The Effect of Structured Exercise Compared with Education on Neuropathic Signs and Symptoms in People at Risk of Neuropathic Diabetic Foot Ulcers: A Randomized Clinical Trial
Abstract
:1. Introduction
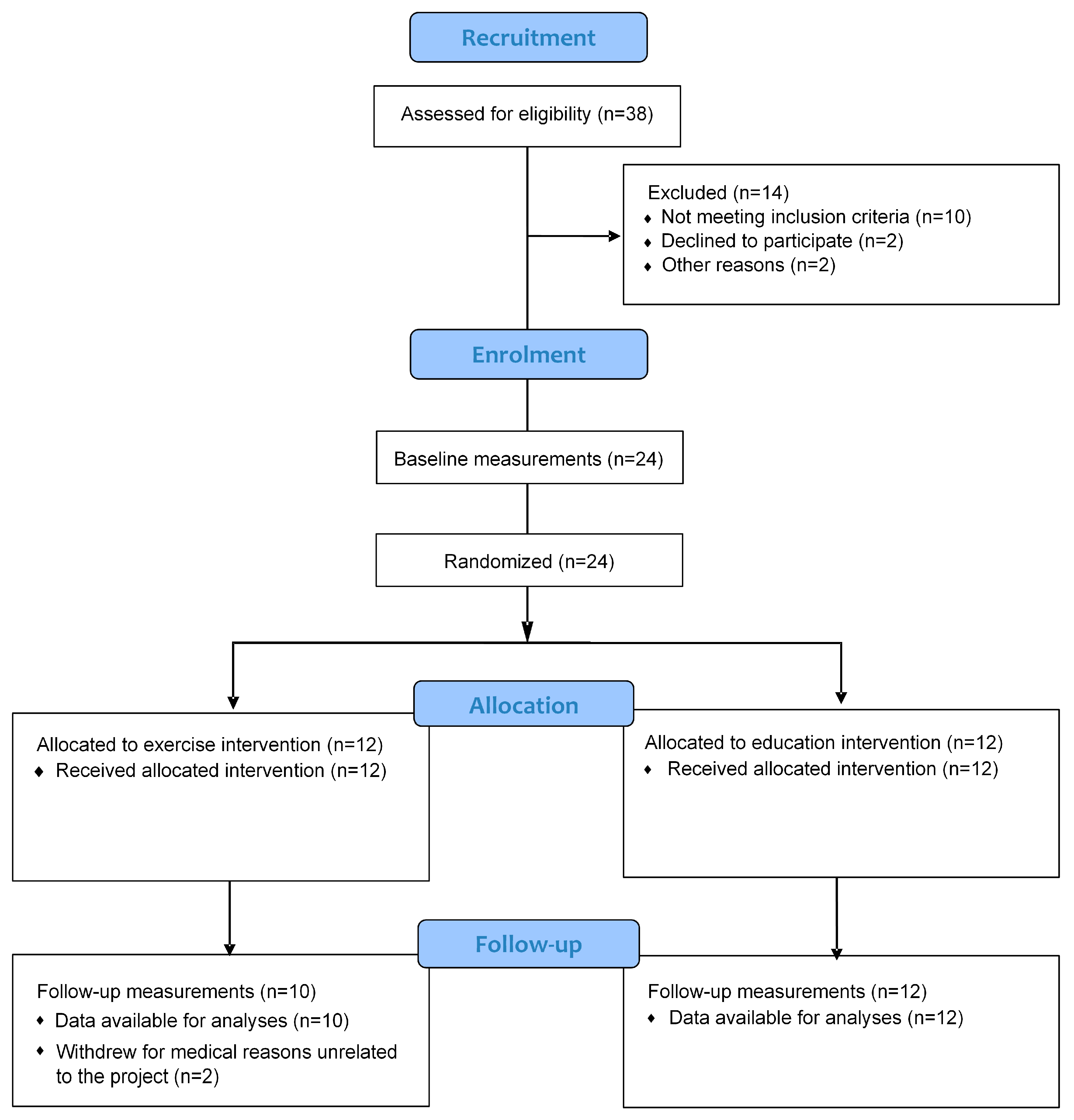
2. Material and Methods
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Boulton, A.J.; Vinik, A.I.; Arezzo, J.C.; Bril, V.; Feldman, E.L.; Freeman, R.; Malik, R.A.; Maser, R.E.; Sosenko, J.M.; Ziegler, D. Diabetic neuropathies: A statement by the American Diabetes Association. Diabetes Care 2005, 28, 956–962. [Google Scholar] [CrossRef] [Green Version]
- Lazzarini, P.; Gurr, J.; Rogers, J.; Schox, A.; Bergin, S. Diabetes foot disease: The Cinderella of Australian diabetes management? J. Foot Ankle Res. 2012, 5, 24. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khunkaew, S.; Fernandez, R.; Sim, J. Health-related quality of life among adults living with diabetic foot ulcers: A meta-analysis. Qual. Life Res. 2019, 28, 1413–1427. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lazzarini, P.A.; van Netten, J.J.; Fitridge, R.A.; Griffiths, I.; Kinnear, E.M.; Malone, M.; Perrin, B.M.; Prentice, J.; Wraight, P.R. Pathway to ending avoidable diabetes-related amputations in Australia. Med. J. Aust. 2018, 209, 288–290. [Google Scholar] [CrossRef] [PubMed]
- Robbins, J.M.; Strauss, G.; Aron, D.; Long, J.; Kuba, J.; Kaplan, Y. Mortality rates and diabetic foot ulcers: Is it time to communicate mortality risk to patients with diabetic foot ulceration? J. Am. Podiatr. Med. Assoc. 2008, 98, 489–493. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Y.; Lazzarini, P.A.; McPhail, S.M.; van Netten, J.J.; Armstrong, D.G.; Pacella, R.E. Global Disability Burdens of Diabetes-Related Lower-Extremity Complications in 1990 and 2016. Diabetes Care 2020, 43, 964–974. [Google Scholar] [CrossRef]
- Kerr, M.; Barron, E.; Chadwick, P.; Evans, T.; Kong, W.M.; Rayman, G.; Sutton-Smith, M.; Todd, G.; Young, B.; Jeffcoate, W.J. The cost of diabetic foot ulcers and amputations to the National Health Service in England. Diabet. Med. 2019, 36, 995–1002. [Google Scholar] [CrossRef]
- American Diabetes Association. 11. Microvascular Complications and Foot Care: Standards of Medical Care in Diabetes-2021. Diabetes Care 2021, 44, S151–S167. [Google Scholar] [CrossRef]
