Sex-Specific Longitudinal Changes in Metabolic, Endocrine, Renal, Cardiovascular, and Inflammatory Biomarkers of Vaccinated COVID-19 Survivors: 30-Month Follow-Up Study
Abstract
1. Introduction
2. Materials and Methods
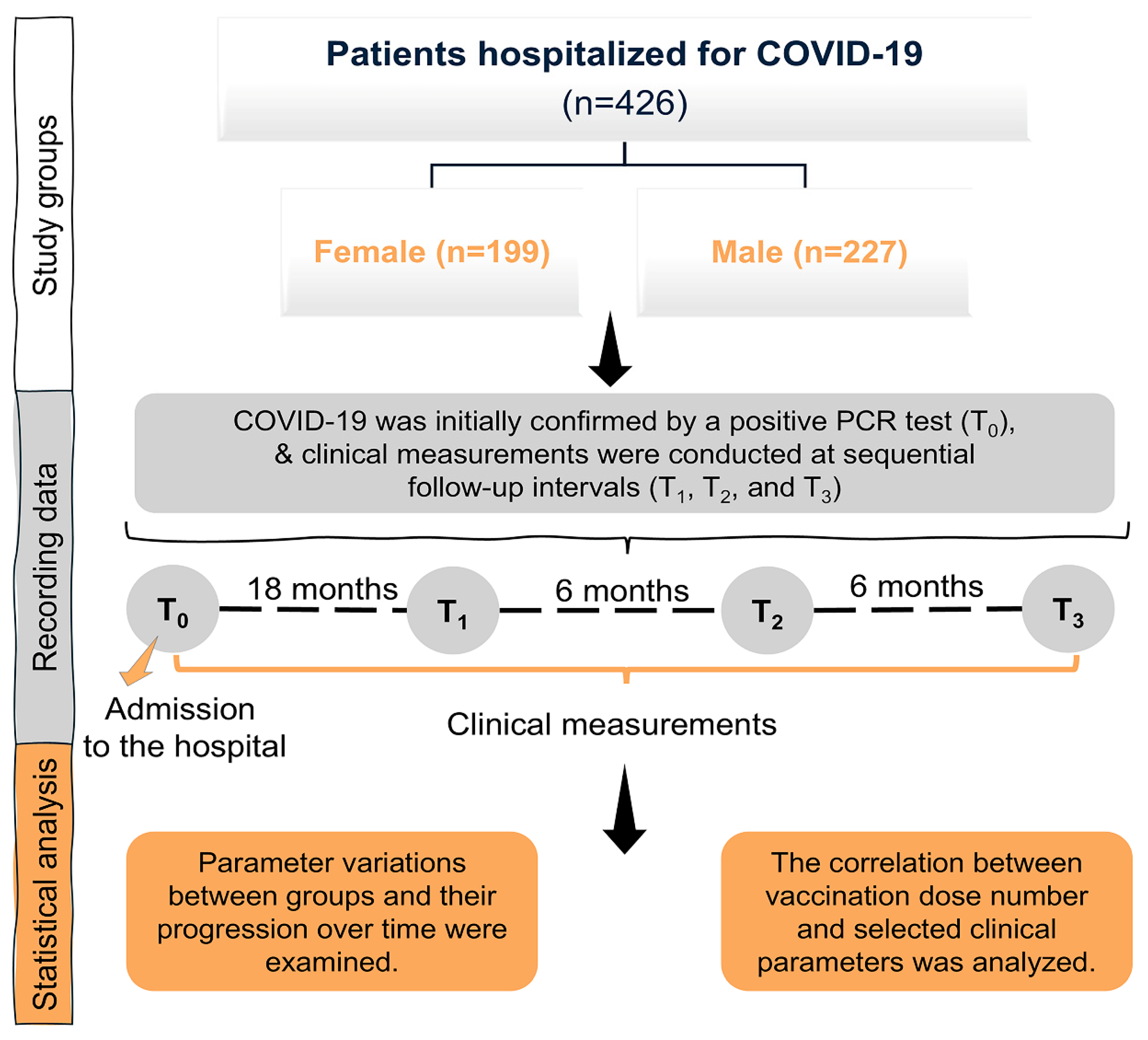
2.1. Study Design and Setting
2.2. Study Population and Cohort Characteristics
2.3. Data Acquisition and Laboratory Assessments
2.4. Ethical Considerations
2.5. Statistical Analysis
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gupta, A.; Madhavan, M.V.; Sehgal, K.; Nair, N.; Mahajan, S.; Sehrawat, T.S.; Bikdeli, B.; Ahluwalia, N.; Ausiello, J.C.; Wan, E.Y.; et al. Extrapulmonary manifestations of COVID-19. Nat. Med. 2020, 26, 1017–1032. [Google Scholar] [CrossRef]
- Nalbandian, A.; Sehgal, K.; Gupta, A.; Madhavan, M.V.; McGroder, C.; Stevens, J.S.; Cook, J.R.; Nordvig, A.S.; Shalev, D.; Sehrawat, T.S.; et al. Post-acute COVID-19 syndrome. Nat. Med. 2021, 27, 601–615. [Google Scholar] [CrossRef]
- Hancı, P.; Uysal, A.; Yüksel, B.; İnal, V. Rox Index Dynamics According to High Flow Nasal Cannula Success in Intensive Care Unit Patients with COVID-19-Related Acute Respiratory Failure. Balkan Med. J. 2023, 40, 111–116. [Google Scholar] [CrossRef]
- Klein, S.L.; Flanagan, K.L. Sex differences in immune responses. Nat. Rev. Immunol. 2016, 16, 626–638. [Google Scholar] [CrossRef]
- Scully, E.P.; Haverfield, J.; Ursin, R.L.; Tannenbaum, C.; Klein, S.L. Considering how biological sex impacts immune responses and COVID-19 outcomes. Nat. Rev. Immunol. 2020, 20, 442–447. [Google Scholar] [CrossRef]
- Haitao, T.; Vermunt, J.V.; Abeykoon, J.; Ghamrawi, R.; Gunaratne, M.; Jayachandran, M.; Narang, K.; Parashuram, S.; Suvakov, S.; Garovic, V.D. COVID-19 and Sex Differences: Mechanisms and Biomarkers. Mayo Clin. Proc. 2020, 95, 2189–2203. [Google Scholar] [CrossRef]
- Takahashi, T.; Ellingson, M.K.; Wong, P.; Israelow, B.; Lucas, C.; Klein, J.; Silva, J.; Mao, T.; Oh, J.E.; Tokuyama, M.; et al. Sex differences in immune responses that underlie COVID-19 disease outcomes. Nature. 2020, 588, 315–320. [Google Scholar] [CrossRef]
- Tejpal, A.; Gianos, E.; Cerise, J.; Hirsch, J.S.; Rosen, S.; Kohn, N.; Lesser, M.; Weinberg, C.; Majure, D.; Satapathy, S.K.; et al. Sex-Based Differences in COVID-19 Outcomes. J. Womens Health 2021, 30, 492–501. [Google Scholar] [CrossRef]
- Brady, E.; Nielsen, M.W.; Andersen, J.P.; Oertelt-Prigione, S. Lack of consideration of sex and gender in COVID-19 clinical studies. Nat. Commun. 2021, 12, 4015. [Google Scholar] [CrossRef]
