Abstract
Background and Objectives: Irreversible visual impairment is mainly caused by retinal degenerative diseases such as age-related macular degeneration and retinitis pigmentosa. Stem cell research has experienced rapid progress in recent years, and researchers and clinical ophthalmologists are trying to implement this promising technology to treat retinal degeneration. The objective of this systematic review is to analyze currently available data from clinical trials applying stem cells to treat human retinal diseases. Materials and Methods: We performed a systematic literature search in PubMed to identify articles related with stem cell therapies to retinal diseases published prior to September 2021. Furthermore, a systematic search in ClinicalTrials (NIH U.S. National Library of Medicine) was performed to identify clinical trials using stem cells to treat retinal diseases. A descriptive analysis of status, conditions, phases, interventions, and outcomes is presented here. Conclusions: To date, no available therapy based on stem cell transplantation is approved for use with patients. However, numerous clinical trials are currently finishing their initial phases and, in general, the outcomes related to implantation techniques and their long-term safety seem promising. In the next few years, we expect to see quantifiable results pertaining to visual function improvement.
1. Introduction
Irreversible visual impairment is mainly caused by retinal degenerative diseases such as age-related macular degeneration (AMD) and retinitis pigmentosa (RP). Macular diseases cause the loss of the retinal pigment epithelium together with the photoreceptors that support it [1]. It has been estimated that AMD had affected 196 million people by 2020 [2,3,4], while RP affects about 2.5 million people worldwide [4,5]. These numbers are projected to heavily increase in the next 20 years, posing substantial social and economic concerns [2,3].
There is currently no surgical or pharmacological solution to regenerate an injured or degenerative retina, and the only approach ophthalmologists can take is to slow the progress of the loss of vision. However, stem cell research has experienced rapid development in recent years. In the last decade, many efforts have been made to take advantage of the promising properties of the stem cell technology and apply them to retinal degenerative diseases. Pluripotent stem cells (PSCs) have the ability to undergo self-renewal and to give rise to all cells of the tissues of the body [6]. There are two types of PSCs depending on their origin, namely embryonic stem cells (ESC) and induced pluripotent stem cells (iPSCs) [7,8]. Since the refinement of methods to obtain large amounts of pluripotent cells from human adult tissues obviates the necessity for the use of embryonic tissue to isolate PSCs, there has been an increased interest in different medical applications of these cell technologies. Advances in cell processing have included the isolation of different adult somatic cells candidates to be dedifferentiated and induced to become PSCs, as well as the identification of new adult stem cells niches (and the isolation of many of them to be differentiate to other cell types). Most effort to date has been on the conversion of mesenchymal stem cells to muscle fibers, cardiomyocytes, B cells of the pancreas, or neurons.
The use of retinal pigment epithelial cells (RPE) and photoreceptor replacement have been investigated in implantation formats, such as an injection of cells or the placing of a sheet of cells over a substrate [9]. In this review, we will focus on the analysis of the currently available clinical trials data involving stem cells to treat human retinal diseases.
2. Methods
We performed a systematic literature search in PubMed to identify articles on stem cell therapies related to retinal diseases published up to September 2021. The search term used was “Stem cell transplant AND retina”. Articles were subsequently selected using the following PubMed filters: “Human”, “Adult: +19 years”, Child: birth–18 years”, and “Clinical Trials”. In addition, a systematic search in ClinicalTrials.gov (NIH U.S. National Library of Medicine) was carried to identify clinical trials using stem cells to treat retinal diseases. The search terms used were: Status “All studies”; Condition or disease “Retina Disorder”, “Retinal degeneration”, “Retinal dystrophies”; Other terms “Stem cells”; Country “Empty”. A descriptive analysis of status, conditions, phases, interventions and outcomes is presented below.
