Intravesical Therapy for Non-Muscle-Invasive Bladder Cancer: What Is the Real Impact of Squamous Cell Carcinoma Variant on Oncological Outcomes?
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design and Patient Population
2.2. Adjuvant Intravesical Therapy and Follow-Up
2.3. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef]
- Abdou Hassan, W.; Shalaby, E.; Abo-Hashesh, M.; Ibrahim Ali, R. Evaluation of the Expression of HER2 and c-KIT Proteins as Prognostic Markers in Superficial Bladder Urothelial Carcinoma. Res. Rep. Urol. 2021, 13, 197–206. [Google Scholar] [CrossRef]
- Peyrottes, A.; Ouzaid, I.; Califano, G.; Hermieu, J.-F.; Xylinas, E. Neoadjuvant Immunotherapy for Muscle-Invasive Bladder Cancer. Medicina 2021, 57, 769. [Google Scholar] [CrossRef]
- Babjuk, M.; Burger, M.; Compérat, E.; Gontero, P.; Liedberg, F.; Masson-Lecomte, A.; Mostafid, A.H.; Palou, J.; van Rhijn, B.W.G.; Rouprêt, M.; et al. EAU Guidelines 2021 on Non-Muscle-Invasive Bladder Cancer. Available online: www.uroweb.org (accessed on 15 August 2021).
- Sawazaki, H.; Arai, Y.; Ito, Y.; Sato, K.; Tsuda, H.; Yamaga, T.; Sakurai, H. Expression of L-Type Amino Acid Transporter 1 is a Predictive Biomarker of Intravesical Recurrence in Patients with Non-Muscle Invasive Bladder Cancer. Res. Rep. Urol. 2021, 13, 603–611. [Google Scholar] [CrossRef]
- Hansel, D.E.; Amin, M.B.; Comperat, E.; Cote, R.J.; Knüchel, R.; Montironi, R.; Reuter, V.E.; Soloway, M.S.; Umar, S.A.; Van der Kwast, T. A Contemporary Update on Pathology Standards for Bladder Cancer: Transurethral Resection and Radical Cystectomy Specimens. Eur. Urol. 2012, 63, 321–332. [Google Scholar] [CrossRef]
