Technical and Medical Aspects of Burn Size Assessment and Documentation
Abstract
1. Introduction
2. Consequences of Inaccurate TBSA-B Assessment
2.1. Over-Resuscitation
2.2. Complications Because of Fluid Aggregation from Burn Edema
2.3. Missing Accuracy in Studies
2.4. Non-Optimal Distribution of Patients
2.4.1. Distribution to Burn Centers
2.4.2. Distribution of Patients in Mass Casualty Situations
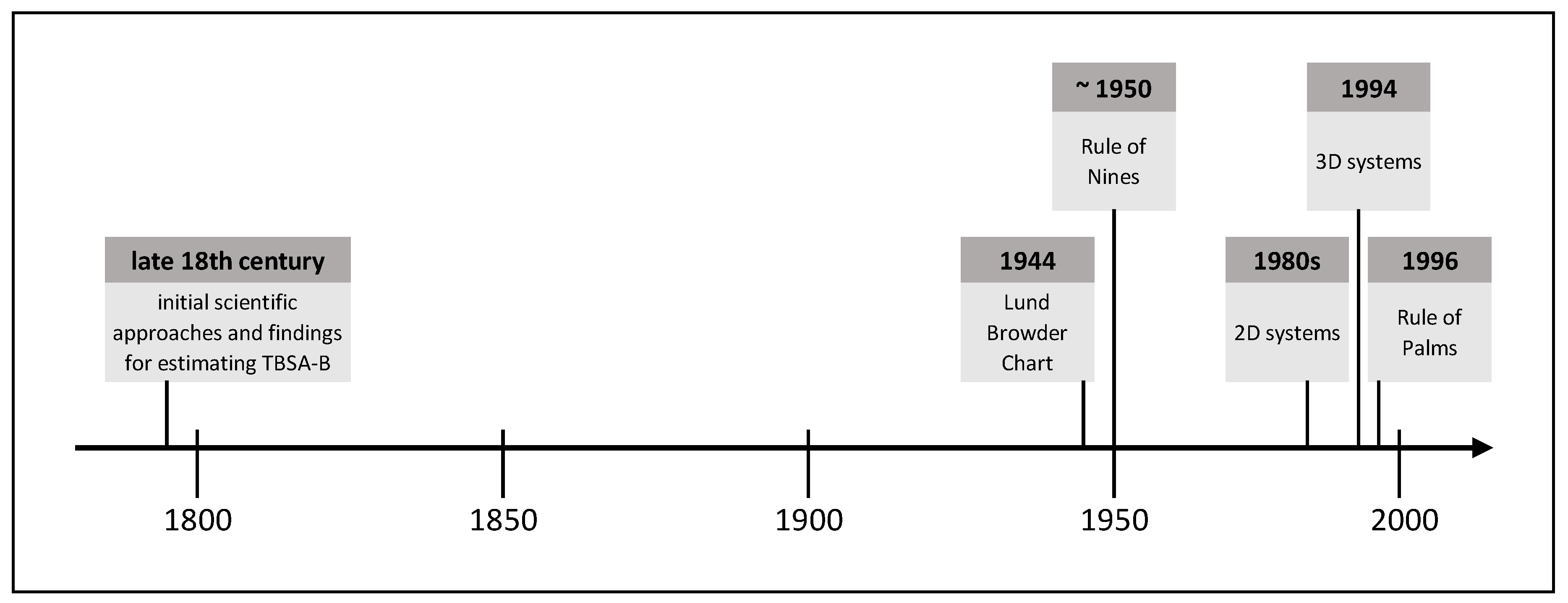
3. Methods for Burn Size Estimation
3.1. Initial Scientific Approaches and Findings for Estimating TBSA-B
3.2. Lund Browder Chart
3.2.1. Description
3.2.2. Estimation Accuracy and Criticism
3.3. Rule of Nines
3.3.1. Description
3.3.2. Estimation Accuracy and Criticism
3.4. Rule of Palms
3.4.1. Description