3. Retinal Therapies Using Stem Cells in the Literature Search
The first level of search using “Stem cell transplant AND retina” keywords produced 1229 publications indexed in PubMed. By using the filter “Human” we excluded 459 of the studies. Two additional filters were subsequently applied in order to ensure that the aim of the studies focused on patient intervention and excluded in vitro or animal experimental procedures. We used the “Adult: 19+ years” PubMed filter followed by “Child: birth- 18 years”, which resulted in a data set of 121 studies. We then identified 12 review articles and 12 clinical trials by activating the corresponding filters in PubMed. Two of the clinical trials were not related to retinal treatments and were excluded from the study. A summary of the conclusions of the 10 papers containing results from clinical trials and published between 2014 and 2021 [10,11,12,13,14,15,16,17,18,19] is presented in Table 1.

Table 1.
Summary of the results of the articles linked to clinical trials using stem cells to treat human retinal diseases.
4. Clinical Trials Using Stem Cells to Treat the Human Retina
The systematic search for clinical trials in the NIH database ClinicalTrials.gov produced 82 results using the searching terms “Retina Disorder” + “Stem cells”. The list was then manually filtered and 13 studies were excluded that did not actually use stem cells as therapy. The 69 remaining records included 31 studies that were also obtained using the search terms “Retinal dystrophies” + “Stem Cells” and 58 which were also found using “Retinal Degeneration” + “Stem Cells”. A list of the clinical trials used for this work is provided as supplementary material “Review of Clinical Trials” and their main characteristics are summarized in Figure 1.
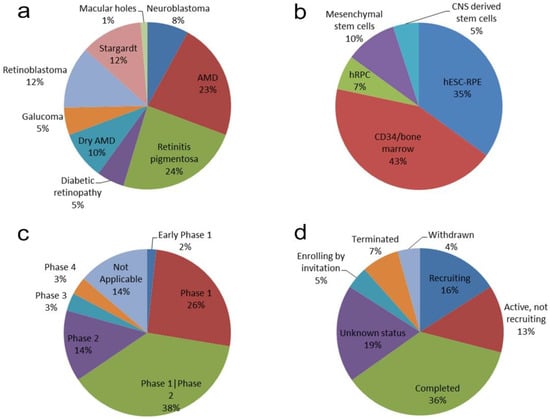
Figure 1.
Summary of findings of the analysis of 69 Clinical trials using human stem cells for transplantation into diseased human retina. Sector graphs showing: (a) clinical trials with respect to the diseases involved; (b) the different sources of the stem cells used; (c) the stage of the trials at the time of the published work; (d) the current status of the trials.
The majority of the clinical trials studied disease conditions related to the degenerative loss of photoreceptors and/or RPE cells, such as AMD (33%), RP (24%), or Stargardt disease (12%), while the most prevalent degenerative diseases of the retina, such as glaucoma, only accounted for a 5% of the studies. Furthermore, 20% of the trials were dedicated to studying ocular tumors (Figure 1a). Thus, it can be seen that the efforts of clinicians and stem cell researchers have focused on photoreceptor and RPE replacement using alternative sources of cells.
Of the 69 clinical trials selected according to our search criteria, 9 were found not to use cells as a transplantation therapy. These include the use of optical coherence tomography (OCT) to evaluate changes in the ganglion cell complex (GCC) in diabetes (NCT02620644) or the evaluation of drugs to treat diabetic retinopathy (NCT01927315; NCT02353923). In addition, the objective of six of the trials was to obtain iPSCs for research purposes from the skin, hair or peripheral blood biopsies of patients with retinoblastoma (NCT02193724), diabetes (NCT03403699), AMD (NCT03372746; NCT02464956; NCT01432847), or other retinal diseases such as macular dystrophies (NCT02162953).
A total of 58.3% of the trials (35 out of 60) used cells from extraocular sources, with autologous bone marrow stem cells being the cell type preferred to prepare the transplantation product (in 26 cases), followed by mesenchymal stem cells of umbilical cord origin (NCT03437759; NCT04224207; NCT04315025; NCT04763369) and adipose tissue (NCT02024269; NCT02144103). Furthermore, there were three clinical trials using neuronal precursors obtained from fetal brains for retinal transplantation (Figure 1b), all of which were sponsored by StemCells, Inc. Two of these studies were terminated on the basis of a business decision unrelated to safety concerns, as reported by the company (NCT02467634; NCT02137915), while, although the other was completed, no results were posted (NCT01632527).
