The Role of C-Reactive Protein and Neutrophil to Lymphocyte Ratio in Predicting the Severity of Odontogenic Infections in Adult Patients
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Ethics
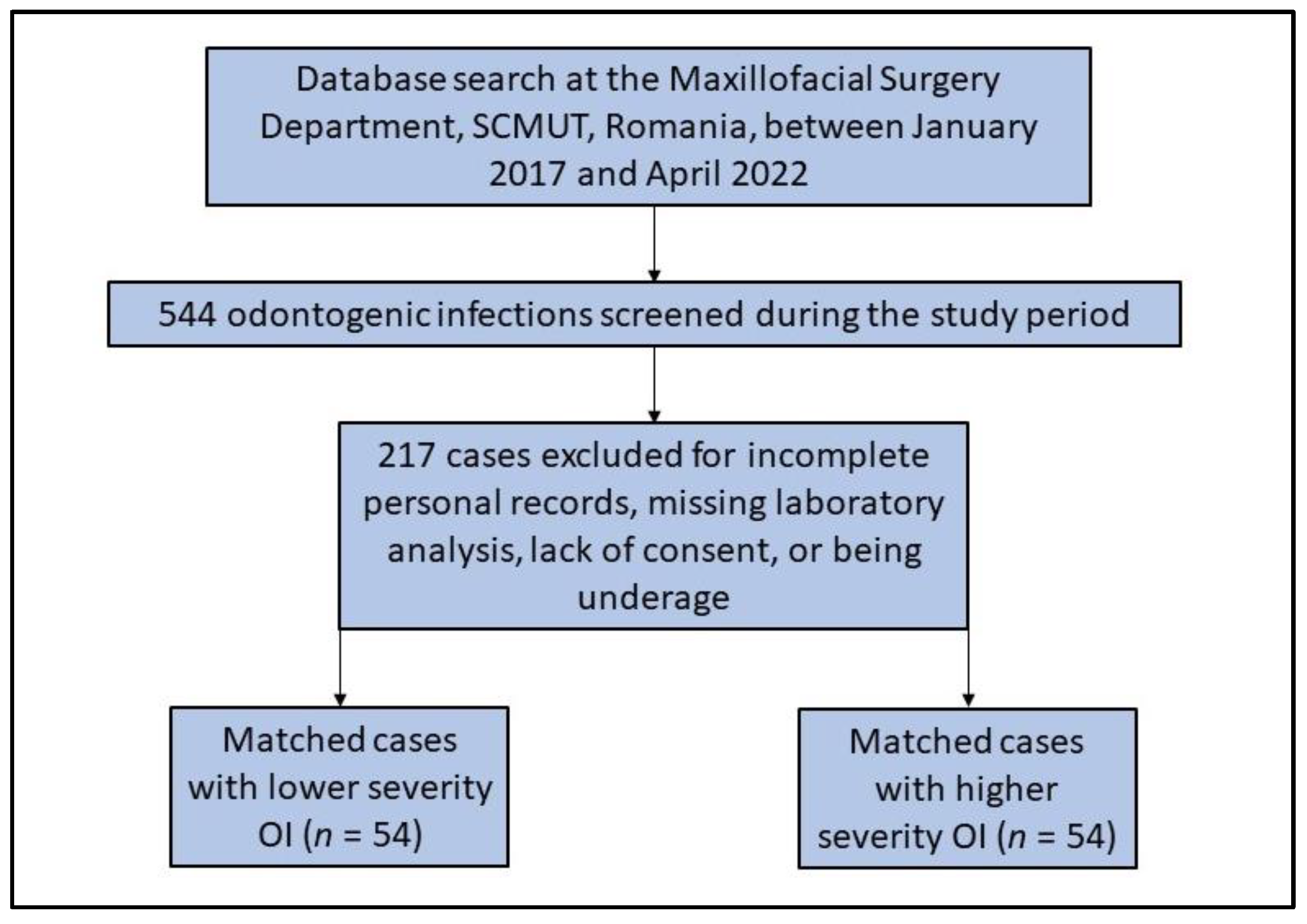
2.2. Patient Selection Process
2.3. Data Collection and Variables
2.4. Statistical Analysis
3. Results
3.1. Demographic Characteristics of the Study Population
3.2. Characteristics of Infection in the Study Population
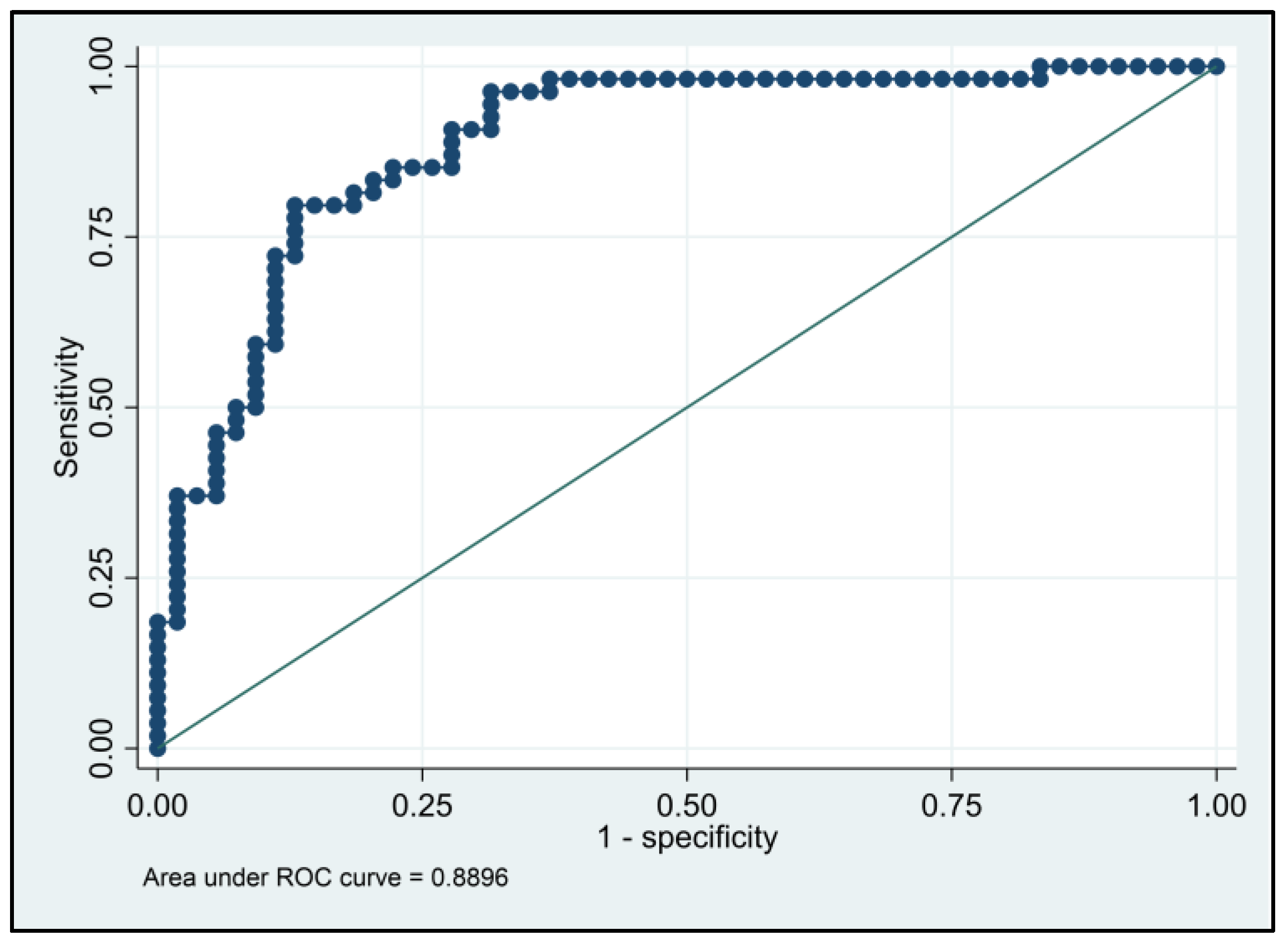
3.3. Risk Assessment in the Study Population
4. Discussion
4.1. Important Findings
4.2. Limitations of the Study
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Alotaibi, N.; Cloutier, L.; Khaldoun, E.; Bois, E.; Chirat, M.; Salvan, D. Criteria for admission of odontogenic infections at high risk of deep neck space infection. Eur. Ann. Otorhinolaryngol. Head Neck Dis. 2015, 132, 261–264. [Google Scholar] [CrossRef]
- Bali, R.K.; Sharma, P.; Gaba, S.; Kaur, A.; Ghanghas, P. A review of complications of odontogenic infections. Natl. J. Maxillofac. Surg. 2015, 6, 136–143. [Google Scholar] [CrossRef]
- Blankson, P.-K.; Parkins, G.; Boamah, M.O.; Abdulai, A.E.; Ahmed, A.-M.; Bondorin, S.; Nuamah, I. Severe odontogenic infections: A 5-year review of a major referral hospital in Ghana. Pan Afr. Med. J. 2019, 32, 71. [Google Scholar] [CrossRef] [PubMed]
- Sánchez, R.; Mirada, E.; Arias, J.; Paño, J.R.; Burgueño Garcia, M. Severe odontogenic infections: Epidemiological, microbiological and therapeutic factors. Med. Oral Patol. Oral Cirugía Bucal 2011, 16, 670–676. [Google Scholar] [CrossRef]
- Stephens, M.B.; Wiedemer, J.P.; Kushner, G.M. Dental Problems in Primary Care. Am. Fam. Physician 2018, 98, 654–660. [Google Scholar]
- Ince, N.; Güçlü, E.; Sungur, M.A.; Karabay, O. Evaluation of neutrophil to lymphocyte ratio, platelet to lymphocyte ratio, and lymphocyte to monocyte ratio in patients with cellulitis. Rev. Assoc. Med. Bras. 2020, 66, 1077–1081. [Google Scholar] [CrossRef]
- Shumilah, A.M.; Othman, A.M.; Al-Madhagi, A.K. Accuracy of neutrophil to lymphocyte and monocyte to lymphocyte ratios as new inflammatory markers in acute coronary syndrome. BMC Cardiovasc. Disord. 2021, 21, 422. [Google Scholar] [CrossRef]
