Biological Potential and Bioaccessibility of Encapsulated Curcumin into Cetyltrimethylammonium Bromide Modified Cellulose Nanocrystals
Abstract
:1. Introduction
2. Results and Discussion
2.1. Characterization of Encapsulated Curcumin Particles
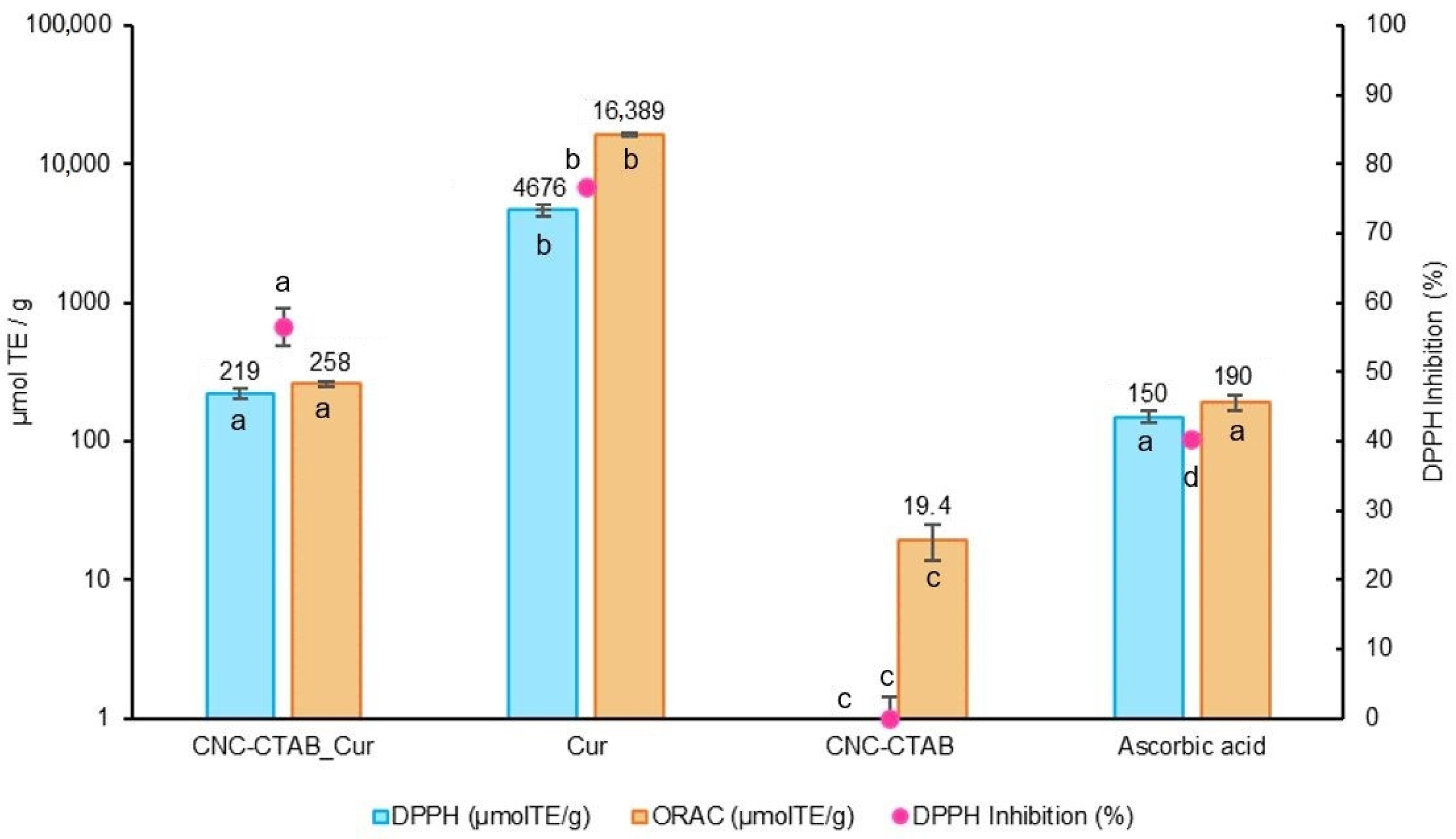
2.2. Biological Potential
2.2.1. Antioxidant Capacity
Production of Reactive Oxygen Species (ROS)
2.2.2. Immunomodulation
2.2.3. Antimicrobial Activity
2.3. Release Profile under Digestion Conditions
2.4. Intestinal Permeability
3. Materials and Methods
3.1. Reagents and Materials
3.2. Curcumin Encapsulation and Spray Drying
3.3. Characterization of Encapsulated Curcumin Particles
3.4. Cell Lines and Culture Conditions
3.5. Antioxidant Potential
3.5.1. 2,2-Diphenyl-1-Picrylhydrazyl-Free-Radical (DPPH) Assay
3.5.2. Oxygen Radical Absorbance Capacity (ORAC) Assay
3.5.3. Production of Reactive Oxygen Species
3.6. Immunomodulation
3.7. Antimicrobial Activity
3.8. Release Profile under Simulated Digestion
3.9. In Vitro Intestinal Permeability
3.10. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Madhavi, D.; Kagan, D. Bioavailability of a Sustained Release Formulation of Curcumin. Integr. Med. 2014, 13, 24–30. [Google Scholar]
- Rafiee, Z.; Nejatian, M.; Daeihamed, M.; Jafari, S.M. Application of Different Nanocarriers for Encapsulation of Curcumin. Crit. Rev. Food Sci. Nutr. 2019, 59, 3468–3497. [Google Scholar] [CrossRef] [PubMed]
- Amalraj, A.; Pius, A.; Gopi, S.; Gopi, S. Biological Activities of Curcuminoids, Other Biomolecules from Turmeric and Their Derivatives—A Review. J. Tradit. Complement. Med. 2017, 7, 205–233. [Google Scholar] [CrossRef] [PubMed]
- Hsu, K.Y.; Ho, C.T.; Pan, M.H. The Therapeutic Potential of Curcumin and Its Related Substances in Turmeric: From Raw Material Selection to Application Strategies. J. Food Drug Anal. 2023, 31, 194. [Google Scholar] [CrossRef] [PubMed]
- Goel, A.; Kunnumakkara, A.B.; Aggarwal, B.B. Curcumin as “Curecumin”: From Kitchen to Clinic. Biochem. Pharmacol. 2008, 75, 787–809. [Google Scholar] [CrossRef] [PubMed]