- Mantica, G.; Tappero, S.; Parodi, S.; Piol, N.; Spina, B.; Malinaric, R.; Balzarini, F.; Borghesi, M.; Van Der Merwe, A.; Suardi, N.; et al. Bladder cancer histological variants: Which parameters could predict the concordance between transurethral resection of bladder tumor and radical cystectomy specimens? Cent. Eur. J. Urol. 2021, 74, 355–361. [Google Scholar]
- Mantica, G.; Simonato, A.; Du Plessis, D.E.; Maffezzini, M.; De Rose, A.F.; van der Merwe, A.; Terrone, C. The pathologist’s role in the detection of rare variants of bladder cancer and analysis of the impact on incidence and type detection. Minerva Urol. Nefrol. 2018, 70, 594–597. [Google Scholar] [CrossRef]
- Baumeister, P.; Zamboni, S.; Mattei, A.; Antonelli, A.; Simeone, C.; Mordasini, L.; DiBona, C.; Moschini, M. Histological variants in non-muscle invasive bladder cancer. Transl. Androl. Urol. 2019, 8, 34–38. [Google Scholar] [CrossRef]
- Goutas, D.; Tzortzis, A.; Gakiopoulou, H.; Vlachodimitropoulos, D.; Giannopoulou, I.; Lazaris, A.C. Contemporary Molecular Classification of Urinary Bladder Cancer. In Vivo 2021, 35, 75–80. [Google Scholar] [CrossRef]
- Seisen, T.; Compérat, E.; Léon, P.; Roupret, M. Impact of histological variants on the outcomes of nonmuscle invasive bladder cancer after transurethral resection. Curr. Opin. Urol. 2014, 24, 524–531. [Google Scholar] [CrossRef]
- Suh, J.; Moon, K.C.; Jung, J.H.; Lee, J.; Song, W.H.; Kang, Y.J.; Jeong, C.W.; Kwak, C.; Kim, H.H.; Ku, J.H. BCG instillation versus radical cystectomy for high-risk NMIBC with squamous/glandular histologic variants. Sci. Rep. 2019, 9, 15268. [Google Scholar] [CrossRef] [Green Version]
