Effect of Anti-Diabetic Medications on Dental Implants: A Scoping Review of Animal Studies and Their Relevance to Humans
Abstract
1. Introduction
2. Results
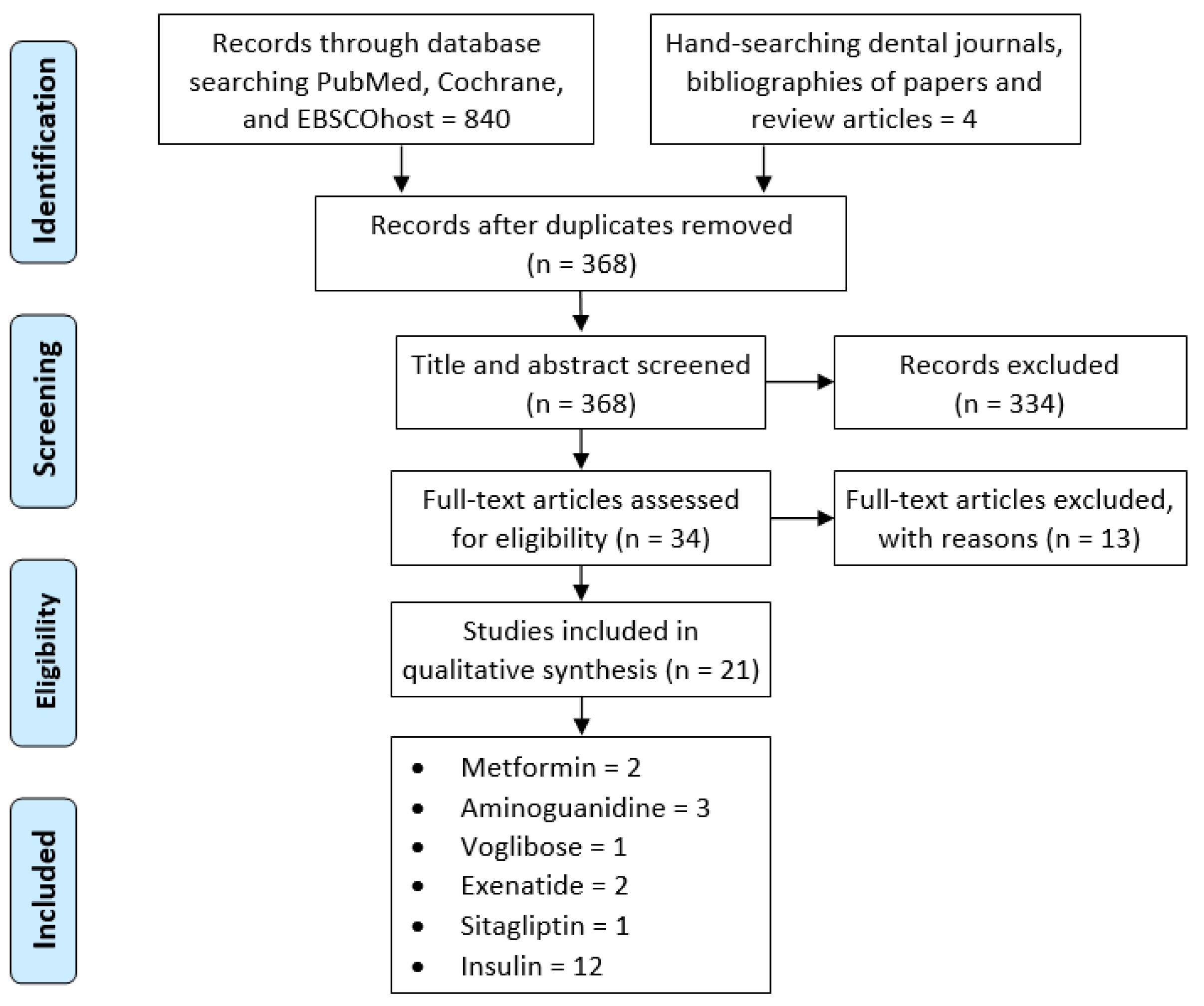
2.1. Study Selection
2.2. Risk of Bias (RoB) Assessment
2.3. Characteristics of the Included Studies
2.4. Effects of Anti-Diabetic Drugs on Implant Outcomes in T1DM Animal Models
2.4.1. Aminoguanidine
2.4.2. Sitagliptin
2.4.3. Insulin
2.5. Effects of Anti-Diabetic Drugs on Implant Outcomes in T2DM Animal Models
2.5.1. Biguanide Drug—Metformin
2.5.2. Alpha-Glucosidase Inhibitor—Voglibose
2.5.3. Exenatide
2.5.4. Insulin
3. Discussion
4. Materials and Methods
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Javed, F.; Romanos, G.E. Chronic Hyperglycemia as a Risk Factor in Implant Therapy. Periodontol. 2000 2019, 81, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Endocrinology, T.L.D. Diabetes: Mapping the Titanic Struggle Ahead. Lancet Diabetes Endocrinol. 2018, 6, 1. [Google Scholar] [CrossRef] [PubMed]
- Bahari, N.I.; Ahmad, N.; Mahmud, M.H.; Baharom, M.; Amir, S.M.; Peng, C.S.; Hassan, M.R.; Nawi, A.M. Issues and Challenges in the Primary Prevention of Type 2 Diabetes Mellitus: A Systematic Review. J. Prev. 2022, 1–21. [Google Scholar] [CrossRef]
- King, S.; Klineberg, I.; Levinger, I.; Brennan-Speranza, T.C. The Effect of Hyperglycaemia on Osseointegration: A Review of Animal Models of Diabetes Mellitus and Titanium Implant Placement. Arch. Osteoporos. 2016, 11, 29. [Google Scholar] [CrossRef]
- Pearce, A.I.; Richards, R.G.; Milz, S.; Schneider, E.; Pearce, S.G. Animal Models for Implant Biomaterial Research in Bone: A Review. Eur. Cells Mater. 2007, 13, 1–10. [Google Scholar] [CrossRef]
- Ko, K.I.; Sculean, A.; Graves, D.T. Diabetic wound healing in soft and hard oral tissues. Transl. Res. 2021, 236, 72–86. [Google Scholar] [CrossRef]
- Moraschini, V.; Barboza, E.S.P.; Peixoto, G.A. The Impact of Diabetes on Dental Implant Failure: A Systematic Review and Meta-Analysis. Int. J. Oral Maxillofac. Surg. 2016, 45, 1237–1245. [Google Scholar] [CrossRef]
- Tan, S.J.; Baharin, B.; Nabil, S.; Mohd, N.; Zhu, Y. Does Glycemic Control Have A Dose-Response Relationship With Implant Outcomes? A Comprehensive Systematic Review And Meta-Analysis. J. Evid. Based Dent. Pract. 2021, 21, 101543. [Google Scholar] [CrossRef]
- Blonde, L.; Umpierrez, G.E.; Reddy, S.S.; McGill, J.B.; Berga, S.L.; Bush, M.; Chandrasekaran, S.; DeFronzo, R.A.; Einhorn, D.; Galindo, R.J.; et al. American Association of Clinical Endocrinology Clinical Practice Guideline: Developing a Diabetes Mellitus Comprehensive Care Plan—2022 Update. Endocr. Pract. 2022, 28, 923–1049. [Google Scholar]
- Jiating, L.; Buyun, J.; Yinchang, Z. Role of Metformin on Osteoblast Differentiation in Type 2 Diabetes. BioMed Res. Int. 2019, 2019, 1–6. [Google Scholar] [CrossRef]
- Chen, X.; Lu, Y.; Fan, Y.; Shen, Y. Chapter 5—Voglibose: An Important Drug for Type 2 Diabetes, Validamycin and Its Derivatives; Elsevier: Amsterdam, The Netherlands, 2017; pp. 237–278. [Google Scholar]
- Patel, V.; Sadiq, M.S.; Najeeb, S.; Khurshid, Z.; Zafar, M.S.; Heboyan, A. Effects of metformin on the bioactivity and osseointegration of dental implants: A systematic review. J. Taibah Univ. Med Sci. 2022, 18, 196–206. [Google Scholar] [CrossRef]
- Kotsovilis, S.; Karoussis, I.K.; Fourmousis, I. A comprehensive and critical review of dental implant placement in diabetic animals and patients. Clin. Oral Implant. Res. 2006, 17, 587–599. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Lu, Q.; Yu, D.S.; Chen, Y.P.; Shang, J.; Zhang, L.Y.; Sun, H.B.; Liu, J. Asiatic Acid Mitigates Hyperglycemia and Reduces Islet Fibrosis in Goto-Kakizaki Rat, a Spontaneous Type 2 Diabetic Animal Model. Chin. J. Nat. Med. 2015, 13, 529–534. [Google Scholar] [CrossRef] [PubMed]
