Investigating the Safety and Efficacy of Platelet-Rich Plasma (PRP) Treatment for Female Androgenetic Alopecia: Review of the Literature
Abstract
1. Introduction
1.1. FAGA: Clinical Features, Pathogenesis and Current Therapeutic Options
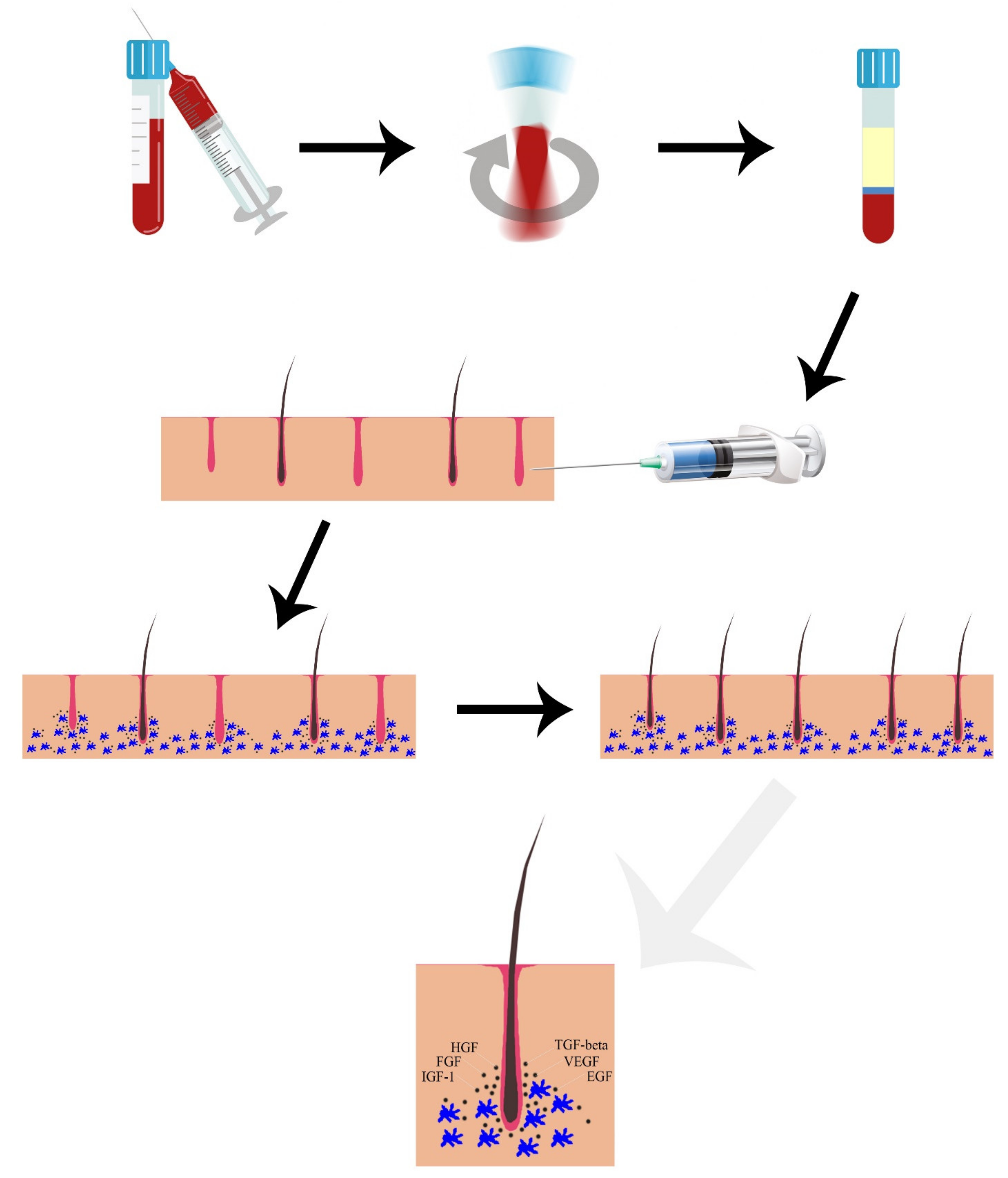
1.2. Platelet Rich Plasma (PRP)
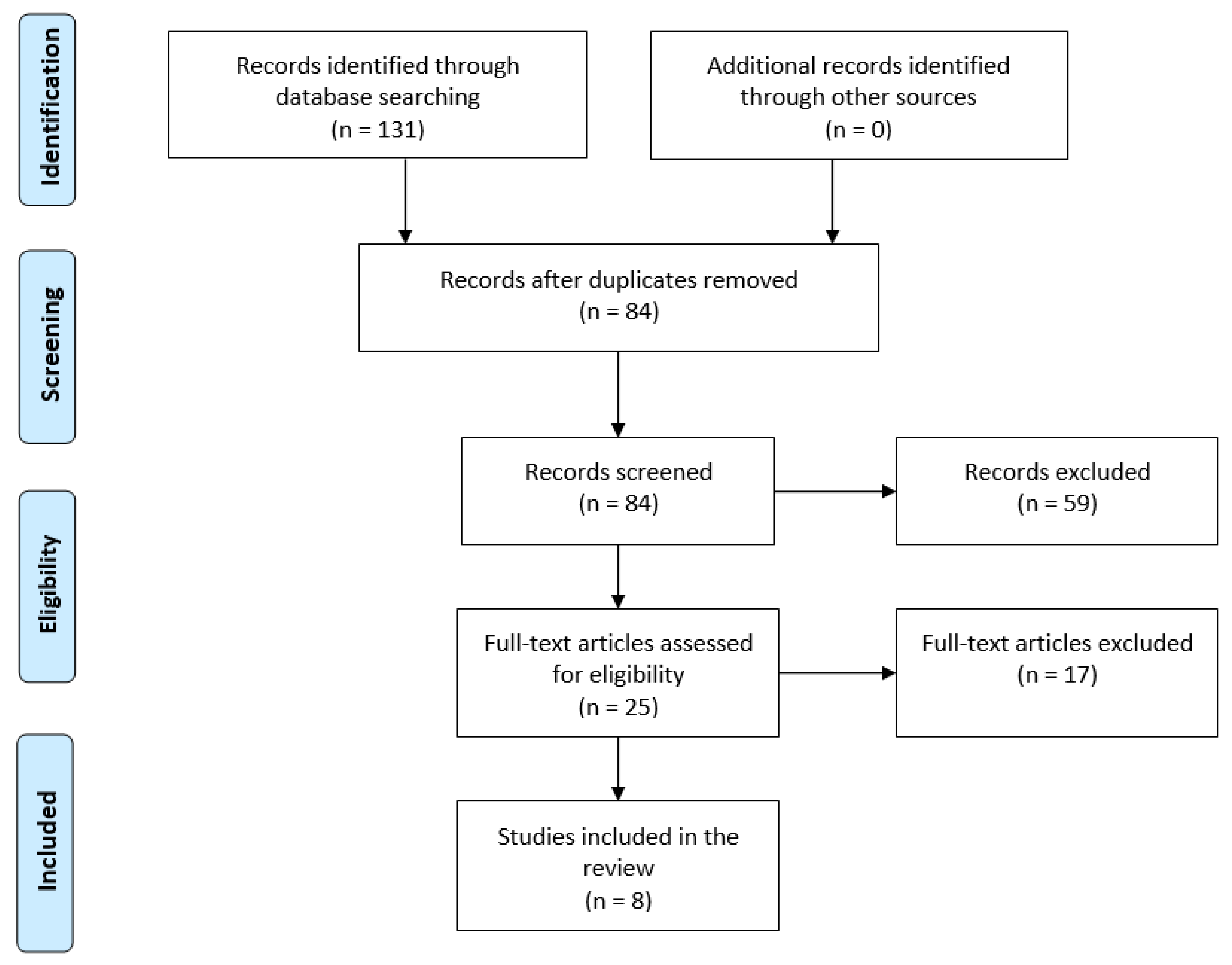
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Jahoda, C.A. Cellular and developmental aspects of androgenetic alopecia. Exp. Dermatol. 1998, 7, 235–248. [Google Scholar]
- Olsen, E.A. Female pattern hair loss. J. Am. Acad. Dermatol. 2001, 45, S70–S80. [Google Scholar] [CrossRef] [PubMed]
- Norwood, O.T.; Lehr, B. Female Androgenetic Alopecia: A Separate Entity. Dermatol. Surg. 2000, 26, 679–682. [Google Scholar] [CrossRef]
- Price, V.H. Androgenetic alopecia in women. J. Investig. Dermatol. Symp. Proc. 2003, 8, 24–27. [Google Scholar] [CrossRef]
- Piraccini, B.M.; Alessandrini, A. Androgenetic alopecia. G Ital. Dermatol. E Venereol. 2014, 149, 15–24. [Google Scholar]
- Shannon, F.; Christa, S.; Lewei, D.; Carolyn, G. Demographics of women with female pattern hair loss and the effectiveness of spironolactone therapy. J. Am. Acad. Dermatol. 2015, 73, 705–706. [Google Scholar] [CrossRef]
- Ellis, J.A.; Stebbing, M.; Harrap, S.B. Polymorphism of the androgen receptor gene is associated with male pattern baldness. J. Investig. Dermatol. 2001, 116, 452–455. [Google Scholar] [CrossRef] [PubMed]
- Tu, Y.-A.; Lin, S.-J.; Chen, P.-L.; Chou, C.-H.; Huang, C.-C.; Ho, H.-N.; Chen, M.-J. HSD3B1 gene polymorphism and female pattern hair loss in women with polycystic ovary syndrome. J. Formos Med. Assoc. 2019, 118, 1225–1231. [Google Scholar] [CrossRef]
- Redler, S.; Birch, M.; Drichel, D.; Dobson, K.; Brockschmidt, F.; Tazi-Ahnini, R.; Giehl, K.; Kluck, N.; Kruse, R.; Lutz, G.; et al. Investigation of variants of the aromatase gene (CYP19A1) in female pattern hair loss. Br. J. Dermatol. 2011, 165, 703–705. [Google Scholar] [CrossRef]
- Redler, S.; Messenger, A.G.; Betz, R.C. Genetics and other factors in the aetiology of female pattern hair loss. Exp. Dermatol. 2017, 26, 510–517. [Google Scholar] [CrossRef] [PubMed]
- Seleit, I.; Bakry, O.A.; Badr, E.; Mabrouk, M. Vitamin D Receptor Gene Polymorphisms Taq-1 and Cdx-1 in Female Pattern Hair Loss. Indian J. Dermatol. 2020, 65, 259–264. [Google Scholar] [CrossRef] [PubMed]
- Yip, L.; Rufaut, N.; Sinclair, R. Role of genetics and sex steroid hormones in male androgenetic alopecia and female pattern hair loss, an update of what we now know. Australas J. Dermatol. 2011, 52, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Randall, V.A. Androgens and hair growth. Dermatol. Ther. 2008, 21, 314–328. [Google Scholar] [CrossRef]
- Itami, S.; Inui, S. Role of androgen in mesenchymal epithelial interactions in human hair follicle. J. Investig. Dermatol. Symp. Proc. 2005, 10, 209–211. [Google Scholar] [CrossRef] [PubMed]
- Kwack, M.H.; Sung, Y.K.; Chung, E.J.; Im, S.U.; Ahn, J.S.; Kim, M.K.; Kim, J.C. Dihydrotestosterone-inducible dickkopf 1 from balding dermal papilla cells causes apoptosis in follicular keratinocytes. J. Investig. Dermatol. 2008, 128, 262–269. [Google Scholar] [CrossRef] [PubMed]
