Correlation of Computed Tomography Parameters with Histology, Stage and Prognosis in Surgically Treated Thymomas
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Histopathologic and Staging Review
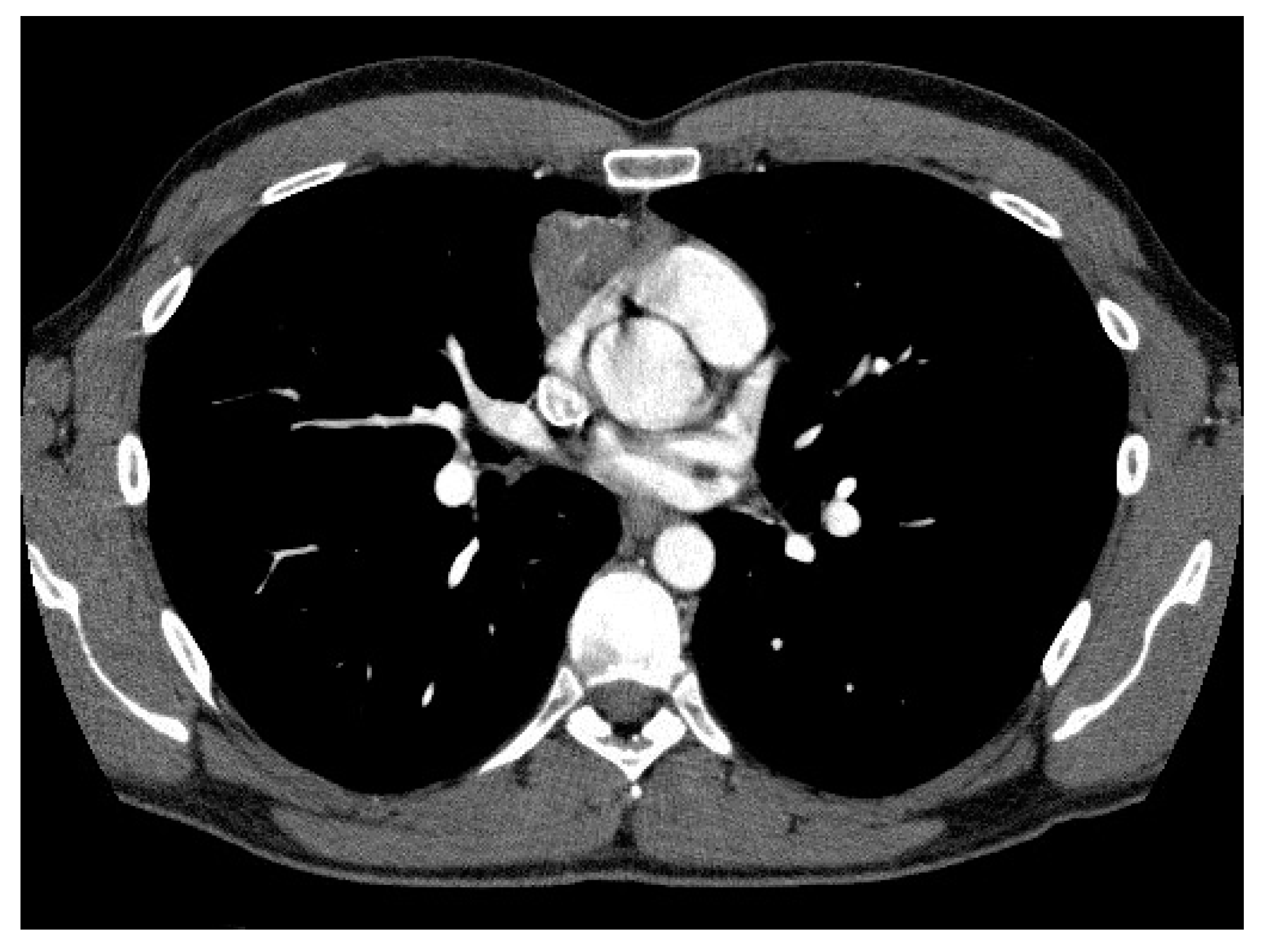
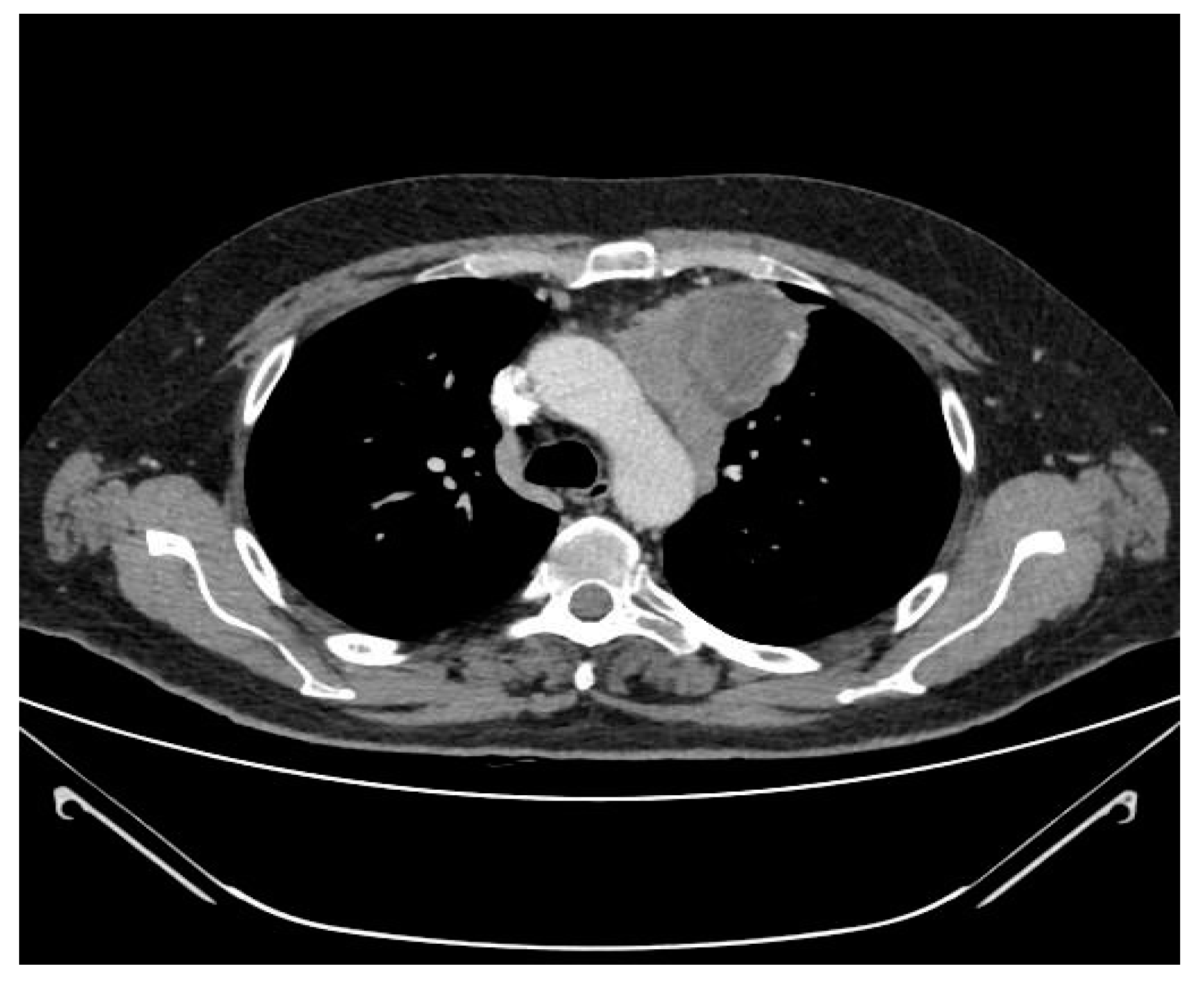
2.3. CT Scan Features and Image Interpretation
2.4. Statistical Analysis
3. Results
3.1. General Clinical Features
3.2. Correlation of Histology and Staging
3.3. Correlation of CT Scan Parameters, Histology and Staging
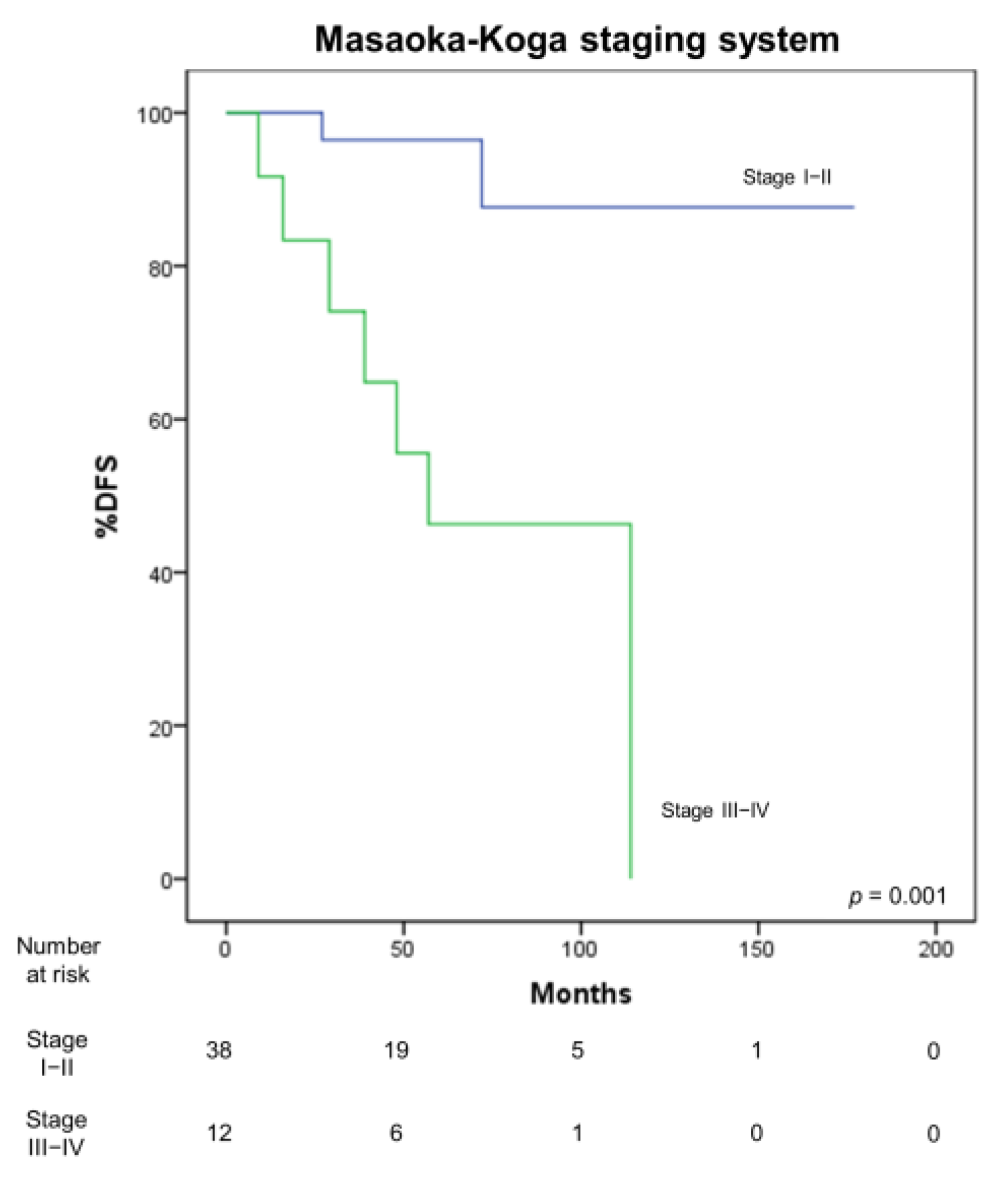
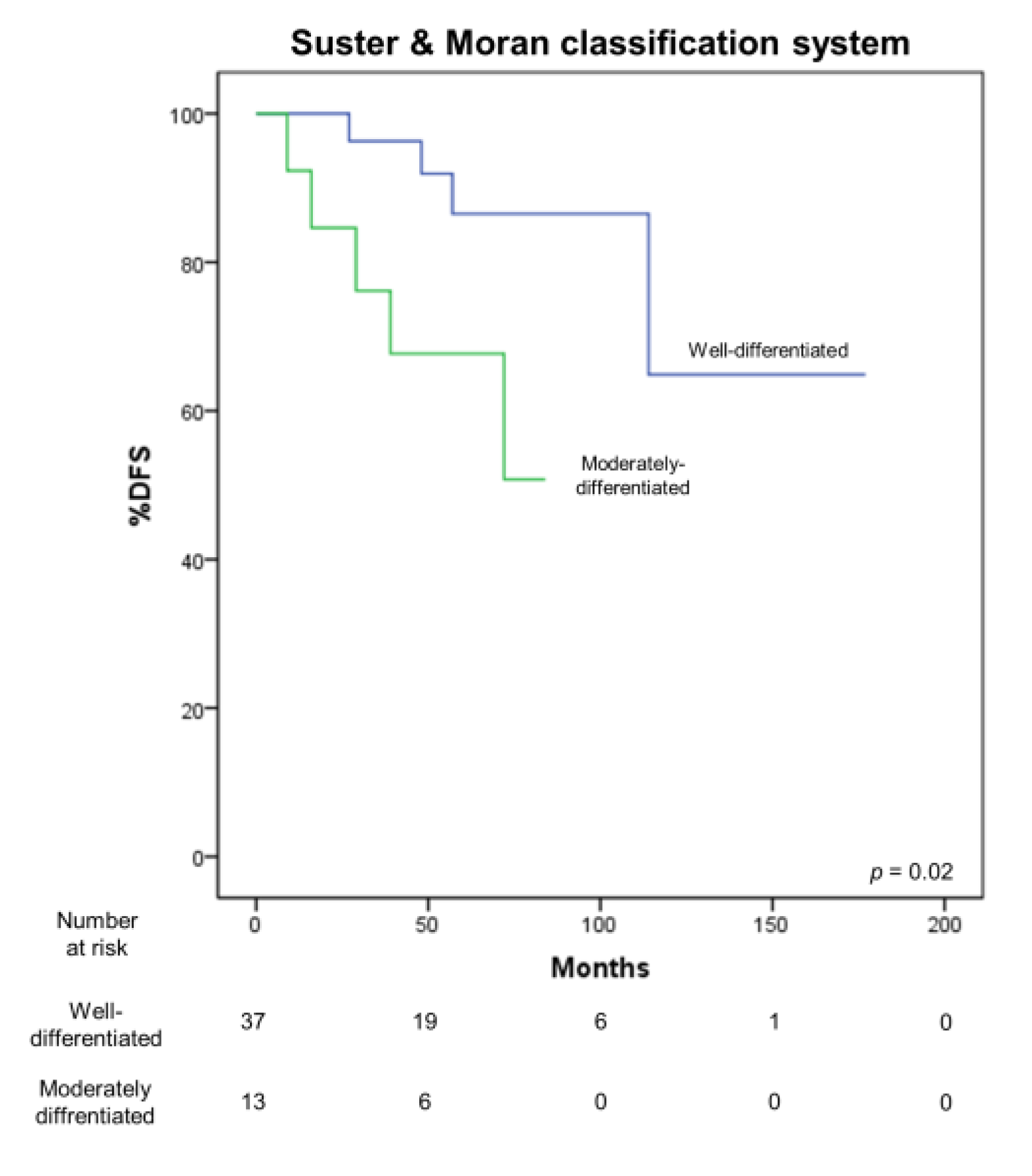
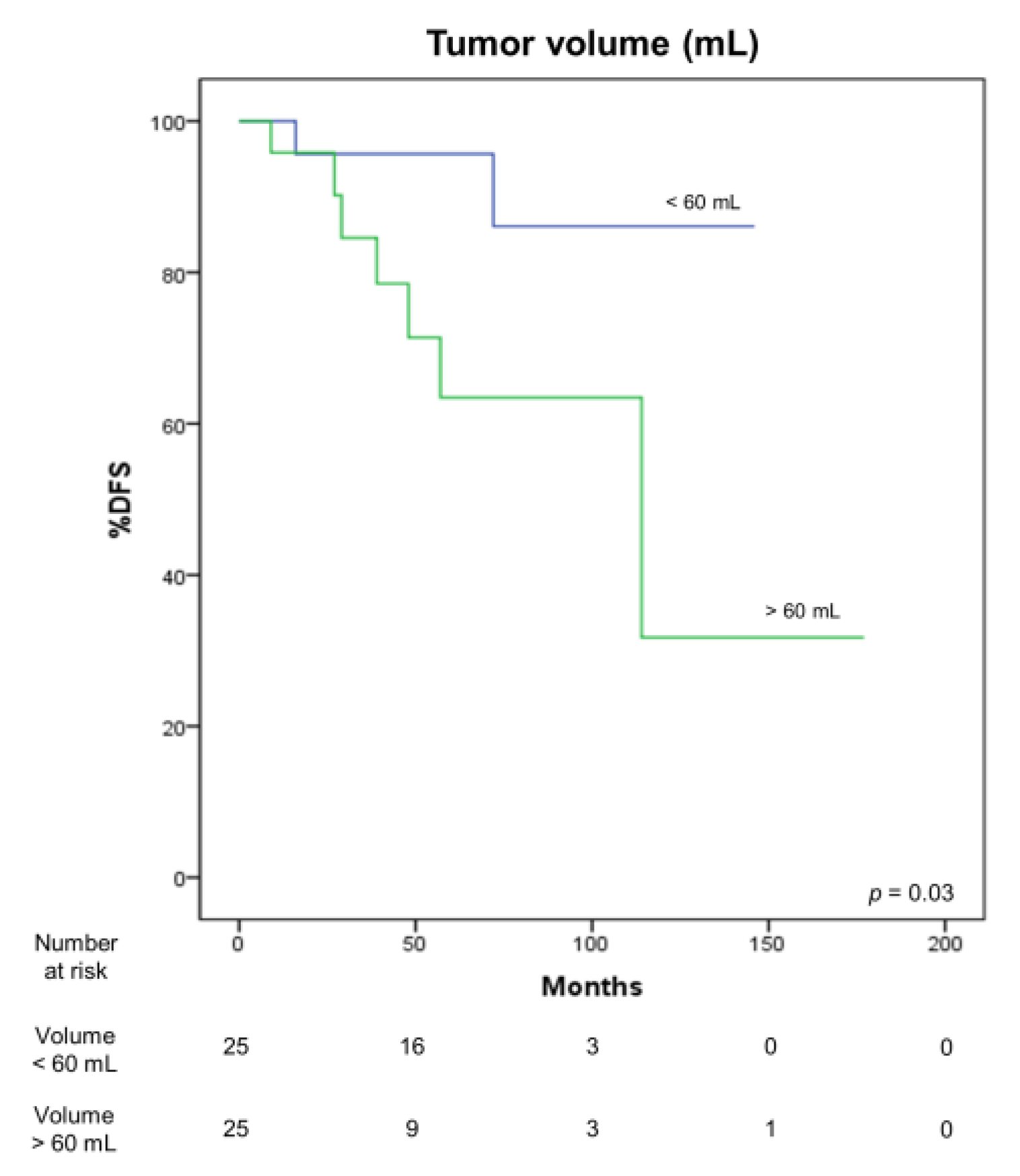
3.4. Correlation with Survival
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jamilloux, Y.; Frih, H.; Bernard, C.; Broussolle, C.; Petiot, P.; Girard, N.; Sève, P. Thymomes et maladies auto-immunes. Rev. Méd. Interne 2018, 39, 17–26. [Google Scholar] [CrossRef] [PubMed]
- Imbimbo, M.; Ottaviano, M.; Vitali, M.; Fabbri, A.; Leuzzi, G.; Fiore, M.; Franceschini, D.; Pasello, G.; Perrino, M.; Schiavon, M.; et al. Best practices for the management of thymic epithelial tumors: A position paper by the Italian collaborative group for ThYmic MalignanciEs (TYME). Cancer Treat. Rev. 2018, 71, 76–87. [Google Scholar] [CrossRef] [PubMed]
- Marx, A.; Chan, J.K.; Coindre, J.-M.; Detterbeck, F.; Girard, N.; Harris, N.L.; Jaffe, E.S.; Kurrer, M.O.; Marom, E.M.; Moreira, A.L.; et al. The 2015 World Health Organization Classification of Tumors of the Thymus: Continuity and Changes. J. Thorac. Oncol. 2015, 10, 1383–1395. [Google Scholar] [CrossRef] [PubMed]
- Marx, A.; Ströbel, P.; Badve, S.S.; Chalabreysse, L.; Chan, J.K.C.; Chen, G.; De Leval, L.; Detterbeck, F.C.; Girard, N.; Huang, J.; et al. ITMIG Consensus Statement on the Use of the WHO Histological Classification of Thymoma and Thymic Carcinoma: Refined Definitions, Histological Criteria, and Reporting. J. Thorac. Oncol. 2014, 9, 596–611. [Google Scholar] [CrossRef]
