duus, J.Ø.; Gotfredsen, C.H.; Bock, K. Carbohydrate Structural Determination by NMR Spectroscopy: Modern Methods and Limitations. Chem. Rev. 2000, 100, 4589–4614. [Google Scholar] [CrossRef] [PubMed]
- Bubb, W.A. NMR spectroscopy in the study of carbohydrates: Characterizing the structural complexity. Concepts Magn. Reson. 2003, 19, 1–19. [Google Scholar] [CrossRef]
- Larsen, F.H.; Engelsen, S.B. Insight into the Functionality of Microbial Exopolysaccharides by NMR Spectroscopy and Molecular Modeling. Front. Microbiol. 2015, 6, 1374. [Google Scholar] [CrossRef] [PubMed]
- Allen, T.W.; Kujucak, S.; Chung, S.H. Molecular Dynamics Study of the KcsA Potassium Channel. Biophys. J. 1999, 77, 2502–2516. [Google Scholar] [CrossRef]
- Stevens, E.S. Circular Dichroism and Conformational Analysis of Biomolecules; Fasman, G.D., Ed.; Plenum Press: Raleigh, NC, USA, 1996; pp. 501–530. [Google Scholar]
- Masuelli, M.; Renard, D. Advances in Physicochemical Properties of Biopolymers; Bentham E-Books: Sharjah, United Arab Emirates, 2017; pp. 75–78. [Google Scholar]
- Baumann, L.; Baumann, P.; Mandel, M.; Allen, R.D. Taxonomy of aerobic marine Eubacteria. J. Bacteriol. 1972, 110, 402–429. [Google Scholar] [PubMed]
- López-Pérez, M.; Rodriguez-Valera, F. The Family Alteromonadaceae. In The Prokaryotes; Rosenberg, E., DeLong, E.F., Lory, S., Stackebrandt, E., Thompson, F., Eds.; Springer: Berlin, Germany, 2014. [Google Scholar]
- Cambon-Bonavita, M.-A.; Rague´ne`s, G.; Jean, J.; Vincent, P.; Guezennec, J. A novel polymer produced by a bacterium isolated from a deep-sea hydrothermal vent polychaete annelid. J. Appl. Microbiol. 2002, 93, 310–315. [Google Scholar] [CrossRef] [PubMed]
- Le Costaouëc, T.; Cérantola, S.; Ropartz, D.; Ratiskol, J.; Sinquin, C.; Colliec-Jouault, S.; Boisset, C. Structural data on a bacterial exopolysaccharide produced by a deep-sea Alteromonas macleodii strain. Carbohydr. Polym. 2012, 90, 49–59. [Google Scholar] [CrossRef] [PubMed]
- Vincent, P.; Pignet, P.; Talmont, F.; Bozzi, L.; Fournet, B.; Guezennec, J.; Jeanthon, C.; Prieur, D. Production and characterization of an exopolysaccharide excreted by a deep-sea hydrothermal vent bacterium isolated from the polychaete annelid Alvinella pompejana. Appl. Environ. Microbiol. 1994, 60, 4134–4141. [Google Scholar] [PubMed]
- Raguenes, G.; Pignet, P.; Gauthier, G.; Peres, A.; Christen, R.; Rougeaux, H.; Barbier, G.; Guezennec, J. Description of a new polymer-secreting bacterium from a deep-sea hydrothermal vent, Alteromonas macleodii subsp. fijiensis and preliminary characterization of the polymer. Appl. Environ. Microbiol. 1996, 62, 67–73. [Google Scholar] [PubMed]
- Rougeaux, H.; Talaga, P.; Carlson, R.W.; Guezennec, J. Structural studies of an exopolysaccharide produced by Alteromonas macleodii subsp. Fijiensis originating from a deep-sea hydrothermal vent. Carbohydr. Res. 1998, 312, 53–59. [Google Scholar] [CrossRef]
- Zhang, Z.; Chen, Y.; Wang, R.; Cai, R.; Fu, Y.; Jiao, N. The Fate of marine bacterial exopolysaccharide in natural marine microbial communities. PLoS ONE 2015, 10, e0142690. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Cai, R.; Zhang, W.; Fu, Y.; Jiao, N. A Novel Exopolysaccharide with Metal Adsorption Capacity Produced by a Marine Bacterium Alteromonas sp. JL2810. Mar. Drugs 2017, 15, 175. [Google Scholar] [CrossRef] [PubMed]
- Raguénès, G.H.C.; Peres, A.; Ruimy, R.; Pignet, P.; Christen, R.; Loaec, M.; Rougeaux, H.; Barbier, G.; Guezennec, J.G. Alteromonas infernus sp. nov., a new polysaccharide-producing bacterium isolated from a deep-sea hydrothermal vent. J. Appl. Microbiol. 1997, 82, 422–430. [Google Scholar] [CrossRef] [PubMed]
- Roger, O.; Kervarec, N.; Ratiskol, J.; Colliec-Jouault, S.; Chevolot, L. Structural studies of the main exopolysaccharide produced by the deep-sea bacterium Alteromonas infernus. Carbohydr. Res. 2004, 339, 2371–2380. [Google Scholar] [CrossRef] [PubMed]
- Mata, J.A.; Be´jar, V.; Bressollier, P.; Tallon, R.; Urdaci, M.C.; Quesada, E.; Llamas, I. Characterization of exopolysaccharides produced by three moderately halophilic bacteria belonging to the family Alteromonadaceae. J. Appl. Microbiol. 2008, 105, 521–528. [Google Scholar] [CrossRef] [PubMed]
- Maugeri, T.L.; Gugliandolo, C.; Caccamo, D.; Panico, A.; Lama, L.; Gambacorta, A.; Nicolaus, B. A halophilic thermotolerant Bacillus isolated from a marine hot spring able to produce a new exopolysaccharide. Biotechnol. Lett. 2002, 24, 515–519. [Google Scholar] [CrossRef]
- Arena, A.; Maugeri, T.; Pavone, B.; Iannello, D.; Gugliandolo, C.; Bisignano, G. Antiviral and immunoregulatory effect of a novel exopolysaccharide from a marine thermotolerant Bacillus licheniformis. Int. Immunopharmacol. 2006, 6, 8–13. [Google Scholar] [CrossRef] [PubMed]
- Nicolaus, B.; Panico, A.; Manca, A.C.; Lama, L.; Gambacorta, A.; Maugeri, T.; Guagliandolo, C.; Caccamo, D. A thermophilic Bacillus isolated from an Eolian shallow hydrothermal vent, able to produce exopolysaccharides. Syst. Appl. Microbiol. 2000, 23, 426–432. [Google Scholar] [CrossRef]
- Mao, R.; Tang, J.; Swanson, B.G. Water holding capacity and microstructure of gellan gums. Carbohydr. Polym. 2001, 46, 365–371. [Google Scholar] [CrossRef]
- Kumar, C.G.; Joo, H.-S.; Choi, J.-W.; Koo, Y.-M.; Chang, C.-S. Purification and characterization of an extracellular polysaccharide from haloalkalophilic Bacillus sp. I-450. Enzyme Microb. Technol. 2004, 34, 673–681. [Google Scholar] [CrossRef]
- Lelchat, F.; Cérantola, S.; Brandily, C.; Colliec-Jouault, S.; Baudoux, A.C.; Ojima, T.; Boisset, C. The marine bacteria Cobetia marina DSMZ 4741 synthesizes an unexpected K-antigen-like exopolysaccharide. Carbohydr. Polym. 2015, 124, 347–356. [Google Scholar] [CrossRef] [PubMed]
