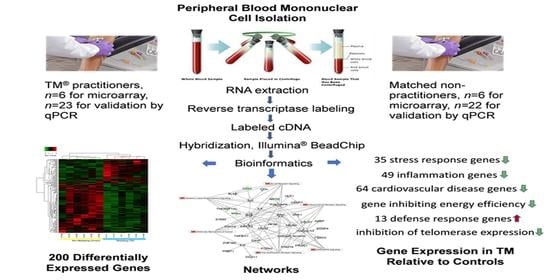
Transcriptomics of Long-Term Meditation Practice: Evidence for Prevention or Reversal of Stress Effects Harmful to Health
Abstract
1. Introduction
2. Materials and Methods
2.1. Research Design and Participants
2.2. PBMC Preparation
2.3. RNA Extraction, Concentration Measurement, and Integrity Check
2.4. Whole-Genome mRNA Expression Using Bead-Based Array
2.5. qPCR Analysis
3. Results
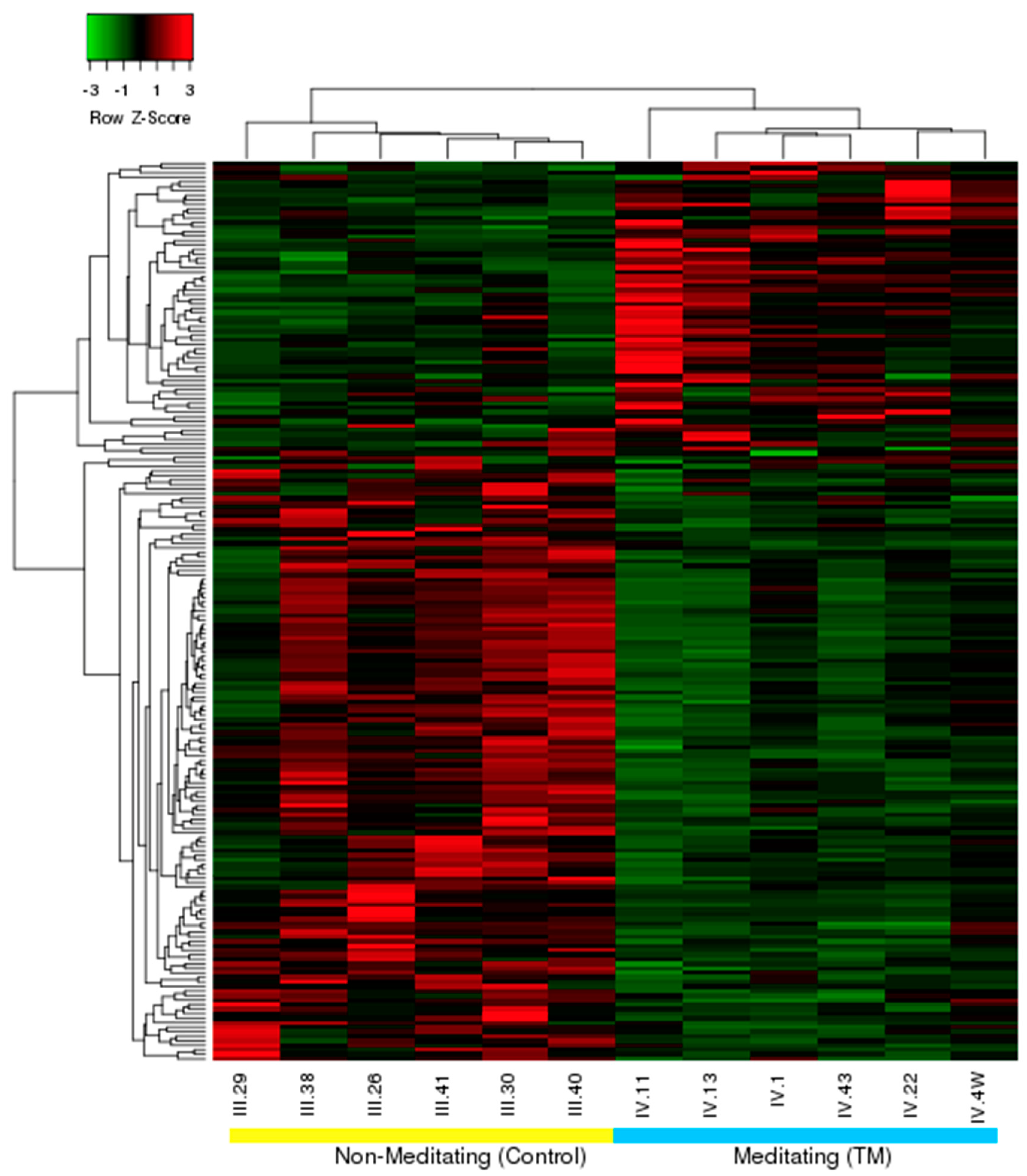
3.1. Differential Expression, Heat Map, and Clustering Analysis from the Microarray Study
3.2. qPCR Validation of Microarray Data
3.3. Top Networks
3.4. Gene Ontological Process Terms
3.5. Gene Classification Based on Associated Disease
3.6. Top Genes Upregulated in the Control Group, and Erythropoiesis-Related Genes
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- McEwen, B.S.; Akil, H. Revisiting the Stress Concept: Implications for Affective Disorders. J. Neurosci. 2020, 40, 12–21. [Google Scholar] [CrossRef] [PubMed]
- Juster, R.P.; McEwen, B.S.; Lupien, S.J. Allostatic load biomarkers of chronic stress and impact on health and cognition. Neurosci. Biobehav. Rev. 2010, 35, 2–16. [Google Scholar] [CrossRef]
- Bairey Merz, C.N.; Dwyer, J.; Nordstrom, C.K.; Walton, K.G.; Salerno, J.W.; Schneider, R.H. Psychosocial stress and cardiovascular disease: Pathophysiological links. Behav. Med. 2002, 27, 141–147. [Google Scholar] [CrossRef] [PubMed]
- Karlamangla, A.S.; Singer, B.H.; Seeman, T.E. Reduction in allostatic load in older adults is associated with lower all-cause mortality risk: MacArthur studies of successful aging. Psychosom. Med. 2006, 68, 500–507. [Google Scholar] [CrossRef] [PubMed]
- Wiley, J.F.; Gruenewald, T.L.; Karlamangla, A.S.; Seeman, T.E. Modeling multisystem physiological dysregulation. Psychosom. Med. 2016, 78, 290–301. [Google Scholar] [CrossRef]
- Hovatta, I.; Juhila, J.; Donner, J. Oxidative stress in anxiety and comorbid disorders. Neurosci. Res. 2010, 68, 261–275. [Google Scholar] [CrossRef]
- Palta, P.; Samuel, L.J.; Miller, E.R., 3rd; Szanton, S.L. Depression and oxidative stress: Results from a meta-analysis of observational studies. Psychosom. Med. 2014, 76, 12–19. [Google Scholar] [CrossRef]
- Shear, J. Transcendental Meditation. In The Experience of Meditation: Experts Introduce the Major Traditions, 1st ed.; Shear, J., Ed.; Paragon House: St. Paul, MN, USA, 2006; pp. 23–48. [Google Scholar]
- Forem, J. Transcendental Meditation: The Essential Teachings of Maharishi Mahesh Yogi; Hay House, Inc.: New York, NY, USA, 2012; p. 361. [Google Scholar]
- Wallace, R.K. Physiological effects of transcendental meditation. Science 1970, 167, 1751–1754. [Google Scholar] [CrossRef] [PubMed]
- Rees, B.; Travis, F.; Shapiro, D.; Chant, R. Reduction in posttraumatic stress symptoms in Congolese refugees practicing transcendental meditation. J. Trauma. Stress 2013, 26, 295–298. [Google Scholar] [CrossRef] [PubMed]
- Nidich, S.; Mills, P.J.; Rainforth, M.; Heppner, P.; Schneider, R.H.; Rosenthal, N.E.; Salerno, J.; Gaylord-King, C.; Rutledge, T. Non-trauma-focused meditation versus exposure therapy in veterans with post-traumatic stress disorder: A randomised controlled trial. Lancet Psychiatry 2018, 5, 975–986. [Google Scholar] [CrossRef]