- Suster, S.; Moran, C.A. Primary thymic epithelial neoplasms: Current concepts and controversies. In Anatomic Pathology; Fechner, R.E., Rosen, P.P., Eds.; ASCP Press: Chicago, IL, USA, 1997; Volume 2, pp. 1–19. [Google Scholar]
- Koga, K.; Matsuno, Y.; Noguchi, M.; Mukai, K.; Asamura, H.; Goya, T.; Shimosato, Y. A review of 79 thymomas: Modification of staging system and reappraisal of conventional division into invasive and non-invasive thymoma. Pathol. Int. 2008, 44, 359–367. [Google Scholar] [CrossRef]
- Asamura, H. Lung, pleural and thymic tumours. In UICC TNM Classification of Malignant Tumours, 8th ed.; Brierley, J.D., Gospodarowicz, M.K., Wittekind, C., Eds.; Wiley: Hoboken, NJ, USA, 2017; pp. 115–118. [Google Scholar]
- Ozawa, Y.; Hara, M.; Shimohira, M.; Sakurai, K.; Nakagawa, M.; Shibamoto, Y. Associations between computed tomography features of thymomas and their pathological classification. Acta Radiol. 2016, 57, 1318–1325. [Google Scholar] [CrossRef]
- Han, X.; Gao, W.; Chen, Y.; Du, L.; Duan, J.; Yu, H.; Guo, R.; Zhang, L.; Ma, G. Relationship Between Computed Tomography Imaging Features and Clinical Characteristics, Masaoka–Koga Stages, and World Health Organization Histological Classifications of Thymoma. Front. Oncol. 2019, 9, 1041. [Google Scholar] [CrossRef]
- Luo, T.; Zhao, H.; Zhou, X. The clinical features, diagnosis and management of recurrent thymoma. J. Cardiothorac. Surg. 2016, 11, 1–7. [Google Scholar] [CrossRef]
- Ruffini, E.; Fang, W.; Guerrera, F.; Huang, J.; Okumura, M.; Kim, D.K.; Girard, N.; Billè, A.; Boubia, S.; Cangir, A.K.; et al. The International Association for the Study of Lung Cancer Thymic Tumors Staging Project: The Impact of the Eighth Edition of the Union for International Cancer Control and American Joint Committee on Cancer TNM Stage Classification of Thymic Tumors. J. Thorac. Oncol. 2020, 15, 436–447. [Google Scholar] [CrossRef]
