Effectiveness of Plasma-Rich Fibrin and De-Epithelialized Free Gingival Graft in the Treatment of Gingival Recessions
Abstract
1. Introduction
2. Materials and Methods
3. Results
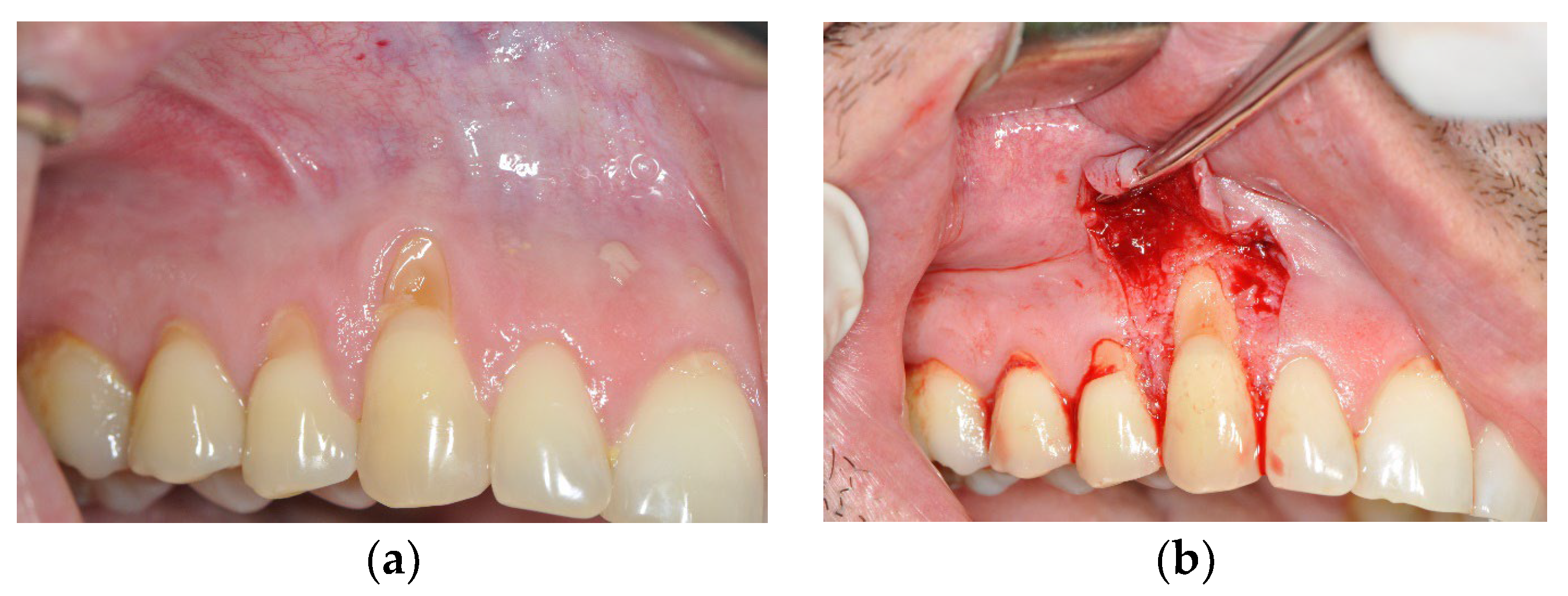
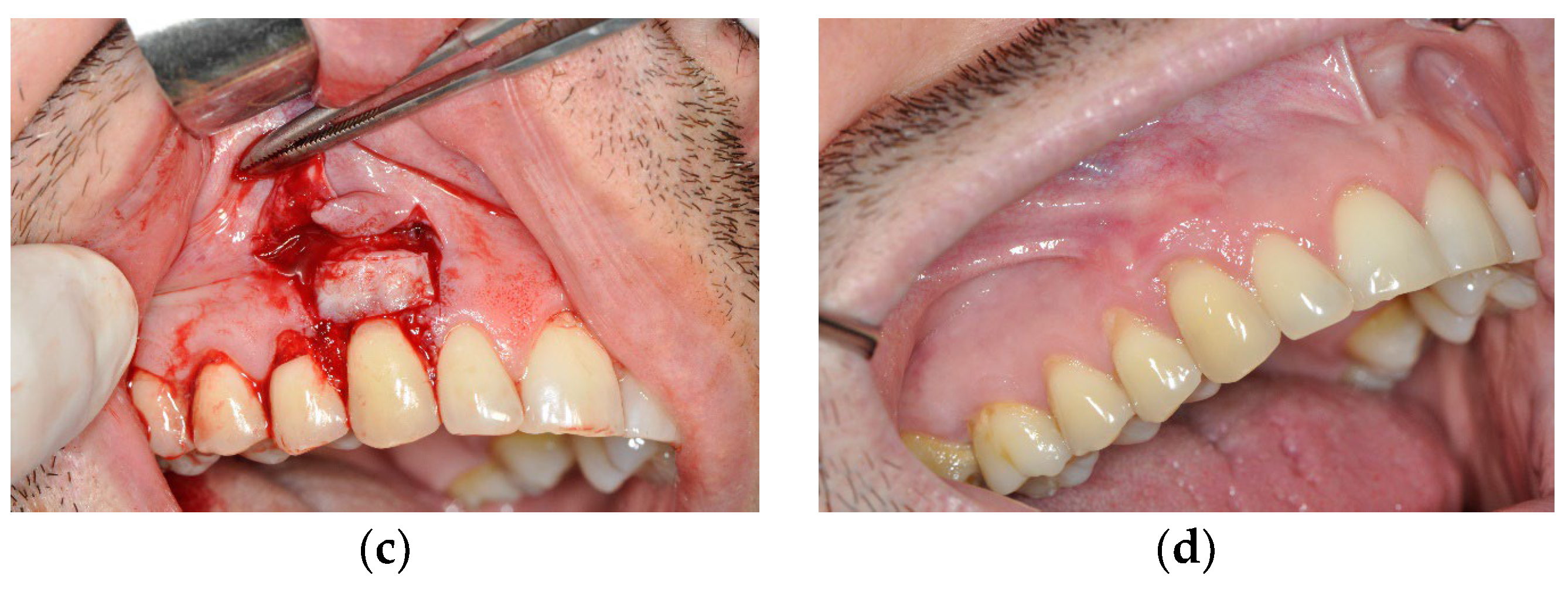
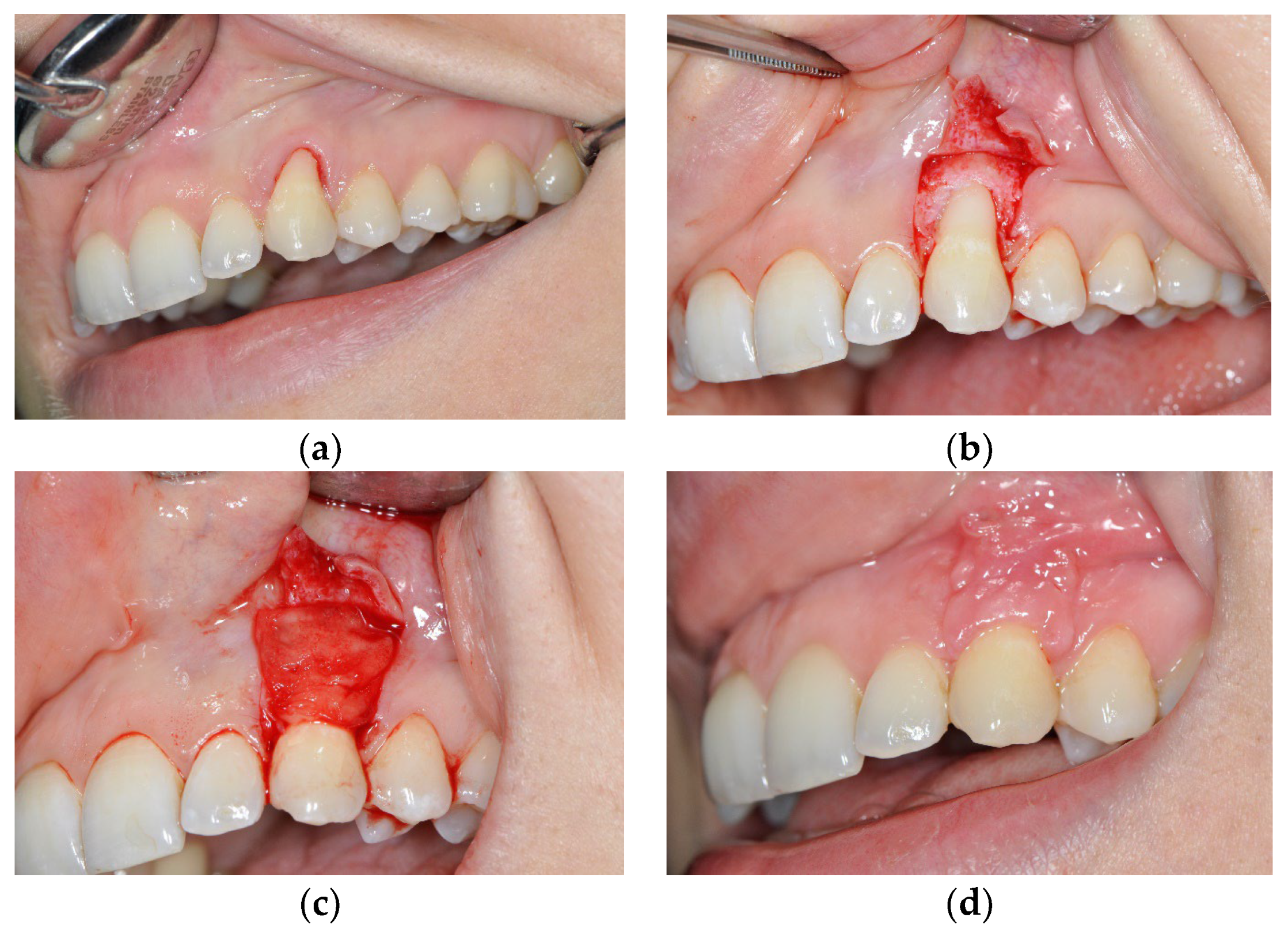
3.1. The Results of Implementing the PRF Technique
3.2. The Results Obtained by Implementing the Surgical Method
3.3. Comparative Analysis of Obtained Data
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zucchelli, G.; Mele, M.; Stefanini, M.; Mazzotti, C.; Mounssif, I.; Marzadori, M.; Montebugnoli, L. Predetermination of root coverage. J. Periodontol. 2010, 81, 1019–1026. [Google Scholar] [CrossRef] [PubMed]
- Jati, A.S.; Furquim, L.Z.; Consolaro, A. Gingival recession: Its causes and types, and the importance of orthodontic treatment. Dental Press J. Orthod. 2016, 21, 18–29. [Google Scholar] [CrossRef]
- Cairo, F.; Nieri, M.; Cattabriga, M.; Cortellini, P.; De Paoli, S.; De Sanctis, M.; Fonzar, A.; Francetti, L.; Merli, M.; Rasperini, G.; et al. Root Coverage Esthetic Score After Treatment of Gingival Recession: An Interrater Agreement Multicenter Study. J. Periodontol. 2010, 81, 1752–1758. [Google Scholar] [CrossRef] [PubMed]
- Miller, P.D., Jr. A classification of marginal tissue recession. Int. J. Periodontics Restor. Dent. 1985, 5, 8–13. [Google Scholar]
- Barootchi, S.; Mancini, L.; Heck, T.; Zucchelli, G.; Stefanini, M.; Kazarian, E.; Tavelli, L. Reliability assessment of the classification for facial peri-implant soft tissue dehiscence/deficiencies (PSTDs): A multi-center inter-rater agreement study of different skill-level practitioners. J. Periodontol. 2022, 93, 1173–1182. [Google Scholar] [CrossRef] [PubMed]
- Skierska, I.; Wyrębek, B.; Górski, B. Clinical and Aesthetic Outcomes of Multiple Gingival Recessions Coverage with Modified Coronally Advanced Tunnel and Subepithelial Connective Tissue Graft in Maxilla and Mandible: A 2-Year Retrospective Study. Int. J. Environ. Res. Public Health 2022, 19, 11024. [Google Scholar] [CrossRef]
- Cairo, F.; Nieri, M.; Cincinelli, S.; Mervelt, J.; Pagliaro, U. The interproximal clinical attachment level to classify gingival recessions and predict root coverage outcomes: An explorative and reliability study. J. Clin. Periodontol. 2011, 38, 661–666. [Google Scholar] [CrossRef] [PubMed]