- Spoto, S.; Lupoi, D.M.; Valeriani, E.; Fogolari, M.; Locorriere, L.; Anguissola, G.B.; Battifoglia, G.; Caputo, D.; Coppola, A.; Costantino, S.; et al. Diagnostic Accuracy and Prognostic Value of Neutrophil-to-Lymphocyte and Platelet-to-Lymphocyte Ratios in Septic Patients outside the Intensive Care Unit. Medicina 2021, 57, 811. [Google Scholar] [CrossRef]
- Gurgus, D.; Grigoras, M.L.; Motoc, A.G.M.; Zamfir, C.L.; Cornianu, M.; Faur, C.I.; Pop, D.L.; Folescu, R. Clinical relevance and accuracy of p63 and TTF-1 for better approach of small cell lung carcinoma versus poorly differentiated nonkeratinizing squamous cell carcinoma. Rom. J. Morphol. Embryol. 2019, 60, 139–143. [Google Scholar]
- Bilgen, Ö.; Atici, T.; Durak, K.; Karaeminoğullari; Bilgen, M.S. C-reactive Protein Values and Erythrocyte Sedimentation Rates after Total Hip and Total Knee Arthroplasty. J. Int. Med. Res. 2001, 29, 7–12. [Google Scholar] [CrossRef]
- Jundt, J.S.; Gutta, R. Characteristics and cost impact of severe odontogenic infections. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2012, 114, 558–566. [Google Scholar] [CrossRef] [PubMed]
- Sutter, R.; Tschudin-Sutter, S.; Grize, L.; Widmer, A.F.; Marsch, S.; Rüegg, S. Acute phase proteins and white blood cell levels for prediction of infectious complications in status epilepticus. Crit. Care 2011, 15, R274. [Google Scholar] [CrossRef] [PubMed]
- Boucher, N.E.; Hanrahan, J.J.; Kihara, F.Y. Occurrence of C-Reactive Protein in Oral Disease. J. Dent. Res. 1967, 46, 624. [Google Scholar] [CrossRef] [PubMed]
- Heimdahl, A.; Nord, C.E. Orofacial infections of odontogenic origin. Scand. J. Infect. Dis. 1983, 39, 86–91. [Google Scholar]
- Stathopoulos, P.; Igoumenakis, D.; Shuttleworth, J.; Smith, W.; Ameerally, P. Predictive factors of hospital stay in patients with odontogenic maxillofacial infections: The role of C-reactive protein. Br. J. Oral Maxillofac. Surg. 2017, 55, 367–370. [Google Scholar] [CrossRef] [PubMed]
- Ioan Faur, C.; Abu-Awwad, A.; Pop, D.L.; Zamfir, C.L.; Gurgus, D.; Hoinoiu, T.; Motoc, A.; Haivas, C.; Grigoraș, M.L.; Folescu, R. Liquid Nitrogen Efficiency in Treatment of Giant Cell Tumor of Bone and Prevention of Recurrence. Appl. Sci. 2020, 10, 6310. [Google Scholar] [CrossRef]
- Pepys, M.B.; Hirschfield, G.M. C-reactive protein: A critical update. J. Clin. Investig. 2003, 111, 1805–1812. [Google Scholar] [CrossRef]
