Therapeutic Potential of Molecular Hydrogen in Metabolic Diseases from Bench to Bedside
Abstract
1. Introduction
2. Protective Effects of H2 against Metabolic Diseases
2.1. Diabetes Mellitus
2.2. Metabolic Syndrome
2.3. Fatty Liver
2.4. Atherosclerosis
2.5. Obesity
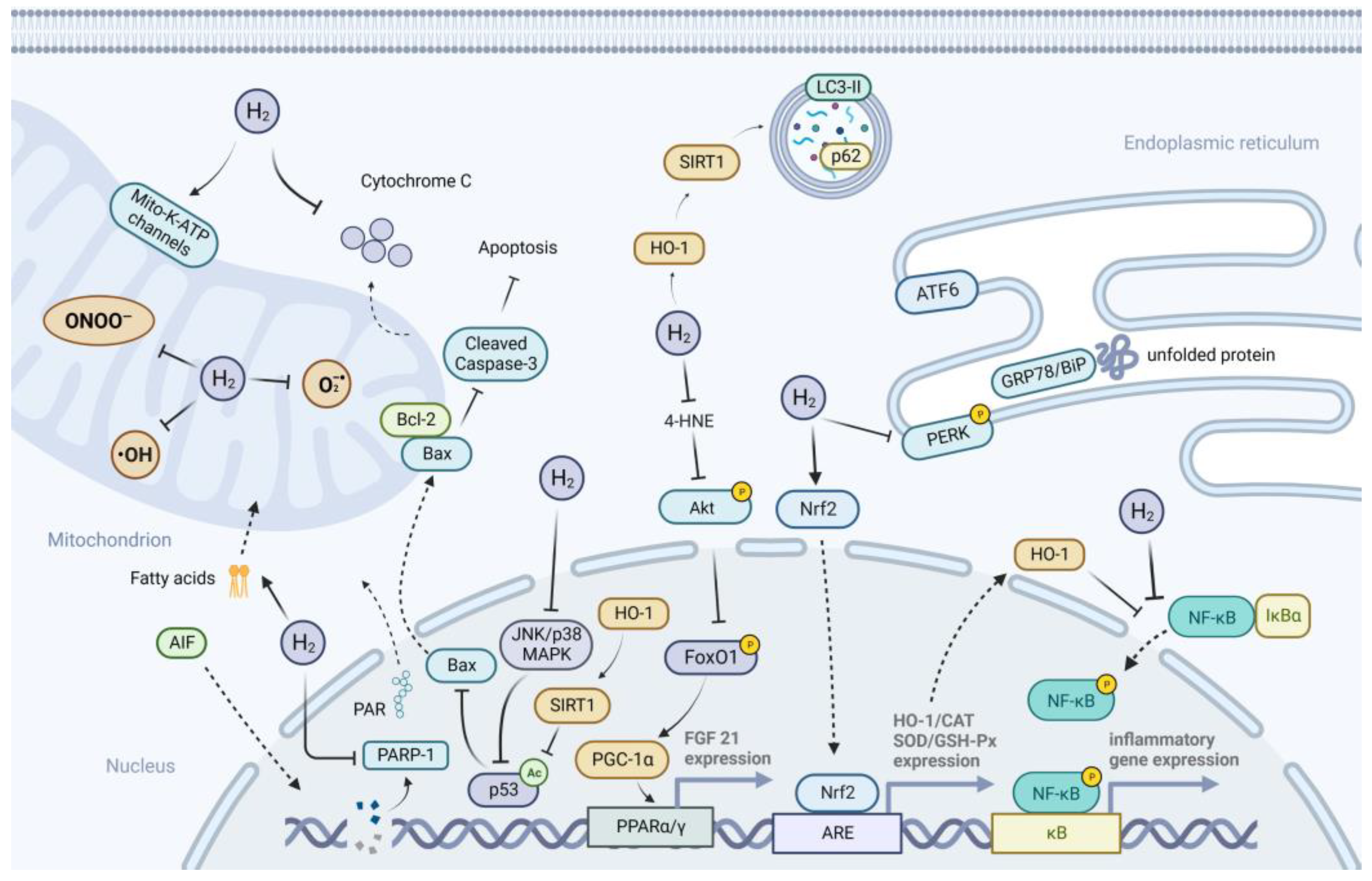
3. Possible Mechanisms of H2 Action on Metabolic Diseases
3.1. Anti-Oxidative Effects of H2
3.2. Anti-Inflammatory Effects of H2
3.3. Anti-Apoptotic Effects of H2
3.4. Suppression of ER Stress
3.5. Activation of Autophagy
3.6. Improvement of Mitochondrial Function
3.7. Regulation of Gut Microbiota
3.8. Other Possible Mechanisms and Potential Molecular Targets of H2
4. Conclusions and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ohta, S. Molecular hydrogen as a novel antioxidant: Overview of the advantages of hydrogen for medical applications. Methods Enzymol. 2015, 555, 289–317. [Google Scholar] [CrossRef] [PubMed]
- Dole, M.; Wilson, F.R.; Fife, W.P. Hyperbaric hydrogen therapy: A possible treatment for cancer. Science 1975, 190, 152–154. [Google Scholar] [CrossRef] [PubMed]
- Gharib, B.; Hanna, S.; Abdallahi, O.M.; Lepidi, H.; Gardette, B.; De Reggi, M. Anti-inflammatory properties of molecular hydrogen: Investigation on parasite-induced liver inflammation. Comptes Rendus Acad. Sci. III 2001, 324, 719–724. [Google Scholar] [CrossRef] [PubMed]
- Ohsawa, I.; Ishikawa, M.; Takahashi, K.; Watanabe, M.; Nishimaki, K.; Yamagata, K.; Katsura, K.; Katayama, Y.; Asoh, S.; Ohta, S. Hydrogen acts as a therapeutic antioxidant by selectively reducing cytotoxic oxygen radicals. Nat. Med. 2007, 13, 688–694. [Google Scholar] [CrossRef] [PubMed]
- Ohta, S. Molecular hydrogen as a preventive and therapeutic medical gas: Initiation, development and potential of hydrogen medicine. Pharmacol. Ther. 2014, 144, 1–11. [Google Scholar] [CrossRef]
- Yadav, U.C.; Rani, V.; Deep, G.; Singh, R.K.; Palle, K. Oxidative stress in metabolic disorders: Pathogenesis, prevention, and therapeutics. Oxid. Med. Cell. Longev. 2016, 2016, 9137629. [Google Scholar] [CrossRef]
- Katsarou, A.; Gudbjörnsdottir, S.; Rawshani, A.; Dabelea, D.; Bonifacio, E.; Anderson, B.J.; Jacobsen, L.M.; Schatz, D.A.; Lernmark, Å. Type 1 diabetes mellitus. Nat. Rev. Dis. Prim. 2017, 3, 17016. [Google Scholar] [CrossRef]
- Galicia-Garcia, U.; Benito-Vicente, A.; Jebari, S.; Larrea-Sebal, A.; Siddiqi, H.; Uribe, K.B.; Ostolaza, H.; Martin, C. Pathophysiology of Type 2 Diabetes Mellitus. Int. J. Mol. Sci. 2020, 21, 6275. [Google Scholar] [CrossRef]
- Harding, J.L.; Pavkov, M.E.; Magliano, D.J.; Shaw, J.E.; Gregg, E.W. Global trends in diabetes complications: A review of current evidence. Diabetologia 2019, 62, 3–16. [Google Scholar] [CrossRef]
- Darenskaya, M.A.; Kolesnikova, L.I.; Kolesnikov, S.I. Oxidative stress: Pathogenetic role in diabetes mellitus and its complications and therapeutic approaches to correction. Bull. Exp. Biol. Med. 2021, 171, 179–189. [Google Scholar] [CrossRef]
- Eguchi, N.; Vaziri, N.D.; Dafoe, D.C.; Ichii, H. The role of oxidative stress in pancreatic beta cell dysfunction in diabetes. Int. J. Mol. Sci. 2021, 22, 1509. [Google Scholar] [CrossRef]
- Zheng, M.; Yu, H.; Xue, Y.; Yang, T.; Tu, Q.; Xiong, K.; Deng, D.; Lu, L.; Huang, N. The protective effect of hydrogen-rich water on rats with type 2 diabetes mellitus. Mol. Cell. Biochem. 2021, 476, 3089–3097. [Google Scholar] [CrossRef]
- Zhang, X.; Liu, J.; Jin, K.; Xu, H.; Wang, C.; Zhang, Z.; Kong, M.; Zhang, Z.; Wang, Q.; Wang, F. Subcutaneous injection of hydrogen gas is a novel effective treatment for type 2 diabetes. J. Diabetes Investig. 2018, 9, 83–90. [Google Scholar] [CrossRef]
- Wang, Q.; Zha, X.; Kang, Z.; Xu, M.; Huang, Q.; Zou, D. Therapeutic effects of hydrogen saturated saline on rat diabetic model and insulin resistant model via reduction of oxidative stress. Chin. Med. J. 2012, 125, 1633–1637. [Google Scholar] [CrossRef]
- Retnaningtyas, E.; Susatia, B.; Arifah, S.N.; Rahayu Lestari, S. The improvement of insulin level after hydrogen-rich water therapy in streptozotocin-induced diabetic rats. Vet. World 2022, 15, 182–187. [Google Scholar] [CrossRef]
- Ming, Y.; Ma, Q.H.; Han, X.L.; Li, H.Y. Molecular hydrogen improves type 2 diabetes through inhibiting oxidative stress. Exp. Ther. Med. 2020, 20, 359–366. [Google Scholar] [CrossRef]
- Kamimura, N.; Nishimaki, K.; Ohsawa, I.; Ohta, S. Molecular hydrogen improves obesity and diabetes by inducing hepatic FGF21 and stimulating energy metabolism in db/db mice. Obesity 2011, 19, 1396–1403. [Google Scholar] [CrossRef]
- Amitani, H.; Asakawa, A.; Cheng, K.; Amitani, M.; Kaimoto, K.; Nakano, M.; Ushikai, M.; Li, Y.; Tsai, M.; Li, J.B.; et al. Hydrogen improves glycemic control in type1 diabetic animal model by promoting glucose uptake into skeletal muscle. PLoS ONE 2013, 8, e53913. [Google Scholar] [CrossRef]