- Aslan, S.; Buduneli, N.; Tavelli, L.; Rasperini, G.; Cortellini, P. Introduction of a Novel Anatomic Recession Ratio in the Treatment of Gingival Recession: A Proof-of-Principle Study. Int. J. Periodontics Restor. Dent. 2022, 42, e103–e112. [Google Scholar] [CrossRef]
- Sculean, A.; Gruber, R.; Bosshardt, D.D. Soft tissue wound healing around teeth and dental implants. J. Clin. Periodontol. 2014, 41, S6–S22. [Google Scholar] [CrossRef]
- Rosenthal, P. Re-covering the exposed necks of teeth by autoplasty. Dent. Cosm. 1912, 54, 377–378. [Google Scholar]
- Chambrone, L.; Sukekava, F.; Araújo, M.G.; Pustiglioni, F.E.; Chambrone, L.A.; Lima, L.A. Root-Coverage Procedures for the Treatment of Localized Recession-Type Defects: A Cochrane Systematic Review. J. Periodontol. 2010, 81, 452–478. [Google Scholar] [CrossRef] [PubMed]
- Zucchelli, G.; Mazzotti, C.; Mounssif, I.; Mele, M.; Stefanini, M.; Montebugnoli, L. A novel surgical–prosthetic approach for soft tissue dehiscence coverage around single implant. Clin. Oral Implant. Res. 2013, 24, 957–962. [Google Scholar] [CrossRef] [PubMed]
- Zuhr, O.; Fickl, S.; Wachtel, H.; Bolz, W.; Hurzeler, M.B. Covering of gingival recessions with a modified microsurgical tunnel technique: Case report. Int. J. Periodontics Restor. Dent. 2007, 27, 457. [Google Scholar]
- Antezack, A.; Ohanessian, R.; Sadowski, C.; Faure-Brac, M.; Brincat, A.; Etchecopar-Etchart, D.; Monnet-Corti, V. Effectiveness of surgical root coverage on dentin hypersensitivity: A systematic review and meta-analysis. J. Clin. Periodontol. 2022, 49, 840–851. [Google Scholar] [CrossRef] [PubMed]
- Zucchelli, G.; Stefanini, M.; Ganz, S.; Mazzotti, C.; Mounssif, I.; Marzadori, M. Coronally Advanced Flap with Different Designs in the Treatment of Gingival Recession: A Comparative Controlled Randomized Clinical Trial. Int. J. Periodontics Restor. Dent. 2016, 36, 319–327. [Google Scholar] [CrossRef]
- Stefanini, M.; Mounssif, I.; Barootchi, S.; Tavelli, L.; Wang, H.L.; Zucchelli, G. An exploratory clinical study evaluating safety and performance of a volume-stable collagen matrix with coronally advanced flap for single gingival recession treatment. Clin. Oral Investig. 2020, 24, 3181–3191. [Google Scholar] [CrossRef]
