Influence of SARS-CoV-2 Virus Infection on the Course of Psoriasis during Treatment with Biological Drugs
Abstract
:1. Introduction
2. Materials and Methods
2.1. Observation Method
2.2. RT-PCR Diagnostics
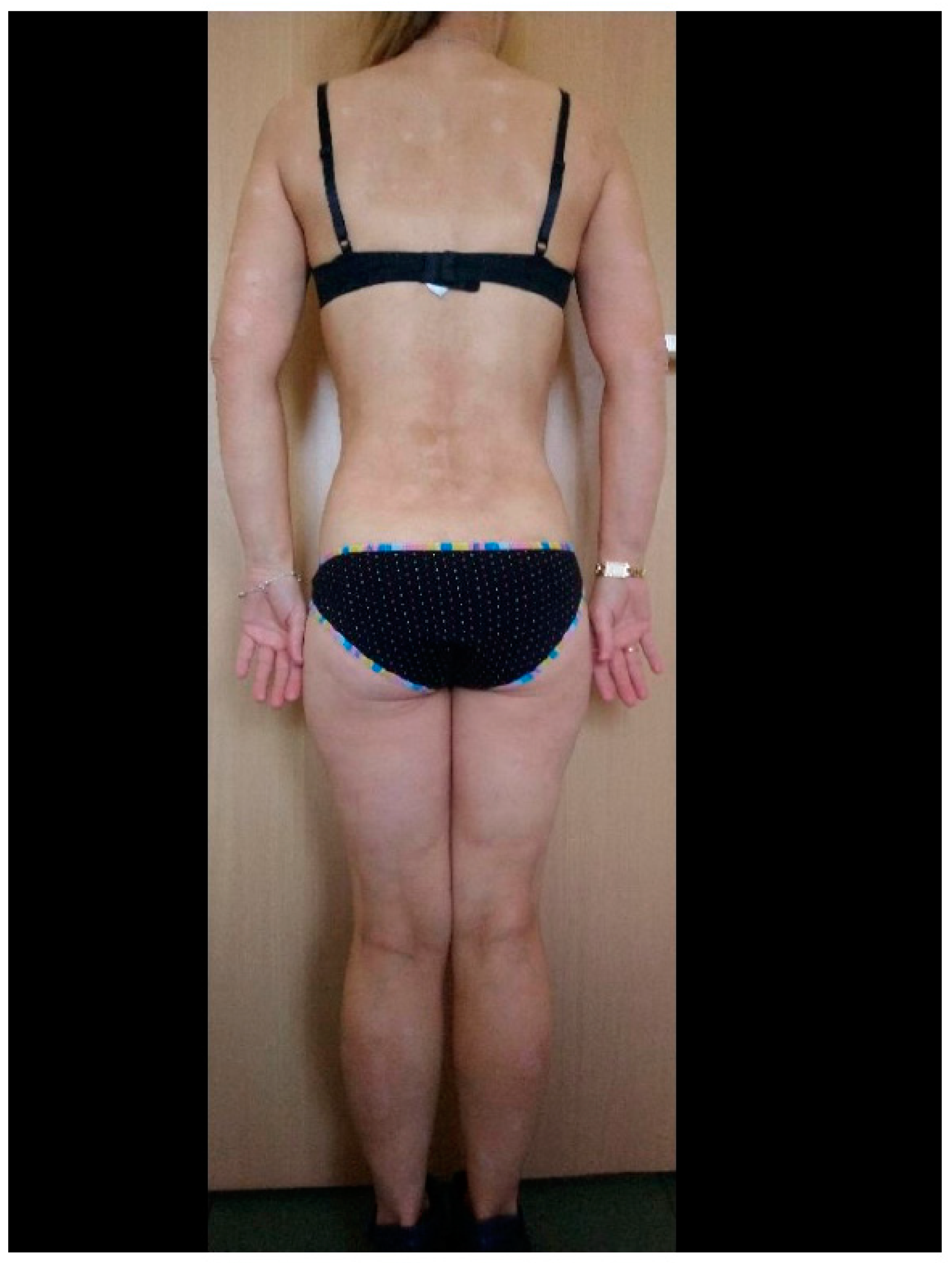
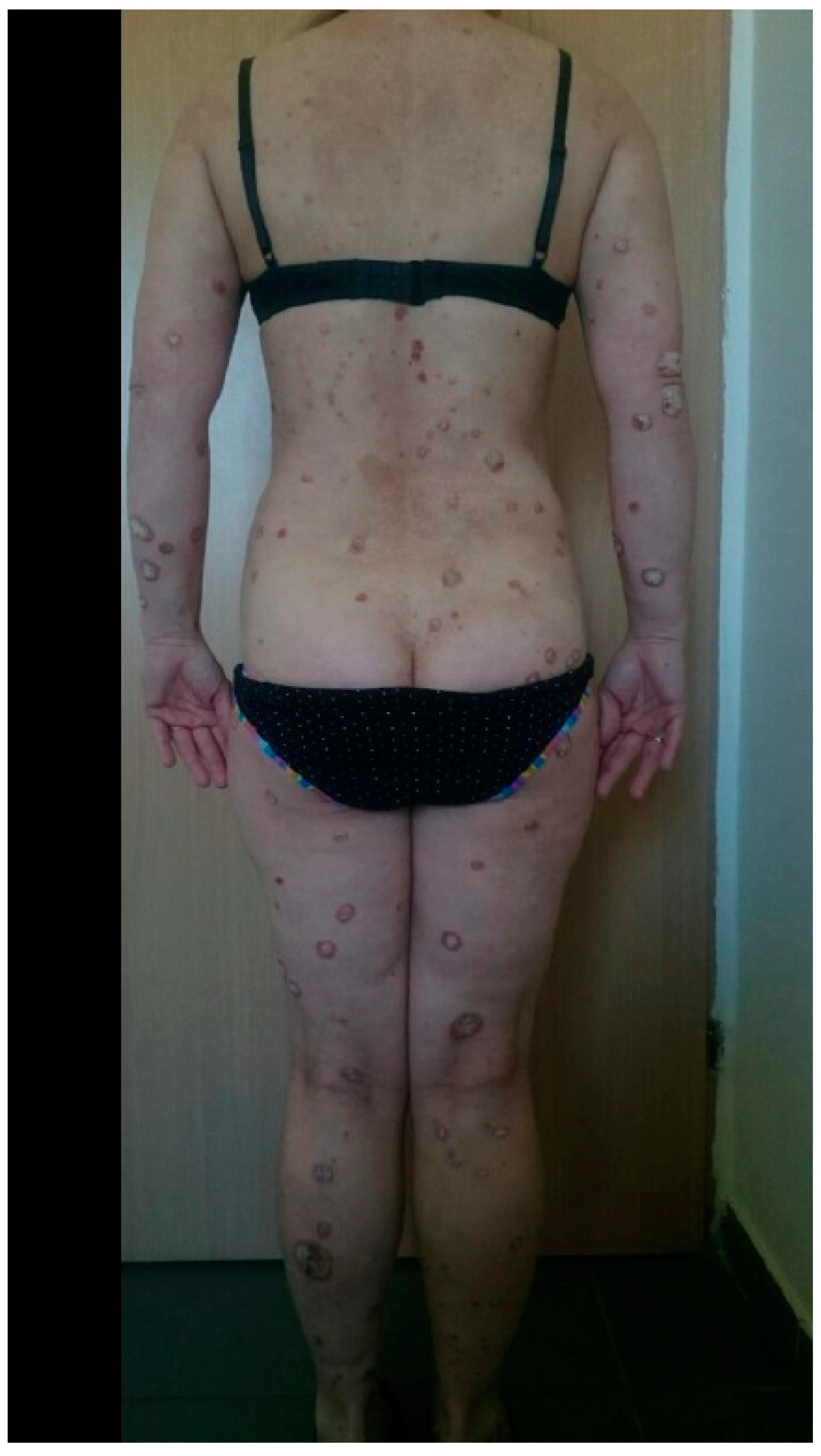
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Braun-Falco, O.; Burgdorf, W.H.C.; Plewig, G.; Wolff, H.H.; Landthaler, M. Dermatology. In Psoriasis, 3rd ed.; Springer: Heidelberg, Germany, 2009. [Google Scholar]
- Armstrong, A.W.; Read, C. Pathophysiology, Clinical Presentation, and Treatment of Psoriasis: A Review. JAMA 2020, 323, 1945–1960. [Google Scholar] [CrossRef] [PubMed]
- Hu, B.; Guo, H.; Zhou, P.; Shi, Z.L. Characteristics of SARS-CoV-2 and COVID-19. Nat. Rev. Microbiol. 2021, 19, 141–154. [Google Scholar] [CrossRef] [PubMed]
- Conforti, C.; Giuffrida, R.; Dianzani, C.; Di Meo, N.; Zalaudek, I. COVID-19 and psoriasis: Is it time to limit treatment with immunosuppressants? A call for action. Dermatol. Ther. 2020, 33, e13298. [Google Scholar] [CrossRef] [PubMed]
- Nasiri, S.; Araghi, F.; Tabary, M.; Gheisari, M.; Mahboubi-Fooladi, Z.; Dadkhahfar, S. A challenging case of psoriasis flare-up after COVID-19 infection. J. Dermatol. Treat. 2020, 31, 448–449. [Google Scholar] [CrossRef] [PubMed]
- Kutlu, Ö.; Metin, A. A case of exacerbation of psoriasis after oseltamivir and hydroxychloroquine in a patient with COVID-19: Will cases of psoriasis increase after COVID-19 pandemic? Dermatol Ther. 2020, 33, e13383. [Google Scholar] [CrossRef] [PubMed]