- Marx, J.G.; Carpenter, S.D.; Deming, J.W. Production of cryoprotectant extracellular polysaccharide substances (EPS) by the marine psychrophilic bacterium Colwellia psychrerythraea strain 34H under extreme conditions. Can. J. Microbiol. 2009, 55, 63–72. [Google Scholar] [CrossRef] [PubMed]
- Casillo, A.; Parrilli, E.; Sannino, F.; Mitchell, D.E.; Gibson, M.I.; Marino, G.; Lanzetta, R.; Parrilli, M.; Cosconati, S.; Novellino, E.; et al. Structure-activity relationship of the exopolysaccharide from a psychrophilic bacterium: A strategy for cryoprotection. Carbohydr. Polym. 2017, 156, 364–371. [Google Scholar] [CrossRef] [PubMed]
- Casillo, A.; Ståhle, J.; Parrilli, E.; Sannino, F.; Mitchell, D.E.; Pieretti, G.; Gibson, M.I.; Marino, G.; Lanzetta, R.; Parrilli, M.; et al. Structural characterization of an all-aminosugar-containing capsular polysaccharide from Colwellia psychrerythraea 34H. Antonie Van Leeuwenhoek 2017, 110, 1377–1387. [Google Scholar] [CrossRef] [PubMed]
- Umezawa, H.; Okami, Y.; Kurasawa, S.; Ohnuki, T.; Ishizuka, M.; Takeuchi, T.U.; Shiio, T.; Yugari, Y. Marinactan, antitumor polysaccharide produced by marine bacteria. J. Antibiot. 1983, 471, 471–477. [Google Scholar] [CrossRef]
- Schiano Moriello, V.; Lama, L.; Poli, A.; Gugliandolo, C.; Maugeri, T.L.; Gambacorta, A.; Nicolaus, B. Production of exopolysaccharides from a thermophilic microorganism isolated from a marine hot spring in flegrean areas. J. Ind. Microbiol. Biotechnol. 2003, 30, 95–101. [Google Scholar] [CrossRef] [PubMed]
- Kambourova, M.; Mandeva, R.; Dimova, D.; Poli, A.; Nicolaus, B.; Tommonaro, G. Production and characterization of a microbial glucan, synthesized by Geobacillus tepidamans V264 isolated from Bulgarian hot spring. Carbohydr. Polym. 2009, 77, 338–343. [Google Scholar] [CrossRef]
- Arena, A.; Gugliandolo, C.; Stassi, G.; Pavone, B.; Iannello, D.; Bisignano, G.; Maugeri, T.L. An exopolysaccharide produced by Geobacillus thermodenitrificans strain B3–72: Antiviral activity on immunocompetent cells. Immunol. Lett. 2009, 123, 132–137. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.K.; Chun, J.; Moon, E.Y.; Ko, S.H.; Lee, D.S.; Lee, H.S.; Bae, K.S. Hahella chejuensis gen. nov., sp. nov., an extracellular-polysaccharide-producing marine bacterium. Int. J. Syst. Evol. Microbiol. 2001, 51, 661–666. [Google Scholar] [CrossRef] [PubMed]
- Yim, J.H.; Kim, S.J.; Aan, S.H.; Lee, H.K. Physicochemical and rheological properties of a novel emulsifier, EPS-R, produced by the marine bacterium Hahella chejuensis. Biotechnol. Bioproc. E 2004, 9, 405. [Google Scholar] [CrossRef]
- Bejar, V.; Calvo, C.; Moliz, J.; Diaz-Martinez, F.; Quesada, E. Effect of growth conditions on the rheological properties and chemical composition of Volcaniella eurihalina exopolysaccharide. Appl. Biochem. Biotechnol. 1996, 59, 77. [Google Scholar] [CrossRef]
- Martínez-Checa, F.; Toledo, F.L.; El Mabrouki, K.; Quesada, E.; Calvo, C. Characteristics of bioemulsifier V2–7 synthesized in culture media added of hydrocarbons: Chemical composition, emulsifying activity and rheological properties. Bioresour. Technol. 2007, 98, 3130–3135. [Google Scholar] [CrossRef] [PubMed]
- Bouchotroch, S.; Quesada, E.; Del Moral, A.; Llamas, I.; Bejar, V. Halomonas maura sp. nov., a novel moderately halophilic, exopolysaccharide-producing bacterium. Int. J. Syst. Evol. Microbiol. 2001, 51, 1625–1632. [Google Scholar] [CrossRef] [PubMed]
- Arias, S.; Del Moral, A.; Ferrer, M.R.; Tallon, R.; Quesada, E.; Béjar, V. Mauran, an exopolysaccharide produced by the halophilic bacterium Halomonas maura, with a novel composition and interesting properties for biotechnology. Extremophiles 2003, 7, 319. [Google Scholar] [CrossRef] [PubMed]
- Mabinya, L.V.; Cosa, S.; Mkwetshana, N.; Okoh, A.I. Halomonas sp. OKOH—A marine bacterium isolated from the bottom sediment of Algoa bay—Produces a polysaccharide bioflocculant: Partial characterization and biochemical analysis of its properties. Molecules 2011, 16, 4358–4370. [Google Scholar] [CrossRef] [PubMed]
- Poli, A.; Kazak, H.; Gürleyendağ, H.; Tommonaro, G.; Pieretti, G.; ToksoyÖner, E.; Nicolaus, B. High level synthesis of levan by a novel Halomonas species growing on defined media. Carbohydr. Polym. 2009, 78, 651–657. [Google Scholar] [CrossRef]
- Küçükaşik, F.; Kazak, H.; Güney, D.; Finore, I.; Poli, A.; Yenigün, O.; Nicolaus, N.; Öner, E.T. Molasses as fermentation substrate for levan production by Halomonas sp. Appl. Microbiol. Biotechnol. 2011, 89, 1729–1740. [Google Scholar] [CrossRef] [PubMed]
- Poli, A.; Schiano Moriello, V.; Esposito, E.; Lama, L.; Gambacorta, A.; Nicolaus, B. Exopolysaccharide production by a new Halomonas strain CRSS isolated from saline lake Cape Russell in Antarctica growing on complex and defined media. Biotechnol. Lett. 2004, 26, 1635–1638. [Google Scholar] [CrossRef] [PubMed]
- Quintero, E.J.; Weiner, R.M. Evidence for the adhesive function of the exopolysaccharide of Hyphomonas strain MHS-3 in its attachment to surfaces. Appl. Environ. Microbiol. 1995, 1897–1903. [Google Scholar]
- Quintero, E.J.; Weiner, R.M. Physical and chemical characterization of the polysaccharide capsule of the marine bacterium, Hyphomonas strain MHS-3. J. Ind. Microbiol. Biotechnol. 1995, 15, 347–351. [Google Scholar] [CrossRef]
- Quintero, E.J.; Langille, S.E.; Weiner, R.M. The polar polysaccharide capsule of Hyphomonas adhaerens MHS-3 has a strong affinity for gold. J. Ind. Microbiol. Biotechnol. 2001, 27, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Langille, S.E.; Weiner, R.M. Spatial and temporal deposition of Hyphomonas strain vp-6 capsules involved in biofilm formation. Appl. Environ. Microbiol. 1998, 2906–2913. [Google Scholar]
