Reclamation of Marine Chitinous Materials for the Production of α-Glucosidase Inhibitors via Microbial Conversion
Abstract
:1. Introduction
2. Results and Discussion
2.1. Screening of Chitin-Containing Materials as C/N for α-Glucosidase Inhibitors Production
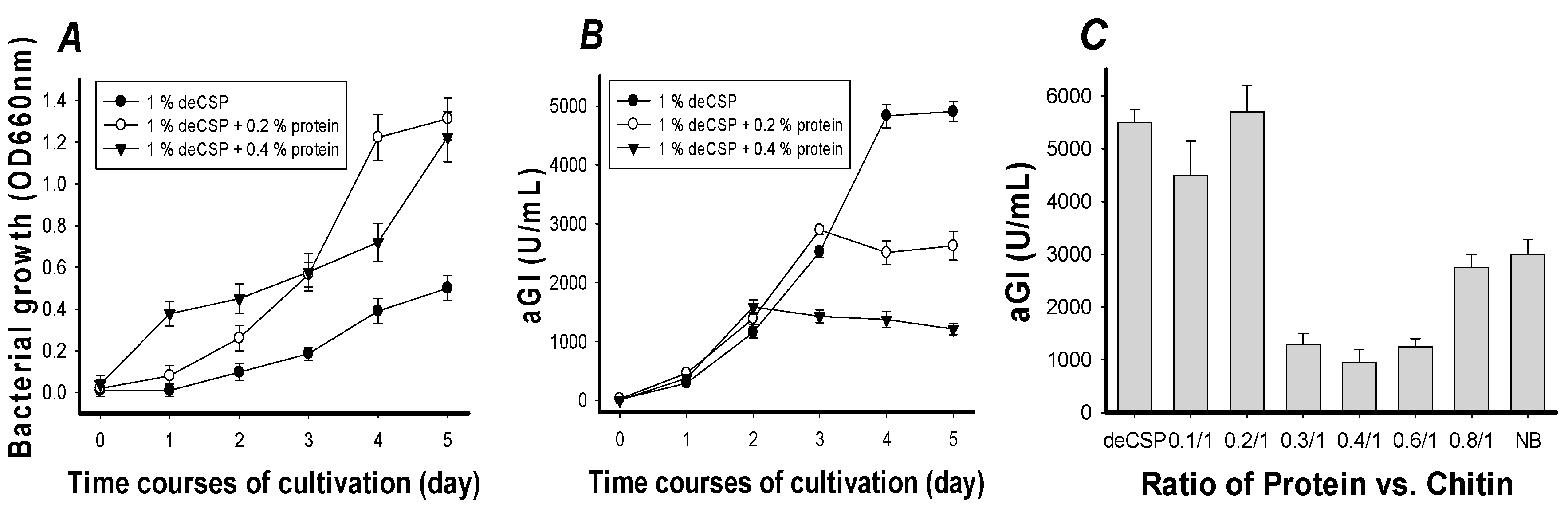
2.2. The Effect of Protein Supplement on α-Glucosidase Inhibitors Production
2.3. Production of aGI from Demineralized Crab Shell Powder by Different Bacteria
2.4. Optimization of Culture Conditions
2.5. Specific α-Glucosidase Inhibitors Activity of Fermented Demineralized Crab Shell Powder
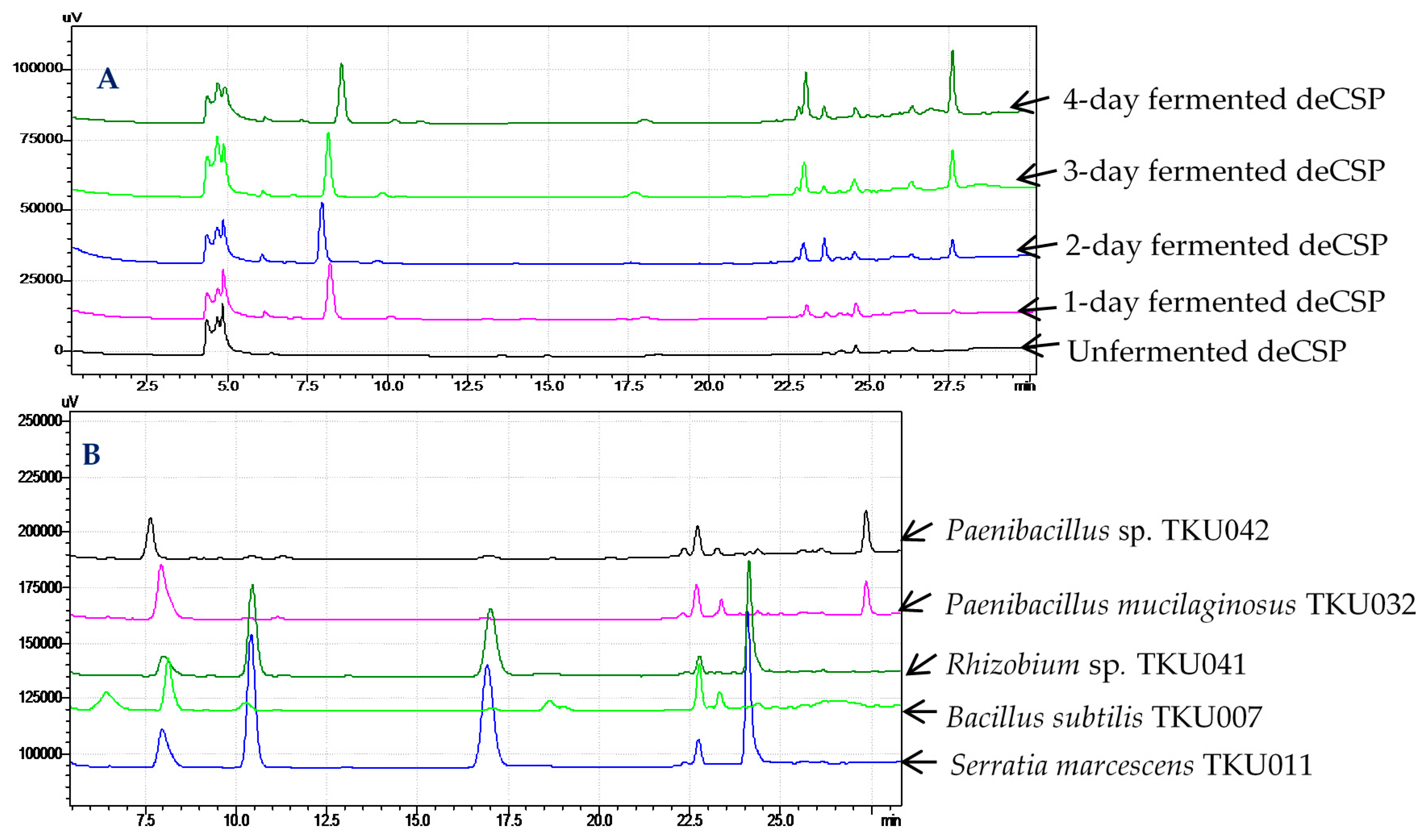
2.6. Confirmation of α-Glucosidase Inhibitors Containing in Fermented Chitin-Containing Media
3. Materials and Methods
3.1. Materials
3.2. Biological Assays of Enzymatic Inhibition
3.3. The Effects of Chitin-Containing Materials and the Protein Supplement on aGI Production Experiments
3.4. Optimization of Some Parameters for α-Glucosidase Inhibitors Productivity Production
3.5. Conditions of High-Performance Liquid Chromatography Fingerprints Analysis
4. Conclusions
Supplementary Materials
Acknowledgments
Authors Contributions
Conflict of Interest
References
- Wang, S.L.; Liang, T. Microbial reclamation of squid pens and shrimp shells. Res. Chem. Intermed. 2017, 43, 3445–3462. [Google Scholar] [CrossRef]
- Nguyen, V.B.; Nguyen, A.D.; Kuo, Y.H.; Wang, S.L. Biosynthesis of α-glucosidase inhibitors by a newly isolated bacterium, Paenibacillus sp. TKU042 and its effect on reducing plasma glucose in mouse model. Int. J. Mol. Sci. 2017, 18, 700. [Google Scholar] [CrossRef] [PubMed]
- Liang, T.W.; Chen, W.T.; Lin, Z.H.; Kuo, Y.H.; Nguyen, A.D.; Pan, P.S.; Wang, S.L. An amphiprotic novel chitosanase from Bacillus mycoides and its application in the production of chitooligomers with their antioxidant and anti-inflammatory evaluation. Int. J. Mol. Sci. 2016, 17, 1302. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, A.D.; Huang, C.C.; Liang, T.W.; Nguyen, V.B.; Pan, P.S.; Wang, S.L. Production and purification of a fungal chitosanase and chitooligomers from Penicillium janthinellum D4 and discovery of the enzyme activators. Carbohydr. Polym. 2014, 108, 331–337. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.L.; Liang, T.W.; Yen, Y.H. Bioconversion of chitin-containing wastes for the production of enzymes and bioactive materials. Carbohydr. Polym. 2011, 84, 732–742. [Google Scholar] [CrossRef]