- Kim, M.J.; Kim, H.K. Anti-diabetic effects of electrolyzed reduced water in streptozotocin-induced and genetic diabetic mice. Life Sci. 2006, 79, 2288–2292. [Google Scholar] [CrossRef]
- Li, Y.; Hamasaki, T.; Nakamichi, N.; Kashiwagi, T.; Komatsu, T.; Ye, J.; Teruya, K.; Abe, M.; Yan, H.; Kinjo, T.; et al. Suppressive effects of electrolyzed reduced water on alloxan-induced apoptosis and type 1 diabetes mellitus. Cytotechnology 2011, 63, 119–131. [Google Scholar] [CrossRef]
- Zhang, M.; Lv, X.Y.; Li, J.; Xu, Z.G.; Chen, L. The characterization of high-fat diet and multiple low-dose streptozotocin induced type 2 diabetes rat model. Exp. Diabetes Res. 2008, 2008, 704045. [Google Scholar] [CrossRef] [PubMed]
- Keane, K.N.; Cruzat, V.F.; Carlessi, R.; de Bittencourt, P.I., Jr.; Newsholme, P. Molecular events linking oxidative stress and inflammation to insulin resistance and beta-cell dysfunction. Oxid. Med. Cell. Longev. 2015, 2015, 181643. [Google Scholar] [CrossRef] [PubMed]
- Lewis, J.E.; Ebling, F.J.P.; Samms, R.J.; Tsintzas, K. Going back to the biology of FGF21: New insights. Trends Endocrinol. Metab. 2019, 30, 491–504. [Google Scholar] [CrossRef] [PubMed]
- LeBaron, T.W.; Sharpe, R.; Ohno, K. Electrolyzed-reduced water: Review I. molecular hydrogen is the exclusive agent responsible for the therapeutic effects. Int. J. Mol. Sci. 2022, 23, 14750. [Google Scholar] [CrossRef]
- Yu, Y.; Ma, X.; Yang, T.A.O.; Li, B.O.; Xie, K.; Liu, D.; Wang, G.; Yu, Y. Protective effect of hydrogen-rich medium against high glucose-induced apoptosis of Schwann cells in vitro. Mol. Med. Rep. 2015, 12, 3986–3992. [Google Scholar] [CrossRef]
- Jiao, Y.; Yu, Y.; Li, B.; Gu, X.; Xie, K.; Wang, G.; Yu, Y. Protective effects of hydrogen-rich saline against experimental diabetic peripheral neuropathy via activation of the mitochondrial ATP-sensitive potassium channel channels in rats. Mol. Med. Rep. 2019, 21, 282–290. [Google Scholar] [CrossRef]
- Li, Q.; Jiao, Y.; Yu, Y.; Wang, G.; Yu, Y. Hydrogen-rich medium alleviates high glucose-induced oxidative stress and parthanatos in rat Schwann cells in vitro. Mol. Med. Rep. 2019, 19, 338–344. [Google Scholar] [CrossRef]
- Wang, P.; You, L.; Li, Q.; Wang, H.; Long, Y.; Chen, W. Hydrogen relieves neuropathic pain in diabetic rats by inhibiting MCP1 and CCR2 expressions. Kafkas Univ. Vet. Fak. Derg. 2022, 22, 169–176. [Google Scholar] [CrossRef]
- Han, X.C.; Ye, Z.H.; Hu, H.J.; Sun, Q.; Fan, D.F. Hydrogen exerts neuroprotective effects by inhibiting oxidative stress in experimental diabetic peripheral neuropathy rats. Med. Gas Res. 2023, 13, 72–77. [Google Scholar] [CrossRef]
- Feng, Y.; Wang, R.; Xu, J.; Sun, J.; Xu, T.; Gu, Q.; Wu, X. Hydrogen-rich saline prevents early neurovascular dysfunction resulting from inhibition of oxidative stress in STZ-diabetic Rats. Curr. Eye Res. 2012, 38, 396–404. [Google Scholar] [CrossRef]
- Xiao, X.; Cai, J.; Xu, J.; Wang, R.; Cai, J.; Liu, Y.; Xu, W.; Sun, X.; Li, R. Protective effects of hydrogen saline on diabetic retinopathy in a streptozotocin-induced diabetic rat model. J. Ocul. Pharmacol. Ther. 2012, 28, 76–82. [Google Scholar] [CrossRef]
- Wu, F.; Qiu, Y.; Ye, G.; Luo, H.; Jiang, J.; Yu, F.; Zhou, W.; Zhang, S.; Feng, J. Treatment with hydrogen molecule attenuates cardiac dysfunction in streptozotocin-induced diabetic mice. Cardiovasc. Pathol. 2015, 24, 294–303. [Google Scholar] [CrossRef]
- Zou, R.; Nie, C.; Pan, S.; Wang, B.; Hong, X.; Xi, S.; Bai, J.; Yu, M.; Liu, J.; Yang, W. Co-administration of hydrogen and metformin exerts cardioprotective effects by inhibiting pyroptosis and fibrosis in diabetic cardiomyopathy. Free Radic. Biol. Med. 2022, 183, 35–50. [Google Scholar] [CrossRef]
- Zheng, Y.; Zhang, Z.; Wang, T.; Zhang, J.; Tian, D.; Zhang, X.; Wu, Z. Photodriven nanoreactor with a hydrogen-insulin double act repairs diabetic wounds through Nrf2 pathway activation. Chem. Eng. J. 2021, 425, 131800. [Google Scholar] [CrossRef]
- Chen, H.; Guo, Y.; Zhang, Z.; Mao, W.; Shen, C.; Xiong, W.; Yao, Y.; Zhao, X.; Hu, Y.; Zou, Z.; et al. Symbiotic algae-bacteria dressing for producing hydrogen to accelerate diabetic wound healing. Nano Lett. 2022, 22, 229–237. [Google Scholar] [CrossRef]
- Wang, P.; Wu, J.; Yang, H.; Liu, H.; Yao, T.; Liu, C.; Gong, Y.; Wang, M.; Ji, G.; Huang, P.; et al. Intelligent microneedle patch with prolonged local release of hydrogen and magnesium ions for diabetic wound healing. Bioact. Mater. 2023, 24, 463–476. [Google Scholar] [CrossRef]
- Guo, J.; Dong, W.; Jin, L.; Wang, P.; Hou, Z.; Zhang, Y. Hydrogen-rich saline prevents bone loss in diabetic rats induced by streptozotocin. Int. Orthop. 2017, 41, 2119–2128. [Google Scholar] [CrossRef]
