Long-Term Efficacy of 5-ALA Photodynamic Therapy in Oral Lichen Planus Patients
Abstract
1. Introduction
2. Results
2.1. Lesion Size
2.2. Subjective Symptoms—VAS Score
2.3. Clinical Index—REU Score
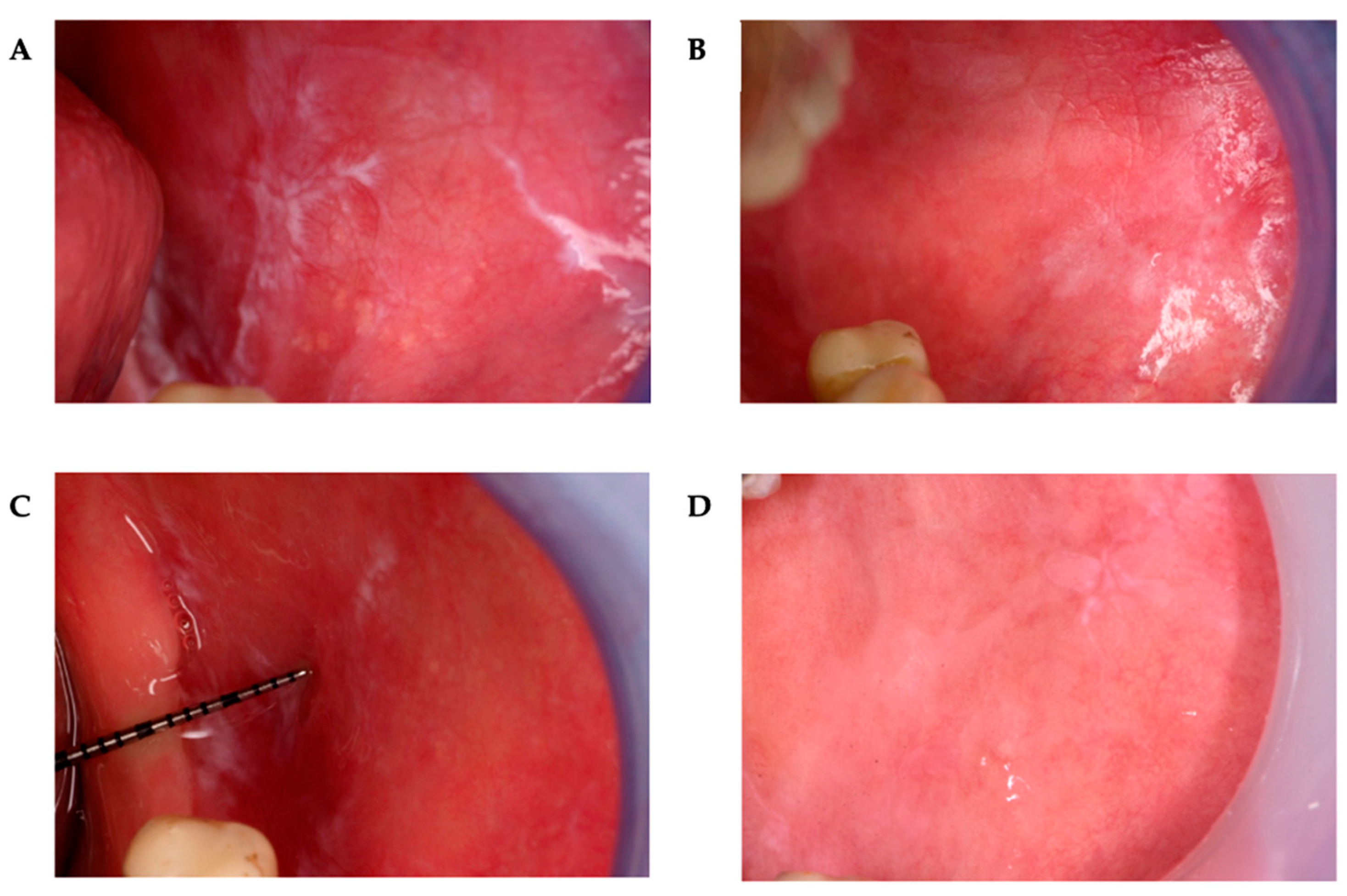
2.4. Healing Rates
3. Discussion
4. Materials and Methods
4.1. Therapeutic Procedure
4.2. Clinical Examination
4.3. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- González-Moles, M.Á.; Ruiz-Ávila, I.; González-Ruiz, L.; Ayen, A.; Gil-Montoya, J.A.; Ramos-Garcia, P. Malignant Transformation Risk of Oral Lichen Planus: A Systematic Review and Comprehensive Meta-analysis. Oral Oncol. 2019, 96, 121–130. [Google Scholar] [CrossRef]
- Osipoff, A.; Carpenter, M.D.; Noll, J.L.; Valdez, J.A.; Gormsen, M.; Brennan, M.T. Predictors of Symptomatic Oral Lichen Planus. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2020, 129, 468–477. [Google Scholar] [CrossRef]
- Abbate, G.; Foscolo, A.M.; Gallotti, M.; Lancella, A.; Mingo, F. Neoplastic Transformation of Oral Lichen: Case Report and Review of the Literature. Acta Otorhinolaryngol. Ital. 2006, 26, 47–52. [Google Scholar]
- Tziotzios, C.; Lee, J.Y.; Brier, T.; Saito, R.; Hsu, C.K.; Bhargava, K.; Stefanato, C.M.; Fenton, D.A.; McGrath, J.A. Lichen Planus and Lichenoid Dermatoses: Clinical Overview and Molecular Basis. J. Am. Acad. Dermatol. 2018, 79, 789–804. [Google Scholar] [CrossRef] [PubMed]
- de Brito Monteiro, B.V.; dos Santos Pereira, J.; Nonaka, C.F.; Godoy, G.P.; da Silveira, É.J.; da Costa Miguel, M.C. Immunoexpression of Th17-Related Cytokines in Oral Lichen Planus. Appl. Immunohistochem. Mol. Morphol. 2015, 23, 409–415. [Google Scholar] [CrossRef] [PubMed]
- Marshall, A.; Celentano, A.; Cirillo, N.; McCullough, M.; Porter, S. Tissue-Specific Regulation of CXCL9/10/11 Chemokines in Keratinocytes: Implications for Oral Inflammatory Disease. PLoS ONE 2017, 12, e0172821. [Google Scholar] [CrossRef] [PubMed]
- Ali, S. Review of the Current Evidence of Non-HLA Gene Polymorphism in Oral Lichen Planus. J. Oral Maxillofac. Surg. Med. Pathol. 2021, 33, 334–339. [Google Scholar] [CrossRef]
- Villa, T.G.; Sánchez-Pérez, Á.; Sieiro, C. Oral Lichen Planus: A Microbiologist Point of View. Int. Microbiol. 2021, 24, 275–289. [Google Scholar] [CrossRef]
- Olson, M.A.; Rogers, R.S., III; Bruce, A.J. Oral Lichen Planus. Clin. Dermatol. 2016, 34, 495–504. [Google Scholar] [CrossRef]
- Carrozzo, M.; Porter, S.; Mercadante, V.; Fedele, S. Oral Lichen Planus: A Disease or a Spectrum of Tissue Reactions? Types, Causes, Diagnostic Algorithms, Prognosis, Management Strategies. Periodontology 2019, 80, 105–125. [Google Scholar] [CrossRef]