The use of neuronal cells to repair retinal degeneration would seem to be the most appropriate strategy due to the similarity between transplanted cells and the degenerated retinal neurons, even when 88.3% of the 60 trials were based on the use of non-neuronal cells (Figure 1b). The more suitable situation would be the use of retinal neuronal cells. One of the best-known stem cell therapies in retinal disease is “photoreceptor replacement therapy”, which can be used in various retinopathies in a way that allows the restoration of visual function [20]. Numerous preclinical studies have reported a modest degree of synaptic integration of the transplanted cells into the host retina, suggesting some capacity for regeneration [21,22,23]. In the process of the degeneration of the mammalian retina, the underlying retinal circuit remains well-preserved for a long period of time, thus allowing these circuits to be reactivated through cell therapy so that the photosensitivity of the degenerating retina could be restored [20]. In stem cell-derived rod photoreceptor precursor transplants, improvements in visual functioning were initially thought to occur since donor cells were integrated into the host’s retina, but later studies showed that what occurs after transplantation is an exchange of cytoplasmic material between the transplanted cells and the cells of the host retina, thereby enabling them to acquire the proteins they lacked [1]. The benefit of neural cell transplantation may, therefore, be driven by the neuroprotective/regenerative effects of the neurotransmitters and growth factors delivered by the transplanted cells. Four clinical trials identified by our systematic search used human retinal precursor cells (hRPCs), which were presumed to be of embryonic neuronal origin. All four used advanced cellular products from the Biotech companies JCyte (NCT04604899; NCT03073733; NCT02320812) and Reneuron (NCT02464436), understandable as hRPC treatment may be more specific in its therapeutic mechanism as compared to bone marrow stem cells or mesenchymal stem cells [9]. The problem with obtaining newborn photoreceptors, however, is that there is not an unlimited source [24]. Furthermore, neuronal cells account for additional difficulties due to their inability to be expanded in vitro as well as their low survival rate after isolation or transplantation. In the studies mentioned here, however, the researchers obtained precursor cells from human fetuses, thus increasing their survival capacity. The counterpoint to this, however, is the difficulty of obtaining the correct in situ differentiation and thus increasing the risk of tumorigenesis. This fact reduces their feasibility in transplantation procedures. At the date of this review, two of the trials had completed phase 1 and phase 2 studies, one of them with results available at ClinicalTrials.gov (https://ClinicalTrials.gov/show/NCT02320812 (accessed on 10 September 2021)). This study involved 28 participants and only one case of serious adverse effects (migratory pain) was found, while all cases reported improved visual acuity, suggesting the safety and feasibility of the procedure. The other two trials are also in phases 1 and 2, providing support for the safety of hRPC transplantation.
However, the preferred source of ocular cells is not a neural cell. Twenty-one trials used RPE cells or cells induced to give rise to RPE (Figure 1b). There is already technical evidence for the successful implantation of RPE cells and reported improvement in visual function. The cases reviewed by Jha and Bharti [25] that used a small piece of autologous RPE-choroid from the peripheral retina for implantation into macular atrophic areas in AMD patients evidenced the long-term integration of the transplant, as well as vision stabilization for several years. A phase 2 clinical trial in which human fetal neural retinal tissue and RPE were co-transplanted in AMD patients (NCT00346060), showed promising results in terms of vision improvement six months post-implantation [26].
Furthermore, RPE cells seem to be more suitable for transplantation as they do not require synaptic connections to adjacent cells [27]. Transplantation of hESC-derived RPE in AMD patients has shown good preliminary results in terms of safety and tolerability. The results from the studies using hESC-derived RPE are promising and supports the notion of exgtending this line of research in the future to randomized multicenter trials using functional measures to appropriately evaluate its efficacy [9].