- Fan, M.; Xu, X.; He, X.; Chen, L.; Qian, L.; Liu, J.; Qing, J.; Chao, Z.; Sun, X. Protective effects of hydrogen-rich saline against erectile dysfunction in a streptozotocin induced diabetic rat model. J. Urol. 2013, 190, 350–356. [Google Scholar] [CrossRef]
- Kajiyama, S.; Hasegawa, G.; Asano, M.; Hosoda, H.; Fukui, M.; Nakamura, N.; Kitawaki, J.; Imai, S.; Nakano, K.; Ohta, M.; et al. Supplementation of hydrogen-rich water improves lipid and glucose metabolism in patients with type 2 diabetes or impaired glucose tolerance. Nutr. Res. 2008, 28, 137–143. [Google Scholar] [CrossRef]
- Ogawa, S.; Ohsaki, Y.; Shimizu, M.; Nako, K.; Okamura, M.; Kabayama, S.; Tabata, K.; Tanaka, Y.; Ito, S. Electrolyzed hydrogen-rich water for oxidative stress suppression and improvement of insulin resistance: A multicenter prospective double-blind randomized control trial. Diabetol. Int. 2022, 13, 209–219. [Google Scholar] [CrossRef]
- Ramadhan, A.; Wicaksono, S.A.; Nugroho, T.E.; Utami, S.B. The effects of alkaline ionized water administration to the total cholesterol levels in patients with type 2 diabetes mellitus accompanied by dyslipidemia. Pak. J. Med. Health Sci. 2021, 15, 1449–1455. [Google Scholar] [CrossRef]
- Nakao, A.; Toyoda, Y.; Sharma, P.; Evans, M.; Guthrie, N. Effectiveness of hydrogen rich water on antioxidant status of subjects with potential metabolic syndrome-an open label pilot study. J. Clin. Biochem. Nutr. 2010, 46, 140–149. [Google Scholar] [CrossRef] [PubMed]
- Song, G.; Li, M.; Sang, H.; Zhang, L.; Li, X.; Yao, S.; Yu, Y.; Zong, C.; Xue, Y.; Qin, S. Hydrogen-rich water decreases serum LDL-cholesterol levels and improves HDL function in patients with potential metabolic syndrome. J. Lipid Res. 2013, 54, 1884–1893. [Google Scholar] [CrossRef] [PubMed]
- LeBaron, T.W.; Singh, R.B.; Fatima, G.; Kartikey, K.; Sharma, J.P.; Ostojic, S.M.; Gvozdjakova, A.; Kura, B.; Noda, M.; Mojto, V.; et al. The effects of 24-Week, high-concentration hydrogen-rich water on body composition, blood lipid profiles and inflammation biomarkers in men and women with metabolic syndrome: A randomized controlled trial. Diabetes Metab. Syndr. Obes. 2020, 13, 889–896. [Google Scholar] [CrossRef] [PubMed]
- Korovljev, D.; Stajer, V.; Ostojic, J.; LeBaron, T.W.; Ostojic, S.M. Hydrogen-rich water reduces liver fat accumulation and improves liver enzyme profiles in patients with non-alcoholic fatty liver disease: A randomized controlled pilot trial. Clin. Res. Hepatol. Gastroenterol. 2019, 43, 688–693. [Google Scholar] [CrossRef]
- Kura, B.; Szantova, M.; LeBaron, T.W.; Mojto, V.; Barancik, M.; Szeiffova Bacova, B.; Kalocayova, B.; Sykora, M.; Okruhlicova, L.; Tribulova, N.; et al. Biological effects of hydrogen water on subjects with NAFLD: A randomized, placebo-controlled trial. Antioxidants 2022, 11, 1935. [Google Scholar] [CrossRef]
- Tao, G.; Zhang, G.; Chen, W.; Yang, C.; Xue, Y.; Song, G.; Qin, S. A randomized, placebo-controlled clinical trial of hydrogen/oxygen inhalation for non-alcoholic fatty liver disease. J. Cell. Mol. Med. 2022, 26, 4113–4123. [Google Scholar] [CrossRef]
- Sakai, T.; Sato, B.; Hara, K.; Hara, Y.; Naritomi, Y.; Koyanagi, S.; Hara, H.; Nagao, T.; Ishibashi, T. Consumption of water containing over 3.5 mg of dissolved hydrogen could improve vascular endothelial function. Vasc. Health Risk Manag. 2014, 10, 591–597. [Google Scholar] [CrossRef]
- Ishibashi, T.; Kawamoto, K.; Matsuno, K.; Ishihara, G.; Baba, T.; Komori, N. Peripheral endothelial function can be improved by daily consumption of water containing over 7 ppm of dissolved hydrogen: A randomized controlled trial. PLoS ONE 2020, 15, e0233484. [Google Scholar] [CrossRef]
- Korovljev, D.; Trivic, T.; Drid, P.; Ostojic, S.M. Molecular hydrogen affects body composition, metabolic profiles, and mitochondrial function in middle-aged overweight women. Ir. J. Med. Sci. 2018, 187, 85–89. [Google Scholar] [CrossRef]
- Asada, R.; Saitoh, Y.; Miwa, N. Effects of hydrogen-rich water bath on visceral fat and skin blotch, with boiling-resistant hydrogen bubbles. Med. Gas Res. 2019, 9, 68–73. [Google Scholar] [CrossRef]
- Hashimoto, M.; Katakura, M.; Nabika, T.; Tanabe, Y.; Hossain, S.; Tsuchikura, S.; Shido, O. Effects of hydrogen-rich water on abnormalities in a SHR.Cg-Leprcp/NDmcr rat—A metabolic syndrome rat model. Med. Gas Res. 2011, 1, 26. [Google Scholar] [CrossRef]
- Katakura, M.; Hashimoto, M.; Tanabe, Y.; Shido, O. Hydrogen-rich water inhibits glucose and α,β -dicarbonyl compound-induced reactive oxygen species production in the SHR.Cg-Leprcp/NDmcr rat kidney. Med. Gas Res. 2012, 2, 18. [Google Scholar] [CrossRef]