- Shapur, N.K.; Katz, R.; Pode, D.; Shapiro, A.; Yutkin, V.; Pizov, G.; Appelbaum, L.; Zorn, K.C.; Duvdevani, M.; Landau, E.H.; et al. Is radical cystectomy mandatory in every patient with variant histology of bladder cancer. Rare Tumors 2011, 3, 67–70. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Miller, J.S.; Epstein, J.I. Noninvasive urothelial carcinoma of the bladder with glandular differentiation: Report of 24 cases. Am. J. Surg. Pathol. 2009, 33, 1241–1248. [Google Scholar] [CrossRef]
- Yorozuya, W.; Nishiyama, N.; Shindo, T.; Kyoda, Y.; Itoh, N.; Sugita, S.; Hasegawa, T.; Masumori, N. Bacillus Calmette-Guérin may have clinical benefit for glandular or squamous differentiation in non-muscle invasive bladder cancer patients: Retrospective multicenter study. Jpn. J. Clin. Oncol. 2018, 48, 661–666. [Google Scholar] [CrossRef]
- Gofrit, O.N.; Yutkin, V.; Shapiro, A.; Pizov, G.; Zorn, K.C.; Hidas, G.; Gielchinsky, I.; Duvdevani, M.; Landau, E.H.; Pode, D. The Response of Variant Histology Bladder Cancer to Intravesical Immunotherapy Compared to Conventional Cancer. Front. Oncol. 2016, 6, 43. [Google Scholar] [CrossRef] [Green Version]
- Lamm, D.L.; Blumenstein, B.A.; Crissman, J.D.; Montie, J.E.; Gottesman, J.E.; Lowe, B.A.; Sarosdy, M.F.; Bohl, R.D.; Grossman, H.B.; Beck, T.M.; et al. Maintenance Bacillus Calmette-Guerin immunotherapy for recurrent TA, T1 and carcinoma in situ transitional cell carcinoma of the bladder: A randomized Southwest Oncology Group Study. J. Urol. 2000, 163, 1124–1129. [Google Scholar] [CrossRef]
- Humphrey, P.A.; Moch, H.; Cubilla, A.L.; Ulbright, T.M.; Reuter, V.E. The 2016 WHO Classification of Tumours of the Urinary System and Male Genital Organs-Part B: Prostate and Bladder Tumours. Eur. Urol. 2016, 70, 106–119. [Google Scholar] [CrossRef] [Green Version]
- American Joint Committee on Cancer (AJCC) Staging Manual, 8th ed; Available online: www.cancerstaging.org (accessed on 15 August 2021).
