Site-Specific, Critical Threshold Barefoot Peak Plantar Pressure Associated with Diabetic Foot Ulcer History: A Novel Approach to Determine DFU Risk in the Clinical Setting
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Participants
2.3. Assessments
2.4. Pressure Analysis
2.5. Statistical Methods
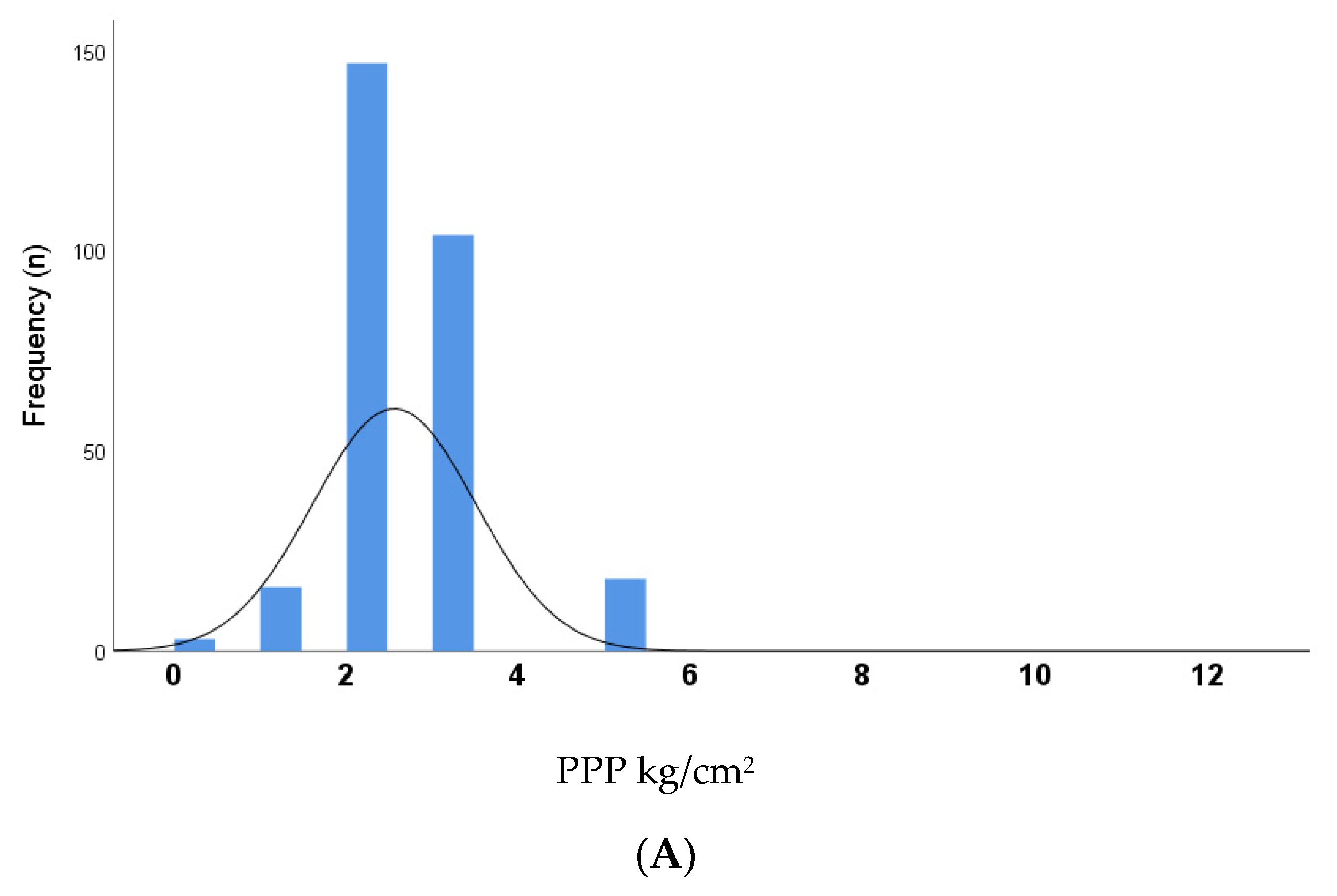
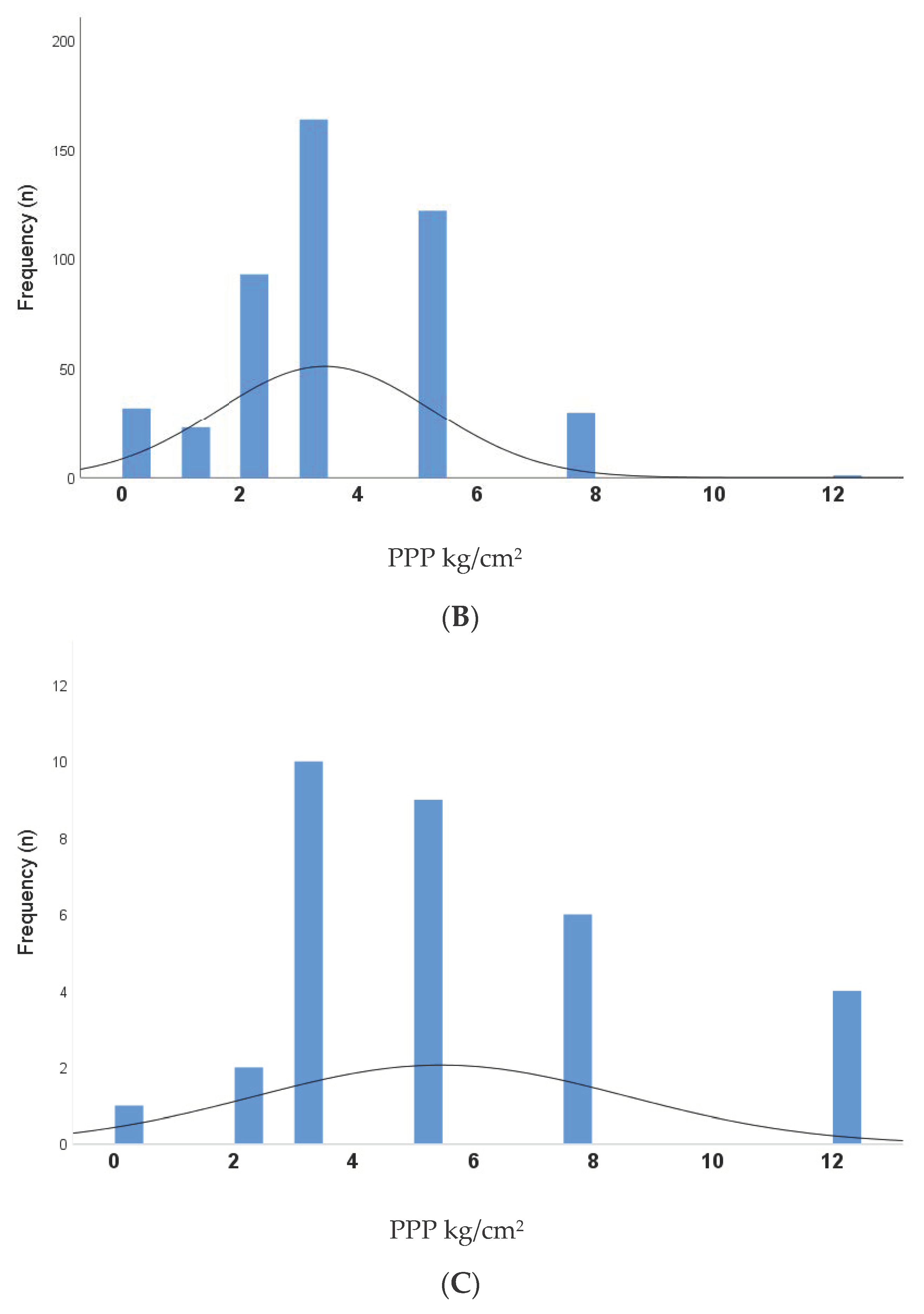
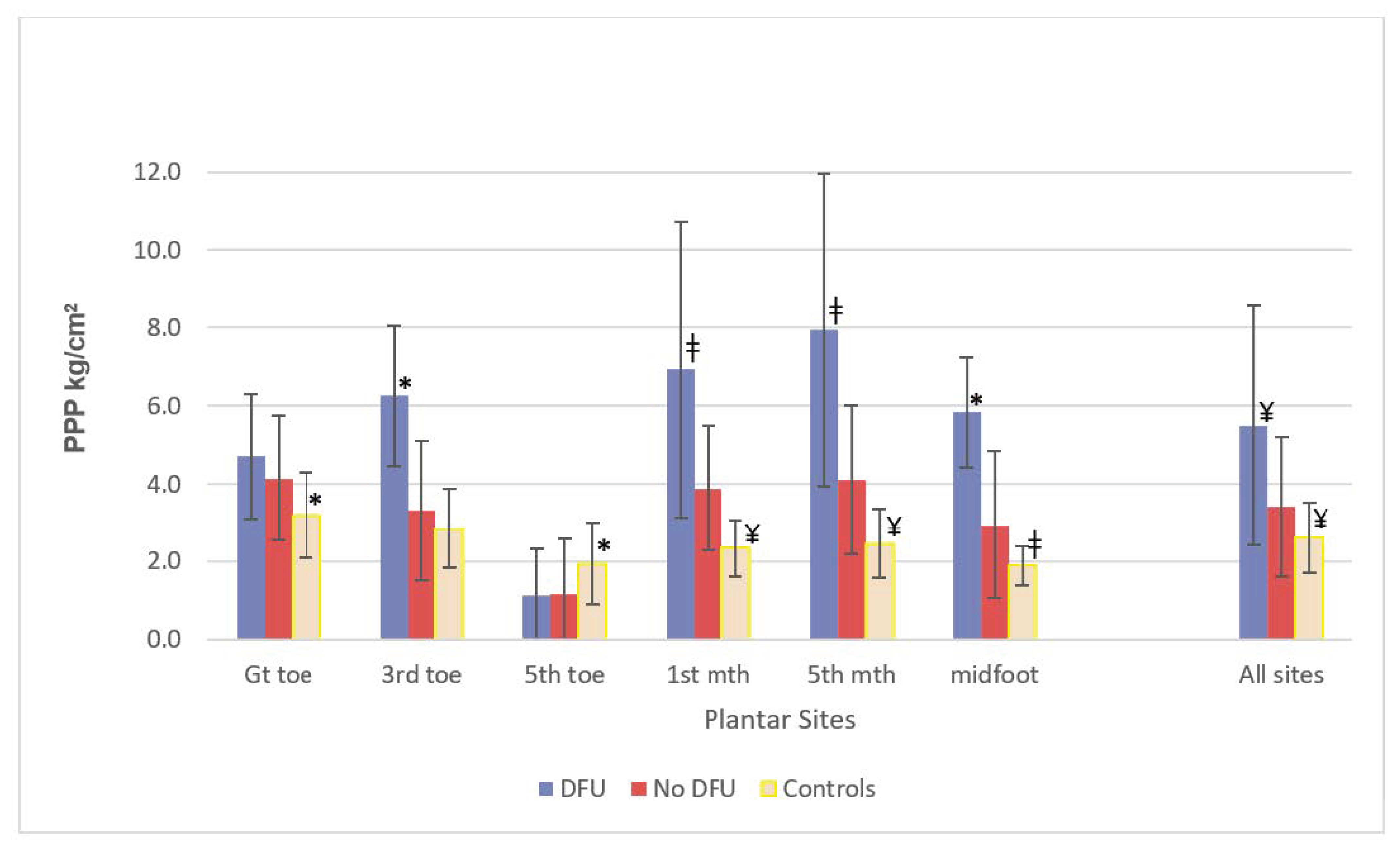
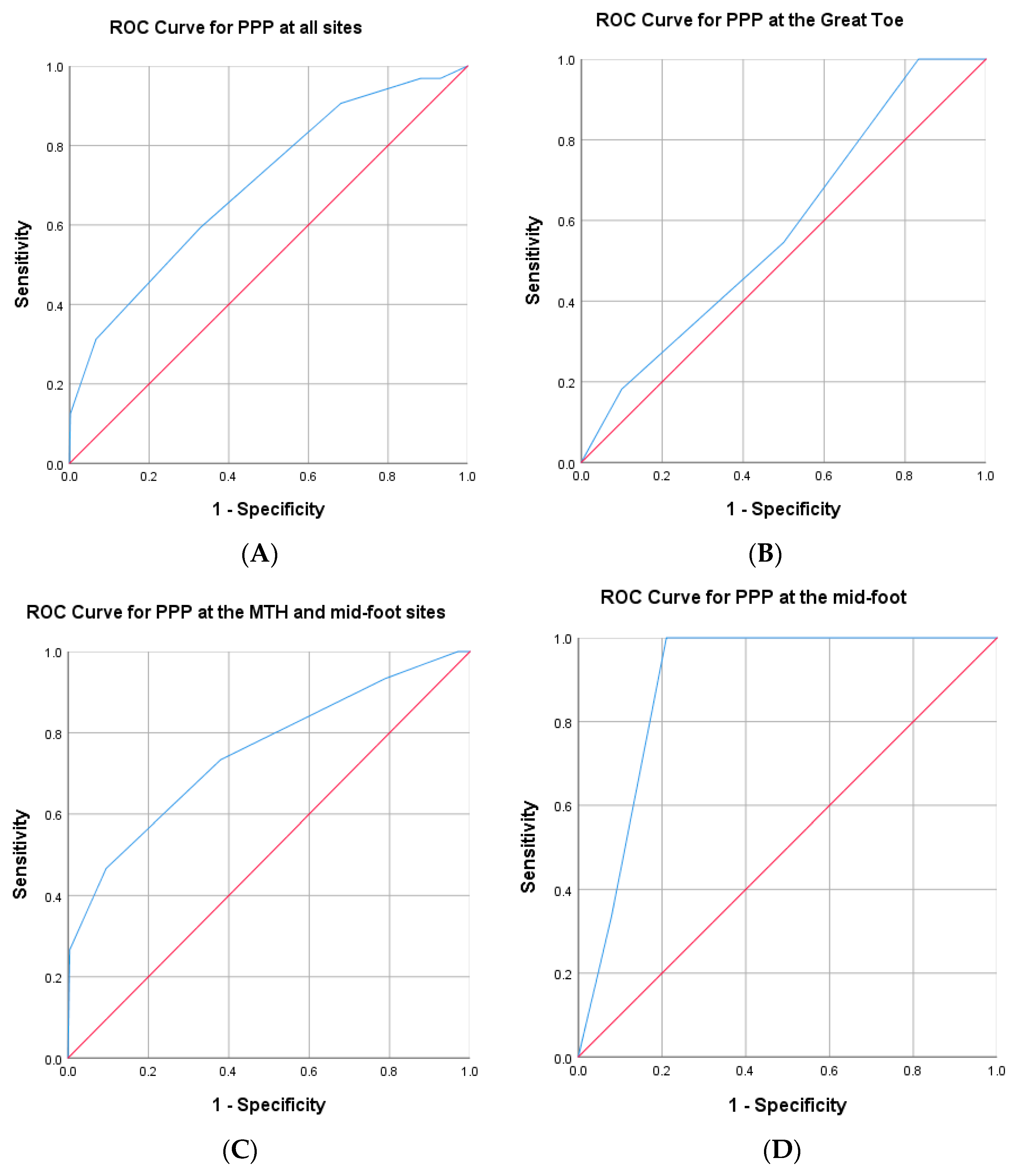
3. Results
3.1. Cross-Sectional Analysis of PPP and DFU Risk
ROC Curves and Critical Threshold for DFU History
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| PPP | peak plantar pressure |
| DFU | diabetic foot ulcer |
| MTH | metatarsal head |
| NDS | neuropathy disability score |
| ICC | intraclass correlation co-efficient |
| ROC | receiver operator characteristic |
References
- Boulton, A.J.M. The Pathway to Ulceration: Aetiopathogenesis and Screening. In The Foot in Diabetes, 5th ed.; Boulton, A.J.M., Rayman, G., Wukich, D.K., Eds.; Wiley: Hoboken, NJ, USA, 2020; pp. 105–124. [Google Scholar]
- Bus, S.A.; Armstrong, D.G.; Gooday, C.; Jarl, G.; Caravaggi, C.; Viswanathan, V.; Lazzarini, P.A.; on behalf of the International Working Group on the Diabetic Foot (IWGDF). Guidelines on offloading foot ulcers in persons with diabetes (IWGDF 2019 update). Diabetes Metab. Res. Rev. 2020, 36 (Suppl. 1). [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stess, R.M.; Jensen, S.R.; Mirmiran, R. The role of dynamic plantar pressures in diabetic foot ulcers. Diabetes Care 1997, 20, 855–858. [Google Scholar] [CrossRef]
- Frykberg, R.G.; Lavery, L.A.; Pham, H.; Harvey, C.; Harkless, L.; Veves, A. Role of neuropathy and high foot pressures in diabetic foot ulceration. Diabetes Care 1998, 21, 1714–1719. [Google Scholar] [CrossRef]