3.4.2. Estimation Accuracy and Criticism
3.5. Two-Dimensional Computer-Aided Systems
3.5.1. Description
3.5.2. Sample Applications
3.5.3. Quality of Estimation Reliability
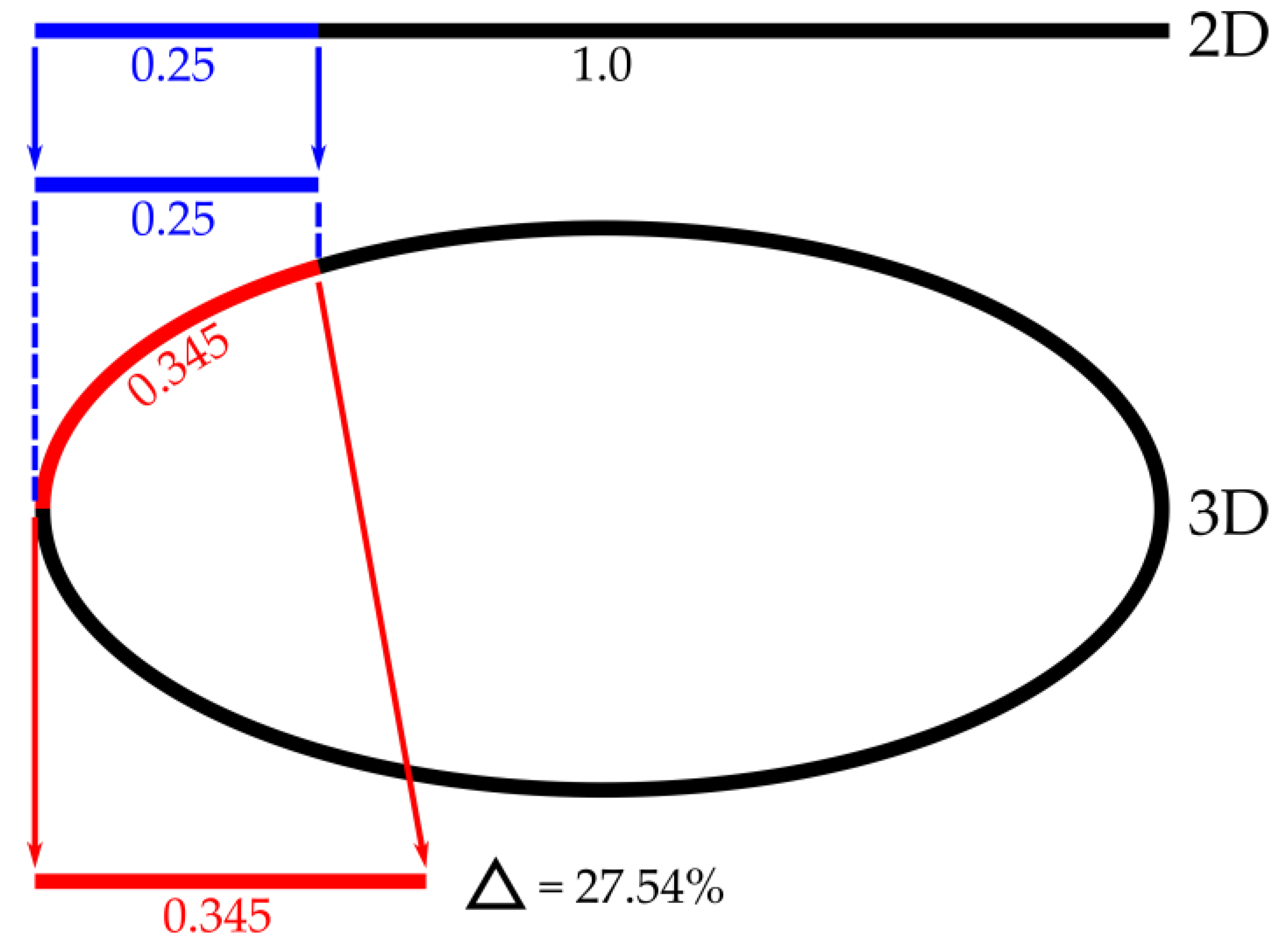
3.6. Three-Dimensional Computer-Aided Systems
3.6.1. Three-Dimensional Images
3.6.2. Three-Dimensional Scans
3.6.3. Sample Applications
3.6.4. Quality of Estimation Reliability
4. Documentation
4.1. Medical Documentation
- Medical history and general status of the patient with all its features;
- Recent and frequent photographic documentation to evaluate changes in the wound;
- Wound assessment with all its features;
- Course of healing;
- Documentation of therapeutic measures and their efficacy;
- Results of follow-ups;
- Traceability and verification of authors.