- Ozaras, R.; Berk, A.; Ucar, D.H.; Duman, H.; Kaya, F.; Mutlu, H. Covid-19 and exacerbation of psoriasis. Dermatol. Ther. 2020, 33, e13632. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M. Cytokine storm and immunomodulatory therapy in COVID-19: Role of chloroquine and anti-IL-6 monoclonal antibodies. Int. J. Antimicrob. Agents 2020, 55, 105982. [Google Scholar] [CrossRef] [PubMed]
- Talamonti, M.; Galluzzo, M.; Chiricozzi, A.; Quaglino, P.; Fabbrocini, G.; Gisondi, P.; Marzano, A.V.; Potenza, C.; Conti, A.; Parodi, A.; et al. Characteristic of chronic plaque psoriasis patients treated with biologics in Italy during the COVID-19 Pandemic: Risk analysis from the PSO-BIO-COVID observational study. Expert Opin. Biol. Ther. 2021, 21, 271–277. [Google Scholar] [CrossRef] [PubMed]
- Camela, E.; Fabbrocini, G.; Cinelli, E.; Lauro, W.; Megna, M. Biologic Therapies, Psoriasis i COVID-19: Our Experience at the Psoriasis Unit of the University of Naples Federico II. Dermatology 2021, 237, 13–14. [Google Scholar] [CrossRef] [PubMed]
- Mahil, S.K.; Dand, N.; Mason, K.J.; Yiu, Z.Z.; Tsakok, T.; Meynell, F.; Coker, B.; McAteer, H.; Moorhead, L.; Mackenzin, T.; et al. Factors associated with adverse COVID-19 outcomes in patients with psoriasis-insights from a global registry-based study. J Allergy Clin. Immunol. 2021, 147, 60–71. [Google Scholar] [CrossRef] [PubMed]
- Attauabi, M.; Seidelin, J.B.; Felding, O.K.; Wewer, M.D.; Arp LK, V.; Sarikaya, M.Z.; Egebergd, A.; Vladimirovae, N.; Bendtsenab, F.; Burischab, J. Coronavirus disease 2019, immune-mediated inflammatory diseases and immunosuppressive therapies—A Danish population-based cohort study. J. Autoimmun. 2021, 118, 102613. [Google Scholar] [CrossRef] [PubMed]
- Polat Ekinci, A.; Pehlivan, G.; Gökalp, M.O. Surveillance of psoriatic patients on biologic treatment during the COVID-19 pandemic: A single-center experience. Dermatol Ther. 2021, 34, e14700. [Google Scholar] [CrossRef] [PubMed]