- De Jager, C.P.; Van Wijk, P.T.; Mathoera, R.B.; De Jongh-Leuvenink, J.; Van Der Poll, T.; Wever, P.C. Lymphocytopenia and neutrophil-lymphocyte count ratio predict bacteremia better than conventional infection markers in an emergency care unit. Crit. Care 2010, 14, R192. [Google Scholar] [CrossRef]
- Lowsby, R.; Gomes, C.; Jarman, I.; Lisboa, P.; Nee, P.A.; Vardhan, M.; Eckersley, T.; Saleh, R.; Mills, H. Neutrophil to lymphocyte count ratio as an early indicator of bloodstream infection in the emergency department. Emerg. Med. J. 2015, 32, 531–534. [Google Scholar] [CrossRef]
- Jiang, J.; Liu, R.; Yu, X.; Yang, R.; Xu, H.; Mao, Z.; Wang, Y. The neutrophil-lymphocyte count ratio as a diagnostic marker for bacteremia: A systematic review and meta-analysis. Am. J. Emerg. Med. 2019, 37, 1482–1489. [Google Scholar] [CrossRef]
- Belei, O.; Ancusa, O.; Mara, A.; Olariu, L.; Amaricai, E.; Folescu, R.; Zamfir, C.L.; Gurgus, D.; Motoc, A.G.; Stanga, L.C.; et al. Current Paradigm of Hepatitis E Virus Among Pediatric and Adult Patients. Front. Pediatr. 2021, 30, 721918. [Google Scholar] [CrossRef] [PubMed]
- Niu, D.; Huang, Q.; Yang, F.; Tian, W.; Li, C.; Ding, L.; Fang, H.-C.; Zhao, Y. Serum biomarkers to differentiate Gram-negative, Gram-positive and fungal infection in febrile patients. J. Med. Microbiol. 2021, 70, 001360. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Hou, M.; Ding, Z.; Liu, X.; Shao, Y.; Li, X. Prognostic Value of Neutrophil-to-Lymphocyte Ratio in Stroke: A Systematic Review and Meta-Analysis. Front. Neurol. 2021, 12, 686983. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.-J.; Park, S.-D.; Kwon, S.W.; Woo, S.-I.; Lee, M.-D.; Shin, S.-H.; Kim, D.-H.; Kwan, J.; Park, K.-S. Relation Between Neutrophil-to-Lymphocyte Ratio and Index of Microcirculatory Resistance in Patients With ST-Segment Elevation Myocardial Infarction Undergoing Primary Percutaneous Coronary Intervention. Am. J. Cardiol. 2016, 118, 1323–1328. [Google Scholar] [CrossRef] [PubMed]
- Adamstein, N.H.; MacFadyen, J.G.; Rose, L.M.; Glynn, R.J.; Dey, A.K.; Libby, P.; Tabas, I.A.; Mehta, N.N.; Ridker, P.M. The neutrophil–lymphocyte ratio and incident atherosclerotic events: Analyses from five contemporary randomized trials. Eur. Heart J. 2021, 42, 896–903. [Google Scholar] [CrossRef]