4.2. Standards Required for Data Analysis
4.3. Existing Documentation Systems
4.3.1. Electronic Documentation
4.3.2. Mobile Documentation
4.3.3. Photo Documentation
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
Abbreviations
| 2D | two-dimensional |
| 3D | three-dimensional |
| ABA | American Burn Association |
| ANP | arterial natriuretic peptide |
| BMI | body mass index |
| CVP | central venous pressure |
| EPRI | Electric Power Research Institute |
| FDA | Food and Drug Administration |
| ISBI | International Society for Burn Injuries |
| IT | information technology |
| MODS | multiple organ dysfunction syndromes |
| NIH | National Institutes of Health (United States) |
| OBOS | One Burn One Standard |
| ODBC | Committee for the Organization and Delivery of Burn Care |
| PACS | picture archiving and communication system |
| RISC | Research Institute for Symbolic Computation |
| SAGE II | Surface Area Graphic Evaluation II |
| TBSA | total body surface area |
| TBSA-B | percentage of the total body surface area burned |
References
- Deutsche Gesellschaft für Verbrennungsmedizin (DGV). Leitlinie Behandlung Thermischer Verletzungen des Erwachsenen. Klasse: S2k. AWMF-Register-Nr.: 044-001 2018. Available online: https://www.verbrennungsmedizin.de/files/dgv_files/pdf/leitlinien/044-001l_S2k_Thermische__Verletzungen_Erwachsene_2018-10.pdf#page=4 (accessed on 30 November 2020).
- Haller, H. Verbrennungstiefe und Ausmaß. In Verbrennungen; Springer: Berlin/Heidelberg, Germany, 2009; pp. 159–167. [Google Scholar]
- Monstrey, S.; Hoeksema, H.; Verbelen, J.; Pirayesh, A.; Blondeel, P. Assessment of Burn Depth and Burn Wound Healing Potential. Burns 2008, 34, 761–769. [Google Scholar] [CrossRef]
- Underhill, F.P. The Significance of Anhydremia in Extensive Superficial Burns. JAMA 1930, 95, 852. [Google Scholar] [CrossRef]
- Baxter, C.R.; Shires, T. Physiological response to crystalloid resuscitation of severe burns. Ann. N. Y. Acad. Sci. 1968, 150, 874–894. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, S.I. Supportive Therapy in Burn Care. Consensus Summary on Fluid Resuscitation. J. Trauma 1979, 19, 876–877. [Google Scholar]
- Velmahos, G.C.; Demetriades, D.; Shoemaker, W.C.; Chan, L.S.; Tatevossian, R.; Wo, C.C.; Vassiliu, P.; Cornwell, E.E.; Murray, J.A.; Roth, B.; et al. Endpoints of Resuscitation of Critically Injured Patients: Normal or Supranormal? Ann. Surg. 2000, 232, 409–418. [Google Scholar] [CrossRef]
- Rhee, P. Shock, Electrolytes, and Fluid. In Textbook of Oral and Maxillofacial Surgery, 19th ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2012; ISBN 9781437715606. [Google Scholar]
- Bruegger, D.; Schwartz, L.; Chappell, D.; Jacob, M.; Rehm, M.; Vogeser, M.; Christ, F.; Reichart, B.; Becker, B.F. Release of Atrial Natriuretic Peptide Precedes Shedding of the Endothelial Glycocalyx Equally in Patients Undergoing On- and off-Pump Coronary Artery Bypass Surgery. Basic Res. Cardiol. 2011, 106, 1111–1121. [Google Scholar] [CrossRef] [PubMed]
- Jacob, M.; Chappell, D. Mythen Und Fakten Der Perioperativen Infusionstherapie. Deutsch 2009, 358–376. [Google Scholar]