- Bandy, C.L.; Dillbeck, M.C.; Sezibera, V.; Taljaard, L.; Wilks, M.; Shapiro, D.; de Reuck, J.; Peycke, R. Reduction of PTSD in South African university students using Transcendental Meditation practice. Psychol. Rep. 2019. [Google Scholar] [CrossRef]
- Orme-Johnson, D.W.; Barnes, V.A. Effects of the transcendental meditation technique on trait anxiety: A meta-analysis of randomized controlled trials. J. Altern. Complement. Med. 2013, 20, 330–341. [Google Scholar] [CrossRef]
- Barnes, V.A.; Orme-Johnson, D.W. Prevention and treatment of cardiovascular disease in adolescents and adults through the Transcendental Meditation((R)) Program: A research review update. Curr. Hypertens. Rev. 2012, 8, 227–242. [Google Scholar] [CrossRef]
- Schneider, R.H.; Grim, C.E.; Rainforth, M.V.; Kotchen, T.; Nidich, S.I.; Gaylord-King, C.; Salerno, J.W.; Kotchen, J.M.; Alexander, C.N. Stress reduction in the secondary prevention of cardiovascular disease: Randomized, controlled trial of transcendental meditation and health education in Blacks. Circ. Cardiovasc. Qual. Outcomes 2012, 5, 750–758. [Google Scholar] [CrossRef] [PubMed]
- Walton, K.G.; Schneider, R.H.; Nidich, S.I.; Salerno, J.W.; Nordstrom, C.K.; Bairey Merz, C.N. Psychosocial stress and cardiovascular disease Part 2: Effectiveness of the Transcendental Meditation program in treatment and prevention. Behav. Med. 2002, 28, 106–123. [Google Scholar] [CrossRef] [PubMed]
- Nidich, S.I.; Rainforth, M.V.; Haaga, D.A.; Hagelin, J.; Salerno, J.W.; Travis, F.; Tanner, M.; Gaylord-King, C.; Grosswald, S.; Schneider, R.H. A randomized controlled trial on effects of the Transcendental Meditation program on blood pressure, psychological distress, and coping in young adults. Am. J. Hypertens. 2009, 22, 1326–1331. [Google Scholar] [CrossRef]
- Orme-Johnson, D. Medical care utilization and the transcendental meditation program. Psychosom. Med. 1987, 49, 493–507. [Google Scholar] [CrossRef]
- Herron, R.E. Changes in physician costs among high-cost transcendental meditation practitioners compared with high-cost nonpractitioners over 5 years. Am. J. Health Promot. 2011, 26, 56–60. [Google Scholar] [CrossRef] [PubMed]
- Travis, F.; Shear, J. Focused attention, open monitoring and automatic self-transcending: Categories to organize meditations from Vedic, Buddhist and Chinese traditions. Conscious. Cogn. 2010, 19, 1110–1118. [Google Scholar] [CrossRef]
- Mahone, M.C.; Travis, F.; Gevirtz, R.; Hubbard, D. fMRI during Transcendental Meditation practice. Brain Cogn. 2018, 123, 30–33. [Google Scholar] [CrossRef]
- Miller, G.E.; Murphy, M.L.; Cashman, R.; Ma, R.; Ma, J.; Arevalo, J.M.; Kobor, M.S.; Cole, S.W. Greater inflammatory activity and blunted glucocorticoid signaling in monocytes of chronically stressed caregivers. Brain Behav. Immun. 2014, 41, 191–199. [Google Scholar] [CrossRef]
- Cole, S.W. Elevating the perspective on human stress genomics. Psychoneuroendocrinology 2010, 35, 955–962. [Google Scholar] [CrossRef]
