Survival and Chemosensitivity in Advanced High Grade Serous Epithelial Ovarian Cancer Patients with and without a BRCA Germline Mutation: More Evidence for Shifting the Paradigm towards Complete Surgical Cytoreduction
Abstract
1. Introduction
2. Materials and Methods
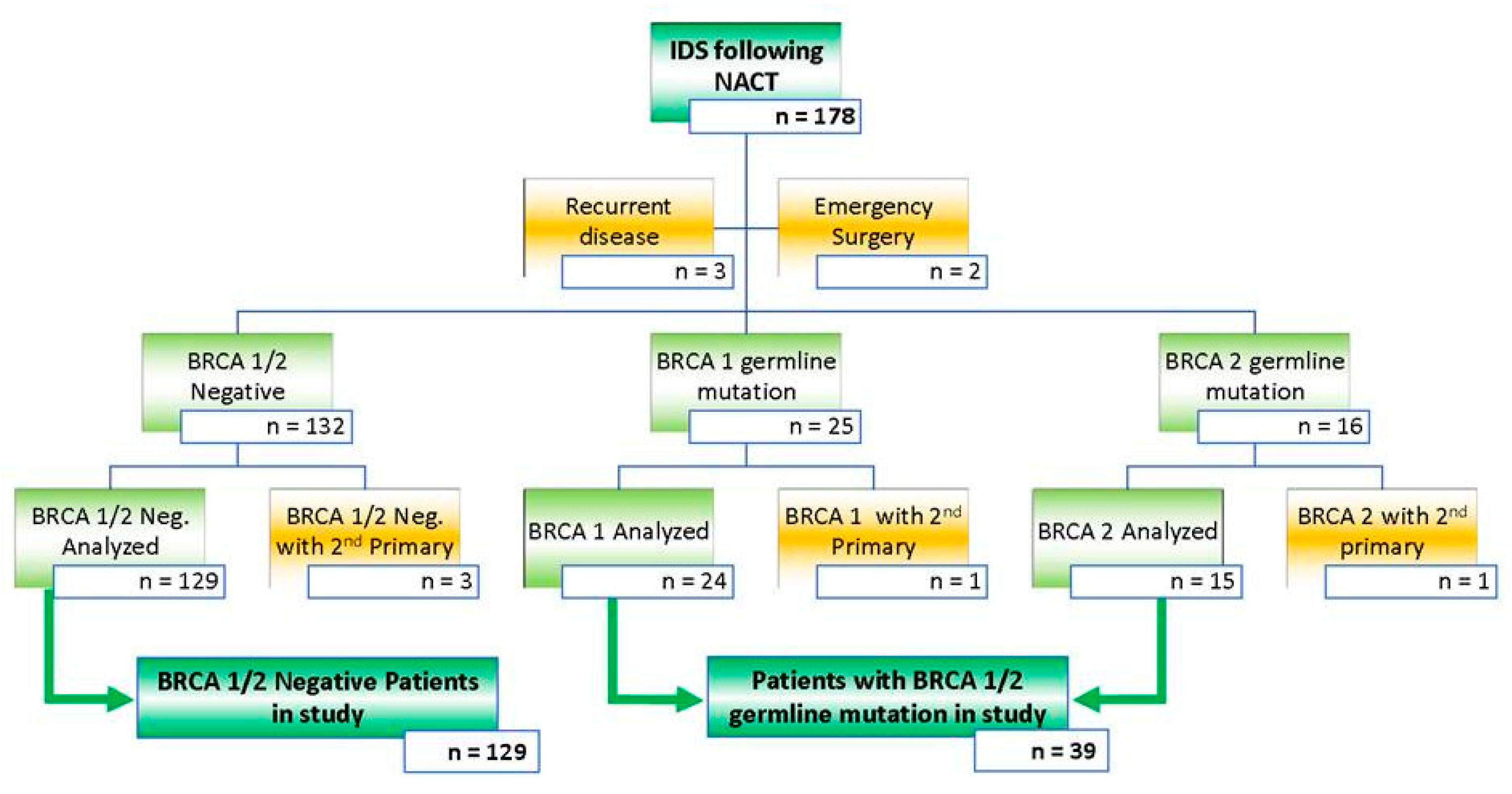
2.1. Selection of Patients, Data Collection, and Study Design
2.2. Workup and Chemotherapy
2.3. Surgical Procedure
2.4. Primary and Secondary Outcome Parameters
2.5. Statistical Analysis
3. Results
3.1. Patient and Tumor Characteristics
3.2. Response to Chemotherapy and Surgical Cytoreduction
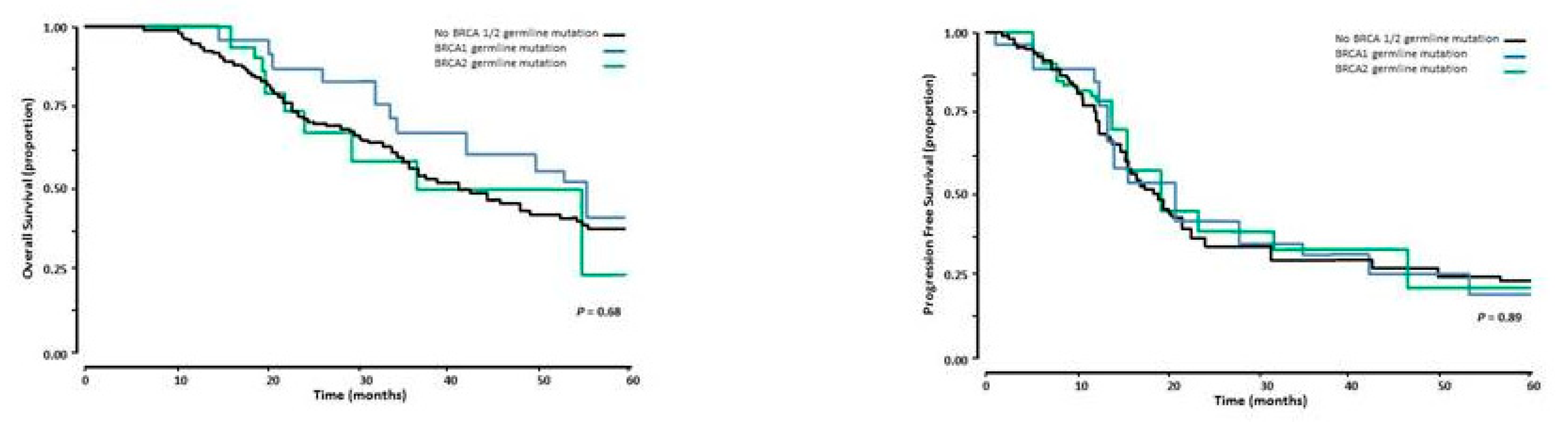
3.3. Overall and Progression-Free Survival
3.4. Neoadjuvant Chemotherapy
3.5. Multivariate Analysis of OS and PFS
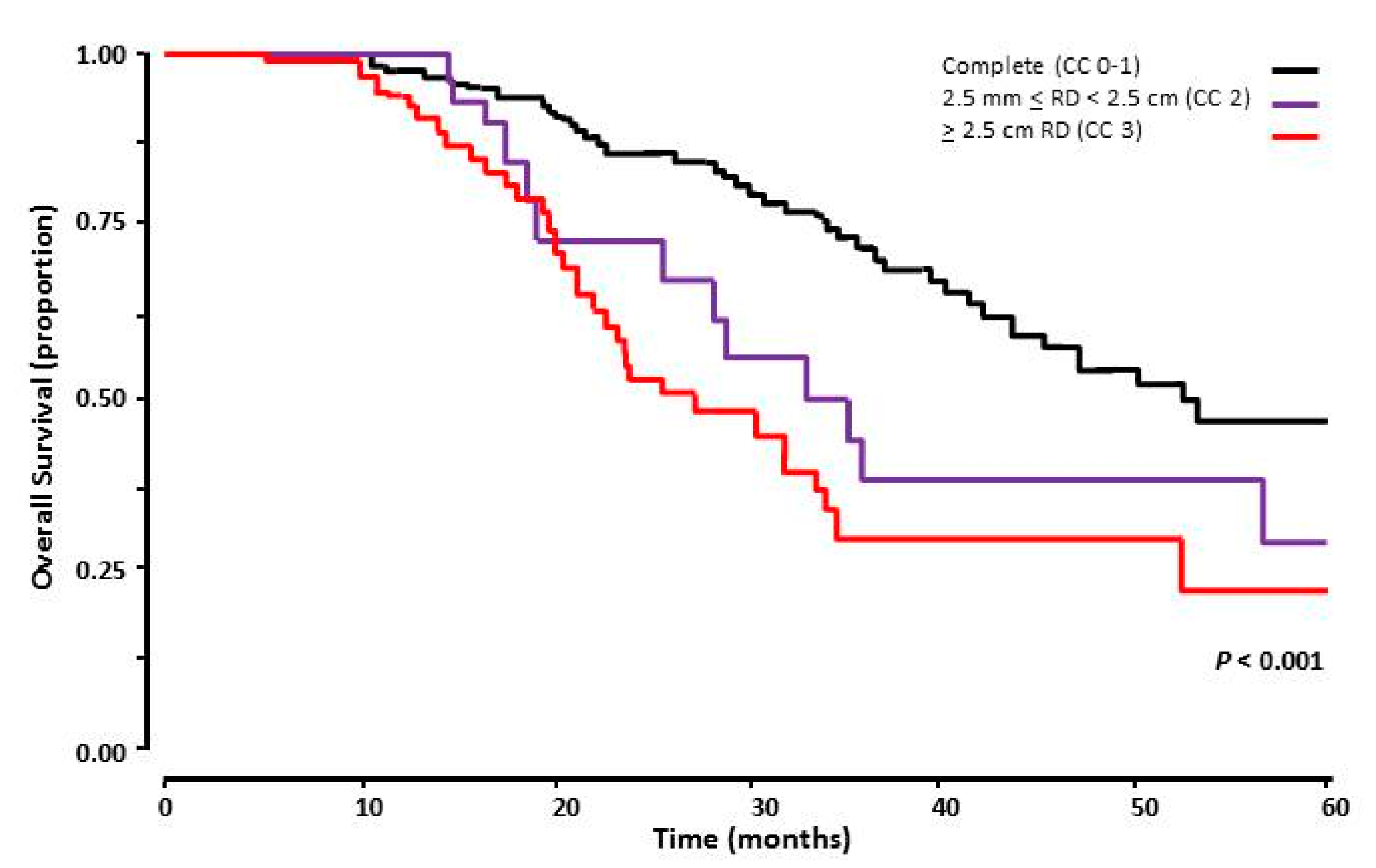
3.6. Surgical Cytoreduction
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Arnold, M.; Rutherford, M.J.; Bardot, A.; Ferlay, J.; Andersson, T.M.; Myklebust, T.Å.; Tervonen, H.; Thursfield, V.; Ransom, D.; Shack, L.; et al. Progress in cancer survival, mortality, and incidence in seven high-income countries 1995-2014 (ICBP SURVMARK-2): A population-based study. Lancet Oncol. 2019, 20, 1493–1505. [Google Scholar] [CrossRef]
- Cancer Research UK. Ovarian Cancer Statistics. 2019. Available online: https://www.cancerresearchuk.org/health-professional/cancer-statistics/statistics-by-cancer-type/ovarian-cancer (accessed on 15 December 2021).
- Moss, E.L.; Evans, T.; Pearmain, P.; Askew, S.; Singh, K.; Chan, K.K.; Ganesan, R.; Hirschowitz, L. Should All Cases of High-Grade Serous Ovarian, Tubal, and Primary Peritoneal Carcinomas Be Reclassified as Tubo-Ovarian Serous Carcinoma? Int. J. Gynecol. Cancer 2015, 25, 1201–1207. [Google Scholar] [CrossRef] [PubMed]
- Girolimetti, G.; Perrone, A.M.; Santini, D.; Barbieri, E.; Guerra, F.; Ferrari, S.; Zamagni, C.; De Iaco, P.; Gasparre, G.; Turchetti, D. BRCA-Associated Ovarian Cancer: From Molecular Genetics to Risk Management. Biomed. Res. Int. 2014, 2014, 787143. [Google Scholar] [CrossRef] [PubMed]
- Gudmundsdottir, K.; Ashworth, A. The roles of BRCA1 and BRCA2 and associated proteins in the maintenance of genomic stability. Oncogene 2006, 25, 5864–5874. [Google Scholar] [CrossRef] [PubMed]
