Fluorinated Protein and Peptide Materials for Biomedical Applications
Abstract
1. Introduction
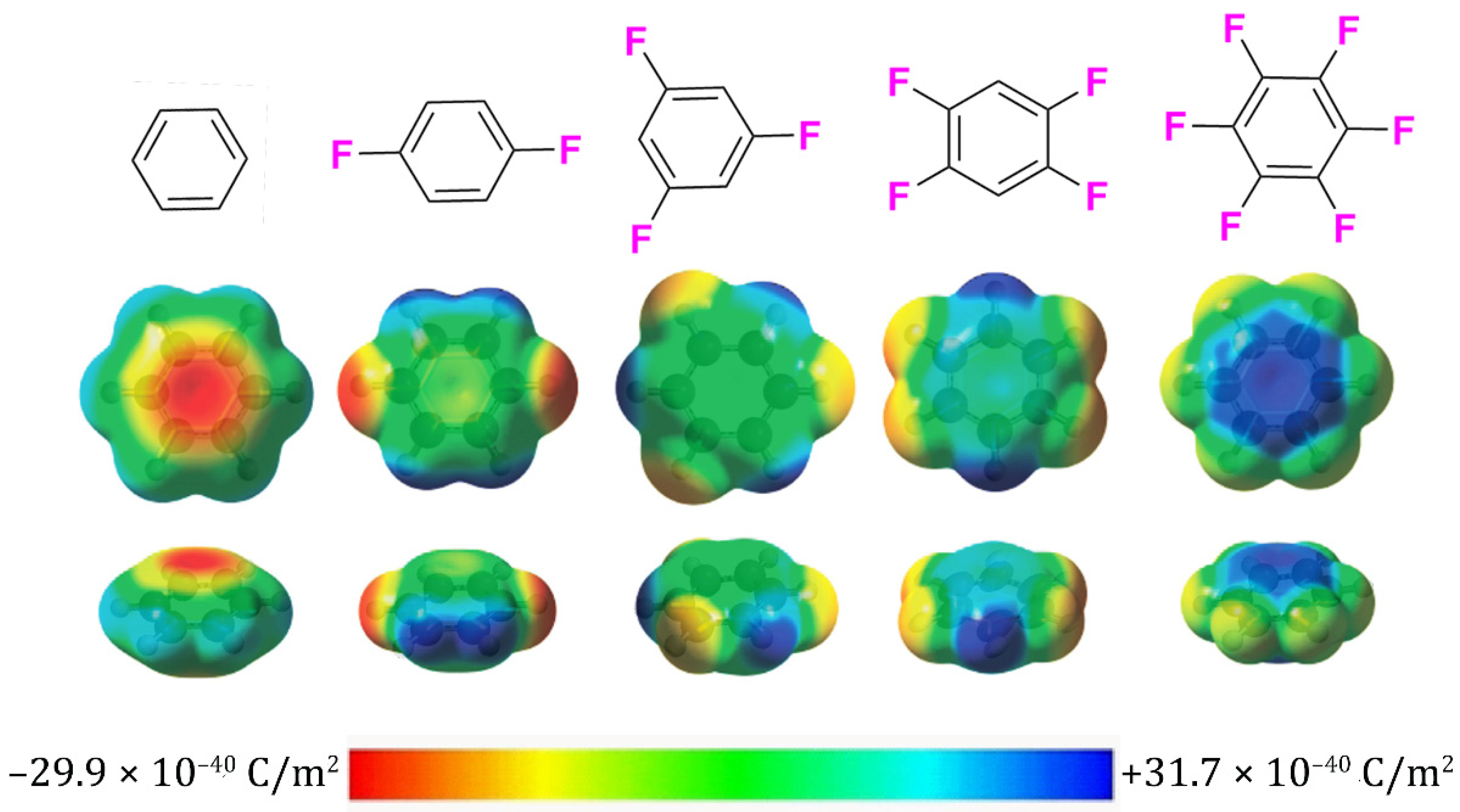
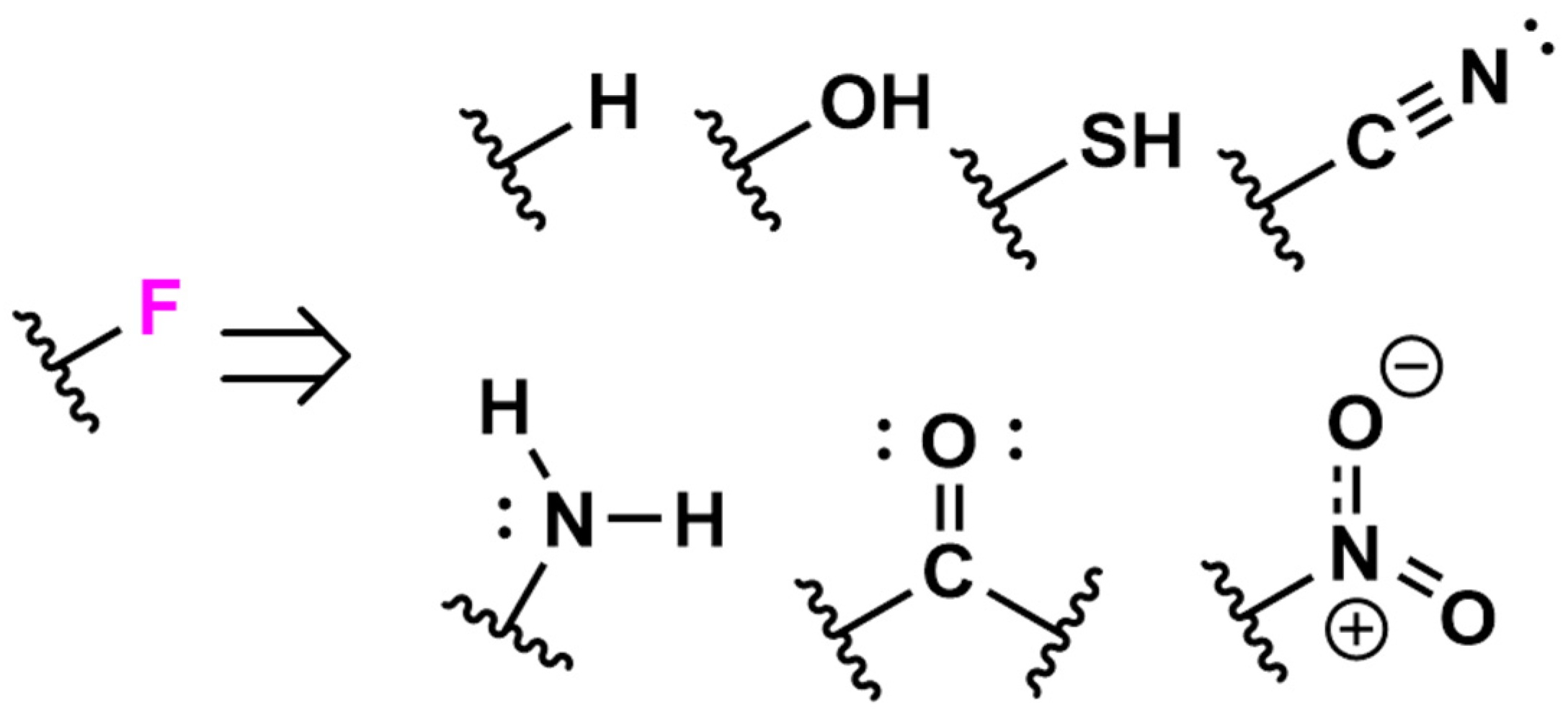
Special Properties of Fluorine
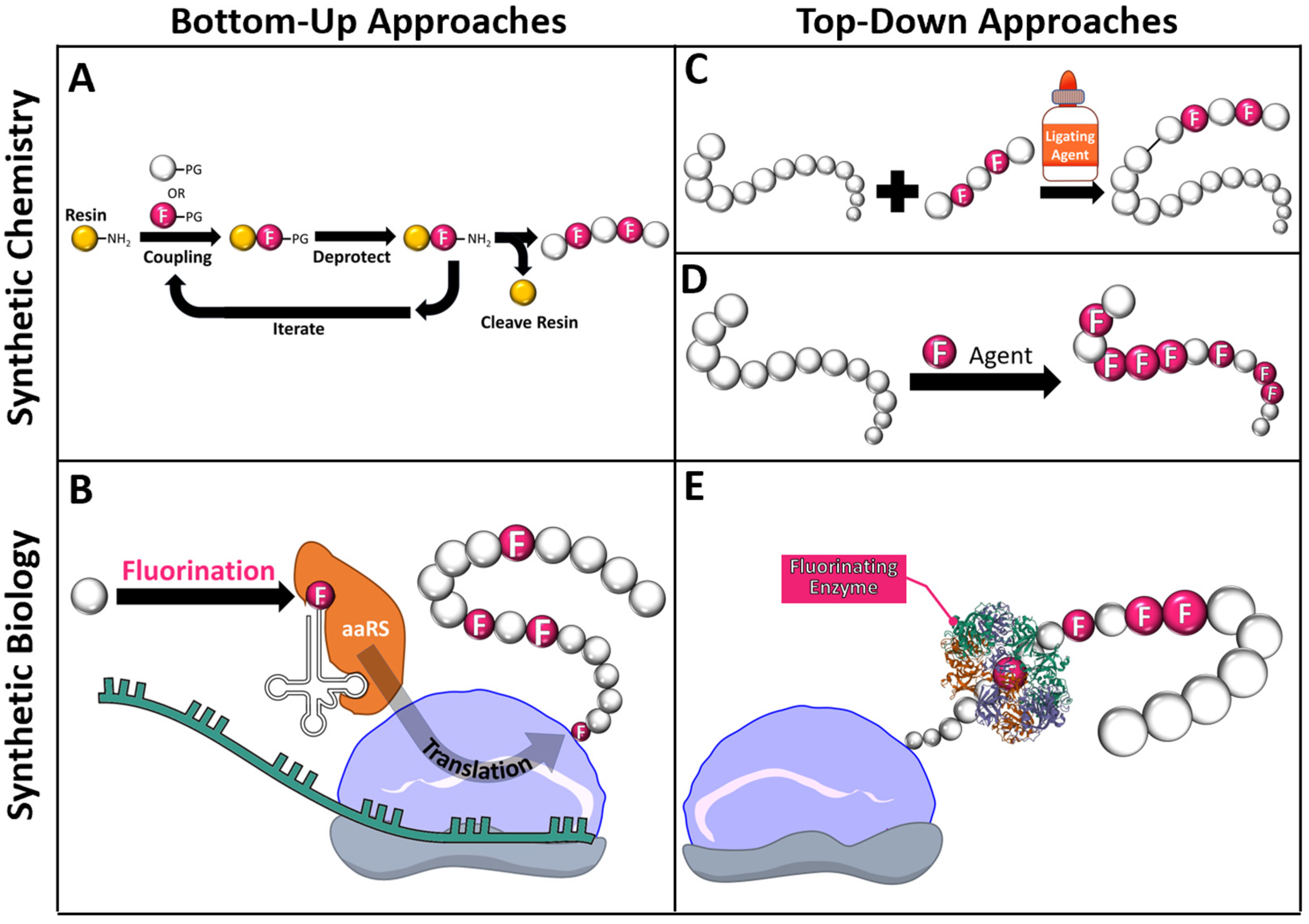
2. Incorporation of Fluorine into Protein and Peptide Materials
2.1. Solid Phase Peptide Synthesis
2.2. Non-Canonical Amino Acid Incorporation
2.3. Protein Fragment Ligation
2.4. Bulk Protein Fluorination via Fluorinating Agents
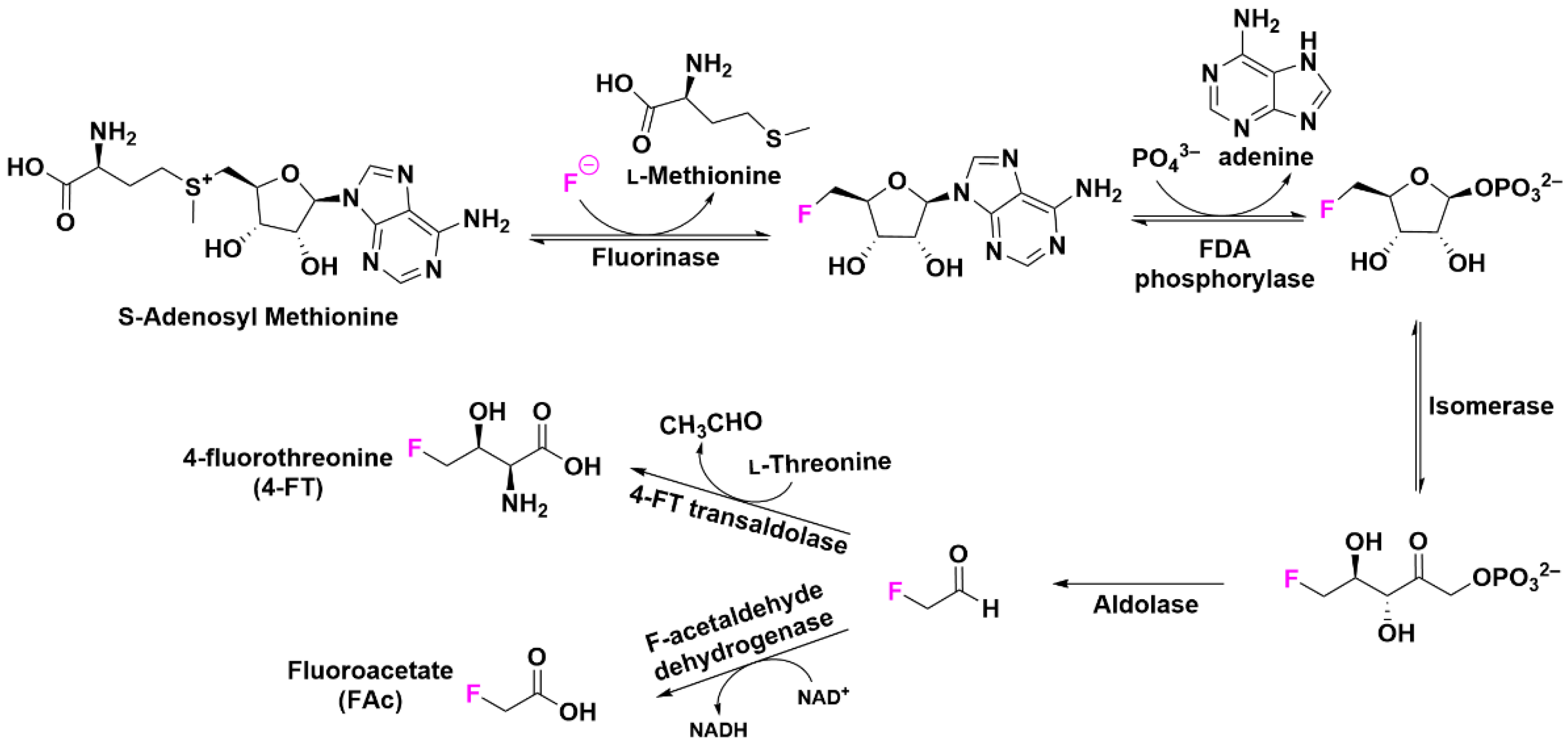
2.5. Post-Translational Modifications with Fluorine
3. Impact of Fluorine on Secondary Structure
3.1. Monomeric Alpha-Helices
3.2. Beta-Sheets
3.3. Intrinsically Disordered Proteins
4. Impact of Fluorine on Supersecondary Structure
4.1. Coiled-Coils
4.2. Collagen Triple-Helices
4.3. Beta Barrels
4.4. Protein Block Copolymers
5. Protein and Peptide Mimetic Therapeutics
| Construct Name | Fluorinated Residue(s) | Number of Fluorines | Position of Fluorines | Synthetic Method | (Super) Secondary Structure(s) | Sequence | References |
|---|---|---|---|---|---|---|---|
| BII1F2 | Leucine (L) | 6 per L (12 total) | Methyl group, δ-carbon | SPPS | Monomer α-helix | TRSSRAGLQFPVGRVHRLLRK | [70] |
| BII5F2 | Leucine (L) | 6 per L (12 total) | Methyl group, δ-carbon | SPPS | Monomer α-helix | RAGLQFPVGRVHRLLRK | [70] |
| BII6F2 | Leucine (L) | 6 per L (12 total) | Methyl group, δ-carbon | SPPS | Monomer α-helix | AGLQFPVGRVHRLLRK | [70] |
| BII10F2 | Leucine (L) | 6 per L (12 total) | Methyl group, δ-carbon | SPPS | Monomer α-helix | FPVGRVHRLLRK | [70] |
| M2F2 | Leucine (L) | 6 per L (12 total) | Methyl group, δ-carbon | SPPS | Monomer α-helix | GIGKFLHAAKKFAKAFVAELMNS | [70] |
| M2F5 | Leucine (L) | 6 per L (30 total) | Methyl group, δ-carbon | SPPS | Monomer α-helix | GIGKFLHALKKFLKAFLAELMNS | [70] |
| DOPA-Phe(4F)-Phe(4F)-OMe | Phenylalanine (F) | 4 per F (8 total) | Aryl ring, all positions | Purchased | Linear Tripeptide | 3OHYFF | [71] |
| 2ZNX (Fluorinated Antibody) | Tryptophan (W) | 1 per W (12 total) | Aryl ring, 5-carbon | Residue-specific ncAA incorporation | Antibody (Combination of α-helices and β-sheets) | Chain A: DIVLTQSPATLSVTPGNSVSLSCRASQSIGNNLHWYQQKSHESPRLLIKYASQSISGIPSRFSGSGSGTDFTLSINSVETEDFGMYFCQQSNSWPYTFGGGTKLEITGGGGSGGGGSGGGGSDIQLQESGPSLVKPSQTLSLTCSVTGDSITSDYWSWIRKFPGNRLEYMGYVSYSGSTYYNPSLKSRISITRDTSKNQYYLDLNSVTTEDTATYYCANWDGDYWGQGTLVTVSAAHHHHHH Chain B: DIVLTQSPATLSVTPGNSVSLSCRASQSIGNNLHWYQQKSHESPRLLIKYASQSISGIPSRFSGSGSGTDFTLSINSVETEDFGMYFCQQSNSWPYTFGGGTKLEITGGGGSGGGGSGGGGSDIQLQESGPSLVKPSQTLSLTCSVTGDSITSDYWSWIRKFPGNRLEYMGYVSYSGSTYYNPSLKSRISITRDTSKNQYYLDLNSVTTEDTATYYCANWDGDYWGQGTLVTVSAAHHHHHH | [72] |
| (flpHypGly)7 | Proline (P) | 1 per P (7 total) | Pyrrolidine ring, 4′-carbon | SPPS | Collagen monomer | PhPGPhPGPhPGPhPGPhPGPhPGPhPG | [73] |
5.1. Bioactive Peptides
5.1.1. Antimicrobial Bioactive Peptides
5.1.2. Antiviral Bioactive Peptides
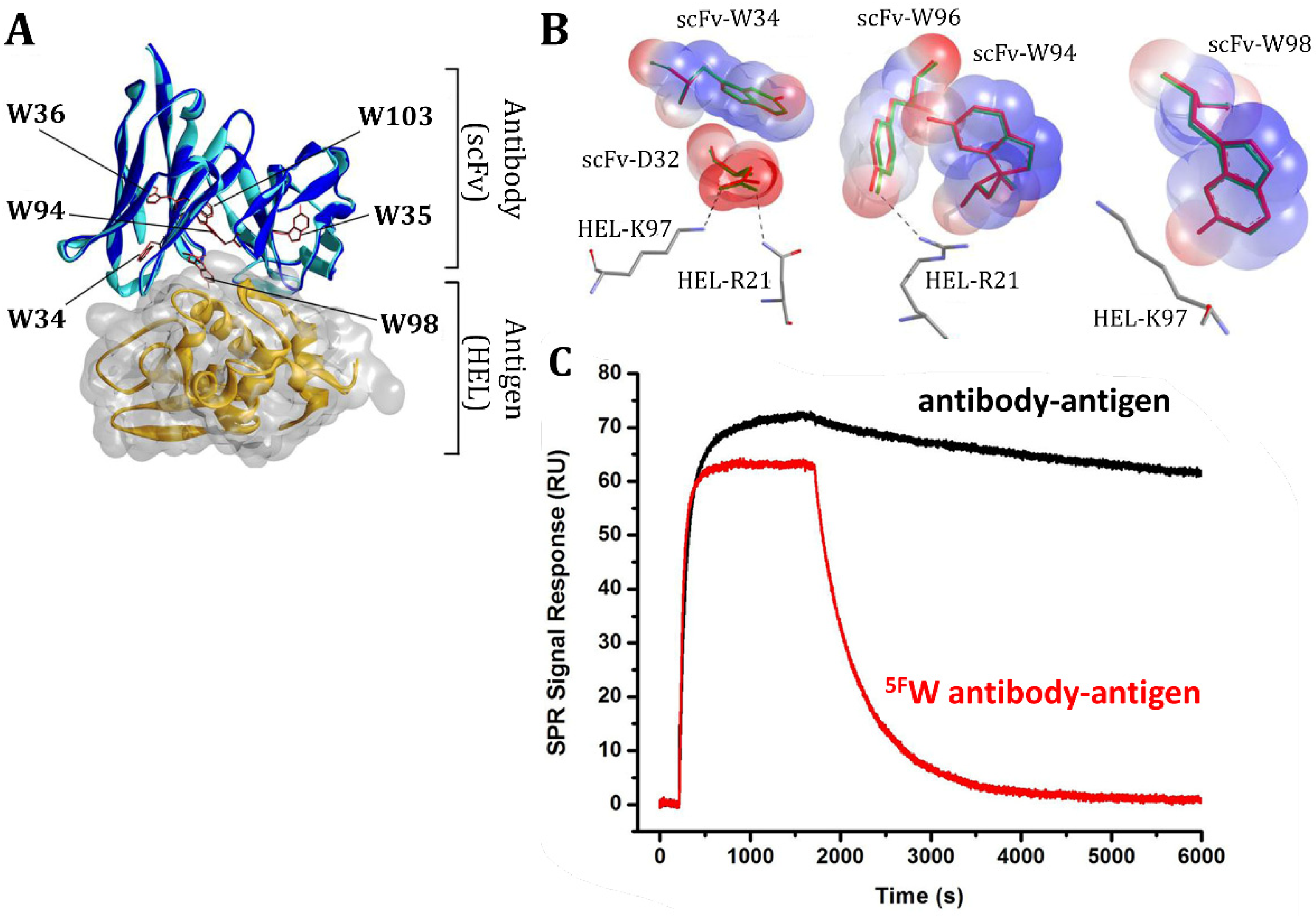
5.2. Fluorinated Antibodies
5.3. Collagen Mimetics for Fiber Repair
6. Drug Delivery
| Construct Name | Fluorinated Residue(s) | Number of Fluorines | Position of Fluorines | Synthetic Method | Assembly Morphology | Sequence | References |
|---|---|---|---|---|---|---|---|
| F-TRAP | Leucine (L) | 3 per L (12 total) | Methyl group, δ-carbon | Residue-specific ncAA incorporation | Protein micelles | MRGSHHHHHHGSACELAARGDATATATATAACGDLAPQMLRELQETNAALQDVRELLRQQVKEITFLKNTVMESDASGLQAARGDATATATATAVDKPIAASAVPGVGVPGVGVPGFGVPGVGVPGVGVPGVGVPGVGVPGFGVPGVGVPGVGVPLEGSGTGAKLN | [81] |
| D1 | (R,R)-β-diaryl (βPhPh) | 1 per βPhPh (1 total) | Aryl ring, 2-ortho position | Ligation | Fibrous nanotubes | AβPhPh | [82] |
| D2 | (S,S)-β-diaryl (βPhPh) | 1 per βPhPh (1 total) | Aryl ring, 2-ortho position | Ligation | Fibrous nanotubes | AβPhPh | [82] |