- Lin, C.P.; Chuang, W.C.; Lu, F.J.; Chen, C.Y. Anti-oxidant and anti-inflammatory effects of hydrogen-rich water alleviate ethanol-induced fatty liver in mice. World J. Gastroenterol. 2017, 23, 4920–4934. [Google Scholar] [CrossRef]
- Zhai, X.; Chen, X.; Lu, J.; Zhang, Y.; Sun, X.; Huang, Q.; Wang, Q. Hydrogen-rich saline improves non-alcoholic fatty liver disease by alleviating oxidative stress and activating hepatic PPARalpha and PPARgamma. Mol. Med. Rep. 2017, 15, 1305–1312. [Google Scholar] [CrossRef]
- Wang, X.; Wang, J. High-content hydrogen water-induced downregulation of miR-136 alleviates non-alcoholic fatty liver disease by regulating Nrf2 via targeting MEG3. Biol. Chem. 2018, 399, 397–406. [Google Scholar] [CrossRef]
- Li, S.; Fujino, M.; Ichimaru, N.; Kurokawa, R.; Hirano, S.; Mou, L.; Takahara, S.; Takahara, T.; Li, X.K. Molecular hydrogen protects against ischemia-reperfusion injury in a mouse fatty liver model via regulating HO-1 and Sirt1 expression. Sci. Rep. 2018, 8, 14019. [Google Scholar] [CrossRef]
- Liu, B.; Xue, J.; Zhang, M.; Wang, M.; Ma, T.; Zhao, M.; Gu, Q.; Qin, S. Hydrogen inhalation alleviates nonalcoholic fatty liver disease in metabolic syndrome rats. Mol. Med. Rep. 2020, 22, 2860–2868. [Google Scholar] [CrossRef]
- Li, S.W.; Takahara, T.; Que, W.; Fujino, M.; Guo, W.Z.; Hirano, S.I.; Ye, L.P.; Li, X.K. Hydrogen-rich water protects against liver injury in nonalcoholic steatohepatitis through HO-1 enhancement via IL-10 and Sirt 1 signaling. Am. J. Physiol. Gastrointest. Liver Physiol. 2021, 320, G450–G463. [Google Scholar] [CrossRef]
- Jin, Z.; Sun, Y.; Yang, T.; Tan, L.; Lv, P.; Xu, Q.; Tao, G.; Qin, S.; Lu, X.; He, Q. Nanocapsule-mediated sustained H2 release in the gut ameliorates metabolic dysfunction-associated fatty liver disease. Biomaterials 2021, 276, 121030. [Google Scholar] [CrossRef]
- Kawai, D.; Takaki, A.; Nakatsuka, A.; Wada, J.; Tamaki, N.; Yasunaka, T.; Koike, K.; Tsuzaki, R.; Matsumoto, K.; Miyake, Y.; et al. Hydrogen-rich water prevents progression of nonalcoholic steatohepatitis and accompanying hepatocarcinogenesis in mice. Hepatology 2012, 56, 912–921. [Google Scholar] [CrossRef]
- Ohsawa, I.; Nishimaki, K.; Yamagata, K.; Ishikawa, M.; Ohta, S. Consumption of hydrogen water prevents atherosclerosis in apolipoprotein E knockout mice. Biochem. Biophys. Res. Commun. 2008, 377, 1195–1198. [Google Scholar] [CrossRef] [PubMed]
- Song, G.; Tian, H.; Liu, J.; Zhang, H.; Sun, X.; Qin, S. H2 inhibits TNF-alpha-induced lectin-like oxidized LDL receptor-1 expression by inhibiting nuclear factor kappaB activation in endothelial cells. Biotechnol. Lett. 2011, 33, 1715–1722. [Google Scholar] [CrossRef] [PubMed]
- Song, G.; Tian, H.; Qin, S.; Sun, X.; Yao, S.; Zong, C.; Luo, Y.; Liu, J.; Yu, Y.; Sang, H.; et al. Hydrogen decreases athero-susceptibility in apolipoprotein B-containing lipoproteins and aorta of apolipoprotein E knockout mice. Atherosclerosis 2012, 221, 55–65. [Google Scholar] [CrossRef] [PubMed]
- Song, G.; Zong, C.; Zhang, Z.; Yu, Y.; Yao, S.; Jiao, P.; Tian, H.; Zhai, L.; Zhao, H.; Tian, S.; et al. Molecular hydrogen stabilizes atherosclerotic plaque in low-density lipoprotein receptor-knockout mice. Free Radic. Biol. Med. 2015, 87, 58–68. [Google Scholar] [CrossRef]
- Yang, S.; He, J.; Li, X.; Liu, H.; Zhao, J.; Liu, M. Hydrogen attenuated oxidized low-density lipoprotein-induced inflammation through the stimulation of autophagy via sirtuin 1. Exp. Ther. Med. 2018, 16, 4042–4048. [Google Scholar] [CrossRef]
- Hu, R.; Dai, C.; Dong, C.; Ding, L.; Huang, H.; Chen, Y.; Zhang, B. Living macrophage-delivered tetrapod PdH nanoenzyme for targeted atherosclerosis management by ROS scavenging, hydrogen anti-inflammation, and autophagy activation. ACS Nano 2022, 16, 15959–15976. [Google Scholar] [CrossRef]
- Ignacio, R.M.; Kang, T.Y.; Kim, C.S.; Kim, S.K.; Yang, Y.C.; Sohn, J.H.; Lee, K.J. Anti-obesity effect of alkaline reduced water in high fat-fed obese mice. Biol. Pharm. Bull. 2013, 36, 1052–1059. [Google Scholar] [CrossRef]
- Kamimura, N.; Ichimiya, H.; Iuchi, K.; Ohta, S. Molecular hydrogen stimulates the gene expression of transcriptional coactivator PGC-1alpha to enhance fatty acid metabolism. NPJ Aging Mech. Dis. 2016, 2, 16008. [Google Scholar] [CrossRef]
- Qiu, X.; Ye, Q.; Sun, M.; Wang, L.; Tan, Y.; Wu, G. Saturated hydrogen improves lipid metabolism disorders and dysbacteriosis induced by a high-fat diet. Exp. Biol. Med. 2020, 245, 512–521. [Google Scholar] [CrossRef]
- Masuda, H.; Sato, A.; Miyata, K.; Shizuno, T.; Oyamada, A.; Ishiwata, K.; Nakagawa, Y.; Asahara, T. Drinking molecular hydrogen water is beneficial to cardiovascular function in diet-induced obesity mice. Biology 2021, 10, 364. [Google Scholar] [CrossRef]