- Vencken, P.M.L.H.; Kriege, M.; Hoogwerf, D.; Beugelink, S.; van der Burg, M.E.L.; Hooning, M.J.; Berns, E.M.; Jager, A.; Collée, M.; Burger, C.W.; et al. Chemosensitivity and outcome of BRCA1- and BRCA2-associated ovarian cancer patients after first-line chemotherapy compared with sporadic ovarian cancer patients. Ann. Oncol. 2011, 22, 1346–1352. [Google Scholar] [CrossRef]
- Bolton, K.L.; Chenevix-Trench, G.; Goh, C.; Sadetzki, S.; Ramus, S.J.; Karlan, B.Y.; Lambrechts, D.; Despierre, E.; Barrowdale, D.; McGuffog, L.; et al. Association between BRCA1 and BRCA2 mutations and survival in women with invasive epithelial ovarian cancer. JAMA 2012, 307, 382–390. [Google Scholar] [CrossRef]
- Makar, A.P.; Tropé, C.G.; Tummers, P.; Denys, H.; Vandecasteele, K. Advanced Ovarian Cancer: Primary or Interval Debulking? Five Categories of Patients in View of the Results of Randomized Trials and Tumor Biology: Primary Debulking Surgery and Interval Debulking Surgery for Advanced Ovarian Cancer. Oncologist 2016, 21, 745–754. [Google Scholar] [CrossRef]
- Vergote, I.; Tropé, C.G.; Amant, F.; Kristensen, G.B.; Ehlen, T.; Johnson, N.; Verheijen, R.H.; van der Burg, M.E.; Lacave, A.J.; Panici, P.B.; et al. Neoadjuvant chemotherapy or primary surgery in stage IIIC or IV ovarian cancer. N. Engl. J. Med. 2010, 363, 943–953. [Google Scholar] [CrossRef]
- Kehoe, S.; Hook, J.; Nankivell, M.; Jayson, G.C.; Kitchener, H.; Lopes, T.; Luesley, D.; Perren, T.; Bannoo, S.; Mascarenhas, M.; et al. Primary chemotherapy versus primary surgery for newly diagnosed advanced ovarian cancer (CHORUS): An open-label, randomised, controlled, non-inferiority trial. Lancet 2015, 386, 249–257. [Google Scholar] [CrossRef]
- Mutch, D.G.; Prat, J. FIGO staging for ovarian, fallopian tube and peritoneal cancer. Gynecol. Oncol. 2014, 133, 401–404. [Google Scholar] [CrossRef]
- Newsham, A.C.; Johnston, C.; Hall, G.; Leahy, M.G.; Smith, A.B.; Vikram, A.; Donnelly, A.M.; Velikova, G.; Selby, P.J.; Fisher, S.E. Development of an advanced database for clinical trials integrated with an electronic patient record system. Comput. Biol. Med. 2011, 41, 575–586. [Google Scholar] [CrossRef]
- Oken, M.M.; Creech, R.H.; Tormey, D.C.; Horton, J.; Davis, T.E.; McFadden, E.T.; Carbone, P.P. Toxicity and response criteria of the Eastern Cooperative Oncology Group. Am. J. Clin. Oncol. 1982, 5, 649–655. [Google Scholar] [CrossRef]
- NICE Clinical Guideline. Familial Breast Cancer: Classification and Care of People at Risk of Familial Breast Cancer and Management of Breast Cancer and Related Risks in People with a Family History of Breast Cancer. National Collaborating Centre for Cancer. 2019. Available online: https://www.nice.org.uk/guidance/cg164/chapter/Recommendations. (accessed on 15 December 2021).