- Bucciarelli, V.; Nasi, M.; Bianco, F.; Seferovic, J.; Ivkovic, V.; Gallina, S.; Mattioli, A.V. Depression pandemic and cardiovascular risk in the COVID-19 era and long COVID syndrome: Gender makes a difference. Trends Cardiovasc. Med. 2022, 32, 12–17. [Google Scholar] [CrossRef]
- Bai, F.; Tomasoni, D.; Falcinella, C.; Barbanotti, D.; Castoldi, R.; Mulè, G.; Augello, M.; Mondatore, D.; Allegrini, M.; Cona, A.; et al. Female gender is associated with long COVID syndrome: A prospective cohort study. Clin. Microbiol. Infect. 2022, 28, e9–e611. [Google Scholar] [CrossRef]
- Ejaz, H.; Alsrhani, A.; Zafar, A.; Javed, H.; Junaid, K.; Abdalla, A.E.; Abosalif, K.O.; Ahmed, Z.; Younas, S. COVID-19 and comorbidities: Deleterious impact on infected patients. J. Infect. Public Health 2020, 13, 1833–1839. [Google Scholar] [CrossRef]
- Jafarzadeh, A.; Nemati, M.; Jafarzadeh, S.; Nozari, P.; Mortazavi, S.M.J. Thyroid dysfunction following vaccination with COVID-19 vaccines: A basic review of the preliminary evidence. J. Endocrinol. Investig. 2022, 45, 1835–1863. [Google Scholar] [CrossRef]
- De Sanctis, J.B.; Balda Noria, G.; García, A.H. Exploring How Adipose Tissue, Obesity, and Gender Influence the Immune Response to Vaccines: A Comprehensive Narrative Review. Int. J. Mol. Sci. 2025, 26, 862. [Google Scholar] [CrossRef]
- Zhao, X.; Jiang, L.; Sun, W.; Tang, S.; Kang, X.; Gao, Q.; Li, Z.; An, X.; Lian, F. Understanding the interplay between COVID-19 and diabetes: Insights for the post-pandemic era. Front. Endocrinol. 2025, 16, 1599969. [Google Scholar] [CrossRef]
- Lazar, M.; Barbu, E.C.; Chitu, C.E.; Anghel, A.-M.; Niculae, C.-M.; Manea, E.-D.; Damalan, A.-C.; Bel, A.-A.; Patrascu, R.-E.; Hristea, A.; et al. Mortality Predictors in Severe SARS-CoV-2 Infection. Medicina 2022, 58, 945. [Google Scholar] [CrossRef]
- Regitz-Zagrosek, V.; Lehmkuhl, E.; Mahmoodzadeh, S. Gender aspects of the role of the metabolic syndrome as a risk factor for cardiovascular disease. Gend. Med. 2007, 4 (Suppl. B), S162–S177. [Google Scholar] [CrossRef]
- Masana, L.; Correig, E.; Ibarretxe, D.; Anoro, E.; Arroyo, J.A.; Jericó, C.; Guerrero, C.; Miret, M.; Näf, S.; Pardo, A.; et al. Low HDL and high triglycerides predict COVID-19 severity. Sci. Rep. 2021, 11, 7217. [Google Scholar] [CrossRef]
- Kaddoura, R.; Ibrahim, M.I.M.; Al-Amri, M.; Nair, A.P.; Alharafsheh, A.; Alyafei, S.A.; Albakri, M. COVID-19-associated hypertriglyceridemia and impact of treatment. Front. Med. 2024, 11, 1326156. [Google Scholar] [CrossRef]