- White, C.M.; Pritchard, J.; Turner-Stokes, L. Exercise for people with peripheral neuropathy. Cochrane Database Syst. Rev. 2004, 4, CD003904. [Google Scholar] [CrossRef]
- Zilliox, L.A.; Russell, J.W. Physical activity and dietary interventions in diabetic neuropathy: A systematic review. Clin. Auton. Res. 2019, 29, 443–455. [Google Scholar] [CrossRef] [PubMed]
- Kluding, P.M.; Bareiss, S.K.; Hastings, M.; Marcus, R.L.; Sinacore, D.R.; Mueller, M.J. Physical training and activity in people with diabetic peripheral neuropathy: Paradigm Shift. Phys. Ther. 2016, 97, 31–43. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Matos, M.; Mendes, R.; Silva, A.B.; Sousa, N. Physical activity and exercise on diabetic foot related outcomes: A systematic review. Diabetes Res. Clin. Pract. 2018, 139, 81–90. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gu, Y.; Dennis, S.M.; Kiernan, M.C.; Harmer, A.R. Aerobic exercise training may improve nerve function in type 2 diabetes and pre-diabetes: A systematic review. Diabetes Metab. Res. Rev. 2019, 35, e3099. [Google Scholar] [CrossRef]
- Vas, P.R.J.; Pafili, K.; Papanas, N. Exercise to improve diabetic peripheral neuropathy: An additional option? Neurophysiol. Clin. 2018, 48, 191–193. [Google Scholar] [CrossRef]
- Schulz, K.F.; Altman, D.G.; Moher, D. CONSORT 2010 Statement: Updated guidelines for reporting parallel group randomised trials. Trials 2010, 11, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Feldman, E.L.; Stevens, M.J.; Thomas, P.K.; Brown, M.B.; Canal, N.; Greene, D.A. A practical two-step quantitative clinical and electrophysiological assessment for the diagnosis and staging of diabetic neuropathy. Diabetes Care 1994, 17, 1281–1289. [Google Scholar] [CrossRef]
- Sigal, R.J.; Kenny, G.P.; Wasserman, D.H.; Castaneda-Sceppa, C.; White, R.D. Physical activity/exercise and type 2 diabetes: A consensus statement from the American Diabetes Association. Diabetes Care 2006, 29, 1433–1438. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- International Physical Activity Questionairre Group. IPAQ_English_Self-Admin_Short. 2013. Available online: https://sites.google.com/site/theipaq/questionnaire_links (accessed on 1 December 2021).