| MfeGlyK16 | Homoalanine (hA) | 1 per hA (8 total) | Ethyl group, γ-carbon | SPPS | Hydrogelator Agent | hAKhAKhAKhAK hAKhAKhAKhAK-BzOH | [83] |
| DfeGlyK16 | Homoalanine (hA) | 2 per hA (16 total) | Ethyl group, γ-carbon | SPPS | Hydrogelator Agent | hAKhAKhAKhAK hAKhAKhAKhAK-BzOH | [83] |
| TfeGlyK16 | Homoalanine (hA) | 3 per hA (24 total) | Ethyl group, γ-carbon | SPPS | Hydrogelator Agent | hAKhAKhAKhAK hAKhAKhAKhAK-BzOH | [83] |
| Fmoc-3F-Phe-DAP | Phenylalanine (F) | 1 per F (1 total) | Aryl ring, 3-meta position | SPPS | Hydrogelator Agent | Fmoc-F-DAP | [84] |
| Fmoc-F5-Phe-DAP | Phenylalanine (F) | 5 per F (5 total) | Aryl ring, all positions | SPPS | Hydrogelator Agent | Fmoc-F-DAP | [84] |
| 4-fluorobenzyl-diphenylalanine | Phenylalanine (F) | 1 per F (1 total) | Aryl ring, 4-para position | SPPS | Hydrogelator Agent | Bz-FF | [85] |
| No name provided | Phenylalanine (F) | 5 per F (15 total) | Aryl ring, all positions | SPPS | Peptide emulsion | FFFGGGCCGGKGRGD | [86] |
6.1. Spherical, Colloidal Fluorinated Peptide and Protein Vehicles
6.1.1. Fluorinated Protein Micelles
6.1.2. Fluorinated Peptide Emulsions
6.2. Fluorinated Fibers
6.3. Fluorinated Protein and Peptide Hydrogels
7. Bioimaging
7.1. Fluorine-19 Magnetic Resonance Imaging
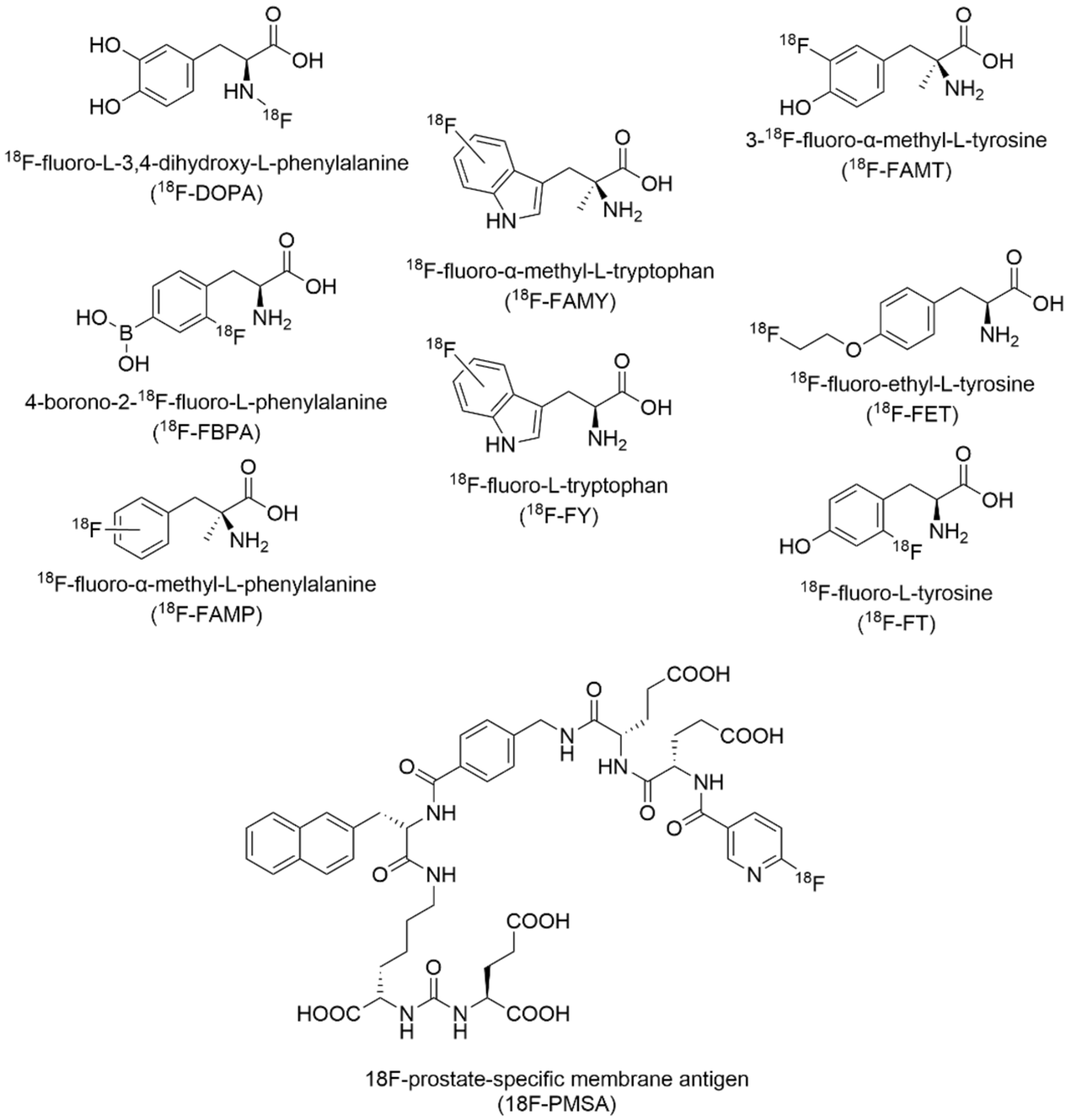
7.2. Fluorine-18 Radiotracers in Positron Emission Tomography Imaging
8. Perspective and Conclusions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Deng, X.; Kokkonda, S.; El Mazouni, F.; White, J.; Burrows, J.N.; Kaminsky, W.; Charman, S.A.; Matthews, D.; Rathod, P.K.; Phillips, M.A. Fluorine Modulates Species Selectivity in the Triazolopyrimidine Class of Plasmodium falciparum Dihydroorotate Dehydrogenase Inhibitors. J. Med. Chem. 2014, 57, 5381–5394. [Google Scholar] [CrossRef] [PubMed]
- Müller, K.; Faeh, C.; Diederich, F. Fluorine in Pharmaceuticals: Looking Beyond Intuition. Science 2007, 317, 1881–1886. [Google Scholar] [CrossRef] [PubMed]
- El’kin, O.V.; Bushuev, A.N.; Tolstobrov, I.V.; Fomin, S.V.; Shirokova, E.S.; Sazanov, A.V.; Kozvonin, V.A.; Kozulin, D.A. Chapter 11—Organofluorine compounds in artificial blood circulation systems. In Fascinating Fluoropolymers and Their Applications; Ameduri, B., Fomin, S., Eds.; Elsevier: Amsterdam, The Netherlands, 2020; pp. 401–424. [Google Scholar]
- Ebnesajjad, S. Applications of Fluorocarbon Gases and Liquids. In Concise Handbook of Fluorocarbon Gases, 1st ed.; Wiley-Scrivener: Hoboken, NJ, USA, 2021; pp. 41–86. [Google Scholar]
- Buer Benjamin, C.; Meagher Jennifer, L.; Stuckey Jeanne, A.; Marsh, E.N.G. Structural basis for the enhanced stability of highly fluorinated proteins. Proc. Natl. Acad. Sci. USA 2012, 109, 4810–4815. [Google Scholar] [CrossRef] [PubMed]
- Tredwell, M.; Gouverneur, V. 1.5 Fluorine in Medicinal Chemistry: Importance of Chirality. In Comprehensive Chirality; Carreira, E.M., Yamamoto, H., Eds.; Elsevier: Amsterdam, The Netherlands, 2012; pp. 70–85. [Google Scholar]
- Charity, R.J.; Webb, T.B.; Elson, J.M.; Hoff, D.E.M.; Pruitt, C.D.; Sobotka, L.G.; Brown, K.W.; Cerizza, G.; Estee, J.; Lynch, W.G.; et al. Observation of the Exotic Isotope 13F Located Four Neutrons beyond the Proton Drip Line. Phys. Rev. Lett. 2021, 126, 132501. [Google Scholar] [CrossRef]
- Rickmeier, J.; Ritter, T. Site-Specific Deoxyfluorination of Small Peptides with [18F]Fluoride. Angew. Chem. Int. Ed. 2018, 57, 14207–14211. [Google Scholar] [CrossRef]
- Leroux, F.R.; Manteau, B.; Vors, J.-P.; Pazenok, S. Trifluoromethyl ethers–synthesis and properties of an unusual substituent. Beilstein J. Org. Chem. 2008, 4, 13. [Google Scholar] [CrossRef]
- Smart, B.E. Characteristics of C-F Systems. In Organofluorine Chemistry: Principles and Commercial Applications; Banks, R.E., Smart, B.E., Tatlow, J.C., Eds.; Springer: Boston, MA, USA, 1994; pp. 57–88. [Google Scholar]
