Historical Evolution of Skin Grafting—A Journey through Time
Abstract
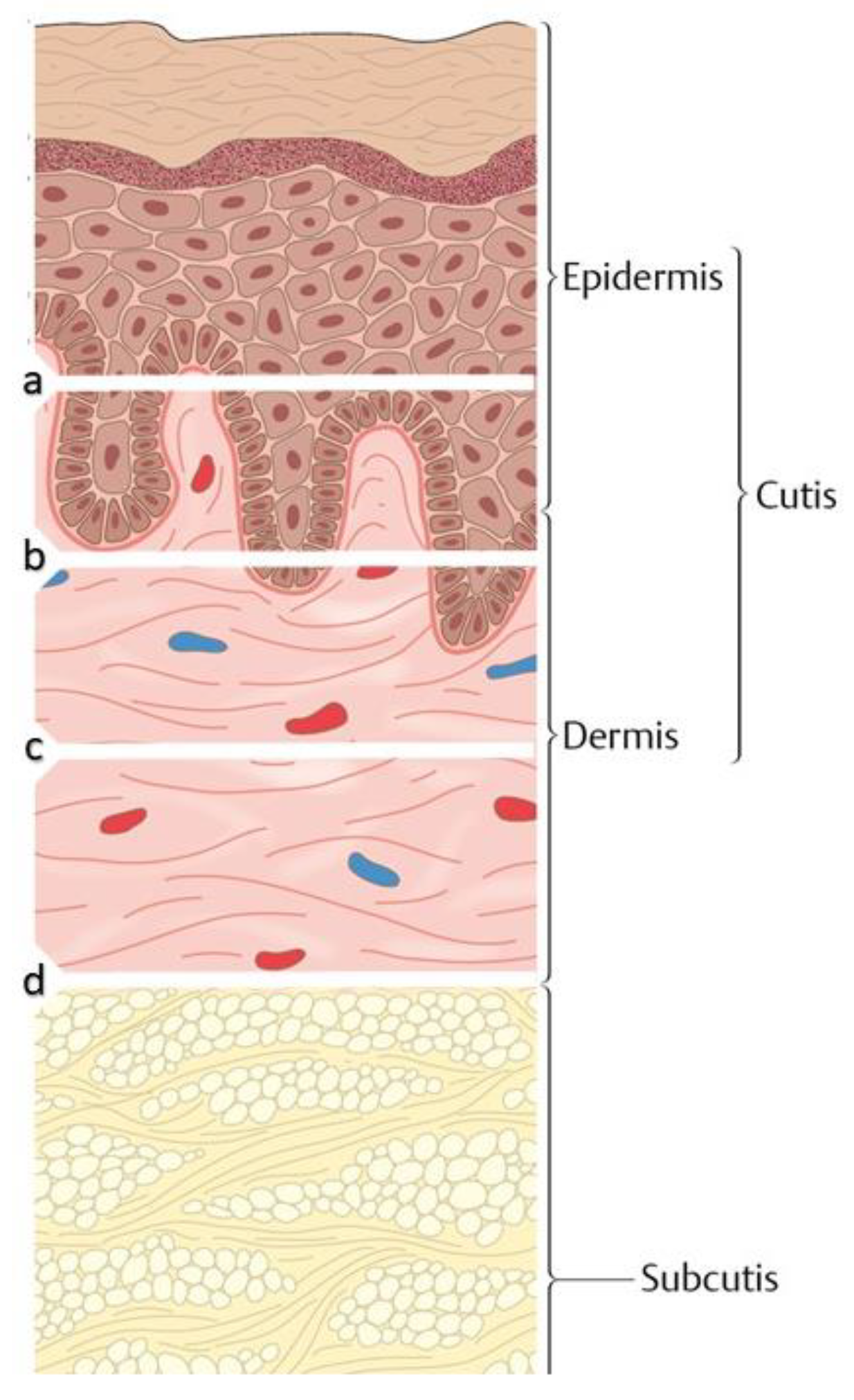
1. Introduction
2. The Origin of Skin Grafting
3. The Split-Thickness Skin Graft
3.1. Sheet Graft
3.2. Mesh Graft
3.3. Meek Technique
4. The Full-Thickness Skin Graft
5. Allogeneic and Xenogeneic Transplants
Sandwich Technique
6. A Brief History of Skin Substitutes and Their Use Today
6.1. Cell Cultures
6.2. Dermal Substitutes
6.3. Cell Suspension
7. The Curse of Donor Site Morbidity
8. The Dermis Graft—A Novel Approach
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Church, D.; Elsayed, S.; Reid, O.; Winston, B.; Lindsay, R. Burn Wound Infections. Clin. Microbiol. Rev. 2006, 19, 403–434. [Google Scholar] [CrossRef]
- Sorg, H.; Tilkorn, D.J.; Hager, S.; Hauser, J.; Mirastschijski, U. Skin Wound Healing: An Update on the Current Knowledge and Concepts. Eur. Surg. Res. 2017, 58, 81–94. [Google Scholar] [CrossRef]
- Brusselaers, N.; Pirayesh, A.; Hoeksema, H.; Richters, C.D.; Verbelen, J.; Beele, H.; Blot, S.I.; Monstrey, S. Skin Replacement in Burn Wounds. J. Trauma Inj. Infect. Crit. Care 2010, 68, 490–501. [Google Scholar] [CrossRef] [PubMed]
- Jeschke, M.G.; Shahrokhi, S.; Finnerty, C.C.; Branski, L.K.; Dibildox, M. Wound Coverage Technologies in Burn Care: Established Techniques. J. Burn. Care Res. 2018, 39, 313–318. [Google Scholar] [CrossRef] [PubMed]
- Jeschke, M.G.; Van Baar, M.E.; Choudhry, M.A.; Chung, K.K.; Gibran, N.S.; Logsetty, S. Burn injury. Nat. Rev. Dis. Prim. 2020, 6, 11. [Google Scholar] [CrossRef] [PubMed]
- Ehrenfried, A. Reverdin and Other Methods of Skin-Grafting: Historical. Boston Med Surg. J. 1909, 161, 911–917. [Google Scholar] [CrossRef]
- Ang, G.C. History of skin transplantation. Clin. Dermatol. 2005, 23, 320–324. [Google Scholar] [CrossRef]
- Hauben, D.J.; Baruchin, A.; Mahler, A. On the History of the Free Skin Graft. Ann. Plast. Surg. 1982, 9, 242–245. [Google Scholar] [CrossRef]
- Hattery, E.; Nguyen, T.; Baker, A.; Palmieri, T. Burn Care in the 1800s. J. Burn. Care Res. 2015, 36, 236–239. [Google Scholar] [CrossRef]
- McDowell, F. Successful attempt of reconstruction of a nose from a completely separated piece of skin from the leg, by Prof. Dr. Bünger, Marburg, Germany. (Journal der Chirurgie and Augenheilkunde, 4: 569, 1822). Translated from the German by Dr. Hans May. Plast. Reconstr. Surg. 1969, 44, 486–490. [Google Scholar]
- Fariña-Pérez, L.A. Jaques-Louis Reverdin (1842–1929): The surgeon and the needle. Arch. Esp. Urol. 2010, 63, 269–274. [Google Scholar] [CrossRef]
- Freshwater, M.F.; Krizek, T.J. Skin grafting of burns: A centennial. A tribute to George David Pollock. J. Trauma 1971, 11, 862–865. [Google Scholar] [CrossRef]
- Freshwater, M.F.; Krizek, T.J. George David Pollock and the Development of Skin Grafting. Ann. Plast. Surg. 1978, 1, 96–102. [Google Scholar] [CrossRef]
- Rogers, B.O. Historical Development of Free Skin Grafting. Surg. Clin. N. Am. 1959, 39, 289–311. [Google Scholar] [CrossRef]
- McDowell, F. Carl Thiersch, microscopy, and skin grafting. Plast. Reconstr. Surg. 1968, 41, 369–370. [Google Scholar] [PubMed]
- Blair, V.P.; Brown, J.B. The use and uses of large split skin grafts of intermediate thickness. Plast. Reconstr. Surg. 1968, 42, 65–75. [Google Scholar] [CrossRef]
- Padgett, E.C. Skin grafting and the “three-quarter”-thickness skin graft for prevention and correction of cicatricial, formation. Ann. Surg. 1941, 113, 1034–1049. [Google Scholar] [CrossRef] [PubMed]
