1. Introduction
Over the last years, the herbal market has rapidly increased and many surveys have been conducted that aim at finding natural ingredients with possible applications as food additives or medicine. In this area, special attention has been given to the sea environment as a rich source of new active compounds [
1]. Among marine botanical organisms, algae have been deeply investigated and exploited [
2]; in contrast, the phytopharmaceutical profile of seagrasses is still almost unknown. However, these latter are high plants sharing adaptive metabolic features with both terrestrial plants and seaweed, resulting in a peculiar phytocomplex possibly endowed with interesting biological properties [
3].
Posidonia oceanica (L.) Delile, family Potamogetonaceae, is a long-living, slow-growing, endemic Mediterranean seagrass forming extensive meadows in coastal shallow waters [
4]. The plant undergoes massive leaf loss in autumn, giving rise in some areas to conspicuous beach deposits. The profile of secondary metabolites is mostly characterized by phenolic compounds, as also testified by the presence of so-called tannin cells [
5]. Chicoric acid is generally reported as the major constituent of leaves [
6,
7], caftaric and gentisic acids are also abundant in leaves, and other phenolic constituents include the aldehyde vanillin, and
p-coumaric, ferulic, caffeic, and cinnamic acids [
8]. Other chemical constituents isolated from the plant include phenol derivatives (e.g., phloroglucinol), benzoic acid derivatives (e.g.,
p-hydroxybenzoic and vanillic acids), calchones, and proanthocyanidins [
9]. Lignin is present in all tissues [
10], while major flavonoids include the flavonols kaempferol, quercetin, isorhamnetin (about 1–3 μg g
−1 dw), and myricetin (about 20 μg g
−1 dw) [
11]. The lipid fraction has been found to contain prevalently palmitic, palmitoleic, oleic, and linoleic acids, in addition to the phytosteroids campesterol, stigmasterol, and β-sitosterol [
12]. A novel sesquiterpenic alcohol named posidozinol has also been isolated from the leaves [
13].
The plant exploitation has mostly concerned beached leaf litter, used in the past as packing material for glassware and pottery, roof insulation, shipping of fishery products, cattle bedding, mattress and pillow filling, and still used in some cases as cattle forage and compost [