- Sun, M.L.; Zhao, F.; Shi, M.; Zhang, X.Y.; Zhou, B.C.; Zhang, Y.Z.; Chen, X.L. Characterization and biotechnological potential analysis of a new exopolysaccharide from the Arctic marine bacterium Polaribacter sp. SM1127. Sci. Rep. 2015, 5, 18435. [Google Scholar] [CrossRef] [PubMed]
- Rougeaux, H.; Guezennec, J.; Carlson, R.W.; Kervarec, N.; Pichon, R.; Talaga, P. Structural determination of the exopolysaccharide of Pseudoalteromonas strain HYD 721 isolated from a deep-sea hydrothermal vent. Carbohydr. Res. 1999, 315, 273–285. [Google Scholar] [CrossRef]
- Gutierrez, T.; Shimmield, T.; Haidon, C.; Black, K.; Green, D.H. Emulsifying and metal ion binding activity of a glycoprotein exopolymer produced by Pseudoalteromonas sp. Strain TG12. Appl. Environ. Microbiol. 2008, 74, 4867–4876. [Google Scholar] [CrossRef] [PubMed]
- Brian-Jaisson, F.; Molmeret, M.; Fahs, A.; Dombrowsky, L.G.; Culioli, G.; Blache, Y.; Cérantola, S.; Ortalo-Magné, A. Characterization and anti-biofilm activity of extracellular polymeric substances produced by the marine biofilm-forming bacterium Pseudoalteromonas ulvae strain TC14. Biofouling 2016, 32, 547–560. [Google Scholar] [CrossRef] [PubMed]
- Saravanan, P.; Jayachandran, S. Preliminary characterization of exopolysaccharides produced by a marine biofilm-forming bacterium Pseudoalteromonas ruthenica (SBT 033). Lett. Appl. Microbiol. 2008, 46, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Roca, C.; Lehmann, M.; Torres, C.A.V.; Baptista, S.; Gaudêncio, S.P.; Freitas, F.; Reis, M.A.M. Exopolysaccharide production by a marine Pseudoalteromonas sp. strain isolated from Madeira Archipelago ocean sediments. New Biotechnol. 2016, 33, 460–466. [Google Scholar] [CrossRef] [PubMed]
- Corsaro, M.M.; Lanzetta, R.; Parrilli, E.; Parrilli, M.; Tutino, M.L.; Ummarino, S. Influence of growth temperature on lipid and phosphate contents of surface polysaccharides from the Antarctic bacterium Pseudoalteromonas haloplanktis TAC 125. J. Bacteriol. 2004, 186, 29–34. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.B.; Chen, X.L.; He, H.L.; Zhang, X.Y.; Xie, B.B.; Yu, Y.; Chen, B.; Zhou, B.C.; Zhang, Y.Z. Structure and ecological roles of a novel exopolysaccharide from the Arctic Sea ice bacterium Pseudoalteromonas sp. strain SM20310. Appl. Environ. Microbiol. 2013, 79, 224–230. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.J.; Yim, H.J. Cryoprotective properties of exopolysaccharide (P-21653) produced by the Antarctic bacterium, Pseudoalteromonas arctica KOPRI 21653. J. Microbiol. 2007, 45, 510–514. [Google Scholar] [PubMed]
- Mancuso Nichols, C.A.; Garon, S.; Bowman, J.P.; Raguénès, G.; Guézennec, J. Production of exopolysaccharides by Antarctic marine bacterial isolates. J. Appl. Microbiol. 2004, 96, 1057–1066. [Google Scholar] [CrossRef] [PubMed]
- Qin, G.; Zhu, L.; Chen, X.; Wang, P.G.; Zhang, Y. Structural characterization and ecological roles of a novel exopolysaccharide from the deep-sea psychrotolerant bacterium Pseudoalteromonas sp. SM9913. Microbiology 2007, 153, 1566–1572. [Google Scholar] [CrossRef] [PubMed]
- Caruso, C.; Rizzo, C.; Mangano, S.; Poli, A.; Di Donato, P.; Nicolaus, B.; Di Marco, G.; Michaud, L.; Lo Giudice, A. Extracellular polymeric substances with metal adsorption capacity produced by Pseudoalteromonas sp. MER144 from Antarctic seawater. Environ. Sci. Pollut. Res. 2017, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Christensen, B.E.; Kjosbakken, J.; Smidsrød, O.L. Partial chemical and physical characterization of two extracellular polysaccharides produced by marine, periphytic Pseudomonas sp. strain NCMB 2021. Appl. Environ. Microbiol. 1985, 50, 837–845. [Google Scholar] [PubMed]
- Wrangstadh, M.; Conway, P.L.; Kjelleberg, S. The production and release of an extracellular polysaccharide during starvation of a marine Pseudomonas sp. and the effect thereof on adhesion. Arch. Microbiol. 1986, 145, 220. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, M.; Yamori, T.; Naitoh, M.; Okutani, K. Structural revision of sulfated polysaccharide b-1 isolated from a marine Pseudomonas species and its cytotoxic activity against human cancer cell lines. Mar. Biotechnol. 2003, 5, 13. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Liu, G.; Jin, W.; Xiu, P.; Sun, C. Antibiofilm and anti-Infection of a marine bacterial exopolysaccharide against Pseudomonas aeruginosa. Front. Microbiol. 2016, 7. [Google Scholar] [CrossRef] [PubMed]
- Carrión, O.; Delgado, L.; Mercade, E. New emulsifying and cryoprotective exopolysaccharide from Antarctic Pseudomonas sp. ID1. Carbohydr. Polym. 2015, 117, 1028–1034. [Google Scholar] [CrossRef] [PubMed]
- Aizawa, T.; Neilan, A.B.; Couperwhite, I.; Urai, M.; Anzai, H.; Iwabuchi, N.; Nakajima, M.; Sunairi, M. Relationship between extracellular polysaccharide and benzene tolerance of Rhodococcus sp. 33. Actinomycetologica 2005, 19, 1–6. [Google Scholar] [CrossRef]
- Urai, M.; Aizawa, T.; Anzai, H.; Ogihara, J.; Iwabuchi, N.; Neilan, B.; Couperwhite, I.; Nakajima, M.; Sunairi, M. Structural analysis of an extracellular polysaccharide produced by a benzene tolerant bacterium, Rhodococcus sp. 33. Carbohydr. Res. 2006, 341, 616–623. [Google Scholar] [CrossRef] [PubMed]
- Urai, M.; Anzai, H.; Ogihara, J.; Iwabuchi, N.; Harayama, S.; Sunairi, M.; Nakajima, M. Structural analysis of an acidic, fatty acid ester-bonded extracellular polysaccharide produced by a pristane-assimilating marine bacterium, Rhodococcus erythropolis PR4. Carbohydr. Res. 2007, 342, 933–942. [Google Scholar] [CrossRef] [PubMed]
- Llamas, I.; Mata, J.A.; Tallon, R.; Bressollier, P.; Urdaci, M.C.; Quesada, E.; Béjar, V. Characterization of the exopolysaccharide produced by Salipiger mucosus A3T, a halophilic species belonging to the Alphaproteobacteria, isolated on the Spanish Mediterranean saboard. Mar. Drugs 2010, 8, 2240–2251. [Google Scholar] [CrossRef] [PubMed]