- Braza, M.E.; Fahrenkopf, M.P. Split-Thickness Skin Grafts; StatPearls Publishing: Treasure Island, FL, USA, 2021. [Google Scholar] [PubMed]
- Janzekovic, Z. A new concept in the early excision and immediate grafting of burns. J. Trauma 1970, 10, 1103–1108. [Google Scholar] [CrossRef] [PubMed]
- Barret, J.P.; Dziewulski, P.; Wolf, S.E.; Desai, M.H.; Nichols, R.J.; Herndon, D.N. Effect of topical and subcutaneous epinephrine in combination with topical thrombin in blood loss during immediate near-total burn wound excision in pediatric burned patients. Burns 1999, 25, 509–513. [Google Scholar] [CrossRef]
- Masella, D.P.C.; Balent, E.M.; Carlson, D.T.L.; Lee, B.K.W.; Pierce, D.L.M. Evaluation of Six Split-thickness Skin Graft Donor-site Dressing Materials in a Swine Model. Plast. Reconstr. Surg. 2013, 1, 1–11. [Google Scholar] [CrossRef]
- Chan, Q.E.; Barzi, F.; Harvey, J.G.; Holland, A.J.A. Functional and Cosmetic Outcome of Full- Versus Split-Thickness Skin Grafts in Pediatric Palmar Surface Burns: A prospective, independent evaluation. J. Burn. Care Res. 2013, 34, 232–236. [Google Scholar] [CrossRef]
- Pripotnev, S.; Papp, A. Split thickness skin graft meshing ratio indications and common practices. Burns 2017, 43, 1775–1781. [Google Scholar] [CrossRef] [PubMed]
- Van Niekerk, G.; Adams, S.; Rode, H. Scalp as a donor site in children: Is it really the best option? Burns 2018, 44, 1259–1268. [Google Scholar] [CrossRef] [PubMed]
- Mimoun, M.; Chaouat, M.; Picovski, D.; Serroussi, D.; Smarrito, S. The Scalp Is an Advantageous Donor Site for Thin-Skin Grafts: A Report on 945 Harvested Samples. Plast. Reconstr. Surg. 2006, 118, 369–373. [Google Scholar] [CrossRef] [PubMed]
- Desai, M.H.; Herndon, D.N.; Rutan, R.L.; Parker, J. An Unusual Donor Site, a Lifesaver in Extensive Burns. J. Burn Care Rehabil. 1988, 9, 637–639. [Google Scholar] [CrossRef]
- Wyrzykowski, D.; Chrzanowska, B.; Czauderna, P. Ten years later—Scalp still a primary donor site in children. Burns 2015, 41, 359–363. [Google Scholar] [CrossRef]
- Nikkhah, D.; Booth, S.; Tay, S.; Gilbert, P.; Dheansa, B. Comparing outcomes of sheet grafting with 1:1 mesh grafting in patients with thermal burns: A randomized trial. Burns 2015, 41, 257–264. [Google Scholar] [CrossRef]
- Greenhalgh, D.G. Management of burns. N. Engl. J. Med. 2019, 380, 2349–2359. [Google Scholar] [CrossRef]
- Archer, S.B.; Henke, A.; Greenhalgh, D.G.; Warden, G.D. The Use of Sheet Autografts to Cover Extensive Burns in Patients. J. Burn. Care Rehabil. 1998, 19, 33–38. [Google Scholar] [CrossRef]
- Brown, J.B.; McDowell, F. Massive repairs of burns with thick split-skin grafts: Emergency dressings with homografts. Ann. Surg. 1942, 115, 658–674. [Google Scholar] [CrossRef]
- Clodius, L. The classic reprint. Die Transplantation Betreffend by Prof. Otto Lanz. Plast. Reconstr. Surg. 1972, 50, 395–397. [Google Scholar] [PubMed]
- Tanner, J.C.; Vandeput, J.; Olley, J.F. The mesh skin graft. Plast. Reconstr. Surg. 1964, 34, 287–292. [Google Scholar]
- Tanner, J.C.; Vandeput, J.J.; Bradley, W.H. Mesh skin grafting: Report of a typical case. J. Occup. Med. 1965, 7, 175–176. [Google Scholar] [CrossRef] [PubMed]
- Salisbury, R.B. Use of the Mesh Skin Graft in Treatment of Massive Casualty Wounds. Plast. Reconstr. Surg. 1967, 40, 161–162. [Google Scholar] [CrossRef] [PubMed]