- Wang, C.L.; Huang, T.H.; Liang, T.W.; Wang, S.L. Production and characterization of exopolysaccharides and antioxidant from Paenibacillus sp. TKU023. New Biotechnol. 2011, 28, 559–565. [Google Scholar] [CrossRef] [PubMed]
- Liang, T.W.; Wu, C.C.; Cheng, W.T.; Chen, Y.C.; Wang, C.L.; Wang, I.L.; Wang, S.L. Exopolysaccharides and antimicrobial biosurfactants produced by Paenibacillus macerans TKU029. Appl. Biochem. Biotechnol. 2014, 172, 933–950. [Google Scholar] [CrossRef] [PubMed]
- Liang, T.W.; Wang, S.L. Recent advances in exopolysaccharides from Paenibacillus spp.: Production, isolation, structure, and bioactivities. Mar. Drugs 2015, 13, 1847–1863. [Google Scholar] [CrossRef] [PubMed]
- Liang, T.W.; Tseng, S.C.; Wang, S.L. Production and characterization of antioxidant properties of exopolysaccharides from Paenibacillus mucilaginosus TKU032. Mar. Drugs 2016, 14, 40. [Google Scholar] [CrossRef] [PubMed]
- Kuo, Y.H.; Liang, T.W.; Liu, K.C.; Hsu, Y.W.; Hsu, H.; Wang, S.L. Isolation and identification of a novel antioxidant with antitumor activity from Serratia ureilytica using squid pen as fermentation substrate. Mar. Biotechnol. 2011, 13, 451–461. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.L.; Wang, C.Y.; Yen, Y.H.; Liang, T.W.; Chen, S.Y.; Chen, C.H. Enhanced production of insecticidal prodigiosin from Serratia marcescens TKU011 in media containing squid pen. Process Biochem. 2012, 47, 1684–1690. [Google Scholar] [CrossRef]
- Liang, T.W.; Chen, C.H.; Wang, S.L. Production of insecticidal materials from Pseudomonas tamsuii. Res. Chem. Intermed. 2015, 41, 7965–7971. [Google Scholar] [CrossRef]