- Serda, M.; Becker, F.G.; Cleary, M.; Team, R.M.; Holtermann, H.; The, D.; Agenda, N.; Science, P.; Sk, S.K.; Hinnebusch, R.; et al. Anticancer Potential of Curcumin: Preclinical and Clinical Studies. Anticancer Res. 2003, 23, 363–398. [Google Scholar]
- Hsu, C.H.; Cheng, A.L. Clinical Studies with Curcumin. Adv. Exp. Med. Biol. 2007, 595, 471–480. [Google Scholar]
- Chen, A.L.; Hsu, C.H.; Lin, J.K.; Hsu, M.M.; Ho, Y.F.; She, T.S.; Ko, J.Y.; Lin, J.T.; Lin, B.R.; Wu, M.S.; et al. Phase I Clinical Trial of Curcumin, a Chemopreventive Agent, in Patients with High-Risk or Pre-Malignant Lesions. Anticancer Res. 2001, 21, 2895–2900. [Google Scholar]
- Sharma, R.A.; Euden, S.A.; Platton, S.L.; Cooke, D.N.; Shafayat, A.; Hewitt, H.R.; Marczylo, T.H.; Morgan, B.; Hemingway, D.; Plummer, S.M.; et al. Phase I Clinical Trial of Oral Curcumin: Biomarkers of Systemic Activity and Compliance. Clin. Cancer Res. 2004, 10, 6847–6854. [Google Scholar] [CrossRef]
- Anand, P.; Kunnumakkara, A.B.; Newman, R.A.; Aggarwal, B.B. Bioavailability of Curcumin: Problems and Promises. Mol. Pharm. 2007, 4, 807–818. [Google Scholar] [CrossRef]
- Omidian, H.; Wilson, R.L.; Chowdhury, S.D. Enhancing Therapeutic Efficacy of Curcumin: Advances in Delivery Systems and Clinical Applications. Gels 2023, 9, 596. [Google Scholar] [CrossRef] [PubMed]
- Casanova, F.; Pereira, C.F.; Ribeiro, A.B.; Freixo, R.; Costa, E.; Pintado, M.E.; Fernandes, J.C.; Ramos, Ó.L. Novel Micro- and Nanocellulose-Based Delivery Systems for Liposoluble Compounds. Nanomaterials 2021, 11, 2593. [Google Scholar] [CrossRef] [PubMed]
- Omidi, S.; Pirhayati, M.; Kakanejadifard, A. Co-Delivery of Doxorubicin and Curcumin by a PH-Sensitive, Injectable, and in Situ Hydrogel Composed of Chitosan, Graphene, and Cellulose Nanowhisker. Carbohydr. Polym. 2020, 231, 115745. [Google Scholar] [CrossRef]
- de Castro, D.O.; Tabary, N.; Martel, B.; Gandini, A.; Belgacem, N.; Bras, J. Controlled Release of Carvacrol and Curcumin: Bio-Based Food Packaging by Synergism Action of TEMPO-Oxidized Cellulose Nanocrystals and Cyclodextrin. Cellulose 2018, 25, 1249–1263. [Google Scholar] [CrossRef]