- Samuel, V.T.; Shulman, G.I. The pathogenesis of insulin resistance: Integrating signaling pathways and substrate flux. J. Clin. Investig. 2016, 126, 12–22. [Google Scholar] [CrossRef]
- Maecker, H.T.; McCoy, J.P.; Nussenblatt, R. Standardizing immunophenotyping for the Human Immunology Project. Nat. Rev. Immunol. 2012, 12, 191–200. [Google Scholar] [CrossRef]
- Duntas, L.H.; Brenta, G. The effect of thyroid disorders on lipid levels and metabolism. Med. Clin. North. Am. 2012, 96, 269–281. [Google Scholar] [CrossRef]
- Verhave, J.C.; Gansevoort, R.T.; Hillege, H.L.; Bakker, S.J.; De Zeeuw, D.; de Jong, P.E. An elevated urinary albumin excretion predicts de novo development of renal function impairment in the general population. Kidney Int. Suppl. 2004, 66, S18–S21. [Google Scholar] [CrossRef]
- Glassock, R.J.; Rule, A.D. Aging and the Kidneys: Anatomy, Physiology and Consequences for Defining Chronic Kidney Disease. Nephron 2016, 134, 25–29. [Google Scholar] [CrossRef]
- Mukhopadhyay, A.; Talmor, N.; Xia, Y.; Berger, J.S.; Iturrate, E.; Adhikari, S.; Pulgarin, C.; Quinones-Camacho, A.; Yuriditsky, E.; Horowitz, J.; et al. Sex differences in the prognostic value of troponin and D-dimer in COVID-19 illness. Heart Lung. 2023, 58, 1–5. [Google Scholar] [CrossRef]
- Vassallo, A.; Shajahan, S.; Harris, K.; Hallam, L.; Hockham, C.; Womersley, K.; Woodward, M.; Sheel, M. Sex and Gender in COVID-19 Vaccine Research: Substantial Evidence Gaps Remain. Front. Glob. Womens Health. 2021, 2, 761511. [Google Scholar] [CrossRef]
- Xie, Y.; Xu, E.; Bowe, B.; Al-Aly, Z. Long-term cardiovascular outcomes of COVID-19. Nat. Med. 2022, 28, 583–590. [Google Scholar] [CrossRef]
- Green, M.S.; Peer, V.; Magid, A.; Hagani, N.; Anis, E.; Nitzan, D. Gender Differences in Adverse Events Following the Pfizer-BioNTech COVID-19 Vaccine. Vaccines 2022, 10, 233. [Google Scholar] [CrossRef]
- Kounis, N.G.; Gogos, C.; de Gregorio, C.; Hung, M.-Y.; Kounis, S.N.; Tsounis, E.P.; Assimakopoulos, S.F.; Pourmasumi, S.; Mplani, V.; Servos, G.; et al. “When,” “Where,” and “How” of SARS-CoV-2 Infection Affects the Human Cardiovascular System: A Narrative Review. Balkan Med. J. 2024, 41, 7–22. [Google Scholar] [CrossRef]

| Total Population (n = 426) | Female (n = 199) | Male (n = 227) | p Value | |||||
|---|---|---|---|---|---|---|---|---|
| Age | Min–Max (Median) | 19–80 (56) | 19–77 (57) | 19–80 (55) 55.36 ± 0.69 | 0.7166 | |||