- Fernando, M.E.; Crowther, R.G.; Pappas, E.; Lazzarini, P.A.; Cunningham, M.; Sangla, K.S.; Buttner, P.; Golledge, J. Plantar pressure in diabetic peripheral neuropathy patients with active foot ulceration, previous ulceration and no history of ulceration: A meta-analysis of observational studies. PLoS ONE 2014, 9, e99050. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abbott, C.A.; Carrington, A.L.; Ashe, H.; Bath, S.; Every, L.C.; Griffiths, J.; Hann, A.W.; Hussein, A.; Jackson, N.; Johnson, K.E.; et al. The North-West Diabetes Foot Care Study: Incidence of, and risk factors for, new diabetic foot ulceration in a community-based patient cohort. Diabet. Med. 2002, 19, 377–384. [Google Scholar] [CrossRef] [PubMed]
- Crawford, F.; Cezard, G.; Chappell, F.M.; Murray, G.D.; Price, J.F.; Sheikh, A.; Simpson, C.R.; Stansby, G.P.; Young, M.J. A systematic review and individual patient data meta-analysis of prognostic factors for foot ulceration in people with diabetes: The international research collaboration for the prediction of diabetic foot ulcerations (PODUS). Health Technol. Assess. 2015, 19, 1–210. [Google Scholar] [CrossRef]
- Armstrong, D.G.; Boulton, A.J.M.; Bus, S.A. Diabetic Foot Ulcers and Their Recurrence. N. Engl. J. Med. 2017, 376, 2367–2375. [Google Scholar] [CrossRef]
- Veves, A.; Murray, H.J.; Young, M.J.; Boulton, A.J. The risk of foot ulceration in diabetic patients with high foot pressure: A prospective study. Diabetologia 1992, 35, 660–663. [Google Scholar] [CrossRef]