- Liu, B.; Zhao, M.; Xue, J.; Gu, Q.; Zhang, X.; Qin, S. Hydrogen influences HDL-associated enzymes and reduces oxidized phospholipids levels in rats fed with a high-fat diet. Life Sci. 2021, 267, 118945. [Google Scholar] [CrossRef]
- Chan, K.L.; Cathomas, F.; Russo, S.J. Central and peripheral inflammation link metabolic syndrome and major depressive disorder. Physiology 2019, 34, 123–133. [Google Scholar] [CrossRef]
- Zhao, L.; Wang, Y.; Zhang, G.; Zhang, T.; Lou, J.; Liu, J. L-Arabinose elicits gut-derived hydrogen production and ameliorates metabolic syndrome in C57BL/6J mice on high-fat-diet. Nutrients 2019, 11, 3054. [Google Scholar] [CrossRef]
- Garcia-Ruiz, C.; Fernandez-Checa, J.C. Mitochondrial oxidative stress and antioxidants balance in fatty liver disease. Hepatol. Commun. 2018, 2, 1425–1439. [Google Scholar] [CrossRef]
- Toshikuni, N.; Tsutsumi, M.; Arisawa, T. Clinical differences between alcoholic liver disease and nonalcoholic fatty liver disease. World J. Gastroenterol. 2014, 20, 8393–8406. [Google Scholar] [CrossRef]
- Eslam, M.; Newsome, P.N.; Sarin, S.K.; Anstee, Q.M.; Targher, G.; Romero-Gomez, M.; Zelber-Sagi, S.; Wai-Sun Wong, V.; Dufour, J.-F.; Schattenberg, J.M.; et al. A new definition for metabolic dysfunction-associated fatty liver disease: An international expert consensus statement. J. Hepatol. 2020, 73, 202–209. [Google Scholar] [CrossRef]
- Makinen, P.; Ruotsalainen, A.K.; Yla-Herttuala, S. Nucleic acid-based therapies for atherosclerosis. Curr. Atheroscler. Rep. 2020, 22, 10. [Google Scholar] [CrossRef]
- Yao, B.C.; Meng, L.B.; Hao, M.L.; Zhang, Y.M.; Gong, T.; Guo, Z.G. Chronic stress: A critical risk factor for atherosclerosis. J. Int. Med. Res. 2019, 47, 1429–1440. [Google Scholar] [CrossRef]
- Lustig, R.H.; Collier, D.; Kassotis, C.; Roepke, T.A.; Kim, M.J.; Blanc, E.; Barouki, R.; Bansal, A.; Cave, M.C.; Chatterjee, S.; et al. Obesity I: Overview and molecular and biochemical mechanisms. Biochem. Pharmacol. 2022, 199, 115012. [Google Scholar] [CrossRef]
- Broskey, N.T.; Zou, K.; Dohm, G.L.; Houmard, J.A. Plasma lactate as a marker for metabolic health. Exerc. Sport Sci. Rev. 2020, 48, 119–124. [Google Scholar] [CrossRef] [PubMed]
- Ishibashi, T. Therapeutic efficacy of molecular hydrogen: A new mechanistic insight. Curr. Pharm. Des. 2019, 25, 946–955. [Google Scholar] [CrossRef] [PubMed]
- Ishihara, G.; Kawamoto, K.; Komori, N.; Ishibashi, T. Molecular hydrogen suppresses superoxide generation in the mitochondrial complex I and reduced mitochondrial membrane potential. Biochem. Biophys. Res. Commun. 2020, 522, 965–970. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Zhang, Z.; Wei, Y.; Li, M.; Zhao, P.; Adzavon, Y.M.; Liu, M.; Zhang, X.; Xie, F.; Wang, A. Mitochondria in higher plants possess H2 evolving activity which is closely related to complex I. arXiv 2020, arXiv:2001.02132. [Google Scholar]
- Matzinger, M.; Fischhuber, K.; Heiss, E.H. Activation of Nrf2 signaling by natural products-can it alleviate diabetes? Biotechnol. Adv. 2018, 36, 1738–1767. [Google Scholar] [CrossRef]
- Ryter, S.W. Heme oxygenase-1: An anti-inflammatory effector in cardiovascular, lung, and related metabolic disorders. Antioxidants 2022, 11, 555. [Google Scholar] [CrossRef]
- Ryter, S.W. Heme oxygenase-1/carbon monoxide as modulators of autophagy and inflammation. Arch. Biochem. Biophys. 2019, 678, 108186. [Google Scholar] [CrossRef]
- Liu, T.; Zhang, L.; Joo, D.; Sun, S.C. NF-kappaB signaling in inflammation. Signal Transduct. Target Ther. 2017, 2, 17023. [Google Scholar] [CrossRef]
- Wardyn, J.D.; Ponsford, A.H.; Sanderson, C.M. Dissecting molecular cross-talk between Nrf2 and NF-kappaB response pathways. Biochem. Soc. Trans. 2015, 43, 621–626. [Google Scholar] [CrossRef]
- You, S.; Zheng, J.; Chen, Y.; Huang, H. Research progress on the mechanism of beta-cell apoptosis in type 2 diabetes mellitus. Front. Endocrinol. 2022, 13, 976465. [Google Scholar] [CrossRef]
- Yue, J.; Lopez, J.M. Understanding MAPK signaling pathways in apoptosis. Int. J. Mol. Sci. 2020, 21, 2346. [Google Scholar] [CrossRef]
- Lemmer, I.L.; Willemsen, N.; Hilal, N.; Bartelt, A. A guide to understanding endoplasmic reticulum stress in metabolic disorders. Mol. Metab. 2021, 47, 101169. [Google Scholar] [CrossRef]
- Bhardwaj, M.; Leli, N.M.; Koumenis, C.; Amaravadi, R.K. Regulation of autophagy by canonical and non-canonical ER stress responses. Semin. Cancer Biol. 2020, 66, 116–128. [Google Scholar] [CrossRef]
- Cullinan, S.B.; Diehl, J.A. Coordination of ER and oxidative stress signaling: The PERK/Nrf2 signaling pathway. Int. J. Biochem. Cell Biol. 2006, 38, 317–332. [Google Scholar] [CrossRef]
- Kitada, M.; Koya, D. Autophagy in metabolic disease and ageing. Nat. Rev. Endocrinol. 2021, 17, 647–661. [Google Scholar] [CrossRef]