| Mean ± SEM | 55.18 ± 0.51 | 54.98 ± 0.76 | ||||||
| BMI | Min–Max (Median) | 18.37–51.02 (29.81) | 18.37–51.02 (31.25) | 19.92–39.79 (28.69) 28.88 ± 0.37 | <0.0001 *** | |||
| Mean ± SEM | 30.09 ± 0.34 | 31.49 ± 0.57 | ||||||
| Smoking | Current/n (%) | 25 (5.87) | 8 (4.02) | 17 (7.49) | <0.0001 *** | |||
| Former/n (%) | 46 (10.80) | 8 (4.02) | 38 (16.77) | |||||
| Never/n (%) | 355 (83.33) | 183 (91.96) | 172 (75.74) | |||||
| Vaccine doses | Vaccine type # | n | Doses | n | Doses | n | Doses | |
| 2 times | Inactivated | 149 | 51 | 82 | 33 | 67 | 18 | |
| mRNA | 247 | 131 | 116 | |||||
| 3 times | Inactivated | 167 | 132 | 74 | 64 | 93 | 68 | |
| mRNA | 369 | 158 | 211 | |||||
| 4 times | Inactivated | 84 | 167 | 36 | 91 | 48 | 76 | |
| mRNA | 169 | 53 | 116 | |||||
| 5 times | Inactivated | 21 | 70 | 6 | 21 | 15 | 49 | |
| mRNA | 35 | 9 | 26 | |||||
| 6 times | Inactivated | 5 | 13 | 1 | 2 | 4 | 11 | |
| mRNA | 17 | 4 | 13 | |||||
| Total Population | Female | Male | |||
|---|---|---|---|---|---|
| Min–Max (Median) | Min–Max (Median) | Min–Max (Median) | p Value | ||
| Mean ± SEM | Mean ± SEM | Mean ± SEM | |||
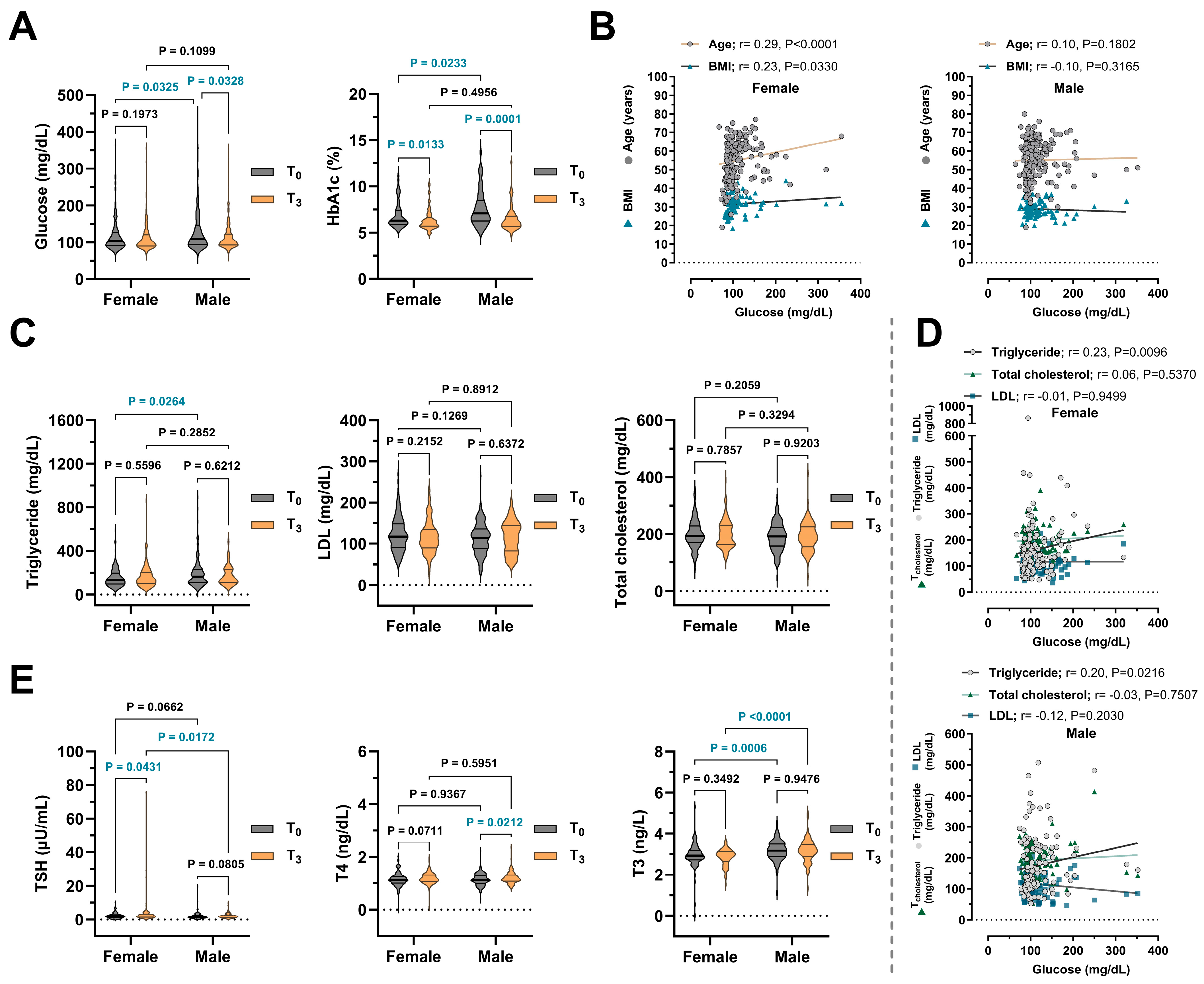
| Glucose (mg/dL) | T0 | 66–444 (105.00) | 66–363 (103.50) | 67–444 (109.50) | 0.0325 * |
| 126.00 ± 2.66 | 119.80 ± 3.40 | 131.30 ± 3.96 | |||
| T1 | 70–504 (99.00) | 72–357 (100.00) | 70–504 (99.00) | 0.4604 | |
| 120.20 ± 2.67 | 119.00 ± 3.81 | 121.40 ± 3.76 | |||
| T2 | 66–540 (100.00) | 66–369 (100.00) | 73–540 (102.00) | 0.2118 | |
| 117.80 ± 2.35 | 114.70 ± 2.94 | 120.70 ± 3.59 | |||
| T3 | 64–355 (101.00) | 67–355 (99.00) | 64–352 (103.00) | 0.1099 | |
| 113.60 ± 2.04 | 111.9 ± 2.94 | 115.20 ± 2.83 | |||
| p Value | 0.0171 * | 0.5191 | 0.0289 * | ||
| HbA1c (%) | T0 | 5.10–13.70 (6.60) | 5.40–10.20 (6.30) | 5.10–13.70 (7.10) | 0.0233 * |
| 7.26 ± 0.17 | 6.89 ± 0.20 | 7.63 ± 0.27 | |||
| T1 | 5.00–12.10 (6.20) | 5.20–12.10 (6.20) | 5.00–11.70 (6.20) | 0.9252 | |
| 6.74 ± 0.11 | 6.71 ± 0.16 | 6.77 ± 0.15 | |||
| T2 | 4.80–13.70 (6.10) | 4.80–10.30 (6.10) | 4.90–13.70 (6.20) | 0.1650 | |
| 6.58 ± 0.12 | 6.41 ± 0.13 | 6.77 ± 0.19 | |||
| T3 | 4.70–12.60 (6.10) | 4.90–10.30 (6.00) | 4.70–12.60 (6.10) | 0.4956 | |
| 6.43 ± 0.10 | 6.35 ± 0.13 | 6.51 ± 0.16 | |||
| p Value | <0.0001 *** | 0.0381 * | 0.0014 ** | ||
| Total Population | Female | Male | |||
|---|---|---|---|---|---|
| Min–Max (Median) | Min–Max (Median) | Min–Max (Median) | p Value | ||