- Pham, H.; Armstrong, D.G.; Harvey, C.; Harkless, L.B.; Giurini, J.M.; Veves, A. Screening techniques to identify people at high risk for diabetic foot ulceration: A prospective multicenter trial. Diabetes Care 2000, 23, 606–611. [Google Scholar] [CrossRef] [Green Version]
- Lavery, L.A.; Armstrong, D.G.; Wunderlich, R.P.; Tredwell, J.; Boulton, A.J. Predictive value of foot pressure assessment as part of a population-based diabetes disease management program. Diabetes Care 2003, 26, 1069–1073. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Waaijman, R.; De Haart, M.; Arts, M.L.; Wever, D.; Verlouw, A.J.; Nollet, F.; Bus, S.A. Risk factors for plantar foot ulcer recurrence in neuropathic diabetic patients. Diabetes Care 2014, 37, 1697–1705. [Google Scholar] [CrossRef] [Green Version]
- Chatwin, K.E.; Abbott, C.A.; Boulton, A.J.M.; Bowling, F.L.; Reeves, N.D. The role of foot pressure measurement in the prediction and prevention of diabetic foot ulceration-A comprehensive review. Diabetes Metab. Res. Rev. 2020, 36, e3258. [Google Scholar] [CrossRef] [PubMed]
- Abbott, C.A.; Garrow, A.P.; Carrington, A.L.; Morris, J.; Van Ross, E.R.; Boulton, A.J. Foot ulcer risk is lower in South-Asian and African-Caribbean compared with European diabetic patients in the U.K.: The North-West diabetes foot care study. Diabetes Care 2005, 28, 1869–1875. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Singh, N.; Armstrong, D.G.; Lipsky, B.A. Preventing foot ulcers in patients with diabetes. JAMA 2005, 293, 217–228. [Google Scholar] [CrossRef]
- Owings, T.M.; Apelqvist, J.; Stenström, A.; Becker, M.; Bus, S.A.; Kalpen, A.; Ulbrecht, J.S.; Cavanagh, P.R. Plantar pressures in diabetic patients with foot ulcers which have remained healed. Diabet. Med. 2009, 26, 1141–1146. [Google Scholar] [CrossRef]