- Zainuddin, N.; Ahmad, I.; Kargarzadeh, H.; Ramli, S. Hydrophobic Kenaf Nanocrystalline Cellulose for the Binding of Curcumin. Carbohydr. Polym. 2017, 163, 261–269. [Google Scholar] [CrossRef] [PubMed]
- Foo, M.L.; Tan, C.R.; Lim, P.D.; Ooi, C.W.; Tan, K.W.; Chew, I.M.L. Surface-Modified Nanocrystalline Cellulose from Oil Palm Empty Fruit Bunch for Effective Binding of Curcumin. Int. J. Biol. Macromol. 2019, 138, 1064–1071. [Google Scholar] [CrossRef]
- Mohanta, V.; Madras, G.; Patil, S. Layer-by-Layer Assembled Thin Films and Microcapsules of Nanocrystalline Cellulose for Hydrophobic Drug Delivery. ACS Appl. Mater. Interfaces 2014, 6, 20093–20101. [Google Scholar] [CrossRef] [PubMed]
- Gunathilake, T.M.S.U.; Ching, Y.C.; Chuah, C.H. Enhancement of Curcumin Bioavailability Using Nanocellulose Reinforced Chitosan Hydrogel. Polymers 2017, 9, 64. [Google Scholar] [CrossRef]
- Ntoutoume, G.M.A.N.; Granet, R.; Mbakidi, J.P.; Brégier, F.; Léger, D.Y.; Fidanzi-Dugas, C.; Lequart, V.; Joly, N.; Liagre, B.; Chaleix, V.; et al. Development of Curcumin-Cyclodextrin/Cellulose Nanocrystals Complexes: New Anticancer Drug Delivery Systems. Bioorganic Med. Chem. Lett. 2016, 26, 941–945. [Google Scholar] [CrossRef]
- Mo, Y.; Guo, R.; Zhang, Y.; Xue, W.; Cheng, B.; Zhang, Y. Controlled Dual Delivery of Angiogenin and Curcumin by Electrospun Nanofibers for Skin Regeneration. Tissue Eng.—Part A 2017, 23, 597–608. [Google Scholar] [CrossRef]
- Casanova, F.; Pereira, C.F.; Ribeiro, A.B.; Costa, E.M.; Freixo, R.; Castro, P.M.; Fernandes, J.C.; Pintado, M.; Ramos, Ó.L. Design of Innovative Biocompatible Cellulose Nanostructures for the Delivery and Sustained Release of Curcumin. Pharmaceutics 2023, 15, 981. [Google Scholar] [CrossRef] [PubMed]
- Casanova, F.; Estevinho, B.N.; Santos, L. Preliminary Studies of Rosmarinic Acid Microencapsulation with Chitosan and Modified Chitosan for Topical Delivery. Powder Technol. 2016, 297, 44–49. [Google Scholar] [CrossRef]
- BUCHI Ibérica Buchi. Available online: https://www.buchi.com/en/spray-drying (accessed on 26 January 2023).