- Kalkur, C.; Sattur, A.P.; Guttal, K.S. Role of Depression, Anxiety and Stress in Patients with Oral Lichen Planus: A Pilot Study. Indian J. Dermatol. 2015, 60, 445–449. [Google Scholar] [CrossRef]
- McAleer, M.A.; Jakasa, I.; Stefanovic, N.; McLean, W.H.I.; Kezic, S.; Irvine, A.D. Topical Corticosteroids Normalize Both Skin and Systemic Inflammatory Markers in Infant Atopic Dermatitis. Br. J. Dermatol. 2021, 185, 153–163. [Google Scholar] [CrossRef]
- Einarsdottir, M.J.; Bankvall, M.; Robledo-Sierra, J.; Rödström, P.O.; Bergthorsdottir, R.; Trimpou, P.; Hasséus, B.; Ragnarsson, O. Topical Clobetasol Treatment for Oral Lichen Planus Can Cause Adrenal Insufficiency. Oral Dis. 2024, 30, 1304–1312. [Google Scholar] [CrossRef]
- Pignatelli, P.; Umme, S.; D’Antonio, D.L.; Piattelli, A.; Curia, M.C. Reactive Oxygen Species Produced by 5-Aminolevulinic Acid Photodynamic Therapy in the Treatment of Cancer. Int. J. Mol. Sci. 2023, 24, 8964. [Google Scholar] [CrossRef]
- Redmond, R.W.; Gamlin, J.N. A Compilation of Singlet Oxygen Yields from Biologically Relevant Molecules. Photochem. Photobiol. 1999, 70, 391–475. [Google Scholar] [CrossRef] [PubMed]
- Correia, J.H.; Rodrigues, J.A.; Pimenta, S.; Dong, T.; Yang, Z. Photodynamic Therapy Review: Principles, Photosensitizers, Applications, and Future Directions. Pharmaceutics 2021, 13, 1332. [Google Scholar] [CrossRef]
- Simões, J.C.; Sarpaki, S.; Papadimitroulas, P.; Therrien, B.; Loudos, G. Conjugated photosensitizers for imaging and PDT in cancer research. J. Med. Chem. 2020, 63, 14119–14150. [Google Scholar] [CrossRef]
- Baptista, M.S.; Cadet, J.; Di Mascio, P.; Ghogare, A.A.; Greer, A.; Hamblin, M.R.; Lorente, C.; Nunez, S.C.; Ribeiro, M.S.; Thomas, A.H.; et al. Type I and Type II Photosensitized Oxidation Reactions: Guidelines and Mechanistic Pathways. Photochem. Photobiol. 2017, 93, 912–919. [Google Scholar] [CrossRef]
- Maciejczyk, M.; Bielas, M.; Zalewska, A.; Gerreth, K. Salivary Biomarkers of Oxidative Stress and Inflammation in Stroke Patients: From Basic Research to Clinical Practice. Oxid. Med. Cell. Longev. 2021, 2021, 5545330. [Google Scholar] [CrossRef]
- Papa, V.; Furci, F.; Minciullo, P.L.; Casciaro, M.; Allegra, A.; Gangemi, S. Photodynamic Therapy in Cancer: Insights into Cellular and Molecular Pathways. Curr. Issues Mol. Biol. 2025, 47, 69. [Google Scholar] [CrossRef]
- Nath, S.; Obaid, G.; Hasan, T. The Course of Immune Stimulation by Photodynamic Therapy: Bridging Fundamentals of Photochemically Induced Immunogenic Cell Death to the Enrichment of T-Cell Repertoire. Photochem. Photobiol. 2019, 95, 1288–1305. [Google Scholar] [CrossRef]