- Mease, P.J.; Calabrese, L.H.; Duffin, K.C.; Haberman, R.H.; Firmino, R.; Scher, J.U.; Schick, L.; Winthrop, K.; Merola, J.F. Psoriasis and Psoriatic Arthritis in the Context of the COVID-19 Pandemic: A Plenary Session from the GRAPPA 2020 Annual Meeting. J. Rheumatol. 2021. [Google Scholar] [CrossRef]
- Announcement of the Polish Society of Rheumatology and the National Consultant in the Field of Rheumatology for Patients and Doctors in Connection with the Spreading COVID-19 Infection Based on the Eular Guidelines of 17/03/2020. Available online: https://www.eular.org/recommendations_management.cfm (accessed on 1 March 2021).
- Gelfand, J.M.; Armstrong, A.W.; Bell, S.; Anesi, G.L.; Blauvelt, A.; Calabrese, C.; Dommasch, E.D.; Feldman, S.R.; Gladman, D.; Kircik, L.; et al. National Psoriasis Foundation COVID-19 Task Force Guidance for Management of Psoriatic Disease during the Pandemic: Version 1. J. Am. Acad. Dermatol. 2020, 83, 1704–1716. [Google Scholar] [CrossRef] [PubMed]
- Talamonti, M.; Galluzzo, M.; Chiricozzi, A.; Quaglino, P.; Fabbrocini, G.; Gisondi, P.; Marzano, A.; Potenza, C.; Conti, A.; Parodi, A.; et al. Management of biological therapies for chronic plaque psoriasis during COVID-19 emergency in Italy. Eur. Acad. Dermatol. Venereol. 2020, 34, e770–e772. [Google Scholar] [CrossRef] [PubMed]
- Miodońska, M.; Bogacz, A.; Mróz, M.; Mućka, S.; Bożek, A. The Effect of SARS-CoV-2 Virus Infection on the Course of Atopic Dermatitis in Patients. Medicina 2021, 22, 521. [Google Scholar] [CrossRef] [PubMed]
- Kalb, R.E.; Fiorentino, D.F.; Lebwohl, M.G.; Toole, J.; Poulin, Y.; Cohen, A.D.; Goyal, K.; Fakharzadeh, S.; Calabro, S.; Chevrier, M.; et al. Risk of Serious Infection with Biologic and Systemic Treatment of Psoriasis: Results from the Psoriasis Longitudinal Assessment and Registry (PSOLAR). JAMA Dermatol. 2015, 151, 961. [Google Scholar] [CrossRef] [PubMed]