- Sledjeski, D.D.; Weiner, R.M. Production and characterization of monoclonal antibodies specific for Shewanella colwelliana exopolysaccharide. Appl. Environ. Microbiol. 1993, 59, 1565–1572. [Google Scholar] [PubMed]
- Rougeaux, H.; Kervarec, N.; Pichon, R.; Guezennec, J. Structure of the exopolysaccharide of Vibrio diabolicus isolated from a deep-sea hydrothermal vent. Carbohydr. Res. 1999, 322, 40–45. [Google Scholar] [CrossRef]
- Zanchetta, P.; Lagarde, N.; Guezennec, J. New bone-healing material: A hyaluronic acid-like bacterial exopolysaccharide. Calcif. Tissue Int. 2003, 72, 74–79. [Google Scholar] [CrossRef] [PubMed]
- Muralidharan, Y.; Jayachandran, S. Physicochemical analyses of the exopolysaccharides produced by a marine biofouling bacterium, Vibrio alginolyticus. Process Biochem. 2003, 38, 841–847. [Google Scholar] [CrossRef]
- Drouillard, S.; Jeacomine, I.; Buon, L.; Boisset, C.; Courtois, A.; Thollas, B.; Morvan, P.Y.; Vallée, R.; Helbert, W. Structure of an amino acid-decorated exopolysaccharide secreted by a Vibrio alginolyticus strain. Mar. Drugs 2015, 13, 6723–6739. [Google Scholar] [CrossRef] [PubMed]
- Bramhachari, P.V.; Dubey, S.K. Isolation and characterization of exopolysaccharide produced by Vibrio harveyi strain VB23. Lett. Appl. Microbio.l 2006, 43, 571. [Google Scholar] [CrossRef] [PubMed]
- Bramhachari, P.V.; Kavi Kishor, P.B.; Ramadevi, R.; Kumar, R.; Rao, B.R.; Dubeyj, S.K. Isolation and characterization of mucous exopolysaccharide (EPS) produced by Vibrio furnissii strain VB0S3. Microbiol. Biotechnol. 2007, 17, 44. [Google Scholar]
- Jiang, P.; Li, J.; Han, F.; Duan, G.; Lu, X.; Gu, Y.; Yu, W. Antibiofilm activity of an exopolysaccharide from marine bacterium Vibrio sp. QY101. PLoS ONE 2011, 6, e18514. [Google Scholar] [CrossRef] [PubMed]
- Deming, J.W.; Somers, L.K.; Straube, W.L.; Swartz, D.G.; Macdonell, M.T. Isolation of an obligately barophilic bacterium and description of a new genus, Colwellia gen. nov. Syst. Appl. Microbiol. 1988, 10, 152–160. [Google Scholar] [CrossRef]
- Kim, Y.; Park, S.; Nam, B.; Jung, Y.; Kim, D.; Yoon, J. Colwellia meonggei sp. nov., a novel gammaproteobacterium isolated from sea squirt Halocynthia roretzi. Antonie Van Leeuwenhoek 2013, 104, 1021–1027. [Google Scholar] [CrossRef] [PubMed]
- Huston, A.L.; Krieger-Brockett, B.B.; Deming, J.W. Remarkably low temperature optima for extracellular enzyme activity from Arctic bacteria and sea ice. Environ. Microbiol. 2000, 2, 383–388. [Google Scholar] [CrossRef] [PubMed]
- Methé, B.A.; Nelson, K.E.; Deming, J.W.; Momen, B.; Melamud, E.; Zhang, X.; Moult, J.; Madupu, R.; Nelson, W.C.; Dodson, R.J.; et al. The psychrophilic lifestyle as revealed by the genome sequence of Colwellia psychrerythraea 34H through genomic and proteomic analyses. Proc. Natl. Acad. Sci. USA 2005, 102, 10913–10918. [Google Scholar] [CrossRef] [PubMed]
- Nunn, B.L.; Slattery, K.V.; Cameron, K.A.; Timmins-Schiffman, E.; Junge, K. Proteomics of Colwellia psychrerythraea at subzero temperatures—A life with limited movement, flexible membranes and vital DNA repair. Environ. Microbiol. 2015, 17, 2319–2335. [Google Scholar] [CrossRef] [PubMed]
- Boetius, A.; Anesio, A.M.; Deming, J.W.; Mikucki, J.A.; Rapp, J.Z. Microbial ecology of the cryosphere: Sea ice and glacial habitats. Nat. Rev. Microbiol. 2015, 13, 677–690. [Google Scholar] [CrossRef] [PubMed]
- Arahal, D.R.; Ventosa, A. The Family Halomonadaceae. In The Prokaryotes: A handbook on the biology of bacteria, 3rd ed.; Springer: Berlin, Germany, 2006; pp. 811–835. [Google Scholar]
- Martínez-Cánovas, M.J.; Quesada, E.; Llamas, I.; Béjar, V. Halomonas ventosae sp. nov., a moderately halophilic, denitrifying, exopolysaccharide-producing bacterium. Int. J. Syst. Evol. Microbiol. 2004, 54, 733–737. [Google Scholar] [CrossRef] [PubMed]
- Byun, B.Y.; Lee, S.J.; Mah, Y.H. Antipathogenic activity and preservative effect of levan (β-2,6-fructan), a multifunctional polysaccharide. Int. J. Food Sci. Technol. 2014, 49, 238–245. [Google Scholar] [CrossRef]
- Abdel-Fattah, A.M.; Gamal-Eldeen, A.M.; Helmy, W.A.; Esawy, M.A. Antitumor and antioxidant activities of levan and its derivative from the isolate Bacillus subtilis. Carbohydr. Polym. 2012, 89, 314–322. [Google Scholar] [CrossRef] [PubMed]
- Abid, Y.; Casillo, A.; Gharsallah, H.; Joulak, I.; Lanzetta, R.; Corsaro, M.M.; Attia, H.; Azabou, S. Production and structural characterization of exopolysaccharides from newly isolated probiotic lactic acid bacteria. Int. J. Biol. Macromol. 2018, 108, 719–728. [Google Scholar] [CrossRef] [PubMed]
- Abraham, W.R.; Rohde, M. The Family Hyphomonadaceae. In The Prokaryotes; Rosenberg, E., DeLong, E.F., Lory, S., Stackebrandt, E., Thompson, F., Eds.; Springer: Berlin, Germany, 2014. [Google Scholar]
- Gauthier, G.; Gauthier, M.; Christen, R. Phylogenetic analysis of the genera Alteromonas, Shewanella and Moritella using genes coding for small-subunit rRNA sequences and division of the genus Alteromonas into two genera, Alteromonas (Emended) and Pseudoalteromonas gen. nov. and proposal of twelve new species combinations. Int. J. Syst. Evol. Microbiol. 1995, 45, 755–761. [Google Scholar] [CrossRef]
- Bosi, E.; Fondi, M.; Maida, M.; Perrin, E.; de Pascale, D.; Tutino, M.L.; Parrilli, E.; Lo Giudice, A.; Filloux, A.; Fani, R. Genome-scale phylogenetic and DNA composition analyses of Antarctic Pseudoalteromonas bacteria reveal inconsistencies in current taxonomic affiliation. Hydrobiologia 2015, 761, 85–95. [Google Scholar] [CrossRef]
- Campo, V.L.; Kawano, D.F.; da Silva, D.B.; Carvalho, I. Carrageenans: Biological properties, chemical modifications and structural analysis—A review. Carbohydr. Polym. 2009, 77, 167–180. [Google Scholar] [CrossRef]