14]. News on medicinal uses date back to ancient Egypt, where it was supposedly used for skin disease [
15], while more recently in the southwestern Mediterranean region the plant has been used as a remedy for acne, leg pain, diabetes, respiratory infections, hypertension, and colitis [
16].
Very little scientific data on
P. oceanica bioactivities and therapeutic properties are available. Antibacterial and antimycotic activities have been found in a rhizome extract [
17], while antidiabetic and vasoprotective effects on alloxan diabetic rats have been observed upon treatment with an ethyl acetate fraction from the aqueous residue of a hydroethanolic leaf extract [
18].
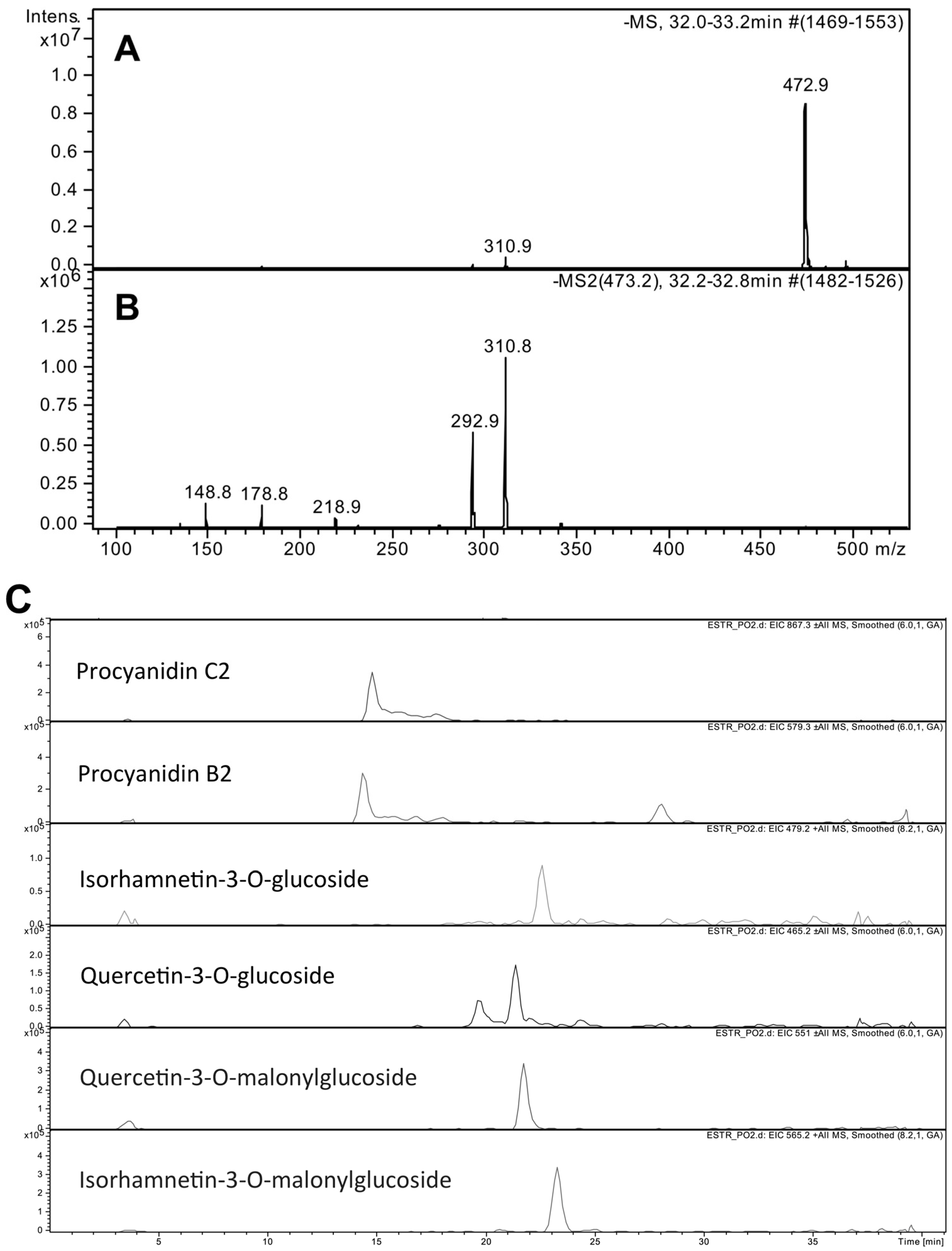
We obtained a P. oceanica ethanolic leaf extract (PEE) characterized by a high concentration of chicoric acid, as determined through HPLC-MS analysis. PEE was subjected to in vitro cell-free and cell-based tests, aimed at showing possible applications for skin care and disease. These experiments revealed stimulation of fibroblast proliferation and collagen production, anti-melanogenic activities, and the stimulation of lipolysis in adipocytes, suggesting that PEE could find applications in skin anti-aging, anti-cellulite and depigmenting products.
3. Discussion
This is the first study concerning the effects of a
P. oceanica extract on skin cells, and one of the very few scientific reports about the biological properties of the
P. oceanica phytocomplex. In agreement with literature reports about this plant, our HPLC-MS characterization showed that the major compound of PEE was chicoric acid. The resultant extract was enriched in chicoric acid, with amounts 4–5-fold higher than those reported for
P. oceanica leaves [
7]. The quantification of PEE radical scavenging activity was remarkably high among plant extracts [
20], consistent with the richness in chicoric acid and other phenolic compounds typical of
P. oceanica. Our analyses also confirmed the chemical affinity of seagrass with seaweed, since the iodine content of PEE rated within the range reported for seaweed [
21]. Considering the usual doses of extracts from natural sources used in products for humans (e.g., 0.1–1%), the PEE iodine content is quite compatible with a maximum tolerable daily intake of 1.0 mg iodine set by the World Health Organization [
22].