- Lobo, D.N.; Stanga, Z.; Aloysius, M.M.; Wicks, C.; Nunes, Q.M.; Ingram, K.L.; Risch, L.; Allison, S.P. Effect of Volume Loading with 1 Liter Intravenous Infusions of 0.9% Saline, 4% Succinylated Gelatine (Gelofusine) and 6% Hydroxyethyl Starch (Voluven) on Blood Volume and Endocrine Responses: A Randomized, Three-Way Crossover Study in Healthy Volunteers. Crit. Care Med. 2010, 38, 464–470. [Google Scholar] [CrossRef] [PubMed]
- Chung, K.K.; Wolf, S.E.; Cancio, L.C.; Alvarado, R.; Jones, J.A.; McCorcle, J.; King, B.T.; Barillo, D.J.; Renz, E.M.; Blackbourne, L.H. Resuscitation of Severely Burned Military Casualties: Fluid Begets More Fluid. J. Trauma Inj. Infect. Crit. Care 2009, 67, 231–237. [Google Scholar] [CrossRef] [PubMed]
- Cancio, L.C.; Chávez, S.; Alvarado-Ortega, M.; Barillo, D.J.; Walker, S.C.; McManus, A.T.; Goodwin, C.W. Predicting Increased Fluid Requirements during the Resuscitation of Thermally Injured Patients. J. Trauma 2004, 56, 404–413. [Google Scholar] [CrossRef]
- Friedrich, J.B.; Sullivan, S.R.; Engrav, L.H.; Round, K.A.; Blayney, C.B.; Carrougher, G.J.; Heimbach, D.M.; Honari, S.; Klein, M.B.; Gibran, N.S. Is Supra-Baxter Resuscitation in Burn Patients a New Phenomenon? Burns 2004, 30, 464–466. [Google Scholar] [CrossRef] [PubMed]
- Engrav, L.H.; Heimbach, D.M.; Rivara, F.P.; Kerr, K.F.; Osler, T.; Pham, T.N.; Sharar, S.R.; Esselman, P.C.; Bulger, E.M.; Carrougher, G.J.; et al. Harborview Burns—1974 to 2009. PLoS ONE 2012, 7, e40086. [Google Scholar] [CrossRef] [PubMed]
- Regan, A.; Hotwagner, D.T. Burn Fluid Management. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2020. [Google Scholar]
- Cartotto, R.; Zhou, A. Fluid Creep: The Pendulum Hasn’t Swung Back Yet! J. Burn Care Res. 2010, 31, 551–558. [Google Scholar] [CrossRef]
- Strang, S.G.; Van Lieshout, E.M.M.; Breederveld, R.S.; Van Waes, O.J.F. A Systematic Review on Intra-Abdominal Pressure in Severely Burned Patients. Burns 2014, 40, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Browning, J.A.; Cindass, R. Burn Debridement, Grafting, and Reconstruction. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2020. [Google Scholar]
- Zhang, J.; Xiang, F.; Tong, D.; Luo, Q.; Yuan, Z.; Yan, H.; Li, X.; Chen, J.; Peng, D.; Luo, G.; et al. Comparative study on the effect of restrictive fluid management strategy on the early pulmonary function of patients with severe burn. Zhonghua Shao Shang Za Zhi 2012, 28, 165–169. [Google Scholar]
- Guilabert, P.; Usúa, G.; Martín, N.; Abarca, L.; Barret, J.P.; Colomina, M.J. Fluid Resuscitation Management in Patients with Burns: Update. Br. J. Anaesth. 2016, 117, 284–296. [Google Scholar] [CrossRef]
- Hickerson, W.L.; Ryan, C.M.; Conlon, K.M.; Harrington, D.T.; Foster, K.; Schwartz, S.; Iyer, N.; Jeschke, M.; Haller, H.L.; Faucher, L.D.; et al. What’s in a Name? Recent Key Projects of the Committee on Organization and Delivery of Burn Care. J. Burn Care Res. 2015, 36, 619–625. [Google Scholar] [CrossRef] [PubMed]
- Serghiou, M.A.; Niszczak, J.; Parry, I.; Li-Tsang, C.W.P.; Van den Kerckhove, E.; Smailes, S.; Edgar, D. One World One Burn Rehabilitation Standard. Burns 2016, 42, 1047–1058. [Google Scholar] [CrossRef]