- Jazaeri, A.A. Molecular profiles of hereditary epithelial ovarian cancers and their implications for the biology of this disease. Mol. Oncol. 2009, 3, 151–156. [Google Scholar] [CrossRef]
- Perren, T.J.; Swart, A.M.; Pfisterer, J.; Ledermann, J.A.; Pujade-Lauraine, E.; Kristensen, G.; Carey, M.S.; Beale, P.; Cervantes, A.; Kurzeder, C.; et al. A phase 3 trial of bevacizumab in ovarian cancer. N. Engl. J. Med. 2011, 365, 2484–2496. [Google Scholar] [CrossRef]
- Eisenhauer, E.A.; Therasse, P.; Bogaerts, J.; Schwartz, L.H.; Sargent, D.; Ford, R.; Dancey, J.; Arbuck, S.; Gwyther, S.; Mooney, M.; et al. New response evaluation criteria in solid tumours: Revised RECIST guideline (version 1.1). Eur. J. Cancer. 2009, 45, 228–247. [Google Scholar] [CrossRef]
- Hamilton, C.A.; Miller, A.; Miller, C.; Krivak, T.C.; Farley, J.H.; Chernofsky, M.R.; Stany, M.P.; Rose, G.S.; Markman, M.; Ozols, R.F.; et al. The impact of disease distribution on survival in patients with stage III epithelial ovarian cancer cytoreduced to microscopic residual: A Gynecologic Oncology Group study. Gynecol. Oncol. 2011, 122, 521–526. [Google Scholar] [CrossRef]
- Böhm, S.; Faruqi, A.; Said, I.; Lockley, M.; Brockbank, E.; Jeyarajah, A.; Fitzpatrick, A.; Ennis, D.; Dowe, T.; Santos, J.L.; et al. Chemotherapy Response Score: Development and Validation of a System to Quantify Histopathologic Response to Neoadjuvant Chemotherapy in Tubo-Ovarian High-Grade Serous Carcinoma. J. Clin. Oncol. 2015, 33, 2457–2463. [Google Scholar] [CrossRef]
- Aletti, G.D.; Santillan, A.; Eisenhauer, E.L.; Hu, J.; Aletti, G.; Podratz, K.C.; Bristow, R.E.; Chi, D.S.; Cliby, W.A. A new frontier for quality of care in gynecologic oncology surgery: Multi-institutional assessment of short-term outcomes for ovarian cancer using a risk-adjusted model. Gynecol. Oncol. 2007, 107, 99–106. [Google Scholar] [CrossRef]
- Sugarbaker, P.H. Cytoreductive surgery and peri-operative intraperitoneal chemotherapy as a curative approach to pseudomyxoma peritonei syndrome. Eur. J. Surg. Oncol. 2001, 27, 239–243. [Google Scholar] [CrossRef]
- Rong, Y.; Li, L. Early clearance of serum HE4 and CA125 in predicting platinum sensitivity and prognosis in epithelial ovarian cancer. J. Ovarian Res. 2021, 14, 2. [Google Scholar] [CrossRef]