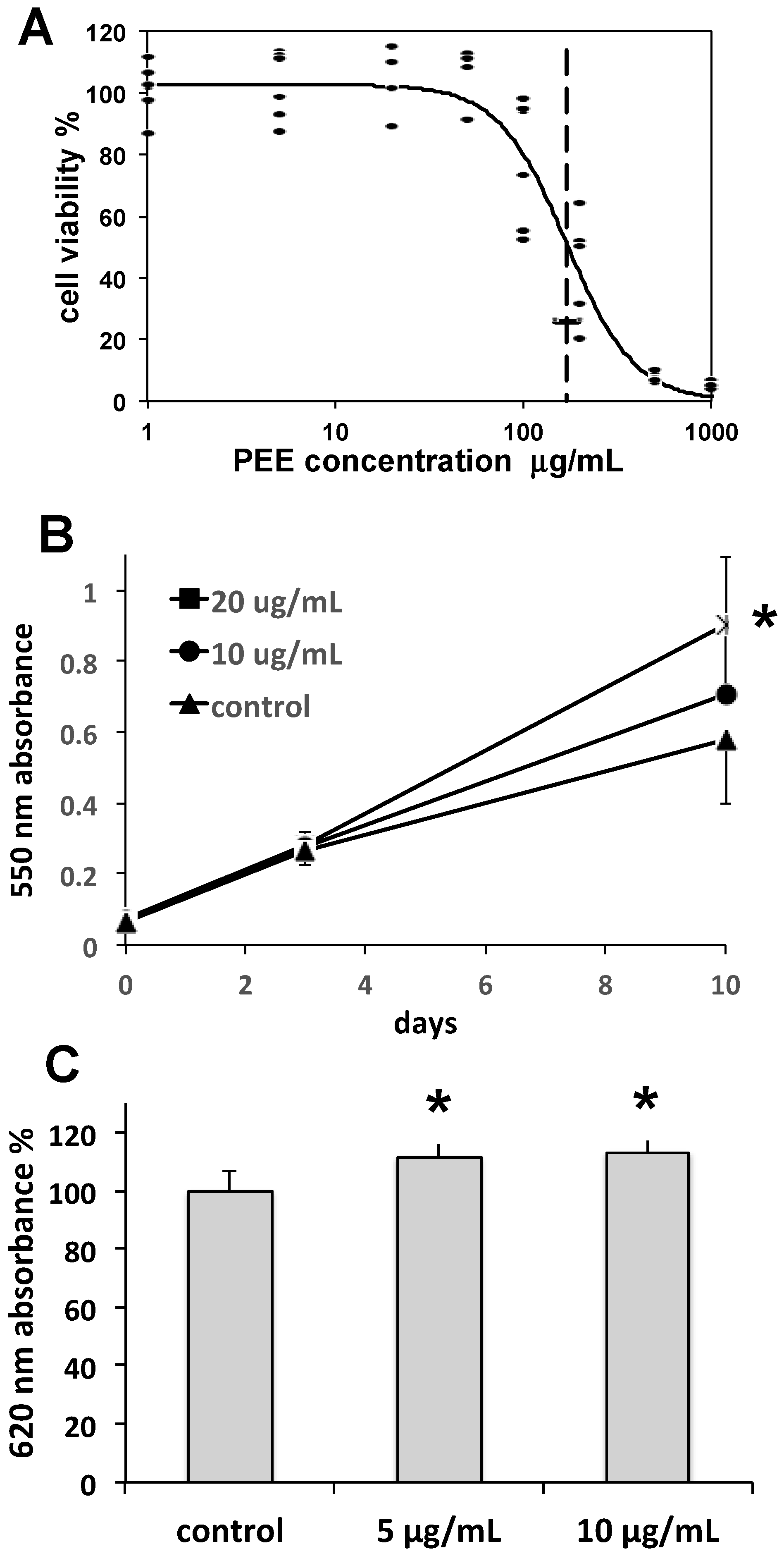
Collagen is the main constituent of the dermal matrix, while collagen type I is the most abundant isoform, forming collagen bundles. Collagen is produced by fibroblasts, is essential for skin tone and turgor, and undergoes physiological turnover through continuous degradation by matrix metalloproteinases and replacement by fibroblast neosynthesis. During skin aging, collagen degradation tends to overwhelm renewal, resulting in the formation of fine lines, wrinkles, and other alterations. Hence, the maintenance of fibroblast function is a prerequisite for contrasting skin aging. In our study, the complex of effects induced by PEE on fibroblast growth rate and collagen synthesis indicate a positive stimulation of fibroblast activity, suggesting the possible use of PEE in anti-wrinkle and anti-aging skin care formulations.