- West, M.A.; Moore, E.E.; Shapiro, M.B.; Nathens, A.B.; Cuschieri, J.; Johnson, J.L.; Harbrecht, B.G.; Minei, J.P.; Bankey, P.E.; Maier, R.V. Inflammation and the Host Response to Injury, a Large-Scale Collaborative Project: Patient-Oriented Research Core—Standard Operating Procedures for Clinical Care VII—Guidelines for Antibiotic Administration in Severely Injured Patients. J. Trauma Inj. Infect. Crit. Care 2008, 65, 1511–1519. [Google Scholar] [CrossRef] [PubMed]
- Silver, G.M.; Klein, M.B.; Herndon, D.N.; Gamelli, R.L.; Gibran, N.S.; Altstein, L.; McDonald-Smith, G.P.; Tompkins, R.G.; Hunt, J.L.; The Inflammation and the Host Response to Trauma, Collaborative Research Program. Standard Operating Procedures for the Clinical Management of Patients Enrolled in a Prospective Study of Inflammation and the Host Response to Thermal Injury. J. Burn Care Res. 2007, 28, 222–230. [Google Scholar] [CrossRef]
- Nichter, L.S.; Williams, J.; Bryant, C.A.; Edlich, R.F. Improving the Accuracy of Burn-Surface Estimation. Plast. Reconstr. Surg. 1985, 76, 428–433. [Google Scholar] [CrossRef]
- Wachtel, T.L.; Berry, C.C.; Wachtel, E.E.; Frank, H.A. The Inter-Rater Reliability of Estimating the Size of Burns from Various Burn Area Chart Drawings. Burn. J. Int. Soc. Burn Inj. 2000, 26, 156–170. [Google Scholar] [CrossRef]
- Berkebile, B.L.; Goldfarb, I.W.; Slater, H. Comparison of Burn Size Estimates Between Prehospital Reports and Burn Center Evaluations. J. Burn Care Rehabil. 1986, 7, 411–412. [Google Scholar] [CrossRef] [PubMed]
- Hammond, J.S.; Ward, C.G. Transfers from Emergency Room to Burn Center: Errors in Burn Size Estimate. J. Trauma 1987, 27, 1161–1165. [Google Scholar] [CrossRef] [PubMed]
- Goverman, J.; Bittner, E.A.; Friedstat, J.S.; Moore, M.; Nozari, A.; Ibrahim, A.E.; Sarhane, K.A.; Chang, P.H.; Sheridan, R.L.; Fagan, S.P. Discrepancy in Initial Pediatric Burn Estimates and Its Impact on Fluid Resuscitation. J. Burn Care Res. 2015, 36, 574–579. [Google Scholar] [CrossRef] [PubMed]
- Hintermüller, C. Estimation of Total Burn Surface Area: A Comparison of Four Different Methods; Paracelsus Medical University: Salzburg, Austria, 2016. [Google Scholar]
- Wurzer, P.; Parvizi, D.; Lumenta, D.B.; Giretzlehner, M.; Branski, L.K.; Finnerty, C.C.; Herndon, D.N.; Tuca, A.; Rappl, T.; Smolle, C.; et al. Smartphone Applications in Burns. Burns 2015, 41, 977–989. [Google Scholar] [CrossRef]
- Vercruysse, G.A.; Ingram, W.L.; Feliciano, D.V. Overutilization of Regional Burn Centers for Pediatric Patientsa Healthcare System Problem That Should Be Corrected. Am. J. Surg. 2011, 202, 802–809. [Google Scholar] [CrossRef]
- Klasen, H.J. Chapter I: Classification of burns. In History of Burns; Erasmus Publishing: Rotterdam, The Netherlands, 2004; pp. 21–66. ISBN 90 5235 168 6. [Google Scholar]
- Riehl, G. Zur Therapie Schwerer Verbrennungen. Wien Klin Wochenschr. 1925, 37, 833–834. [Google Scholar]
- Dubois, D.; Dubois, E. A Formula to Estimate the Approximate Surface Area If Height and Weight Be Known. Arch. Intern. Med. 1916, 17, 863–871. [Google Scholar] [CrossRef]
- Lund, C.C.; Browder, N.C. The Estimation of Areas of Burns. Surg. Gynecol. Obstet. 1944, 79, 352–358. [Google Scholar]
- Boyd, E. The Growth of the Surface Area of the Human Body; University of Minnesota Press: Minnesota, MN, USA, 1935. [Google Scholar]