- Powell, N.D.; Sloan, E.K.; Bailey, M.T.; Arevalo, J.M.; Miller, G.E.; Chen, E.; Kobor, M.S.; Reader, B.F.; Sheridan, J.F.; Cole, S.W. Social stress up-regulates inflammatory gene expression in the leukocyte transcriptome via beta-adrenergic induction of myelopoiesis. Proc. Natl. Acad. Sci. USA 2013, 110, 16574–16579. [Google Scholar] [CrossRef]
- Cole, S.W. Human social genomics. PLoS Genet. 2014, 10, e1004601. [Google Scholar] [CrossRef] [PubMed]
- Picard, M.; McEwen, B.S.; Epel, E.S.; Sandi, C. An energetic view of stress: Focus on mitochondria. Front. Neuroendocrinol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Picard, M.; McManus, M.J.; Gray, J.D.; Nasca, C.; Moffat, C.; Kopinski, P.K.; Seifert, E.L.; McEwen, B.S.; Wallace, D.C. Mitochondrial functions modulate neuroendocrine, metabolic, inflammatory, and transcriptional responses to acute psychological stress. Proc. Natl. Acad. Sci. USA 2015, 112, E6614–E6623. [Google Scholar] [CrossRef]
- Picard, M.; Prather, A.A.; Puterman, E.; Cuillerier, A.; Coccia, M.; Aschbacher, K.; Burelle, Y.; Epel, E.S. A Mitochondrial Health Index Sensitive to Mood and Caregiving Stress. Biol. Psychiatry 2018. [Google Scholar] [CrossRef] [PubMed]
- Kauppila, T.E.S.; Kauppila, J.H.K.; Larsson, N.G. Mammalian Mitochondria and Aging: An Update. Cell Metab. 2017, 25, 57–71. [Google Scholar] [CrossRef] [PubMed]
- Picard, M.; McEwen, B.S. Psychological Stress and Mitochondria: A Systematic Review. Psychosom. Med. 2018, 80, 141–153. [Google Scholar] [CrossRef]
- Black, D.S.; Cole, S.W.; Irwin, M.R.; Breen, E.; St Cyr, N.M.; Nazarian, N.; Khalsa, D.S.; Lavretsky, H. Yogic meditation reverses NF-kappaB and IRF-related transcriptome dynamics in leukocytes of family dementia caregivers in a randomized controlled trial. Psychoneuroendocrinology 2013, 38, 348–355. [Google Scholar] [CrossRef]
- Epel, E.S.; Puterman, E.; Lin, J.; Blackburn, E.H.; Lum, P.Y.; Beckmann, N.D.; Zhu, J.; Lee, E.; Gilbert, A.; Rissman, R.A.; et al. Meditation and vacation effects have an impact on disease-associated molecular phenotypes. Transl. Psychiatry 2016, 6, e880. [Google Scholar] [CrossRef] [PubMed]
- Bhasin, M.K.; Denninger, J.W.; Huffman, J.C.; Joseph, M.G.; Niles, H.; Chad-Friedman, E.; Goldman, R.; Buczynski-Kelley, B.; Mahoney, B.A.; Fricchione, G.L.; et al. Specific Transcriptome Changes Associated with Blood Pressure Reduction in Hypertensive Patients After Relaxation Response Training. J. Altern. Complement. Med. 2018, 24, 486–504. [Google Scholar] [CrossRef]
- Chaix, R.; Alvarez-Lopez, M.J.; Fagny, M.; Lemee, L.; Regnault, B.; Davidson, R.J.; Lutz, A.; Kaliman, P. Epigenetic clock analysis in long-term meditators. Psychoneuroendocrinology 2017, 85, 210–214. [Google Scholar] [CrossRef] [PubMed]
- Buric, I.; Farian, M.; Jong, J.; Mee, C.; Brazil, I.A. What is the molecular signature of mind-body interventions? A systematic review of gene expression changes induced by meditation and related practices. Front. Immunol. 2017, 8, 1–17. [Google Scholar] [CrossRef]
- Kaliman, P. Epigenetics and meditation. Curr. Opin. Psychol. 2019, 28, 76–80. [Google Scholar] [CrossRef]