- Macmillan, B.G. The Use of Mesh Grafting in Treating Burns. Surg. Clin. N. Am. 1970, 50, 1347–1359. [Google Scholar] [CrossRef]
- Meek, C.P. Successful microdermagrafting using the Meek-Wall microdermatome. Am. J. Surg. 1958, 96, 557–558. [Google Scholar] [CrossRef]
- Ottomann, C.; Hartmann, B.; Branski, L.; Krohn, C. A tribute to Cicero Parker Meek. Burns 2015, 41, 1660–1663. [Google Scholar] [CrossRef]
- Meek, C.P. Medical Debridement and Microdermagrafting of Burns. South. Med J. 1963, 56, 1074–1076. [Google Scholar] [CrossRef]
- Meek, C.P. Microdermagrafting: The Meek technic. Hosp. Top. 1965, 43, 114–116. [Google Scholar] [CrossRef]
- Kreis, R.; Mackie, D.; Vloemans, A.; Hermans, R.; Hoekstra, M. Widely expanded postage stamp skin grafts using a modified Meek technique in combination with an allograft overlay. Burns 1993, 19, 142–145. [Google Scholar] [CrossRef]
- Lumenta, D.B.; Kamolz, L.-P.; Frey, M. Adult Burn Patients With More Than 60% TBSA Involved–Meek and Other Techniques to Overcome Restricted Skin Harvest Availability–The Viennese Concept. J. Burn. Care Res. 2009, 30, 231–242. [Google Scholar] [CrossRef] [PubMed]
- Dahmardehei, M.; Vaghardoost, R.; Saboury, M.; Zarei, H.; Saboury, S.; Molaei, M.; Seyyedi, J.; Maleknejad, A.; Hospital, I.F. Comparison of Modified Meek Technique with Standard Mesh Method in Patients with Third Degree Burns. World J. Plast. Surg. 2020, 9, 267–273. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.Z.; Halim, A.S.; Sulaiman, W.A.W.; Saad, A.Z.M. Outcome of the Modified Meek Technique in the Management of Major Pediatric Burns. Ann. Plast. Surg. 2018, 81, 295–301. [Google Scholar] [CrossRef] [PubMed]
- Lari, A.R.; Gang, R.K. Expansion technique for skin grafts (Meek technique) in the treatment of severely burned patients. Burns 2001, 27, 61–66. [Google Scholar] [CrossRef]
- Wanjala, N.F.; Paul, O.J.; Sephania, O.R. Meek Micro-grafting Technique in Reduction of Mortality and Hospital Stay in Patients with Extensive Burns in a Resource Constrained Setting. J. Surg. 2018, 6, 154. [Google Scholar] [CrossRef]
- Sánchez-García, A.; Vanaclocha, N.; García-Vilariño, E.; Salmerón-González, E.; Vicente-Pardo, A.; Pérez-Del Caz, M.D. Use of the Meek Micrografting Technique for Coverage of Extensive Burns: A Case Report. Plast. Surg. Nurs. 2019, 39, 44–47. [Google Scholar] [CrossRef]
- Houschyar, K.S.; Tapking, C.; Nietzschmann, I.; Rein, S.; Weissenberg, K.; Chelliah, M.P.; Duscher, D.; Maan, Z.N.; Philipps, H.M.; Sheckter, C.C.; et al. Five Years Experience With Meek Grafting in the Management of Extensive Burns in an Adult Burn Center. Plast. Surg. 2018, 27, 44–48. [Google Scholar] [CrossRef]
- Lee, S.Z.; Halim, A.S. Superior long term functional and scar outcome of Meek micrografting compared to conventional split thickness skin grafting in the management of burns. Burns 2019, 45, 1386–1400. [Google Scholar] [CrossRef]
- Munasinghe, N.; Wasiak, J.; Ives, A.; Cleland, H.; Lo, C.H. Retrospective review of a tertiary adult burn centre’s experience with modified Meek grafting. Burn. Trauma 2016, 4, 6. [Google Scholar] [CrossRef]
- Kilner, T.P. The Full-Thickness Skin Graft. Postgrad. Med J. 1935, 11, 279–282. [Google Scholar] [CrossRef][Green Version]
- Padgett, E.C. Indications for determination of the thickness of split skin grafts. Am. J. Surg. 1946, 72, 683–693. [Google Scholar] [CrossRef]