- Muriana, P.; Carretta, A.; Ciriaco, P.; Bandiera, A.; Negri, G. Assessment of the prognostic role of neutrophil-to-lymphocyte ratio following complete resection of thymoma. J. Cardiothorac. Surg. 2018, 13, 119. [Google Scholar] [CrossRef]
- Voulaz, E.; Veronesi, G.; Infante, M.; Cariboni, U.; Testori, A.; Novellis, P.; Bottoni, E.; Passera, E.; Morenghi, E.; Alloisio, M. Radical thymectomy versus conservative thymomectomy in the surgical treatment of thymic malignancies. J. Thorac. Dis. 2018, 10, 4127–4136. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.-H.; Marx, A.; Wen-Hu, C.; Yong, J.; Puppe, B.; Stroebel, P.; Mueller-Hermelink, H.K. New WHO histologic classification predicts prognosis of thymic epithelial tumors: A clinicopathologic study of 200 thymoma cases from China. Cancer 2002, 95, 420–429. [Google Scholar] [CrossRef] [PubMed]
- Müller-Hermelink, H.K.; Engel, P.; Kuo, T.T.; Ströbel, P.; Marx, A.; Harris, N.L.; Möller, P.; Menestrina, F.; Shimosato, Y.; Asamura, H.; et al. Thymomas. In Pathology and Genetics of Tumors of the Lung, Pleura, Thymus and Heart; Travis, W.D., Brambilla, E., Muller-Hermelink, H.K., Harris, C.C., Eds.; IARC Press: Lyon, France, 2004. [Google Scholar]
- Rieker, R.J.; Hoegel, J.; Morresi-Hauf, A.; Hofmann, W.J.; Bläker, H.; Penzel, R.; Otto, H.F. Histologic classification of thymic epithelial tumors: Comparison of established classification schemes. Int. J. Cancer 2002, 98, 900–906. [Google Scholar] [CrossRef] [PubMed]
