Are We Overtesting? Rethinking Routine Blood Work After Low-Risk Laparoscopic Cholecystectomy: A Retrospective Study
Abstract
1. Introduction
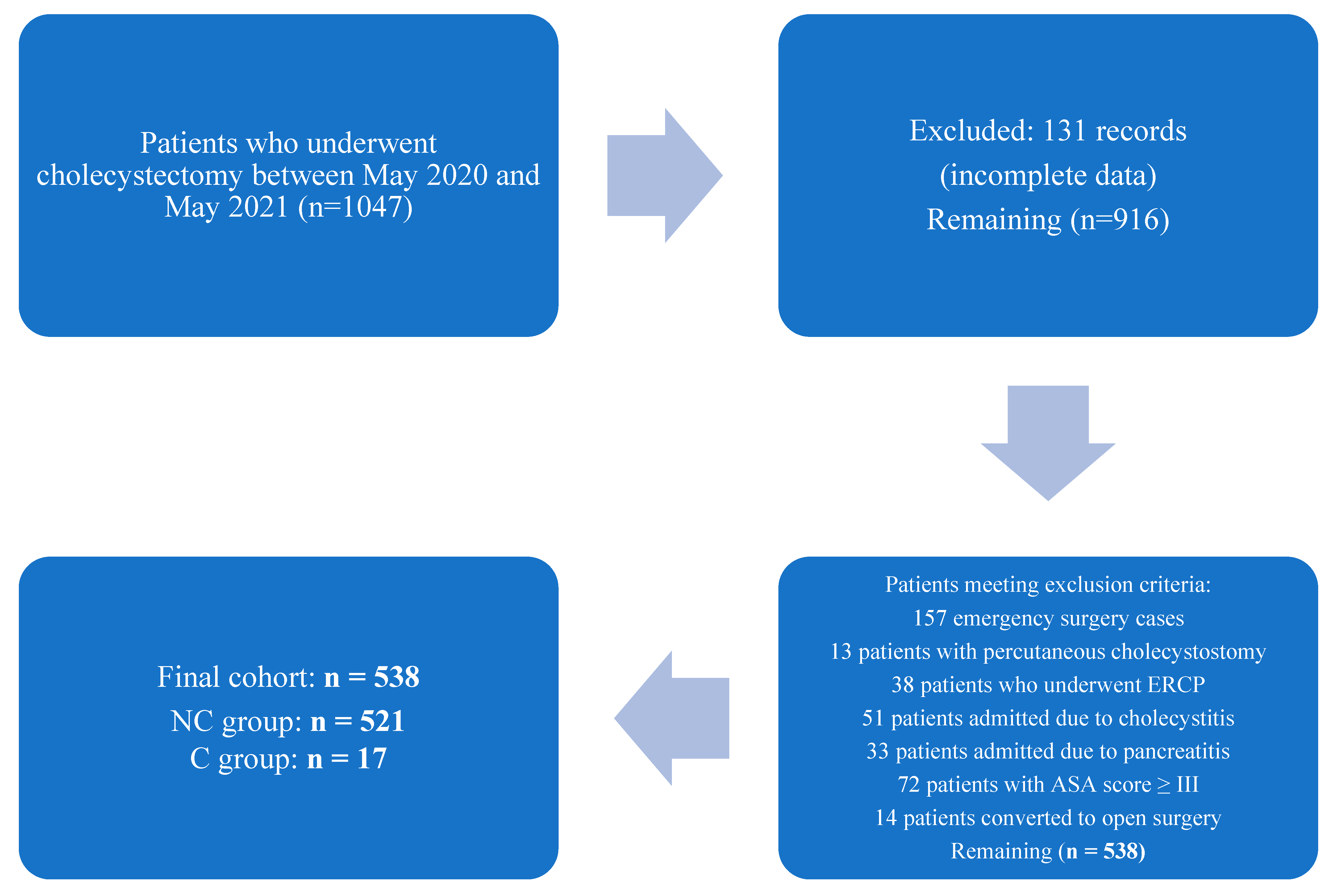
2. Materials and Method
2.1. Surgical Technique
2.2. Biostatistical Data Analysis
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Everhart, J.E.; Ruhl, C.E. Burden of digestive diseases in the United States Part III: Liver, biliary tract, and pancreas. Gastroenterology 2009, 136, 1134–1144. [Google Scholar] [CrossRef]
- Kimura, Y.; Takada, T.; Strasberg, S.M.; Pitt, H.A.; Gouma, D.J.; Garden, O.J.; Büchler, M.W.; Windsor, J.A.; Mayumi, T.; Yoshida, M.; et al. TG13 current terminology, etiology, and epidemiology of acute cholangitis and cholecystitis. J. Hepato-Biliary-Pancreat. Sci. 2013, 20, 8–23. [Google Scholar] [CrossRef]
- Nimptsch, U.; Mansk, T. Deaths Following Cholecystectomy and Herniotomy: An Analysis of Nationwide German Hospital Discharge Data From 2009 to 2013. Dtsch. Ärzteblatt Int. 2015, 112, 535–543. [Google Scholar] [CrossRef][Green Version]
- Pochhammer, J.; Blumenstock, G.; Schäffer, M. Routine ultrasound and blood tests after laparoscopic cholecystectomy-are they worthwhile? A diagnostic accuracy study. Langenbeck’s Arch. Surg. 2016, 401, 489–494. [Google Scholar] [CrossRef]
- Scollay, J.M.; Mullen, R.; McPhillips, G.; Thompson, A.M. Mortality associated with the treatment of gallstone disease: A 10-year contemporary national experience. World J. Surg. 2011, 35, 643–647. [Google Scholar] [CrossRef] [PubMed]
- Shamiyeh, A.; Wayand, W. Laparoscopic cholecystectomy: Early and late complications and their treatment. Langenbeck’s Arch. Surg. 2004, 389, 164–171. [Google Scholar] [CrossRef]
- Sahajpal, A.K.; Chow, S.C.; Dixon, E.; Greig, P.D.; Gallinger, S.; Wei, A.C. Bile duct injuries associated with laparoscopic cholecystectomy: Timing of repair and long-term outcomes. Arch. Surg. 2010, 145, 757–763. [Google Scholar] [CrossRef]
- Wakabayashi, G.; Iwashita, Y.; Hibi, T.; Takada, T.; Strasberg, S.M.; Asbun, H.J.; Endo, I.; Umezawa, A.; Asai, K.; Suzuki, K.; et al. Tokyo Guidelines 2018: Surgical management of acute cholecystitis: Safe steps in laparoscopic cholecystectomy for acute cholecystitis (with videos). J. Hepato-Biliary-Pancreat. Sci. 2018, 25, 73–86. [Google Scholar] [CrossRef] [PubMed]
- Scurr, J.R.; Brigstocke, J.R.; Shields, D.A.; Scurr, J.H. Medicolegal claims following laparoscopic cholecystectomy in the UK and Ireland. Ann. R. Coll. Surg. Engl. 2010, 92, 286–291. [Google Scholar] [CrossRef] [PubMed]