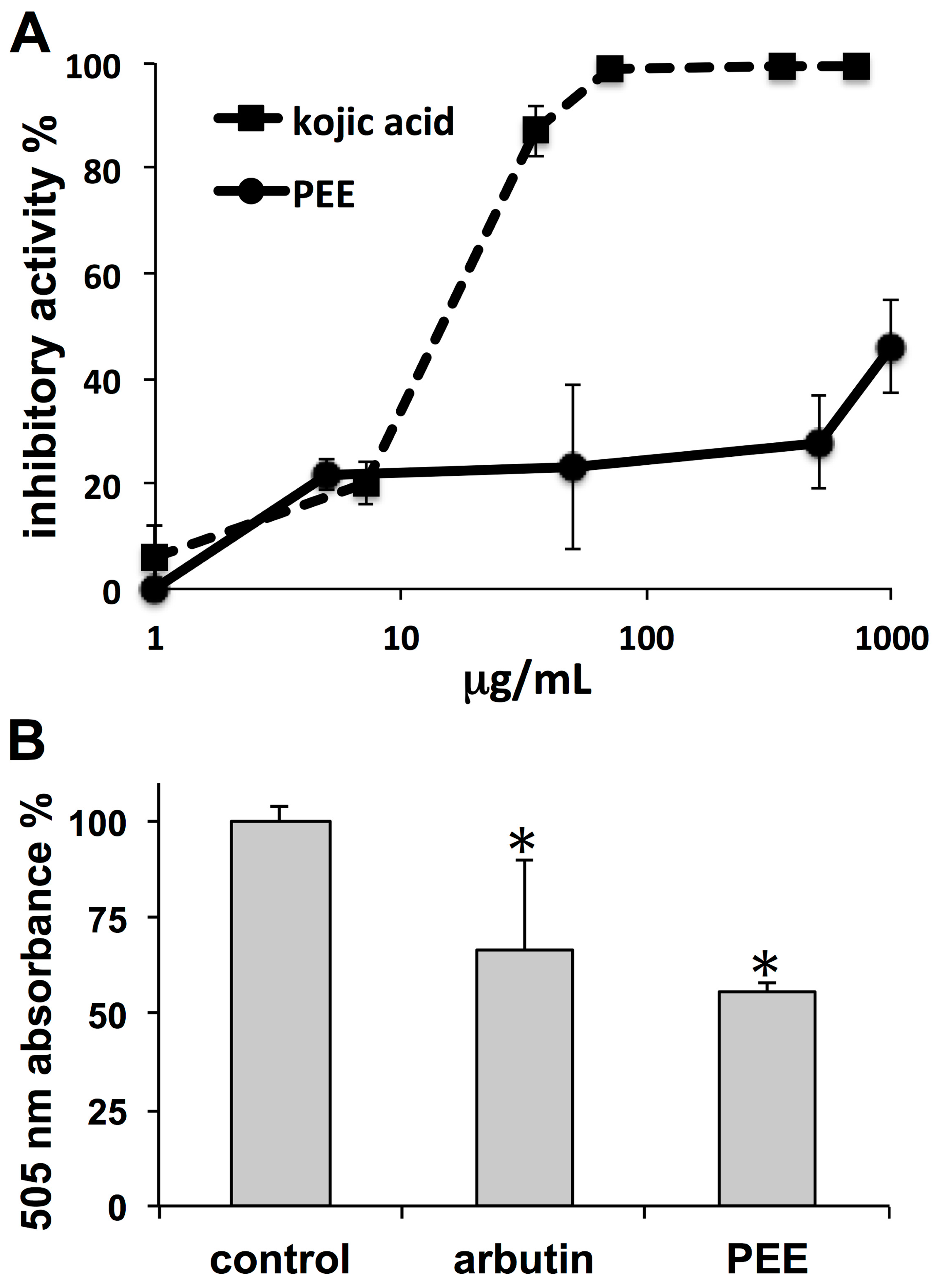
The observed PEE inhibitory activities on both tyrosinase and melanoma pigmentation indicate skin whitening properties. In tyrosinase activity assays, the strength of PEE inhibition was much lower than that of the reference compound kojic acid. Even though a different inhibition profile could be possible using
l-dopa instead of
l-tyrosine as a substrate, the dose-dependent curve of kojic acid fits a logistic trend, consistent with the effect of a single agent, whereas that of PEE diverges from this model, suggesting that the dose-dependent tyrosinase inhibition of PEE could be the result of multiple agents with different inhibitory patterns. In tests on melanoma cells, the PEE demelanizing effect was stronger than that of the reference compound arbutin, confirming that the PEE whitening effect could occur through a complex mechanism, with a component involving tyrosinase inhibition, and another one targeting pathways that regulate cell melanization such as MC1R-MITF [
23]. However, regardless of the mechanism, the data indicate the possible use of PEE in formulations aimed at contrasting hyperpigmentation conditions, such as melasma associated with age, freckling, age spots, post-inflammatory melanization, and sites of actinic damage.