- Jaksik, R.; Iwanaszko, M.; Rzeszowska-Wolny, J.; Kimmel, M. Microarray experiments and factors which affect their reliability. Biol. Direct. 2015, 10, 46. [Google Scholar] [CrossRef]
- Okamoto, T.; Okabe, S. Ultraviolet absorbance at 260 and 280 nm in RNA measurement is dependent on measurement solution. Int. J. Mol. Med. 2000, 5, 657–659. [Google Scholar] [CrossRef]
- Du, P.; Kibbe, W.A.; Lin, S.M. lumi: A pipeline for processing Illumina microarray. Bioinformatics 2008, 24, 1547–1548. [Google Scholar] [CrossRef] [PubMed]
- Galili, T. dendextend: An R package for visualizing, adjusting and comparing trees of hierarchical clustering. Bioinformatics 2015, 31, 3718–3720. [Google Scholar] [CrossRef] [PubMed]
- Kramer, A.; Green, J.; Pollard, J., Jr.; Tugendreich, S. Causal analysis approaches in Ingenuity Pathway Analysis. Bioinformatics 2014, 30, 523–530. [Google Scholar] [CrossRef]
- Da Huang, W.; Sherman, B.T.; Lempicki, R.A. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat. Protoc. 2009, 4, 44–57. [Google Scholar] [CrossRef]
- Schmittgen, T.D.; Livak, K.J. Analyzing real-time PCR data by the comparative C(T) method. Nat. Protoc. 2008, 3, 1101–1108. [Google Scholar] [CrossRef] [PubMed]
- Miller, G.E.; Chen, E.; Sze, J.; Marin, T.; Arevalo, J.M.; Doll, R.; Ma, R.; Cole, S.W. A functional genomic fingerprint of chronic stress in humans: Blunted glucocorticoid and increased NF-kappaB signaling. Biol. Psychiatry 2008, 64, 266–272. [Google Scholar] [CrossRef]
- MacLean, C.R.; Walton, K.G.; Wenneberg, S.R.; Levitsky, D.K.; Mandarino, J.P.; Waziri, R.; Hillis, S.L.; Schneider, R.H. Effects of the Transcendental Meditation program on adaptive mechanisms: Changes in hormone levels and responses to stress after 4 months of practice. Psychoneuroendocrinology 1997, 22, 277–295. [Google Scholar] [CrossRef]
- Walton, K.G.; Pugh, N.D.; Gelderloos, P.; Macrae, P. Stress reduction and preventing hypertension: Preliminary support for a psychoneuroendocrine mechanism. J. Altern. Complement. Med. 1995, 1, 263–283. [Google Scholar] [CrossRef] [PubMed]
- Jevning, R.; Wallace, R.K.; Beidebach, M. The physiology of meditation: A review. A wakeful hypometabolic integrated response. Neurosci. Biobehav. Rev. 1992, 16, 415–424. [Google Scholar] [CrossRef]
- Chaves de Souza, J.A.; Nogueira, A.V.; Chaves de Souza, P.P.; Kim, Y.J.; Silva Lobo, C.; Pimentel Lopes de Oliveira, G.J.; Cirelli, J.A.; Garlet, G.P.; Rossa, C., Jr. SOCS3 expression correlates with severity of inflammation, expression of proinflammatory cytokines, and activation of STAT3 and p38 MAPK in LPS-induced inflammation in vivo. Mediators Inflamm. 2013, 2013, 650812. [Google Scholar] [CrossRef]
- Kuparinen, T.; Marttila, S.; Jylhava, J.; Tserel, L.; Peterson, P.; Jylha, M.; Hervonen, A.; Hurme, M. Cytomegalovirus (CMV)-dependent and -independent changes in the aging of the human immune system: A transcriptomic analysis. Exp. Gerontol. 2013, 48, 305–312. [Google Scholar] [CrossRef]
- McEwen, B.S.; Stellar, E. Stress and the individual. Mechanisms leading to disease. Arch. Intern. Med. 1993, 153, 2093–2101. [Google Scholar] [CrossRef]