- Spartan 2018; Wavefunction, Inc.: Irvine, CA, USA, 2018.
- Merrifield, R.B. Solid Phase Peptide Synthesis. I. The Synthesis of a Tetrapeptide. J. Am. Chem. Soc. 1963, 85, 2149–2154. [Google Scholar] [CrossRef]
- Hartrampf, N.; Saebi, A.; Poskus, M.; Gates, Z.P.; Callahan, A.J.; Cowfer, A.E.; Hanna, S.; Antilla, S.; Schissel, C.K.; Quartararo, A.J.; et al. Synthesis of proteins by automated flow chemistry. Science 2020, 368, 980–987. [Google Scholar] [CrossRef]
- Wang, P.; Tang, Y.; Tirrell, D.A. Incorporation of Trifluoroisoleucine into Proteins in Vivo. J. Am. Chem. Soc. 2003, 125, 6900–6906. [Google Scholar] [CrossRef]
- Son, S.; Tanrikulu, I.C.; Tirrell, D.A. Stabilization of bzip Peptides through Incorporation of Fluorinated Aliphatic Residues. ChemBioChem 2006, 7, 1251–1257. [Google Scholar] [CrossRef]
- Wang, P.; Fichera, A.; Kumar, K.; Tirrell, D.A. Alternative Translations of a Single RNA Message: An Identity Switch of (2S,3R)-4,4,4-Trifluorovaline between Valine and Isoleucine Codons. Angew. Chem. Int. Ed. 2004, 43, 3664–3666. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Brock, A.; Herberich, B.; Schultz Peter, G. Expanding the Genetic Code of Escherichia coli. Science 2001, 292, 498–500. [Google Scholar] [CrossRef]
- Liu, C.C.; Schultz, P.G. Adding New Chemistries to the Genetic Code. Annu. Rev. Biochem. 2010, 79, 413–444. [Google Scholar] [CrossRef] [PubMed]
- Voloshchuk, N.; Montclare, J.K. Incorporation of unnatural amino acids for synthetic biology. Mol. BioSyst. 2010, 6, 65–80. [Google Scholar] [CrossRef] [PubMed]
- Dawson, P.E.; Muir, T.W.; Clark-Lewis, I.; Kent, S.B.H. Synthesis of Proteins by Native Chemical Ligation. Science 1994, 266, 776–779. [Google Scholar] [CrossRef]
- Bode, J.W. Chemical Protein Synthesis with the α-Ketoacid–Hydroxylamine Ligation. Acc. Chem. Res. 2017, 50, 2104–2115. [Google Scholar] [CrossRef]
- Schmidt, M.; Toplak, A.; Quaedflieg, P.J.L.M.; Nuijens, T. Enzyme-mediated ligation technologies for peptides and proteins. Curr. Opin. Chem. Biol. 2017, 38, 1–7. [Google Scholar] [CrossRef]
- Meyer, D.; Jangra, H.; Walther, F.; Zipse, H.; Renaud, P. A third generation of radical fluorinating agents based on N-fluoro-N-arylsulfonamides. Nat. Commun. 2018, 9, 4888. [Google Scholar] [CrossRef]
- Robalo, J.R.; Vila Verde, A. Unexpected trends in the hydrophobicity of fluorinated amino acids reflect competing changes in polarity and conformation. Phys. Chem. Chem. Phys. 2019, 21, 2029–2038. [Google Scholar] [CrossRef]
- Wang, Y.; Deng, Z.; Qu, X. Characterization of a SAM-dependent fluorinase from a latent biosynthetic pathway for fluoroacetate and 4-fluorothreonine formation in Nocardia brasiliensis [version 1; peer review: 2 approved]. F1000Research 2014, 3, 61. [Google Scholar] [CrossRef]
- McMurry, J.L.; Chang, M.C.Y. Fluorothreonyl-tRNA deacylase prevents mistranslation in the organofluorine producer Streptomyces cattleya. Proc. Natl. Acad. Sci. USA 2017, 114, 11920–11925. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Tong, M.H.; Raab, A.; Fang, Q.; Wang, S.; Kyeremeh, K.; Yu, Y.; Deng, H. An unusual metal-bound 4-fluorothreonine transaldolase from Streptomyces sp. MA37 catalyses promiscuous transaldol reactions. Appl. Microbiol. Biotechnol. 2020, 104, 3885–3896. [Google Scholar] [CrossRef] [PubMed]
- Lyu, Z.; Zhao, Y.; Buuh, Z.Y.; Gorman, N.; Goldman, A.R.; Islam, M.S.; Tang, H.-Y.; Wang, R.E. Steric-Free Bioorthogonal Labeling of Acetylation Substrates Based on a Fluorine–Thiol Displacement Reaction. J. Am. Chem. Soc. 2021, 143, 1341–1347. [Google Scholar] [CrossRef] [PubMed]
- Bondi, A. van der Waals Volumes and Radii. J. Phys. Chem. 1964, 68, 441–451. [Google Scholar] [CrossRef]