- Park, J.M. Neutrophil-to-lymphocyte ratio in trauma patients. J. Trauma Acute Care Surg. 2017, 82, 225–226. [Google Scholar] [CrossRef]
- Lee, P.Y.; Oen, K.Q.X.; Lim, G.R.S.; Hartono, J.L.; Muthiah, M.; Huang, D.Q.; Teo, F.S.W.; Li, A.Y.; Mak, A.; Chandran, N.S.; et al. Neutrophil-to-Lymphocyte Ratio Predicts Development of Immune-Related Adverse Events and Outcomes from Immune Checkpoint Blockade: A Case-Control Study. Cancers 2021, 13, 1308. [Google Scholar] [CrossRef]
- Fest, J.; Ruiter, T.R.; Koerkamp, B.G.; Rizopoulos, D.; Ikram, M.A.; Van Eijck, C.H.J.; Stricker, B.H. The neutrophil-to-lymphocyte ratio is associated with mortality in the general population: The Rotterdam Study. Eur. J. Epidemiol. 2019, 34, 463–470. [Google Scholar] [CrossRef]
- Miloro, M.; Ghali, G.E.; Larsen, P.; Waite, P. Peterson’s Principles of Oral and Maxillofacial Surgery, 3rd ed.; People’s Medical Publishing House: Shelton, CT, USA, 2012; pp. 841–861. [Google Scholar]
- Sainuddin, S.; Hague, R.; Howson, K.; Clark, S. New admission scoring criteria for patients with odontogenic infections: A pilot study. J. Oral Maxilloc. Surg. 2016, 55, 86–89. [Google Scholar] [CrossRef]
- Steindel, S.J. International classification of diseases, 10th edition, clinical modification and procedure coding system: Descriptive overview of the next generation HIPAA code sets. J. Am. Med. Inform. Assoc. 2010, 17, 274–282. [Google Scholar] [CrossRef]
- Lambden, S.; Laterre, P.F.; Levy, M.M.; Francois, B. The SOFA score—Development, utility and challenges of accurate assessment in clinical trials. Crit. Care 2019, 23, 374. [Google Scholar] [CrossRef] [PubMed]
- Seppänen, L.; Rautemaa, R.; Lindqvist, C.; Lauhio, A.; Richardson, R. Changing clinical features of odontogenic maxillofacial infections. Clin. Oral Investig. 2010, 14, 459–465. [Google Scholar] [CrossRef] [PubMed]
- Burnham, R.; Bhandari, R.; Bridle, C. Changes in admission rates for spreading odontogenic infection resulting from changes in government policy about the dental schedule and remunerations. Br. J. Oral Maxillofac. Surg. 2011, 49, 26–28. [Google Scholar] [CrossRef] [PubMed]
- Edman-Wallér, J.; Ljungström, L.; Jacobsson, G.; Andersson, R.; Werner, M. Systemic symptoms predict presence or development of severe sepsis and septic shock. Infect. Dis. 2015, 48, 209–214. [Google Scholar] [CrossRef]