- Wang, L.; Chen, Q.; Liu, D. Development of Photodynamic Therapy in Treating Oral Diseases. Front. Oral Health 2025, 5, 1506407. [Google Scholar] [CrossRef]
- Joseph, B.; Mauramo, M.; Vijayakumary, B.K.; Waltimo, T.; Anil, S.; Sorsa, T. Photodynamic Therapy for Oral Mucositis in Cancer Patients—A Systematic Review and Meta-Analysis. Photodiag. Photodyn. Ther. 2024, 50, 104424. [Google Scholar] [CrossRef] [PubMed]
- Shang, Q.; Wang, Z.; Dong, Y.; Cai, L.; Mao, F.; Deng, J.; Dan, H.; Zeng, X.; Zhou, Y.; Chen, Q. Photodynamic Therapy in the Treatment of Oral Lichen Planus with Moderate-to-Severe Dysplasia: A Case Report. Dermatol. Ther. 2020, 33, e14490. [Google Scholar] [CrossRef] [PubMed]
- Jerjes, W.; Upile, T.; Hamdoon, Z.; Mosse, C.A.; Akram, S.; Hopper, C. Photodynamic Therapy Outcome for Oral Dysplasia. Lasers Surg. Med. 2011, 43, 192–199. [Google Scholar] [CrossRef] [PubMed]
- Binnal, A.; Tadakamadla, J.; Rajesh, G.; Tadakamadla, S.K. Photodynamic Therapy for Oral Potentially Malignant Disorders: A Systematic Review and Meta-Analysis. Photodiagn. Photodyn. Ther. 2022, 37, 102713. [Google Scholar] [CrossRef]
- Lopez, R.F.V.; Lange, N.; Guy, R.; Bentley, M.V.L.B. Photodynamic Therapy of Skin Cancer: Controlled Drug Delivery of 5-ALA and Its Esters. Adv. Drug Deliv. Rev. 2004, 56, 77–94. [Google Scholar] [CrossRef]
- Pascale, R.M.; Calvisi, D.F.; Simile, M.M.; Feo, C.F.; Feo, F. The Warburg Effect 97 Years after Its Discovery. Cancers 2020, 12, 2819. [Google Scholar] [CrossRef]
- Juzeniene, A.; Iani, V.; Moan, J. Clearance Mechanism of Protoporphyrin IX from Mouse Skin after Application of 5-Aminolevulinic Acid. Photodiagn. Photodyn. Ther. 2013, 10, 538–545. [Google Scholar] [CrossRef]
- Wachowska, M.; Muchowicz, A.; Firczuk, M.; Gabrysiak, M.; Winiarska, M.; Wańczyk, M.; Bojarczuk, K.; Golab, J. Aminolevulinic Acid (ALA) as a Prodrug in Photodynamic Therapy of Cancer. Molecules 2011, 16, 4140–4164. [Google Scholar] [CrossRef]
- Algorri, J.F.; Ochoa, M.; Roldán-Varona, P.; Rodríguez-Cobo, L.; López-Higuera, J.M. Light Technology for Efficient and Effective Photodynamic Therapy: A Critical Review. Cancers 2021, 13, 3484. [Google Scholar] [CrossRef] [PubMed]
- Wiegell, S.R.; Stender, I.M.; Na, R.; Wulf, H.C. Pain Associated with Photodynamic Therapy Using 5-Aminolevulinic Acid or 5-Aminolevulinic Acid Methylester on Tape-Stripped Normal Skin. Arch. Dermatol. 2003, 139, 1173–1177. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Deng, J.; Zhao, Y.; Tao, H.; Dan, H.; Xu, H.; Chen, Q. Efficacy Evaluation of Photodynamic Therapy for Oral Lichen Planus: A Systematic Review and Meta-Analysis. BMC Oral Health 2020, 20, 302. [Google Scholar] [CrossRef] [PubMed]