- Monde, K.; Miura, N.; Hashimoto, M.; Taniguchi, T.; Inabe, T. Conformational Analysis of Chiral Helical Perfluoroalkyl Chains by VCD. J. Am. Chem. Soc. 2006, 128, 6000–6001. [Google Scholar] [CrossRef]
- Meanwell, N.A. Fluorine and Fluorinated Motifs in the Design and Application of Bioisosteres for Drug Design. J. Med. Chem. 2018, 61, 5822–5880. [Google Scholar] [CrossRef]
- Chiu, H.-P.; Suzuki, Y.; Gullickson, D.; Ahmad, R.; Kokona, B.; Fairman, R.; Cheng, R.P. Helix Propensity of Highly Fluorinated Amino Acids. J. Am. Chem. Soc. 2006, 128, 15556–15557. [Google Scholar] [CrossRef]
- Horng, J.-C.; Raleigh, D.P. Φ-Values beyond the Ribosomally Encoded Amino Acids: Kinetic and Thermodynamic Consequences of Incorporating Trifluoromethyl Amino Acids in a Globular Protein. J. Am. Chem. Soc. 2003, 125, 9286–9287. [Google Scholar] [CrossRef]
- Clark, G.A.; Baleja, J.D.; Kumar, K. Cross-Strand Interactions of Fluorinated Amino Acids in β-Hairpin Constructs. J. Am. Chem. Soc. 2012, 134, 17912–17921. [Google Scholar] [CrossRef][Green Version]
- Chiu, H.-P.; Kokona, B.; Fairman, R.; Cheng, R.P. Effect of Highly Fluorinated Amino Acids on Protein Stability at a Solvent-Exposed Position on an Internal Strand of Protein G B1 Domain. J. Am. Chem. Soc. 2009, 131, 13192–13193. [Google Scholar] [CrossRef]
- Kirberger, S.E.; Maltseva, S.D.; Manulik, J.C.; Einstein, S.A.; Weegman, B.P.; Garwood, M.; Pomerantz, W.C.K. Synthesis of Intrinsically Disordered Fluorinated Peptides for Modular Design of High-Signal 19F MRI Agents. Angew. Chem. Int. Ed. 2017, 56, 6440–6444. [Google Scholar] [CrossRef] [PubMed]
- Beck, K.; Brodsky, B. Supercoiled Protein Motifs: The Collagen Triple-Helix and the α-Helical Coiled Coil. J. Struct. Biol. 1998, 122, 17–29. [Google Scholar] [CrossRef] [PubMed]
- Babu, I.R.; Hamill, E.K.; Kumar, K. A Highly Stereospecific and Efficient Synthesis of Homopentafluoro-phenylalanine. J. Org. Chem. 2004, 69, 5468–5470. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.-Y.; Lee, K.-H.; Al-Hashimi, H.M.; Marsh, E.N.G. Modulating Protein Structure with Fluorous Amino Acids: Increased Stability and Native-like Structure Conferred on a 4-Helix Bundle Protein by Hexafluoroleucine. J. Am. Chem. Soc. 2006, 128, 337–343. [Google Scholar] [CrossRef]
- Tang, Y.; Ghirlanda, G.; Petka, W.A.; Nakajima, T.; DeGrado, W.F.; Tirrell, D.A. Fluorinated Coiled-Coil Proteins Prepared In Vivo Display Enhanced Thermal and Chemical Stability. Angew. Chem. Int. Ed. 2001, 40, 1494–1496. [Google Scholar] [CrossRef]
- Erdbrink, H.; Nyakatura, E.K.; Huhmann, S.; Gerling, U.I.M.; Lentz, D.; Koksch, B.; Czekelius, C. Synthesis of enantiomerically pure (2S,3S)-5,5,5-trifluoroisoleucine and (2R,3S)-5,5,5-trifluoro-allo-isoleucine. Beilstein J. Org. Chem. 2013, 9, 2009–2014. [Google Scholar] [CrossRef] [PubMed]
- Berger, A.A.; Völler, J.-S.; Budisa, N.; Koksch, B. Deciphering the Fluorine Code—The Many Hats Fluorine Wears in a Protein Environment. Acc. Chem. Res. 2017, 50, 2093–2103. [Google Scholar] [CrossRef] [PubMed]
- Reeb, J.; Rost, B. Secondary Structure Prediction. In Encyclopedia of Bioinformatics and Computational Biology; Ranganathan, S., Gribskov, M., Nakai, K., Schönbach, C., Eds.; Academic Press: Oxford, UK, 2019; pp. 488–496. [Google Scholar]
- Syud, F.A.; Stanger, H.E.; Gellman, S.H. Interstrand Side Chain−Side Chain Interactions in a Designed β-Hairpin: Significance of Both Lateral and Diagonal Pairings. J. Am. Chem. Soc. 2001, 123, 8667–8677. [Google Scholar] [CrossRef]
- Widyarani; Sari, Y.W.; Ratnaningsih, E.; Sanders, J.P.M.; Bruins, M.E. Production of hydrophobic amino acids from biobased resources: Wheat gluten and rubber seed proteins. Appl. Microbiol. Biotechnol. 2016, 100, 7909–7920. [Google Scholar] [CrossRef]
- Uversky, V.N. The alphabet of intrinsic disorder. Intrinsically Disord. Proteins 2013, 1, e24684. [Google Scholar] [CrossRef]