- Póvoa, P.; Coelho, L.; Almeida, E.; Fernandes, A.; Mealha, R.; Moreira, P.; Sabino, H. Early identification of intensive care unit-acquired infections with daily monitoring of C-reactive protein: A prospective observational study. Crit. Care 2006, 10, R63. [Google Scholar] [CrossRef]
- Bagul, R.; Chandan, S.; Sane, V.D.; Patil, S.; Yadav, D. Comparative Evaluation of C-Reactive Protein and WBC Count in Fascial Space Infections of Odontogenic Origin. J. Maxillofac. Oral Surg. 2016, 16, 238–242. [Google Scholar] [CrossRef]
- John, C.R.; Gandhi, S.; Singh, I.; James, T.T. Efficacy of C-Reactive protein as a marker in patients with odontogenic fascial space infection: A prospective analytical study. J. NTR Univ. Health. Sci. 2021, 10, 76–81. [Google Scholar]
- Barreto, V.T.; Isaac, A.; Bhimidi, P.; Nguyen, C.; Jones, G. Trends of C-Reactive Protein Laboratory Values With White Blood Cell Count Levels in Maxillofacial Infections. J. Oral Maxillofac. Surg. 2013, 71, 31–32. [Google Scholar] [CrossRef]
- Huang, Z.; Fu, Z.; Huang, W.; Huang, K. Prognostic value of neutrophil-to-lymphocyte ratio in sepsis: A meta-analysis. Am. J. Emerg. Med. 2020, 38, 641–647. [Google Scholar] [CrossRef]
- Josse, J.M.; Cleghorn, M.C.; Ramji, K.M.; Jiang, H.; Elnahas, A.; Jackson, T.D.; Okrainec, A.; Quereshy, F.A. The neutrophil/lymphocyte ratio predicts major perioperative complications in patients undergoing colorectal surgery. Color. Dis. 2016, 18, 236–242. [Google Scholar] [CrossRef]
- Silberman, S.; Abu-Yunis, U.; Tauber, R.; Shavit, L.; Grenader, T.; Fink, D.; Bitran, D.; Merin, O. Neutrophil-Lymphocyte Ratio: Prognostic Impact in Heart Surgery. Early Outcomes and Late Survival. Ann. Thorac. Surg. 2018, 105, 581–586. [Google Scholar] [CrossRef] [PubMed]
- Hajibandeh, S.; Hajibandeh, S.; Hobbs, N.; Mansour, M. Neutrophil-to-lymphocyte ratio predicts acute appendicitis and distinguishes between complicated and uncomplicated appendicitis: A systematic review and meta-analysis. Am. J. Surg. 2020, 219, 154–163. [Google Scholar] [CrossRef] [PubMed]
- Gallagher, N.; Collyer, J.; Bowe, C.M. Neutrophil to Lymphocyte Ratio as a Prognostic Marker of Deep Neck Space Infections Secondary to Odontogenic Infection. Br. J. Oral Maxillofac. Surg. 2021, 59, 228–232. [Google Scholar] [CrossRef] [PubMed]