- Jevning, R.; Wilson, A.F.; Pirkle, H.; O’Halloran, J.P.; Walsh, R.N. Metabolic control in a state of decreased activation: Modulation of red cell metabolism. Am. J. Physiol. 1983, 245, C457–C461. [Google Scholar] [CrossRef]
- Steinberg, G.R.; McAinch, A.J.; Chen, M.B.; O’Brien, P.E.; Dixon, J.B.; Cameron-Smith, D.; Kemp, B.E. The suppressor of cytokine signaling 3 inhibits leptin activation of AMP-kinase in cultured skeletal muscle of obese humans. J. Clin. Endocrinol. Metab. 2006, 91, 3592–3597. [Google Scholar] [CrossRef]
- Dzamko, N.L.; Steinberg, G.R. AMPK-dependent hormonal regulation of whole-body energy metabolism. Acta Physiol. 2009, 196, 115–127. [Google Scholar] [CrossRef]
- Herzig, S.; Shaw, R.J. AMPK: Guardian of metabolism and mitochondrial homeostasis. Nat. Rev. Mol. Cell Biol. 2018, 19, 121–135. [Google Scholar] [CrossRef]
- Yang, Z.; Hulver, M.; McMillan, R.P.; Cai, L.; Kershaw, E.E.; Yu, L.; Xue, B.; Shi, H. Regulation of insulin and leptin signaling by muscle suppressor of cytokine signaling 3 (SOCS3). PLoS ONE 2012, 7, e47493. [Google Scholar] [CrossRef]
- Cao, L.; Wang, Z.; Wan, W. Suppressor of Cytokine Signaling 3: Emerging role linking central insulin resistance and Alzheimer’s disease. Front. Neurosci. 2018, 12, 417. [Google Scholar] [CrossRef]
- Carow, B.; Rottenberg, M.E. SOCS3, a Major Regulator of Infection and Inflammation. Front. Immunol. 2014, 5, 58. [Google Scholar] [CrossRef]
- Wegrzyn, J.; Potla, R.; Chwae, Y.J.; Sepuri, N.B.; Zhang, Q.; Koeck, T.; Derecka, M.; Szczepanek, K.; Szelag, M.; Gornicka, A.; et al. Function of mitochondrial Stat3 in cellular respiration. Science 2009, 323, 793–797. [Google Scholar] [CrossRef] [PubMed]
- Park, H.Y.; Hwang, H.; Park, J.; Lee, S.; Lim, K. The effects of altitude/hypoxic training on oxygen delivery capacity of the blood and aerobic exercise capacity in elite athletes—A meta-analysis. J. Exerc. Nutrition Biochem. 2016, 20, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Nogueira-Pedro, A.; dos Santos, G.G.; Oliveira, D.C.; Hastreiter, A.A.; Fock, R.A. Erythropoiesis in vertebrates: From ontogeny to clinical relevance. Front. Biosci. 2016, 8, 100–112. [Google Scholar]
- Rogers, H.; Wang, L.; Yu, X.; Alnaeeli, M.; Cui, K.; Zhao, K.; Bieker, J.J.; Prchal, J.; Huang, S.; Weksler, B.; et al. T-cell acute leukemia 1 (TAL1) regulation of erythropoietin receptor and association with excessive erythrocytosis. J. Biol. Chem. 2012, 287, 36720–36731. [Google Scholar] [CrossRef]
- De Punder, K.; Heim, C.; Wadhwa, P.D.; Entringer, S. Stress and immunosenescence: The role of telomerase. Psychoneuroendocrinology 2019, 101, 87–100. [Google Scholar] [CrossRef] [PubMed]
- Terme, J.M.; Mocquet, V.; Kuhlmann, A.S.; Zane, L.; Mortreux, F.; Wattel, E.; Duc Dodon, M.; Jalinot, P. Inhibition of the hTERT promoter by the proto-oncogenic protein TAL1. Leukemia 2009, 23, 2081–2089. [Google Scholar] [CrossRef] [PubMed]
- Wenuganen, S.; Walton, K.G.; Travis, F.; Wallace, R.K.; Fagan, J. (Maharishi International University, Fairfield, IA, USA); Stalder, T.; Kirschbaum, C. (Technical University of Dresden, Dresden, Germany); Shrivastava, M. (Uniformed Services University of the Health Sciences, Bethesda, MD, USA). Personal communication, 2020.