- Pelley, J.W. 3-Protein Structure and Function. In Elsevier’s Integrated Review Biochemistry, 2nd ed.; Pelley, J.W., Ed.; W. B. Saunders: Philadelphia, PA, USA, 2012; pp. 19–28. [Google Scholar]
- Ulrich, T.; Rapaport, D. Biogenesis of beta-barrel proteins in evolutionary context. Int. J. Med. Microbiol. 2015, 305, 259–264. [Google Scholar] [CrossRef] [PubMed]
- Rabotyagova, O.S.; Cebe, P.; Kaplan, D.L. Protein-Based Block Copolymers. Biomacromolecules 2011, 12, 269–289. [Google Scholar] [CrossRef] [PubMed]
- Liang, H.; Chen, H.; Fan, K.; Wei, P.; Guo, X.; Jin, C.; Zeng, C.; Tang, C.; Lai, L. De Novo Design of a βαβ Motif. Angew. Chem. Int. Ed. 2009, 48, 3301–3303. [Google Scholar] [CrossRef]
- Buer, B.C.; Marsh, E.N.G. Fluorine: A new element in protein design. Protein Sci. 2012, 21, 453–462. [Google Scholar] [CrossRef]
- Jäckel, C.; Salwiczek, M.; Koksch, B. Fluorine in a Native Protein Environment—How the Spatial Demand and Polarity of Fluoroalkyl Groups Affect Protein Folding. Angew. Chem. Int. Ed. 2006, 45, 4198–4203. [Google Scholar] [CrossRef]
- Tang, Y.; Ghirlanda, G.; Vaidehi, N.; Kua, J.; Mainz, D.T.; Goddard, W.A.; DeGrado, W.F.; Tirrell, D.A. Stabilization of Coiled-Coil Peptide Domains by Introduction of Trifluoroleucine. Biochemistry 2001, 40, 2790–2796. [Google Scholar] [CrossRef] [PubMed]
- Montclare, J.K.; Son, S.; Clark, G.A.; Kumar, K.; Tirrell, D.A. Biosynthesis and Stability of Coiled-Coil Peptides Containing (2S,4R)-5,5,5-Trifluoroleucine and (2S,4S)-5,5,5-Trifluoroleucine. ChemBioChem 2009, 10, 84–86. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.; Tirrell, D.A. Biosynthesis of a Highly Stable Coiled-Coil Protein Containing Hexafluoroleucine in an Engineered Bacterial Host. J. Am. Chem. Soc. 2001, 123, 11089–11090. [Google Scholar] [CrossRef]
- More, H.T.; Zhang, K.S.; Srivastava, N.; Frezzo, J.A.; Montclare, J.K. Influence of Fluorination on Protein-Engineered Coiled-Coil Fibers. Biomacromolecules 2015, 16, 1210–1217. [Google Scholar] [CrossRef]
- Bilgiçer, B.; Fichera, A.; Kumar, K. A Coiled Coil with a Fluorous Core. J. Am. Chem. Soc. 2001, 123, 4393–4399. [Google Scholar] [CrossRef]
- Cejas, M.A.; Kinney, W.A.; Chen, C.; Vinter, J.G.; Almond, H.R., Jr.; Balss, K.M.; Maryanoff, C.A.; Schmidt, U.; Breslav, M.; Mahan, A.; et al. Thrombogenic collagen-mimetic peptides: Self-assembly of triple helix-based fibrils driven by hydrophobic interactions. Proc. Natl. Acad. Sci. USA 2008, 105, 8513–8518. [Google Scholar] [CrossRef] [PubMed]
- Welte, H.; Zhou, T.; Mihajlenko, X.; Mayans, O.; Kovermann, M. What does fluorine do to a protein? Thermodynamic, and highly-resolved structural insights into fluorine-labelled variants of the cold shock protein. Sci. Rep. 2020, 10, 2640. [Google Scholar] [CrossRef] [PubMed]
- Yuvienco, C.; More, H.T.; Haghpanah, J.S.; Tu, R.S.; Montclare, J.K. Modulating Supramolecular Assemblies and Mechanical Properties of Engineered Protein Materials by Fluorinated Amino Acids. Biomacromolecules 2012, 13, 2273–2278. [Google Scholar] [CrossRef] [PubMed]
- Shoulders, M.D.; Raines, R.T. Collagen Structure and Stability. Annu. Rev. Biochem. 2009, 78, 929–958. [Google Scholar] [CrossRef]
- Chorghade, M.S.; Mohapatra, D.K.; Sahoo, G.; Gurjar, M.K.; Mandlecha, M.V.; Bhoite, N.; Moghe, S.; Raines, R.T. Practical syntheses of 4-fluoroprolines. J. Fluor. Chem. 2008, 129, 781–784. [Google Scholar] [CrossRef] [PubMed]
- Hodges, J.A.; Raines, R.T. Stereoelectronic Effects on Collagen Stability: The Dichotomy of 4-Fluoroproline Diastereomers. J. Am. Chem. Soc. 2003, 125, 9262–9263. [Google Scholar] [CrossRef]
- Lalonde, J.M.; Bernlohr, D.A.; Banaszak, L.J. The up-and-down β-barrel proteins. FASEB J. 1994, 8, 1240–1247. [Google Scholar] [CrossRef]