- Yang, Z.J.; Zhao, B.B.; Li, L. The significance of the change pattern of serum CA125 level for judging prognosis and diagnosing recurrences of epithelial ovarian cancer. J. Ovarian Res. 2016, 9, 57. [Google Scholar] [CrossRef]
- Fader, A.N.; Java, J.; Krivak, T.C.; Bristow, R.E.; Tergas, A.I.; Bookman, M.A.; Armstrong, D.K.; Tanner, E.J.; Gershenson, D.M. The prognostic significance of pre- and post-treatment CA-125 in grade 1 serous ovarian carcinoma: A gynecologic Oncology Group study. Gynecol. Oncol. 2014, 132, 560–565. [Google Scholar] [CrossRef] [PubMed]
- Le, T.; Hopkins, L.; Faught, W.; Fung-Kee-Fung, M. The lack of significance of CA125 response in epithelial ovarian cancer patients treated with neoadjuvant chemotherapy and delayed primary surgical debulking. Gynecol. Oncol. 2007, 105, 712–715. [Google Scholar] [CrossRef] [PubMed]
- Ramspott, J.P.; Baert, T.; MacKintosh, M.L.; Traut, A.; Ataseven, B.; Bommert, M.; Heitz, F.; Plett, H.; Schneider, S.; Waltering, K.U.; et al. Response evaluation after neoadjuvant therapy: Evaluation of chemotherapy response score and serological and/or radiological assessment of response in ovarian cancer patients. Arch. Gynecol. Obstet. 2021, 304, 1021–1032. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.J.; Kim, H.S.; Rim, J.H.; Lee, J.Y.; Nam, E.J.; Kim, S.W.; Kim, S.; Kim, Y.T. Germline BRCA, chemotherapy response scores, and survival in the neoadjuvant treatment of ovarian cancer. BMC Cancer 2020, 20, 185. [Google Scholar] [CrossRef]
- Whitley, N.; Brenner, D.; Francis, A.; Kwon, T.; Villasanta, U.; Aisner, J.; Wiernik, P.; Whitley, J. Use of the computed tomographic whole body scanner to stage and follow patients with advanced ovarian carcinoma. Investig. Radiol. 1981, 16, 479–486. [Google Scholar] [CrossRef]
- Morgan, R.D.; McNeish, I.A.; Cook, A.D.; James, E.C.; Lord, R.; Dark, G.; Glasspool, R.M.; Krell, J.; Parkinson, C.; Poole, C.J.; et al. Objective responses to first-line neoadjuvant carboplatin-paclitaxel regimens for ovarian, fallopian tube, or primary peritoneal carcinoma (ICON8): Post-hoc exploratory analysis of a randomised, phase 3 trial. Lancet Oncol. 2021, 22, 277–288. [Google Scholar] [CrossRef]