- de Souza, H.J.B.; Botrel, D.A.; de Barros Fernandes, R.V.; Borges, S.V.; Campelo Felix, P.H.; Viana, L.C.; Lago, A.M.T. Hygroscopic, Structural, and Thermal Properties of Essential Oil Microparticles of Sweet Orange Added with Cellulose Nanofibrils. J. Food Process. Preserv. 2020, 44, e14365. [Google Scholar] [CrossRef]
- Kolakovic, R.; Laaksonen, T.; Peltonen, L.; Laukkanen, A.; Hirvonen, J. Spray-Dried Nanofibrillar Cellulose Microparticles for Sustained Drug Release. Int. J. Pharm. 2012, 430, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Esatbeyoglu, T.; Huebbe, P.; Ernst, I.M.A.; Chin, D.; Wagner, A.E.; Rimbach, G. Curcumin—From Molecule to Biological Function. Angew. Chem. Int. Ed. 2012, 51, 5308–5332. [Google Scholar] [CrossRef] [PubMed]
- Jastrząb, A.; Jarocka-Karpowicz, I.; Skrzydlewska, E. The Origin and Biomedical Relevance of Cannabigerol. Int. J. Mol. Sci. 2022, 23, 7929. [Google Scholar] [CrossRef]
- Thaipong, K.; Boonprakob, U.; Crosby, K.; Cisneros-Zevallos, L.; Hawkins Byrne, D. Comparison of ABTS, DPPH, FRAP, and ORAC Assays for Estimating Antioxidant Activity from Guava Fruit Extracts. J. Food Compos. Anal. 2006, 19, 669–675. [Google Scholar] [CrossRef]
- Choudhury, A.K.; Raja, S.; Mahapatra, S.; Nagabhushanam, K.; Majeed, M. Synthesis and Evaluation of the Anti-Oxidant Capacity of Curcumin Glucuronides, the Major Curcumin Metabolites. Antioxidants 2015, 4, 750–767. [Google Scholar] [CrossRef]
- Nikolic, I.; Mitsou, E.; Damjanovic, A.; Papadimitriou, V.; Antic-Stankovic, J.; Stanojevic, B.; Xenakis, A.; Savic, S. Curcumin-Loaded Low-Energy Nanoemulsions: Linking EPR Spectroscopy-Analysed Microstructure and Antioxidant Potential with in Vitro Evaluated Biological Activity. J. Mol. Liq. 2020, 301, 112479. [Google Scholar] [CrossRef]
- Schaich, K.M.; Tian, X.; Xie, J. Hurdles and Pitfalls in Measuring Antioxidant Efficacy: A Critical Evaluation of ABTS, DPPH, and ORAC Assays. J. Funct. Foods 2015, 14, 111–125. [Google Scholar] [CrossRef]
- Priyadarsini, K.I. The Chemistry of Curcumin: From Extraction to Therapeutic Agent. Molecules 2014, 19, 20091–20112. [Google Scholar] [CrossRef] [PubMed]
- Jovanovic, S.V.; Boone, C.W.; Steenken, S.; Trinoga, M.; Kaskey, R.B. How Curcumin Works Preferentially with Water Soluble Antioxidants. J. Am. Chem. Soc. 2001, 123, 3064–3068. [Google Scholar] [CrossRef] [PubMed]
- Schrader, C.; Schiborr, C.; Frank, J.; Rimbach, G. Curcumin Induces Paraoxonase 1 in Cultured Hepatocytes in Vitro but Not in Mouse Liver in Vivo. Br. J. Nutr. 2011, 105, 167–170. [Google Scholar] [CrossRef] [PubMed]
- Balogun, E.; Hoque, M.; Gong, P.; Killeen, E.; Green, C.J.; Foresti, R.; Alam, J.; Motterlini, R. Curcumin Activates the Haem Oxygenase-1 Gene via Regulation of Nrf2 and the Antioxidant-Responsive Element. Biochem. J. 2003, 371, 887–895. [Google Scholar] [CrossRef] [PubMed]