- Bhatti, J.S.; Bhatti, G.K.; Reddy, P.H. Mitochondrial dysfunction and oxidative stress in metabolic disorders—A step towards mitochondria based therapeutic strategies. Biochim. Biophys. Acta Mol. Basis Dis. 2017, 1863, 1066–1077. [Google Scholar] [CrossRef]
- Liang, W.; Chen, M.; Zheng, D.; Li, J.; Song, M.; Zhang, W.; Feng, J.; Lan, J. The opening of ATP-sensitive K+ channels protects H9c2 cardiac cells against the high glucose-induced injury and inflammation by inhibiting the ROS-TLR4-necroptosis pathway. Cell. Physiol. Biochem. 2017, 41, 1020–1034. [Google Scholar] [CrossRef]
- Facundo, H.T.; de Paula, J.G.; Kowaltowski, A.J. Mitochondrial ATP-sensitive K+ channels are redox-sensitive pathways that control reactive oxygen species production. Free Radic. Biol. Med. 2007, 42, 1039–1048. [Google Scholar] [CrossRef]
- Adzavon, Y.M.; Xie, F.; Yi, Y.; Jiang, X.; Zhang, X.; He, J.; Zhao, P.; Liu, M.; Ma, S.; Ma, X. Long-term and daily use of molecular hydrogen induces reprogramming of liver metabolism in rats by modulating NADP/NADPH redox pathways. Sci. Rep. 2022, 12, 3904. [Google Scholar] [CrossRef]
- Aw, W.; Fukuda, S. Understanding the role of the gut ecosystem in diabetes mellitus. J. Diabetes Investig. 2018, 9, 5–12. [Google Scholar] [CrossRef]
- Agus, A.; Clement, K.; Sokol, H. Gut microbiota-derived metabolites as central regulators in metabolic disorders. Gut 2021, 70, 1174–1182. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Zhao, L.; Liu, M.; Xie, F.; Ma, X.; Zhao, P.; Liu, Y.; Li, J.; Wang, M.; Yang, Z.; et al. Oral intake of hydrogen-rich water ameliorated chlorpyrifos-induced neurotoxicity in rats. Toxicol. Appl. Pharmacol. 2014, 280, 169–176. [Google Scholar] [CrossRef] [PubMed]
- Yi, Y.; Zhang, X.; Guo, B.; Xie, F.; Zhang, Z.; Ma, S.; Zhao, P.; Li, y.; Ma, X. Effect of hydrogen on horseradish peroxidase activity and its mechanism. Chin. J. Biochem. Mol. Biol. 2020, 36, 811–819. [Google Scholar]
- Jin, Z.; Zhao, P.; Gong, W.; Ding, W.; He, Q. Fe-porphyrin: A redox-related biosensor of hydrogen molecule. Nano Res. 2022, 16, 2020–2025. [Google Scholar] [CrossRef]
- Spiro, T.G.; Soldatova, A.V.; Balakrishnan, G. CO, NO and O2 as Vibrational Probes of Heme Protein Interactions. Coord. Chem. Rev. 2013, 257, 511–527. [Google Scholar] [CrossRef]
- Bieza, S.; Mazzeo, A.; Pellegrino, J.; Doctorovich, F. H2S/Thiols, NO•, and NO−/HNO: Interactions with Iron Porphyrins. ACS Omega 2022, 7, 1602–1611. [Google Scholar] [CrossRef]
- Ohta, S. Molecular hydrogen may activate the transcription factor Nrf2 to alleviate oxidative stress through the hydrogen targeted porphyrin. Aging Pathobiol. Ther. 2023, 5, 25–32. [Google Scholar] [CrossRef]
- Zhu, L.; Luo, M.; Zhang, Y.; Fang, F.; Li, M.; An, F.; Zhao, D.; Zhang, J. Free radical as a double-edged sword in disease: Deriving strategic opportunities for nanotherapeutics. Coord. Chem. Rev. 2023, 475, 214875. [Google Scholar] [CrossRef]
- Andres, C.M.C.; Perez de la Lastra, J.M.; Andres Juan, C.; Plou, F.J.; Perez-Lebena, E. Superoxide Anion Chemistry—Its Role at the Core of the Innate Immunity. Int. J. Mol. Sci. 2023, 24, 1841. [Google Scholar] [CrossRef]
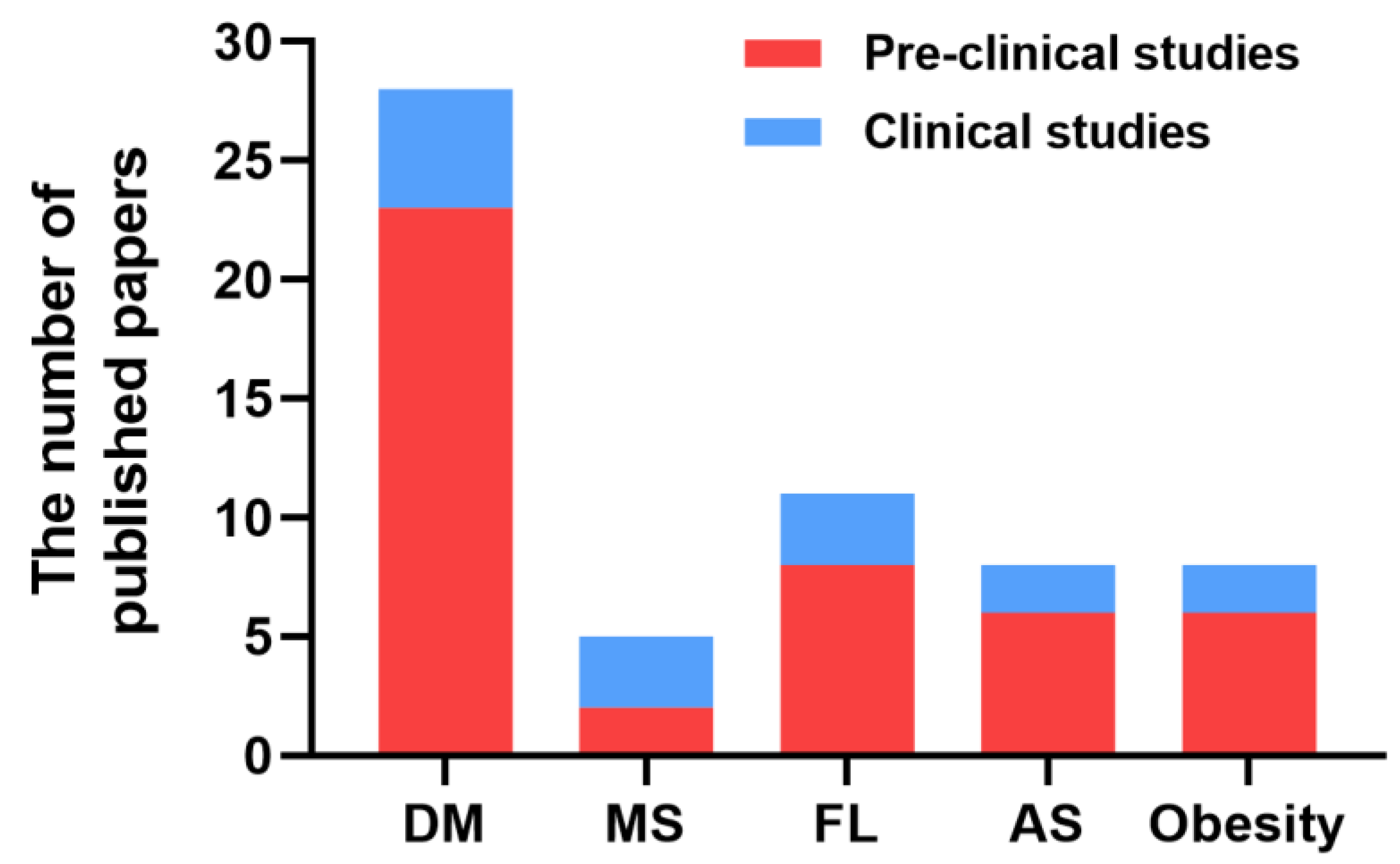

| Disease Type | Model System | Routes of H2 Delivery | H2 Concentration | Duration | Initial FBG Levels (mmol/L) | Reduction (%) | Main Effects | Reference |
|---|---|---|---|---|---|---|---|---|
| T2DM | HFD (4 weeks) and low-dose STZ (25 mg/kg)-induced DM rat | drinking HRW | >0.6 mM | 3 weeks | ~22 | ~14 | FBG, TG, TC, LDL-c, HDL-c, IL-1β, hepatic fat, renal and spleen tissue damage: ↓ GHb: NSC | [12] |
| T2DM | HFD (4 weeks) and STZ (100 mg/kg)-induced DM mice | subcutaneous injection of H2 gas | ~0.8 mM | 4 weeks | ~24 | ~40 | FBG, insulin, TG, LDL-c: ↓ HDL-c: ↑ glucose tolerance, insulin sensitivity: ↑ diabetic renal injury: ↓ | [13] |
| T2DM | HGHFD-induced (8 weeks) insulin resistance rat model, high glucose and HFD (4 weeks) and low-dose STZ (20 mg/kg)-induced DM rat | oral gavage of HRS | NA | 8 weeks | NA | IR: 12.50%; DM: 19.63% | IR: FBG, insulin, TG, TC, LDL-c: ↓, insulin sensitivity: ↑ DM: FBG, TG, TC, LDL-c: ↓ | [14] |
| T2DM | HFD (30 days) and low-dose STZ (35 mg/kg)-induced DM rat | drinking HRW | 0.5 mM | 2 weeks | NA | NA | insulin: ↑ IRs expression in the adipose and skeletal muscle tissues: ↑ | [15] |
| T2DM | HFD (4 weeks) and low-dose STZ (30 mg/kg)-induced DM rat | intravenous injection of HRS | >0.6 mM | 80 days | ~28 | ~50 | FBG, TG, TC, LDL-c: ↓ HDL-c: ↑ insulin resistance, pancreatic islets and glomeruli damage: ↓ insulin: NSC | [16] |
| T2DM | Leprdb/db mice | drinking HRW | 0.8 mM | 3 months | NA | 20.75 | FBG, insulin, TG, hepatic fat: ↓ | [17] |
| T2DM | HFD-induced mice, Leprdb/db mice | drinking HRW | 0.8 mM | HFD-induced mice: 25 weeks; db/db mice: 18 weeks | HFD-induced mice): 9.61; db/db mice: 16.56 | NA | FBG, GA, lipid parameters: NSC; | [18] |