- Ledoux, W.R.; Shofer, J.B.; Cowley, M.S.; Ahroni, J.H.; Cohen, V.; Boyko, E.J. Diabetic foot ulcer incidence in relation to plantar pressure magnitude and measurement location. J. Diabetes Complicat. 2013, 27, 621–626. [Google Scholar] [CrossRef] [Green Version]
- Petersen, B.J.; Rothenberg, G.M.; Lakhani, P.J.; Zhou, M.; Linders, D.R.; Bloom, J.D.; Wood, K.A.; Armstrong, D.G. Ulcer metastasis? Anatomical locations of recurrence for patients in diabetic foot remission. J. Foot Ankle Res. 2020, 13, 1. [Google Scholar] [CrossRef] [PubMed]
- Örneholm, H.; Apelqvist, J.; Larsson, J.; Eneroth, M. Recurrent and other new foot ulcers after healed plantar forefoot diabetic ulcer. Wound Repair Regen. 2017, 25, 309–315. [Google Scholar] [CrossRef] [PubMed]
- Abbott, C.A.; Chatwin, K.E.; Foden, P.; Hasan, A.N.; Sange, C.; Rajbhandari, S.M.; Reddy, P.N.; Vileikyte, L.; Bowling, F.L.; Boulton, A.J.; et al. Innovative intelligent insole system reduces diabetic foot ulcer recurrence at plantar sites: A prospective, randomised, proof-of-concept study. Lancet Digit. Health 2019, 1, 308–318. [Google Scholar] [CrossRef] [Green Version]
- Van Schie, C.H.; Abbott, C.A.; Vileikyte, L.; Shaw, J.E.; Hollis, S.; Boulton, A.J. A comparative study of the Podotrack, a simple semiquantitative plantar pressure measuring device, and the optical pedobarograph in the assessment of pressures under the diabetic foot. Diabet. Med. 1999, 16, 154–159. [Google Scholar] [CrossRef]
- Oyibo, S.O.; Jude, E.B.; Tarawneh, I.; Nguyen, H.C.; Harkless, L.B.; Boulton, A.J. A comparison of two diabetic foot ulcer classification systems: The Wagner and the University of Texas wound classification systems. Diabetes Care 2001, 24, 84–88. [Google Scholar] [CrossRef] [Green Version]