- Kvaal, S.I.; Angell-Petersen, E.; Warloe, T. Photodynamic Treatment of Oral Lichen Planus. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2013, 115, 62–70. [Google Scholar] [CrossRef]
- Maloth, K.N.; Velpula, N.; Kodangal, S.; Sangmesh, M.; Vellamchetla, K.; Ugrappa, S.; Meka, N. Photodynamic Therapy—A Non-Invasive Treatment Modality for Precancerous Lesions. J. Lasers Med. Sci. 2016, 7, 30–36. [Google Scholar] [CrossRef]
- Sulewska, M.; Pietruska, A.; Tomaszuk, J.; Szymańska, E.; Winnicka, K.; Narolewska, J.; Pietruska, M. Efficacy of Photodynamic Therapy with 5-Aminolevulinic Acid-Loaded Oromucosal Emulgel in Patients with Oral Lichen Planus: A Randomized Controlled Clinical Study. Int. Dent. J. 2025, 75, 103925. [Google Scholar] [CrossRef]
- Zborowski, J.; Kida, D.; Karolewicz, B.; Jurczyszyn, K.; Konopka, T. A Comparison of Photodynamic Therapy and Topical Clobetasol in Treatment of Oral Lichen Planus: A Split-Mouth Randomized Controlled Study. J. Clin. Med. 2025, 14, 681. [Google Scholar] [CrossRef]
- Bakhtiari, S.; Azari-Marhabi, S.; Mojahedi, S.M.; Namdari, M.; Rankohi, Z.E.; Jafari, S. Comparing Clinical Effects of Photodynamic Therapy as a Novel Method with Topical Corticosteroid for Treatment of Oral Lichen Planus. Photodiagn. Photodyn. Ther. 2017, 20, 159–164. [Google Scholar] [CrossRef]
- Mostafa, D.; Moussa, E.; Alnouaem, M. Evaluation of Photodynamic Therapy in Treatment of Oral Erosive Lichen Planus in Comparison with Topically Applied Corticosteroids. Photodiagn. Photodyn. Ther. 2017, 19, 56–66. [Google Scholar] [CrossRef]
- Mirza, S.; Rehman, N.; Alrahlah, A.; Alamri, W.A.R.; Vohra, F. Efficacy of Photodynamic Therapy or Low-Level Laser Therapy against Steroid Therapy in the Treatment of Erosive-Atrophic Oral Lichen Planus. Photodiagn. Photodyn. Ther. 2018, 21, 404–408. [Google Scholar] [CrossRef]
- Salinas-Gilabert, C.; Gómez García, F.; Galera Molero, F.; Pons-Fuster, E.; Vander Beken, S.; Lopez Jornet, P. Photodynamic Therapy, Photobiomodulation and Acetonide Triamcinolone 0.1% in the Treatment of Oral Lichen Planus: A Randomized Clinical Trial. Pharmaceutics 2023, 15, 30. [Google Scholar] [CrossRef] [PubMed]
- Sobaniec, S.; Bernaczyk, P.; Pietruski, J.; Cholewa, M.; Skurska, A.; Dolińska, E.; Duraj, E.; Tokajuk, G.; Paniczko, A.; Olszewska, E.; et al. Clinical Assessment of the Efficacy of Photodynamic Therapy in the Treatment of Oral Lichen Planus. Lasers Med. Sci. 2013, 28, 311–316. [Google Scholar] [CrossRef] [PubMed]