| Study Group n = 57 | Control Group n = 68 | |
|---|---|---|
| age, mean, SD (range) | 50 ± 12.34 (22–74) | 48 ± 15.21 (23–73) |
| sex | (%) | (%) |
| women | 31.58 (18) | 38.24 (26) |
| men | 68.42 (39) | 61.76 (42) |
| type of psoriasis according to ICD-10 | ||
| L.40.0 psoriasis vulgaris | 47 (82) | 54 (80) |
| L. 40.5 articular psoriasis | 10 (18) | 14 (15) |
| mean PASI before treatment | 56 ± 11 | 49 ± 16 |
| mean PASI after a year of treatment | 8 ± 6 | 10 ± 6 |
| type of systemic treatment | ||
| secukinumab | 29(51) | - |
| adalimumab | 17 (30) | - |
| ustekinumab | 8 (14) | - |
| ixekizumab | 2 (4) | - |
| risankizumab | 1 (2) | - |
| Cyclosporine a | 10 (15) | |
| Methotrexate b | 21(31) | |
| Retinoids c | 3(4) | |
| PUVA d | 20 (29) | |
| retinoids +PUVA | 10 (15) | |
| only topical treatment | 4 (6) |
| N S/C | Sex | Age | Type of Psoriasis According to ICD-10 | Drug Name | Exacerbation of Psoriasis PASI Score before vs. after | Concomitant Diseases | Time of Exacerbation (days) |
|---|---|---|---|---|---|---|---|
| 1S | M | 72 | L40.0 | adalimumab | No 32–28 | Arterial hypertension | - |
| 2S | M | 60 | L40.0 | secukinumab | No 29–31 | - | - |
| 3S | W | 50 | L40.0 | ustekinumab | Yes 4–64 | Obesity | 12 |
| 4S | M | 44 | L40.0 | ustekinumab | Yes 23–51 | - | 24 |
| 5S | W | 43 | L40.5 | adalimumab | Yes 18–33 | - | 28 |
| 6S | M | 50 | L40.0 | risankizumab | Yes 4–31 | Arterial hypertension | 28 |
| 7S | W | 52 | L40.0 | ustekinumab | Yes 8–44 | - | 21 |
| 8S | W | 52 | L40.0 | adalimumab | Yes 15–25 | - | 36 |
| 1C | M | 51 | L40.5 | cyclosporin | No 17–15 | - | - |