- Wang, S.L.; Huang, T.Y.; Wang, C.Y.; Liang, T.W.; Yen, Y.H.; Sakata, Y. Bioconversion of squid pen by Lactobacillus paracasei subsp paracasei TKU010 for the production of proteases and lettuce enhancing biofertilizers. Biores. Technol. 2008, 99, 5436–5443. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.L.; Chen, S.Y.; Yen, Y.H.; Liang, T.W. Utilization of chitinous materials in pigment adsorption. Food Chem. 2012, 135, 1134–1140. [Google Scholar] [CrossRef] [PubMed]
- Liang, T.W.; Lo, B.C.; Wang, S.L. Chitinolytic bacteria-assisted conversion of squid pen and its effect on dyes and adsorption. Mar. Drugs 2015, 13, 4576–4593. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, V.B.; Nguyen, A.D.; Wang, S.L. Utilization of fishery processing byproduct squid pens for Paenibacillus sp. fermentation on producing potent α-glucosidase inhibitors. Mar. Drugs 2017, 15, 274. [Google Scholar] [CrossRef] [PubMed]
- Kameda, Y.; Asano, N.; Yoshikawa, M.; Takeuchi, M.; Yamaguchi, T.; Matsui, K.; Horii, S.; Fukase, H. Valiolamine, a new α-glucosidase inhibiting amino-cyclitol produced by Streptomyces hygroscopicus. J. Antibiot. 1984, 37, 1301–1307. [Google Scholar] [CrossRef] [PubMed]
- Ezure, Y.; Maruo, S.; Miyazaki, K.; Kawamata, M. Moranoline (1-deoxynojirimycin) fermentation and its improvement. Agric. Biol. Chem. Tokyo 1985, 49, 1119–1125. [Google Scholar]
- Cho, Y.S.; Park, Y.S.; Lee, J.Y.; Kang, K.D.; Hwang, K.Y.; Seong, S.I. Hypoglycemic effect of culture broth of Bacillus subtilis S10 producing 1-deoxynojirimycin. J. Korean Soc. Food Sci. Nutr. 2008, 37, 1401–1407. [Google Scholar] [CrossRef]
- Zhu, Y.P.; Yamaki, K.; Yoshihashi, T.; Ohnishi, K.M.; Li, X.T.; Cheng, Y.Q.; Mori, Y.; Li, L.T. Purification and identification of 1-deoxynojirimycin (DNJ) in okara fermented by Bacillus subtilis B2 from Chinese traditional food (meitaoza). J. Agric. Food Chem. 2010, 58, 4097–4103. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.S.; Lee, J.Y.; Hwang, K.Y.; Cho, Y.S.; Park, Y.S.; Kang, K.D.; Seong, S.I. Isolation and identification of a Bacillus sp. producing α-glucosidase inhibitor 1-deoxynojirimycin. Korean J. Microbiol. Biotechnol. 2011, 39, 49–55. [Google Scholar]
- Onose, S.; Ikeda, R.; Nakagawa, K.; Kimura, T.; Yamagishi, K.; Higuchi, O.; Miyazawa, T. Production of the α-glycosidase inhibitor 1-deoxynojirimycin from Bacillus species. Food Chem. 2013, 138, 516–523. [Google Scholar] [CrossRef] [PubMed]