- Rybaczek, T.; Tangl, S.; Dobsak, T.; Gruber, R.; Kuchler, U. The Effect of Parathyroid Hormone on Osseointegration in Insulin-Treated Diabetic Rats. Implant Dent. 2015, 24, 392–396. [Google Scholar] [CrossRef]
- Wang, F.; Song, Y.; Li, C.; Li, D.; Zhang, H.; Ma, A.; Xi, X.; Zhang, N.; Wang, B.; Wang, Y.; et al. Sustained Release of Insulin-like Growth Factor-1 from Poly(Lactide-Co-Glycolide) Microspheres Improves Osseointegration of Dental Implants in Type 2 Diabetic Rats. Eur. J. Pharmacol. 2010, 640, 226–232. [Google Scholar] [CrossRef]
- Cirano, F.R.; Pimentel, S.P.; Casati, M.Z.; Correa, M.G.; Pino, D.S.; Messora, M.R.; Silva, P.H.F.; Ribeiro, F.V. Effect of Curcumin on Bone Tissue in the Diabetic Rat: Repair of Peri-Implant and Critical-Sized Defects. Int. J. Oral Maxillofac. Surg. 2018, 47, 1495–1503. [Google Scholar] [CrossRef]
- Corrêa, M.G.; Ribeiro, F.V.; Pimentel, S.P.; Benatti, B.B.; Felix Silva, P.H.; Casati, M.Z.; Cirano, F.R. Impact of Resveratrol in the Reduction of the Harmful Effect of Diabetes on Peri-Implant Bone Repair: Bone-Related Gene Expression, Counter-Torque and Micro-CT Analysis in Rats. Acta Odontol. Scand. 2020, 1–8. [Google Scholar] [CrossRef]
- Lu, L.; Zhijian, H.; Lei, L.; Wenchuan, C.; Zhimin, Z. Berberine in Combination with Insulin Has Additive Effects on Titanium Implants Osseointegration in Diabetes Mellitus Rats. Evid. Based. Complement. Alternat. Med. 2015, 2015, 824259. [Google Scholar] [CrossRef]
- Takeshita, F.; Murai, K.; Iyama, S.; Ayukawa, Y.; Suetsugu, T. Uncontrolled Diabetes Hinders Bone Formation Around Titanium Implants in Rat Tibiae. A Light and Fluorescence Microscopy, and Image Processing Study. J. Periodontol. 1998, 69, 314–320. [Google Scholar] [CrossRef]
- Zhou, W.; Tangl, S.; Reich, K.M.; Kirchweger, F.; Liu, Z.; Zechner, W.; Ulm, C.; Rausch-Fan, X. The Influence of Type 2 Diabetes Mellitus on the Osseointegration of Titanium Implants with Different Surface Modifications-A Histomorphometric Study in High-Fat Diet/Low-Dose Streptozotocin-Treated Rats. Implant Dent. 2019, 28, 11–19. [Google Scholar] [CrossRef] [PubMed]
- Saito, N.; Mikami, R.; Mizutani, K.; Takeda, K.; Kominato, H.; Kido, D.; Ikeda, Y.; Buranasin, P.; Nakagawa, K.; Takemura, S.; et al. Impaired Dental Implant Osseointegration in Rat with Streptozotocin-Induced Diabetes. J. Periodontal Res. 2022, 57, 412–424. [Google Scholar] [CrossRef] [PubMed]
- Sun, R.; Liang, C.; Sun, Y.; Xu, Y.; Geng, W.; Li, J. Effects of Metformin on the Osteogenesis of Alveolar BMSCs from Diabetic Patients and Implant Osseointegration in Rats. Oral Dis. 2022, 28, 1170–1180. [Google Scholar] [CrossRef] [PubMed]
- Goodman, W.G.; Hori, M.T. Diminished Bone Formation in Experimental Diabetes. Relationship to Osteoid Maturation and Mineralization. Diabetes 1984, 33, 825–831. [Google Scholar] [CrossRef] [PubMed]
- Stefan Tangl, W.Z.; Xilong Yue, F.C. Effects of Delayed Release Microsphere of Exenatide on Peri-Implant Bone Response of Diabetic Rats. Dentistry 2015, 5, 1–4. [Google Scholar] [CrossRef]
- Kopman, J.A.; Kim, D.M.; Rahman, S.S.; Arandia, J.A.; Karimbux, N.Y.; Fiorellini, J.P. Modulating the Effects of Diabetes on Osseointegration with Aminoguanidine and Doxycycline. J. Periodontol. 2005, 76, 614–620. [Google Scholar] [CrossRef]
- de Molon, R.S.; Morais-Camilo, J.A.N.D.; Verzola, M.H.A.; Faeda, R.S.; Pepato, M.T.; Marcantonio, E.J. Impact of Diabetes Mellitus and Metabolic Control on Bone Healing around Osseointegrated Implants: Removal Torque and Histomorphometric Analysis in Rats. Clin. Oral Implants Res. 2013, 24, 831–837. [Google Scholar] [CrossRef]