- Dimitrov, D.S. Therapeutic Proteins. In Therapeutic Proteins: Methods and Protocols; Voynov, V., Caravella, J.A., Eds.; Humana Press: Totowa, NJ, USA, 2012; pp. 1–26. [Google Scholar]
- Murray, J.E.; Laurieri, N.; Delgoda, R. Chapter 24-Proteins. In Pharmacognosy; Badal, S., Delgoda, R., Eds.; Academic Press: Boston, MA, USA, 2017; pp. 477–494. [Google Scholar]
- Li Petri, G.; Di Martino, S.; De Rosa, M. Peptidomimetics: An Overview of Recent Medicinal Chemistry Efforts toward the Discovery of Novel Small Molecule Inhibitors. J. Med. Chem. 2022, 65, 7438–7475. [Google Scholar] [CrossRef]
- Hagmann, W.K. The Many Roles for Fluorine in Medicinal Chemistry. J. Med. Chem. 2008, 51, 4359–4369. [Google Scholar] [CrossRef]
- Shah, P.; Westwell, A.D. The role of fluorine in medicinal chemistry. J. Enzym. Inhib. Med. Chem. 2007, 22, 527–540. [Google Scholar] [CrossRef]
- Meng, H.; Kumar, K. Antimicrobial Activity and Protease Stability of Peptides Containing Fluorinated Amino Acids. J. Am. Chem. Soc. 2007, 129, 15615–15622. [Google Scholar] [CrossRef]
- Hu, T.; Agazani, O.; Nir, S.; Cohen, M.; Pan, S.; Reches, M. Antiviral Activity of Peptide-Based Assemblies. ACS Appl. Mater. Interfaces 2021, 13, 48469–48477. [Google Scholar] [CrossRef] [PubMed]
- Acchione, M.; Lee, Y.-C.; DeSantis, M.E.; Lipschultz, C.A.; Wlodawer, A.; Li, M.; Shanmuganathan, A.; Walter, R.L.; Smith-Gill, S.; Barchi, J.J. Specific Fluorine Labeling of the HyHEL10 Antibody Affects Antigen Binding and Dynamics. Biochemistry 2012, 51, 6017–6027. [Google Scholar] [CrossRef] [PubMed]
- Dones, J.M.; Tanrikulu, I.C.; Chacko, J.V.; Schroeder, A.B.; Hoang, T.T.; Gibson, A.L.F.; Eliceiri, K.W.; Raines, R.T. Optimization of interstrand interactions enables burn detection with a collagen-mimetic peptide. Org. Biomol. Chem. 2019, 17, 9906–9912. [Google Scholar] [CrossRef] [PubMed]
- Mardirossian, M.; Rubini, M.; Adamo, M.F.A.; Scocchi, M.; Saviano, M.; Tossi, A.; Gennaro, R.; Caporale, A. Natural and Synthetic Halogenated Amino Acids—Structural and Bioactive Features in Antimicrobial Peptides and Peptidomimetics. Molecules 2021, 26, 7401. [Google Scholar] [CrossRef]
- Mora, L.; Aristoy, M.C.; Toldrá, F. Bioactive Peptides. In Encyclopedia of Food Chemistry; Melton, L., Shahidi, F., Varelis, P., Eds.; Academic Press: Oxford, UK, 2019; pp. 381–389. [Google Scholar]
- Lutz, H.U. Homeostatic roles of naturally occurring antibodies: An overview. J. Autoimmun. 2007, 29, 287–294. [Google Scholar] [CrossRef]
- Berger, M.; Shankar, V.; Vafai, A. Therapeutic Applications of Monoclonal Antibodies. Am. J. Med. Sci. 2002, 324, 14–30. [Google Scholar] [CrossRef]
- Ellison, A.J.; Tanrikulu, I.C.; Dones, J.M.; Raines, R.T. Cyclic Peptide Mimetic of Damaged Collagen. Biomacromolecules 2020, 21, 1539–1547. [Google Scholar] [CrossRef]
- Wen, H.; Jung, H.; Li, X. Drug Delivery Approaches in Addressing Clinical Pharmacology-Related Issues: Opportunities and Challenges. AAPS J. 2015, 17, 1327–1340. [Google Scholar] [CrossRef] [PubMed]
- Chu, S.; Wang, A.L.; Bhattacharya, A.; Montclare, J.K. Protein based biomaterials for therapeutic and diagnostic applications. Prog. Biomed. Eng. 2021, 4, 012003. [Google Scholar] [CrossRef]
- Hill, L.K.; Frezzo, J.A.; Katyal, P.; Hoang, D.M.; Gironda, Z.B.Y.; Xu, C.; Xie, X.; Delgado-Fukushima, E.; Wadghiri, Y.Z.; Montclare, J.K. Protein-Engineered Nanoscale Micelles for Dynamic 19F Magnetic Resonance and Therapeutic Drug Delivery. ACS Nano 2019, 13, 2969–2985. [Google Scholar] [CrossRef] [PubMed]
- Bonetti, A.; Pellegrino, S.; Das, P.; Yuran, S.; Bucci, R.; Ferri, N.; Meneghetti, F.; Castellano, C.; Reches, M.; Gelmi, M.L. Dipeptide Nanotubes Containing Unnatural Fluorine-Substituted β2,3-Diarylamino Acid and l-Alanine as Candidates for Biomedical Applications. Org. Lett. 2015, 17, 4468–4471. [Google Scholar] [CrossRef] [PubMed]