- Caselli, A.; Pham, H.; Giurini, J.M.; Armstrong, D.G.; Veves, A. The Forefoot-to-Rearfoot Plantar Pressure Ratio Is Increased in Severe Diabetic Neuropathy and Can Predict Foot Ulceration. Diabetes Care 2002, 25, 1066–1071. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Armstrong, D.G.; Peters, E.J.; Athanasiou, K.A.; Lavery, L.A. Is there a critical level of plantar foot pressure to identify patients at risk for neuropathic foot ulceration? J. Foot Ankle Surg. 1998, 37, 303–307. [Google Scholar] [CrossRef]
- Molines-Barroso, R.J.; Lázaro-Martínez, J.L.; Beneit-Montesinos, J.V.; Álvaro-Afonso, F.J.; García-Morales, E.; García-Álvarez, Y. Predictors of Diabetic Foot Reulceration beneath the Hallux. J. Diabetes Res. 2019, 2019, 9038171. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yavuz, M.; Master, H.; Garrett, A.; Lavery, L.A.; Adams, L.S. Peak Plantar Shear and Pressure and Foot Ulcer Locations: A Call to Revisit Ulceration Pathomechanics. Diabetes Care 2015, 38, e184–e185. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bacarin, T.A.; Sacco, I.C.; Hennig, E.M. Plantar pressure distribution patterns during gait in diabetic neuropathy patients with a history of foot ulcers. Clinics 2009, 64, 113–120. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Armstrong, D.G.; Lavery, L.A. Elevated peak plantar pressures in patients who have Charcot arthropathy. J. Bone Jt. Surg. Am. 1998, 80, 365–369. [Google Scholar] [CrossRef] [PubMed]
- Owings, T.M.; Woerner, J.L.; Frampton, J.D.; Cavanagh, P.R.; Botek, G. Custom therapeutic insoles based on both foot shape and plantar pressure measurement provide enhanced pressure relief. Diabetes Care 2008, 31, 839–844. [Google Scholar] [CrossRef] [Green Version]