- Chalabreysse, L.; Roy, P.; Cordier, J.-F.; Loire, R.; Gamondes, J.-P.; Thivolet-Bejui, F. Correlation of the WHO schema for the classification of thymic epithelial neoplasms with prognosis: A retrospective study of 90 tumors. Am. J. Surg. Pathol. 2002, 26, 1605–1611. [Google Scholar] [CrossRef] [PubMed]
- Moran, C.A.; Suster, S. The World Health Organization (WHO) Histologic Classification of Thymomas: A Reanalysis. Curr. Treat. Options Oncol. 2008, 9, 288–299. [Google Scholar] [CrossRef] [PubMed]
- Suster, S.; Moran, C.A. Thymoma, Atypical Thymoma, and Thymic Carcinoma: A Novel Conceptual Approach to the Classification of Thymic Epithelial Neoplasms. Am. J. Clin. Pathol. 1999, 111, 826–833. [Google Scholar] [CrossRef] [PubMed]
- Ried, M.; Eicher, M.-M.; Neu, R.; Sziklavari, Z.; Hofmann, H.-S. Evaluation of the new TNM-staging system for thymic malignancies: Impact on indication and survival. World J. Surg. Oncol. 2017, 15, 214. [Google Scholar] [CrossRef] [PubMed]
- Jackson, M.W.; Palma, D.A.; Camidge, D.R.; Jones, B.L.; Robin, T.P.; Sher, D.J.; Koshy, M.; Kavanagh, B.D.; Gaspar, L.E.; Rusthoven, C.G. The Impact of Postoperative Radiotherapy for Thymoma and Thymic Carcinoma. J. Thorac. Oncol. 2017, 12, 734–744. [Google Scholar] [CrossRef]
- Hwang, Y.; Kang, C.H.; Park, S.; Lee, H.J.; Park, I.K.; Kim, Y.T.; Lee, G.D.; Kim, H.R.; Choi, S.H.; Kim, Y.-H.; et al. Impact of Lymph Node Dissection on Thymic Malignancies: Multi-Institutional Propensity Score Matched Analysis. J. Thorac. Oncol. 2018, 13, 1949–1957. [Google Scholar] [CrossRef]
- Kondo, K.; Monden, Y. Lymphogenous and hematogenous metastasis of thymic epithelial tumors. Ann. Thorac. Surg. 2003, 76, 1859–1864. [Google Scholar] [CrossRef]
- Gu, Z.; Wei, Y.; Fu, J.; Tan, L.; Zhang, P.; Han, Y.; Chen, C.; Zhang, R.; Li, Y.; Chen, K.; et al. Lymph node metastases in thymic malignancies: A Chinese Alliance for Research in Thymomas retrospective database analysis†. Interact. Cardiovasc. Thorac. Surg. 2017, 25, 455–461. [Google Scholar] [CrossRef] [PubMed]