- Chowdhary, S.; Schmidt, R.F.; Sahoo, A.K.; tom Dieck, T.; Hohmann, T.; Schade, B.; Brademann-Jock, K.; Thünemann, A.F.; Netz, R.R.; Gradzielski, M.; et al. Rational design of amphiphilic fluorinated peptides: Evaluation of self-assembly properties and hydrogel formation. Nanoscale 2022, 14, 10176–10189. [Google Scholar] [CrossRef] [PubMed]
- Raymond, D.M.; Abraham, B.L.; Fujita, T.; Watrous, M.J.; Toriki, E.S.; Takano, T.; Nilsson, B.L. Low-Molecular-Weight Supramolecular Hydrogels for Sustained and Localized in Vivo Drug Delivery. ACS Appl. Bio. Mater. 2019, 2, 2116–2124. [Google Scholar] [CrossRef]
- Wu, F.-Y.; Hsu, S.-M.; Cheng, H.; Hsu, L.-H.; Lin, H.-C. The effect of fluorine on supramolecular hydrogelation of 4-fluorobenzyl-capped diphenylalanine. New J. Chem. 2015, 39, 4240–4243. [Google Scholar] [CrossRef]
- Medina, S.H.; Michie, M.S.; Miller, S.E.; Schnermann, M.J.; Schneider, J.P. Fluorous Phase-Directed Peptide Assembly Affords Nano-Peptisomes Capable of Ultrasound-Triggered Cellular Delivery. Angew. Chem. Int. Ed. 2017, 56, 11404–11408. [Google Scholar] [CrossRef]
- Liu, Y.; Tan, J.; Thomas, A.; Ou-Yang, D.; Muzykantov, V.R. The shape of things to come: Importance of design in nanotechnology for drug delivery. Ther. Deliv. 2012, 3, 181–194. [Google Scholar] [CrossRef]
- Kashapov, R.; Gaynanova, G.; Gabdrakhmanov, D.; Kuznetsov, D.; Pavlov, R.; Petrov, K.; Zakharova, L.; Sinyashin, O. Self-Assembly of Amphiphilic Compounds as a Versatile Tool for Construction of Nanoscale Drug Carriers. Int. J. Mol. Sci. 2020, 21, 6961. [Google Scholar] [CrossRef]
- Nasr, M.; Nawaz, S.; Elhissi, A. Amphotericin B lipid nanoemulsion aerosols for targeting peripheral respiratory airways via nebulization. Int. J. Pharm. 2012, 436, 611–616. [Google Scholar] [CrossRef]
- Arbain, N.H.; Basri, M.; Salim, N.; Wui, W.T.; Abdul Rahman, M.B. Development and Characterization of Aerosol Nanoemulsion System Encapsulating Low Water Soluble Quercetin for Lung Cancer Treatment. Mater. Today Proc. 2018, 5, S137–S142. [Google Scholar] [CrossRef]
- Asmawi, A.A.; Salim, N.; Abdulmalek, E.; Abdul Rahman, M.B. Modeling the Effect of Composition on Formation of Aerosolized Nanoemulsion System Encapsulating Docetaxel and Curcumin Using D-Optimal Mixture Experimental Design. Int. J. Mol. Sci. 2020, 21, 4357. [Google Scholar] [CrossRef]
- Sloand, J.N.; Nguyen, T.T.; Zinck, S.A.; Cook, E.C.; Zimudzi, T.J.; Showalter, S.A.; Glick, A.B.; Simon, J.C.; Medina, S.H. Ultrasound-Guided Cytosolic Protein Delivery via Transient Fluorous Masks. ACS Nano 2020, 14, 4061–4073. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.; Singh, I.; Sharma, A.K.; Kumar, P. Ultrashort Peptide Self-Assembly: Front-Runners to Transport Drug and Gene Cargos. Front. Bioeng. Biotechnol. 2020, 8, 504. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Erbas, A.; Tantakitti, F.; Palmer, L.C.; Jackman, J.A.; Olvera de la Cruz, M.; Cho, N.-J.; Stupp, S.I. Co-assembly of Peptide Amphiphiles and Lipids into Supramolecular Nanostructures Driven by Anion−π Interactions. J. Am. Chem. Soc. 2017, 139, 7823–7830. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Lou, S.; Yu, Z. Polar−π Interactions Promote Self-assembly of Dipeptides into Laminated Nanofibers. Langmuir 2019, 35, 4710–4717. [Google Scholar] [CrossRef]
- Hamley, I.W. Peptide Nanotubes. Angew. Chem. Int. Ed. 2014, 53, 6866–6881. [Google Scholar] [CrossRef] [PubMed]
- Handelman, A.; Lavrov, S.; Kudryavtsev, A.; Khatchatouriants, A.; Rosenberg, Y.; Mishina, E.; Rosenman, G. Nonlinear Optical Bioinspired Peptide Nanostructures. Adv. Opt. Mater. 2013, 1, 875–884. [Google Scholar] [CrossRef]
- Yuran, S.; Razvag, Y.; Reches, M. Coassembly of Aromatic Dipeptides into Biomolecular Necklaces. ACS Nano 2012, 6, 9559–9566. [Google Scholar] [CrossRef]