- Alvarez-Maestro, M.; Chierigo, F.; Mantica, G.; Quesada-Olarte, J.M.; Carrion, D.M.; Gomez-Rivas, J.; Pinto-Marin, A.; Bazan, A.A.; Martinez-Piñeiro, L. The effect of neoadjuvant chemotherapy among patients undergoing radical cystectomy for variant histology bladder cancer: A systematic review. Arab. J. Urol. 2021, 1–13. [Google Scholar] [CrossRef]
- Sánchez-Rodríguez, C.; Cruces, K.P.; Ayora, J.R.; Martín-Sanz, E.; Sanz-Fernandez, R. BCG immune activation reduces growth and angiogenesis in an in vitro model of head and neck squamous cell carcinoma. Vaccine 2017, 35, 6395–6403. [Google Scholar] [CrossRef]
- Medina, J.E. The Controversial Role of BCG in the Treatment of Squamous Cell Carcinoma of the Head and Neck. Arch. Otolaryngol. 1983, 109, 543. [Google Scholar] [CrossRef]
- Raez, L.E.; Fein, S.; Podack, E.R. Lung cancer immunotherapy. Clin. Med. Res. 2005, 3, 221–228. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Podarala, V.; Lakshmi, M.P.; Venkata, S.K.R.; Devalam, R.P. Efficacy of BCG vaccine and Mitomycin C for the treatment of ocular squamous cell carcinoma in bovines. Res. Veter. Sci. 2020, 133, 48–52. [Google Scholar] [CrossRef] [PubMed]
- Shokeir, A. Squamous cell carcinoma of the bladder: Pathology, diagnosis and treatment. BJU Int. 2004, 93, 216–220. [Google Scholar] [CrossRef] [PubMed]

| Characteristic | Overall, n = 353 | TCC, n = 321 | TCC + SCC, n = 32 | p-Value |
|---|---|---|---|---|
| Age | 72 (65, 78) | 72 (65, 78) | 72 (66, 79) | 0.72 |
| Smoking, n (%) | 0.10 | |||
| No | 42 (12%) | 40 (12%) | 2 (6.2%) | |
| Yes | 196 (56%) | 182 (57%) | 14 (44%) | |
| Ex | 115 (33%) | 99 (31%) | 16 (50%) | |
| Single vs. Multiple, n (%) | 0.60 | |||
| Single | 203 (58%) | 186 (58%) | 17 (53%) | |
| Multiple | 150 (42%) | 135 (42%) | 15 (47%) | |
| Dimensions, n (%) | 0.40 | |||