- Toida, T.; Chaidedgumjorn, A.; Linhardt, R.J. Structure and bioactivity of sulfated polysaccharides. Trends Glycosci. Glycotechnol. 2003, 15, 29–46. [Google Scholar] [CrossRef]
- Joshi, P.M.; Juwarkar, A.A. In vivo studies to elucidate the role of extracellular polymeric substances from Azotobacter in immobilization of heavy metals environ. Sci. Technol. 2009, 43, 5884–5889. [Google Scholar] [CrossRef]
- Ha, J.S.; Lim, H.M.; Park, S.S. Extracellular hydrogen peroxide contributes to oxidative glutamate toxicity. Brain Res. 2010, 1359, 291–297. [Google Scholar] [CrossRef] [PubMed]
- Médigue, C.; Krin, E.; Pascal, G.; et al. Coping with cold: The genome of the versatile marine Antarctica bacterium Pseudoalteromonas haloplanktis TAC125. Genome Res. 2005, 15, 1325–1335. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Li, H.; Zeng, Y.; Chen, B. Extracellular enzymes of cold-adapted bacteria from Arctic sea ice, Canada basin. Polar Biol. 2009, 32, 1539–1547. [Google Scholar] [CrossRef]
- Wu, J.F.; Boyle, E.; Sunda, W.; Wen, L.S. Soluble and colloidal iron in the oligotrophic North Atlantic and North Pacific. Science 2001, 293, 847–849. [Google Scholar] [CrossRef] [PubMed]
- Jiao, G.; Yu, G.; Zhang, J.; Ewart, H.S. Chemical structures and bioactivities of sulfated polysaccharides from marine algae. Mar. Drugs 2011, 9, 196–223. [Google Scholar] [CrossRef] [PubMed]
- Luz, M.; Paje, F.; Neilan, A.B.; Couperwhite, I. A Rhodococcus species that thrives on medium saturated with liquid benzene. Microbiology 1997, 143, 2975–2981. [Google Scholar]
- Iwabuchi, N.; Sunairi, M.; Urai, M.; Itoh, C.; Anzai, H.; Nakajima, M.; Harayama, S. Extracellular polysaccharides of Rhodococcus rhodochrous S-2 stimulate the degradation of aromatic components in crude oil by indigenous marine bacteria. Appl. Environ. Microbiol. 2002, 68, 2337–2343. [Google Scholar] [CrossRef] [PubMed]
- Urai, M.; Anzai, H.; Iwabuchi, N.; Sunairi, M.; Nakajima, M. A novel moisture-absorbing extracellular polysaccharide from Rhodococcus rhodochrous SM-1. Actinomycetologica 2002, 16, 26–31. [Google Scholar] [CrossRef]
- Urai, M.; Anzai, H.; Iwabuchi, N.; Sunairi, M.; Nakajima, M. A novel viscous extracellular polysaccharide containing fatty acids from Rhodococcus rhodochrous ATCC 53968. Actinomycetologica 2004, 18, 15–17. [Google Scholar] [CrossRef]
- Urai, M.; Anzai, H.; Ogihara, J.; Iwabuchi, N.; Harayama, S.; Sunairi, M.; Nakajima, M. Structural analysis of an extracellular polysaccharide produced by Rhodococcus rhodochrous strain S-2. Carbohydr. Res. 2006, 341, 766–775. [Google Scholar] [CrossRef] [PubMed]
- DeLong, E.F.; Franks, D.G.; Yayanos, A.A. Evolutionary relationship of cultivated psychrophilic and barophilic deep-sea bacteria. Appl. Environ. Microbiol. 1997, 63, 2105–2108. [Google Scholar] [PubMed]
- Nogi, Y. Microbial life in the deep sea: Psychropiezophiles. In Psychrophiles: from biodiversity to biotechnology, 2nd ed.; Margesin, R., Ed.; Springer: Berlin, Germany, 2017; pp. 259–284. [Google Scholar]
- Murray, A.E.; Lies, D.; Li, G.; Nealson, K.; Zhou, J.; Tiedje, J.M. DNA/DNA hybridization to microarrays reveals gene-specific differences between closely related microbial genomes. Proc. Natl. Acad. Sci. USA 2001, 98, 9853–9858. [Google Scholar] [CrossRef] [PubMed]
- Weiner, R.M.; Coyne, V.E.; Brayton, P.; West, P.; Raiken, S.F. Alteromonas colwelliana sp. nov., an isolate from oyster habitats. Int. J. Syst. Bacteriol. 1988, 38, 240–244. [Google Scholar] [CrossRef]
- Weiner, R.M.; Segall, A.M.; Colwell, R.C. Characterization of a marine bacterium associated with Crassostrea virginica (the Eastern Oyster). Appl. Environ. Microbiol. 1985, 49, 83–90. [Google Scholar] [PubMed]
- Reen, F.J.; Almagro-Moreno, S.; Ussery, D.; Boyd, E.F. The genomic code: Inferring Vibrionaceae niche specialization. Nat. Rev. Microbiol. 2006, 4, 697–704. [Google Scholar] [CrossRef] [PubMed]
- Romalde, J.L.; Diéguez, A.L.; Lasa, A.; Balboa, S. New Vibrio species associated to molluscan microbiota: A review. Front. Microbiol. 2014, 4. [Google Scholar] [CrossRef] [PubMed]
- Casillo, A.; Ziaco, M.; Lindner, B.; Parrilli, E.; Schwudke, D.; Holgado, A.; Verstrepen, L.; Sannino, F.; Beyaert, R.; Lanzetta, R.; et al. Unusual lipid A from a cold-adapted bacterium: Detailed structural characterization. ChemBioChem 2017, 18, 1845–1854. [Google Scholar] [CrossRef] [PubMed]
- Phillips, N.J.; Adin, D.M.; Stabb, E.V.; McFall-Ngai, M.J.; Apicella, M.A.; Gibson, B.W. The Lipid A from Vibrio fischeri lipopolysaccharide: A unique structure bearing a phosphoglycerol moiety. J. Biol. Chem. 2011, 21203–21219. [Google Scholar] [CrossRef] [PubMed]
- Song, K.; Yu, W.G.; Han, F.; Han, W.J.; Li, J.B. Purification and characterization of alginate lyase from marine bacterium Vibrio sp. QY101. Sheng Wu Hua Xue Yu Sheng Wu Wu Li Xue Bao 2003, 35, 473–477. [Google Scholar] [PubMed]
- Cobet, A.B.; Jones, G.E.; Albright, J.; Simon, H.; Wirsen, C. The effect of nickel on a marine bacterium: Fine structure of Arthrobacter marinus. J. Gen. Microbiol. 1971, 66, 185–196. [Google Scholar] [CrossRef] [PubMed]
- Bernardet, J.F.; Bowman, J.P. The Genus Flavobacterium. In The Prokaryotes; Dworkin, M., Falkow, S., Rosenberg, E., Schleifer, K.H., Stackebrandt, E., Eds.; Springer: New York, NY, USA, 2006. [Google Scholar]
- Dong, S.; Yang, J.; Zhang, X.Y.; Shi, M.; Song, X.Y.; Chen, X.L.; Zhang, Y.Z. Cultivable alginate lyase-excreting bacteria associated with the arctic brown alga Laminaria. Mar. Drugs 2012, 10, 2481–2491. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Cánovas, M.J.; Quesada, E.; Martínez-Checa, F.; del Moral, A.; Béjar, V. Salipiger mucosus gen. nov., sp. nov., a moderately halophilic, exopolysaccharide-producing bacteriumisolated from hypersaline soil, belonging to the α-Proteobacteria. Int. J. Syst. Evol. Microbiol. 2004, 54, 1735–1740. [Google Scholar] [CrossRef] [PubMed]