- Zuhr, O.; Rebele, S.F.; Cheung, S.L.; Hürzeler, M.B.; Research Group on Oral Soft Tissue Biology and Wound Healing. Surgery without papilla incision: Tunneling flap procedures in plastic periodontal and implant surgery. Periodontol. 2000 2018, 77, 123–149. [Google Scholar] [CrossRef]
- Zucchelli, G.; Marzadori, M.; Mounssif, I.; Mazzotti, C.; Stefanini, M. Coronally advanced flap + connective tissue graft techniques for the treatment of deep gingival recession in the lower incisors. A controlled randomized clinical trial. J. Clin. Periodontol. 2014, 41, 806–813. [Google Scholar] [CrossRef]
- Ghanaati, S.; Herrera-Vizcaino, C.; Al-Maawi, S.; Lorenz, J.; Miron, R.J.; Nelson, K.; Schwarz, F.; Choukroun, J.; Sader, R. Fifteen Years of Platelet Rich Fibrin in Dentistry and Oromaxillofacial Surgery: How High is the Level of Scientific Evidence? J. Oral Implantol. 2018, 44, 471–492. [Google Scholar] [CrossRef]
- Dohan, D.M.; Choukroun, J.; Diss, A.; Dohan, S.L.; Dohan, A.J.; Mouhyi, J.; Gogly, B. Platelet-rich fibrin (PRF): A second-generation platelet concentrate. Part I: Technological concepts and evolution. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontol. 2006, 101, e37–e44. [Google Scholar] [CrossRef]
- Zuhr, O.; Bäumer, D.; Hürzeler, M. The addition of soft tissue replacement grafts in plastic periodontal and implant surgery: Critical elements in design and execution. J. Clin. Periodontol. 2014, 41, S123–S142. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, G.V.O.; Santos, N.B.M.; Siqueira, R.A.C.; Wang, H.L.; Blanco-Carrion, J.; Fernandes, J.C.H. Autologous Platelet Concentrate of 2nd and 3rd generations efficacy in the surgical treatment of gingival recession: An overview of systematic reviews. J. Indian Soc. Periodontol. 2021, 25, 463–479. [Google Scholar] [CrossRef] [PubMed]
- Gluckman, H. Platelet Rich Fibrin as an Adjunct to Implant Dentistry. In Platelet Rich Fibrin in Regenerative Dentistry: Biological Background and Clinical Indications; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2017; Volume 44, pp. 145–157. [Google Scholar]
- Miron, R.J.; Zucchelli, G.; Pikos, M.A.; Salama, M.; Lee, S.; Guillemette, V.; Fujioka-Kobayashi, M.; Bishara, M.; Zhang, Y.; Wang, H.-L.; et al. Use of platelet-rich fibrin in regenerative dentistry: A systematic review. Clin. Oral Investig. 2017, 21, 1913–1927. [Google Scholar] [CrossRef] [PubMed]