- Winiarska, A.; Mandt, N.; Kamp, H.; Hossini, A.; Seltmann, H.; Zouboulis, C.; Blume-Peytavi, U. Effect of 5alpha-dihydrotestosterone and testosterone on apoptosis in human dermal papilla cells. Skin Pharmacol. Physiol. 2006, 19, 311–321. [Google Scholar] [CrossRef]
- Norwood, O.T. Incidence of female androgenetic alopecia (female pattern alopecia). Dermatol. Surg. 2001, 27, 53–54. [Google Scholar]
- Vexiau, P.; Chaspoux, C.; Boudou, P.; Fiet, J.; Abramovici, Y.; Rueda, M.; Hardy, N.; Reygagne, P. Role of Androgens in Female-Pattern Androgenetic Alopecia, Either Alone or Associated With Other Symptoms of Hyperandrogenism. Arch. Dermatol. Res. 2000, 292, 598–604. [Google Scholar] [CrossRef]
- Olsen, E.A. Female Pattern Hair Loss and its Relationship to Permanent/Cicatricial Alopecia: A New Perspective. J. Investig. Dermatol. Symp. Proc. 2005, 10, 217–221. [Google Scholar] [CrossRef]
- El-Husseiny, R.M.; Saleh, H.M.; Moustafa, A.A.; Salem, S.A. Comparison between single—Versus double-spin prepared platelet-rich plasma injection in treatment of female pattern hair loss: Clinical effect and relation to vascular endothelial growth factor. Arch. Dermatol. Res. 2020, 1–10. [Google Scholar] [CrossRef]
- Sawaya, M.E.; Price, V.H. Different levels of 5alpha-reductase type I and II, aromatase, and androgen receptor in hair follicles of women and men with androgenetic alopecia. J. Investig. Dermatol. 1997, 109, 296–300. [Google Scholar] [CrossRef]
- Ludwig, E. Classification of the types of androgenetic alopecia (common baldness) occurring in the female sex. Br. J. Dermatol. 1977, 97, 247–254. [Google Scholar] [CrossRef] [PubMed]
- Rathnayake, D.; Sinclair, R. Innovative use of spironolactone as an antiandrogen in the treatment of female pattern hair loss. Dermatol. Clin. 2010, 28, 611–618. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, J.B.; Lindmaier, A.; Trenz, A.; Schurz, B.; Spona, J. Hormone studies in females with androgenic hairloss. Gynecol. Obstet. Investig. 1991, 31, 235–239. [Google Scholar] [CrossRef] [PubMed]
- Orme, S.; Cullen, D.R.; Messenger, A.G. Diffuse female hair loss: Are androgens necessary? Br. J. Dermatol. 1999, 141, 521–523. [Google Scholar] [CrossRef]
- Starace, M.; Orlando, G.; Alessandrini, A.; Piraccini, B.M. Female Androgenetic Alopecia: An Update on Diagnosis and Management. Am. J. Clin. Dermatol. 2020, 21, 69–84. [Google Scholar] [CrossRef] [PubMed]
- Russo, P.M.; Fino, E.; Mancini, C.; Mazzetti, M.; Starace, M.; Piraccini, B.M. HrQoL in hair loss-affected patients with alopecia areata, androgenetic alopecia and telogen effluvium: The role of personality traits and psychosocial anxiety. J. Eur. Acad. Dermatol. Venereol. JEADV 2019, 33, 608–611. [Google Scholar] [CrossRef]
- Sinclair, R.; Jolley, D.; Mallari, R.; Magee, J. The reliability of horizontally sectioned scalp biopsies in the diagnosis of chronic diffuse telogen hair loss in women. J. Am. Acad. Dermatol. 2004, 51, 189–199. [Google Scholar] [CrossRef]
- Ross, E.K.; Vincenzi, C.; Tosti, A. Videodermoscopy in the evaluation of hair and scalp disorders. J. Am. Acad. Dermatol. 2006, 55, 799–806. [Google Scholar] [CrossRef]