- Dugan, P.R.; Stoner, D.L.; Pickrum, H.M. The Genus Zoogloea. In The Prokaryotes; Balows, A., Trüper, H.G., Dworkin, M., Harder, W., Schleifer, K.H., Eds.; Springer: New York, NY, USA, 1992. [Google Scholar]
- Lim, D.J.; Kim, J.D.; Kim, M.Y.; Yoo, S.H.; Kong, J.Y. Physicochemical properties of the exopolysaccharides produced by marine bacterium Zoogloea sp. KCCM10036. J. Microbiol. Biotechnol. 2007, 17, 979–984. [Google Scholar] [PubMed]
- Poli, A.; Di Donato, P.; Abbamondi, G.R.; Nicolaus, B. Synthesis, production and biotechnological applications of exopolysaccharides and polyhydroxyalkanoates by Archaea. Archaea 2011, 693253, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Nicolaus, B.; Lama, L.; Esposito, E.; Manca, M.C.; Improt, R.; Bellitti, M.R.; Duckworth, A.W.; Grant, W.D.; Gambacorta, A. Haloarcula spp able to biosynthesize exo- and endopolymers. J. Ind. Microbiol. Biotechnol. 1999, 23, 489–496. [Google Scholar] [CrossRef]
- Lü, Y.; Lu, H.; Wang, S.; Han, J.; Xiang, H.; Jin, C. An acidic exopolysaccharide from Haloarcula hispanica ATCC33960 and two genes responsible for its synthesis. Archea 2017, 2017, 5842958. [Google Scholar] [CrossRef]
- Anton, J.; Meseguer, I.; Rodriguez-Valera, F. Production of an extracellular polysaccharide by Haloferax mediterranei. Appl. Environ. Microbiol. 1988, 54, 2381–2386. [Google Scholar] [PubMed]
- Parolis, H.; Parolis, L.A.S.; Boán, I.F.; Rodriguez-Valera, F.; Widmalm, G.; Manca, M.M.; Jansson, P.E.; Sutherland, I.W. The structure of the exopolysaccharide produced by the halophilic Archaeon Haloferax mediterranei strain R4 (ATCC 33500). Carhohydr. Res. 1996, 295, 147–156. [Google Scholar] [CrossRef]
- Paramonova, N.A.; Parolis, L.A.S.; Parolis, H.; Boán, I.F.; Anton, J.; Rodríguez-Valera, F. The structure of the exocellular polysaccharide produced by the Archaeon Haloferax gibbonsii (ATCC 33959). Carhohydr. Res. 1998, 309, 89–94. [Google Scholar] [CrossRef]
- Parolis, L.A.S.; Parolis, H.; Paramonova, N.A.; Boán, I.F.; Anton, J.; Rodríguez-Valera, F. Structural studies on the acidic exopolysaccharide from Haloferax denitrificans ATCC 35960 Carhohydr. Res. 1999, 319, 133–140. [Google Scholar] [CrossRef]
- Rinker, K.D.; Kelly, R.M. Growth physiology of the hyperthermophilic Archaeon Thermococcus litoralis: Development of a sulfur-free defined medium, characterization of an exopolysaccharide and evidence of biofilm formation. Appl. Environ. Microbiol. 1996, 4478–4485. [Google Scholar]
- Sunagawa, S.; Coelho, L.P.; Chaffron, S.; Kultima, J.R.; Labadie, K.; Salazar, G.; Djahanschiri, B.; Zeller, G.; et al. Structure and function of the global ocean microbiome. Science 2015, 348, 1261359. [Google Scholar] [CrossRef] [PubMed]
- BeMiller, J.N. Gum Arabic and Other Exudate Gums. In Carbohydrate Chemistry for Food Scientists; AACC International: St. Paul, MN, USA, 2007; pp. 313–320. [Google Scholar]
- Gutierrez, T.; Mulloy, B.; Black, K.; Green, D.H. Glycoprotein emulsifiers from two marine Halomonasspecies: Chemical and physical characterization. J. Appl. Microbiol. 2007, 103, 1716–1727. [Google Scholar] [CrossRef] [PubMed]
- Chin, W.C.; Orellana, M.V.; Verdugo, P. Formation of microgels by spontaneous assembly of dissolved marine polymers. Nature 1998, 391, 568–572. [Google Scholar]
- Azam, F.; Malfatti, F. Microbial structuring of marine ecosystems. Nature Rev. Microbiol. 2007, 5, 782–791. [Google Scholar] [CrossRef] [PubMed]
- Smith, D.C.; Simon, M.; Alldredge, A.L.; Azam, F. Intense hydrolytic enzyme activity on marine aggregates and implications for rapid particle dissolution. Nature 1992, 359, 139–142. [Google Scholar] [CrossRef]
- Hassler, C.S.; Schoemann, V.; Mancuso Nichols, C.; Butler, E.C.V.; Boyd, P.W. Saccharides enhance iron bioavailability to Southern Ocean phytoplankton. Proc. Natl. Acad. Sci. USA 2011, 108, 1076–1081. [Google Scholar] [CrossRef] [PubMed]
- Gledhill, N.; Buck, K.N. The organic complexation of iron in the marine environment:a review. Front. Microbiol. 2012, 3, 69. [Google Scholar] [CrossRef] [PubMed]
- Gledhill, M.; Milton, J.A.; Tarran, G.A.; Ussher, S.; Thompson, A.; Wolff, G.A.; Worsfold, P.J.; Achterberg, E.P. Hydroxamate siderophores: Occurrence and importance in the Atlantic Ocean. Environ. Sci. Technol. 2008, 42, 8675–8680. [Google Scholar] [CrossRef]
- Velasquez, I.; Nuun, B.L.; Ibisanmi, E.; Goodlett, D.R.; Hunter, K.A.; Sander, S.G. Detection of hydroxamate siderophores in coastal and Sub-Antarctic waters off the South Eastern Coast of New Zealand. Mar. Chem. 2011, 126, 97–107. [Google Scholar] [CrossRef]
- Norman, L.; Worms, I.A.M.; Angles, E.; Bowie, A.R.; Mancuso Nichols, C.; Pham, A.N.; Slaveykova, V.I.; Townsend, A.T.; Waite, T.D.; Hassler, C.S. The role of bacterial and algal exopolymeric substances in iron chemistry. Mar. Chem. 2015, 173, 148–161. [Google Scholar] [CrossRef]
- Bhaskar, P.V.; Bhosle, N.B. Microbial extracellular polymeric substances in marine biogeochemical processes. Curr. Sci. 2005, 88, 45–53. [Google Scholar]
- Decho, A.W. Microbial biofilms in intertidal systems: An overview. Cont. Shelf Res. 2000, 20, 1257–1273. [Google Scholar] [CrossRef]
- Doney, S.C.; Fabry, V.J.; Feely, R.A.; Kleypas, J.A. Ocean Acidification: The Other CO2 Problem. Annu. Rev. Mar. Sci. 2009, 1, 169–192. [Google Scholar] [CrossRef] [PubMed]
- Dupraz, C.; Reid, R.P.; Braissant, O.; Decho, A.W.; Norman, R.S.; Visscher, P.T. Processes of carbonate precipitation in modern microbial mats, Earth-Sci. Rev. 2009, 96, 141–162. [Google Scholar] [CrossRef]