- Botaitis, S.; Pitiakoudis, M.; Perente, S.; Tripsianis, G.; Polychronidis, A.; Simopoulos, C. Laparoscopic cholecystectomy in acute cholecystitis: An analysis of the risk factors. S. Afr. J. Surg. 2012, 50, 62–68. [Google Scholar] [CrossRef]
- Lee, N.W.; Collins, J.; Britt, R.; Britt, L.D. Evaluation of preoperative risk factors for converting laparoscopic to open cholecystectomy. Am. Surg. 2012, 78, 831–833. [Google Scholar] [CrossRef]
- Cwik, G.; Skoczylas, T.; Wyroślak-Najs, J.; Wallner, G. The value of percutaneous ultrasound in predicting conversion from laparoscopic to open cholecystectomy due to acute cholecystitis. Surg. Endosc. 2013, 27, 2561–2568. [Google Scholar] [CrossRef]
- Ábrahám, S.; Németh, T.; Benkő, R.; Matuz, M.; Váczi, D.; Tóth, I.; Ottlakán, A.; Andrási, L.; Tajti, J.; Kovács, V.; et al. Evaluation of the conversion rate as it relates to preoperative risk factors and surgeon experience: A retrospective study of 4013 patients undergoing elective laparoscopic cholecystectomy. BMC Surg. 2021, 21, 151. [Google Scholar] [CrossRef] [PubMed]
- Chin, X.; Mallika Arachchige, S.; Orbell-Smith, J.; Wysocki, A.P. Preoperative and Intraoperative Risk Factors for Conversion of Laparoscopic Cholecystectomy to Open Cholecystectomy: A Systematic Review of 30 Studies. Cureus 2023, 15, e47774. [Google Scholar] [CrossRef] [PubMed]
- Asai, K.; Watanabe, M.; Kusachi, S.; Matsukiyo, H.; Saito, T.; Kodama, H.; Kiribayashi, T.; Enomoto, T.; Nakamura, Y.; Okamoto, Y.; et al. Risk factors for conversion of laparoscopic cholecystectomy to open surgery associated with the severity characteristics according to the Tokyo guidelines. Surg. Today 2014, 44, 2300–2304. [Google Scholar] [CrossRef] [PubMed]
- Cox, M.R.; Budge, J.P.; Eslick, G.D. Timing and nature of presentation of unsuspected retained common bile duct stones after laparoscopic cholecystectomy: A retrospective study. Surg. Endosc. 2015, 29, 2033–2038. [Google Scholar] [CrossRef]
- Müller, S.; Falch, C.; Kreuzer, J.; Storz, P.; Konigsrainer, A.; Kirschniak, A. Utility of routine ultrasound after laparoscopic cholecystectomy to detect early postoperative complication. Dig. Surg. 2012, 29, 183–186. [Google Scholar] [CrossRef]
- Videhult, P.; Sandblom, G.; Rudberg, C.; Rasmussen, I.C. Are liver function tests, pancreatitis and cholecystitis predictors of common bile duct stones? Results of a prospective, population-based, cohort study of 1171 patients undergoing cholecystectomy. HPB 2011, 13, 519–527. [Google Scholar] [CrossRef]