| Mean ± SEM | Mean ± SEM | Mean ± SEM | |||
| LDL (mg/dL) | T0 | 31–271 (1171.00) | 51–271 (117.00) | 31–265 (114.00) | 0.1269 |
| 118.30 ± 2.57 | 123.70 ± 3.89 | 113.30 ± 3.36 | |||
| T1 | 34–269 (117.00) | 34–264 (117.00) | 39–269 (116.00) | 0.9093 | |
| 118.60 ± 2.24 | 118.80 ± 3.31 | 118.40 ± 3.06 | |||
| T2 | 28–275 (119.00) | 28–275 (118.50) | 32–268 (119.50) | 0.8960 | |
| 118.70 ± 2.24 | 119.50 ± 3.38 | 118.00 ± 2.99 | |||
| T3 | 25–234 (115.00) | 25–234 (110.00) | 46–203 (118.00) | 0.8912 | |
| 115.80 ± 2.51 | 116.40 ± 3.57 | 115.10 ± 3.54 | |||
| p Value | 0.8264 | 0.6261 | 0.7223 | ||
| Triglyceride (mg/dL) | T0 | 41–892 (143.00) | 41–588 (135.00) | 41–892 (150.00) | 0.0264 * |
| 174.60 ± 6.83 | 156.20 ± 7.66 | 191.30 ± 10.86 | |||
| T1 | 34–1263 (142.00) | 34–525 (140.00) | 42–1263 (152.00) | 0.4293 | |
| 170.10 ± 6.28 | 160.70 ± 7.05 | 178.30 ± 10.00 | |||
| T2 | 33–602 (138.00) | 33–602 (129.00) | 34–585 (143.00) | 0.4579 | |
| 162.00 ± 5.20 | 159.00 ± 7.68 | 164.40 ± 7.09 | |||
| T3 | 48–862 (142.00) | 48–862 (141.00) | 52–507 (141.00) | 0.2852 | |
| 169.80 ± 6.26 | 165.60 ± 9.46 | 174.00 ± 8.22 | |||
| p Value | 0.5726 | 0.9133 | 0.3010 | ||
| Total cholesterol (mg/dL) | T0 | 47–357 (193.00) | 76–357 (193.00) | 47–328 (191.50) | 0.7179 |
| 194.60 ± 3.13 | 200.10 ± 4.70 | 189.90 ± 4.16 | |||
| T1 | 69–377 (195.50) | 69–377 (197.50) | 90–321 (192.00) | 0.1149 | |
| 196.20 ± 2.52 | 199.30 ± 3.56 | 193.50 ± 3.53 | |||
| T2 | 53–331 (192.00) | 103–314 (195.50) | 53–331 (191.00) | 0.8209 | |
| 192.70 ± 2.39 | 195.50 ± 3.39 | 190.50 ± 3.35 | |||
| T3 | 52–413 (193.00) | 112–390 (194.50) | 52–413 (191.00) | 0.9736 | |
| 195.30 ± 3.10 | 199.40 ± 4.36 | 191.30 ± 4.39 | |||
| p Value | 0.6734 | 0.8518 | 0.6984 | ||
| TSH (μU/mL) | T0 | 0.06–19.90 (1.67) | 0.06–10.04 (1.85) | 0.08–19.90 (1.51) | 0.0662 |
| 2.11 ± 0.12 | 2.18 ± 0.13 | 2.05 ± 0.18 | |||
| T1 | 0.02–55.10 (1.79) | 0.02–55.10 (1.91) | 0.04–12.30 (1.72) | 0.1448 | |
| 2.25 ± 0.16 | 2.46 ± 0.31 | 2.07 ± 0.12 | |||
| T2 | 0.05–35.80 (1.91) | 0.05–27.40 (2.04) | 0.18–35.80 (1.61) | 0.1006 | |
| 2.38 ± 0.15 | 2.48 ± 0.20 | 2.29 ± 0.22 | |||
| T3 | 0.01–75.40 (1.89) | 0.10–75.40 (2.12) | 0.25–10.20 (1.77) | 0.0172 * | |
| 2.69 ± 0.30 | 3.26 ± 0.58 | 2.14 ± 0.13 | |||
| p Value | 0.0575 | 0.1849 | 0.3372 | ||
| T3 (ng/L) | T0 | 0.02–5.30 (3.04) | 0.02–5.30 (2.93) | 1.11–4.52 (3.18) | 0.0006 *** |
| 3.07 ± 0.04 | 2.95 ± 0.06 | 3.18 ± 0.05 | |||
| T1 | 1.43–5.44 (3.08) | 1.43–4.19 (2.98) | 1.87–5.44 (3.12) | 0.0061 ** | |