- Russell, C.D.; Parajuli, A.; Gale, H.J.; Bulteel, N.S.; Schuetz, P.; de Jager, C.P.; Loonen, A.J.; Merekoulias, G.I.; Baillie, J.K. The utility of peripheral blood leucocyte ratios as biomarkers in infectious diseases: A systematic review and meta-analysis. J. Infect. 2019, 78, 339–348. [Google Scholar] [CrossRef] [PubMed]
- Vatankhah, N.; Jahangiri, Y.; Landry, G.J.; McLafferty, R.B.; Alkayed, N.J.; Moneta, G.L.; Azarbal, A.F. Predictive value of neutrophil-to lymphocyte ratio in diabetic wound healing. J. Vasc. Surg. 2017, 65, 478–483. [Google Scholar] [CrossRef]
- Dogruel, F.; Gonen, Z.-B.; Gunay-Canpolat, D.; Zararsiz, G.; Alkan, A. The Neutrophil-to-Lymphocyte ratio as a marker of recovery status in patients with severe dental infection. Med. Oral Patol. Oral Cir. Bucal. 2017, 22, 440–445. [Google Scholar] [CrossRef]
- Fu, B.; McGowan, K.; Sun, J.H.; Batstone, M. Increasing frequency and severity of odontogenic infection requiring hospital admission and surgical management. Br. J. Oral Maxillofac. Surg. 2020, 58, 409–415. [Google Scholar] [CrossRef]
| Criteria | Score | Max Score | |
|---|---|---|---|
| Systemic Inflammatory Response Syndrome (SIRS) | Temperature > 38.3 °C | 1 | 4 |
| Heart rate > 90 bpm | 1 | ||
| RR 20/min | 1 | ||
| WBC < 4 or >12 × 109 | 1 | ||
| Trismus | Moderate < 2 cm | 3 | 4 |
| Severe < 1 cm | 4 | ||
| Dysphagia | Mild—able to swallow most foods | 2 | 5 |
| Moderate—unable to swallow fluids | 4 | ||
| Severe—drooling saliva | 5 | ||
| Collection in 1 fascial space | Low severity (canine, vestibular) | 1 | 5 |
| Moderate severity (buccal) | 2 | ||
| High severity (all other spaces) | 4 | ||
| Collection in 2 or more fascial spaces | 5 | ||
| Sign of dehydration (↓BP/↑Urea/↓Skin turgor) | 1 | 2 | |
| Comorbidities: diabetes mellitus, immunocompromised status, known or suspected chronic alcohol misuser | 1 | ||
| Total Score | 20 | ||
| Variables | Group A (n = 54) | Group B (n = 54) | Significance |
|---|---|---|---|
| Gender | 0.236 | ||
| Men | 30 (55.6%) | 36 (66.7%) | |
| Women | 24 (44.4%) | 18 (33.3%) | |
| Age, mean (mean ± SD) | 46.7 ± 17.9 | 51.7 ± 18.1 | 0.150 |
| Age range | 18–81 | 20–85 | NA |
| Place of origin | 0.019 | ||