| Demographic Variables | Control Group | TM Group |
|---|---|---|
| n | 6 | 6 |
| Age (years ± SD) | 65.0 ± 4.9 | 64.5 ± 5.4 |
| Sex (number of males) | 4 | 5 |
| Non-vegetarians (number of) | 5 | 5 |
| Non-smokers (number of) | 6 | 6 |
| Non-drinkers (number of) | 6 | 6 |
| Moderate exercisers (number of) | 6 | 6 |
| Subjective SES * (mean) | 3.0 | 3.1 |
| Demographic Variables | Control Group | TM Group |
|---|---|---|
| n | 22 | 23 |
| Age (years ± SD) | 62.2 ± 4.64 | 63.6 ± 3.92 |
| Sex (number of males) | 10 | 14 |
| Non-vegetarians (number of) | 20 | 8 |
| Non-smokers (number of) | 17 | 23 |
| Non-drinkers (number of) | 17 | 21 |
| Moderate exercisers (number of) | 18 | 23 |
| Subjective SES * | N/A | N/A |
| Microarray Output (N = 12) | qPCR Output (N = 10 *) | |||
|---|---|---|---|---|
| Genes Downregulated in the TM Group Relative to the Control Group | ||||
| Gene | p-Value | Expression Ratio ** | p-Value | “Fold Difference” *** |
| AHSP | 0.002 | 5.45 | <0.001 | 14.58 |
| ALOX12 | 0.006 | 2.45 | 0.071 | 2.76 |
| CD22 | 0.037 | 2.48 | 0.248 | 2.01 |
| ITGB3 | 0.013 | 2.11 | 0.015 | 3.15 |
| ITGB5 | 0.006 | 2.00 | 0.019 | 2.47 |
| LMNA | 0.004 | 2.16 | 0.063 | 2.35 |
| MYL9 | 0.017 | 2.88 | 0.161 | 2.01 |
| SOCS3 | 0.030 | 2.33 | 0.003 | 6.50 |
| TAL1 | 0.002 | 2.36 | 0.073 | 2.19 |
| Genes Upregulated in the TM Group Relative to the Control Group | ||||
| CDKN1C | 0.017 | 2.79 | 0.003 | 2.28 |
| CXCL10 | 0.014 | 2.89 | 0.084 | 2.33 |
| HES4 | 0.047 | 2.00 | 0.748 | 1.12 |
| IGFBP7 | 0.026 | 2.08 | 0.449 | 1.43 |
| IL1B | 0.012 | 3.54 | 0.126 | 1.77 |
| TLR4 | 0.002 | 2.23 | 0.741 | 1.11 |
| Genes Downregulated in the TM Group Relative to the Control Group | ||||||||
|---|---|---|---|---|---|---|---|---|
| Gene | Group | Mean Ct | Std. Dev. | N | df | F | p-Value | “Fold Difference” * |
| AHSP | Control TM | 32.5668 34.5163 | 2.1561 1.4674 | 22 23 | 1 | 12.674 | 0.001 | 3.86 |
| ALOX12 | Control TM | 30.6055 31.1267 | 0.7140 1.1756 | 22 23 | 1 | 3.194 | 0.081 | 1.44 |
| CD22 | Control TM | 30.7102 31.0753 | 0.8499 1.1344 | 22 23 | 1 | 1.482 | 0.230 | 1.29 |
| ITGB3 | Control TM | 26.4452 27.0511 | 0.7939 1.1830 | 22 23 | 1 | 4.031 | 0.051 | 1.52 |
| ITGB5 | Control TM | 30.9789 31.4723 | 0.6233 0.7281 | 22 23 | 1 | 5.938 | 0.019 | 1.41 |
| LMNA | Control TM | 29.8050 30.2638 | 0.7014 0.9137 | 22 23 | 1 | 3.546 | 0.066 | 1.37 |
| MYL9 | Control TM | 27.1693 27.5611 | 0.6926 1.1940 | 22 23 | 1 | 1.792 | 0.188 | 1.31 |
| SOCS3 | Control TM | 31.4033 32.5206 | 0.9121 1.3372 | 22 23 | 1 | 10.626 | 0.002 | 2.17 |
| TAL1 | Control TM | 30.7461 31.1823 | 0.6561 0.8969 | 22 23 | 1 | 3.441 | 0.070 | 1.35 |
| Genes Upregulated in the TM Group Relative to the Control Group | ||||||||
| CDKN1C | Control TM | 30.2448 29.9343 | 0.7156 0.7260 | 22 23 | 1 | 2.085 | 0.156 | 1.24 |
| CXCL10 | Control TM | 32.7365 31.7125 | 0.9466 0.9613 | 22 23 | 1 | 12.950 | 0.001 | 2.03 |
| HES4 | Control TM | 34.1132 34.3267 | 0.6820 0.7179 | 22 23 | 1 | 1.044 | 0.313 | 1.16 |