| 3.06 ± 0.03 | 2.97 ± 0.05 | 3.15 ± 0.04 | |||
| T2 | 1.89–4.45 (3.10) | 1.94–4.45 (3.01) | 1.89–4.25 (3.23) | <0.0001 *** | |
| 3.10 ± 0.03 | 2.98 ± 0.04 | 3.20 ± 0.04 | |||
| T3 | 1.34–4.99 (3.05) | 1.53–3.64 (2.94) | 1.34–4.99 (3.18) | <0.0001 *** | |
| 3.02 ± 0.04 | 2.85 ± 0.05 | 3.17 ± 0.06 | |||
| p Value | 0.5073 | 0.4780 | 0.6687 | ||
| T4 (ng/dL) | T0 | 0.01–2.22 (1.13) | 0.01–2.01 (1.13) | 0.40–2.22 (1.13) | 0.9367 |
| 1.15 ± 0.02 | 1.15 ± 0.02 | 1.15 ± 0.02 | |||
| T1 | 0.37–2.15 (1.17) | 0.37–1.98 (1.16) | 0.64–2.15 (1.17) | 0.8525 | |
| 1.19 ± 0.01 | 1.20 ± 0.02 | 1.18 ± 0.01 | |||
| T2 | 0.07–2.06 (1.17) | 0.07–1.84 (1.18) | 0.68–2.06 (1.16) | 0.3274 | |
| 1.19 ± 0.01 | 1.21 ± 0.02 | 1.18 ± 0.01 | |||
| T3 | 0.09–2.36 (1.17) | 0.09–1.96 (1.16) | 0.82–2.36 (1.18) | 0.5951 | |
| 1.20 ± 0.02 | 1.19 ± 0.02 | 1.21 ± 0.02 | |||
| p Value | 0.0104 * | 0.0715 | 0.1365 | ||
| Total Population | Female | Male | |||
|---|---|---|---|---|---|
| Min–Max (Median) | Min–Max (Median) | Min–Max (Median) | p Value | ||
| Mean ± SEM | Mean ± SEM | Mean ± SEM | |||
| Urea (mg/dL) | T0 | 11.80–141.20 (30.25) | 11.80–141.20 (27.60) | 13.90–132.00 (32.40) | 0.0002 *** |
| 33.75 ± 0.78 | 32.23 ± 1.21 | 35.09 ± 1.00 | |||
| T1 | 11.70–114.50 (29.80) | 11.70–114.50 (27.90) | 15.10–103.80 (31.80) | <0.0001 *** | |
| 32.66 ± 0.64 | 31.25 ± 1.05 | 33.93 ± 0.77 | |||
| T2 | 11.70–163.60 (30.20) | 11.70–163.60 (28.05) | 17.30–136.40 (31.80) | <0.0001 *** | |
| 33.55 ± 0.82 | 32.61 ± 1.38 | 34.38 ± 0.94 | |||
| T3 | 12.00–176.00 (30.60) | 12.00–124.60 (28.60) | 18.10–176.00 (32.50) | 0.0007 *** | |
| 33.72 ± 0.84 | 31.80 ± 1.14 | 35.42 ± 1.21 | |||
| p Value | 0.8980 | 0.9214 | 0.9814 | ||
| Creatinine (mg/dL) | T0 | 0.34–11.02 (0.76) | 0.34–11.02 (0.65) | 0.42–4.64 (0.87) | <0.0001 *** |
| 0.87 ± 0.03 | 0.81 ± 0.07 | 0.92 ± 0.03 | |||
| T1 | 0.31–10.17 (0.79) | 0.31–10.17 (0.68) | 0.33–3.47 (0.89) | <0.0001 *** | |
| 0.89 ± 0.03 | 0.83 ± 0.06 | 0.95 ± 0.02 | |||
| T2 | 0.33–6.87 (0.80) | 0.33–6.87 (0.68) | 0.52–4.55 (0.90) | <0.0001 *** | |
| 0.90 ± 0.03 | 0.82 ± 0.05 | 0.98 ± 0.03 | |||
| T3 | 0.37–7.79 (0.83) | 0.37–7.79 (0.69) | 0.46–4.13 (0.91) | <0.0001 *** | |
| 0.94 ± 0.03 | 0.84 ± 0.06 | 1.02 ± 0.03 | |||
| p Value | 0.0017 ** | 0.0349 * | 0.0031 ** | ||
| D-dimer (μg/mL) | T1 | 0.01–15.28 (0.29) | 0.01–6.72 (0.31) | 0.01–15.28 (0.27) | 0.4619 |
| 0.61 ± 0.06 | 0.55 ± 0.07 | 0.66 ± 0.10 | |||