- Stewart, A.; Marfell-Jones, M.; Olds, T.; de Ridder, H. International Standards for Anthropometric Assessment; International Society for the Advancement of Kinanthropometry: Lower Hutt, New Zealand, 2011. [Google Scholar]
- American College of Sports Medicine. ACSM’s Guidelines for Exercise Testing and Prescription, 9th ed.; Lippincott Williams & Wilkins: Baltimore, MD, USA; Philadelphia, PA, USA, 2013. [Google Scholar]
- Hile, E.S.; Brach, J.S.; Perera, S.; Wert, D.M.; VanSwearingen, J.M.; Studenski, S.A. Interpreting the Need for Initial Support to Perform Tandem Stance Tests of Balance. Phys. Ther. 2012, 92, 1316–1328. [Google Scholar] [CrossRef]
- Jessie Jones, C.; Rikli, R.E.; Beam, W.C. A 30-s chair-stand test as a measure of lower body strength in community-residing older adults. Res. Q. Exerc. Sport 1999, 70, 113–119. [Google Scholar] [CrossRef]
- International Diabetes Federation. Diabetes Conversations. 2013. Available online: https://www.idf.org/node/23553 (accessed on 30 July 2013).
- Department of Health and Ageing. Allied Health Group Services under Medicare for Patients with Type 2 Diabetes. 2013. Available online: https://www.health.gov.au/internet/main/publishing.nsf/Content/mbsprimary-ahgs-diabetes.htm (accessed on 1 December 2021).
- Armstrong, D.G.; Lavery, L.A.; Vela, S.A.; Quebedeaux, T.L.; Fleischli, J.G. Choosing a practical screening instrument to identify patients at risk for diabetic foot ulceration. Arch. Intern. Med. 1998, 158, 289–292. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dixit, S.; Maiya, A.; Shastry, B.A. Effects of Aerobic Exercise on Vibration Perception Threshold in Type 2 Diabetic Peripheral Neuropathy Population Using 3-sites Method: Single-blind Randomized Controlled Trial. Altern. Ther. Health Med. 2019, 25, 36–41. [Google Scholar] [PubMed]
- Balducci, S.; Iacobellis, G.; Parisi, L.; Di Biase, N.; Calandriello, E.; Leonetti, F.; Fallucca, F. Exercise training can modify the natural history of diabetic peripheral neuropathy. J. Diabetes Complicat. 2006, 20, 216–223. [Google Scholar] [CrossRef]
- Dixit, S.; Maiya, A.G.; Shastry, B.A. Effect of aerobic exercise on peripheral nerve functions of population with diabetic peripheral neuropathy in type 2 diabetes: A single blind, parallel group randomized controlled trial. J. Diabetes Complicat. 2014, 28, 332–339. [Google Scholar] [CrossRef]
- Kluding, P.M.; Pasnoor, M.; Singh, R.; Jernigan, S.; Farmer, K.; Rucker, J.; Sharma, N.K.; Wright, D.E. The effect of exercise on neuropathic symptoms, nerve function, and cutaneous innervation in people with diabetic peripheral neuropathy. J. Diabetes Complicat. 2012, 26, 424–429. [Google Scholar] [CrossRef] [Green Version]
- Hennigan, A.; O’Callaghan, R.M.; Kelly, Á.M. Neurotrophins and their receptors: Roles in plasticity, neurodegeneration and neuroprotection. Biochem. Soc. Trans. 2007, 35, 424–427. [Google Scholar] [CrossRef]
- Vincent, A.M.; Russell, J.W.; Low, P.; Feldman, E.L. Oxidative stress in the pathogenesis of diabetic neuropathy. Endocr. Rev. 2004, 25, 612–628. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.-S. Effects of exercise training and detraining on cutaneous microvascular function in man: The regulatory role of endothelium-dependent dilation in skin vasculature. Eur. J. Appl. Physiol. 2005, 93, 429–434. [Google Scholar] [CrossRef]
- Ang, E.T.; Wong, P.T.H.; Moochhala, S.; Ng, Y.K. Neuroprotection associated with running: Is it a result of increased endogenous neurotrophic factors? Neuroscience 2003, 118, 335–345. [Google Scholar] [CrossRef]
- Tuomilehto, J.; Schwarz, P.; Lindström, J. Long-term benefits from lifestyle interventions for type 2 diabetes prevention: Time to expand the efforts. Diabetes Care 2011, 34 (Suppl. S2), S210–S214. [Google Scholar] [CrossRef] [Green Version]
- Department of Health. Diabetes Type 2—Allied Health. Available online: https://www1.health.gov.au/internet/main/publishing.nsf/Content/mbsprimary-ahgs-diabetes.htm (accessed on 18 November 2021).