- Rakesh, N.; Clint, J.B.; Reddy, S.S.; Nagi, R.; Chauhan, P.; Sharma, S.; Sharma, P.; Kaur, A.; Shetty, B.; Ashwini, S.; et al. Clinical Evaluation of Photodynamic Therapy for the Treatment of Refractory Oral Lichen Planus—A Case Series. Photodiagn. Photodyn. Ther. 2018, 24, 280–285. [Google Scholar] [CrossRef] [PubMed]
- Gulzar, M.A.; Gul, N.; Alvi, F.D.; Khattak, Y.R.; Hasan, U.S.; Haneef, M.B.; Ahmad, I. Comparison of Photodynamic Therapy and Corticosteroid Therapy in Management of Oral Lichen Planus: A Systematic Review of Randomized Controlled Trials. Photodiagn. Photodyn. Ther. 2023, 44, 103747. [Google Scholar] [CrossRef]
- Nagi, R.; Muthukrishnan, A.; Rakesh, N. Effectiveness of Photodynamic Therapy (PDT) in the Management of Symptomatic Oral Lichen Planus: A Systematic Review. J. Oral Biol. Craniofac. Res. 2023, 13, 353–359. [Google Scholar] [CrossRef]
- Lavaee, F.; Shadmanpour, M. Comparison of the Effect of Photodynamic Therapy and Topical Corticosteroid on Oral Lichen Planus Lesions. Oral Dis. 2019, 25, 1954–1963. [Google Scholar] [CrossRef]
- Romano, A.; Di Stasio, D.; Lauritano, D.; Lajolo, C.; Fiori, F.; Gentile, E.; Lucchese, A. Topical Photodynamic Therapy in the Treatment of Benign Oral Mucosal Lesions: A Systematic Review. J. Oral Pathol. Med. 2021, 50, 639–648. [Google Scholar] [CrossRef]
- Sulewska, M.; Duraj, E.; Sobaniec, S.; Graczyk, A.; Milewski, R.; Wróblewska, M.; Pietruski, J.; Pietruska, M. A Clinical Evaluation of Efficacy of Photodynamic Therapy in Treatment of Reticular Oral Lichen Planus: A Case Series. Photodiagn. Photodyn. Ther. 2019, 25, 50–57. [Google Scholar] [CrossRef]
- Sulewska, M.; Duraj, E.; Sobaniec, S.; Graczyk, A.; Milewski, R.; Wróblewska, M.; Pietruski, J.; Pietruska, M. A Clinical Evaluation of the Efficacy of Photodynamic Therapy in the Treatment of Erosive Oral Lichen Planus: A Case Series. Photodiagn. Photodyn. Ther. 2017, 18, 12–19. [Google Scholar] [CrossRef]
- Sulewska, M.E.; Tomaszuk, J.; Sajewicz, E.; Pietruski, J.; Starzyńska, A.; Pietruska, M. Treatment of Reticular Oral Lichen Planus with Photodynamic Therapy: A Case Series. J. Clin. Med. 2023, 12, 875. [Google Scholar] [CrossRef]
- Gollnick, S.O.; Owczarczak, B.; Maier, P. Photodynamic Therapy and Anti-Tumor Immunity. Lasers Surg. Med. 2006, 38, 509–515. [Google Scholar] [CrossRef]
- Cosgarea, R.; Pollmann, R.; Sharif, J.; Schmidt, T.; Stein, R.; Bodea, A.; Auschill, T.; Sculean, A.; Eming, R.; Greene, B.; et al. Photodynamic Therapy in Oral Lichen Planus: A Prospective Case-Controlled Pilot Study. Sci. Rep. 2020, 10, 1667. [Google Scholar] [CrossRef]