- Rakowska, A.; Slowinska, M.; Kowalska-Oledzka, E.; Olszewska, M.; Rudnicka, L. Dermoscopy in female androgenic alopecia: Method standardization and diagnostic criteria. Int. J. Trichol. 2009, 1, 123–130. [Google Scholar] [CrossRef] [PubMed]
- Mubki, T.; Rudnicka, L.; Olszewska, M.; Shapiro, J. Evaluation and diagnosis of the hair loss patient: Part II. Trichoscopic and laboratory evaluations. J. Am. Acad. Dermatol. 2014, 71, 431.e1–431.e11. [Google Scholar] [CrossRef]
- Ummiti, A.; Priya, P.S.; Chandravathi, P.L.; Kumar, C.S. Correlation of Trichoscopic Findings in Androgenetic Alopecia and the Disease Severity. Int. J. Trichol. 2019, 11, 118–122. [Google Scholar] [CrossRef]
- Kanti, V.; Messenger, A.; Dobos, G.; Reygagne, P.; Finner, A.; Blumeyer, A.; Trakatelli, M.; Tosti, A.; Del Marmol, V.; Piraccini, B.; et al. Evidence-based (S3) guideline for the treatment of androgenetic alopecia in women and in men—Short version. J. Eur. Acad. Dermatol. Venereol. JEADV 2018, 32, 11–22. [Google Scholar] [CrossRef]
- Van Neste, D. Female patients complaining about hair loss: Documentation of defective scalp hair dynamics with contrast-enhanced phototrichogram. Skin Res. Technol. 2006, 12, 83–88. [Google Scholar] [CrossRef] [PubMed]
- Rushton, D.H.; de Brouwer, B.; de Coster, W.; van Neste, D.J. Comparative evaluation of scalp hair by phototrichogram and unit area trichogram analysis within the same subjects. Acta Derm. Venereol. 1993, 73, 150–153. [Google Scholar] [CrossRef] [PubMed]
- Van Neste, D.J. Contrast enhanced phototrichogram (CE-PTG): An improved non-invasive technique for measurement of scalp hair dynamics in androgenetic alopecia--validation study with histology after transverse sectioning of scalp biopsies. Eur. J. Dermatol. EJD 2001, 11, 326–331. [Google Scholar]
- Hoffmann, R. TrichoScan: Combining epiluminescence microscopy with digital image analysis for the measurement of hair growth in vivo. Eur. J. Dermatol. EJD 2001, 11, 362–368. [Google Scholar]
- Riedel-Baima, B.; Riedel, A. Use of the TrichoScan to assess female pattern hair loss. Dermatol. Surg. 2009, 35, 651–655. [Google Scholar] [CrossRef]
- López, V.; Martín, J.M.; Sánchez, R.; Ortega, C.; Ricart, J.M. Usefulness of TrichoScan professional in the evaluation of hair loss in females. Report of 180 cases. J. Eur. Acad. Dermatol. Venereol. JEADV 2011, 25, 1068–1072. [Google Scholar] [CrossRef] [PubMed]
- Gassmueller, J.; Rowold, E.; Frase, T.; Hughes-Formella, B. Validation of TrichoScan technology as a fully-automated tool for evaluation of hair growth parameters. Eur. J. Dermatol. EJD 2009, 19, 224–231. [Google Scholar] [CrossRef] [PubMed]
- Carmina, E.; Azziz, R.; Bergfeld, W.; Escobar-Morreale, H.F.; Futterweit, W.; Huddleston, H.; Lobo, R.; Olsen, E. Female Pattern Hair Loss and Androgen Excess: A Report From the Multidisciplinary Androgen Excess and PCOS Committee. J. Clin. Endocrinol. Metab. 2019, 104, 2875–2891. [Google Scholar] [CrossRef] [PubMed]
- Suchonwanit, P.; Thammarucha, S.; Leerunyakul, K. Minoxidil and its use in hair disorders: A review. Drug Des. Dev. Ther. 2019, 13, 2777–2786. [Google Scholar] [CrossRef] [PubMed]