- Pifl, M.; Bertl, K.; Hirtler, L.; Rendl, B.; Nürnberger, S.; Ulm, C. Histomorphometric analysis of the palatal soft tissue as donor region for retrieval of connective tissue grafts. In Proceedings of the Österreichische Zahnärzte Kongress, Rust, Austria, 25–27 September 2014. [Google Scholar]
- Hämmerle, C.H.; Giannobile, W.V.; Abrahamsson, I.; Blanco, J.; Bosshardt, D.; Dahlin, C.; Working Group 1 of the European Workshop on Periodontology. Biology of soft tissue wound healing and regeneration—Consensus report of Group 1 of the 10th European Workshop on Periodontology. J. Clin. Periodontol. 2014, 41, S1–S5. [Google Scholar] [CrossRef] [PubMed]
- Zucchelli, G.; Mounssif, I.; Mazzotti, C.; Montebugnoli, L.; Sangiorgi, M.; Mele, M.; Stefanini, M. Does the dimension of the graft influence patient morbidity and root coverage outcomes? A randomized controlled clinical trial. J. Clin. Periodontol. 2014, 41, 708–716. [Google Scholar] [CrossRef] [PubMed]
- Du Toit, J.; Siebold, A.; Dreyer, A.; Gluckman, H. Choukroun platelet-rich fibrin as an autogenous graft biomaterial in preimplant surgery: Results of a preliminary randomized, human histomorphometric, split-mouth study. Int. J. Periodontics Restor. Dent. 2016, 36 (Suppl. 36), s75–s86. [Google Scholar] [CrossRef] [PubMed]
- Jepsen, K.; Jepsen, S.; Zucchelli, G.; Stefanini, M.; De Sanctis, M.; Baldini, N.; Greven, B.; Heinz, J.; Wennström, B.; Cassel, F.; et al. Treatment of gingival recession defects with a coronally advanced flap and a xenogeneic collagen matrix: A multicenter randomized clinical trial. J. Clin. Periodontol. 2013, 40, 82–89. [Google Scholar] [CrossRef]
- Zucchelli, G.; Mazzotti, C.; Tirone, F.; Mele, M.; Bellone, P.; Mounssif, I. The connective tissue graft wall technique and enamel matrix derivative to improve root coverage and clinical attachment levels in Miller Class IV gingival recession. Int. J. Periodontics Restor. Dent. 2014, 34, 601–609. [Google Scholar] [CrossRef]
- Kahn, S.; Araújo, I.T.E.; Dias, A.T.; Souza, A.B.; Chambrone, L.; Fernandes, G.V.O. Histologic and histomorphometric analysis of connective tissue grafts harvested by the parallel incision method: A pilot randomized controlled trial comparing macro-and microsurgical approaches. Quintessence Int. 2021, 52, 772–778. [Google Scholar] [CrossRef]