- Çeliköz, B.; Deveci, M.; Duman, H.; Nişanci, M. Recontruction of facial defects and burn scars using large size freehand full-thickness skin graft from lateral thoracic region. Burns 2001, 27, 174–178. [Google Scholar] [CrossRef]
- Somma, A.M.; Somma, L.M. John Reissberg Wolfe (1823–1904): A plastic surgeon in Garibaldi’s Army. J. Med Biogr. 2010, 18, 77–80. [Google Scholar] [CrossRef] [PubMed]
- Sykes, P.J. Wolfe’s Part in the Italian Risorgimento and His Skin Graft. Ann. Plast. Surg. 2012, 69, 228–231. [Google Scholar] [CrossRef] [PubMed]
- Bogdanov, S.B.; Gilevich, I.V.; Melkonyan, K.I.; Sotnichenko, A.S.; Alekseenko, S.N.; Porhanov, V.A. Total full-thickness skin grafting for treating patients with extensive facial burn injury: A 10-year experience. Burns 2020. [Google Scholar] [CrossRef]
- Weeks, D.; Kasdan, M.L.; Wilhelmi, B.J. Forty-Year Follow-up of Full-Thickness Skin Graft After Thermal Burn Injury to the Volar Hand. Eplasty 2016, 16, e21. [Google Scholar]
- Merrell, S.W.; Saffle, J.R.; Schnebly, W.A.; Kravitz, M.; Warden, G.D. Full-Thickness Skin Grafting for Contact Burns of the Palm in Children. J. Burn. Care Rehabil. 1986, 7, 501–507. [Google Scholar] [CrossRef]
- Seghers, M.J.; Longacre, J.J. Paul Bert and his animal grafts. Plast. Reconstr. Surg. 1964, 33, 178–186. [Google Scholar] [CrossRef]
- Cooper, D.K.; Ekser, B.; Tector, A.J. A brief history of clinical xenotransplantation. Int. J. Surg. 2015, 23 Pt B, 205–210. [Google Scholar] [CrossRef]
- Bromberg, B.E.; Song, I.C.; Mohn, M.P. The use of pig skin as a temporary biological dressing. Plast. Reconstr. Surg. 1965, 36, 80–90. [Google Scholar] [CrossRef]
- Yamamoto, T.; Iwase, H.; King, T.W.; Hara, H.; Cooper, D.K. Skin xenotransplantation: Historical review and clinical potential. Burns 2018, 44, 1738–1749. [Google Scholar] [CrossRef]
- Rowan, M.P.; Cancio, L.C.; Elster, E.A.; Burmeister, D.M.; Rose, L.F.; Natesan, S.; Chan, R.K.; Christy, R.J.; Chung, K.K. Burn wound healing and treatment: Review and advancements. Crit. Care 2015, 19, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Zhang, F.; Lineaweaver, W.C. Clinical Applications of Allograft Skin in Burn Care. Ann. Plast. Surg. 2020, 84, S158–S160. [Google Scholar] [CrossRef] [PubMed]
- Blome-Eberwein, S.; Jester, A.; Kuentscher, M.; Raff, T.; Germann, G.; Pelzer, M. Clinical practice of glycerol preserved allograft skin coverage. Burns 2002, 28, 10–12. [Google Scholar] [CrossRef]
- Hoekstra, M.J.; Kreis, R.W.; du Pont, J.S. History of the Euro Skin Bank: The innovation of preservation technologies. Burns 1994, 20, S43–S47. [Google Scholar] [CrossRef]
- Hermans, M.H. Preservation methods of allografts and their (lack of) influence on clinical results in partial thickness burns. Burns 2011, 37, 873–881. [Google Scholar] [CrossRef]
- Kua, E.H.J.; Goh, C.Q.; Ting, Y.; Chua, A.; Song, C. Comparing the use of glycerol preserved and cryopreserved allogenic skin for the treatment of severe burns: Differences in clinical outcomes and in vitro tissue viability. Cell Tissue Bank. 2012, 13, 269–279. [Google Scholar] [CrossRef] [PubMed]
- Debeer, S.; Le Luduec, J.-B.; Kaiserlian, D.; Laurent, P.; Nicolas, J.-F.; Dubois, B.; Kanitakis, J. Comparative histology and immunohistochemistry of porcine versus human skin. Eur. J. Dermatol. 2013, 23, 456–466. [Google Scholar] [CrossRef]
- Sykes, D.S. Transplanting organs from pigs to humans Megan. Physiol. Behav. 2016, 176, 100–106. [Google Scholar] [CrossRef]