- Sampath, M.; Pichaimani, A.; Kumpati, P.; Sengottuvelan, B. The Remarkable Role of Emulsifier and Chitosan, Dextran and PEG as Capping Agents in the Enhanced Delivery of Curcumin by Nanoparticles in Breast Cancer Cells. Int. J. Biol. Macromol. 2020, 162, 748–761. [Google Scholar] [CrossRef]
- Meng, Q.; Cooney, M.; Yepuri, N.; Cooney, R.N. L-Arginine Attenuates Interleukin-1β (IL-1β) Induced Nuclear Factor Kappa-Beta (NF-ΚB) Activation in Caco-2 Cells. PLoS ONE 2017, 12, e0174441. [Google Scholar] [CrossRef]
- Costa, E.M.; Silva, S.; Pereira, C.F.; Ribeiro, A.B.; Casanova, F.; Freixo, R.; Pintado, M.; Ramos, Ó.L. Carboxymethyl Cellulose as a Food Emulsifier: Are Its Days Numbered? Polymers 2023, 15, 2408. [Google Scholar] [CrossRef]
- Reimund, J.; Wittersheim, C.; Dumont, S.; Muller, C.D.; Kenney, J.S.; Baumann, R.; Poindron, P.; Reimund Dumont P Poindron, J.S.; Muller J S Kenney Antibody Solutions, C.D.; Moon Bay, H.; et al. Increased Production of Tumour Necrosis Factor-Alpha Interleukin-1 Beta, and Interleukin-6 by Morphologically Normal Intestinal Biopsies from Patients with Crohn’s Disease. Gut 1996, 39, 684. [Google Scholar] [CrossRef]
- Zhong, F.; Chen, H.; Han, L.; Jin, Y.; Wang, W. Curcumin Attenuates Lipopolysaccharide-Induced Renal Inflammation. Biol. Pharm. Bull. 2011, 34, 226–232. [Google Scholar] [CrossRef]
- Mackenzie, G.G.; Queisser, N.; Wolfson, M.L.; Fraga, C.G.; Adamo, A.M.; Oteiza, P.I. Curcumin Induces Cell-Arrest and Apoptosis in Association with the Inhibition of Constitutively Active NF-ΚB and STAT3 Pathways in Hodgkin’s Lymphoma Cells. Int. J. Cancer 2008, 123, 56–65. [Google Scholar] [CrossRef]
- Shishodia, S.; Amin, H.M.; Lai, R.; Aggarwal, B.B. Curcumin (Diferuloylmethane) Inhibits Constitutive NF-ΚB Activation, Induces G1/S Arrest, Suppresses Proliferation, and Induces Apoptosis in Mantle Cell Lymphoma. Biochem. Pharmacol. 2005, 70, 700–713. [Google Scholar] [CrossRef] [PubMed]
- Bharti, A.C.; Donato, N.; Singh, S.; Aggarwal, B.B. Curcumin (Diferuloylmethane) down-Regulates the Constitutive Activation of Nuclear Factor–ΚB and IκBα Kinase in Human Multiple Myeloma Cells, Leading to Suppression of Proliferation and Induction of Apoptosis. Blood 2003, 101, 1053–1062. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Aggarwal, B.B. Activation of Transcription Factor NF-ΚB Is Suppressed by Curcumin (Diferuloylmethane). J. Biol. Chem. 1995, 270, 24995–25000. [Google Scholar] [CrossRef] [PubMed]
- Ameruoso, A.; Palomba, R.; Palange, A.L.; Cervadoro, A.; Lee, A.; Di Mascolo, D.; Decuzzi, P. Ameliorating Amyloid-β Fibrils Triggered Inflammation via Curcumin-Loaded Polymeric Nanoconstructs. Front. Immunol. 2017, 8, 31. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Yadav, A.; Cwiklinski, K.; Quaye, E.; Aalinkeel, R.; Mahajan, S.D.; Schwartz, S.A.; Sharma, R.K. In-Vitro Studies of Curcumin Encapsulated Mesoporous Fe-Phenanthroline Nanocluster for Reduction of Amyloid β Plaque. J. Drug Deliv. Sci. Technol. 2019, 54, 101314. [Google Scholar] [CrossRef]