| T1DM | STZ (50 mg/kg/day for 5 days)-induced T1DM mice | drinking HRW or intraperitoneal injection of HRS | 0.8 mM | HRW: 18 weeks; HRS: 4 weeks | NA | HRS: 19.31; HRW: NSC | HRS: FBG, GHb, TG: ↓ HRW: FBG: NSC; GHb, TG: ↓ | [18] |
| T2DM | Leprdb/db mice | drinking ERW | 0.15–0.3 mM | 4 weeks | ~9 | ~41 | FBG: ↓ insulin: ↑; glucose tolerance: NSC | [19] |
| T1DM | STZ (60 mg/kg/day for 5 days)-induced T1DM mice | drinking ERW | 0.15–0.3 mM | 6 weeks | NA | 38.98 | FBG: ↓ insulin: NSC glucose tolerance: ↑ | [19] |
| T1DM | Alloxan (100 mg/kg/injection, 3 injections)-induced T1DM mice | drinking ERW | ~0.5 mM | 9 weeks | 5.39 | 56.35 | FBG: ↓ insulin: ↑ | [20] |
| Disease/Objective | Subjects No. | Routes of H2 Delivery | H2 Concentration | Duration | Main Effects | Clinical Registration No. | Reference | |
|---|---|---|---|---|---|---|---|---|
| Male | Female | |||||||
| T2DM/IGT | 18 | 18 | drinking HRW | ~0.6 mM | 8 weeks | sdLDL, emLDL, u-IsoP: ↓, oxLDL: ⇓ glucose tolerance in 4 of 6 IGT patients: ↑ TG, TC, LDL, HDL, FBG, HbA1C, insulin: NSC | NA | [39] |
| T2DM | 25 | 20 | drinking ERW | NA | 12 weeks | lactate: ↓, FBG, insulin, HOMA-IR: NSC the rate of change in lactate was positively correlated with the rate of change in HOMA-IR, FBG and insulin in the HOMA-IR ≥1.73 group, in the FBG ≥6.1 mM group: insulin: ↓, FBG: ⇓ | UMIN000019032 | [40] |
| T2DM | 15 | 15 | drinking ERW | NA | 12 days | TC, TG, LDL: ↓, HDL: ↑ FBG: ⇓ | NA | [41] |
| MS | 10 | 10 | drinking HRW | 0.55–0.65 mM | 8 weeks | SOD: ↑, TBARS in urine: ↓ HDL: ↑ (week 4) LDL, TC/HDL: ↓ (week 4) TC, TG, FBG: NSC | NA | [42] |
| MS | 12 | 8 | drinking HRW | 0.2–0.25 mM | 10 weeks | TC, LDL, apoB100, apoE: ↓ TG, HDL, FBG: NSC MDA: ↓, SOD: ↑ TNF-α, IL-6: NSC | NA | [43] |
| MS | 30 | 30 | drinking high-concentration HRW (>5.5 mM) | >5.5 mmol H2 per day | 24 weeks | BMI, WHC, TG, TC, LDL, HDL, FBG, HbA1c: ↓ TNF-α, IL-6, CRP: ↓ MDA: ↓, TBARS: NSC Vitamin E, Vitamin C: ↑ | NA | [44] |
| NAFLD | 5 | 7 | drinking HRW | 3 mM | 4 weeks | liver fat, AST: ↓ BW, BMI, body composition, lipid profiles, FBG: NSC | NCT03625362 | [45] |
| NAFLD | 13 | 17 | drinking HRW | >2 mM | 8 weeks | BW, BMI, SBP, AST, ALT, LDH, NF-κB, HSP70, MMP-9: ⇓ CRP, ALB, ALP, TG, TC, HDL, LDL, TG/HDL ratio, 8-OHdG, MDA: ⇑ | NCT05325398 | [46] |
| NAFLD | 24 | 19 | Hydrogen/oxygen inhalation | 66% | 13 weeks | TC, TG, HDL: NSC LDL, AST, ALT, MDA, TNF-α, IL-6: ↓ SOD: ↑ liver fat in moderate–severe cases: ↓ | ChiCTR-IIR-16009114 | [47] |
| Vascular endothelial function | 18 | 16 | drinking high-concentration HRW (3.5 mM) | >3.5 mM | 1 time | the ratio of the changes in FMD of the BA: ↑ | NA | [48] |
| Vascular endothelial function | 24 | 44 | drinking high-concentration HRW (3.5 mM) | >3.5 mM | 2 weeks | Ln_RHI value in the low Ln_RHI group: ↑ Ln_RHI value in the high Ln_RHI group: NSC | NA | [49] |
| Obesity | 0 | 10 | oral intake of H2 generating minerals | ~6 ppm of H2 per day | 4 weeks | body fatness, arm fat index, TG, insulin: ↓ ghrelin: ⇑, lactate: ↓ BW, FBG, other lipid parameters: NSC | NCT02832219 | [50] |
| Obesity | 2 | 2 | HRW bath (41 °C, 0.15 mM) | ~0.15 mM | 4–24 weeks | visceral fat, LDL in two women: ↓ | NA | [51] |
| Disease Type | Model System | Routes of H2 Delivery | H2 Concentration | Duration | Main Effects | Reference |
|---|---|---|---|---|---|---|
| MS | SHR-cp rats | drinking HRW | 0.15–0.2 mM | 16 weeks | plasma BUN, creatinine:↓, BAP: ↑ plasma TG, TC, FBG, 8-OHdG: NSC, glomerulosclerosis score: ↓ 24 h water intake and urine flow: ↑ urinary albumin to creatinine ratio: ↓ | [53] |
| MS | SHR-cp rats | drinking HRW | 0.15–0.2 mM | 16 weeks | renal glyoxal, methylglyoxal, and 3-deoxyglucosone levels: ↓ ROS production in kidney: ↓ | [55] |
| ALD | chronic-binge ethanol-fed mice model | drinking HRW (0.25 mM) | 0.25–0.3 mM | 13 weeks | EtOH-induced anorexia and liver enlargement: ↓ serum TG, TC, ALT, TNF-α, IL-6, oxidative stress: ↓, IL-10, IL-22: ↑ hepatic TG, TC, TNF-α, IL-6: ↓ | [56] |
| NAFLD | STZ (25 mg/kg, single dose) + HFHSD (8 weeks)-induced rats model | intraperitoneal injection of HRS | >0.6 mM | 8 weeks | serum ALT, TBIL, TC, TG, FBG, insulin: ↓ insulin sensitivity, glucose tolerance: ↑ hepatocyte apoptosis: ↓ Inflammation, oxidative stress: ↓ | [57] |
| NAFLD | HFD-induced mice model | drinking HRW | 0.8 mM | 8 weeks | serum ALT, AST, TC, TG: ↓ hepatic expression of MEG3, Nrf2: ↑, miR-136: ↓ | [58] |
| NAFLD | MCDHF diet-induced mice model | HRS (3.5 mM) spread in the liver after clipping hepatic portal vein with a microvascular clamp for ischemia | 3.5 mM | 1 time | serum ALT, AST: ↓ ischemia–reperfusion injury in fatty liver: ↓ hepatic expression of TNF-α, IL-6, TLR-4, Nlrp3: ↓ | [59] |