- Maia, V.T.G.; Kahn, S.; de Souza, A.B.; Fernandes, G.V.O. Deepithelialized Connective Tissue Graft and the Remaining Epithelial Content After Harvesting by the Harris Technique: A Histological and Morphometrical Case Series. Clin. Adv. Periodontics 2021, 11, 150–154. [Google Scholar] [CrossRef]
- Marques, T.; Ramos, S.; dos Santos, N.B.M.; Borges, T.; Montero, J.; Correia, A.; Fernandes, G.V.D.O. A 3D Digital Analysis of the Hard Palate Wound Healing after Free Gingival Graft Harvest: A Pilot Study in the Short Term. Dent. J. 2022, 10, 109. [Google Scholar] [CrossRef] [PubMed]
- Stefanini, M.; Barootchi, S.; Tavelli, L.; Marzadori, M.; Mazzotti, C.; Mounssif, I.; Sangiorgi, M.; Sabri, H.; Wang, H.L.; Zucchelli, G. Difficulty score for the treatment of isolated gingival recessions with the coronally advanced flap: A preliminary reliability study. Clin. Oral Investig. 2023, 27, 559–569. [Google Scholar] [CrossRef] [PubMed]
- Azar, E.L.; Rojas, M.A.; Mandalunis, P.; Gualtieri, A.; Carranza, N. Histological evaluation of subepithelial connective tissue grafts harvested by two different techniques: Preliminary study in humans. Acta Odontol. Latinoam. 2019, 32, 10–16. [Google Scholar] [PubMed]
- Zucchelli, G.; Gori, G.; Mele, M.; Stefanini, M.; Mazzotti, C.; Marzadori, M.; Montebugnoli, L.; De Sanctis, M. Non-carious cervical lesions associated with gingival recessions: A decision-making process. J. Periodontol. 2011, 82, 1713–1724. [Google Scholar] [CrossRef]
- Fickl, S.; Fischer, K.R.; Jockel-Schneider, Y.; Stappert, C.F.; Schlagenhauf, U.; Kebschull, M. Early wound healing and patient morbidity after single-incision vs. trap-door graft harvesting from the palate—A clinical study. Clin. Oral Investig. 2014, 18, 2213–2219. [Google Scholar] [CrossRef]
- Kloukos, D.; Eliades, T.; Sculean, A.; Katsaros, C. Indication and timing of soft tissue augmentation at maxillary and mandibular incisors in orthodontic patients. A systematic review. Eur. J. Orthod. 2014, 36, 442–449. [Google Scholar] [CrossRef]