- de Morais, J.A.N.D.; Trindade-Suedam, I.K.; Pepato, M.T.; Marcantonio, E.J.; Wenzel, A.; Scaf, G. Effect of Diabetes Mellitus and Insulin Therapy on Bone Density around Osseointegrated Dental Implants: A Digital Subtraction Radiography Study in Rats. Clin. Oral Implants Res. 2009, 20, 796–801. [Google Scholar] [CrossRef]
- Yamazaki, S.; Masaki, C.; Nodai, T.; Tsuka, S.; Tamura, A.; Mukaibo, T.; Kondo, Y.; Ono, K.; Hosokawa, R. The Effects of Hyperglycaemia on Peri-Implant Tissues after Osseointegration. J. Prosthodont. Res. 2020, 64, 217–223. [Google Scholar] [CrossRef]
- Kilkenny, C.; Browne, W.J.; Cuthill, I.C.; Emerson, M.; Altman, D.G. Improving Bioscience Research Reporting: The ARRIVE Guidelines for Reporting Animal Research. J. Pharmacol. Pharmacother. 2010, 1, 94–99. [Google Scholar] [CrossRef]
- Guimarães, R.P.; de Oliveira, P.A.D.; Oliveira, A.M.S.D. Effects of Induced Diabetes and the Administration of Aminoguanidine in the Biomechanical Retention of Implants: A Study in Rats. J. Periodontal Res. 2011, 46, 691–696. [Google Scholar] [CrossRef]
- Aiala, G.F.; Oliveira, A.M.S.D.; Costa, F.O.; Fialho, D.L.; Cunha, A.S.J.; Oliveira, P.A.D. Effect of Local Application of Aminoguanidine on the Biomechanical Retention of Implants in Rats with Induced Diabetes. Int. J. Oral Maxillofac. Implants 2013, 28, 1272–1277. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bautista, C.R.G.; Santos, I.V.D.; Moraes, R.M.; Chiba, F.Y.; Sumida, D.H.; de Moraes, M.B.; de Vasconcellos, L.M.R.; Anbinder, A.L. Sitagliptin’s Effects on Bone Tissue and Osseointegration in Diabetic Rats. Arch. Oral Biol. 2019, 102, 238–243. [Google Scholar] [CrossRef] [PubMed]
- Matsubara, F.; Shirota, T.; Yamazaki, M.; Motohashi, M.; Ohno, K.; Michi, K.I. The Effect of Insulin Therapy on Bone Tissue around Implants in Streptozotocin-Induced Diabetic Rats. Oral Ther. Pharmacol. 2001, 20, 173–180. [Google Scholar]
- Zhang, J.; Wang, Y.N.; Jia, T.; Huang, H.; Zhang, D.; Xu, X. Genipin and Insulin Combined Treatment Improves Implant Osseointegration in Type 2 Diabetic Rats. J. Orthop. Surg. Res. 2021, 16, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Kwon, P.T.; Rahman, S.S.; Kim, D.M.; Kopman, J.A.; Karimbux, N.Y.; Fiorellini, J.P. Maintenance of Osseointegration Utilizing Insulin Therapy in a Diabetic Rat Model. J. Periodontol. 2005, 76, 621–626. [Google Scholar] [CrossRef] [PubMed]
- Margonar, R.; Sakakura, C.E.; Holzhausen, M.; Pepato, M.T.; Alba; junior, R.C.; Marcantonio, E., Jr. The Influence of Diabetes Mellitus and Insulin Therapy on Biomechanical Retention around Dental Implants: A Study in Rabbits. Implant Dent. 2003, 12, 333–339. [Google Scholar] [CrossRef]
- Siqueira, J.T.; Cavalher-Machado, S.C.; Arana-Chavez, V.E.; Sannomiya, P. Bone Formation around Titanium Implants in the Rat Tibia: Role of Insulin. Implant Dent. 2003, 12, 242–251. [Google Scholar] [CrossRef]
- Fiorellini, J.P.; Nevins, M.L.; Norkin, A.; Weber, H.P.; Karimbux, N.Y. The Effect of Insulin Therapy on Osseointegration in a Diabetic Rat Model. Clin. Oral Implants Res. 1999, 10, 362–368. [Google Scholar] [CrossRef]
- McCracken, M.S.; Aponte-Wesson, R.; Chavali, R.; Lemons, J.E. Bone Associated with Implants in Diabetic and Insulin-Treated Rats. Clin. Oral Implants Res. 2006, 17, 495–500. [Google Scholar] [CrossRef]
- Han, Y.; Zeng, Q.E.L.; Wang, D.; He, H.; Liu, H. Sustained Topical Delivery of Insulin from Fibrin Gel Loaded with Poly(Lactic-Co-Glycolic Acid) Microspheres Improves the Biomechanical Retention of Titanium Implants in Type 1 Diabetic Rats. J. Oral Maxillofac. Surg. 2012, 70, 2299–2308. [Google Scholar] [CrossRef]