- Picchio, M.; Lucarelli, P.; Di Filippo, A.; De Angelis, F.; Stipa, F.; Spaziani, E. Meta-analysis of drainage versus no drainage after laparoscopic cholecystectomy. JSLS 2014, 18, e2014.00242. [Google Scholar] [CrossRef]
- Lee, S.J.; Choi, I.S.; Moon, J.I.; Yoon, D.S.; Choi, W.J.; Lee, S.E.; Sung, N.S.; Kwon, S.U.; Bae, I.E.; Roh, S.J.; et al. Optimal drain management following complicated laparoscopic cholecystectomy for acute cholecystitis: A propensity-matched comparative study. J. Minim. Invasive Surg. 2022, 25, 63–72. [Google Scholar] [CrossRef]
- Tan, M.; Xu, F.F.; Peng, J.S.; Li, D.M.; Chen, L.H.; Lv, B.J.; Zhao, Z.X.; Huang, C.; Zheng, C.X. Changes in the level of serum liver enzymes after laparoscopic surgery. World J. Gastroenterol. 2003, 9, 364–367. [Google Scholar] [CrossRef]
- Ahmad, N.Z. Routine testing of liver function before and after elective laparoscopic cholecystectomy: Is it necessary? JSLS 2011, 15, 65–69. [Google Scholar] [CrossRef]
- Válek, V.; Kala, Z.; Kysela, P. Biliary tree and cholecyst: Post surgery imaging. Eur. J. Radiol. 2005, 53, 433–440. [Google Scholar] [CrossRef]
- Hobbs, M.S.; Mai, Q.; Knuiman, M.W.; Fletcher, D.R.; Ridout, S.C. Surgeon experience and trends in intraoperative complications in laparoscopic cholecystectomy. Br. J. Surg. 2006, 93, 844–853. [Google Scholar] [CrossRef]
- Strohäker, J.; Wiegand, L.; Beltzer, C.; Königsrainer, A.; Ladurner, R.; Bachmann, R. Routine postoperative blood tests fail to reliably predict procedure-related complications after laparoscopic cholecystectomy. Langenbeck’s Arch. Surg. 2021, 406, 1155–1163. [Google Scholar] [CrossRef] [PubMed]
- Ben-Ishay, O.; Zeltser, M.; Kluger, Y. Utility of routine blood tests after elective laparoscopic cholecystectomy for symptomatic gallstones. World J. Gastrointest. Surg. 2017, 9, 149–152. [Google Scholar] [CrossRef] [PubMed]
- Vyas, F.L.; Nayak, S.; Perakath, B.; Pradhan, N.R. Gallbladder remnant and cystic duct stump calculus as a cause of postcholecystectomy syndrome. Trop. Gastroenterol. 2005, 26, 159–160. [Google Scholar]
- Kim, J.Y.; Kim, K.W.; Ahn, C.S.; Hwang, S.; Lee, Y.J.; Shin, Y.M.; Lee, M.G. Spectrum of biliary and nonbiliary complications after laparoscopic cholecystectomy: Radiologic findings. AJR Am. J. Roentgenol. 2008, 191, 783–789. [Google Scholar] [CrossRef] [PubMed]
- Walsh, R.M.; Chung, R.S.; Grundfest-Broniatowski, S. Incomplete excision of the gallbladder during laparoscopic cholecystectomy. Surg. Endosc. 1995, 9, 67–70. [Google Scholar] [CrossRef]