- Choukroun, J.; Diss, A.; Simonpieri, A.; Girard, M.O.; Schoeffler, C.; Dohan, S.L.; Dohan, A.; Mouhyi, A.; Dohan, D.M. Platelet-rich fibrin (PRF): A second-generation platelet concentrate. Part V: Histologic evaluations of PRF effects on bone allograft maturation in sinus lift. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontol. 2006, 101, 299–303. [Google Scholar] [CrossRef]
- Toit, J.D.; Gluckman, H.; Salama, M. Platelet-Rich Fibrin (PRF): A growth factor-rich biomaterial. part 1-the platelet concentrates milieu & review of the literature. Int. Dent. -Afr. Ed. 2015, 5, 62–70. [Google Scholar]
- Pagni, G.; Tavelli, L.; Rasperini, G. The Evolution of Surgical Techniques and Biomaterials for Periodontal Regeneration. Dent. Clin. 2022, 66, 75–85. [Google Scholar] [CrossRef]
- Butera, A.; Gallo, S.; Pascadopoli, M.; Maiorani, C.; Milone, A.; Alovisi, M.; Scribante, A. Paraprobiotics in Non-Surgical Periodontal Therapy: Clinical and Microbiological Aspects in a 6-Month Follow-Up Domiciliary Protocol for Oral Hygiene. Microorganisms 2022, 10, 337. [Google Scholar] [CrossRef]
| Statistics\Method Applied | PRF | DGG | ||||
|---|---|---|---|---|---|---|
| Value | CI 95% | Value | CI 95% | |||
| Average before the intervention, | 6.25 | 5.77 | 6.73 | 6.17 | 5.65 | 6.69 |
| Average after the intervention, | 2.68 | 2.43 | 2.93 | 0.37 | 0.09 | 0.65 |
| Mean difference, − | 3.57 | 3.03 | 4.11 | 5.80 | 5.21 | 6.39 |
| Standard error of the mean, SEM1 (before intervention) | 0.23 | 0.25 | ||||
| Standard error of the mean, SEM2 (after intervention) | 0.12 | 0.13 | ||||
| Sample size | 20 | 20 | ||||
| Variance before the intervention, s12 | 1.06 | 1.24 | ||||
| Standard deviation before the intervention, s1 | 1.03 | 1.11 | ||||
| Variance after the intervention, s22 | 0.29 | 0.36 | ||||
| Standard deviation after the intervention, s2 | 0.54 | 0.60 | ||||
| Pooled variance, s1,22(pooled) | 0.67 | 0.80 | ||||
| Difference within groups before and after intervention, t | 5.6 | 6.8 | ||||
| p value | <0.01 | <0.01 | ||||
| Statistics\Method Applied | PRF vs. DGG (Comparative Differences between Two Applied Methods) | |||||
| Value | CI 95% | |||||
| Mean difference (before intervention), (PRF) − (DGG) | 0.08 | −0.63 | 0.79 | |||
| Mean difference (after intervention), (PRF) − (DGG) | 2.31 | 1.99 | 2.63 | |||
| Pooled variance, s12PRF/DDG(pooled) | 1.15 | |||||