- Gentile, P.; Dionisi, L.; Pizzicannella, J.; De Angelis, B.; De Fazio, D.; Garcovich, S. A randomized blinded retrospective study: The combined use of micro-needling technique, low-level laser therapy and autologous non-activated platelet-rich plasma improves hair re-growth in patients with androgenic alopecia. Expert Opin. Biol. Ther. 2020, 20, 1099–1109. [Google Scholar] [CrossRef] [PubMed]
- Hesseler, M.J.; Shyam, N. Platelet-Rich Plasma and Its Utilities in Alopecia: A Systematic Review. Dermatol. Surg. 2020, 46, 93–102. [Google Scholar] [CrossRef] [PubMed]
- Ehrenfest, D.M.D.; Bielecki, T.; Mishra, A.; Borzini, P.; Inchingolo, F.; Sammartino, G.; Rasmusson, L.; Evert, P.A. In Search of a Consensus Terminology in the Field of Platelet Concentrates for Surgical Use: Platelet-Rich Plasma (PRP), Platelet-Rich Fibrin (PRF), Fibrin Gel Polymerization and Leukocytes. Curr. Pharm. Biotechnol. 2012, 13, 1131–1137. [Google Scholar] [CrossRef]
- Mercuri, S.R.; Vollono, L.; Paolino, G. The Usefulness of Platelet-Rich Plasma (PRP) for the Treatment of Vitiligo: State of the Art and Review. Drug Des. Dev. Ther. 2020, 14, 1749–1755. [Google Scholar] [CrossRef]
- Dhurat, R.; Sukesh, M. Principles and Methods of Preparation of Platelet-Rich Plasma: A Review and Author’s Perspective. J. Cutan Aesthetic Surg. 2014, 7, 189–197. [Google Scholar] [CrossRef]
- Gupta, A.K.; Carviel, J.A. Mechanistic Model of Platelet-Rich Plasma Treatment for Androgenetic Alopecia. Dermatol. Surg. 2016, 42, 1335–1339. [Google Scholar] [CrossRef]
- Li, Z.J.; Choi, H.-I.; Choi, D.-K.; Sohn, K.-C.; Im, M.; Seo, Y.-J.; Lee, Y.-H.; Lee, J.-H. Autologous Platelet-Rich Plasma: A Potential Therapeutic Tool for Promoting Hair Growth. Dermatol. Surg. 2012, 38, 1040–1046. [Google Scholar] [CrossRef] [PubMed]
- Pierce, G.F.; A Mustoe, T.; Lingelbach, J.; Masakowski, V.R.; Griffin, G.L.; Senior, R.M.; Deuel, T.F. Platelet-derived growth factor and transforming growth factor-beta enhance tissue repair activities by unique mechanisms. J. Cell Biol. 1989, 109, 429–440. [Google Scholar] [CrossRef] [PubMed]
- Mecklenburg, L.; Tobin, D.J.; Müller-Röver, S.; Handjiski, B.; Wendt, G.; Peters, E.M.; Pohl, S.; Moll, I.; Paus, R. Active Hair Growth (Anagen) is Associated with Angiogenesis. J. Investig. Dermatol. 2000, 114, 909–916. [Google Scholar] [CrossRef]
- Giudice, A.; Esposito, M.; Bennardo, F.; Brancaccio, Y.; Buti, J.; Fortunato, L. Dental extractions for patients on oral antiplatelet: A within-person randomised controlled trial comparing haemostatic plugs, advanced-platelet-rich fibrin (A-PRF+) plugs, leukocyte- and platelet-rich fibrin (L-PRF) plugs and suturing alone. Int. J. Oral Implant 2019, 12, 77–87. [Google Scholar]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; PRISMA Group. Preferredreporting items for systematic reviews and meta-analyses: The PRISMAstatement. BMJ 2009, 339, b2535. [Google Scholar] [CrossRef] [PubMed]
- Gupta, M.; Mysore, V. Classifications of Patterned Hair Loss: A Review. J. Cutan Aesthetic Surg. 2016, 9, 3–12. [Google Scholar] [CrossRef]
- Lee, S.H.; Zheng, Z.; Kang, J.S.; Kim, D.Y.; Oh, S.H.; Cho, S.B. Therapeutic efficacy of autologous platelet-rich plasma and polydeoxyribonucleotide on female pattern hair loss. Wound Repair Regen. 2015, 23, 30–36. [Google Scholar] [CrossRef]