| Rural | 25 (46.3%) | 37 (68.5%) | |
| Urban | 29 (53.7%) | 17 (31.5%) | |
| Smoking | 0.028 | ||
| Yes | 9 (16.7%) | 19 (35.2%) | |
| No | 45 (83.3%) | 35 (64.8%) | |
| Comorbidities | |||
| Diabetes mellitus | 10 (18.5%) | 28 (51.9%) | <0.001 |
| Obesity | 31 (57.4%) | 37 (68.5%) | 0.231 |
| Chronic kidney disease | 14 (25.9%) | 17 (31.5%) | 0.523 |
| Malignancy | 5 (9.3%) | 7 (13.0%) | 0.540 |
| Others | 2 (3.7%) | 4 (7.4%) | 0.401 |
| Variables | Group A (n = 54) | Group B (n = 54) | Significance |
|---|---|---|---|
| Reason for hospitalization | <0.001 | ||
| Abscess | 38 (70.4%) | 17 (31.5%) | |
| Cellulitis | 5 (9.3%) | 7 (13.0%) | |
| Association of abscess and cellulitis | 11 (20.4%) | 30 (55.6%) | |
| Infection site | |||
| Peri-maxillary | 13 (24.1%) | 10 (18.5%) | 0.480 |
| Peri-mandibular | 14 (25.9%) | 18 (33.3%) | 0.399 |
| Superficial lodges | 22 (40.7%) | 26 (48.1%) | 0.438 |
| Deep lodges | 1 (1.9%) | 2 (3.7%) | 0.558 |
| Fascial | 5 (9.3%) | 3 (5.6%) | 0.462 |
| Outcomes | |||
| Sepsis | 4 (7.4%) | 12 (22.2%) | 0.030 |
| ICU admission | 0 (0.0%) | 4 (7.4%) | 0.041 |
| Duration of hospitalization, median (IQR) | 4.1 (2.8) | 12.0 (5.7) | <0.001 |
| Severe complications | 2 (3.7%) | 9 (16.7%) | 0.025 |
| Mortality | 0 (0.0%) | 3 (5.6%) | 0.078 |
| Variables | Group A (n = 54) | Group B (n = 54) | Significance |
|---|---|---|---|
| SIRS score | <0.001 | ||
| 0 | 14 (25.9%) | 5 (9.2%) | |
| 1 | 18 (33.3%) | 8 (14.8%) | |
| 2 | 10 (18.5%) | 8 (14.8%) | |
| 3 | 6 (11.1%) | 19 (35.2%) | |
| 4 | 1 (1.8%) | 19 (35.2%) | |
| Trismus score | <0.001 | ||
| Normal | 30 (55.6%) | 12 (22.2%) | |
| Moderate | 19 (35.2%) | 15 (27.8%) | |
| Severe | 5 (9.3%) | 27 (50.0%) | |
| Dysphagia score | 0.028 | ||
| Normal | 5 (9.3%) | 18 (33.3%) | |
| Mild | 21 (38.9%) | 16 (29.6%) | |
| Moderate | 17 (31.5%) | 29 (53.7%) | |
| Severe | 0 (0.0%) | 2 (3.7%) | |
| Fascial space score | <0.001 | ||
| Low risk | 39 (0.0%) | 10 (18.5%) | |
| Moderate risk | 23 (42.6%) | 27 (50.0%) | |
| Severe risk | 0 (0.0%) | 8 (14.8%) | |
| Dehydration/Comorbid | 0.001 | ||
| No dehydration and comorbid | 28 (51.9%) | 13 (24.1%) | |
| Dehydration or comorbid | 26 (48.1%) | 22 (40.7%) | |
| Dehydration and comorbid | 3 (5.6%) | 16 (29.6%) |
| Variables | Group A (n = 54) | Group B (n = 54) | Significance |