- Júnior, E.M.L.; Filho, M.O.D.M.; Costa, B.A.; Rohleder, A.V.P.; Rocha, M.B.S.; Fechine, F.V.; Forte, A.J.; Alves, A.P.N.N.; Júnior, F.R.S.; Martins, C.B.; et al. Innovative Burn Treatment Using Tilapia Skin as a Xenograft: A Phase II Randomized Controlled Trial. J. Burn. Care Res. 2020, 41, 585–592. [Google Scholar] [CrossRef]
- Alam, K.; Jeffery, S.L. Acellular Fish Skin Grafts for Management of Split Thickness Donor Sites and Partial Thickness Burns: A Case Series. Mil. Med. 2019, 184, 16–20. [Google Scholar] [CrossRef]
- Costa, B.A.; Júnior, E.M.L. Use of Tilapia Skin as a Xenograft for Pediatric Burn Treatment: A Case Report. J. Burn Care Res. 2020, 40, 714–717. [Google Scholar] [CrossRef] [PubMed]
- Alexander, J.W.; Macmillan, B.G.; Law, E.; Kittur, D.S. Treatment of severe burns with widely meshed skin autograft and meshed skin allograft overlay. J. Trauma 1981, 21, 433–438. [Google Scholar] [PubMed]
- Phipps, A.R.; Clarke, J.A. The use of intermingled autograft and parental allograft skin in the treatment of major burns in children. Br. J. Plast. Surg. 1991, 44, 608–611. [Google Scholar] [CrossRef]
- Qaryoute, S.; Mirdad, I.; Hamail, A. Usage of autograft and allograft skin in treatment of burns in children. Burns 2001, 27, 599–602. [Google Scholar] [CrossRef]
- Horch, R.E.; Stark, G.; Kopp, J.; Spilker, G. Cologne Burn Centre experiences with glycerol-preserved allogeneic skin: Part I: Clinical experiences and histological findings (overgraft and sandwich technique). Burns 1994, 20, S23–S26. [Google Scholar] [CrossRef]
- Rheinwatd, J.G.; Green, H. Serial cultivation of strains of human epidermal keratinocytes: The formation of keratinizing colonies from single cells. Cell 1975, 6, 331–343. [Google Scholar] [CrossRef]
- O’Connor, N.; Mulliken, J.; Banks-Schlegel, S.; Kehinde, O.; Green, H. Grafting of burns with cultured epithelium prepared from autologous epidermal cells. Lancet 1981, 317, 75–78. [Google Scholar] [CrossRef]
- Barret, J.P.; Wolf, S.E.; Desai, M.H.; Herndon, D.N. Cost-Efficacy of Cultured Epidermal Autografts in Massive Pediatric Burns. Ann. Surg. 2000, 231, 869–876. [Google Scholar] [CrossRef]
- Matsumura, H.; Matsushima, A.; Ueyama, M.; Kumagai, N. Application of the cultured epidermal autograft “JACE®” for treatment of severe burns: Results of a 6-year multicenter surveillance in Japan. Burns 2016, 42, 769–776. [Google Scholar] [CrossRef] [PubMed]
- Ottomann, C.; Küntscher, M.V.; Hartmann, B. The Combination of Cultured Epidermal Autograft (CEA) and Split Thickness Skin Graft Technique (Meek) in Therapy for Severe Burns. Osteosynth. Trauma Care 2007, 15, 29–33. [Google Scholar] [CrossRef]
- Menon, S.; Li, Z.; Harvey, J.G.; Holland, A.J. The use of the Meek technique in conjunction with cultured epithelial autograft in the management of major paediatric burns. Burns 2013, 39, 674–679. [Google Scholar] [CrossRef] [PubMed]
- Akita, S.; Hayashida, K.; Yoshimoto, H.; Fujioka, M.; Senju, C.; Morooka, S.; Nishimura, G.; Mukae, N.; Kobayashi, K.; Anraku, K.; et al. Novel Application of Cultured Epithelial Autografts (CEA) with Expanded Mesh Skin Grafting Over an Artificial Dermis or Dermal Wound Bed Preparation. Int. J. Mol. Sci. 2018, 19, 57. [Google Scholar] [CrossRef] [PubMed]
- Dimitropoulos, G.; Jafari, P.; de Buys Roessingh, A.; Hirt-Burri, N.; Raffoul, W.; Applegate, L.A. Burn patient care lost in good manufacturing practices? Ann. Burn. Fire Disasters 2016, 29, 111–115. [Google Scholar]