5. Different Administration Routes for Different Types of Stem Cells
Intravitreal injection of cell suspensions is the method of choice for transplantation of bone marrow stem cells in the majority of clinical trials. Also, 3 of the studies reviewed here used this route to deliver RPC into the eye. The intravitreal injection of a suspension of cells does, however, have drawbacks such as the need for the cells to migrate from the injection site to the site of cell loss, incomplete maturation and differentiation into specific phenotypes, and the fact that they have to form connections between each other, as well as with other cells in the diseased eye, in order to remain functional.
The subretinal space is the location selected by all of the clinical trials using RPE-based strategies (21 out of 60) and also by one study using RPC. Injections of cell suspensions of human RPE and of photoreceptors alone in the subretinal space have demonstrated partial vision restoration in preclinical models of early retinal degeneration [28,29,30,31] and the applicability of these injections in patients is currently in phase 1/2 clinical trials. The transplantation of cells as a sheet partially overcomes the inconveniences of injections, as they can immobilize and support the cells. Transplanted retinal sheets have shown better survival compared to RPE injected as a suspension [32], as well as the formation of synapses with other neuronal retinal cells months after implantation (and even visual recovery, in some cases). Polymeric biomaterials such as parylene, polyester, and poly (lactide-co-glycolic acid) (PLGA) sheets have been used to seed hESC-derived RPE cells and iPSC-derived RPE cells in phase 1 and 1/2 studies of implantation in AMD patients (NCT02590692; NCT04339764; NCT01691261).
6. Current Status of Clinical Trials
The majority of the trials (68%) were in the initial phases of their studies (phases 1 and 2), which correspond to safety and feasibility outcomes. Only 2 of 69 trials reported reaching phase 4 and other 2 has reached phase 3 (Figure 1c). Eight studies were withdrawn or terminated prior to the scheduled end-date. Only 14 of the 69 clinical trials were currently recruiting or enrolling patients while other 9 trials were active but not recruiting patients. Only 3 trials had posted results at ClinicalTrials.gov. Although 36% of the trials were completed, only two of them had posted results. Seven articles from our PubMed search had published results linked with 8 of the clinical trials [10,11,13,14,15,16,19].
7. Future Directions
The problem with obtaining viable human compatible cells (photoreceptors and RPE) is that there is not an unlimited source, since so many groups have focused on obtaining this [24]. hiPSCs have been explored as a near-unlimited cell source for cell replacement of both types of retinal cells, photoreceptors and RPE cells [7,33,34,35,36,37]. Indeed, a safety and tolerability prospective clinical trial is currently underway to evaluate subretinal transplantation of iPSC-derived RPE cell sheets in AMD patients (NCT04339764). Additionally, five clinical trials are active or completed whose objective was to obtain hiPSCs for transplantation into diseased retinas, although none of the studies involve surgical procedures in patients (NCT03403699; NCT03372746; NCT02464956; NCT02162953; NCT01432847). Due to the intrinsic complexity of neural tissue reconnection, the use of neuronal precursors or in vitro differentiated neuronal constructs in retinal transplantation seems to still require further preclinical development. One interesting use of stem cells is the creation of organoids, which consist of a tiny three-dimensional mass of tissue developed in the laboratory by growing stem cells. It is possible to grow organoids that resemble human tissues or organs, or a specific type of tumor (NIH). Consequently, iPSCs can be reprogrammed to generate a retina from which an enormous amount of information about the transcriptional and epigenetic regulation of retinal development and maintenance can be obtained [38]. In addition, they serve to advance cell therapies to treat diseases such as AMD and glaucoma [38]. Besides, self-organizing optic cups and 3D neural retinas that could be clinically applied for transplantation therapy in retinal degeneration have been successfully created from hiPSCs by 3D differentiation culture [36,37,39].
8. Conclusions
To date there is no available therapy based on stem cell transplantation that has been approved for use with patients. Numerous clinical trials are currently finishing the early phases of their studies and, in general, the outcomes related with implantation techniques and long-term safety seem promising, including at five-year follow-up after transplantation. In the next few years, we expect to see quantifiable results in terms of visual function improvement. The use of stem cells in eye regeneration is slowly becoming positioned as an exciting retinal strategy for treating degenerative diseases. However, there is still a need for more in-depth research to increase our understanding of the mechanisms underlying stem cell differentiation into the different retinal neuronal phenotypes and to develop techniques to achieve synaptic integration and circuitry functional restoration.