- Wiśniewski, P.; Sulewska, M.; Tomaszuk, J.; Zalewska, A.; Zięba, S.; Pietruska, A.; Szymańska, E.; Winnicka, K.; Maciejczyk, M.; Żendzian-Piotrowska, M.; et al. Effects of Photodynamic Therapy and Glucocorticosteroids on Salivary Oxidative Stress in Oral Lichen Planus: A Randomized Clinical Trial. Antioxidants 2025, 14, 1017. [Google Scholar] [CrossRef]
- Chiriac, A.P.; Diaconu, A.; Nita, L.E.; Tudorachi, N.; Mititelu-Tartau, L.; Creteanu, A.; Dragostin, O.; Rusu, D.; Popa, G. The Influence of Excipients on Physical and Pharmaceutical Properties of Oral Lyophilisates Containing a Pregabalin-Acetaminophen Combination. Expert Opin. Drug Deliv. 2017, 14, 589–599. [Google Scholar] [CrossRef]
- González-Moles, M.Á.; Ramos-García, P. Oral Lichen Planus and Related Lesions. What should we accept based on the available evidence? Oral Dis. 2023, 29, 2624–2637. [Google Scholar] [CrossRef]
- Park, H.K.; Hurwitz, S.; Woo, S.B. Oral Lichen Planus: REU Scoring System Correlates with Pain. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2012, 114, 75–82. [Google Scholar] [CrossRef]
- Wang, J.; Van der Waal, I. Disease Scoring Systems for Oral Lichen Planus; A Critical Appraisal. Med. Oral Patol. Oral Cir. Bucal 2015, 20, e199–e204. [Google Scholar] [CrossRef]

| Variable | Number of patients (n) | Percentage (%) |
| Sex | ||
| Women | 34 | 77.3% |
| Men | 10 | 22.7% |
| Number of lesion sites | ||
| Single-site lesions | 5 | 11.4% |
| Multifocal lesions | 39 | 88.6% |
| Clinical form of lesions | ||
| Erosive form | 7 | 15.91% |
| Reticular form | 37 | 84.09% |
| Variable | Number of lesions (n) | Percentage (%) |
| Lesion localization | ||
| Keratinized oral mucosa | 31 | 28.18% |
| Non-keratinized oral mucosa | 79 | 71.82% |
| Clinical form of lesions | ||
| Erosive form | 16 | 11.82% |
| Reticular form | 94 | 88.18% |
| Group | Time Point | Number of Lesions | Mean ± SD | 95% CI | Min–Max | Friedman Test (p) |
|---|---|---|---|---|---|---|
| Total group | T0 | 110 | 4.05 ± 3.86 | 3.32–4.78 | 0.09–18.00 | <0.0001 |
| T1 | 1.51 ± 1.98 | 1.13–1.88 | 0.00–9.24 | |||
| T2 | 0.69 ± 1.39 | 0.42–0.95 | 0.00–9.36 | |||
| T3 | 0.67 ± 1.43 | 0.40–0.94 | 0.00–8.20 | |||
| Reticular | T0 | 94 | 4.47 ± 3.99 | 3.66–5.29 | 0.24–18.00 | <0.0001 |
| T1 | 1.49 ± 1.99 | 1.08–1.90 | 0.00–9.24 | |||
| T2 | 0.78 ± 1.47 | 0.47–1.08 | 0.00–9.36 | |||
| T3 | 0.77 ± 1.53 | 0.45–1.08 | 0.00–8.20 | |||
| Erosive | T0 | 16 | 1.60 ± 1.51 | 0.79–2.41 | 0.09–5.00 | <0.0001 |
| T1 | 1.62 ± 1.98 | 0.57–2.68 | 0.00–5.67 | |||
| T2 | 0.18 ± 0.53 | −0.09–0.46 | 0.00–2.10 | |||
| T3 | 0.10 ± 0.21 | −0.01–0.21 | 0.00–0.81 |
| Group | Time Point | n | Median | Min | Max | Lower Quartile | Upper Quartile | Friedman Test (p) |