- Serrão, C.; Bastos, M.; Cruz, D.; Malta, F.; Vallim, P.; Duarte, P. Role of Metformin in Reversing the Negative Impact of Hyperglycemia on Bone Healing Around Implants Inserted in Type 2 Diabetic Rats. Int. J. Oral Maxillofac. Implants 2017, 32, 547–554. [Google Scholar] [CrossRef] [PubMed]
- Inouye, K.A.S.; Bisch, F.C.; Elsalanty, M.E.; Zakhary, I.; Khashaba, R.M.; Borke, J.L. Effect of Metformin on Periimplant Wound Healing in a Rat Model of Type 2 Diabetes. Implant Dent. 2014, 23, 319–327. [Google Scholar] [CrossRef] [PubMed]
- Hashiguchi, C.; Kawamoto, S.; Kasai, T.; Nishi, Y.; Nagaoka, E. Influence of an Antidiabetic Drug on Biomechanical and Histological Parameters around Implants in Type 2 Diabetic Rats. Implant Dent. 2014, 23, 264–269. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Zhou, W.; Tangl, S.; Liu, S.; Xu, X.; Rausch-Fan, X. Potential Mechanism for Osseointegration of Dental Implants in Zucker Diabetic Fatty Rats. Br. J. Oral Maxillofac. Surg. 2015, 53, 748–753. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.; Liu, Z.; Yao, J.; Chi, F.; Dong, K.; Yue, X.; Teng, T.; Rausch, X. The Effects of Exenatide Microsphere on Serum BGP and ALP Levels in ZDF Rats after Implantation. Clin. Implant Dent. Relat. Res. 2015, 17, 765–770. [Google Scholar] [CrossRef]
- Wang, B.; Song, Y.; Wang, F.; Li, D.; Zhang, H.; Ma, A.; Huang, N. Effects of Local Infiltration of Insulin around Titanium Implants in Diabetic Rats. Br. J. Oral Maxillofac. Surg. 2011, 49, 225–229. [Google Scholar] [CrossRef]
- Zhang, X.Z.; Shi, M.Z.; Dang, G.D. Effect of Insulin Therapy on Abnormal Bone and Mineral Metabolism in Chronic Streptozotocin-Induced Diabetic Rat. Zhonghua Nei Ke Za Zhi 1992, 31, 674–677, 729. [Google Scholar]
- Bolker, J.A. Animal Models in Translational Research: Rosetta Stone or Stumbling Block? In BioEssays; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2017; p. 1700089. [Google Scholar]
- Omar, N.I.; Baharin, B.; Lau, S.F.; Ibrahim, N.; Mohd, N.; Ahmad Fauzi, A.; Muhammad, N.; Fernandez, N.M. The Influence of Ficus Deltoidea in Preserving Alveolar Bone in Ovariectomized Rats. Vet. Med. Int. 2020, 2020, 8862489. [Google Scholar] [CrossRef]
- Blanc-Sylvestre, N.; Bouchard, P.; Chaussain, C.; Bardet, C. Pre-Clinical Models in Implant Dentistry: Past, Present, Future. Biomedicines 2021, 9, 1538. [Google Scholar] [CrossRef]
- Prabhakar, S. Translational Research Challenges: Finding the Right Animal Models. J. Investig. Med. 2012, 60, 1141–1146. [Google Scholar] [CrossRef]
- Mohd, N.; Razali, M.; Ghazali, M.J.; Kasim, N.H.A. 3D-Printed Hydroxyapatite and Tricalcium Phosphates-Based Scaffolds for Alveolar Bone Regeneration in Animal Models: A Scoping Review. Materials 2022, 15, 2621. [Google Scholar] [CrossRef] [PubMed]
- Mohd, N.; Razali, M.; Ghazali, M.J.; Kasim, N.H.A. Current Advances of Three-Dimensional Bioprinting Application in Dentistry: A Scoping Review. Materials 2022, 15, 6398. [Google Scholar] [CrossRef]
- Musskopf, M.L.; Stadler, A.F.; Wikesjö, U.M.E.; Susin, C. The Minipig Intraoral Dental Implant Model: A Systematic Review and Meta-Analysis. PLoS ONE 2022, 17, e0264475. [Google Scholar] [CrossRef] [PubMed]
- Coelho, P.G.; Pippenger, B.; Tovar, N.; Koopmans, S.-J.; Plana, N.M.; Graves, D.T.; Engebretson, S.; van Beusekom, H.M.M.; Oliveira, P.G.F.P.; Dard, M. Effect of Obesity or Metabolic Syndrome and Diabetes on Osseointegration of Dental Implants in a Miniature Swine Model: A Pilot Study. J. Oral Maxillofac. Surg. 2018, 76, 1677–1687. [Google Scholar] [CrossRef] [PubMed]
- Borg, D.J.; Forbes, J.M. Targeting Advanced Glycation with Pharmaceutical Agents: Where Are We Now? Glycoconj. J. 2016, 33, 653–670. [Google Scholar] [CrossRef]