| IGFBP7 | Control TM | 29.8499 29.5057 | 0.8188 0.8956 | 22 23 | 1 | 1.805 | 0.186 | 1.27 |
| IL1B | Control TM | 31.4125 31.0576 | 1.0132 0.8328 | 22 23 | 1 | 1.655 | 0.205 | 1.28 |
| TLR4 | Control TM | 29.0808 28.8832 | 0.7732 0.7260 | 22 23 | 1 | 0.782 | 0.381 | 1.15 |
| Term | p-Value | Genes Downregulated in the TM Group |
| Blood Coagulation | 8.40 × 10−8 | F2RL3, CAV1, ITGB3, MMRN1, GP9, VWF, GP5, GP6, THBS1, TREML1, PROS1, ALOX12, ITGA2B, HBD |
| Cell Activation | 1.30 × 10−4 | F2RL3, CAV1, BCR, SNCA, TNFRSF13C, PAWR, ITGB3, GP9, VWF, GP5, GP6, THBS1, TREML1, WNT7A, ALOX12, ITGA2B |
| Response to Stress | 1.70 × 10−4 | SLC8A3, F2RL3, CAV1, CDC14B, SNCA, SLC6A4, FSTL1, PAWR, ITGB3, MMRN1, TRIM10, GP9, GP5, ALAS2, GP6, DDX11, PLOD2, CFH, MGLL, THBS1, HBD, BCR, PTPRF, SOCS3, LMNA, HBA2, HBA1, PTPRN, VWF, SH2D3C, TGFB1I1, TREML1, PROS1, ITGA2B, ALOX12 |
| Exocytosis | 4.00 × 10−4 | VWF, ANK1, BCR, SNCA, SYTL4, ITGB3, THBS1, MMRN1, PROS1, ITGA2B |
| Cell Adhesion | 8.70 × 10−4 | CAV1, PTPRF, CALD1, TNFRSF13C, ITGB5, ITGB3, PAWR, MMRN1, GP9, KIFC3, VWF, GP5, CD22, CNTNAP2, SGCE, TGFB1I1, LAMC1, THBS1, JAM3, ALOX12, ITGA2B |
| Hematopoiesis | 1.30 × 10−3 | TAL1, ALAS2, AHSP, BCL11A, ZNF160 |
| Term | p-Value | Genes Upregulated in the TM Group |
| Defense Response | 2.40 × 10−4 | OAS1, CCL4L1, CXCL10, MICA, CASP5, FPR2, IFIT3, IL1B, LILRB2, METRNL, PTPN2, TLR4, VNN1 |
| Response to External Stimuli | 3.30 × 10−4 | CCL4L1, CXCL10, MICA, ATF3, BATF3, CASP5, FPR2, IFIT3, IL1B, LILRB2, METRNL, KCNJ2, PTPN2, TLR4 |
| Inflammatory Response | 9.30 × 10−4 | CCL4L1, CXCL10, FPR2, IL1B, METRNL, PTPN2, TLR4, VNN1 |
| Immune System Processes | 1.20 × 10−3 | OAS1, CCL4L1, CXCL10, FCGR3B, MICA, FPR2, IFIT3, IL1B, LILRB2, PTPN2, TLR4, VNN1 |
| Homeostatic Processes | 1.90 × 10−3 | CXCL10, CKB, FPR2, IL1B, METRNL, KCNJ2, PTPN2, SLC31A2, SLC4A8, TLR4, UTS2 |
| Cell Chemotaxis | 2.50 × 10−2 | GPR44, CCRL2, FPR2, CXCL10 |
| Disease | p-Value | Genes |
|---|---|---|
| Hematologic Diseases | 1.5 × 10−10 | TRIM10, VPREB3, CFH, MGP, F2RL3, AHSP, TAL1, ITGB3, HBD, GATM, HRASLS, TMCC2, OSBP2, TNFRSF13C, PTCRA, DMTN, GP9, TNS1, CAV1, JAM3, CMTM5, BCL11A, SOCS3, SNCA, HBA2, SH2D3C, PTPRN, NR1I2, TGFB1I1, HDC, HBM, DDX11, PAWR, ITGB5, LMNA, ITGA2B, CABP5, THBS1, FOXP4, HBA1, ANK1, GP5, MAP1A, SLC35D3, CALD1, CD22, SLC4A1, BCR, LAMC1, ALOX12, HOMER2, TREML1, ALAS2, CA1, ABCB4, XK, EBF1, PTPRF, MMRN1, PLOD2, VWF, GP6 |
| Coronary Artery Disease | 9.7 × 10−8 | CFH, MGP, F2RL3, ITGB3, MGLL, TNFRSF13C, CAV1, JAM3, SOCS3, PNOC, FSTL1, NDUFAF3, NR1I2, LMNA, ITGA2B, THBS1, HBA1, GP5, CALD1, PEAR1, EBF1, PTPRF, SLC6A4, VWF, GP6, FHL1 |
| Diabetes Complications | 2.2 × 10−7 | CFH, MGP, F2RL3, ITGB3, GATM, SGCE, DMTN, CAV1, SOCS3, PNOC, FSTL1, HBA2, NDUFAF3, PTPRN, TGFB1I1, HDC, CNTNAP2, LMNA, ITGA2B, THBS1, SELENBP1, HBA1, ANK1, GP5, CALD1, CD22, SLC4A1, LAMC1, ALOX12, CA1, PTPRF, SLC6A4, VWF, GP6 |