- Hellmig, S.; Katsoulis, S.; Fölsch, U. Symptomatic cholecystolithiasis after laparoscopic cholecystectomy. Surg. Endosc. 2004, 18, 347. [Google Scholar] [CrossRef]
- Gustafsson, A.; Enochsson, L.; Tingstedt, B.; Olsson, G. Bile leakage and the number of metal clips on the cystic duct during laparoscopic cholecystectomy. Scand. J. Surg. 2022, 111, 14574969221102284. [Google Scholar] [CrossRef]
- Ai, X.M.; Ho, L.C.; Yang, N.Y.; Han, L.L.; Lu, J.J.; Yue, X. A comparative study of ultrasonic scalpel (US) versus conventional metal clips for closure of the cystic duct in laparoscopic cholecystectomy (LC): A meta-analysis. Medicine 2018, 97, e13735. [Google Scholar] [CrossRef] [PubMed]
- Chowbey, P.; Sharma, A.; Goswami, A.; Afaque, Y.; Najma, K.; Baijal, M.; Soni, V.; Khullar, R. Residual gallbladder stones after cholecystectomy: A literature review. J. Minimal Access Surg. 2015, 11, 223–230. [Google Scholar] [CrossRef]
- Li, L.B.; Cai, X.J.; Mou, Y.P.; Wei, Q. Reoperation of biliary tract by laparoscopy: Experiences with 39 cases. World J. Gastroenterol. 2008, 14, 3081–3084. [Google Scholar] [CrossRef]
- Ravendran, K.; Elmoraly, A.; Kagiosi, E.; Henry, C.S.; Joseph, J.M.; Kam, C. Converting From Laparoscopic Cholecystectomy to Open Cholecystectomy: A Systematic Review of Its Advantages and Reasoning. Cureus 2024, 16, e64694. [Google Scholar] [CrossRef]
- Salky, B.A.; Edye, M.B. The difficult cholecystectomy: Problems related to concomitant diseases. Semin. Laparosc. Surg. 1998, 5, 107–114. [Google Scholar] [CrossRef]
- Amreek, F.; Hussain, S.Z.M.; Mnagi, M.H.; Rizwan, A. Retrospective Analysis of Complications Associated with Laparoscopic Cholecystectomy for Symptomatic Gallstones. Cureus 2019, 11, e5152. [Google Scholar] [CrossRef] [PubMed]
- Karayiannakis, A.J.; Makri, G.G.; Mantzioka, A.; Karousos, D.; Karatzas, G. Postoperative pulmonary function after laparoscopic and open cholecystectomy. Br. J. Anaesth. 1996, 77, 448–452. [Google Scholar] [CrossRef] [PubMed]
- Gautam, N.; Harjai, M.; Sharma, P.; Rai, S.; Tripathi, M.; Malviya, D.; Singh, A.K.; Gautam Sr, N.; Harjai, M. Anaesthetic Management and Physiologic Effects of Pneumoperitoneum in Patients With Chronic Obstructive Pulmonary Disease Undergoing Laparoscopic Cholecystectomy. Cureus 2023, 15, e46458. [Google Scholar] [CrossRef] [PubMed]
- Bablekos, G.D.; Michaelides, S.A.; Analitis, A.; Charalabopoulos, K.A. Effects of laparoscopic cholecystectomy on lung function: A systematic review. World J. Gastroenterol. 2014, 20, 17603–17617. [Google Scholar] [CrossRef]