| NAFLD | HFFD-induced rats model | hydrogen inhalation | 4% or 67% | 10 weeks | BW, BMI, abdominal fat index, liver index: ↓ glucose tolerance: ↓ serum TG, ALT, AST, LDH: ↓, TC: NSC hepatic fat: ↓ | [60] |
| NASH and fibrosis | CDAA diet (20 weeks)-induced mice model | drinking HRW | 3.5 mM | 4, 8, or 20 weeks | serum AST, ALT, TBARS: ↓ hepatic fibrosis, macrophage recruitment, HSC activation, expression of inflammatory cytokines: ↓ | [61] |
| NAFLD | HFD (16 weeks)-induced mice model and db/db mice (6 weeks) | oral intake of hydrogen nanocapsule-mixed HFD | >104 times higher than that of HRW | 16 or 6 weeks | BW, WAT mass, liver weight, liver steatosis: ↓ plasma TC, FBG, insulin: ↓, glucose tolerance: ↑ food intake, plasma TG: NSC | [62] |
| NASH and HCC | MCD diet (8 weeks)-induced mice model and HFD (8 weeks) induced-STAM® mice model | drinking HRW | 0.175–0.225 mM | 8 weeks | plasma ALT, hepatic TC, plasma and hepatic oxidative stress, hepatocyte apoptosis, NAS: ↓ hepatic expression of free fatty acid uptake-related enzymes, inflammatory cytokines, PPARα: ↓ hepatocarcinogenesis: ↓ | [63] |
| AS | apoE knockout mice model | drinking HRW | >0.6 mM | 8 weeks | atherosclerotic lesion: ↓ oxidative stress level of aorta: ↓ | [64] |
| AS | apoE knockout mice model | intraperitoneal injection of HRS | >0.6 mM | 8 weeks | LOX-1 expression and NF-κB activation in thoracic aorta: ↓ | [65] |
| AS | apoE knockout mice model | intraperitoneal injection of HRS | >0.6 mM | 8 weeks | plasma TC, non-HDL-C, MDA, SAA: ↓, PON-1: ↑ plasma and hepatic apoB: ↓ aortic inflammation: ↓ RCT-related genes expression: ↑ oxidation of non-HDL: ↓, HDL quality: ↑ | [66] |
| AS | Leprdb/db mice fed an atherogenic diet | intraperitoneal injection of HRS | >0.6 mM | 28 weeks | atherosclerotic plaque stability: ↑ the formation and apoptosis of macrophage-derived foam cells: ↓ serum ox-LDL, ROS in aorta, peritoneal macrophages: ↓ | [67] |
| AS | mouse macrophage-like cell line | cultured in HRM | 0.6 mM | 24 h | ox-LDL-induced inflammation: ↓ autophagic flux: ↑ | [68] |
| AS | apoE knockout mice model | intravenously injected with tetrapod needle-like PdH nanozyme loaded by macrophages | NA | 8 weeks | plaque area and the necrotic cores in aortic sinus sections: ↓ macrophage infiltration and MMP-9 expression in atherosclerotic plaques: ↓ stability of atherosclerotic plaques: ↑ | [69] |
| obesity | Leprdb/db mice, HFD-induced obesity mice | drinking HRW | 0.8 mM | 12 weeks | hepatic oxidative stress and lipid accumulation: ↓ BW, serum FBG, TG, insulin: ↓ TC, LDL, HDL: NSC | [17] |
| obesity | HFD-induced obesity mice | drinking ERW | 0.05 mM | 12 weeks | BW, epididymal fat, liver fat: ↓ blood neutrophils and lymphocytes: ↑ serum adiponectin: ↑ | [70] |
| obesity | HFD-induced obesity mice | gavage of MgH2 suspension | NA | 74 weeks | plasma TG: ↓ the average of lifespan: ↑ | [71] |
| obesity | HFD-induced mice | gavage of HRS | >0.6 mM | 2, 4, 6 weeks | BW, blood TC, TG, LDL: ↓, HDL: ↑ intestinal integrity: ↑ abundance of Bacteroides, Bifidobacteria, Lactobacillus in feces: ↑, Enterobacter cloacae: ↓, ICL activity: ↓ | [72] |
| obesity | HFD-induced mice | drinking HRW | ~0.45 mM | 2 weeks | heart weight: ↓, BAT mass: ↑ cardiac function, vascular bioactivity: ↑ FBG, BW, WAT mass, HR: NSC | [73] |
| obesity | HFD-induced rats | hydrogen inhalation | 4% | 10 weeks | hepatic FFA, MDA, 4-HNE, PGPC, PONPC, PAzPC: ↓, TC, TG: ⇓ plasma PONPC: ↓, TC, TG, FFA, MDA, 4-HNE: NSC plasma HDL antioxidant activity: ↑, Lp-PLA2 activity: ↓, PON-1 activity, LCAT expression: ↑ | [74] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xie, F.; Song, Y.; Yi, Y.; Jiang, X.; Ma, S.; Ma, C.; Li, J.; Zhanghuang, Z.; Liu, M.; Zhao, P.; et al. Therapeutic Potential of Molecular Hydrogen in Metabolic Diseases from Bench to Bedside. Pharmaceuticals 2023, 16, 541. https://doi.org/10.3390/ph16040541
Xie F, Song Y, Yi Y, Jiang X, Ma S, Ma C, Li J, Zhanghuang Z, Liu M, Zhao P, et al. Therapeutic Potential of Molecular Hydrogen in Metabolic Diseases from Bench to Bedside. Pharmaceuticals. 2023; 16(4):541. https://doi.org/10.3390/ph16040541
Chicago/Turabian StyleXie, Fei, Yifei Song, Yang Yi, Xue Jiang, Shiwen Ma, Chen Ma, Junyu Li, Ziyi Zhanghuang, Mengyu Liu, Pengxiang Zhao, and et al. 2023. "Therapeutic Potential of Molecular Hydrogen in Metabolic Diseases from Bench to Bedside" Pharmaceuticals 16, no. 4: 541. https://doi.org/10.3390/ph16040541
APA StyleXie, F., Song, Y., Yi, Y., Jiang, X., Ma, S., Ma, C., Li, J., Zhanghuang, Z., Liu, M., Zhao, P., & Ma, X. (2023). Therapeutic Potential of Molecular Hydrogen in Metabolic Diseases from Bench to Bedside. Pharmaceuticals, 16(4), 541. https://doi.org/10.3390/ph16040541