- Hwang, Y.; Park, I.K.; Park, S.; Kim, E.R.; Kang, C.H.; Kim, Y.T. Lymph Node Dissection in Thymic Malignancies: Implication of the ITMIG Lymph Node Map, TNM Stage Classification, and Recommendations. J. Thorac. Oncol. 2016, 11, 108–114. [Google Scholar] [CrossRef] [PubMed]
- Fang, W.; Wang, Y.; Pang, L.; Gu, Z.; Wei, Y.; Liu, Y.; Zhang, P.; Chen, C.; Zhou, X.; Liu, Y.; et al. Lymph node metastasis in thymic malignancies: A Chinese multicenter prospective observational study. J. Thorac. Cardiovasc. Surg. 2018, 156, 824–833.e1. [Google Scholar] [CrossRef] [PubMed]
- Tseng, Y.-C.; Hsieh, C.-C.; Huang, H.-Y.; Huang, C.; Hsu, W.-H.; Huang, B.-S.; Huang, M.-H.; Hsu, H.-S. Is Thymectomy Necessary in Nonmyasthenic Patients with Early Thymoma? J. Thorac. Oncol. 2013, 8, 952–958. [Google Scholar] [CrossRef] [PubMed]


| Total No. | 50 |
|---|---|
| Age (±SD; range) | 57.5 ± 12.7; 27–83 |
| Gender (males/female) | 27/23 |
| Myasthenia gravis | 14 (28%) |
| Induction treatment | 4 (8%) |
| Surgical access | |
| Median sternotomy | 43 (86%) |
| Thoracotomy | 2 (4%) |
| Video-assisted thoracic surgery (VATS) | 5 (10%) |
| WHO classification | |
| A | 4 (8%) |
| AB | 14 (28%) |
| B1 | 11 (22%) |
| B2 | 8 (16%) |
| B3 | 13 (26%) |
| Masaoka stage | |
| I | 13 (26%) |
| II | 25 (50%) |
| III | 10 (20%) |
| IV | 2 (4%) |
| Adjuvant therapy | 30 (60%) |
| Masaoka Stage | |||||
|---|---|---|---|---|---|
| I | II | III | IV | Total No. | |
| TNM stage | |||||
| I | 13 | 25 | 1 | 0 | 39 (78%) |
| II | 0 | 0 | 2 | 0 | 2 (4%) |
| IIIA | 0 | 0 | 7 | 1 | 8 (16%) |
| IVA | 0 | 0 | 0 | 1 | 1 (2%) |
| Total | 13 (26%) | 25 (50%) | 10 (20%) | 2 (4%) | |
| WHO | ||||||
|---|---|---|---|---|---|---|
| A | AB | B1 | B2 | B3 | ||
| TNM | I | 4 (8%) | 14 (28%) | 11 (22%) | 6 (12%) | 4 (8%) |