- Bolton, W.K.; Cattran, D.C.; Williams, M.E.; Adler, S.G.; Appel, G.B.; Cartwright, K.; Foiles, P.G.; Freedman, B.I.; Raskin, P.; Ratner, R.E.; et al. Randomized Trial of an Inhibitor of Formation of Advanced Glycation End Products in Diabetic Nephropathy. Am. J. Nephrol. 2004, 24, 32–40. [Google Scholar] [CrossRef] [PubMed]
- Eskow, C.C.; Oates, T.W. Dental Implant Survival and Complication Rate over 2 Years for Individuals with Poorly Controlled Type 2 Diabetes Mellitus. Clin. Implant Dent. Relat. Res. 2017, 19, 423–431. [Google Scholar] [CrossRef]
- Tricco, A.C.; Lillie, E.; Zarin, W.; O’Brien, K.K.; Colquhoun, H.; Levac, D.; Moher, D.; Peters, M.D.J.; Horsley, T.; Weeks, L.; et al. PRISMA Extension for Scoping Reviews (PRISMA-ScR): Checklist and Explanation. Ann. Intern. Med. 2018, 169, 467–473. [Google Scholar] [CrossRef]
- Hooijmans, C.R.; Rovers, M.M.; De Vries, R.B.M.; Leenaars, M.; Ritskes-Hoitinga, M.; Langendam, M.W. SYRCLE’s Risk of Bias Tool for Animal Studies. BMC Med. Res. Methodol. 2014, 14, 1–9. [Google Scholar] [CrossRef]
| Author and Year | Signaling Questions * | Summary | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | ||
| Guimarães et al., 2011 [31] | + | ? | − | − | − | − | − | + | + | ? | ? |
| Aiala et al., 2013 [32] | + | + | + | − | − | − | − | + | + | − | ? |
| Kopman et al., 2005 [26] | + | + | ? | − | − | + | + | + | + | − | + |
| Bautista et al., 2019 [33] | − | + | − | − | − | − | + | + | + | − | − |
| Matsubara et al., 2001 [34] | − | + | − | − | − | − | − | + | + | + | ? |
| Zhang et al., 2021 [35] | + | + | ? | − | − | − | − | + | + | + | ? |
| de Molon et al., 2013 [27] | + | + | ? | − | − | + | + | + | + | + | + |
| de Morais et al., 2009 [28] | + | + | ? | − | − | + | + | + | + | + | + |
| Kwon et al., 2005 [36] | − | − | − | − | − | − | + | + | + | ? | ? |
| Margonar et al., 2003 [37] | + | + | ? | − | − | − | − | + | + | + | ? |
| Siqueira et al., 2003 [38] | − | + | − | − | − | − | − | + | + | − | − |
| Fiorellini et al., 1999 [39] | − | + | − | − | − | − | − | + | + | + | ? |
| McCracken et al., 2006 [40] | − | + | − | − | − | − | − | − | + | − | − |
| Yamazaki et al., 2020 [29] | + | + | + | − | − | − | + | + | + | + | + |
| Han et al., 2012 [41] | − | + | − | − | − | − | − | + | + | − | − |
| Serrão et al., 2017 [42] | + | + | ? | ? | + | − | ? | + | − | − | ? |
| Inouye et al., 2014 [43] | − | − | − | − | − | − | − | + | + | − | − |
| Hashiguchi et al., 2014 [44] | − | + | − | − | − | − | − | + | + | − | − |
| Liu et al., 2015 [45] | − | + | − | − | − | − | − | + | + | ? | ? |
| Zhou et al., 2015 [46] | − | + | − | − | − | − | − | + | + | ? | ? |
| Wang et al., 2011 [47] | − | + | − | − | − | − | − | + | + | ? | ? |
| Author and Year | Animal Model | Sex | Age | Number of Implants | Type of DM and Medication | Induction of DM | Timing of Diabetes Induction | Timing of Anti-Diabetic Medication |
|---|---|---|---|---|---|---|---|---|
| Guimarães et al., 2011 [31] | Wistar rats | Not specified | 20 weeks | 6 per group, total 36 | T1DM (aminoguanidine) | Intraperitoneal alloxan injection, 84 mg/kg | Before implant placement | Started the day of implant placement |
| Aiala et al., 2013 [32] | Wistar rats | Male | 28 weeks | 8 per group, total 48 | T1DM (aminoguanidine) | 70 mg/kg streptozocin | Before implant placement | Started the day of implant placement |
| Kopman et al., 2005 [26] | Sprague–Dawley rats | Male | Not specified | 8 per group, total 32 | T1DM (aminoguanidine) | 70 mg/kg streptozocin | Before implant placement | Started the day of implant placement |
| Bautista et al., 2019 [33] | Wistar rats | Male | 16 weeks | 8 per group, total 32 | T1DM (sitagliptin) | 40 mg/kg streptozocin | Before implant placement | Started the day after implant placement |