- Nam, H.; Jung, H.; Karuppasamy, S.; Park, Y.S.; Cho, Y.S.; Lee, J.Y.; Seong, S.; Suh, J.G. Anti-diabetic effect of the soybean extract fermented by Bacillus subtilis MORI in db/db mice. Food Sci. Biotechnol. 2012, 21, 1669–1676. [Google Scholar] [CrossRef]
- Zheng, Y.G.; Xue, Y.P.; Shen, Y.C. Production of valienamine by a newly isolated strain: Stenotrophomonas maltrophilia. Enzyme Microb. Technol. 2006, 39, 1060–1065. [Google Scholar] [CrossRef]
- Schmidt, D.D.; Frommer, W.; Junge, B.; Müller, L.; Wingender, W.; Truscheit, E.; Schäfer, D. α-Glucosidase inhibitors, new complex oligosaccharides of microbial origin. Naturwissenschaften 1977, 64, 535–536. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.L.; Yeh, P.Y. Production of a surfactant- and solvent-stable alkaliphilic protease by bioconversion of shrimp shell wastes fermented by Bacillus subtilis TKU007. Process Biochem. 2006, 41, 1545–1552. [Google Scholar] [CrossRef]
- Wang, S.L.; Yeh, P.Y. Purification and characterization of a chitosanase from a nattokinase producing strain Bacillus subtilis TKU007 using shrimp shell powder as a medium. Process Biochem. 2008, 43, 132–138. [Google Scholar] [CrossRef]
- Wang, S.L.; Wu, Y.Y.; Liang, T.W. Purification and biochemical characterization of a nattokinase by bioconversion of shrimp shell with Bacillus subtilis TKU007. New Biotechnol. 2011, 28, 196–202. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.L.; Kao, D.Y.; Wang, C.L.; Yen, Y.H.; Chern, M.K.; Chen, Y.H. A solvent stable metalloprotease produced by Bacillus sp. TKU004 and its application in the deproteinization of squid pen for beta-chitin preparation. Enzyme Microb. Technol. 2006, 39, 724–731. [Google Scholar] [CrossRef]
- Hong, C.E.; Kwon, S.Y.; Park, J.M. Biocontrol activity of Paenibacillus polymyxa AC-1 against Pseudomonas syringae and its interaction with Arabidopsis thaliana. Microbiol. Res. 2016, 185, 13–21. [Google Scholar] [CrossRef] [PubMed]