- Perrin, B.M.; Gardner, M.; Kennett, S. The foot-health of people with diabetes in a regional Australian population: A prospective clinical audit. J. Foot Ankle Res. 2012, 5, 6. [Google Scholar] [CrossRef] [Green Version]
- Bergin, S.; Brand, C.; Colman, P.; Campbell, D. The impact of socio-economic disadvantage on rates of hospital separations for diabetes-related foot disease in Victoria, Australia. J. Foot Ankle Res. 2011, 4, 17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Perrin, B.M.; Allen, P.; Gardner, M.J.; Chappell, A.; Phillips, B.; Massey, C.; Skinner, I.; Skinner, T.C. The foot-health of people with diabetes in regional and rural Australia: Baseline results from an observational cohort study. J. Foot Ankle Res. 2019, 12, 56. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Galiero, R.; Ricciardi, D.; Pafundi, P.C.; Todisco, V.; Tedeschi, G.; Cirillo, G.; Sasso, F.C. Whole plantar nerve conduction study: A new tool for early diagnosis of peripheral diabetic neuropathy. Diabetes Res. Clin. Pract. 2021, 176, 108856. [Google Scholar] [CrossRef] [PubMed]

| Variable | Exercise (n = 12) | Education (n = 12) | All (n = 24) | p Value |
|---|---|---|---|---|
| Male sex (%) | 58.3 | 50.0 | 54.2 | 0.68 |
| Age (years) | 70.1 (12.9) | 72.2 (7.1) | 71.1 (10.3) | 0.63 |
| Body Mass Index | 35.6 (14.6) | 31.9 (5.4) | 33.6 (10.5) | 0.46 |
| Resting heart rate (bpm) | 68.8 (11.4) | 73.8 (9.2) | 71.3 (10.5) | 0.25 |
| Resting systolic blood pressure (mm Hg) | 137.8 (14.2) | 132.1 (14.5) | 134.9 (14.4) | 0.35 |
| Resting diastolic blood pressure (mm Hg) | 74.2 (8.4) | 77.4 (10.5) | 75.8 (9.3) | 0.41 |
| Waist circumference (cm) | 107.6 (19.1) | 104.1 (10.6) | 105.8 (15.1) | 0.59 |
| Type 2 diabetes (%) | 91.7 | 100.0 | 95.8 | 0.31 |
| Duration of diabetes (years) | 9.8 (9.5) | 14.2 (9.9) | 12.0 (9.7) | 0.28 |
| Method of Control (%) Oral medication Insulin Diet Combination | 16.7 16.7 41.7 25.0 | 41.7 16.7 25.0 16.7 | 29.2 16.7 33.3 20.8 | 0.58 |
| Adhered to exercise recommendations *(%) | 41.7 | 54.5 | 47.8 | 0.68 |
| Michigan Neuropathy Screening Instrument. signs (/8) Michigan Neuropathy Screening Instrument. symptoms (/13) VPT (/50) | 4.9 (0.9) 5.7 (2.5) 39.4 (6.2) | 4.1 (0.8) 4.5 (2.0) 33.9 (7.9) | 4.5 (0.9) 5.1 (2.3) 36.6 (7.5) | 0.04 ^ 0.23 0.07 |
| Balance: tandem stance (s) | 37.0 (23.6) | 40.6 (24.6) | 38.8 (23.7) | 0.72 |
| Maximum oxygen uptake (mL·kg−1·min−1) | 19.2 (3.2) | 20.3 (4.6) | 19.8 (3.9) | 0.53 |
| Sit to Stand (number 30 s) | 16.4 (11.1) | 14.8 (12.3) | 15.6 (11.5) | 0.75 |
| Variable | Group | Baseline | Post Intervention | Mean Difference | 95% CI |