| Difference between groups before intervention, t | 0.2 | |||||
| p value | <0.05 | |||||
| Pooled variance, s22 (PRF/DDGpooled) | 0.32 | |||||
| Difference between groups after intervention PRF vs. DGG, t | 12.8 | |||||
| p value | >0.05 | |||||
| Statistics\Method Applied | PRF | DGG | ||||
|---|---|---|---|---|---|---|
| Value | CI 95% | Value | CI 95% | |||
| Average before the intervention, | 5.63 | 5.31 | 5.95 | 5.55 | 5.23 | 5.87 |
| Average after the intervention, | 1.95 | 1.76 | 2.14 | 0.43 | 0.24 | 0.62 |
| Mean difference, − | 3.68 | 3.31 | 4.05 | 5.12 | 4.75 | 5.49 |
| Standard error of the mean, SEM1 (before intervention) | 0.15 | 0.15 | ||||
| Standard error of the mean, SEM2 (after intervention) | 0.09 | 0.09 | ||||
| Sample size | 20 | 20 | ||||
| Variance before the intervention, s12 | 0.47 | 0.47 | ||||
| Standard deviation before the intervention, s1 | 0.69 | 0.69 | ||||
| Variance after the intervention, s22 | 0.17 | 0.17 | ||||
| Standard deviation after the intervention, s2 | 0.41 | 0.41 | ||||
| Pooled variance, s1,22(pooled) | 0.32 | 0.32 | ||||
| Difference within groups before and after intervention, t | 4.4 | 6.6 | ||||
| p value | <0.01 | <0.01 | ||||
| Statistics\Method Applied | PRF vs.DGG (Comparative Differences between Two Applied Methods) | |||||
| Value | CI 95% | |||||
| Mean difference (before intervention), (PRF) − (DGG) | 0.08 | −0.37 | 0.53 | |||
| Mean difference (after intervention), (PRF) − (DGG) | 1.52 | 1.12 | 1.19 | |||
| Pooled variance, s12PRF/DDG(pooled) | 0.47 | |||||
| Difference between groups before intervention, t | 0.4 | |||||
| p value | <0.05 | |||||
| Pooled variance, s22 (PRF/DDGpooled) | 0.17 | |||||
| Difference between groups after intervention PRF vs. DGG, t | 11.8 | |||||
| p value | >0.05 | |||||
| Statistics\Method Applied | PRF | DGG | ||||
|---|---|---|---|---|---|---|
| Value | CI 95% | Value | CI 95% | |||
| Average before the intervention, | 1.38 | 1.24 | 1.52 | 1.24 | 1.12 | 1.36 |
| Average after the intervention, | 3.13 | 2.71 | 3.55 | 4.12 | 3.66 | 4.58 |
| Mean difference, − | 1.75 | 1.31 | 2.19 | 2.88 | 2.40 | 3.36 |
| Standard error of the mean, SEM1 (before intervention) | 0.07 | 0.06 | ||||
| Standard error of the mean, SEM2 (after intervention) | 0.20 | 0.22 | ||||