- Seo, D.J.; Lee, Y.S.; Kim, K.Y.; Jung, W.J. Antifungal activity of chitinase obtained from Paenibacillus ehimensis MA2012 against conidial of Collectotrichum gloeosporioides in vitro. Microb. Pathog. 2016, 96, 10–14. [Google Scholar] [CrossRef] [PubMed]
- Lal, S.; Tabacchioni, S. Ecology and biotechnological potential of Paenibacillus polymyxa: A minirevie. Indian J. Microbiol. 2009, 49, 2–10. [Google Scholar] [CrossRef] [PubMed]
- Puri, A.; Padda, K.P.; Chanway, C.P. Evidence of nitrogen fixation and growth promotion in canola (Brassica napus L.) by an endophytic diazotroph Paenibacillus polymyxa P2b-2R. Biol. Fertil. Soils 2016, 52, 119–125. [Google Scholar] [CrossRef]
- Wang, S.L.; Li, H.T.; Zhang, L. J.; Lin, Z.H.; Kuo, Y.H. Conversion of squid pen to homogentisic acid via Paenibacillus sp. TKU036 and the antioxidant and anti-inflammatory activities of homogentisic acid. Mar. Drugs 2016, 14, 183. [Google Scholar] [CrossRef] [PubMed]
- Kwon, Y.I.; Vattem, D.A.; Shetty, K. Evaluation of clonal herbs Lamiaceae species for management of diabetes and hypertension. Asia Pac. J. Clin. Nutr. 2006, 15, 107–118. [Google Scholar] [PubMed]
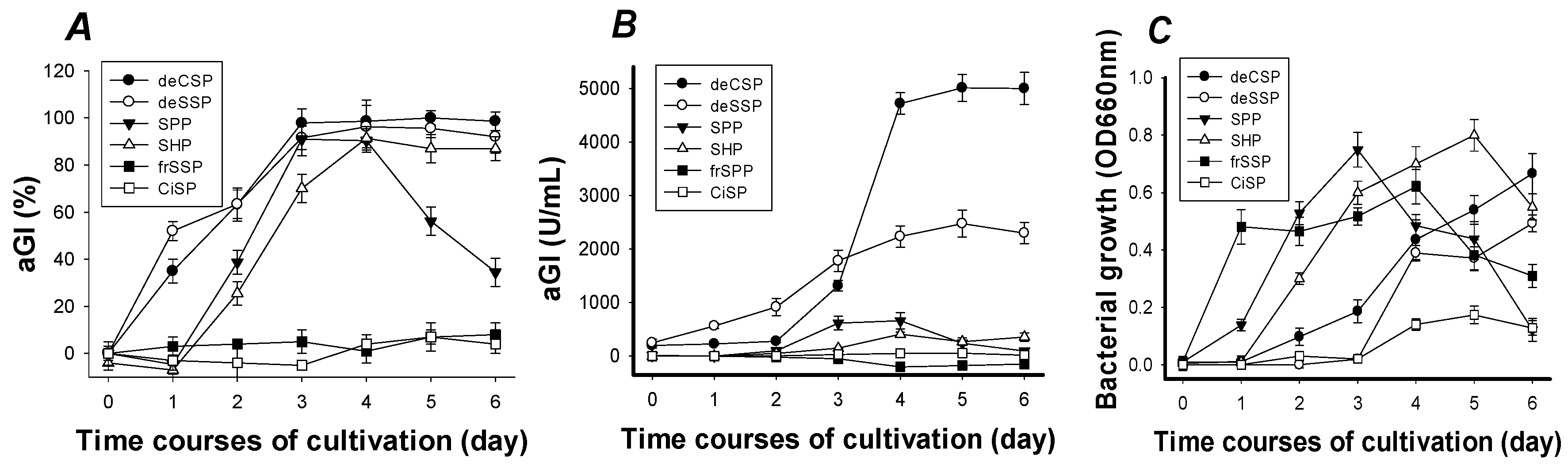
| FCMs | Cultivation Time (Day) | Yield of Production (kU/g) a | α-Glucosidase Inhibition | |
|---|---|---|---|---|
| IC50 (µg/mL) | Maximal Inhibition (%) b | |||
| deCSP | 4 | 26,316 | 38 ± 4.1 | 98 ± 3.7 |
| deSSP | 4 | 9259 | 108 ± 5.2 | 89 ± 3.6 |
| SPP | 3 | 2370 | 422 ± 19 | 98 ± 1.9 |
| SHP | 4 | 2198 | 455 ± 42 | 92 ± 4.3 |
| FNB c | 4 | 12,346 | 81 ± 4.3 | 93 ± 4.2 |
| acarbose d | 913 | 1095 ± 93 | 74 ± 3.4 | |
| No. | Bacterial Strain | Sources of α-Glucosidase | ||
|---|---|---|---|---|
| S | B | R | ||
| 1 | Bacillus licheniformis TKU004 | - | - | - |
| 2 | Bacillus subtilis TKU007 | - | - | - |
| 3 | Bacillus mycoides TKU038 | - | - | - |
| 4 | Bacillus mycoildes TKU040 | - | - | - |
| 5 | Paenibacillus macerans TKU029 | 96 | 90 | 92 |
| 6 | Paenibacillus mucilaginosus TKU032 | 98 | 95 | 95 |
| 7 | Paenibacillus sp. TKU037 | 99 | 95 | 90 |
| 8 | Paenibacillus sp. TKU042 | 99 | 97 | 96 |
| 9 | Serratia marcescens TKU011 | - | - | - |
| 10 | Serratia ureilytica TKU013 | - | - | - |
| 11 | Serratia marcescens TKU019 | - | - | - |
| 12 | Lactobacillus paracasei TKU010 | - | - | - |