- Gurusamy, K.S.; Koti, R.; Davidson, B.R. Abdominal lift for laparoscopic cholecystectomy. Cochrane Database Syst. Rev. 2013, 2013, CD006574. [Google Scholar] [CrossRef] [PubMed]
- Gamo, G.O.; Reichardt, G.S.; Guetter, C.R.; Pimentel, S.K. Risk factors for surgical wound infection after elective laparoscopic cholecystectomy. Arq. Bras. Cir. Dig. 2022, 35, e1675. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Ospina, J.C.; Zapata-Copete, J.A.; Bejarano, M.; García-Perdomo, H.A. Antibiotic Prophylaxis in Elective Laparoscopic Cholecystectomy: A Systematic Review and Network Meta-Analysis. J. Gastrointest. Surg. 2018, 22, 1193–1203. [Google Scholar] [CrossRef]
- Ely, S.; Rothenberg, K.A.; Beattie, G.; Gologorsky, R.C.; Huyser, M.R.; Chang, C.K. Modern Elective Laparoscopic Cholecystectomy Carries Extremely Low Postoperative Infection Risk. J. Surg. Res. 2020, 246, 506–511. [Google Scholar] [CrossRef] [PubMed]
| Patient | n = 538 |
| Age (yr) * | 45.1 (18–85) |
| Gender ** | |
| ● Female | 379 (70.4%) |
| ● Male | 159 (29.6%) |
| Duration of surgery (min) * | 58.4 (48–69) |
| Postoperative complications ** | 17 (3.15%) |
| Variables | Group NC (n = 521) | Group C (n = 17) | p Value |
|---|---|---|---|
| Age (yr) * | 45.4 (18–85) | 47.1 (39–54) | p = 0.616 |
| Etiology ** | p = 0.663 | ||
| Gallbladder stones | 515 (98.85%) | 17 (100%) | |
| Gallbladder polyps | 1 (0.19%) | 0 (0) | |
| Gallbladder stones and polyps | 5 (0.96%) | 0 (0) | |
| Length of hospital stay * | 1.41 (1–4) | 6.41 (2–10) | p = 0.0009 |
| Pre-op blood test parameters * | |||
| WBC (4.49–12.68 × 109/L) | 7.38 (6.1–8.56) | 8.02 (6.73–9.6) | p = 0.94 |
| CRP (0.5–5 mg/L) | 1.7 (1–4.1) | 3.4 (1.15–4.6) | p = 0.329 |
| T. bilirubin (0–1.2 mg/dL) | 0.39 (0.26–0.59) | 0.42 (0.33–0.61) | p = 0.182 |
| D. bilirubin (0–0.3 mg/dL) | 0.15 (0.11–0.22) | 0.15 (0.07–0.21) | p = 0.731 |
| ALT (0–35 U/L) | 17 (12.5–26) | 25 (15–35) | p = 0.062 |
| AST (10–35 U/L) | 17 (14–21.5) | 24 (18–31) | p = 0.003 |
| ALP (35–104 U/L) | 68 (55–84) | 63 (49–85) | p = 0.495 |
| GGT (5–36 U/L) | 21 (14–35) | 18 (14–38.5) | p = 0.982 |
| Amylase (28–100 U/L) | 57 (47–71) | 55 (35.5–78.5) | p = 0.525 |
| Lipase (13–60 U/L) | 29 (22–40) | 37 (29.5–46) | p = 0.068 |
| Variables | Group NC (n = 521) | Group C (n = 17) |
|---|---|---|
| Cystic duct ligation type * | ||
| ● Endoclip | 500 (95.97%) | 17 (100%) |
| ● Hemoclip | 20 (3.84%) | 0 (0) |
| ● Stapler | 1 (0.19%) | 0 (0) |
| Drain * | ||
| ● Yes | 301 (57.77%) | 10 (58.82%) |
| ● No | 220 (42.23%) | 7 (41.18%) |