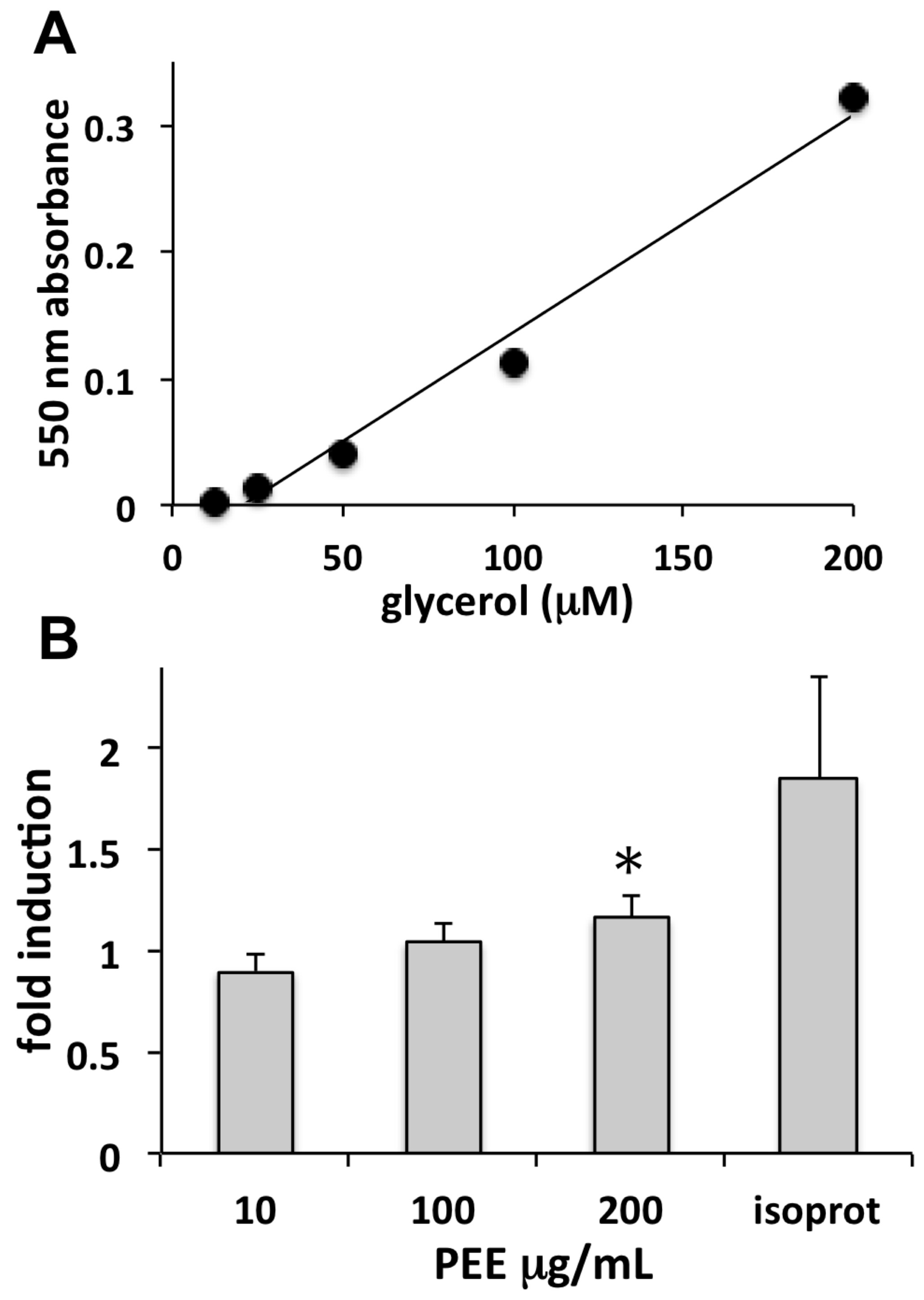
Cellulite is a skin condition associated with hypodermal fat accumulation, commonly occurring as lumps and dimples on women’s thighs. Adipocytes are fat storage cells of adipose tissue that may undergo excessive fat load, leading to the appearance of cellulite. The lipolytic induction of PEE observed in our study is indicative of a possible use in topically applied products aimed at reducing fat accumulation in adipocytes and unaesthetic cellulite.