| Matsubara et al., 2001 [34] | Wistar rats | Male | 12 weeks | 12 per group, total 36 | T1DM (insulin) | 60 mg/kg streptozocin | Before implant placement | Before implant placement |
| Zhang et al., 2021 [35] | Sprague–Dawley rats | Male | 11 weeks | 6 per group, total 30 | T1DM (insulin) | 30 mg/kg streptozocin | Before implant placement | Before implant placement |
| de Molon et al., 2013 [27] | Wistar rats | Male | 16 weeks | 20 per group, total 80 | T1DM (insulin) | 40 mg/kg streptozocin | 2 months after implant placement | Day 2 of DM |
| de Morais et al., 2009 [28] | Wistar rats | Male | 16 weeks | 10 per group, total 40 | T1DM (insulin) | 40 mg/kg streptozocin | 2 months after implant placement | Day 2 of DM |
| Kwon et al., 2005 [36] | Sprague–Dawley rats | Male | 4 weeks | 4 per group, total 32 | T1DM (insulin) | 70 mg/kg streptozocin | 28 days after implant placement | 28 days after implant placement |
| Margonar et al., 2003 [37] | New Zealand rabbits | Female | 20 weeks | 9 per group, total 27 | T1DM (insulin) | Intraperitoneal alloxan injection 115 mg/kg | Before implant placement | Day 2 of DM |
| Siqueira et al., 2003 [38] | Wistar rats | Male | 12 weeks | Control 17, diabetic 18, insulin 8, total 43 | T1DM (insulin) | Intraperitoneal alloxan injection 42 mg/kg | Before implant placement | 10 days after implant placement |
| Fiorellini et al., 1999 [39] | Sprague–Dawley rats | Male | 5 weeks | 5 per group, total 10 | T1DM (insulin) | 70 mg/kg streptozocin | Before implant placement | Day 2 of DM |
| McCracken et al., 2006 [40] | Sprague–Dawley rats | Not specified | Not specified | 60 diabetic, 60 healthy, 32 insulin, total 152 | T1DM (insulin) | 60 mg/kg streptozocin | Before implant placement | Ongoing insulin treatment |
| Yamazaki et al., 2020 [29] | Wistar rats | Male | 5 weeks | 12 per group, total 36 | T1DM (insulin) | 50 mg/kg streptozocin | 5 weeks after implant placement | Day 3 of DM |
| Han et al., 2012 [41] | Wistar rats | Male | Not specified | 6 per group, total 48 | T1DM (insulin) | 80 mg/kg streptozocin | Before implant placement | Slow release insulin coated implant at placement |
| Serrão et al., 2017 [42] | Wistar rats | Male | 12 weeks | 10 per group, total 30 | T2DM (metformin) | 10% fructose and 40 mg/kg streptozocin | Implant placement in spontaneously diabetic animal | 2 weeks after implant placement |
| Inouye et al., 2014 [43] | Goto–Kakizaki (GK) rats | Male | 12 weeks | 12 per group, total 36 | T2DM (metformin) | Genetically modified, spontaneous T2DM | Implant placement in spontaneously diabetic animal | Ongoing metformin treatment |
| Hashiguchi et al., 2014 [44] | Goto–Kakizaki (GK) rats | Male | Not specified | 10 per group, total 20 | T2DM (voglibose) | Genetically modified, spontaneous T2DM | Implant placement in spontaneously diabetic animal | Ongoing voglibose treatment |
| Liu et al., 2015 [45] | Zucker diabetic fatty (ZDF) rats | Male | 12 weeks | 11 per group, total 33 rats, 66 implants | T2DM (exenatide) | Genetically modified, spontaneous T2DM | Implant placement in spontaneously diabetic animal | Ongoing subcutaneous injection |
| Zhou et al., 2015 [46] | Zucker diabetic fatty (ZDF) rats | Male | 12 weeks | 11 per group, total 33 rats, 66 implants | T2DM (exenatide) | Genetically modified, spontaneous T2DM | Implant placement in spontaneously diabetic animal | Ongoing subcutaneous injection |
| Wang et al., 2011 [47] | Goto–Kakizaki (GK) rats | Not specified | Not specified | 10 per group, total 20, also 10 control Sprague–Dawley rats | T2DM (insulin) | Genetically modified, spontaneous T2DM | Implant placement in spontaneously diabetic animal | Slow release insulin coated implant at placement |