- Hamilton, C.A.; Miller, A.; Casablanca, Y.; Horowitz, N.S.; Rungruang, B.; Krivak, T.C.; Richard, S.D.; Rodriguez, N.; Birrer, M.J.; Backes, F.J.; et al. Clinicopathologic characteristics associated with long-term survival in advanced epithelial ovarian cancer: An NRG Oncology/Gynecologic Oncology Group ancillary data study. Gynecol. Oncol. 2018, 148, 275–280. [Google Scholar] [CrossRef]
- Hyman, D.M.; Long, K.C.; Tanner, E.J.; Grisham, R.N.; Arnold, A.G.; Bhatia, J.; Phillips, M.F.; Spriggs, D.R.; Soslow, R.A.; Kauff, N.D.; et al. Outcomes of primary surgical cytoreduction in patients with BRCA-associated high-grade serous ovarian carcinoma. Gynecol. Oncol. 2012, 126, 224–228. [Google Scholar] [CrossRef]
- Gallagher, D.J.; Konner, J.A.; Bell-McGuinn, K.M.; Bhatia, J.; Sabbatini, P.; Aghajanian, C.A.; Offit, K.; Barakat, R.R.; Spriggs, D.R.; Kauff, N.D. Survival in epithelial ovarian cancer: A multivariate analysis incorporating BRCA mutation status and platinum sensitivity. Ann. Oncol. 2011, 22, 1127–1132. [Google Scholar] [CrossRef]
- Zweemer, R.P.; Verheijen, R.H.; Coebergh, J.W.; Jacobs, I.J.; van Diest, P.J.; Gille, J.J.; Skates, S.; Menko, F.H.; Ten Kate, L.P.; Kenemans, P. Survival analysis in familial ovarian cancer, a case control study. Eur. J. Obstet. Gynecol. Reprod. Biol. 2001, 98, 219–223. [Google Scholar] [CrossRef]
- McLaughlin, J.R.; Rosen, B.; Moody, J.; Pal, T.; Fan, I.; Shaw, P.A.; Risch, H.A.; Sellers, T.A.; Sun, P.; Narod, S.A. Long-term ovarian cancer survival associated with mutation in BRCA1 or BRCA2. J. Natl. Cancer Inst. 2013, 105, 141–148. [Google Scholar] [CrossRef] [PubMed]
- Brozek, I.; Ochman, K.; Debniak, J.; Morzuch, L.; Ratajska, M.; Stepnowska, M.; Stukan, M.; Emerich, J.; Limon, J. High frequency of BRCA1/2 germline mutations in consecutive ovarian cancer patients in Poland. Gynecol. Oncol. 2008, 108, 433–437. [Google Scholar] [CrossRef] [PubMed]
- Lyons, Y.A.; Reyes, H.D.; McDonald, M.E.; Newtson, A.; Devor, E.; Bender, D.P.; Goodheart, M.J.; Gonzalez Bosquet, J. Interval debulking surgery is not worth the wait: A National Cancer Database study comparing primary cytoreductive surgery versus neoadjuvant chemotherapy. Int J. Gynecol. Cancer 2020, 30, 845–852. [Google Scholar] [CrossRef] [PubMed]
- Fagotti, A.; Ferrandina, M.G.; Vizzielli, G.; Pasciuto, T.; Fanfani, F.; Gallotta, V.; Margariti, P.A.; Chiantera, V.; Costantini, B.; Gueli Alletti, S.; et al. Randomized trial of primary debulking surgery versus neoadjuvant chemotherapy for advanced epithelial ovarian cancer (SCORPION-NCT01461850). Int. J. Gynecol. Cancer 2020, 30, 1657–1664. [Google Scholar] [CrossRef] [PubMed]