Among the compounds of PEE that could be responsible for the observed effects, chicoric acid, the most abundant acid found in PEE, has been shown to possess antioxidant, antiangiogenic, anti-inflammatory, antiallergic, antidiabetic, and anti-HIV activities [
24,
25,
26,
27]. Among these, only the strong antioxidant power of chicoric acid can be matched to our data, whereas the other findings from our experiments are completely new for
P. oceanica, and have not been reported for chicoric acid so far. Due to its abundance in the plant and in PEE, the complex consisting of chicoric acid and flavonoids is a major candidate for at least part of the observed PEE effects on skin cells and activities.
In conclusion, we have collected data concerning the biological effects on skin cells of a chicoric acid-rich, ethanolic extract of the seagrass P. oceanica. The obtained results represent a novel discovery among the studies on the biological effects of plant phytocomplexes, since they concern a seagrass that is poorly investigated from this point of view. Our findings indicate that the P. oceanica phytocomplex can be profitably used in the development of products for contrasting wrinkle formation and skin aging, undesired hyperpigmentation, and cellulite.
4. Materials and Methods
4.1. Reagents and Plant Material
Cell culture reagents and other chemicals were from Sigma-Aldrich (St. Louis, MO, USA), unless otherwise indicated. Fresh, beached residues of Posidonia oceanica (L.) Delile seagrass were collected in autumn 2015 at Favignana Island, Sicily, under the supervision of the “Area Marina Protetta Isole Egadi” natural reserve (Favignana, Italy). Plants were determined by one of the authors (LC) and voucher specimens were deposited at the Herbarium of DISTAV, University of Genova, Genova, Italy (GE s.n.).
4.2. Extraction
Soon after collection, leaves were separated from shoots, cleaned manually of basal sheath and epiphytes and rinsed in seawater, dehydrated for 36 h in a forced-ventilation oven at 42 °C, and grounded to a particle size of about 1–2 mm. The pulverized material (100 g) was put in a beaker, extracted under shaking in 60% aq. ethanol (1 L), and acidified with formic acid (pH 4.0) at room temperature for 4 h. The residual of the first extraction was separated from the supernatant and subjected to a second extraction as above. The supernatants of the two extraction steps were mixed together, cloth filtered, and then filtered through a Duran® sintered glass filter disc (DURAN Group GmbH, Wertheim, Germany) and vacuum-dried in a Buchi Rotavapor R-114 (Buchi Italia s.r.l., Cornaredo, Italy) under controlled temperature (<45 °C). The dried P. oceanica ethanolic extract (PEE) was finely pulverized with a mortar and stored at −20 °C until use. The total extraction yield was about 10% (dw/dw).
4.3. Radical Scavenging and Total Polyphenol Assays
Radical scavenging activity of PEE was quantified by the DPPH (2,2-diphenyl-1-picrylhydrazyl) assay, following a well-established protocol [
20]. We used an EtOH solution of DPPH 10
−4 M; a 4:1 DPPH: sample ratio, a reaction time of 30 min, and the absorbance was recorded at 516 nm. After linear regression, the antioxidant activity was calculated as IC
50, i.e., the concentration needed to reduce the initial DPPH absorbance value to half. The above data were also compared to those recorded for a standard solution of ascorbic acid, in order to express the antioxidant power of extracts as ascorbic acid equivalents (AAE) [
28].