- Pontes-Quero, G.M.; Benito-Garzón, L.; Pérez Cano, J.; Aguilar, M.R.; Vázquez-Lasa, B. Amphiphilic Polymeric Nanoparticles Encapsulating Curcumin: Antioxidant, Anti-Inflammatory and Biocompatibility Studies. Mater. Sci. Eng. C 2021, 121, 111793. [Google Scholar] [CrossRef] [PubMed]
- Zorofchian Moghadamtousi, S.; Abdul Kadir, H.; Hassandarvish, P.; Tajik, H.; Abubakar, S.; Zandi, K. A Review on Antibacterial, Antiviral, and Antifungal Activity of Curcumin. BioMed Res. Int. 2014, 2014, 186864. [Google Scholar] [CrossRef]
- Hussain, Y.; Alam, W.; Ullah, H.; Dacrema, M.; Daglia, M.; Khan, H.; Arciola, C.R. Antimicrobial Potential of Curcumin: Therapeutic Potential and Challenges to Clinical Applications. Antibiotics 2022, 11, 322. [Google Scholar] [CrossRef]
- Gunes, H.; Gulen, D.; Mutlu, R.; Gumus, A.; Tas, T.; Topkaya, A.E. Antibacterial Effects of Curcumin: An in Vitro Minimum Inhibitory Concentration Study. Toxicol. Ind. Health 2016, 32, 246–250. [Google Scholar] [CrossRef]
- Keyvan, E.; Tutun, H.; Kahraman, H.A.; Şen, E.; Demirtaş, A.; Dönmez, S.; Akyüz, A.Ö. Determination of Time Dependent Antibacterial Activities of Curcumin, Carvacrol and Styrax Liquidus on Salmonella Enteritidis. Ank. Üniv. Vet. Fak. Derg. 2022, 69, 355–360. [Google Scholar] [CrossRef]
- Sandikci Altunatmaz, S.; Yilmaz Aksu, F.; Issa, G.; Basaran Kahraman, B.; Dulger Altiner, D.; Buyukunal, S.K. Antimicrobial Effects of Curcumin against L. Monocytogenes, S. Aureus, S. Typhimurium and E. coli O157:H7 Pathogens in Minced Meat. Vet. Med. 2016, 61, 256–262. [Google Scholar] [CrossRef]
- Bespalova, Y.; Kwon, D.; Vasanthan, N. Surface Modification and Antimicrobial Properties of Cellulose Nanocrystals. J. Appl. Polym. Sci. 2017, 134, 44789. [Google Scholar] [CrossRef]
- Bucci, A.R.; Marcelino, L.; Mendes, R.K.; Etchegaray, A. The Antimicrobial and Antiadhesion Activities of Micellar Solutions of Surfactin, CTAB and CPCl with Terpinen-4-Ol: Applications to Control Oral Pathogens. World J. Microbiol. Biotechnol. 2018, 34, 86. [Google Scholar] [CrossRef] [PubMed]
- No, D.S.; Algburi, A.; Huynh, P.; Moret, A.; Ringard, M.; Comito, N.; Drider, D.; Takhistov, P.; Chikindas, M.L. Antimicrobial Efficacy of Curcumin Nanoparticles against Listeria Monocytogenes Is Mediated by Surface Charge. J. Food Saf. 2017, 37, e12353. [Google Scholar] [CrossRef]
- Kalischuk, L.D.; Buret, A.G. A Role for Campylobacter Jejuni-Induced Enteritis in Inflammatory Bowel Disease? Am. J. Physiol.—Gastrointest. Liver Physiol. 2010, 298, G1–G9. [Google Scholar] [CrossRef] [PubMed]
- Brodkorb, A.; Egger, L.; Alminger, M.; Alvito, P.; Assunção, R.; Ballance, S.; Bohn, T.; Bourlieu-Lacanal, C.; Boutrou, R.; Carrière, F.; et al. INFOGEST Static in Vitro Simulation of Gastrointestinal Food Digestion. Nat. Protoc. 2019, 14, 991–1014. [Google Scholar] [CrossRef] [PubMed]