|---|---|---|---|
| Severity scores, (mean ± SD) | |||
| SS | 6.1 ± 1.8 | 13.6 ± 3.9 | <0.001 * |
| SII | 696.3 ± 35.2 | 2312.4 ± 66.0 | <0.001 * |
| Biomarker scores (median, IQR) | |||
| WBC, (median, IQR) | 9.34 (7.92–11.50) | 12.02 (10.3–17) | <0.001 ** |
| WBC_Ne, (median, IQR) | 6.26 (4.68–10.23) | 8.05 (6.73–10.61) | 0.012 ** |
| WBC_Ly, (median, IQR) | 2.04 (1.41–2.7) | 2.56 (2.06–3) | 0.037 ** |
| NLR, (median, IQR) | 3.01 (2.10–4.83) | 3.31 (3–4.37) | 0.239 ** |
| CRP, (median, IQR) | 22 (9–47) | 99 (86–118) | <0.001 ** |
| CRP-NLR, (median, IQR) | 79.00 (22.14–191.4) | 341.47 (256.97–526.30) | <0.001 ** |
| Variables | Risk (95% CI) | Significance |
|---|---|---|
| SS (dependent variable) | ||
| WBC | 5.54 (3.18–7.90) | <0.001 |
| WBC_Ne | 7.10 (5.19–9.01) | <0.001 |
| WBC_Ly | 8.62 (7.44–9.81) | <0.001 |
| NLR | 4.46 (3.53–5.40) | <0.001 |
| CRP | 6.65 (5.61–7.70) | <0.001 |
| CRP-NLR | 7.28 (4.83–10.16) | <0.001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rosca, O.; Bumbu, B.A.; Ancusa, O.; Talpos, S.; Urechescu, H.; Ursoniu, S.; Bloanca, V.; Pricop, M. The Role of C-Reactive Protein and Neutrophil to Lymphocyte Ratio in Predicting the Severity of Odontogenic Infections in Adult Patients. Medicina 2023, 59, 20. https://doi.org/10.3390/medicina59010020
Rosca O, Bumbu BA, Ancusa O, Talpos S, Urechescu H, Ursoniu S, Bloanca V, Pricop M. The Role of C-Reactive Protein and Neutrophil to Lymphocyte Ratio in Predicting the Severity of Odontogenic Infections in Adult Patients. Medicina. 2023; 59(1):20. https://doi.org/10.3390/medicina59010020
Chicago/Turabian StyleRosca, Ovidiu, Bogdan Andrei Bumbu, Oana Ancusa, Serban Talpos, Horatiu Urechescu, Sorin Ursoniu, Vlad Bloanca, and Marius Pricop. 2023. "The Role of C-Reactive Protein and Neutrophil to Lymphocyte Ratio in Predicting the Severity of Odontogenic Infections in Adult Patients" Medicina 59, no. 1: 20. https://doi.org/10.3390/medicina59010020
APA StyleRosca, O., Bumbu, B. A., Ancusa, O., Talpos, S., Urechescu, H., Ursoniu, S., Bloanca, V., & Pricop, M. (2023). The Role of C-Reactive Protein and Neutrophil to Lymphocyte Ratio in Predicting the Severity of Odontogenic Infections in Adult Patients. Medicina, 59(1), 20. https://doi.org/10.3390/medicina59010020