| T2 | 0.01–10.10 (0.30) | 0.01–2.20 (0.31) | 0.01–10.10 (0.31) | 0.4281 | |
| 0.45 ± 0.04 | 0.41 ± 0.03 | 0.49 ± 0.07 | |||
| T3 | 0.01–12.50 (0.30) | 0.01–12.50 (0.36) | 0.01–1.71 (0.32) | 0.0239 * | |
| 0.45 ± 0.05 | 0.48 ± 0.10 | 0.35 ± 0.02 | |||
| p Value | 0.9050 | 0.5902 | 0.8610 | ||
| Fibrinogen (mg/dL) | T1 | 94–935 (424.50) | 163–751 (450.00) | 94–935 (406.00) | 0.0114 * |
| 447.20 ± 6.65 | 457.20 ± 7.97 | 438.30 ± 10.34 | |||
| T2 | 168–736 (397.00) | 168–666 (406.00) | 203–736 (382.50) | 0.0008 ** | |
| 407.00 ± 5.12 | 422.20 ± 6.82 | 393.80 ± 7.40 | |||
| T3 | 189–685 (383.00) | 219–666 (398.00) | 189–685 (378.50) | 0.0201 * | |
| 394.10 ± 5.46 | 407.40 ± 7.82 | 382.90 ± 7.51 | |||
| p Value | <0.0001 *** | <0.0001 *** | 0.0010 ** | ||
| Troponin (pg/mL) | T1 | 0–1008.00 (4.30) | 0–318.10 (3.50) | 0–1008.00 (4.84) | 0.0226 * |
| 12.17 ± 4.23 | 8.44 ± 2.88 | 15.23 ± 7.33 | |||
| T2 | 0–261.00 (4.86) | 1.40–75.67 (4.13) | 0–261.00 (5.87) | 0.0517 | |
| 11.38 ± 2.14 | 7.04 ± 1.17 | 14.85 ± 3.70 | |||
| T3 | 1.00–175.30 (6.81) | 1.00–89.82 (5.77) | 1.40–175.30 (7.32) | 0.1756 | |
| 10.84 ± 1.73 | 9.20 ± 1.70 | 12.30 ± 2.89 | |||
| p Value | <0.0001 *** | 0.0002 *** | 0.0033 ** | ||
| Yes/n (%) | Yes/n (%) | Yes/n (%) | p Value | ||
| No/n (%) | No/n (%) | No/n (%) | |||
| Hyper-tension | T0 | 170 (39.91) | 89 (44.72) | 81 (35.68) | 0.0603 |
| 256 (60.09) | 110 (55.28) | 146 (64.32) | |||
| T3 | 179 (42.02) | 93 (46.73) | 86 (37.89) | 0.0766 | |
| 247 (57.98) | 106 (53.27) | 141 (62.11) | |||
| p Value | 0.5773 | 0.3289 | 0.6971 | ||
| Total Population | Female | Male | |||
|---|---|---|---|---|---|
| Min–Max (Median) | Min–Max (Median) | Min–Max (Median) | p Value | ||
| Mean ± SEM | Mean ± SEM | Mean ± SEM | |||
| CRP (mg/dL) | T1 | 0.02–309.80 (4.34) | 0.02–151.40 (5.53) | 0.27–309.80 (3.52) | 0.0702 |
| 13.51 ± 1.35 | 11.21 ± 1.31 | 15.53 ± 2.26 | |||
| T2 | 0.27–276.70 (3.37) | 0.27–117.90 (4.43) | 0.28–276.70 (2.77) | 0.0159 * | |
| 8.42 ± 1.47 | 7.68 ± 0.96 | 9.07 ± 2.02 | |||
| T3 | 0.21–210.60 (3.14) | 0.21–146.60 (3.98) | 0.23–210.60 (2.47) | 0.0003 *** | |
| 7.64 ± 0.88 | 7.32 ± 0.95 | 7.92 ± 1.42 | |||
| p Value | 0.0003 *** | 0.0435 * | 0.0036 * | ||
| WBC Count (×109/L) | T1 | 0.88–24.10 (6.87) | 1.76–20.13 (6.54) | 0.88–24.10 (7.20) | 0.0007 *** |
| 7.26 ± 0.12 | 6.82 ± 0.14 | 7.65 ± 0.18 | |||
| T2 | 1.10–24.42 (6.87) | 3.34–24.42 (6.72) | 1.10–16.48 (7.16) | 0.0025 ** | |
| 7.28 ± 0.11 | 7.04 ± 0.17 | 7.50 ± 0.14 | |||