| II | 0 | 0 | 0 | 0 | 2 (4%) | |
| IIIA | 0 | 0 | 0 | 2 (4%) | 6 (12%) | |
| IVA | 0 | 0 | 0 | 0 | 1 (2%) | |
| MASAOKA–KOGA | p | TNM | p | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| I | II | III | IV | I | II | III-A | IV-A | ||||
| Long axis (median, mm) | <53.5 | 10 (76.9%) | 9 (36%) | 5 (50%) | 1 (50%) | 0.13 | 20 (51.3%) | 0 | 4 (50%) | 1 (100%) | 0.39 |
| >53.5 | 3 (23.1%) | 16 (64%) | 5 (50%) | 1 (50%) | 19 (48.7%) | 2 (100%) | 4 (50%) | 0 | |||
| Short axis (median, mm) | <36.5 | 9 (69.2%) | 11 (44%) | 4 (40%) | 1 (50%) | 0.44 | 20 (51.3%) | 0 | 4 (50%) | 1 (100%) | 0.39 |
| >36.5 | 4 (30.8%) | 14 (56%) | 6 (60%) | 1 (50%) | 19 (48.7%) | 2 (100%) | 4 (50%) | 0 | |||
| Volume (median, mm3) | <60 | 10 (76,9%) | 11 (44%) | 3 (30%) | 1 (50%) | 0.13 | 21 (53.8%) | 0 | 3 (37.5%) | 1 (100%) | 0.29 |
| >60 | 3 (23.1%) | 14 (56%) | 7 (70%) | 1 (50%) | 18 (46.2%) | 2 (100%) | 5 (62.5%) | 0 | |||
| Length of pleural contact (median, mm) | <64 | 10 (76,9%) | 11 (44%) | 3 (30%) | 1 (50%) | 0.13 | 21 (53.8%) | 1 (50%) | 2 (25%) | 1 (100%) | 0.36 |
| >64 | 3 (23.1%) | 14 (56%) | 7 (70%) | 1 (50%) | 18 (46.2%) | 1 (50%) | 6 (75%) | 0 | |||
| Shape | Ovalar | 13 (100%) | 13 (52%) | 4 (40%) | 0 | 0.003 * | 27 (69.2%) | 0 | 3 (37.5%) | 0 | 0.06 |
| Irregular | 0 | 12 (48%) | 6 (60%) | 2 (100%) | 12 (30.8%) | 2 (100%) | 5 (62.5%) | 1 (100%) | |||
| Contours | Regular | 7 (53.8%) | 11 (44%) | 1 (10%) | 0 | 0.09 | 19 (48.7%) | 0 | 0 | 0 | 0.034 * |
| Irregular | 6 (46.2%) | 14 (56%) | 9 (90%) | 2 (100%) | 20 (51.3%) | 2 (100%) | 8 (100%) | 1 (100%) | |||
| Necrosis | Yes | 5 (38.5%) | 11 (44%) | 6 (60%) | 2 (100%) | 0.34 | 17 (43.6%) | 2 (100%) | 4 (50%) | 1 (100%) | 0.31 |
| No | 8 (61.5%) | 14 (56%) | 4 (40%) | 0 | 22 (56.4%) | 0 | 4 (50%) | 0 | |||