Supplementary Materials
The following are available online at https://www.mdpi.com/article/10.3390/medicina58010102/s1.
Author Contributions
Conceptualization, I.A. and S.D.O.-A.; resources, I.A. and C.S.-F.; data curation, I.A., C.M., N.D.P. and C.S.-F.; writing—original draft preparation, I.A., N.D.P. and C.S.-F.; writing—review and editing, I.A. and N.J.-D.; supervision, J.M.-L. and G.V.G. All authors have read and agreed to the published version of the manuscript.
Funding
This study was funded in part by Foundation Caja Rural de Asturias (FIO#21001).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Data used to write this review were taken from the public database ClinicalTrials.gov (https://clinicaltrials.gov/ (accessed on 10 September 2021)) and PubMed.gov (https://pubmed.ncbi.nlm.nih.gov/ (accessed on 10 September 2021)). All data supporting reported results can be found using the search terms published in this article.
Acknowledgments
The authors would like to acknowledge the careful reading of the manuscript by Luis M. Quirós and Fivos Panetsos and thank them for their support.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Gonzalez-Cordero, A.; Kruczek, K.; Naeem, A.; Fernando, M.; Kloc, M.; Ribeiro, J.; Goh, D.; Duran, Y.; Blackford, S.J.I.; Abelleira-Hervas, L.; et al. Recapitulation of Human Retinal Development from Human Pluripotent Stem Cells Generates Transplantable Populations of Cone Photoreceptors. Stem Cell Rep. 2017, 9, 820–837. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wong, W.L.; Su, X.; Li, X.; Cheung, C.M.G.; Klein, R.; Cheng, C.-Y.; Wong, T.Y. Global Prevalence of Age-Related Macular Degeneration and Disease Burden Projection for 2020 and 2040: A Systematic Review and Meta-Analysis. Lancet Glob. Health 2014, 2, e106–e116. [Google Scholar] [CrossRef] [Green Version]
- Jemni-Damer, N.; Guedan-Duran, A.; Fuentes-Andion, M.; Serrano-Bengoechea, N.; Alfageme-Lopez, N.; Armada-Maresca, F.; Guinea, G.V.; Perez-Rigueiro, J.; Rojo, F.; Gonzalez-Nieto, D.; et al. Biotechnology and Biomaterial-Based Therapeutic Strategies for Age-Related Macular Degeneration. Part II: Cell and Tissue Engineering Therapies. Front. Bioeng. Biotechnol. 2020, 8, 1419. [Google Scholar] [CrossRef]
- Jemni-Damer, N.; Guedan-Duran, A.; Fuentes-Andion, M.; Serrano-Bengoechea, N.; Alfageme-Lopez, N.; Armada-Maresca, F.; Guinea, G.V.; Pérez-Rigueiro, J.; Rojo, F.; Gonzalez-Nieto, D.; et al. Biotechnology and Biomaterial-Based Therapeutic Strategies for Age-Related Macular Degeneration. Part I: Biomaterials-Based Drug Delivery Devices. Front. Bioeng. Biotechnol. 2020, 8, 1257. [Google Scholar] [CrossRef]
- Dias, M.F.; Joo, K.; Kemp, J.A.; Fialho, S.L.; da Silva Cunha, A.J.; Woo, S.J.; Kwon, Y.J. Molecular Genetics and Emerging Therapies for Retinitis Pigmentosa: Basic Research and Clinical Perspectives. Prog. Retin. Eye Res. 2018, 63, 107–131. [Google Scholar] [CrossRef]