The total polyphenol content of PEE was determined by the Folin-Ciocalteu method as previously reported [
29]. A standard curve was obtained from gallic acid samples and total polyphenols in PEE were expressed as mg GAE per g of dry extract.
4.4. ICP-MS
Total iodine determination in PEE was performed by inductively coupled plasma-mass spectrometry, using a Thermo Scientific X Series 2 ICP-MS equipped with a perfluoroalkoxy (PFA) micro-flow concentric nebulizer (Thermo Fisher Scientific, Waltham, MA, USA). The inlet system included a PC3 Peltier chiller (Elemental Scientific, Omaha, NE, USA) and a cyclonic spray chamber. Plasma worked at the power of 1400 W; coolant, auxiliary, and nebulizer flows were set at 14.0, 0.82, and 0.96 L/min, respectively. The peristaltic pump ran at 30%, except for uptake and washout of the sample, set as fast pump (100%). The Collision Cell Technology-Kinetic Energy Discrimination (CCT-KED) mode used an H2/He 8/92 mixture, set at a flow of 5.0 mL/min.
An aliquot of 10 mg of PEE was weighed and put in a glass test tube with 0.15 mL of 69% HNO3, 1.0 mL of 30% H2O2, and 3.0 mL of ultrapure H2O. The test tube was heated at 90 °C in a quartz sand bath for 2 h, and then the volume of the digested solution was increased to 5.0 mL with ultrapure water. Quantitative analysis was performed by means of calibration standard solutions in the range of 1–200 μg/L.
4.5. HPLC-MS
High-performance liquid chromatography coupled with mass spectrometry (HPLC-MS/MS) was performed using an Agilent 1100 HPLC-MSD Ion Trap XCT system, equipped with an electrospray ion source (HPLC-ESI-MS) (Agilent Technologies, Santa Clara, CA, USA). Separation of PEE was performed on a Symmetry C18 column 1 mm × 150 mm with 3 μm particle size (Waters Corporation, Milford, MA, USA). Eluents used were water (eluent A) and MeOH (eluent B), both added with 0.1% formic acid. The gradient employed was: 5% eluent B for 5 min, linear to 40% eluent B in 35 min, then linear gradient to 95% in 15 min, and finally hold at 95% eluent B for another 5 min. The flow rate was set to 30 µL/min and the column temperature was set to 30 °C. The injection volume was 8 μL. Ions were detected in the positive and negative ion mode, in the 200–1000 m/z range, and ion charged control with a target ion value of 200,000 and an accumulation time of 300 ms. A capillary voltage of 3300 V, nebulizer pressure of 15 psi, drying gas of 8 L/min, dry temperature of 325 °C, and 2 rolling averages (averages: 5) were the parameters set for the MS detection. MS/MS analysis was conducted using an amplitude optimized time by time for each compound.
4.6. Cell Culture
Stabilized human dermal fibroblasts (46BR.1N, Sigma-Aldrich, St. Louis, MO, USA) were used for cell proliferation and collagen production assays. Fibroblasts were grown in DMEM, supplemented with 10% fetal bovine serum (FBS) at 37 °C, in a 5% CO2, fully humidified atmosphere.
Cells of the MeWo human melanoma cell line (ATCC, HTB-65) were used for melanin assays. These cells were cultured in RPMI supplemented with 10% FBS, 1% glutamine, and 1% antibiotic mix, at 37 °C in humidified 5% CO2.
Subcutaneous human preadipocytes (Zen-Bio Inc., Research Triangle Park, NC, USA) were used for lipolysis assays. Preadipocytes were grown in Preadipocyte Medium (cat# PM-1, Zen-Bio Inc.) according to the manufacturer’s protocol, at 37 °C in humidified 5% CO2.