- Abdel-Sayed, P.; Michetti, M.; Scaletta, C.; Flahaut, M.; Hirt-Burri, N.; Roessingh, A.D.B.; Raffoul, W.; Applegate, L.A. Cell therapies for skin regeneration: An overview of 40 years of experience in burn units. Swiss Med Wkly. 2019, 149, w20079. [Google Scholar] [CrossRef]
- Gardien, K.L.M.; Marck, R.E.; Bloemen, M.C.T.; Waaijman, T.; Gibbs, S.; Ulrich, M.M.W.; Middelkoop, E. Outcome of Burns Treated with Autologous Cultured Proliferating Epidermal Cells: A Prospective Randomized Multicenter Intrapatient Comparative Trial. Cell Transplant. 2016, 25, 437–448. [Google Scholar] [CrossRef]
- Arno, A.I.; Jeschke, M.G. The Use of Dermal Substitutes in Burn Surgery: Acute Phase. Dermal Replace. Gen. Burn Plast. Surg. Tissue Eng. Clin. Pr. 2014, 9783709115, 193–210. [Google Scholar] [CrossRef]
- Yannas, I.V.; Burke, J.F. Design of an artificial skin. I. Basic design principles. J. Biomed. Mater. Res. 1980, 14, 65–81. [Google Scholar] [CrossRef]
- Yannas, I.V.; Burke, J.F.; Gordon, P.L.; Huang, C.; Rubenstein, R.H. Design of an artificial skin. II. Control of chemical composition. J. Biomed. Mater. Res. 1980, 14, 107–132. [Google Scholar] [CrossRef]
- Heimbach, D.M.; Warden, G.D.; Luterman, A.; Jordan, M.H.; Ozobia, N.; Ryan, C.M.; Voigt, D.W.; Hickerson, W.L.; Saffle, J.R.; Declement, F.A.; et al. Multicenter Postapproval Clinical Trial of Integra® Dermal Regeneration Template for Burn Treatment. J. Burn. Care Rehabil. 2003, 24, 42–48. [Google Scholar] [CrossRef]
- Branski, L.K.; Herndon, D.N.; Pereira, C.; Mlcak, R.P.; Celis, M.M.; Lee, J.O.; Sanford, A.P.; Norbury, W.B.; Zhang, X.-J.; Jeschke, M.G. Longitudinal assessment of Integra in primary burn management: A randomized pediatric clinical trial. Crit. Care Med. 2007, 35, 2615–2623. [Google Scholar] [CrossRef] [PubMed]
- Danin, A.; Georgesco, G.; Le Touze, A.; Penaud, A.; Quignon, R.; Zakine, G. Assessment of burned hands reconstructed with Integra® by ultrasonography and elastometry. Burns 2012, 38, 998–1004. [Google Scholar] [CrossRef]
- Cuadra, Á.; Correa, G.; Roa, R.; Piñeros, J.L.; Norambuena, H.; Searle, S.; Heras, R.L.; Calderon, W. Functional results of burned hands treated with Integra®. J. Plast. Reconstr. Aesthetic Surg. 2012, 65, 228–234. [Google Scholar] [CrossRef]
- Koenen, W.; Felcht, M.; Vockenroth, K.; Sassmann, G.; Goerdt, S.; Faulhaber, J. One-stage reconstruction of deep facial defects with a single layer dermal regeneration template. J. Eur. Acad. Dermatol. Venereol. 2010, 25, 788–793. [Google Scholar] [CrossRef] [PubMed]
- Jackson, S.R.; Roman, S. Matriderm and Split Skin Grafting for Full-Thickness Pediatric Facial Burns. J. Burn Care Res. 2019, 40, 251–254. [Google Scholar] [CrossRef]
- Phillips, G.S.A.; Nizamoglu, M.; Wakure, A.; Barnes, D.; Dziewulski, P. The use of dermal regeneration templates for primary burns surgery in a UK regional burns centre. Ann. Burn. Fire Disasters 2020, XXXIII, 245–252. [Google Scholar]
- Ryssel, H.; Gazyakan, E.; Germann, G.; Öhlbauer, M. The use of MatriDerm® in early excision and simultaneous autologous skin grafting in burns—A pilot study. Burns 2008, 34, 93–97. [Google Scholar] [CrossRef]
- Ryssel, H.; Germann, G.; Kloeters, O.; Gazyakan, E.; Radu, C. Dermal substitution with Matriderm® in burns on the dorsum of the hand. Burns 2010, 36, 1248–1253. [Google Scholar] [CrossRef]
- Greenhalgh, D.G.; Hinchcliff, K.; Sen, S.; Palmieri, T.L. A Ten-Year Experience with Pediatric Face Grafts. J. Burn. Care Res. 2010, 34, 576–584. [Google Scholar] [CrossRef] [PubMed]