- Romito, A.; Cobellis, G. Pluripotent Stem Cells: Current Understanding and Future Directions. Stem Cells Int. 2016, 2016, 9451492. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Takahashi, K.; Tanabe, K.; Ohnuki, M.; Narita, M.; Ichisaka, T.; Tomoda, K.; Yamanaka, S. Induction of Pluripotent Stem Cells from Adult Human Fibroblasts by Defined Factors. Cell 2007, 131, 861–872. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thomson, J.A.; Itskovitz-Eldor, J.; Shapiro, S.S.; Waknitz, M.A.; Swiergiel, J.J.; Marshall, V.S.; Jones, J.M. Embryonic Stem Cell Lines Derived from Human Blastocysts. Science 1998, 282, 1145–1147. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Singh, M.S.; Park, S.S.; Albini, T.A.; Canto-Soler, M.V.; Klassen, H.; MacLaren, R.E.; Takahashi, M.; Nagiel, A.; Schwartz, S.D.; Bharti, K. Retinal Stem Cell Transplantation: Balancing Safety and Potential. Prog. Retin. Eye Res. 2020, 75, 100779. [Google Scholar] [CrossRef]
- da Cruz, L.; Fynes, K.; Georgiadis, O.; Kerby, J.; Luo, Y.H.; Ahmado, A.; Vernon, A.; Daniels, J.T.; Nommiste, B.; Hasan, S.M.; et al. Phase 1 Clinical Study of an Embryonic Stem Cell-Derived Retinal Pigment Epithelium Patch in Age-Related Macular Degeneration. Nat. Biotechnol. 2018, 36, 328–337. [Google Scholar] [CrossRef] [Green Version]
- Kashani, A.H.; Uang, J.; Mert, M.; Rahhal, F.; Chan, C.; Avery, R.L.; Dugel, P.; Chen, S.; Lebkowski, J.; Clegg, D.O.; et al. Surgical Method for Implantation of a Biosynthetic Retinal Pigment Epithelium Monolayer for Geographic Atrophy: Experience from a Phase 1/2a Study. Ophthalmol. Retin. 2020, 4, 264–273. [Google Scholar] [CrossRef]
- Park, S.S.; Bauer, G.; Abedi, M.; Pontow, S.; Panorgias, A.; Jonnal, R.; Zawadzki, R.J.; Werner, J.S.; Nolta, J. Intravitreal Autologous Bone Marrow CD34+ Cell Therapy for Ischemic and Degenerative Retinal Disorders: Preliminary Phase 1 Clinical Trial Findings. Investig. Ophthalmol. Vis. Sci. 2014, 56, 81–89. [Google Scholar] [CrossRef]
- Song, W.K.; Park, K.-M.; Kim, H.-J.; Lee, J.H.; Choi, J.; Chong, S.Y.; Shim, S.H.; Del Priore, L.V.; Lanza, R. Treatment of Macular Degeneration Using Embryonic Stem Cell-Derived Retinal Pigment Epithelium: Preliminary Results in Asian Patients. Stem Cell Rep. 2015, 4, 860–872. [Google Scholar] [CrossRef]
- Schwartz, S.D.; Regillo, C.D.; Lam, B.L.; Eliott, D.; Rosenfeld, P.J.; Gregori, N.Z.; Hubschman, J.-P.; Davis, J.L.; Heilwell, G.; Spirn, M.; et al. Human Embryonic Stem Cell-Derived Retinal Pigment Epithelium in Patients with Age-Related Macular Degeneration and Stargardt’s Macular Dystrophy: Follow-up of Two Open-Label Phase 1/2 Studies. Lancet 2015, 385, 509–516. [Google Scholar] [CrossRef]
- Mehat, M.S.; Sundaram, V.; Ripamonti, C.; Robson, A.G.; Smith, A.J.; Borooah, S.; Robinson, M.; Rosenthal, A.N.; Innes, W.; Weleber, R.G.; et al. Transplantation of Human Embryonic Stem Cell-Derived Retinal Pigment Epithelial Cells in Macular Degeneration. Ophthalmology 2018, 125, 1765–1775. [Google Scholar] [CrossRef] [Green Version]