| <3 cm | 196 (56%) | 176 (55%) | 20 (62%) | |
| ≥3 cm | 157 (44%) | 145 (45%) | 12 (38%) | |
| Histology, n (%) | 0.57 | |||
| pTa | 186 (53%) | 172 (54%) | 14 (44%) | |
| pT1 | 144 (41%) | 127 (40%) | 17 (53%) | |
| pT2 | 3 (0.8%) | 3 (0.9%) | 0 (0%) | |
| Cis | 19 (5.4%) | 18 (5.6%) | 1 (3.1%) | |
| Tx | 1 (0.3%) | 1 (0.3%) | 0 (0%) | |
| Grading, n (%) | 0.40 | |||
| Low | 100 (28%) | 93 (29%) | 7 (22%) | |
| High | 253 (72%) | 228 (71%) | 25 (78%) | |
| Concomitant Cis | 10 (2.8%) | 9 (2.8%) | 1 (3.1%) | >0.99 |
| Therapy, n (%) | 0.65 | |||
| MMC | 178 (50%) | 163 (51%) | 15 (47%) | |
| BCG | 159 (45%) | 142 (44%) | 17 (53%) | |
| Epirubicin | 16 (4.5%) | 16 (4.9%) | 0 (0%) | |
| Histology at Recurrence, n (%) | 0.066 | |||
| Ta | 98 (64%) | 91 (65%) | 7 (50%) | |
| T1 | 33 (22%) | 29 (21%) | 4 (29%) | |
| T2 | 4 (2.6%) | 2 (1.4%) | 2 (14%) | |
| Cis | 18 (12%) | 17 (12%) | 1 (7.1%) | |
| Tx | 0 (0%) | 0 (0%) | 0 (0%) | |
| Grading at Recurrence, n (%) | 0.29 | |||
| Low | 64 (42%) | 60 (43%) | 4 (29%) | |
| High | 89 (58%) | 79 (57%) | 10 (71%) | |
| Concomitant Cis at Recurrence | 5 (3.3%) | 4 (2.9%) | 1 (7.1%) | 0.39 |
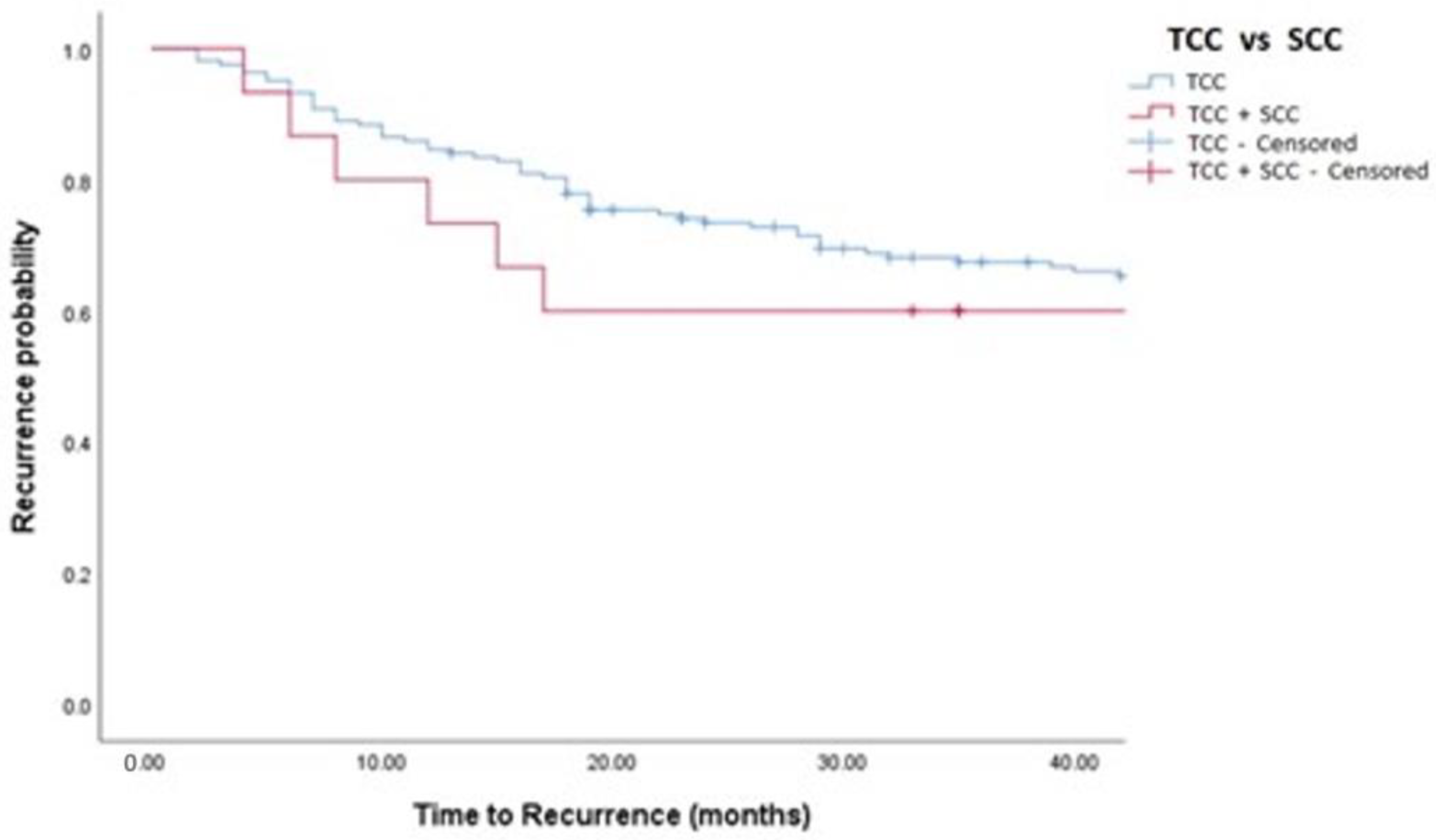
| Time To Recurrence | 35 (15, 58) | 33 (16, 57) | 40 (13, 78) | 0.44 |
| Recurrence, n (%) | 150 (42%) | 136 (42%) | 14 (44%) | 0.88 |
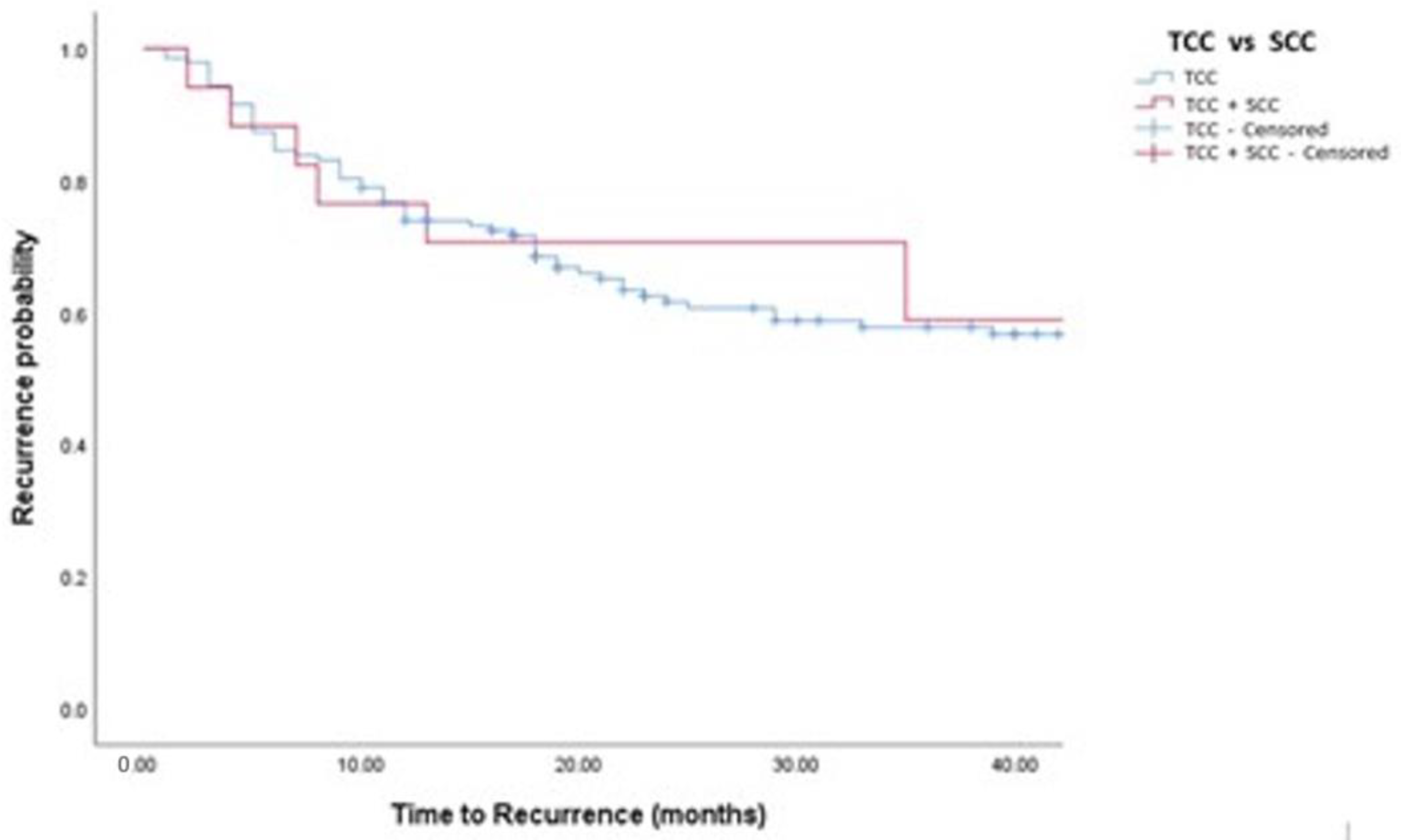
| Progression, n (%) | 42 (12%) | 38 (12%) | 4 (12%) | 0.78 |
| Time To Progression | 36 (16, 58) | 36 (16, 57) | 40 (13, 78) | 0.51 |
| Univariable | Multivariable | |||
|---|---|---|---|---|
| HR (95% CI) | p-Value | HR (95% CI) | p-Value | |
| Age | 1.00 (0.99, 1.01) | 0.896 | 1.00 (0.99, 1.01) | 0.947 |
| Smoking (No as Ref) | 0.592 | 0.643 | ||
| Yes | 0.96 (0.58, 1.59) | 0.872 | 0.93 (0.55, 1.58) | 0.792 |