- Neaman, K.C.; Andres, L.A.; McClure, A.M.; Burton, M.E.; Kemmeter, P.R.; Ford, R.D. A New Method for Estimation of Involved BSAs for Obese and Normal-Weight Patients with Burn Injury. J. Burn Care Res. 2011, 32, 421–428. [Google Scholar] [CrossRef] [PubMed]
- Wilson, G.R.; Fowler, C.A.; Housden, P.L. A New Burn Area Assessment Chart. Burns 1987, 13, 401–405. [Google Scholar] [CrossRef]
- Miminas, D.A. A Critical Evaluation of the Lund and Browder Chart. Wounds 2007, 3, 58–68. [Google Scholar]
- Haller, H.L.; Giretzlehner, M.; Thumfart, S. Burn Size Estimation, Challenges, and Novel Technology. In Handbook of Burns Volume 1: Acute Burn Care; Jeschke, M.G., Kamolz, L.-P., Sjöberg, F., Wolf, S.E., Eds.; Springer International Publishing: Cham, Switzerland, 2020; pp. 181–197. ISBN 978-3-030-18940-2. [Google Scholar]
- Collis, N.; Smith, G.; Fenton, O.M. Accuracy of Burn Size Estimation and Subsequent Fluid Resuscitation Prior to Arrival at the Yorkshire Regional Burns Unit. A Three Year Retrospective Study. Burns 1999, 25, 345–351. [Google Scholar] [CrossRef]
- Freiburg, C.; Igneri, P.; Sartorelli, K.; Rogers, F. Effects of Differences in Percent Total Body Surface Area Estimation on Fluid Resuscitation of Transferred Burn Patients. J. Burn Care Res. 2007, 28, 42–48. [Google Scholar] [CrossRef] [PubMed]
- Irwin, L.R.; Reid, C.A.; McLean, N.R. Burns in Children: Do Casualty Officers Get It Right? Injury 1993, 24, 187–188. [Google Scholar] [CrossRef]
- Klippel, C.H. Surface Area versus Skin Area. N. Engl. J. Med. 1979, 301, 730. [Google Scholar] [PubMed]
- Wallace, A.B. The Exposure Treatment of Burns. Lancet 1951, 257, 501–504. [Google Scholar] [CrossRef]
- Berry, C.C.; Wachtel, T.; Frank, H.A. Differences in Burn Size Estimates Between Community Hospitals and a Burn Center. J. Burn Care Rehabil. 1982, 3, 176–178. [Google Scholar] [CrossRef]
- Giretzlehner, M.; Dirnberger, J.; Owen, R.; Haller, H.L.; Lumenta, D.B.; Kamolz, L.-P. The Determination of Total Burn Surface Area: How Much Difference? Burn. J. Int. Soc. Burn Inj. 2013, 39, 1–7. [Google Scholar] [CrossRef]
- Livingston, E.H.; Lee, S. Percentage of Burned Body Surface Area Determination in Obese and Nonobese Patients. J. Surg. Res. 2000, 91, 106–110. [Google Scholar] [CrossRef] [PubMed]
- Williams, R.Y.; Wohlgemuth, S.D. Does the “Rule of Nines” Apply to Morbidly Obese Burn Victims? J. Burn Care Res. Off. Publ. Am. Burn Assoc. 2013, 34, 447–452. [Google Scholar] [CrossRef] [PubMed]
- Yu, C.-Y.; Lin, C.-H.; Yang, Y.-H. Human Body Surface Area Database and Estimation Formula. Burn. J. Int. Soc. Burn Inj. 2010, 36, 616–629. [Google Scholar] [CrossRef] [PubMed]
- Rossiter, N.D.; Chapman, P.; Haywood, I.A. How Big Is a Hand? Burn. J. Int. Soc. Burn Inj. 1996, 22, 230–231. [Google Scholar] [CrossRef]
- Nagel, T.R.; Schunk, J.E. Using the Hand to Estimate the Surface Area of a Burn in Children. Pediatric Emerg. Care 1997, 13, 254–255. [Google Scholar] [CrossRef]
- Berry, M.G.; Evison, D.; Roberts, A.H. The Influence of Body Mass Index on Burn Surface Area Estimated from the Area of the Hand. Burn. J. Int. Soc. Burn Inj. 2001, 27, 591–594. [Google Scholar] [CrossRef]