- Tawfik, A.A.; Osman, M.A.R. The effect of autologous activated platelet-rich plasma injection on female pattern hair loss: A randomized placebo-controlled study. J. Cosmet. Dermatol. 2018, 17, 47–53. [Google Scholar] [CrossRef]
- Starace, M.; Alessandrini, A.; D’Acunto, C.; Melandri, D.; Bruni, F.; Pr, A.P.; Pr, B.M.P. Platelet-rich plasma on female androgenetic alopecia: Tested on 10 patients. J. Cosmet. Dermatol. 2018, 18, 59–64. [Google Scholar] [CrossRef]
- Dubin, D.P.; Lin, M.J.; Leight, H.M.; Farberg, A.S.; Torbeck, R.L.; Burton, W.B.; Khorasani, H. The effect of platelet-rich plasma on female androgenetic alopecia: A randomized controlled trial. J. Am. Acad. Dermatol. 2020, 83, 1294–1297. [Google Scholar] [CrossRef] [PubMed]
- Puig, C.J.; Reese, R.; Peters, M. Double-Blind, Placebo-Controlled Pilot Study on the Use of Platelet-Rich Plasma in Women with Female Androgenetic Alopecia. Dermatol. Surg. 2016, 42, 1243–1247. [Google Scholar] [CrossRef] [PubMed]
- BBruce, A.J.; Pincelli, T.P.; Heckman, M.G.; Desmond, C.M.; Arthurs, J.R.; Diehl, N.N.; Douglass, E.J.; Bruce, C.J.; Shapiro, S.A. A Randomized, Controlled Pilot Trial Comparing Platelet-Rich Plasma to Topical Minoxidil Foam for Treatment of Androgenic Alopecia in Women. Dermatol. Surg. 2020, 46, 826–832. [Google Scholar] [CrossRef]
- Laird, M.E.; Sicco, K.I.L.; Reed, M.L.; Brinster, N.K. Platelet-Rich Plasma for the Treatment of Female Pattern Hair Loss: A Patient Survey. Dermatol. Surg. 2018, 44, 130–132. [Google Scholar] [CrossRef] [PubMed]
- Goldman, B.E.; Fisher, D.M.; Ringler, S.L. Transcutaneous Po2 of the Scalp in Male Pattern Baldness: A New Piece to the Puzzle. Plast. Reconstr. Surg. 1996, 97, 1109–1116. [Google Scholar] [CrossRef]
- Li, W.; Enomoto, M.; Ukegawa, M.; Hirai, T.; Sotome, S.; Wakabayashi, Y.; Shinomiya, K.; Okawa, A. Subcutaneous Injections of Platelet-Rich Plasma into Skin Flaps Modulate Proangiogenic Gene Expression and Improve Survival Rates. Plast. Reconstr. Surg. 2012, 129, 858–866. [Google Scholar] [CrossRef] [PubMed]
- Gkini, M.-A.; Kouskoukis, A.-E.; Tripsianis, G.; Rigopoulos, D.; Kouskoukis, K. Study of platelet-rich plasma injections in the treatment of androgenetic alopecia through an one-year period. J. Cutan. Aesthetic Surg. 2014, 7, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Butt, G.; Hussain, I.; Ahmed, F.J.; Choudhery, M.S. Efficacy of platelet-rich plasma in androgenetic alopecia patients. J. Cosmet Dermatol. 2019, 18, 996–1001. [Google Scholar] [CrossRef] [PubMed]
- Qu, Q.; Shi, P.; Yi, Y.; Fan, Z.; Liu, X.; Zhu, D.; Chen, J.; Ye, K.; Miao, Y.; Hu, Z. Efficacy of Platelet-rich Plasma for Treating Androgenic Alopecia of Varying Grades. Clin. Drug Investig. 2019, 39, 865–872. [Google Scholar] [CrossRef]
- Schiavone, G.; Raskovic, D.; Greco, J.; Abeni, D. Platelet-Rich Plasma for Androgenetic Alopecia. Dermatol. Surg. 2014, 40, 1010–1019. [Google Scholar] [CrossRef]
- Shapiro, J.; Ho, A.; Sukhdeo, K.; Yin, L.; Sicco, K.L. Evaluation of platelet-rich plasma as a treatment for androgenetic alopecia: A randomized controlled trial. J. Am. Acad. Dermatol. 2020, 83, 1298–1303. [Google Scholar] [CrossRef] [PubMed]