| Calcifications | Yes | 3 (23.1%) | 5 (20%) | 5 (50%) | 0 | 0.24 | 9 (23.1%) | 1 (50%) | 3 (37.5%) | 0 | 0.64 |
| No | 10 (76.9%) | 20 (80%) | 5 (50%) | 2 (100%) | 30 (76.9%) | 1 (50%) | 5 (62.5%) | 1 (100%) | |||
| Lymph node enlargements | Yes | 1 (7.7%) | 5 (20%) | 4 (40%) | 0 | 0.51 | 6 (15.4%) | 1 (50%) | 3 (37.5%) | 0 | 0.73 |
| No | 12 (92.3%) | 20 (80%) | 6 (60%) | 2 (100%) | 33 (84.6%) | 1 (50%) | 5 (62.5%) | 1 (100%) | |||
| Pleural effusion | Yes | 2 (15.4%) | 1 (4%) | 1 (10%) | 0 | 0.63 | 3 (7.7%) | 0 | 1 (12.5%) | 0 | 0.92 |
| No | 11 (84.6%) | 24 (96%) | 9 (90%) | 2 (100%) | 36 (92.3%) | 2 (100%) | 7 (87.5%) | 1 (100%) | |||
| Pericardial effusion | Yes | 0 | 3 (12%) | 2 (20%) | 0 | 0.41 | 3 (7.7%) | 0 | 2 (25%) | 0 | 0.46 |
| No | 13 (100%) | 22 (88%) | 8 (80%) | 2 (100%) | 36 (92.3%) | 2 (100%) | 6 (75%) | 1 (100%) | |||
| Invasion of mediastinal fat | Yes | 2 (15.4%) | 12 (48%) | 7 (70%) | 2 (100%) | 0.022 * | 14 (35.9%) | 2 (100%) | 6 (75%) | 1 (100%) | 0.05 |
| No | 11 (84.6%) | 13 (52%) | 3 (30%) | 0 | 25 (64.1%) | 0 | 2 (25%) | 0 | |||
| Contact with mediastinal vessels | Yes | 8 (61.5%) | 24 (96%) | 10 (100%) | 2 (100%) | 0.008 * | 33 (84.6%) | 2 (100%) | 8 (100%) | 1 (100%) | 0.59 |
| No | 5 (38.5%) | 1 (4%) | 0 | 0 | 6 (15.4%) | 0 | 0 | 0 | |||
| Pericardial contact | Yes | 7 (53.8%) | 21 (84%) | 10 (100%) | 2 (100%) | 0.032 * | 29 (74.4%) | 2 (100%) | 8 (100%) | 1 (100%) | 0.32 |
| No | 6 (46.2%) | 4 (16%) | 0 | 0 | 10 (25.6%) | 0 | 0 | 0 | |||
| Lung invasion | Yes | 1 (7.7%) | 0 | 1 (10%) | 0 | 0.47 | 1 (2.6%) | 1 (50%) | 0 | 0 | 0.009 * |
| No | 12 (92.3%) | 25 (100%) | 9 (90%) | 2 (100%) | 38 (97.4%) | 1 (50%) | 8 (100%) | 1 (100%) | |||
| Pleural invasion | Yes | 1 (7.7%) | 1 (4%) | 0 | 0 | 0.81 | 2 (5.1%) | 0 | 0 | 0 | 0.89 |
| No | 12 (92.3%) | 24 (96%) | 10 (100%) | 2 (100%) | 37 (94.9%) | 2 (100%) | 8 (100%) | 1 (100%) | |||