- Butz, D.R.; Collier, Z.; O’Connor, A.; Magdziak, M.; Gottlieb, L.J.; Connor, A.O.; Magdziak, M.; Gottlieb, L.J. Is Palmar Surface Area a Reliable Tool to Estimate Burn Surface Areas in Obese Patients ? J. Burn Care Res. Off. Publ. Am. Burn Assoc. 2015, 36, 87–91. [Google Scholar] [CrossRef] [PubMed]
- Laing, J.H.; Morgan, B.D.; Sanders, R. Assessment of Burn Injury in the Accident and Emergency Department: A Review of 100 Referrals to a Regional Burns Unit. Ann. R. Coll. Surg. Engl. 1991, 73, 329–331. [Google Scholar] [PubMed]
- Cone, J.B. What’s New in General Surgery: Burns and Metabolism. J. Am. Coll. Surg. 2005, 200, 607–615. [Google Scholar] [CrossRef]
- Sheng, W.; Zeng, D.; Wan, Y.; Yao, L.; Tang, H.; Xia, Z. BurnCalc Assessment Study of Computer-Aided Individual Three-Dimensional Burn Area Calculation. J. Transl. Med. 2014, 12, s12967–s014. [Google Scholar] [CrossRef][Green Version]
- Wachtel, T.L.; Brimm, J.E.; Knight, M.A.; Heisterkamp, S.; Frank, H.A.; Inancsi, W. Research: Computer Assisted Estimation of the Size of Burns. J. Burn Care Rehabil. 1983, 4, 255–259. [Google Scholar] [CrossRef]
- Barnes, J.; Duffy, A.; Hamnett, N.; McPhail, J.; Seaton, C.; Shokrollahi, K.; James, M.I.; McArthur, P.; Pritchard Jones, R. The Mersey Burns App: Evolving a Model of Validation. Emerg. Med. J. 2015, 32, 637–641. [Google Scholar] [CrossRef]
- Morris, R.; Javed, M.; Bodger, O.; Gorse, S.H.; Williams, D. A Comparison of Two Smartphone Applications and the Validation of Smartphone Applications as Tools for Fluid Calculation for Burns Resuscitation. Burns 2014, 40, 826–834. [Google Scholar] [CrossRef]
- Neuwalder, J.M.; Sampson, C.; Breuing, K.H.; Orgill, D.P. A Review of Computer-Aided Body Surface Area Determination: SAGE II and EPRI’s 3D Burn Vision. J. Burn Care Rehabil. 2002, 23, 55–59. [Google Scholar] [CrossRef]
- Knaysi, G.A.; Crikelair, G.F.; Cosman, B. The Role of Nines: Its History and Accuracy. Plast. Reconstr. Surg. 1968, 41, 560–563. [Google Scholar] [CrossRef] [PubMed]
- Lee, R.C.; Kieska, G.; Mankani, M.H. A Three-Dimensional Computerized Burn Chart: Stage I: Development of Three-Dimensional Renderings. J. Burn Care Rehabil. 1994, 15, 80–83. [Google Scholar] [CrossRef]
- Mankani, M.H.; Kicska, G.; Lee, R.C. A Three-Dimensional Computerized Burn Chart: Stage II: Assessment of Accuracy. J. Burn Care Rehabil. 1994, 15, 191–192. [Google Scholar] [CrossRef]
- Parvizi, D.; Giretzlehner, M.; Wurzer, P.; Klein, L.D.; Shoham, Y.; Bohanon, F.J.; Haller, H.L.; Tuca, A.; Branski, L.K.; Lumenta, D.B.; et al. BurnCase 3D Software Validation Study: Burn Size Measurement Accuracy and Inter-Rater Reliability. Burns 2016, 42, 329–335. [Google Scholar] [CrossRef]
- Yu, C.-Y.; Lo, Y.-H.; Chiou, W.-K. The 3D Scanner for Measuring Body Surface Area: A Simplified Calculation in the Chinese Adult. Appl. Ergon. 2003, 34, 273–278. [Google Scholar] [CrossRef]
- Stockton, K.A.; McMillan, C.M.; Storey, K.J.; David, M.C.; Kimble, R.M. 3D Photography Is as Accurate as Digital Planimetry Tracing in Determining Burn Wound Area. Burns 2015, 41, 80–84. [Google Scholar] [CrossRef] [PubMed]
- Wurzer, P.; Giretzlehner, M.; Kamolz, L.-P. 3D Photography Is an Accurate Technique for Measuring Small Wound Areas. Burns 2015, 41, 196–197. [Google Scholar] [CrossRef]