| 2C | M | 42 | L40.5 | PUVA | Yes 32–46 | Arterial hypertension | 24 |
| 3C | M | 45 | L40.0 | cyclosporin | Yes 28–39 | - | 38 |
| 4C | W | 62 | L40.0 | PUVA | Yes 12–54 | - | 42 |
| 5C | W | 55 | L40.0 | cyclosporin | No 19–42 | - | - |
| 6C | M | 54 | L40.5 | cyclosporin | Yes 33–63 | Arterial hypertension | 32 |
| 7C | M | 46 | L40.0 | methotrexate | No 8–7 | - | - |
| 8C | W | 69 | L40.0 | methotrexate | Yes 5–26 | Diabetes Arterial hypertension | 25 |
| 9C | M | 38 | L40.0 | cyclosporin | Yes 9–52 | - | 60 |
| 10C | M | 44 | L40.0 | PUVA | Yes 12–33 | - | 24 |
| 11C | W | 49 | L40.0 | methotrexate | Yes 25–38 | - | 32 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mroz, M.; Mućka, S.; Miodońska, M.; Ziolkowska, D.; Hadas, E.; Bożek, A. Influence of SARS-CoV-2 Virus Infection on the Course of Psoriasis during Treatment with Biological Drugs. Medicina 2021, 57, 881. https://doi.org/10.3390/medicina57090881
Mroz M, Mućka S, Miodońska M, Ziolkowska D, Hadas E, Bożek A. Influence of SARS-CoV-2 Virus Infection on the Course of Psoriasis during Treatment with Biological Drugs. Medicina. 2021; 57(9):881. https://doi.org/10.3390/medicina57090881
Chicago/Turabian StyleMroz, Magdalena, Szymon Mućka, Martyna Miodońska, Dominika Ziolkowska, Ewa Hadas, and Andrzej Bożek. 2021. "Influence of SARS-CoV-2 Virus Infection on the Course of Psoriasis during Treatment with Biological Drugs" Medicina 57, no. 9: 881. https://doi.org/10.3390/medicina57090881
APA StyleMroz, M., Mućka, S., Miodońska, M., Ziolkowska, D., Hadas, E., & Bożek, A. (2021). Influence of SARS-CoV-2 Virus Infection on the Course of Psoriasis during Treatment with Biological Drugs. Medicina, 57(9), 881. https://doi.org/10.3390/medicina57090881