- Endres, S.; Galgani, L.; Riebesell, U.; Schulz, K.-G.; Engel, A. Stimulated Bacterial Growth under Elevated pCO2: Results from an Off-Shore Mesocosm Study. PLoS ONE 2014, 9, e99228. [Google Scholar] [CrossRef] [PubMed]
- Lidbury, I.; Johnson, V.; Hall-Spencer, J.M.; Munn, C.B.; Cunliffe, M. Community-level response of coastal microbial biofilms to ocean acidification in a natural carbon dioxide vent ecosystem. Mar. Pollut. Bull. 2012, 64, 1063–1066. [Google Scholar] [CrossRef] [PubMed]
- Underwood, G.J.C.; Fietz, S.; Papadimitriou, S.; Thomas, D.N.; Dieckmann, G.S. Distribution and composition of dissolved extracellular polymeric substances (EPS) in Antarctic sea ice. Mar. Ecol. Prog. Ser. 2010, 404, 1–19. [Google Scholar] [CrossRef]
- Underwood, G.J.C.; Aslam, S.N.; Niemi, A.; Norman, L.; Meiners, K.M.; Laybourn-Parry, J.; Paterson, H.; Thomas, D.N. Broad-scale predictability of carbohydrates and EPS in Antarctic and Arctic sea ice. Proc. Natl. Acad. Sci. USA 2013, 110, 15734–15739. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Lu, J.; Lu, L.; Liu, Y.; Wang, F.; Xiao, M. Isolation, structural characterization and immunological activity of an exopolysaccharide produced by Bacillus licheniformis 8-37-0-1. Bioresour. Technol. 2010, 101, 5528–5533. [Google Scholar] [CrossRef] [PubMed]
- Leung, M.Y.K.; Liu, C.; Koon, J.C.M.; Fung, K.P. Polysaccharide biological response modifiers. Immunol. Lett. 2006, 105, 101–114. [Google Scholar] [CrossRef] [PubMed]
- De Ruiter, G.A.; Rudolph, B. Carrageenan biotechnology. Trends Food Sci. Technol. 1997, 8, 389–395. [Google Scholar] [CrossRef]
- Li, D.; Chen, L.; Yi, X.; Zhang, X.; Ye, N. Pyrolytic characteristics and kinetics of two brown algae and sodium alginate. Bioresour. Technol. 2010, 101, 7131–7136. [Google Scholar] [CrossRef] [PubMed]
- Nicolaus, B.; Kambourova, M.; Oner, E.T. Exopolysaccharides from extremophiles: From fundamentals to biotechnology. Environ. Technol. 2010, 31, 1145–1158. [Google Scholar] [CrossRef] [PubMed]
- Aquino, R.S.; Grativol, C.; Mourão, P.A.S. Rising from the Sea: Correlations between Sulfated Polysaccharides and Salinity in Plants. PLoS ONE 2011, 6, e18862. [Google Scholar] [CrossRef] [PubMed]
| Monosaccharides | Example |
|---|---|
| Pentoses | arabinose (Ara), ribose (Rib), xylose (Xyl) |
| Hexoses | glucose (Glc), galactose (Gal), mannose (Man) |
| Deoxy-hexoses | quinovose (Qui), fucose (Fuc), rhamnose (Rha) |
| Uronic acids | glucuronic acid (GlcA), galacturonic acid (GalA), mannuronic acid (ManA) |
| Amino sugars | glucosamine (GlcN), galactosamine (GalN), mannosamine (ManN) |
| Uncommon sugars | 3-deoxy-d-manno-2-octulosonic acid (Kdo), neuraminic acid (Neu) |
| Microorganism | Source | EPS Structure or Monosaccharide Composition | Functions and Applications | Reference |
|---|---|---|---|---|
| Alteromonas | ||||
| A. macleodii sub. fijiensis biovar deepsane HYD 657 | Deep-sea hydrothermal vent polychaete annelid | Fuc:Rha:Glc:Gal:Man:GlcA:GalA 1:2.5:2.6:5.9:1.4:2:1.9 Sulphate, lactate and pyruvate groups | Cosmetic, keratinocytes protection | [62,63] |
| A. strain HYD-1545 | Hydrothermal vent polychaete annelid | Gal, Glc, GlcA, GalA, GalX 2.5:3:2:2:1 X = pyruvate | - | [64] |
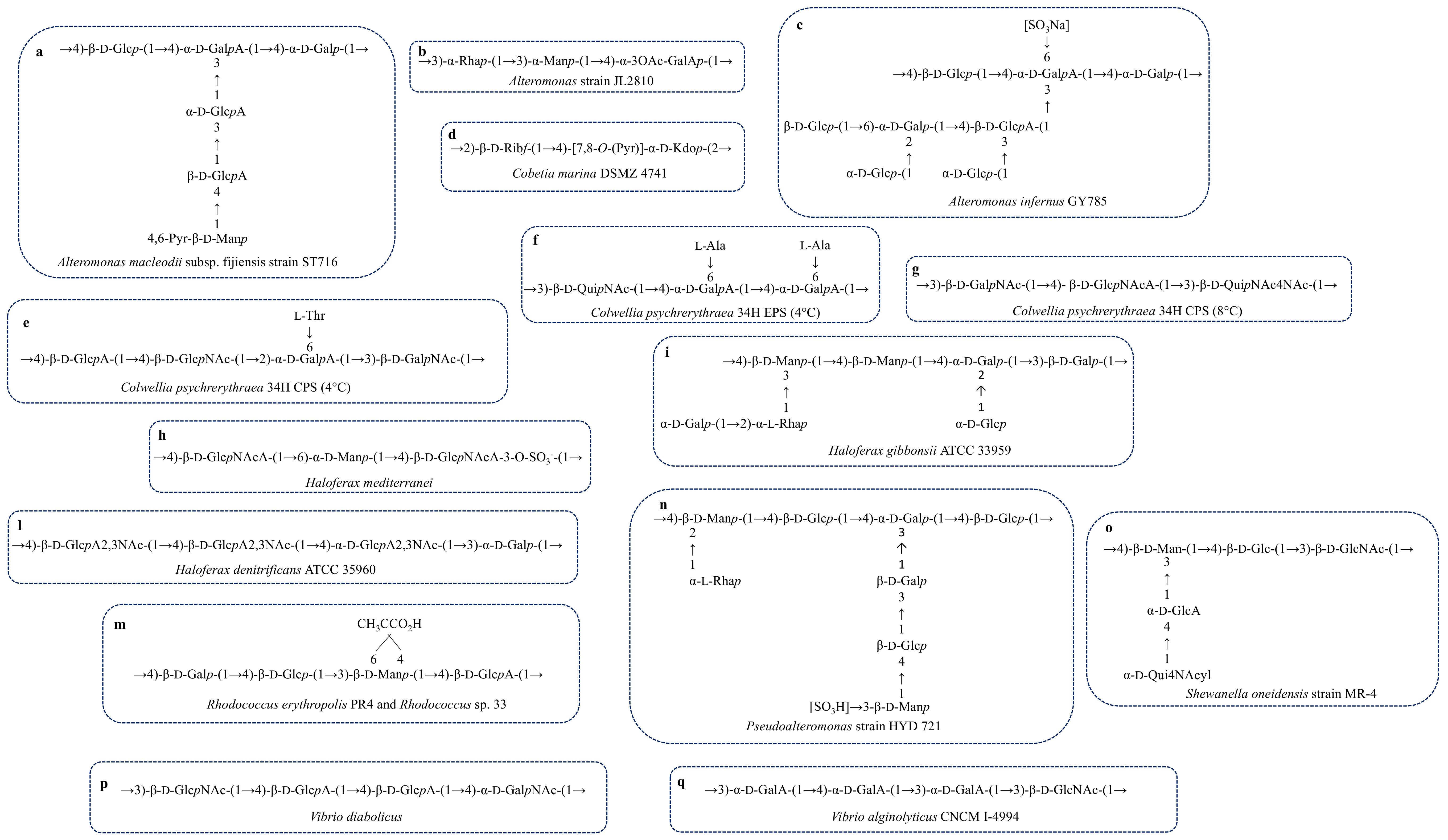
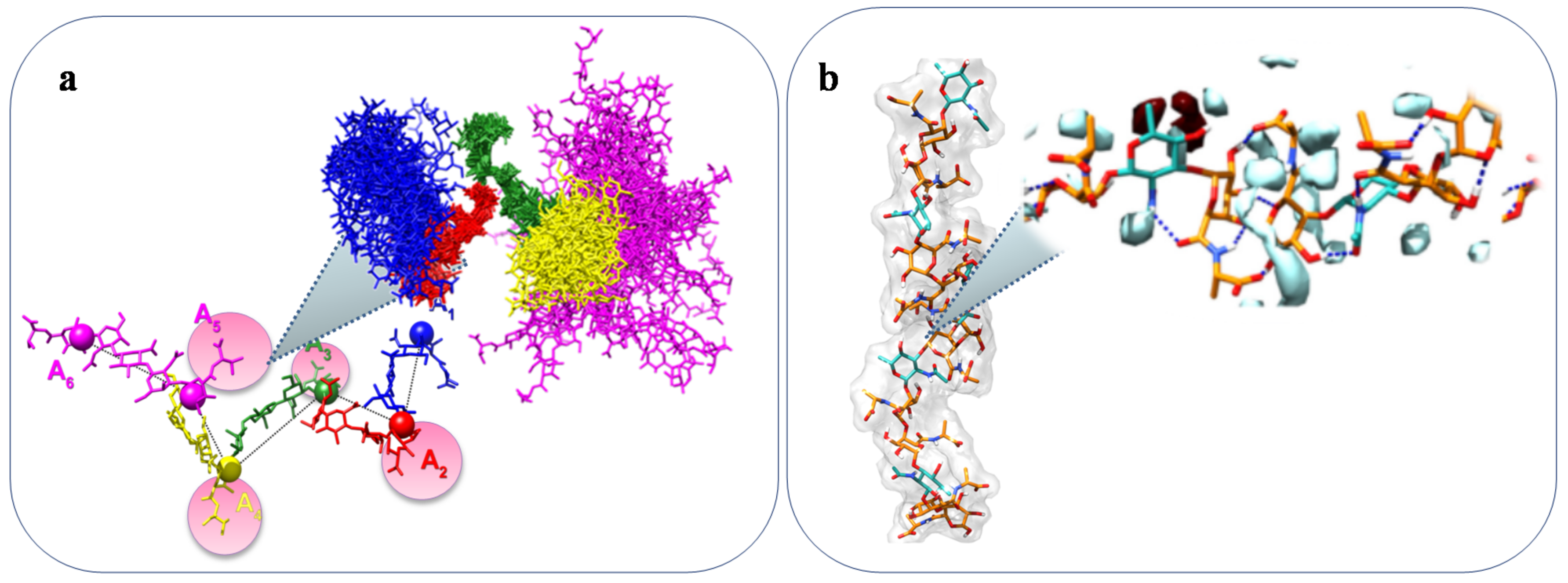
| A. macleodii sub. fijiensis strain ST716 | Deep-sea hydrothermal vent | Structure, Figure 4a | Gel forming | [65,66] |
| A. strain JL2810 | Sea water | Structure, Figure 4b | Biosorption of heavy metal | [67,68] |
| A. infernus GY785 | Hydrothermal vent | Structure, Figure 4c | Metal recover | [69,70] |
| A. hispanica F32 | Hypersaline inland | Glc:Man:Rha:Xyl 18:63:7:12 Sulphate and phosphate groups | - | [71] |
| Bacillus | ||||
| B. strain B3-15 halophile | Marine hot spring | Man | - | [72,73] |
| B. strain B3-72 thermophile | Hydrothermal vent | EPS1, Man:Glc 0.3:1 EPS2, Man:Glc1:0.2 | - | [74] |
| B. strain I-450 haloalkaliphile | Mudflats | Glc, Gal, Fru | Gel forming | [75,76] |
| Cobetia marina DSMZ 4741 | Coast | Structure, Figure 4d | - | [77] |
| Colwellia | ||||
| C. psychrerythraea 34H psychrophile | Sea sediments, sea ice | Structure, Figure 4e | Anti-freeze | [37] |
| C. psychrerythraea 34H | Sea sediments, sea ice | Structure, Figure 4f | Anti-freeze | [78,79] |
| C. psychrerythraea 34H | Sea sediments, sea ice | Structure, Figure 4g | No anti-freeze activity | [80] |
| Flavobacterium uliginosum MP-55 | Sea weed | Glc:Man:Fuc 7:2:1. Marinactan | [81] | |
| Geobacillus | ||||
| G. strain 4004 thermophile | Sea water | EPS1, Man:Glc:Gal 0.5:1:0.3 EPS2, Man:Glc:Gal 1:0.3:traces EPS3, Gal:Man:GlcN:Ara 1:0.8:0.4:0.2 | - | [82] |
| G. tepidamans V264 thermophile | Hot spring | Glucan | Immunomodulant, anti-viral | [83] |
| G. thermodenitrificans thermophile | Shallow marine vent | Man, Glc | [84] | |
| Hahella chejuensis strain 96CJ1035 | Marine sediments | Glc:Gal 1:6.8 Traces of xylose and ribose | Emulsifier | [85,86] |
| Halomonas | ||||
| H. eurihalina F2-7 | Dead Sea | Glc:Man:Rha 2.9:1.5:1 Uronic acids, amino sugars, sulphate groups | Emulsifier | [87,88] |
| H. maura halophile | Solar saltern | Man:Gal:Glc:GlcA 34.8:14:29.3:21.9 Sulphate groups | Viscous, pseudoplastic | [89,90] |
| H. sp. OKOH halophile | Bottom sediments | No structure | Flocculant | [91] |
| H. sp AAD6 (JCM 15723) | Soil saltern | Levan | - | [92,93] |
| H. alkaliantarctica strain CRSS | Salt lake | EPS1, Mannan EPS2, Xylo-mannan EPS3, Fructo-glucan | - | [94] |
| Hyphomonas | ||||
| H. strain MHS-3 | Shallow marine sediments | GalNAcA | Adhesion | [95,96,97] |
| H. strainVP-6 | Vent region | No structure | Adhesion | [98] |
| Idiomarina | ||||
| I. fontislapidosi F23T | Lagoon | Glc, Man, Xyl, Rha Sulphate and phosphate groups | Emulsifier, metal binding | [71] |