| Ex-smoker | 1.16 (0.68, 1.97) | 0.591 | 1.11 (0.63, 1.96) | 0.710 |
| Single vs. Multi (Single as Ref) | 1.07 (0.76, 1.49) | 0.702 | 1.07 (0.76, 1.53) | 0.691 |
| Dimension (<3 cm as Ref) | 0.99 (0.71, 1.38) | 0.958 | 1.01 (0.71, 1.44) | 0.948 |
| TCC vs. TCC + SCC | 1.03 (0.59, 1.79) | 0.915 | 0.88 (0.50, 1.56) | 0.663 |
| Histology (Ta as Ref) | 0.041 | 0.033 | ||
| T1 | 1.59 (0.64, 3.95) | 0.315 | 1.30 (0.85, 2.00) | 0.229 |
| T2 | 1.76 (0.71, 4.41) | 0.224 | 5.20 (1.53, 17.69) | 0.008 |
| Cis | 7.48 (1.77, 31.60) | 0.006 | 0.73 (0.28, 1.92) | 0.528 |
| Grading (Low as Ref) | 0.79 (0.55, 1.12) | 0.183 | 0.52 (0.31, 0.88) | 0.015 |
| Concomitant Cis (No as Ref) | 1.22 (0.52, 2.87) | 0.653 | 1.20 (0.51, 2.87) | 0.674 |
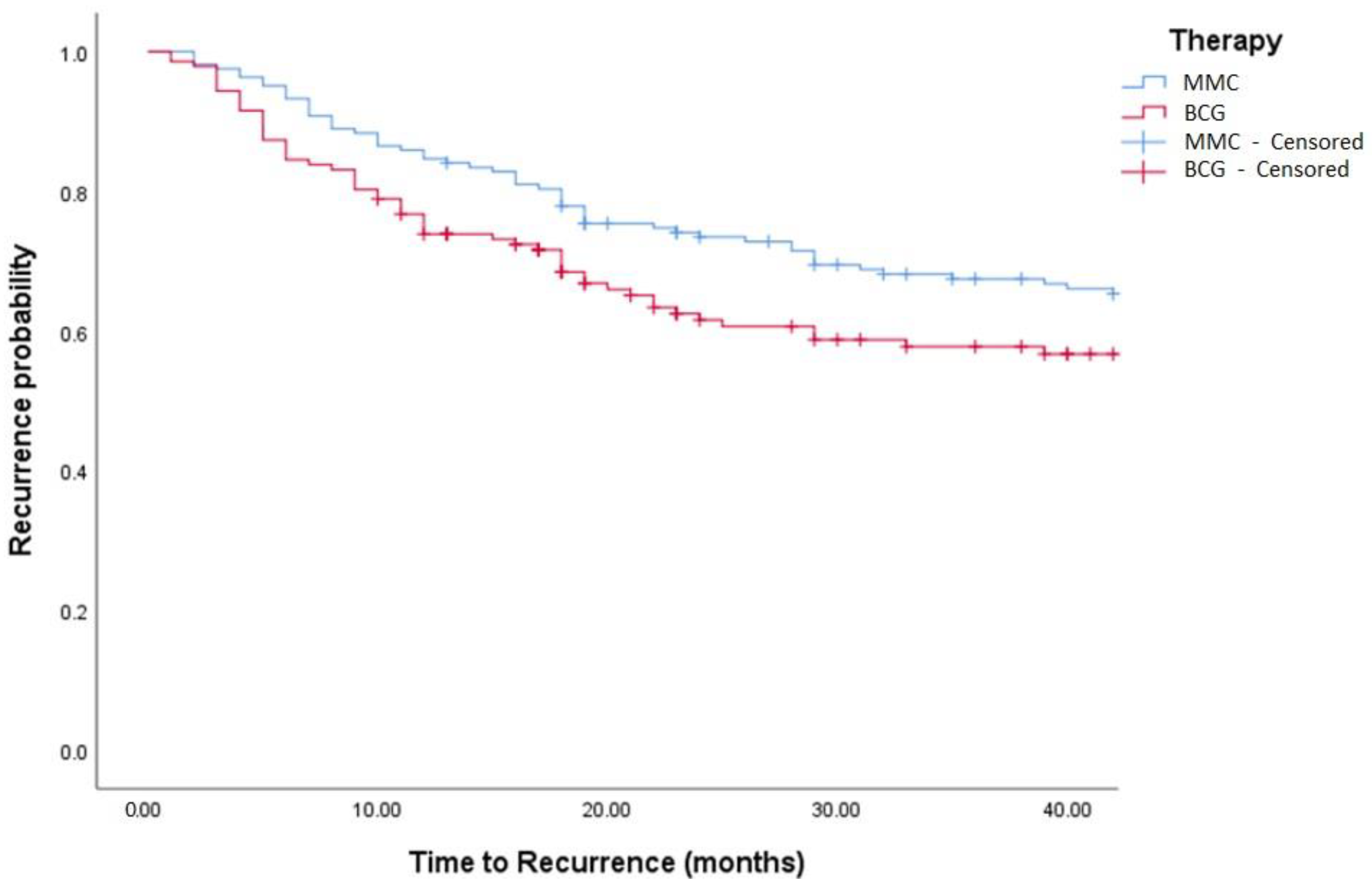
| BCG vs. MMC (MMC as Ref) | 1.17 (0.84, 1.64) | 0.362 | 1.47 (0.93, 2.35) | 0.103 |
| Univariable | Multivariable | |||
|---|---|---|---|---|
| HR (95% CI) | p-Value | HR (95% CI) | p-Value | |
| Age | 1.01 (0.99, 1.04) | 0.369 | 1.01 (0.99, 1.04) | 0.303 |
| Smoking (No as Ref) | 0.118 | 0.090 | ||
| Yes | 1.21 (0.41, 3.58) | 0.728 | 1.20 (0.39, 3.64) | 0.753 |
| Ex-smoker | 2.26 (0.75, 6.83) | 0.147 | 2.40 (0.76, 7.58) | 0.135 |
| Single vs. Multi (Single as Ref) | 0.94 (0.50, 1.79) | 0.856 | 0.80 (0.40, 1.57) | 0.509 |
| Dimension (<3 cm as Ref) | 1.06 (0.57, 1.95) | 0.865 | 1.03 (0.54, 1.97) | 0.936 |
| TCC vs. TCC + SCC | 1.06 (0.38, 3.01) | 0.907 | 0.74 (0.25, 2.17) | 0.582 |
| Histology (Ta as Ref) | 0.072 | 0.298 | ||
| T1 | 1.88 (0.97, 3.65) | 0.061 | 1.67 (0.76, 3.68) | 0.203 |
| T2 | 9.42 (1.21, 73.23) | 0.032 | 6.48 (0.75, 55.63) | 0.089 |
| Cis | 1.89 (0.55, 6.55) | 0.316 | 1.82 (0.46, 7.10) | 0.392 |
| Grading (Low as Ref) | 0.55 (0.24, 1.26) | 0.158 | 0.88 (0.29, 2.66) | 0.814 |