- Alves, R.; Grimalt, R. Randomized Placebo-Controlled, Double-Blind, Half-Head Study to Assess the Efficacy of Platelet-Rich Plasma on the Treatment of Androgenetic Alopecia. Dermatol. Surg. 2016, 42, 491–497. [Google Scholar] [CrossRef]
- Torabi, P.; Behrangi, E.; Goodarzi, A.; Rohaninasab, M. A systematic review of the effect of platelet-rich plasma on androgenetic alopecia of women. Dermatol. Ther. 2020, 33, e13835. [Google Scholar] [CrossRef]
- Gupta, A.K.; Cole, J.; Deutsch, D.P.; Everts, P.A.; Niedbalski, R.P.; Panchaprateep, R.; Rinaldi, F.; Rose, P.T.; Sinclair, R.; Vogel, J.E.; et al. Platelet-Rich Plasma as a Treatment for Androgenetic Alopecia. Dermatol. Surg. 2019, 45, 1262–1273. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.K.; Renaud, H.J.; Bamimore, M. Platelet-rich plasma for androgenetic alopecia: Efficacy differences between men and women. Dermatol. Ther. 2020, 33, e14143. [Google Scholar] [CrossRef] [PubMed]
- Ho, A.; Sukhdeo, K.; Lo Sicco, K.; Shapiro, J. Trichologic response of platelet-rich plasma in androgenetic alopecia is maintained during combination therapy. J. Am. Acad. Dermatol. 2020, 82, 478–479. [Google Scholar] [CrossRef]
- Alves, R.; Grimalt, R. Platelet-Rich Plasma in Combination With 5% Minoxidil Topical Solution and 1 mg Oral Finasteride for the Treatment of Androgenetic Alopecia: A Randomized Placebo-Controlled, Double-Blind, Half-Head Study. Dermatol. Surg. 2018, 44, 126–130. [Google Scholar] [CrossRef] [PubMed]
- Lachgar, S.; Charveron, M.; Gall, Y.; Bonafe, J.L. Minoxidil upregulates the expression of vascular endothelial growth factor in human hair dermal papilla cells. Br. J. Dermatol. 1998, 138, 407–411. [Google Scholar] [CrossRef]
- Bennardo, F.; Bennardo, L.; Del Duca, E.; Patruno, C.; Fortunato, L.; Giudice, A.; Nisticò, S.P. Autologous platelet-rich fibrin injections in the management of facial cutaneous sinus tracts secondary to medication-related osteonecrosis of the jaw. Dermatol. Ther. 2020, 33, e13334. [Google Scholar] [CrossRef] [PubMed]
| Adverse Events | Results | Endpoints | Assessment Methods | Duration of the Study (Weeks) | N° of Sessions | Time Session | PRP Technique | Other Concurrent Treatments | Intervention | Ludwig Stage | Mean Age | N° of Subjects | Type of Study | Year | Author | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Transient edema and tenderness (=4), mild itching (N = 2) | Improvement in both hair count and hair diameter in both groups; greater improvement in hair thickness than hair count in Group A compared to Group B. | Hair count; hair diameter | Phototrichograms using Follioscope PT® software at BL and w14 | 14 | 1 | Single session | 60 mL of peripheral blood in g 8 mL of 4% sodium citrate solution centrifuged with the SmartPReP® platelet concentrate system; 4 mL injected in the scalp (intra/perifollicular areas) | No (naïve or 6 months wash-out) | NA-L-PRP followed by 12 weeks of weekly polydeoxirybonucleotide (PDRN) (Group A) vs. 12 weeks of weekly PDRN only (Group B) | NR | 33.9 (20–60) | 40 | Randomized, controlled comparative trial | 2015 | Lee, S. | ||||||