- Sung, Y.; Lee, M.J.; Choi, J.; Jung, S.Y.; Chong, S.Y.; Sung, J.H.; Shim, S.H.; Song, W.K. Long-Term Safety and Tolerability of Subretinal Transplantation of Embryonic Stem Cell-Derived Retinal Pigment Epithelium in Asian Stargardt Disease Patients. Br. J. Ophthalmol. 2021, 105, 829–837. [Google Scholar] [CrossRef]
- Oner, A.; Gonen, Z.B.; Sinim, N.; Cetin, M.; Ozkul, Y. Subretinal Adipose Tissue-Derived Mesenchymal Stem Cell Implantation in Advanced Stage Retinitis Pigmentosa: A Phase I Clinical Safety Study. Stem Cell Res. Ther. 2016, 7, 178. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, Y.; Chen, S.J.; Li, S.Y.; Qu, L.H.; Meng, X.H.; Wang, Y.; Xu, H.W.; Liang, Z.Q.; Yin, Z.Q. Long-Term Safety of Human Retinal Progenitor Cell Transplantation in Retinitis Pigmentosa Patients. Stem Cell Res. Ther. 2017, 8, 209. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, S.-Y.; Liu, Y.; Wang, L.; Wang, F.; Zhao, T.-T.; Li, Q.-Y.; Xu, H.-W.; Meng, X.-H.; Hao, J.; Zhou, Q.; et al. A Phase I Clinical Trial of Human Embryonic Stem Cell-Derived Retinal Pigment Epithelial Cells for Early-Stage Stargardt Macular Degeneration: 5-Years’ Follow-Up. Cell Prolif. 2021, 54, e13100. [Google Scholar] [CrossRef]
- Decembrini, S.; Koch, U.; Radtke, F.; Moulin, A.; Arsenijevic, Y. Derivation of Traceable and Transplantable Photoreceptors from Mouse Embryonic Stem Cells. Stem Cell Rep. 2014, 2, 853–865. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- del Cerro, M.; Ison, J.R.; Bowen, G.P.; Lazar, E.; del Cerro, C. Intraretinal Grafting Restores Visual Function in Light-Blinded Rats. Neuroreport 1991, 2, 529–532. [Google Scholar] [CrossRef] [PubMed]
- Gouras, P.; Du, J.; Kjeldbye, H.; Kwun, R.; Lopez, R.; Zack, D.J. Transplanted Photoreceptors Identified in Dystrophic Mouse Retina by a Transgenic Reporter Gene. Investig. Ophthalmol. Vis. Sci. 1991, 32, 3167–3174. [Google Scholar]
- Gouras, P.; Du, J.; Gelanze, M.; Lopez, R.; Kwun, R.; Kjeldbye, H.; Krebs, W. Survival and Synapse Formation of Transplanted Rat Rods. J. Neural Transpl. Plast. 1991, 2, 91–100. [Google Scholar] [CrossRef] [PubMed]
- Lamba, D.A.; Gust, J.; Reh, T.A. Transplantation of Human Embryonic Stem Cell-Derived Photoreceptors Restores Some Visual Function in Crx-Deficient Mice. Cell Stem Cell 2009, 4, 73–79. [Google Scholar] [CrossRef] [Green Version]
- Jha, B.S.; Bharti, K. Regenerating Retinal Pigment Epithelial Cells to Cure Blindness: A Road Towards Personalized Artificial Tissue. Curr. Stem Cell Rep. 2015, 1, 79–91. [Google Scholar] [CrossRef] [PubMed]
- Radtke, N.D.; Aramant, R.B.; Petry, H.M.; Green, P.T.; Pidwell, D.J.; Seiler, M.J. Vision Improvement in Retinal Degeneration Patients by Implantation of Retina Together with Retinal Pigment Epithelium. Am. J. Ophthalmol. 2008, 146, 172–182. [Google Scholar] [CrossRef] [Green Version]
- Strauss, O. The Retinal Pigment Epithelium in Visual Function. Physiol. Rev. 2005, 85, 845–881. [Google Scholar] [CrossRef] [Green Version]
- Gasparini, S.J.; Llonch, S.; Borsch, O.; Ader, M. Transplantation of Photoreceptors into the Degenerative Retina: Current State and Future Perspectives. Prog. Retin. Eye Res. 2019, 69, 1–37. [Google Scholar] [CrossRef]