| I. ramblicola R22T | Water-course | Glc, Man, Xyl, Rha Sulphate and phosphate groups | Emulsifier, metal binding | [71] |
| Polaribacter sp. SM1127 psychrophile | Artic brown alga Laminaria | Rha:Fuc:GlcA:Man:Glc:GlcN 0.8:7.4:21.4:23.4:17.3:1.6:28.0 | Cryoprotectant, anti-oxidant | [99] |
| Pseudoalteromonas | ||||
| P. strain HYD721 | Deep-sea hydrothermal vent | Structure, Figure 4n | - | [100] |
| P. strain TG12 | Sea-water | Rha:Fuc:Gal:GalNAc:Glc:GlcNAc 1.2:0.5:0.9:0.7:10.4:24.8 Man:Xyl:MurA:GalA:GlcA 4.8:27.7:0.3:23.1:5.6 | Metal binding | [101] |
| P. ulvae TC14 | Marine biofilm | LB-EPS, Glc, uronic acids TB-EPS, Glc, uronic acids Sol-EPS, Glc | Anti-biofilm | [102] |
| P. ruthenica | Sea-water | Man, traces of uronic acids | Pseudoplastic | [103] |
| P. sp. strain MD12-642 | Marine sediments | GalA:GlcA:Rha:GlcNAc 44:28:15:14 | Viscosity | [104] |
| P. haloplanktis TAC125 psychrophile | Antarctic sea water | Man, Glc Phosphate groups | - | [105] |
| P. sp. strain SM20310 psychrophile | Arctic sea-ice | Rha:Xyl:Man:Gal:Glc:GalNAc:GlcNAc 2.1:0.9:71.7:9.0:10.7:1.5:4.0 | Cryoprotectant | [106] |
| P. arctica KOPRI 21653 psychrophile | Sea-side sediments | Glc:Gal 1.5:1 | Cryoprotectant | [107] |
| P. elyakovii Arcpo 15 psychrophile | Not reported | Man:GalA 3.3:1 | Cryoprotectant | [40] |
| P. sp. CAM025 psychrophile | Particles form Antarctic sea | Glc:GalA:Rha:Gal 1:0.5:0.1:0.08 | Adhesion | [108] |
| P. sp. CAM036 psychrophile | Particles from Southern Ocean | GalA:Glc:Man:GalNAc:Ara 1:0.8:0.84:0.36:0.13 | - | [108] |
| P. sp. SM9913 psychrophile | Deep-sea sediment | Glc, Gal, Xyl, Ara | Metal binding | [109] |
| P. sp. MER144 psychrophile | Terra Nova Bay, Ross SeaAntarctic | Glc:Man:GalN:Ara:GlcA:GalA:Gal 1:0.36:0.26:0.06:0.06:0.05:0.03 | - | [110] |
| Pseudomonas | ||||
| P. sp. NCMB 2021 | Not reported | Pol A, Glc:Gal:GlcA:GalA 1.45:1.18:0.64:0.43 Pol B, GlcNAc, deoxy-Hex, Kdo | metal binding (A) Adhesion (B) | [111] |
| P. sp. S9 psychrophile | Polar basin | Glc:GlcNAc:GalNAc 28:35:37 | Adhesion | [112] |
| P. sp. WAK1 | Brown seaweed Undaria pinnatifida | Gal:Glc 2:1 Sulphate groups | Anti-cancer | [113] |
| P. stutzeri 273 | Marine sediments | GlcN:Rha:Glc:Man 35.4:28.6:27.2:8.7 | Anti-biofilm, anti-biofouling, antioxidant | [114] |
| P. sp. ID1 | Marine sediments | Glc:Gal:Fuc 2:1:1 | Cryoprotectant | [115] |
| Rhodococcus | ||||
| R. sp. 33 | Contaminate site near a chemical plant | Structure, Figure 4m | - | [116,117] |
| R. erythropolis PR4 | Ocean | FR2 Structure, Figure 4m | Emulsifier | [118] |
| FR1 Glc:GlcN:GlcA:Fuc 2:1:1:1 | - | |||
| Salipinger mucosus A3Thalophile | Solar saltern | Glc:Man:Gal:Fuc 1.5:2.5:2.5:1 Sulphate and phosphate groups | Metal binding, emulsifier, pseudoplastic | [119] |
| Shewanella | ||||
| S. oneidensis MR-4 | Dead Sea | Structure, Figure 4o | - | [38] |
| S. colwelliana | Associate bivalve | Man:Glc:Gal:pyruvate 1:2:2:4 | - | [120] |
| Vibrio | ||||
| V. diabolicus | Deep-Sea hydrothermal ventA. pompejana | Structure, Figure 4p | Filler of bone defects in rat calvaria | [121,122] |
| V. alginolyticus | Sea water | Glc:Xyl:RibN:AraN 2:1:9:1 | Shearing properties | [123] |
| V. alginolyticus CNCM I 4994 | Sea water | Structure, Figure 4q | - | [124] |
| V. harveji VB23 | Sea water | Gal:Glc:Rha:Fuc:Man:Rib:Ara:Xyl 10.08:3.6:0.7:0.15:1.56:0.2:0.3:0.45 | Emusilfier | [125] |
| V. furnissii strain VB0S3 | Sea water | Gal:Glc:Rha:Fuc:Man:Rib:Ara 5.21:4.68:1.0:0.79:1.43:0.16:0.19 | Emusilfier | [126] |
| V. sp. QY101 | Laminaria thallus | Glc:Gal:GlcA:GalA:Rha:Fuc:GlcN:Man 6.57:6.89:21.47:23.05:23.9:3.61:12.15:2.36 | Anti-biofilm | [127] |
| Zooglea sp. KCCM100376 | Seaweed Undaria | CBP, Glc:Gal:Man 1:2:2 WSP, Glc:Gal:Man 2:2:3 | Water-holding capacity | [39] |
| Microorganism | Source | EPS Structure or Monosaccharide Composition | Functions and Applications | Reference |
|---|---|---|---|---|
| Haloarcula | ||||
| H. japonica T5 halophile | Marine saltern | Man:Gal:GlcA 2:1:3 | - | [172] |
| H. japonica T6–T7 halophile | Marine saltern | Man:Gal:Glc:GlcA 1:0.2:0.2:traces | - | [172] |
| H. hispanica ATCC 33960 halophile | Solar saltern | Man:Gal:Glc 55.9:43.2:0.9 Sulphate groups | - | [173] |
| Haloferax | ||||
| H. mediterranei R-4 ATCC 33500 halophile | Salt ponds | Structure, Figure 4h | Pseudoplastic | [174,175] |
| H. gibbonsii ATCC 33959 halophile | Marine saltern Dead Sea | Structure, Figure 4i | - | [176] |
| H. denitrificans ATCC 35960 halophile | Saltern | Structure, Figure 4l | - | [177] |
| Thermococcus litoralis DSM5 473 e DSM 3638 | Shallow marine Thermal spring | Man | Adhesion | [178] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Casillo, A.; Lanzetta, R.; Parrilli, M.; Corsaro, M.M. Exopolysaccharides from Marine and Marine Extremophilic Bacteria: Structures, Properties, Ecological Roles and Applications. Mar. Drugs 2018, 16, 69. https://doi.org/10.3390/md16020069
Casillo A, Lanzetta R, Parrilli M, Corsaro MM. Exopolysaccharides from Marine and Marine Extremophilic Bacteria: Structures, Properties, Ecological Roles and Applications. Marine Drugs. 2018; 16(2):69. https://doi.org/10.3390/md16020069
Chicago/Turabian StyleCasillo, Angela, Rosa Lanzetta, Michelangelo Parrilli, and Maria Michela Corsaro. 2018. "Exopolysaccharides from Marine and Marine Extremophilic Bacteria: Structures, Properties, Ecological Roles and Applications" Marine Drugs 16, no. 2: 69. https://doi.org/10.3390/md16020069
APA StyleCasillo, A., Lanzetta, R., Parrilli, M., & Corsaro, M. M. (2018). Exopolysaccharides from Marine and Marine Extremophilic Bacteria: Structures, Properties, Ecological Roles and Applications. Marine Drugs, 16(2), 69. https://doi.org/10.3390/md16020069