- Yu, C.-Y.; Hsu, Y.-W.; Chen, C.-Y. Determination of Hand Surface Area as a Percentage of Body Surface Area by 3D Anthropometry. Burn. J. Int. Soc. Burn Inj. 2008, 34, 1183–1189. [Google Scholar] [CrossRef]
- Goldberg, H.; Klaff, J.; Spjut, A.; Milner, S. A Mobile App for Measuring the Surface Area of a Burn in Three Dimensions: Comparison to the Lund and Browder Assessment. J. Burn Care Res. 2014, 35, 480–483. [Google Scholar] [CrossRef]
- Thumfart, S.; Giretzlehner, M.; Wurzer, P.; Höller, J.; Ehrenmüller, M.; Pfurtscheller, K.; Haller, H.L.; Kamolz, L.-P.; Schmitt, K.; Furthner, D. Burn Size Measurement Using Proportionally Correct 3D Models of Pediatric Patients. In Proceedings of the Supplement to Journal of Burn Care & Research, Las Vegas, NV, USA, 3–6 May 2016; Volume 37, p. 80. [Google Scholar]
- Siegel, J.B.; Wachtel, T.L.; Brimm, J.E. Automated Documentation and Analysis of Burn Size. J. Trauma Inj. Infect. Crit. Care 1986, 26, 44–46. [Google Scholar] [CrossRef] [PubMed]
- Kinnunen, U.-M.; Saranto, K.; Ensio, A.; Iivanainen, A.; Dykes, P. Developing the Standardized Wound Care Documentation Model: A Delphi Study to Improve the Quality of Patient Care Documentation. J. Wound Ostomy Cont. Nurs. 2012, 39, 397–407. [Google Scholar] [CrossRef]
- Panfil, E.; Linde, E. Kriterien Zur Wunddokumentation–Literaturanalyse; Hessisches Institut Für Pflegeforschung: Frankfurt, Germany, 2006. [Google Scholar]
- Giretzlehner, M.; Haller, H.L.; Faucher, L.D.; Pressman, M.A.; Salinas, J.; Jeng, J.C. One Burn, One Standard. J. Burn Care Res. 2014, 35, e372. [Google Scholar] [CrossRef]
- Törnvall, E.; Wahren, L.K.; Wilhelmsson, S. Advancing Nursing Documentation--an Intervention Study Using Patients with Leg Ulcer as an Example. Int. J. Med. Inf. 2009, 78, 605–617. [Google Scholar] [CrossRef]
- Hübner, U.; Flemming, D.; Schultz-Gödker, A. Software Zur Digitalen Wunddokumentation: Marktübersicht und Bewertungskriterien. Wundmanagement 2009, 3, 16–25. [Google Scholar]
- Ingenerf, J. Computergestützte Strukturierte Befundung Am Beispiel der Wunddokumentation. Wundmanagement 2009, 3, 104–108. [Google Scholar]
- Parvizi, D.; Giretzlehner, M.; Dirnberger, J.; Owen, R.; Haller, H.L.; Schintler, M.V.; Wurzer, P.; Lumenta, D.B.; Kamolz, L.P. The Use of Teleemedicine in Burn Care: Development of a Mobile System for Tbsa Documentation and Remote Assessment. Ann. Burn. Fire Disasters 2014, 7, 94. [Google Scholar]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Giretzlehner, M.; Ganitzer, I.; Haller, H. Technical and Medical Aspects of Burn Size Assessment and Documentation. Medicina 2021, 57, 242. https://doi.org/10.3390/medicina57030242
Giretzlehner M, Ganitzer I, Haller H. Technical and Medical Aspects of Burn Size Assessment and Documentation. Medicina. 2021; 57(3):242. https://doi.org/10.3390/medicina57030242
Chicago/Turabian StyleGiretzlehner, Michael, Isabell Ganitzer, and Herbert Haller. 2021. "Technical and Medical Aspects of Burn Size Assessment and Documentation" Medicina 57, no. 3: 242. https://doi.org/10.3390/medicina57030242
APA StyleGiretzlehner, M., Ganitzer, I., & Haller, H. (2021). Technical and Medical Aspects of Burn Size Assessment and Documentation. Medicina, 57(3), 242. https://doi.org/10.3390/medicina57030242