- Binning, J.; Woodburn, J.; Bus, S.A.; Barn, R. Motivational interviewing to improve adherence behaviours for the prevention of diabetic foot ulceration. Diabetes Metab. Res. Rev. 2019, 35, e3105. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Characteristics | Diabetes Group (n = 21) | Control Group (n = 12) |
|---|---|---|
| Age (years) | 60.7 ± 9∙5 | 58.5 ± 8.3 |
| Male gender, n (%) | 20 (95.2%) | 4 (33.3%) |
| Type 2 diabetes, n (%) | 11 (52.4%) | |
| Diabetes duration (years) | 28.8 ± 13.4 | |
| HbA1c (mmol/mol) (%) | 64.7 ± 10.4 8.1 ± 3.1 | |
| Ethnicity, n (%): | ||
| White British/Other White | 17 (81.0%) | 10 (83.3%) |
| South Asian | 2 (9.6%) | 1 (8.3%) |
| Black/Mixed race | 2 (9.6%) | 1 (8.3%) |
| Weight (kg) | 97.9 ± 22.2 | 71.9 ± 11.9 |
| BMI (kg/m2) | 30.9 ± 6.7 | 26.2 ± 3.8 |
| Neuropathy Disability Score (NDS): | 8.3 ± 2.1 | |
| Previous toe amputations | ||
| Hallux | 1 (4.8%) | |
| 2nd—5th toe | 1 (4.8%) | |
| Previous DFU on left foot: | ||
| Hallux | 6 (28.6%) | |
| 2nd—5th toe | 2 (9.5%) | |
| 1st—5th metatarsal heads | 6 (28.6%) | |
| Midfoot | 1 (4.8%) | |
| Heel | 1 (4.8%) | |
| Previous DFU on right foot: | ||
| Hallux | 5 (23.8%) | |
| 2nd—5th toe | 3 (14.3%) | |
| 1st—5th metatarsal heads | 6 (28.6%) | |
| Midfoot | 2 (9.5%) | |
| Heel | 0 (0.0%) | |
| Claw/hammer toes—Left | 13 (61.9%) | |
| Claw/hammer toes—Right | 10 (47.6%) | |
| Prominent metatarsal heads—Left | 10 (47.6%) | |
| Prominent metatarsal heads—Right | 11 (52.4%) | |
| Charcot foot—Left | 5 (25.0%) | |
| Charcot foot—Right | 2 (9.5%) | |
| Limited Joint Mobility—Left | 11 (52.4%) | |
| Limited Joint Mobility—Right | 11 (52.4%) | |
| Foot Deformity Score—Left | 2.5 ± 1.6 | |