- Barber, A.C.; Hippert, C.; Duran, Y.; West, E.L.; Bainbridge, J.W.B.; Warre-Cornish, K.; Luhmann, U.F.O.; Lakowski, J.; Sowden, J.C.; Ali, R.R.; et al. Repair of the Degenerate Retina by Photoreceptor Transplantation. Proc. Natl. Acad. Sci. USA 2013, 110, 354–359. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pearson, R.A.; Barber, A.C.; Rizzi, M.; Hippert, C.; Xue, T.; West, E.L.; Duran, Y.; Smith, A.J.; Chuang, J.Z.; Azam, S.A.; et al. Restoration of Vision after Transplantation of Photoreceptors. Nature 2012, 485, 99–103. [Google Scholar] [CrossRef] [Green Version]
- Lu, B.; Malcuit, C.; Wang, S.; Girman, S.; Francis, P.; Lemieux, L.; Lanza, R.; Lund, R. Long-Term Safety and Function of RPE from Human Embryonic Stem Cells in Preclinical Models of Macular Degeneration. Stem Cells 2009, 27, 2126–2135. [Google Scholar] [CrossRef]
- Ben M’Barek, K.; Habeler, W.; Plancheron, A.; Jarraya, M.; Regent, F.; Terray, A.; Yang, Y.; Chatrousse, L.; Domingues, S.; Masson, Y.; et al. Human ESC-Derived Retinal Epithelial Cell Sheets Potentiate Rescue of Photoreceptor Cell Loss in Rats with Retinal Degeneration. Sci. Transl. Med. 2017, 9, eaai7471. [Google Scholar] [CrossRef] [Green Version]
- Yu, J.; Vodyanik, M.A.; Smuga-Otto, K.; Antosiewicz-Bourget, J.; Frane, J.L.; Tian, S.; Nie, J.; Jonsdottir, G.A.; Ruotti, V.; Stewart, R.; et al. Induced Pluripotent Stem Cell Lines Derived from Human Somatic Cells. Science 2007, 318, 1917–1920. [Google Scholar] [CrossRef]
- Jin, Z.-B.; Okamoto, S.; Mandai, M.; Takahashi, M. Induced Pluripotent Stem Cells for Retinal Degenerative Diseases: A New Perspective on the Challenges. J. Genet. 2009, 88, 417–424. [Google Scholar] [CrossRef]
- Jin, Z.-B.; Takahashi, M. Generation of Retinal Cells from Pluripotent Stem Cells. Prog. Brain Res. 2012, 201, 171–181. [Google Scholar] [CrossRef]
- Zhong, X.; Gutierrez, C.; Xue, T.; Hampton, C.; Vergara, M.N.; Cao, L.-H.; Peters, A.; Park, T.S.; Zambidis, E.T.; Meyer, J.S.; et al. Generation of Three-Dimensional Retinal Tissue with Functional Photoreceptors from Human IPSCs. Nat. Commun. 2014, 5, 4047. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhu, J.; Reynolds, J.; Garcia, T.; Cifuentes, H.; Chew, S.; Zeng, X.; Lamba, D.A. Generation of Transplantable Retinal Photoreceptors from a Current Good Manufacturing Practice-Manufactured Human Induced Pluripotent Stem Cell Line. Stem Cells Transl. Med. 2018, 7, 210–219. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Roberts, J.M.; Vetter, M.L. From Retina to Stem Cell and Back Again: Memories of a Chromatin Journey. Cell Rep. 2018, 22, 2519–2520. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tu, H.-Y.; Watanabe, T.; Shirai, H.; Yamasaki, S.; Kinoshita, M.; Matsushita, K.; Hashiguchi, T.; Onoe, H.; Matsuyama, T.; Kuwahara, A.; et al. Medium- to Long-Term Survival and Functional Examination of Human IPSC-Derived Retinas in Rat and Primate Models of Retinal Degeneration. EBioMedicine 2019, 39, 562–574. [Google Scholar] [CrossRef] [Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).