| Inflammation | 7.1 × 10−6 | CFH, MGP, CTDSPL, F2RL3, TAL1, ITGB3, MGLL, GATM, HRASLS, SLC8A3, TNFRSF13C, DMTN, WNT7A, GP9, CAV1, JAM3, SOCS3, SNCA, PNOC, FSTL1, HBA2, SH2D3C, PTPRN, NR1I2, HDC, PAWR, ITGB5, LMNA, ITGA2B, THBS1, SELENBP1, HBA1, ANK1, GP5, MAP1A, CALD1, CD22, SLC4A1, BCR, ALOX12, TREML1, ALAS2, CA1, ABCB4, EBF1, PTPRF, SLC6A4, MMRN1, VWF |
| Cardiovascular Disease | 4.4 × 10−4 | VPREB3, CFH, MGP, CTDSPL, F2RL3, AHSP, TAL1, ITGB3, HBD, MGLL, GATM, HRASLS, SGCE, SLC8A3, TNFRSF13C, DMTN, WNT7A, GP9, CAV1, JAM3, CMTM5, SOCS3, SNCA, PNOC, FSTL1, HBA2, NDUFAF3, PTPRN, NR1I2, TGFB1I1, HDC, DDX11, PAWR, CNTNAP2, ITGB5, LMNA, ITGA2B, THBS1, SELENBP1, FOXP4, HBA1, ANK1, GP5, MAP1A, CALD1, CD22, SLC4A1, BCR, LAMC1, ALOX12, PEAR1, ALAS2, CA1, ABCB4, XK, EBF1, PTPRF, SLC6A4, MMRN1, PLOD2, ZNF160, VWF, GP6, FHL1 |
| Gene | Control Mean | Control SD | TM Mean | TM SD | Expression Ratio | p-Value |
|---|---|---|---|---|---|---|
| HBM | 93.53 | 57.09 | 9.53 | 15.76 | 9.8 | 0.004 |
| SLC4A1 | 72.47 | 49.02 | 8.13 | 10.54 | 8.9 | 0.002 |
| ALAS2 | 631.43 | 579.80 | 78.12 | 67.67 | 8.1 | 0.002 |
| CA1 | 85.37 | 77.98 | 12.40 | 9.03 | 6.9 | 0.002 |
| AHSP | 203.13 | 186.36 | 37.27 | 20.60 | 5.5 | 0.002 |
| HBD | 586.23 | 457.93 | 139.48 | 59.29 | 4.2 | 0.002 |
| GATA1 | 19.22 | 8.31 | 11.17 | 4.27 | 1.7 | 0.049 |
| GATA2 | 33.72 | 6.23 | 21.82 | 9.74 | 1.6 | 0.030 |
| TAL1 | 64.28 | 9.70 | 27.18 | 6.22 | 2.4 | 0.002 |
| EPOR | 83.67 | 11.01 | 70.27 | 14.03 | 1.2 | 0.108 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wenuganen, S.; Walton, K.G.; Katta, S.; Dalgard, C.L.; Sukumar, G.; Starr, J.; Travis, F.T.; Wallace, R.K.; Morehead, P.; Lonsdorf, N.K.; et al. Transcriptomics of Long-Term Meditation Practice: Evidence for Prevention or Reversal of Stress Effects Harmful to Health. Medicina 2021, 57, 218. https://doi.org/10.3390/medicina57030218
Wenuganen S, Walton KG, Katta S, Dalgard CL, Sukumar G, Starr J, Travis FT, Wallace RK, Morehead P, Lonsdorf NK, et al. Transcriptomics of Long-Term Meditation Practice: Evidence for Prevention or Reversal of Stress Effects Harmful to Health. Medicina. 2021; 57(3):218. https://doi.org/10.3390/medicina57030218
Chicago/Turabian StyleWenuganen, Supaya, Kenneth G. Walton, Shilpa Katta, Clifton L. Dalgard, Gauthaman Sukumar, Joshua Starr, Frederick T. Travis, Robert Keith Wallace, Paul Morehead, Nancy K. Lonsdorf, and et al. 2021. "Transcriptomics of Long-Term Meditation Practice: Evidence for Prevention or Reversal of Stress Effects Harmful to Health" Medicina 57, no. 3: 218. https://doi.org/10.3390/medicina57030218
APA StyleWenuganen, S., Walton, K. G., Katta, S., Dalgard, C. L., Sukumar, G., Starr, J., Travis, F. T., Wallace, R. K., Morehead, P., Lonsdorf, N. K., Srivastava, M., & Fagan, J. (2021). Transcriptomics of Long-Term Meditation Practice: Evidence for Prevention or Reversal of Stress Effects Harmful to Health. Medicina, 57(3), 218. https://doi.org/10.3390/medicina57030218