| Intraoperative findings | ||
| Floppy gallbladder | 487 | 11 |
| Adhesion (mild or high) | 8 | 2 |
| Anatomic difficulty ** | 11 | 2 |
| Buried gallbladder | 9 | 1 |
| Impacted stone > 1 cm | 6 | 1 |
| Variables | Group NC * (n = 521) | Group C * (n = 17) | p Value |
|---|---|---|---|
| Post-op blood test parameters * | |||
| WBC (4.49–12.68 × 109/L) | 10.15 (8.44–12.36) | 10.35 (8.5–12.33) | p = 0.966 |
| CRP (0.5–5 mg/L) | 6.7 (3.5–12) | 11.3 (4.62–18.7) | p = 0.171 |
| T. bilirubin (0–1.2 mg/dL) | 0.46 (0.31–0.66) | 0.7 (0.48–0.93) | p = 0.005 |
| D. bilirubin (0–0.3 mg/dL) | 0.18 (0.14–0.27) | 0.28 (0.14–0.46) | p = 0.062 |
| ALT (0–35 U/L) | 34 (23–53) | 61 (33.25–148.5) | p = 0.002 |
| AST (10–35 U/L) | 33 (24–46) | 54 (33.25–147.5) | p = 0.002 |
| ALP (35–104 U/L) | 65 (55–81) | 72.5 (57.75–97) | p = 0.205 |
| GGT (5–36 U/L) | 24 (14–47) | 62 (23–98) | p = 0.02 |
| Amylase (28–100 U/L) | 48 (37–63) | 65.5 (46.5–97.75) | p = 0.034 |
| Lipase (13–60 U/L) | 23 (17–32) | 28 (21.5–41.25) | p = 0.184 |
| Overall complications ** | n = 0 | n = 17 (3.15%) | |
| Wound infection | 0 | 1 | |
| Bile leakage | 0 | 1 | |
| Choledocholithiasis | 0 | 2 | |
| Cardiac complications | 0 | 2 | |
| Pulmonary complications | 0 | 9 | |
| Hemorrhage | 0 | 2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Published by MDPI on behalf of the Lithuanian University of Health Sciences. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Demir, M.; Kilavuz, H.; Gungor, F.; Yaman, S.; Ekci, B.; Kurtulus, I. Are We Overtesting? Rethinking Routine Blood Work After Low-Risk Laparoscopic Cholecystectomy: A Retrospective Study. Medicina 2025, 61, 1555. https://doi.org/10.3390/medicina61091555
Demir M, Kilavuz H, Gungor F, Yaman S, Ekci B, Kurtulus I. Are We Overtesting? Rethinking Routine Blood Work After Low-Risk Laparoscopic Cholecystectomy: A Retrospective Study. Medicina. 2025; 61(9):1555. https://doi.org/10.3390/medicina61091555
Chicago/Turabian StyleDemir, Murat, Huseyin Kilavuz, Feyyaz Gungor, Sibel Yaman, Baki Ekci, and Idris Kurtulus. 2025. "Are We Overtesting? Rethinking Routine Blood Work After Low-Risk Laparoscopic Cholecystectomy: A Retrospective Study" Medicina 61, no. 9: 1555. https://doi.org/10.3390/medicina61091555
APA StyleDemir, M., Kilavuz, H., Gungor, F., Yaman, S., Ekci, B., & Kurtulus, I. (2025). Are We Overtesting? Rethinking Routine Blood Work After Low-Risk Laparoscopic Cholecystectomy: A Retrospective Study. Medicina, 61(9), 1555. https://doi.org/10.3390/medicina61091555