| NR | No significant difference in the main hair count or hair mass between Group A and B; higher perceived improvement in hair loss in Group A compared to Group B (13.3% vs. 0%); higher perceived hair mass in Group A compared to Group B (26.7% vs. 18.2%) | Hair count; hair mass index; patient satisfaction | Global photography; Cohen hair | check system; patient survey | 27 | 1 | Single session | 60 mL of peripheral blood centrifuged in Angel PRP system (Cytomedix®); produced PRP diluted with platelet-poor plasma; 10 mL of low-concentration, leukocyte- and | erythrocyte-free PRP for injection | No (naïve or 2 months wash-out) | NA-P-PRP (Group A) vs. saline injections (Group B) | II | NR | 26 | Randomized, double-blind, placebo-controlled trial | 2016 | Puig, C. | ||||
| Swollen forehead/face (29%); sore/painful scalp over 3 days (26%) | 58% patients claiming to be satisfied with the treatment, with 65% of them reporting either “marked” or “exceptional” improvement. | Patient satisfaction | Custom survey | 1 to 3 | Every 8–12 weeks | NR | NR; patients who underwent treatment with supplements, topical minoxidil and oral finasteride | u-PRP | NR | 51 | 41 | Patient survey | 2017 | Laird, M. | |||||||
| Temporary pain and pinpoint bleeding at the injection sites | Significant increase in hair density and hair thickness, improvement in hair-pull test in the PRP-treated areas compared with placebo areas; higher overall patient’s satisfaction for PRP areas compared to placebo areas. | Hair density (number of | hairs/cm2); hair diameter; hair volume; patient’s satisfaction | Global photography; hair pull test; patient’s satisfaction scale; phototrichograms | 28 | 1 to 4 | Every other week | 10 mL of peripheral blood in 1.5 mL of sodium citrate solution centrifuged at 1200 rpm for 15 min; buffy coat collected and centrifuged at 2000 rpm for 10 min; lower layer collected | No | AA-L-PRP (Group A) vs. saline injections (Group B) | I-II-III | 29.3 (20–45) | 30 | Randomized, intrapatient split-side, placebo-controlled trial | 2018 | Tawfik, A. | |||||
| NR | Significant increase in hair count, hair density and hair diameter; reduction in the vellus hair number | hair count; hair density (frontal-central-vertex); hair diameter; vellus hair count (frontal-central-vertex) | Global photography; hair pull test; phototrichograms using Trichoscan® software | 24 | 4 | Every 2 weeks | 10 mL of peripheral blood in acid-citrate-dextrose solution in MyCells® system centrifuged at 2500 rpm for 10 min; 5 mL of pure and concentrated autologous platelet-rich plasma injected in the scalp (perifollicular areas) | NR; patients not responding to treatment with | topical minoxidil and/or oral finasteride lasting for at least 1 year | NA-L-PRP | I; I-II; II; II-III | 47.1 (33–64) | 10 | Open-label, not randomized single-group clinical study | 2019 | Starace, M. | |||||