| Foot deformity score—Right | 2.3 ± 1.5 | |
| Callus severity score—Left | 2.6 ± 3.1 | |
| Callus severity score—Right | 1.6 ± 2.3 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abbott, C.A.; Chatwin, K.E.; Rajbhandari, S.M.; John, K.M.; Pabbineedi, S.; Bowling, F.L.; Boulton, A.J.M.; Reeves, N.D. Site-Specific, Critical Threshold Barefoot Peak Plantar Pressure Associated with Diabetic Foot Ulcer History: A Novel Approach to Determine DFU Risk in the Clinical Setting. Medicina 2022, 58, 166. https://doi.org/10.3390/medicina58020166
Abbott CA, Chatwin KE, Rajbhandari SM, John KM, Pabbineedi S, Bowling FL, Boulton AJM, Reeves ND. Site-Specific, Critical Threshold Barefoot Peak Plantar Pressure Associated with Diabetic Foot Ulcer History: A Novel Approach to Determine DFU Risk in the Clinical Setting. Medicina. 2022; 58(2):166. https://doi.org/10.3390/medicina58020166
Chicago/Turabian StyleAbbott, Caroline A., Katie E. Chatwin, Satyan M. Rajbhandari, Kanwal M. John, Sushma Pabbineedi, Frank L. Bowling, Andrew J. M. Boulton, and Neil D. Reeves. 2022. "Site-Specific, Critical Threshold Barefoot Peak Plantar Pressure Associated with Diabetic Foot Ulcer History: A Novel Approach to Determine DFU Risk in the Clinical Setting" Medicina 58, no. 2: 166. https://doi.org/10.3390/medicina58020166
APA StyleAbbott, C. A., Chatwin, K. E., Rajbhandari, S. M., John, K. M., Pabbineedi, S., Bowling, F. L., Boulton, A. J. M., & Reeves, N. D. (2022). Site-Specific, Critical Threshold Barefoot Peak Plantar Pressure Associated with Diabetic Foot Ulcer History: A Novel Approach to Determine DFU Risk in the Clinical Setting. Medicina, 58(2), 166. https://doi.org/10.3390/medicina58020166