|---|---|---|---|---|---|
| MNSI Signs (0–8) | Exercise Education | 5.0 (0.9) 4.1 (0.8) | 3.7 (1.6) 3.3 (1.5) | −0.42 | −1.72 to 0.90 |
| Combined | 4.5 (0.9) | 3.5 (1.5) | −1.04 ** | −1.68 to −0.40 | |
| MNSI Symptoms (0–13) | Exercise Education | 5.0 (2.2) 4.5 (2.0) | 3.7 (1.9) 3.6 (1.2) | −0.38 | −1.96 to 1.2 |
| Combined | 4.8 (2.1) | 3.6 (1.6) | −1.11 ** | −1.89 to −0.33 | |
| VPT (0–50) | Exercise Education | 39.7 (6.8) 33.9 (7.9) | 33.3 (13.7) 31.8 (8.5) | −4.22 | −12.09 to 3.65 |
| Combined | 36.8 (7.8) | 32.5 (10.9) | −4.22 * | −8.04 to −0.40 | |
| Maximum oxygen uptake (mL·kg−1·min−1) | Exercise Education | 19.2 (3.4) 19.5 (3.7) | 22.2 (6.1) 21.5 (5.3) | 1.64 | −2.72 to 6.01 |
| Combined | 19.3 (3.5) | 21.5 (5.3) | 2.18 * | 0.18 to 4.18 | |
| Tandem Stance (s) | Exercise Education | 37.2 (23.5) 38.8 (25.0) | 39.8 (22.4) 42.3 (22.0) | −0.94 | −11.44 to 9.57 |
| Combined | 38.5 (23.7) | 40.6 (21.7) | 3.07 | −12.21 to 8.35 | |
| 30 s sit-to-stand (repetitions) | Exercise Education | 15.4 (12.8) 15.1 (10.6) | 22.2 (12.0) 21.2 (11.3) | 0.71 | −8.60 to 10.01 |
| Combined | 15.2 (11.4) | 21.7 (11.3) | 6.44 ** | 1.84 to 11.05 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Perrin, B.M.; Southon, J.; McCaig, J.; Skinner, I.; Skinner, T.C.; Kingsley, M.I.C. The Effect of Structured Exercise Compared with Education on Neuropathic Signs and Symptoms in People at Risk of Neuropathic Diabetic Foot Ulcers: A Randomized Clinical Trial. Medicina 2022, 58, 59. https://doi.org/10.3390/medicina58010059
Perrin BM, Southon J, McCaig J, Skinner I, Skinner TC, Kingsley MIC. The Effect of Structured Exercise Compared with Education on Neuropathic Signs and Symptoms in People at Risk of Neuropathic Diabetic Foot Ulcers: A Randomized Clinical Trial. Medicina. 2022; 58(1):59. https://doi.org/10.3390/medicina58010059
Chicago/Turabian StylePerrin, Byron M., Jessica Southon, Jane McCaig, Isabelle Skinner, Timothy C. Skinner, and Michael I. C. Kingsley. 2022. "The Effect of Structured Exercise Compared with Education on Neuropathic Signs and Symptoms in People at Risk of Neuropathic Diabetic Foot Ulcers: A Randomized Clinical Trial" Medicina 58, no. 1: 59. https://doi.org/10.3390/medicina58010059
APA StylePerrin, B. M., Southon, J., McCaig, J., Skinner, I., Skinner, T. C., & Kingsley, M. I. C. (2022). The Effect of Structured Exercise Compared with Education on Neuropathic Signs and Symptoms in People at Risk of Neuropathic Diabetic Foot Ulcers: A Randomized Clinical Trial. Medicina, 58(1), 59. https://doi.org/10.3390/medicina58010059