4.7. Cell Viability and Proliferation Assays
PEE effects on cell viability were determined on 46BR.1N fibroblasts, MeWo cells, and adipocytes by the MTT assay, as previously reported [
30]. Cells were settled in 96-well plates for 24 h, 10,000 cells per well, and then exposed for 48 h to a logarithmic series of PEE concentrations in DMEM, ranging between 1 and 1000 µg/mL. After the MTT reaction, plates were read at 550 nm in a VMax microplate reader (Molecular Devices, Sunnyvale, CA, USA).
Cell growth rates were determined on 46BR.1N fibroblasts by settling cells in 96-well plates at a density of 5000 cells per well, and then exposing them for periods of 3 and 10 days to 10 or 20 µg/mL PEE. At the end of exposures, an MTT assay was carried out as above, obtaining cell growth curves.
4.8. Collagen Production
The production of collagen type I by 46BR.1N fibroblasts was evaluated by ELISA as previously reported [
30]. Briefly, cells were settled in 96-well plates and incubated with 5, 10, or 20 µg/mL PEE for 48 h. Thereafter, cells were fixed with 3.7% paraformaldehyde, blocked with BSA, probed with mouse anti-human collagen type I (ab6308, Abcam, Cambridge, UK), and then with HRP-conjugated rabbit anti-mouse IgG (ab97046, Abcam), incubated with Pierce 1Step™ Ultra TMB ELISA Substrate Solution (Thermo Fisher Scientific), blocked with 2 M sulfuric acid, and read at 620 nm in the microplate reader.
4.9. Cell-Free Tyrosinase Inhibition Assay
Depigmenting properties of PEE were assessed by the use of an in vitro mushroom tyrosinase inhibition assay [
31]. Aliquots of 10 µL of a solution composed of 125 U/mL mushroom tyrosinase in phosphate buffer (pH 6.8) were added to 96-well plates, followed by 70 µL of phosphate buffer and 60 µL of ultrapure water, or PEE dissolved in ultrapure water, in order to obtain a series of final concentrations ranging between 5 and 1000 µg/mL of PEE. Kojic acid was used instead of PEE as a positive control. Thereafter, 70 µL of 0.3 mg/mL
l-tyrosine in ultrapure water was added. Blanks without enzyme were also included for all conditions. Plates were then incubated at 30 °C for 30 min and absorbance was read at 505 nm in the microplate reader. Percent inhibitory activity (
I%) was calculated according to the formula:
where
Aex/en = absorbance of assay mixture with extract and enzyme;
Aex = absorbance of assay mixture with extract and without enzyme;
Aen = absorbance of assay mixture with enzyme and without extract;
Abk = absorbance of assay mixture without enzyme and extract (blank).
4.10. Melanin Assay
Confluent MeWo cells were suspended and plated in 24-well plates (100,000 cells/well), allowed to settle for 24 h, and then exposed to PEE at a dose of 50 µg/mL for 72 h. Treatment with arbutin (1 mg/mL) was used as a positive control. Thereafter, the culture medium was removed, cells were washed with PBS, trypsinized, centrifuged, and the pellet was subjected to freeze-thawing. The pellet was then dissolved in 100 µL of 1 N NaOH and the crude extract was assayed with the microplate reader at 505 nm to determine the melanin content. All tests were performed in triplicate.
4.11. Lipolysis Assay
The lipolytic effect of PEE was evaluated on primary human adipocytes using the ZenBio Cellulite Treatment Screening Kit Human Adipocyte Lipolysis Assay Kit (cat# LIP-1-L1; LIP-1-NCL1, Zen-Bio Inc., Research Triangle Park, NC, USA) for the detection of free glycerol. Pre-adipocytes were grown as above, settled in 96-well plates, differentiated into adipocytes for one week in Adipocyte Differentiation Medium (cat# DM-2), and maintained for a further week in Adipocyte Medium (cat# AM-1). Fully differentiated adipocytes were incubated for 3 h with 10, 100, or 200 µg/mL PEE, and samples of conditioned medium were then assayed for glycerol as previously reported [
29]. Samples were read in the microplate reader at 550 nm. Absorbance increase is proportional to glycerol concentration in the sample.
4.12. Statistics
Data were analyzed with the R package, version 3.0.1 (
http://www.r-project.org/foundation/), using Student’s
t-test with Bonferroni’s correction for multiple comparisons. The difference between two conditions was considered significant if
p < 0.05.