| T3 | 2.69–20.36 (7.02) | 2.69–20.36 (6.98) | 3.41–16.80 (7.09) | 0.3913 | |
| 7.41 ± 0.11 | 7.31 ± 0.17 | 7.49 ± 0.14 | |||
| p Value | 0.4225 | 0.0931 | 0.9035 | ||
| NEU Count (×103/µL) | T1 | 5.87–93.40 (60.05) | 5.87–89.70 (60.20) | 36.30–93.40 (59.90) | 0.9535 |
| 60.42 ± 0.49 | 59.95 ± 0.72 | 60.84 ± 0.67 | |||
| T2 | 5.49–91.20 (59.25) | 25.30–91.20 (58.30) | 5.49–85.10 (60.10) | 0.1503 | |
| 59.33 ± 0.46 | 58.81 ± 0.64 | 59.78 ± 0.66 | |||
| T3 | 19.90–90.40 (58.80) | 19.90–90.40 (59.10) | 40.10–84.00 (58.15) | 0.5921 | |
| 59.25 ± 0.44 | 59.19 ± 0.67 | 59.32 ± 0.59 | |||
| p Value | 0.1939 | 0.2267 | 0.2909 | ||
| LY Count (%) | T1 | 2.30–76.40 (29.70) | 8.20–76.40 (30.20) | 2.30–53.80 (29.40) | 0.3054 |
| 29.82 ± 0.43 | 30.56 ± 0.62 | 29.16 ± 0.60 | |||
| T2 | 6.20–72.50 (30.80) | 6.20–72.50 (32.07) | 10.70–59.40 (30.02) | 0.0064 ** | |
| 30.98 ± 0.41 | 32.07 ± 0.59 | 30.02 ± 0.56 | |||
| T3 | 7.40–78.00 (31.30) | 7.40–78.00 (31.30) | 7.90–51.50 (31.25) | 0.4812 | |
| 31.07 ± 0.42 | 31.72 ± 0.65 | 30.50 ± 0.55 | |||
| p Value | 0.0692 | 0.0972 | 0.2235 | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Published by MDPI on behalf of the Lithuanian University of Health Sciences. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gur, C.; Solak, S.K.; Gundogan, E.; Pektas, F.; Uzun, H. Sex-Specific Longitudinal Changes in Metabolic, Endocrine, Renal, Cardiovascular, and Inflammatory Biomarkers of Vaccinated COVID-19 Survivors: 30-Month Follow-Up Study. Medicina 2025, 61, 1510. https://doi.org/10.3390/medicina61091510
Gur C, Solak SK, Gundogan E, Pektas F, Uzun H. Sex-Specific Longitudinal Changes in Metabolic, Endocrine, Renal, Cardiovascular, and Inflammatory Biomarkers of Vaccinated COVID-19 Survivors: 30-Month Follow-Up Study. Medicina. 2025; 61(9):1510. https://doi.org/10.3390/medicina61091510
Chicago/Turabian StyleGur, Ceren, Sezen Kumas Solak, Erdal Gundogan, Fatih Pektas, and Hafize Uzun. 2025. "Sex-Specific Longitudinal Changes in Metabolic, Endocrine, Renal, Cardiovascular, and Inflammatory Biomarkers of Vaccinated COVID-19 Survivors: 30-Month Follow-Up Study" Medicina 61, no. 9: 1510. https://doi.org/10.3390/medicina61091510
APA StyleGur, C., Solak, S. K., Gundogan, E., Pektas, F., & Uzun, H. (2025). Sex-Specific Longitudinal Changes in Metabolic, Endocrine, Renal, Cardiovascular, and Inflammatory Biomarkers of Vaccinated COVID-19 Survivors: 30-Month Follow-Up Study. Medicina, 61(9), 1510. https://doi.org/10.3390/medicina61091510