| Author and Year | Drug Used and Method of Administration | Titanium Fixture Location | Follow-up Duration | Glucose Level of Diabetic Animals after Medication (mg/dL) | BIC (%) of Healthy Animals | Control BIC (%) (Untreated Diabetic) | Experimental BIC (%) (Treated Diabetic) | Other Outcomes of Interest |
|---|---|---|---|---|---|---|---|---|
| Guimarães et al., 2011 [31] | Aminoguanidine (intraperitoneal) | Implant in femur | 4 weeks | Value provided as variation from baseline, no actual value given | - | - | - | Counter-torque values (N/cm) Healthy rats: 2.24 ± 0.58 Diabetic + medication: 2.5 ± 0.6 Diabetic rats: 1.18 ± 0.15 |
| Aiala et al., 2013 [32] | Aminoguanidine (drug-loaded chitosan membrane) | Implant in femur | 4 weeks | 436.6 mg/dL (hyperglycemic) | - | - | - | Counter-torque values (N/cm) Healthy rats: 2.66 ± 0.91 Diabetic + medication: 1.8 ± 0.7 Diabetic rats: 1.26 ± 0.19 |
| Kopman et al., 2005 [26] | Aminoguanidine (intraperitoneal) | Implant in femur | 4 weeks | 525.4 ± 46.9 mg/dL (hyperglycemic) | 64.3 ± 3.7 | 22.2 ± 3.9 | 55.3 ± 6.1 | Aminoguanidine augments osseointegration in diabetic rats |
| Bautista et al., 2019 [33] | Sitagliptin (oral) | Implant in tibia | 4 weeks | 590.7 ± 30.2 mg/dL (hyperglycemic) | - | - | - | Bone area fraction occupancy significantly lower in diabetic rats |
| Matsubara et al., 2001 [34] | Insulin (systemic delivery) | Implant in tibia | 8 weeks | 156 mg/dL (normoglycemic) | 65.5 | 56.0 | 66.2 | Lower BIC and bone volume in untreated diabetic rats |
| Zhang et al., 2021 [35] | Insulin (systemic delivery) | Implant in femur | 12 weeks | Less than 180 mg/dL (normoglycemic) | 61 | 20 | Almost 50 | Higher pull-out test values in insulin-treated rats; however, insulin alone does not adequately reverse effects of DM |
| de Molon et al., 2013 [27] | Insulin (systemic delivery) | Implant in tibia | 16 weeks | 75 ± 11 (normoglycemic) | 63.37 ± 5.88 | 60.81 ± 6.83 | 66.97 ± 4.13 | Bone area (%): Diabetic rats: 69.34 ± 5.00 Insulin-treated rats: 79.63 ± 4.97 Counter torque (N/cm): Diabetic rats: 12.91 ± 2.51 Insulin-treated rats: 17.10 ± 3.06 |
| de Morais et al., 2009 [28] | Insulin (systemic delivery) | Implant in tibia | 16 weeks | 42 ± 8 (normoglycemic) | - | - | - | Digital subtraction radiography: Significantly lower peri-implant bone density and more bone loss in untreated diabetic rats |
| Kwon et al., 2005 [36] | Insulin (systemic delivery) | Implant in femur | 16 weeks | HbA1c at 4.2 ± 0.1 (normoglycemic) | - | 41.7 ± 2.3 | 61.4 ± 2.4 | Untreated diabetic rats show gradual decrease in BIC over 4 months |
| Margonar et al., 2003 [37] | Insulin (systemic delivery) | Implant in tibia | 12 weeks | 99.2 ± 72.2 (normoglycemic) | - | - | - | Counter removal torque (N/cm): Diabetic rabbits: 22.0 ± 6.0 Insulin-treated rabbits: 25.2 ± 4.4 |
| Siqueira et al., 2003 [38] | Insulin (systemic delivery) | Implant in tibia | 21 days | 295 ± 24 (hyperglycemic) | About 50 | Less BIC (about 20) | Significantly improved BIC (about 50) | Need to control blood glucose for successful osseointegration |
| Fiorellini et al., 1999 [39] | Insulin (systemic delivery) | Implant in femur | 4 weeks | 145 ± 64.4 (8 mmol/L—slightly hyperglycemic) | 50.4 ± 1.3 | 50.4 ± 1.3 (non-diabetic) | 52.4 ± 0.0 | Insulin administration for diabetic rats ameliorates bone formation to similar level with healthy controls |
| McCracken et al., 2006 [40] | Insulin (systemic delivery) | Implant in tibia | 24 days | Less than 200 mg/dL (normoglycemic) | - | - | - | Bone volume (%): Diabetic rats: 22.6 ± 10 (significant) Insulin-treated rats: 17 ± 7 |