| Sample size | 20 | 20 | ||||
| Variance before the intervention, s12 | 0.09 | 0.07 | ||||
| Standard deviation before the intervention, s1 | 0.30 | 0.26 | ||||
| Variance after the intervention, s22 | 0.81 | 0.97 | ||||
| Standard deviation after the intervention, s2 | 0.90 | 0.99 | ||||
| Pooled variance, s1,22(pooled) | 0.45 | 0.52 | ||||
| Difference within the group before and after intervention, t | 5.4 | 5.2 | ||||
| p value | <0.01 | <0.01 | ||||
| Statistics\Method Applied | PRF vs. DGG (Comparative Differences between Two Applied Methods) | |||||
| Value | CI 95% | |||||
| Mean difference (before intervention), (PRF) − (DGG) | 0.14 | −0.04 | 0.32 | |||
| Mean difference (after intervention), (PRF) − (DGG) | −0.99 | −1.82 | −0.16 | |||
| Pooled variance, s12PRF/DDG(pooled) | 0.08 | |||||
| Difference between groups before intervention, t | 1.6 | |||||
| p value | <0.05 | |||||
| Pooled variance, s22 (PRF/DDGpooled) | 0.89 | |||||
| Difference between groups after intervention PRF vs. DGG, t | −3.3 | |||||
| p value | >0.05 | |||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jovičić, B.; Matijević, S.; Veličković, S.; Stevanović, M.; Mišić, A.; Stanojević, S.; Bubalo, M. Effectiveness of Plasma-Rich Fibrin and De-Epithelialized Free Gingival Graft in the Treatment of Gingival Recessions. Medicina 2023, 59, 447. https://doi.org/10.3390/medicina59030447
Jovičić B, Matijević S, Veličković S, Stevanović M, Mišić A, Stanojević S, Bubalo M. Effectiveness of Plasma-Rich Fibrin and De-Epithelialized Free Gingival Graft in the Treatment of Gingival Recessions. Medicina. 2023; 59(3):447. https://doi.org/10.3390/medicina59030447
Chicago/Turabian StyleJovičić, Bojan, Stevo Matijević, Stefan Veličković, Momir Stevanović, Aleksandra Mišić, Slavoljub Stanojević, and Marija Bubalo. 2023. "Effectiveness of Plasma-Rich Fibrin and De-Epithelialized Free Gingival Graft in the Treatment of Gingival Recessions" Medicina 59, no. 3: 447. https://doi.org/10.3390/medicina59030447
APA StyleJovičić, B., Matijević, S., Veličković, S., Stevanović, M., Mišić, A., Stanojević, S., & Bubalo, M. (2023). Effectiveness of Plasma-Rich Fibrin and De-Epithelialized Free Gingival Graft in the Treatment of Gingival Recessions. Medicina, 59(3), 447. https://doi.org/10.3390/medicina59030447