- Johnson, R.L.; Laios, A.; Jackson, D.; Nugent, D.; Orsi, N.M.; Theophilou, G.; Thanagavelu, A.; De Jong, D. The uncertain benefit of adjuvant chemotherapy in advanced low-grade serous ovarian cancer and the pivotal role of surgical cytoreduction. J. Clin. Med. 2021, 10, 5927. [Google Scholar] [CrossRef]
- De Jong, D.; Eijkemans, M.J.; Lie Fong, S.; Gerestein, C.G.; Kooi, G.S.; Baalbergen, A.; Van der Burg, M.E.; Burger, C.W.; Ansink, A.C. Preoperative predictors for residual tumor after surgery in patients with ovarian carcinoma. Oncology 2007, 72, 293–301. [Google Scholar] [CrossRef]
- Laios, A.; Gryparis, A.; de Jong, D.; Hutson, R.; Theophilou, G.; Leach, C. Predicting complete cytoreduction for advanced ovarian cancer patients using nearest-neighbor models. J. Ovarian Res. 2020, 13, 117. [Google Scholar] [CrossRef]
- Glajzer, J.; Castillo-Tong, D.C.; Richter, R.; Vergote, I.; Kulbe, H.; Vanderstichele, A.; Ruscito, I.; Trillsch, F.; Mustea, A.; Kreuzinger, C.; et al. Impact of BRCA Mutation Status on Tumor Dissemination Pattern, Surgical Outcome and Patient Survival in Primary and Recurrent High-Grade Serous Ovarian Cancer: A Multicenter Retrospective Study by the Ovarian Cancer Therapy-Innovative Models Prolong Survival (OCTIPS) Consortium. Ann. Surg. Oncol. 2022. [CrossRef]
- González-Martín, A.; Pothuri, B.; Vergote, I.; DePont Christensen, R.; Graybill, W.; Mirza, M.R.; Mc Cormick, C.; Lorusso, D.; Hoskins, P.; Freyer, G.; et al. Niraparib in Patients with Newly Diagnosed Advanced Ovarian Cancer. N. Engl. J. Med. 2019, 38, 2391–2402. [Google Scholar] [CrossRef]
| BRCA 1/2 Mutation | No BRCA 1/2 Mutation | Total | p-Value | |||
|---|---|---|---|---|---|---|
| Patients | n = 39 | 23.2% | n = 129 | 76.8% | n = 168 | |
| Age (yrs) | 58.0 ± 7.79 | 66.3 ± 8.79 | 64.4 ± 9.79 | 0.00001 | ||
| WHO PS | 0.815 | |||||
| PS 0 | 18 | 46.2% | 57 | 44.2% | 75 | |
| PS 1 | 13 | 33.3% | 47 | 36.4% | 60 | |
| PS 2 | 7 | 17.9% | 18 | 14.0% | 25 | |
| PS 3 | 1 | 2.6% | 7 | 5.4% | 8 | |
| CA125 (U/mL) * | 1245 (96–31,600) | 791 (23–17,900) | 1019 (23–31,600) | 0.247 | ||
| Primary Tumor | 0.722 | |||||
| Ovary | 21 | 53.8% | 60 | 46.5% | 81 | |
| Peritoneum | 14 | 35.9% | 53 | 41.1% | 67 | |
| Fallopian Tube | 4 | 10.3% | 16 | 12.4% | 20 | |
| FIGO Stage | 0.584 | |||||
| III A-B | 1 | 2.6% | 5 | 3.9% | 6 | |
| III C | 24 | 61.5% | 82 | 63.6% | 106 | |