- Minekus, M.; Alminger, M.; Alvito, P.; Ballance, S.; Bohn, T.; Bourlieu, C.; Carrière, F.; Boutrou, R.; Corredig, M.; Dupont, D.; et al. A Standardised Static in Vitro Digestion Method Suitable for Food-an International Consensus. Food Funct. 2014, 5, 1113–1124. [Google Scholar] [CrossRef] [PubMed]
- Iurciuc-Tincu, C.E.; Atanase, L.I.; Ochiuz, L.; Jérôme, C.; Sol, V.; Martin, P.; Popa, M. Curcumin-Loaded Polysaccharides-Based Complex Particles Obtained by Polyelectrolyte Complexation and Ionic Gelation. I-Particles Obtaining and Characterization. Int. J. Biol. Macromol. 2020, 147, 629–642. [Google Scholar] [CrossRef]
- Löbmann, K.; Svagan, A.J. Cellulose Nanofibers as Excipient for the Delivery of Poorly Soluble Drugs. Int. J. Pharm. 2017, 533, 285–297. [Google Scholar] [CrossRef]
- Wahlang, B.; Pawar, Y.B.; Bansal, A.K. Identification of Permeability-Related Hurdles in Oral Delivery of Curcumin Using the Caco-2 Cell Model. Eur. J. Pharm. Biopharm. 2011, 77, 275–282. [Google Scholar] [CrossRef]
- Robaina Cabrera, C.L.; Keir-Rudman, S.; Horniman, N.; Clarkson, N.; Page, C. The Anti-Inflammatory Effects of Cannabidiol and Cannabigerol Alone, and in Combination. Pulm. Pharmacol. Ther. 2021, 69, 102047. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Z.; Shen, Z.L.; Zhai, S.; Xu, J.L.; Liang, H.; Shen, Q.; Li, Q.Y. Transport of Curcumin Derivatives in Caco-2 Cell Monolayers. Eur. J. Pharm. Biopharm. 2017, 117, 123–131. [Google Scholar] [CrossRef] [PubMed]
- Rashwan, A.K.; Karim, N.; Xu, Y.; Hanafy, N.A.N.; Li, B.; Mehanni, A.H.E.; Taha, E.M.; Chen, W. An Updated and Comprehensive Review on the Potential Health Effects of Curcumin-Encapsulated Micro/Nanoparticles. Crit. Rev. Food Sci. Nutr. 2022, 63, 9731–9751. [Google Scholar] [CrossRef] [PubMed]
- Pan, F.; Han, L.; Zhang, Y.; Yu, Y.; Liu, J. Optimization of Caco-2 and HT29 Co-Culture in Vitro Cell Models for Permeability Studies. Int. J. Food Sci. Nutr. 2015, 66, 680–685. [Google Scholar] [CrossRef] [PubMed]
- Guri, A.; Gülseren, I.; Corredig, M. Utilization of Solid Lipid Nanoparticles for Enhanced Delivery of Curcumin in Cocultures of HT29-MTX and Caco-2 Cells. Food Funct. 2013, 4, 1410–1419. [Google Scholar] [CrossRef] [PubMed]
- Madureira, A.R.; Campos, D.A.; Oliveira, A.; Sarmento, B.; Pintado, M.M.; Gomes, A.M. Insights into the Protective Role of Solid Lipid Nanoparticles on Rosmarinic Acid Bioactivity during Exposure to Simulated Gastrointestinal Conditions. Colloids Surf. B Biointerfaces 2016, 139, 277–284. [Google Scholar] [CrossRef] [PubMed]
- Khanvilkar, K.; Donovan, M.D.; Flanagan, D.R. Drug Transfer through Mucus. Adv. Drug Deliv. Rev. 2001, 48, 173–193. [Google Scholar] [CrossRef]
- Samad, T.; Witten, J.; Grodzinsky, A.J.; Ribbeck, K. Spatial Configuration of Charge and Hydrophobicity Tune Particle Transport through Mucus. Biophys. J. 2022, 121, 277–287. [Google Scholar] [CrossRef]