- Graça, M.F.; Miguel, S.P.; Cabral, C.S.; Correia, I.J. Hyaluronic acid—Based wound dressings: A review. Carbohydr. Polym. 2020, 241, 116364. [Google Scholar] [CrossRef]
- Longinotti, C. The use of hyaluronic acid based dressings to treat burns: A review. Burn. Trauma 2014, 2, 162–168. [Google Scholar] [CrossRef] [PubMed]
- Gravante, G.; Sorge, R.; Merone, A.; Tamisani, A.M.; Di Lonardo, A.; Scalise, A.; Doneddu, G.; Melandri, D.; Stracuzzi, G.; Onesti, M.G.; et al. Hyalomatrix PA in Burn Care Practice: Results from a national retrospective survey, 2005 to 2006. Ann. Plast. Surg. 2010, 64, 69–79. [Google Scholar] [CrossRef]
- Erbatur, S.; Coban, Y.K.; Aydın, E.N. Comparision of clinical and histopathological results of hyalomatrix usage in adult patients. Int. J. Burn. Trauma 2012, 2, 118–125. [Google Scholar]
- Gravante, G.; Delogu, D.; Giordan, N.; Morano, G.; Montone, A.; Esposito, G. The Use of Hyalomatrix PA in the Treatment of Deep Partial-Thickness Burns. J. Burn. Care Res. 2007, 28, 269–274. [Google Scholar] [CrossRef] [PubMed]
- Faga, A.; Nicoletti, G.; Brenta, F.; Scevola, S.; Abatangelo, G.; Brun, P. Hyaluronic acid three-dimensional scaffold for surgical revision of retracting scars: A human experimental study. Int. Wound J. 2013, 10, 329–335. [Google Scholar] [CrossRef] [PubMed]
- Mangoldt, F. Die Epithelsaat zum Verschlußeiner großen Wundfläche. Med. Wochenschr 1895, 21, 798–903. [Google Scholar] [CrossRef][Green Version]
- Hunyadi, J.; Farkas, B.; Bertényi, C.; Oláh, J.; Dobozy, A. Keratinocyte Grafting: Covering of Skin Defects by Separated Autologous Keratinocytes in a Fibrin Net. J. Investig. Dermatol. 1987, 89, 119–120. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Holmes, J.; Molnar, J.; Shupp, J.; Hickerson, W.; King, B.T.; Foster, K.; Cairns, B.; Carter, J. Demonstration of the safety and effectiveness of the RECELL® System combined with split-thickness meshed autografts for the reduction of donor skin to treat mixed-depth burn injuries. Burns 2019, 45, 772–782. [Google Scholar] [CrossRef] [PubMed]
- Gravante, G.; Di Fede, M.; Araco, A.; Grimaldi, M.; De Angelis, B.; Arpino, A.; Cervelli, V.; Montone, A. A randomized trial comparing ReCell® system of epidermal cells delivery versus classic skin grafts for the treatment of deep partial thickness burns. Burns 2007, 33, 966–972. [Google Scholar] [CrossRef]
- Iv, J.H.H.; Molnar, J.; Carter, J.; Hwang, J.; Cairns, B.; King, B.T.; Smith, D.J.; Cruse, C.W.; Foster, K.N.; Peck, M.D.; et al. A Comparative Study of the ReCell® Device and Autologous Split-Thickness Meshed Skin Graft in the Treatment of Acute Burn Injuries. J. Burn. Care Res. 2018, 39, 694–702. [Google Scholar] [CrossRef]
- Otene, C.; Olaitan, P.B.; Ogbonnaya, I.S.; Nnabuko, R. Donor site morbidity following harvest of split-thickness skin grafts in south eastern nigeria. J. West Afr. Coll. Surg. 2016, 1, 86–96. [Google Scholar]
- Asuku, M.; Yan, Q.; Yu, T.-C.; Boing, E.; Hahn, H.; Hovland, S.; Donelan, M.B. 522 Skin Graft Donor-site Morbidity: A Systematic Literature Review. J. Burn. Care Res. 2020, 41, S98–S99. [Google Scholar] [CrossRef]
- Rotatori, R.M.; Starr, B.; Peake, M.; Fowler, L.; James, L.; Nelson, J.; Dale, E.L. Prevalence and Risk Factors for Hypertrophic Scarring of Split Thickness Autograft Donor Sites in a Pediatric Burn Population. Burns 2019, 45, 1066–1074. [Google Scholar] [CrossRef] [PubMed]