| IV A | 7 | 17.9% | 12 | 9.3% | 19 | |
| IV B | 7 | 17.9% | 30 | 23.3% | 37 | |
| Tumor Differentiation | 1.000 | |||||
| Well | 0 | 0% | 0 | 0% | 0 | |
| Moderate | 0 | 0% | 1 | 0.8% | 1 | |
| Poor | 39 | 100% | 128 | 99.2% | 167 | |
| Agent NACT | 0.829 | |||||
| CP | 2 | 5.1% | 7 | 5.4% | 9 | |
| CPT | 37 | 94.9% | 121 | 94.6% | 158 | |
| BRCA 1/2 Mutation | No BRCA 1/2 Mutation | Total | p-Value | |||
|---|---|---|---|---|---|---|
| Patients | n = 39 | 23.2% | n = 129 | 76.8% | n = 168 | |
| ΔCA125 (U/mL) * | 1111 (27–30715) | 641 (2–15897) | 801 (2–30,715) | 0.981 | ||
| RECIST after NACT | 0.677 | |||||
| Complete | 0 | 0.0% | 3 | 2.3% | 3 | |
| Partial | 35 | 89.7% | 108 | 83.7% | 143 | |
| Stable disease | 3 | 7.7% | 16 | 12.4% | 19 | |
| Prog. Disease | 1 | 2.6% | 2 | 1.6% | 3 | |
| Preoperative Extent of Disease | 0.478 | |||||
| MD | 1 | 2.6% | 8 | 6.2% | 9 | |
| APD | 33 | 84.6% | 98 | 76.0% | 131 | |
| UAD | 5 | 12.8% | 23 | 12.4% | 28 | |
| Histological response | 0.593 | |||||
| CRS 1 | 17 | 43.6% | 48 | 37.2% | 65 | |
| CRS 2 | 18 | 46.1% | 60 | 46.5% | 78 | |
| CRS 3 | 4 | 10.3% | 21 | 16.3% | 25 | |
| SCS Group | 0.836 | |||||
| Low | 29 | 74.4% | 98 | 76.0% | 127 | |
| Intermediate | 8 | 20.5% | 27 | 20.9% | 35 | |
| High | 2 | 5.1% | 4 | 3.1% | 6 | |
| Residual Disease (RD) | 0.226 | |||||
| 0–2.5 mm (CC 0–1) | 22 | 56.4% | 76 | 58.9% | 98 | |
| ≥2.5 mm (CC 2) | 7 | 18.0% | 11 | 8.5% | 18 | |
| ≥2.5 cm (CC 3) | 10 | 25.6% | 42 | 32.6% | 52 | |
| Multivariate Analysis OS | Multivariate Analysis PFS | |||||
|---|---|---|---|---|---|---|
| Covariates | HR | p | 95% CI | HR | p | 95% CI |
| Performance Score | ||||||
| PS 0 | 1.000 | <0.001 | 1.00 | <0.001 | ||
| PS 1 | 0.134 | <0.001 | 0.051–0.353 | 0.205 | <0.001 | 0.082–0.514 |
| PS 2 | 0.490 | 0.019 | 0.209–0.929 | 0.218 | 0.004 | 0.077–0.618 |
| Result of Cytoreduction | ||||||
| Completete (CC 0–1) | 1.000 | 0.017 | 1.00 | 0.540 | ||
| ≥2.5 mm RD (CC 2) | 0.446 | 0.175 | 0.139–1.430 | 0.710 | 0.537 | 0.239–2.107 |
| ≥2.5 cm RD (CC 3) | 0.701 | 0.428 | 0.292–1.685 | 0.853 | 1.087 | 0.449–2.636 |
| Chemotherapy Response Score | ||||||
| CRS 1 | 1.000 | <0.001 | 1.00 | <0.001 | ||
| CRS 2 | 7.496 | <0.001 | 2.523–22.27 | 3.898 | <0.001 | 1.873–8.112 |
| CRS 3 | 4.069 | 0.011 | 1.388–11.93 | 2.000 | 0.003 | 1.433–5.862 |
| Preoperative Extent of Disease | ||||||
| MD | 1.00 | 0.17 | 1.00 | 0.105 | ||
| APD | 0.235 | 0.082 | 0.046–1.201 | 0.902 | 0.845 | 0.320–2.542 |