| Concomitant Cis (No as Ref) | 1.78 (0.23, 13.90) | 0.58 | 0.41 (0.05, 3.21) | 0.397 |
| BCG vs. MMC (MMC as Ref) | 2.08 (1.09, 3.97) | 0.026 | 1.81 (0.79, 4.15) | 0.159 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mantica, G.; Chierigo, F.; Malinaric, R.; Smelzo, S.; Ambrosini, F.; Beverini, M.; Guano, G.; Caviglia, A.; Rigatti, L.; De Rose, A.F.; et al. Intravesical Therapy for Non-Muscle-Invasive Bladder Cancer: What Is the Real Impact of Squamous Cell Carcinoma Variant on Oncological Outcomes? Medicina 2022, 58, 90. https://doi.org/10.3390/medicina58010090
Mantica G, Chierigo F, Malinaric R, Smelzo S, Ambrosini F, Beverini M, Guano G, Caviglia A, Rigatti L, De Rose AF, et al. Intravesical Therapy for Non-Muscle-Invasive Bladder Cancer: What Is the Real Impact of Squamous Cell Carcinoma Variant on Oncological Outcomes? Medicina. 2022; 58(1):90. https://doi.org/10.3390/medicina58010090
Chicago/Turabian StyleMantica, Guglielmo, Francesco Chierigo, Rafaela Malinaric, Salvatore Smelzo, Francesca Ambrosini, Martina Beverini, Giovanni Guano, Alberto Caviglia, Lorenzo Rigatti, Aldo Franco De Rose, and et al. 2022. "Intravesical Therapy for Non-Muscle-Invasive Bladder Cancer: What Is the Real Impact of Squamous Cell Carcinoma Variant on Oncological Outcomes?" Medicina 58, no. 1: 90. https://doi.org/10.3390/medicina58010090
APA StyleMantica, G., Chierigo, F., Malinaric, R., Smelzo, S., Ambrosini, F., Beverini, M., Guano, G., Caviglia, A., Rigatti, L., De Rose, A. F., Tafuri, A., De Marchi, D., Gaboardi, F., Suardi, N., & Terrone, C. (2022). Intravesical Therapy for Non-Muscle-Invasive Bladder Cancer: What Is the Real Impact of Squamous Cell Carcinoma Variant on Oncological Outcomes? Medicina, 58(1), 90. https://doi.org/10.3390/medicina58010090