- Karlsson, M.; Elmasry, M.; Steinvall, I.; Sjöberg, F.; Olofsson, P.; Thorfinn, J. Scarring At Donor Sites After Split-Thickness Skin Graft: A Prospective, Longitudinal, Randomized Trial. Adv. Ski. Wound Care 2018, 31, 183–188. [Google Scholar] [CrossRef]
- Legemate, C.M.; Ooms, P.J.; Trommel, N.; Middelkoop, E.; van Baar, M.E.; Goei, H.; van der Vlies, C.H. Patient-reported scar quality of donor-sites following split-skin grafting in burn patients: Long-term results of a prospective cohort study. Burns 2021, 47, 315–321. [Google Scholar] [CrossRef] [PubMed]
- Bradow, B.P.; Hallock, G.G.; Wilcock, S.P. Immediate Regrafting of the Split Thickness Skin Graft Donor Site Assists Healing. Plast. Reconstr. Surg. Glob. Open 2017, 5, e1339. [Google Scholar] [CrossRef]
- Goverman, J.; Kraft, C.T.; Fagan, S.; Levi, B. Back Grafting the Split-Thickness Skin Graft Donor Site. J. Burn. Care Res. 2017, 38, e443–e449. [Google Scholar] [CrossRef]
- Bian, Y.; Sun, C.; Zhang, X.; Li, Y.; Li, W.; Lv, X.; Li, J.; Jiang, L.; Li, J.; Feng, J.; et al. Wound-healing improvement by resurfacing split-thickness skin donor sites with thin split-thickness grafting. Burns 2016, 42, 123–130. [Google Scholar] [CrossRef]
- Legemate, C.M.; Lucas, Y.; Oen, I.M.M.H.; Van Der Vlies, C.H. Regrafting of the Split-Thickness Skin Graft Donor-Site: Is It Beneficial? J. Burn. Care Res. 2020, 41, 211–214. [Google Scholar] [CrossRef]
- Miyanaga, T.; Kishibe, M.; Yamashita, M.; Kaneko, T.; Kinoshita, F.; Shimada, K. Minced Skin Grafting for Promoting Wound Healing and Improving Donor-Site Appearance after Split-Thickness Skin Grafting: A Prospective Half-Side Comparative Trial. Plast. Reconstr. Surg. 2019, 144, 475–483. [Google Scholar] [CrossRef]
- Kumar, P.; Ajai, K.S.; Sharma, R.K. The role of recruited minced skin grafting in improving the quality of healing at the donor site of split-thickness skin graft—A comparative study. Burns 2019, 45, 923–928. [Google Scholar] [CrossRef]
- Miyanaga, T.; Haseda, Y.; Sakagami, A. Minced skin grafting for promoting epithelialization of the donor site after split-thickness skin grafting. Burns 2017, 43, 819–823. [Google Scholar] [CrossRef] [PubMed]
- Han, S.-K.; Yoon, T.-H.; Kim, J.-B.; Kim, W.-K. Dermis Graft for Wound Coverage. Plast. Reconstr. Surg. 2007, 120, 166–172. [Google Scholar] [CrossRef] [PubMed]
- Lindford, A.J.; Kaartinen, I.S.; Virolainen, S.; Kuokkanen, H.O.; Vuola, J. The dermis graft: Another autologous option for acute burn wound coverage. Burns 2012, 38, 274–282. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kohlhauser, M.; Luze, H.; Nischwitz, S.P.; Kamolz, L.P. Historical Evolution of Skin Grafting—A Journey through Time. Medicina 2021, 57, 348. https://doi.org/10.3390/medicina57040348
Kohlhauser M, Luze H, Nischwitz SP, Kamolz LP. Historical Evolution of Skin Grafting—A Journey through Time. Medicina. 2021; 57(4):348. https://doi.org/10.3390/medicina57040348
Chicago/Turabian StyleKohlhauser, Michael, Hanna Luze, Sebastian Philipp Nischwitz, and Lars Peter Kamolz. 2021. "Historical Evolution of Skin Grafting—A Journey through Time" Medicina 57, no. 4: 348. https://doi.org/10.3390/medicina57040348
APA StyleKohlhauser, M., Luze, H., Nischwitz, S. P., & Kamolz, L. P. (2021). Historical Evolution of Skin Grafting—A Journey through Time. Medicina, 57(4), 348. https://doi.org/10.3390/medicina57040348