- Akbari, A.; Lavasanifar, A.; Wu, J. Interaction of Cruciferin-Based Nanoparticles with Caco-2 Cells and Caco-2/HT29-MTX Co-Cultures. Acta Biomater. 2017, 64, 249–258. [Google Scholar] [CrossRef]
- Coscueta, E.R.; Campos, D.A.; Osório, H.; Nerli, B.B.; Pintado, M. Enzymatic Soy Protein Hydrolysis: A Tool for Biofunctional Food Ingredient Production. Food Chem. X 2019, 1, 100006. [Google Scholar] [CrossRef]
- Lea, T. Caco-2 Cell Line; Springer: Berlin/Heidelberg, Germany, 2015; ISBN 9783319161044. [Google Scholar]
- Antunes, F.; Andrade, F.; Araújo, F.; Ferreira, D.; Sarmento, B. Establishment of a Triple Co-Culture in Vitro Cell Models to Study Intestinal Absorption of Peptide Drugs. Eur. J. Pharm. Biopharm. 2013, 83, 427–435. [Google Scholar] [CrossRef] [PubMed]
- Batista, P.; Castro, P.M.; Madureira, A.R.; Sarmento, B.; Pintado, M. Preparation, Characterization and Evaluation of Guar Films Impregnated with Relaxing Peptide Loaded into Chitosan Microparticles. Appl. Sci. 2021, 11, 9849. [Google Scholar] [CrossRef]





| Delivery System | Yield (%) | EE (%) | LC (%) | Zeta Potential (mV) | Particle Size | |
|---|---|---|---|---|---|---|
| Dv 50 (µm) | D 4:3 (µm) | |||||
| CNC-CTAB_Curcumin | 81.97 | 82.61 ± 0.45 | 28.63 ± 0.31 | −17.73 ± 0.30 | 7.33 | 9.34 |
| Sample | E. coli | S. enteritidis | L. innocua | C. jejuni |
|---|---|---|---|---|
| CNC-CTAB_Cur | − | − | − | + |
| Curcumin | − | − | − | − |
| CNC-CTAB | − | − | − | + |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Casanova, F.; Pereira, C.F.; Ribeiro, A.B.; Castro, P.M.; Freixo, R.; Martins, E.; Tavares-Valente, D.; Fernandes, J.C.; Pintado, M.E.; Ramos, Ó.L. Biological Potential and Bioaccessibility of Encapsulated Curcumin into Cetyltrimethylammonium Bromide Modified Cellulose Nanocrystals. Pharmaceuticals 2023, 16, 1737. https://doi.org/10.3390/ph16121737
Casanova F, Pereira CF, Ribeiro AB, Castro PM, Freixo R, Martins E, Tavares-Valente D, Fernandes JC, Pintado ME, Ramos ÓL. Biological Potential and Bioaccessibility of Encapsulated Curcumin into Cetyltrimethylammonium Bromide Modified Cellulose Nanocrystals. Pharmaceuticals. 2023; 16(12):1737. https://doi.org/10.3390/ph16121737
Chicago/Turabian StyleCasanova, Francisca, Carla F. Pereira, Alessandra B. Ribeiro, Pedro M. Castro, Ricardo Freixo, Eva Martins, Diana Tavares-Valente, João C. Fernandes, Manuela E. Pintado, and Óscar L. Ramos. 2023. "Biological Potential and Bioaccessibility of Encapsulated Curcumin into Cetyltrimethylammonium Bromide Modified Cellulose Nanocrystals" Pharmaceuticals 16, no. 12: 1737. https://doi.org/10.3390/ph16121737
APA StyleCasanova, F., Pereira, C. F., Ribeiro, A. B., Castro, P. M., Freixo, R., Martins, E., Tavares-Valente, D., Fernandes, J. C., Pintado, M. E., & Ramos, Ó. L. (2023). Biological Potential and Bioaccessibility of Encapsulated Curcumin into Cetyltrimethylammonium Bromide Modified Cellulose Nanocrystals. Pharmaceuticals, 16(12), 1737. https://doi.org/10.3390/ph16121737