| UAD | 1.750 | 0.132 | 0.846–3.622 | 1.713 | 0.084 | 0.931–3.150 |
| Radiological Response | ||||||
| Complete (RECIST) | 1.00 | 0.762 | 1.00 | 0.487 | ||
| Partial/Stable (RECIST) | 4.271 | 0.254 | 0.352–51.86 | 0.503 | 0.533 | 0.058–4.367 |
| Progressive (RECIST) | 0.176 | 0.124 | 0.019–1.608 | 0.444 | 0.197 | 0.129–1.526 |
| Surgical Complexity Score | ||||||
| Low Complexity | 1.00 | 0.542 | 1.00 | 0.893 | ||
| Intermediate Complexity | 0.886 | 0.847 | 0.258–3.038 | 1.240 | 0.646 | 0.494–3.112 |
| High Complexity | 1.240 | 0.746 | 0.338–4.544 | 1.191 | 0.727 | 0.447–3.173 |
| ΔCA125 (U/mL) | 1.107 | 0.695 | 0.666–1.839 | 1.179 | 0.404 | 0.800–1.739 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
De Jong, D.; Otify, M.; Chen, I.; Jackson, D.; Jayasinghe, K.; Nugent, D.; Thangavelu, A.; Theophilou, G.; Laios, A. Survival and Chemosensitivity in Advanced High Grade Serous Epithelial Ovarian Cancer Patients with and without a BRCA Germline Mutation: More Evidence for Shifting the Paradigm towards Complete Surgical Cytoreduction. Medicina 2022, 58, 1611. https://doi.org/10.3390/medicina58111611
De Jong D, Otify M, Chen I, Jackson D, Jayasinghe K, Nugent D, Thangavelu A, Theophilou G, Laios A. Survival and Chemosensitivity in Advanced High Grade Serous Epithelial Ovarian Cancer Patients with and without a BRCA Germline Mutation: More Evidence for Shifting the Paradigm towards Complete Surgical Cytoreduction. Medicina. 2022; 58(11):1611. https://doi.org/10.3390/medicina58111611
Chicago/Turabian StyleDe Jong, Diederick, Mohamed Otify, Inga Chen, David Jackson, Kelum Jayasinghe, David Nugent, Amudha Thangavelu, Georgios Theophilou, and Alexandros Laios. 2022. "Survival and Chemosensitivity in Advanced High Grade Serous Epithelial Ovarian Cancer Patients with and without a BRCA Germline Mutation: More Evidence for Shifting the Paradigm towards Complete Surgical Cytoreduction" Medicina 58, no. 11: 1611. https://doi.org/10.3390/medicina58111611
APA StyleDe Jong, D., Otify, M., Chen, I., Jackson, D., Jayasinghe, K., Nugent, D., Thangavelu, A., Theophilou, G., & Laios, A. (2022). Survival and Chemosensitivity in Advanced High Grade Serous Epithelial Ovarian Cancer Patients with and without a BRCA Germline Mutation: More Evidence for Shifting the Paradigm towards Complete Surgical Cytoreduction. Medicina, 58(11), 1611. https://doi.org/10.3390/medicina58111611