| Pain/discomfort/bruising (N = 4) | Greater degree of improvement in hair count in Group B compared to Group A; mild improvement in hair density and cumulative thickness in Group A compared to improvement in Group B. Similar degree of improvement in vellus hair density in both groups; significantly greater improvement in QOL responses in Group A compared to Group B | Hair count; terminal hair density; vellus hair density; total thickness; patient’s quality of life | Phototrichograms using Trichoscan® software; 16-item qualityof | life | (QOL) questionnaire; 7-item questionnaire | 48 | 3 | Every 4 weeks; washout among treatment regimens: 8 weeks | 60 mL of peripheral blood in 8 mL of citrate dextrose solution centrifuged at 1500 rpm for 10 min followed by 10 further mins at 3500 rpm after removal of the red cell layer; 5 mL of platelet-poor plasma injected in the scalp | No (naïve or 3 months wash-out) | NA-PRP followed by topical minoxidil 1/day for 12 weeks (Group A) vs. topical minoxidil 1/day for 12 weeks then PRP (Group B) | I-II | NR | 20 | Randomized, controlled, comparative trial | 2020 | Bruce, AJ | ||||
| Scalp tightness (N = 10); swelling, redness, post-injection bleeding, tingling (Group A and B) | Significant improvement in hair density in Group A compared to no improvement in Group B; significant improvement in mean hair diameter and global clinical appearance in Group A compared to Group B | Hair density; hair diameter; global clinical appearance | Global photography; magnified photography | 24 | 3 | Every 4 weeks | 22 mL of peripheral blood in Eclipse® PRP system centrifuged at 3500 rpm for 10 min; removal of the uppermost platelet-poor plasma suprenatant; remaining 4.0 mL platelet-rich plasma injected in the scalp | No (naïve or 12 months wash-out) | NA-L-PRP (Group A) vs. saline injections (Group B) | I-II-III | 50 (27–85) | 30 | Randomized, controlled trial | 2020 | Dubin, D | ||||||
| / | / | Severity of alopecia; hair count; hair density; hair diameter; anagen to telogen ratio; vellus to terminal ratio; patient’s | quality of life | Severity of Alopecia Tool (SALT) score; phototrichogram using Trichoscan® software; Dermatology Life Quality Index (DLQI) | 64 (ensimated) | / | / | / | No (naïve or 2 weeks washout) | / | / | / | 16 (enstimated) | Randomized, double blind, intrapatient, split-side, placebo control trial | 2021 (enstimated) | ClinicalTrials.gov identifier: NCT03474718 | |||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mercuri, S.R.; Paolino, G.; Di Nicola, M.R.; Vollono, L. Investigating the Safety and Efficacy of Platelet-Rich Plasma (PRP) Treatment for Female Androgenetic Alopecia: Review of the Literature. Medicina 2021, 57, 311. https://doi.org/10.3390/medicina57040311
Mercuri SR, Paolino G, Di Nicola MR, Vollono L. Investigating the Safety and Efficacy of Platelet-Rich Plasma (PRP) Treatment for Female Androgenetic Alopecia: Review of the Literature. Medicina. 2021; 57(4):311. https://doi.org/10.3390/medicina57040311
Chicago/Turabian StyleMercuri, Santo Raffaele, Giovanni Paolino, Matteo Riccardo Di Nicola, and Laura Vollono. 2021. "Investigating the Safety and Efficacy of Platelet-Rich Plasma (PRP) Treatment for Female Androgenetic Alopecia: Review of the Literature" Medicina 57, no. 4: 311. https://doi.org/10.3390/medicina57040311
APA StyleMercuri, S. R., Paolino, G., Di Nicola, M. R., & Vollono, L. (2021). Investigating the Safety and Efficacy of Platelet-Rich Plasma (PRP) Treatment for Female Androgenetic Alopecia: Review of the Literature. Medicina, 57(4), 311. https://doi.org/10.3390/medicina57040311