|---|---|---|---|---|---|---|---|---|
| Total group | T0 | 44 | 4.00 | 0.00 | 9.00 | 2.00 | 6.00 | <0.0001 |
| T1 | 2.00 | 0.00 | 8.00 | 1.00 | 4.00 | |||
| T2 | 1.00 | 0.00 | 6.00 | 0.00 | 3.00 | |||
| T3 | 1.00 | 0.00 | 6.00 | 0.00 | 2.00 | |||
| Reticular form | T0 | 37 | 4.00 | 0.00 | 9.00 | 1.00 | 6.00 | <0.0001 |
| T1 | 2.00 | 0.00 | 8.00 | 1.00 | 4.00 | |||
| T2 | 1.00 | 0.00 | 6.00 | 0.00 | 3.00 | |||
| T3 | 1.00 | 0.00 | 6.00 | 0.00 | 2.00 | |||
| Erosive form | T0 | 7 | 4.00 | 2.00 | 7.00 | 4.00 | 6.00 | 0.0216 |
| T1 | 3.00 | 0.00 | 5.00 | 2.00 | 4.00 | |||
| T2 | 2.00 | 0.00 | 5.00 | 0.00 | 3.00 | |||
| T3 | 2.00 | 1.00 | 4.00 | 1.00 | 4.00 |
| Time Point | n | Mean | SD | 95% CI (Lower) | 95% CI (Upper) | Min | Max | Friedman Test (p) |
|---|---|---|---|---|---|---|---|---|
| T0 | 44 | 4.86 | 3.46 | 3.81 | 5.91 | 1.00 | 19.00 | <0.0001 |
| T1 | 3.72 | 2.89 | 2.84 | 4.60 | 0.00 | 11.00 | ||
| T2 | 2.93 | 2.67 | 2.11 | 3.74 | 0.00 | 9.50 | ||
| T3 | 1.68 | 1.83 | 1.12 | 2.24 | 0.00 | 8.00 |
| Group | Healed/Not Healed | Non-Keratinized Mucosa n (%) | Keratinized Mucosa n (%) | Chi-Square Test p |
|---|---|---|---|---|
| Total | Healed | 46 (58.23%) | 19 (61.29%) | 0.7688 |
| Not healed | 33 (41.77%) | 12 (38.71%) | ||
| Reticular form | Healed | 40 (57.97%) | 15 (60.00%) | 0.86 |
| Not healed | 29 (42.03%) | 10 (40.00%) | ||
| Erosive form | Healed | 6 (60.00%) | 4 (66.67%) | 1.00 |
| Not healed | 4 (40.00%) | 2 (33.33%) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sulewska, M.; Wróblewska, M.; Wiśniewski, P.; Duraj, E.; Tomaszuk, J.; Pietruska, A.; Pietruska, M. Long-Term Efficacy of 5-ALA Photodynamic Therapy in Oral Lichen Planus Patients. Pharmaceuticals 2025, 18, 1676. https://doi.org/10.3390/ph18111676
Sulewska M, Wróblewska M, Wiśniewski P, Duraj E, Tomaszuk J, Pietruska A, Pietruska M. Long-Term Efficacy of 5-ALA Photodynamic Therapy in Oral Lichen Planus Patients. Pharmaceuticals. 2025; 18(11):1676. https://doi.org/10.3390/ph18111676
Chicago/Turabian StyleSulewska, Magdalena, Marta Wróblewska, Patryk Wiśniewski, Ewa Duraj, Jagoda Tomaszuk, Aleksandra Pietruska, and Małgorzata Pietruska. 2025. "Long-Term Efficacy of 5-ALA Photodynamic Therapy in Oral Lichen Planus Patients" Pharmaceuticals 18, no. 11: 1676. https://doi.org/10.3390/ph18111676
APA StyleSulewska, M., Wróblewska, M., Wiśniewski, P., Duraj, E., Tomaszuk, J., Pietruska, A., & Pietruska, M. (2025). Long-Term Efficacy of 5-ALA Photodynamic Therapy in Oral Lichen Planus Patients. Pharmaceuticals, 18(11), 1676. https://doi.org/10.3390/ph18111676