| Yamazaki et al., 2020 [29] | Insulin (systemic delivery) | Implant in healed extraction site | 8 weeks | Less than 100 mg/dL (normoglycemic) | - | - | - | Bone loss measured in micro-CT: Diabetic rats: 0.42 ± 0.05 mm Insulin-treated rats: 0.39 ± 0.10 mm (not significant) |
| Han et al., 2012 [41] | Insulin (local delivery)—FGIPM | Implant in tibia | 8 weeks | 383.4 ± 56.5 (hyperglycemic) | - | - | - | Counter torque (N/cm): Normal rats: 4.82 ± 0.5419 Diabetic rats: 3.07 ± 0.3266 Insulin-treated rats: 4.67 ± 0.3386 (significant) |
| Serrão et al., 2017 [42] | Metformin (oral) | Implant in tibia | 4 weeks | 154 ± 27 (normoglycemic) | 70.17 ± 5.36 | 50.85 ± 6.26 | 48.58 ± 12.41 | Increased levels of OPG, decreased RANKL/OPG ratio in medullary bone |
| Inouye et al., 2014 [43] | Metformin (oral) | Implant in healed extraction site | 4 weeks | Fluctuates, 293.8 ± 28.0 mg/dL (hyperglycemic) | 49.18 + 5.57 | 23.36 ± 6.44 | 31.57 ± 8.29 | No significant difference in BIC at week 4 due to hyperglycemia at that point (fluctuation) |
| Hashiguchi et al., 2014 [44] | Voglibose (oral) | Implant in tibia | 9 weeks | About 250 mg/dL (from graph) (hyperglycemic) | 71.7 ± 11.2 | 55.640 ±16.578 | 40.999 ± 10.682 | No significant difference in BIC and counter torque values |
| Zhou et al., 2015 [46] | Exenatide (subcutaneous) | Implant in femur | 4 weeks, 8 weeks | Constant glucose concentration less than 288 mg/dL—unclear level of glycemia | - | - | - | At 4 weeks, incomplete peri-implant bone fill in diabetic controls, while woven bone and direct BIC surrounds implants with exenatide treatment |
| Liu et al., 2015 [45] | Exenatide (subcutaneous) | Implant in femur | 4 weeks, 8 weeks | Constant glucose concentration less than 288 mg/dL—unclear level of glycemia | - | - | - | Higher bone density, less inflammatory cells, stronger expression of integrin α5β1 and fibronectin in exenatide group versus untreated diabetic rats |
| Wang et al., 2011 [47] | Insulin (local delivery)—insulin-containing PLGA microcapsules | Implant in tibiae | 6 weeks | Not specified | 66 ± 3 | 51 ± 3 | 58 ± 3 | Significantly more BIC in insulin-treated group |
| PICO Elements | Framework Details |
|---|---|
| Animal model representing patient population | Diabetic animals (T1DM and T2DM) that received titanium implants |
| Intervention reflecting clinical practice | Diabetic animals that received anti-diabetic treatment using approved medications in human clinical practice guidelines |
| Comparator without treatment or intervention | Diabetic animals that did not receive treatment for diabetes |
| Outcome (biological effect or mechanism) | Implant osseointegration outcomes (bone-implant contact, bone volume, bone area, counter-torque values, amount of bone loss, bone biomarkers level) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tan, S.J.; Baharin, B.; Mohd, N.; Nabil, S. Effect of Anti-Diabetic Medications on Dental Implants: A Scoping Review of Animal Studies and Their Relevance to Humans. Pharmaceuticals 2022, 15, 1518. https://doi.org/10.3390/ph15121518
Tan SJ, Baharin B, Mohd N, Nabil S. Effect of Anti-Diabetic Medications on Dental Implants: A Scoping Review of Animal Studies and Their Relevance to Humans. Pharmaceuticals. 2022; 15(12):1518. https://doi.org/10.3390/ph15121518
Chicago/Turabian StyleTan, Sze Jun, Badiah Baharin, Nurulhuda Mohd, and Syed Nabil. 2022. "Effect of Anti-Diabetic Medications on Dental Implants: A Scoping Review of Animal Studies and Their Relevance to Humans" Pharmaceuticals 15, no. 12: 1518. https://doi.org/10.3390/ph15121518
APA StyleTan, S. J., Baharin, B., Mohd, N., & Nabil, S. (2022). Effect of Anti-Diabetic Medications on Dental Implants: A Scoping Review of Animal Studies and Their Relevance to Humans. Pharmaceuticals, 15(12), 1518. https://doi.org/10.3390/ph15121518