| 13 | Pseudomonas tamsuii TKU015 | - | - | - |
| 14 | Serratia sp. TKU016 | - | - | - |
| 15 | Serratia sp. TKU020 | - | - | - |
| 16 | Rhizobium sp. TKU041 | - | - | - |
| Control (medium only) | - | - | - | |
| Compared Factors | Before Optimization * | After Optimization |
|---|---|---|
| C/N source | NB | deCSP |
| Cultivation temperature (°C) | 30 | 30 |
| C/N Concentration (%) | 0.8 | 1.6 |
| Cultivation time (day) | 4 | 4 |
| Medium/flask volume ratio | 100/250 | 130/250 |
| Seed culture (%) | 1 | 2 |
| Inhibition (IC50 μg/mL) | 81 ± 4.3 | 6.7 ± 0.31 |
| aGI productivity (U/mL) | 5000 | 12,400 |
| Enzyme | Inhibition by deCSP | Inhibition by Acarbose | ||
|---|---|---|---|---|
| IC50 (µg/mL) | Max. Inh. Activity (%) * | IC50 (µg/mL) | Max. Inh. Activity (%) * | |
| S. cerevisiae α-glucosidase | 6.7 ± 0.31 | 99 ± 2.2 | 1095 | 74 ± 3.4 ** |
| Rat intestinal α-glucosidase | 15.9 ± 0.7 | 97 ± 2.7 | 78 ± 3.2 | 91 ± 3.1 |
| B. stearothermophilus α-glucosidase | 6.6 ± 0.22 | 95 ± 2.3 | 0.042 ± 0.003 | 99 ± 1.7 |
| Rice α-glucosidase | 6.7 ± 0.25 | 96 ± 1.9 | 3.04 ± 0.82 | 100 ± 2.1 |
| Porcine pancreatic α-amylase | - | - | ND | ND |
| B. subtilis α-amylase | - | - | ND | ND |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nguyen, V.B.; Wang, S.-L. Reclamation of Marine Chitinous Materials for the Production of α-Glucosidase Inhibitors via Microbial Conversion. Mar. Drugs 2017, 15, 350. https://doi.org/10.3390/md15110350
Nguyen VB, Wang S-L. Reclamation of Marine Chitinous Materials for the Production of α-Glucosidase Inhibitors via Microbial Conversion. Marine Drugs. 2017; 15(11):350. https://doi.org/10.3390/md15110350
Chicago/Turabian StyleNguyen, Van Bon, and San-Lang Wang. 2017. "Reclamation of Marine Chitinous Materials for the Production of α-Glucosidase Inhibitors via Microbial Conversion" Marine Drugs 15, no. 11: 350. https://doi.org/10.3390/md15110350
APA StyleNguyen, V. B., & Wang, S.-L. (2017). Reclamation of Marine Chitinous Materials for the Production of α-Glucosidase Inhibitors via Microbial Conversion. Marine Drugs, 15(11), 350. https://doi.org/10.3390/md15110350