| Contrast enhancement diffusion pattern | Homogeneous | 4 (57.1%) | 5 (27.8%) | 2 (25%) | 0 | 0.42 | 9 (34.6%) | 0 | 2 (33.3%) | 0 | 0.60 |
| Inhomogeneous | 3 (42.9%) | 13 (72.2%) | 6 (75%) | 1 (100%) | 17 (65.4%) | 2 (100%) | 4 (66.7%) | 0 | |||
| Laterality | Right | 4 (30.8%) | 6 (24%) | 3 (30%) | 0 | 0.25 | 11 (28.2%) | 1 (50%) | 1 (12.5%) | 0 | 0.15 |
| Left | 9 (69.2%) | 14 (56%) | 3 (30%) | 1 (50%) | 23 (59%) | 0 | 4 (50%) | 0 | |||
| Median | 0 | 5 (20%) | 4 (40%) | 1 (50%) | 5 (12.8%) | 1 (50%) | 3 (37.5%) | 1 (100%) | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Carretta, A.; Viscardi, S.; Muriana, P.; Ciriaco, P.; Bandiera, A.; Varagona, R.; Colombo, M.; Negri, G. Correlation of Computed Tomography Parameters with Histology, Stage and Prognosis in Surgically Treated Thymomas. Medicina 2021, 57, 10. https://doi.org/10.3390/medicina57010010
Carretta A, Viscardi S, Muriana P, Ciriaco P, Bandiera A, Varagona R, Colombo M, Negri G. Correlation of Computed Tomography Parameters with Histology, Stage and Prognosis in Surgically Treated Thymomas. Medicina. 2021; 57(1):10. https://doi.org/10.3390/medicina57010010
Chicago/Turabian StyleCarretta, Angelo, Stefano Viscardi, Piergiorgio Muriana, Paola Ciriaco, Alessandro Bandiera, Roberto Varagona, Michele Colombo, and Giampiero Negri. 2021. "Correlation of Computed Tomography Parameters with Histology, Stage and Prognosis in Surgically Treated Thymomas" Medicina 57, no. 1: 10. https://doi.org/10.3390/medicina57010010
APA StyleCarretta, A., Viscardi, S., Muriana, P., Ciriaco, P., Bandiera, A., Varagona, R., Colombo, M., & Negri, G. (2021). Correlation of Computed Tomography Parameters with Histology, Stage and Prognosis in Surgically Treated Thymomas. Medicina, 57(1), 10. https://doi.org/10.3390/medicina57010010