- Alavijeh, M.S.; Chishty, M.; Qaiser, M.Z.; Palmer, A.M. Drug metabolism and pharmacokinetics, the blood-brain barrier, and central nervous system drug discovery. NeuroRX 2005, 2, 554–571. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, W.; Gong, C.; Liu, B.; Li, Y.; Wang, L.; Su, Z.; Wei, G. Recent advances in the fabrication, functionalization, and bioapplications of peptide hydrogels. Soft Matter 2020, 16, 10029–10045. [Google Scholar] [CrossRef]
- Bashir, S.; Hina, M.; Iqbal, J.; Rajpar, A.H.; Mujtaba, M.A.; Alghamdi, N.A.; Wageh, S.; Ramesh, K.; Ramesh, S. Fundamental Concepts of Hydrogels: Synthesis, Properties, and Their Applications. Polymers 2020, 12, 2702. [Google Scholar] [CrossRef] [PubMed]
- Eggeling, C. Advances in bioimaging—Challenges and potentials. J. Phys. D Appl. Phys. 2018, 51, 040201. [Google Scholar] [CrossRef]
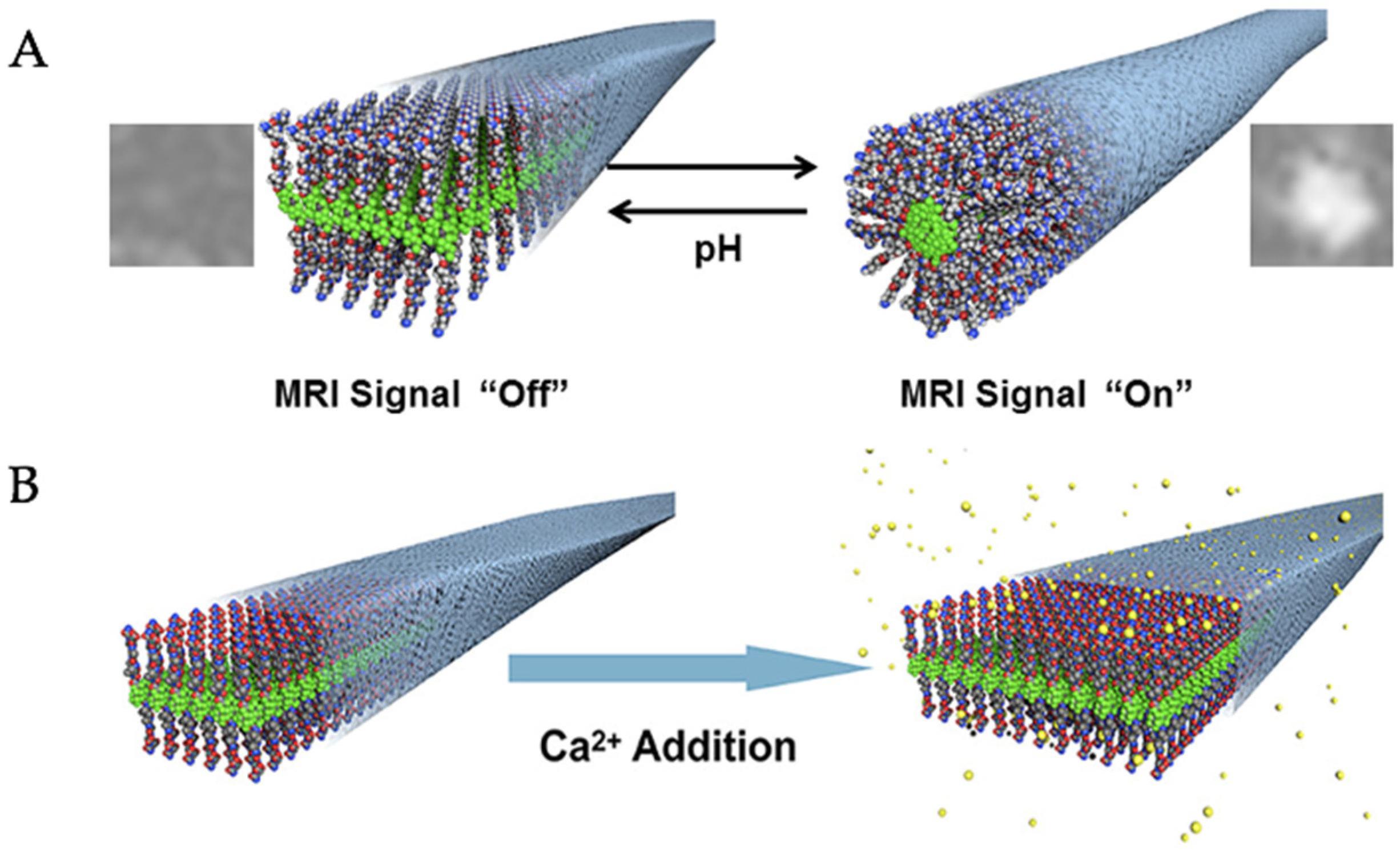
- Preslar, A.T.; Tantakitti, F.; Park, K.; Zhang, S.; Stupp, S.I.; Meade, T.J. 19F Magnetic Resonance Imaging Signals from Peptide Amphiphile Nanostructures Are Strongly Affected by Their Shape. ACS Nano 2016, 10, 7376–7384. [Google Scholar] [CrossRef] [PubMed]
- Preslar, A.T.; Lilley, L.M.; Sato, K.; Zhang, S.; Chia, Z.K.; Stupp, S.I.; Meade, T.J. Calcium-Induced Morphological Transitions in Peptide Amphiphiles Detected by 19F-Magnetic Resonance Imaging. ACS Appl. Mater. Interfaces 2017, 9, 39890–39894. [Google Scholar] [CrossRef]
- Yuan, Y.; Sun, H.; Ge, S.; Wang, M.; Zhao, H.; Wang, L.; An, L.; Zhang, J.; Zhang, H.; Hu, B.; et al. Controlled Intracellular Self-Assembly and Disassembly of 19F Nanoparticles for MR Imaging of Caspase 3/7 in Zebrafish. ACS Nano 2015, 9, 761–768. [Google Scholar] [CrossRef]
- Sloand, J.N.; Miller, M.A.; Medina, S.H. Fluorinated peptide biomaterials. Pept. Sci. 2021, 113, e24184. [Google Scholar] [CrossRef]
- Fox, M.S.; Gaudet, J.M.; Foster, P.J. Fluorine-19 MRI Contrast Agents for Cell Tracking and Lung Imaging. Magn. Reson. Insights 2015, 8s1. [Google Scholar] [CrossRef]
- Shukla, A.; Kumar, U. Positron emission tomography: An overview. J. Med. Phys. 2006, 31, 13–21. [Google Scholar] [CrossRef]
- Fletcher, J.W.; Djulbegovic, B.; Soares, H.P.; Siegel, B.A.; Lowe, V.J.; Lyman, G.H.; Coleman, R.E.; Wahl, R.; Paschold, J.C.; Avril, N.; et al. Recommendations on the Use of 18F-FDG PET in Oncology. J. Nucl. Med. 2008, 49, 480–508. [Google Scholar] [CrossRef]
- Berger, A. Positron emission tomography. BMJ 2003, 326, 1449. [Google Scholar] [CrossRef]
- Awenat, S.; Piccardo, A.; Carvoeiras, P.; Signore, G.; Giovanella, L.; Prior, J.O.; Treglia, G. Diagnostic Role of 18F-PSMA-1007 PET/CT in Prostate Cancer Staging: A Systematic Review. Diagnostics 2021, 11, 552. [Google Scholar] [CrossRef] [PubMed]
- Huynh, P.T.; Soni, N.; Pal, R.; Sarkar, S.; Jung, J.-M.; Lee, W.; Yoo, J. Direct radiofluorination of a heat-sensitive antibody by Al–18F complexation. New J. Chem. 2019, 43, 15389–15395. [Google Scholar] [CrossRef]
- Krishnan, H.S.; Ma, L.; Vasdev, N.; Liang, S.H. 18F-Labeling of Sensitive Biomolecules for Positron Emission Tomography. Chem. –A Eur. J. 2017, 23, 15553–15577. [Google Scholar] [CrossRef]
- Paulus, A.; Desai, P.; Carney, B.; Carlucci, G.; Reiner, T.; Brand, C.; Weber, W.A. Development of a clickable bimodal fluorescent/PET probe for in vivo imaging. EJNMMI Res. 2015, 5, 43. [Google Scholar] [CrossRef] [PubMed]
- Makaravage, K.J.; Brooks, A.F.; Mossine, A.V.; Sanford, M.S.; Scott, P.J.H. Copper-Mediated Radiofluorination of Arylstannanes with [18F]KF. Org. Lett. 2016, 18, 5440–5443. [Google Scholar] [CrossRef] [PubMed]
- Stadmiller, S.S.; Aguilar, J.S.; Waudby, C.A.; Pielak, G.J. Rapid Quantification of Protein-Ligand Binding via 19F NMR Lineshape Analysis. Biophys. J. 2020, 118, 2537–2548. [Google Scholar] [CrossRef]
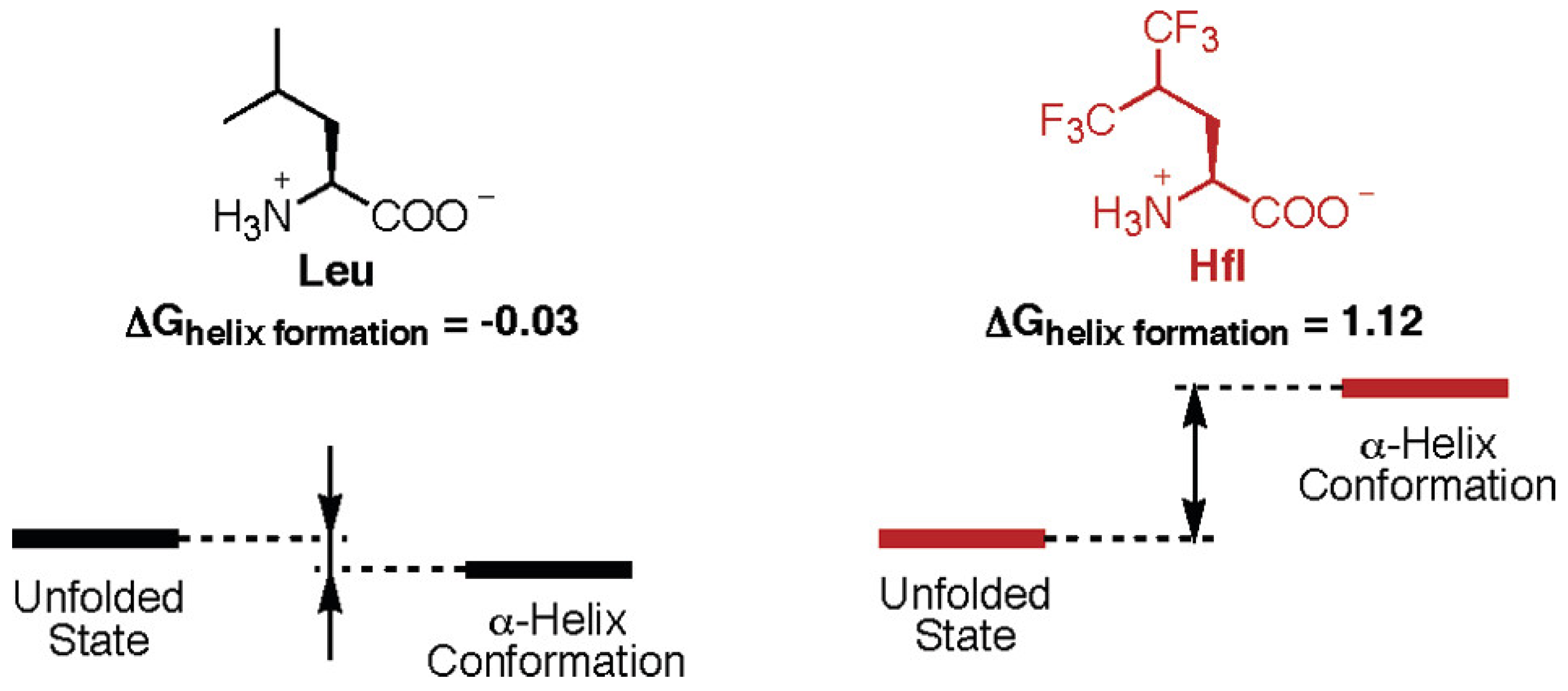



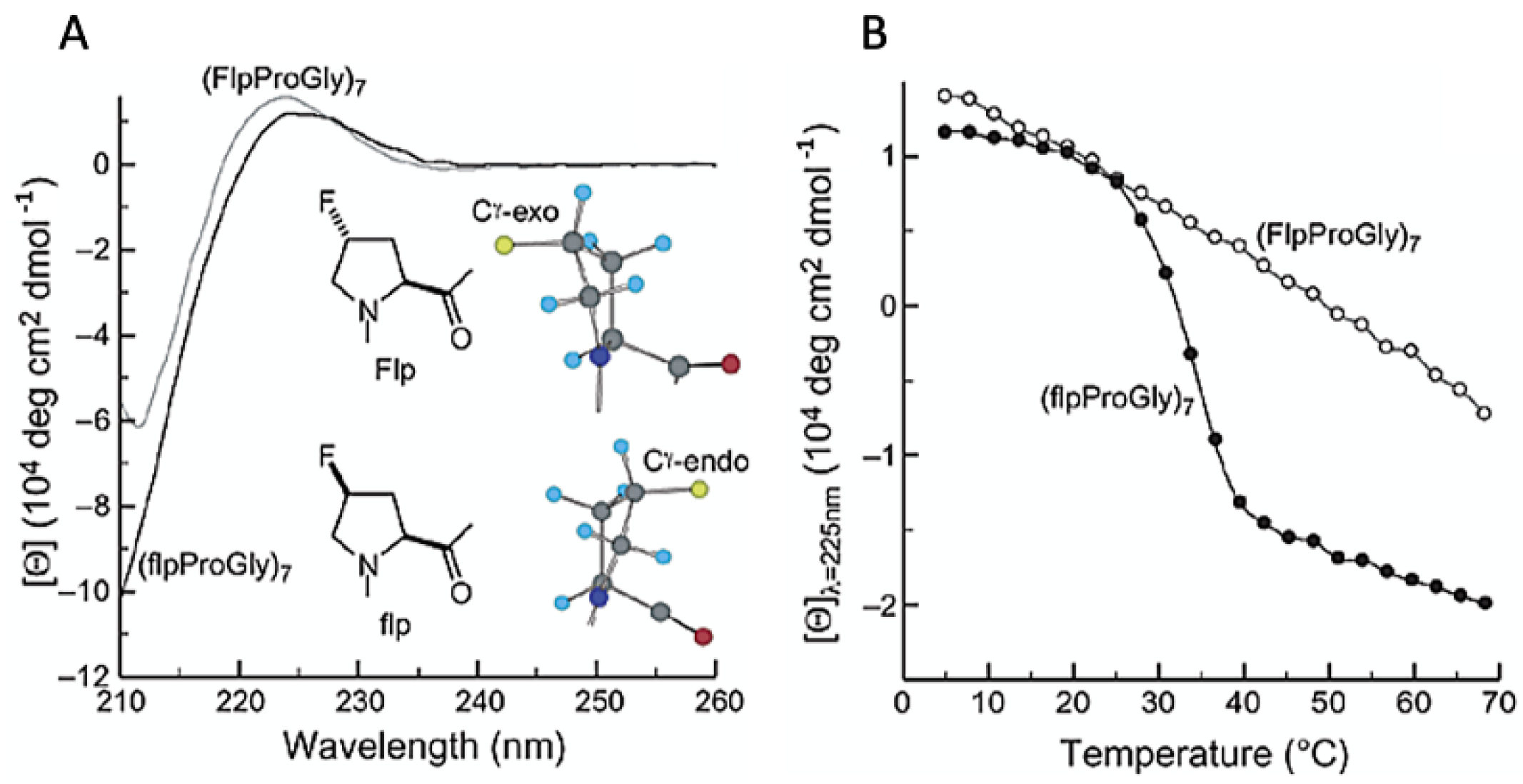
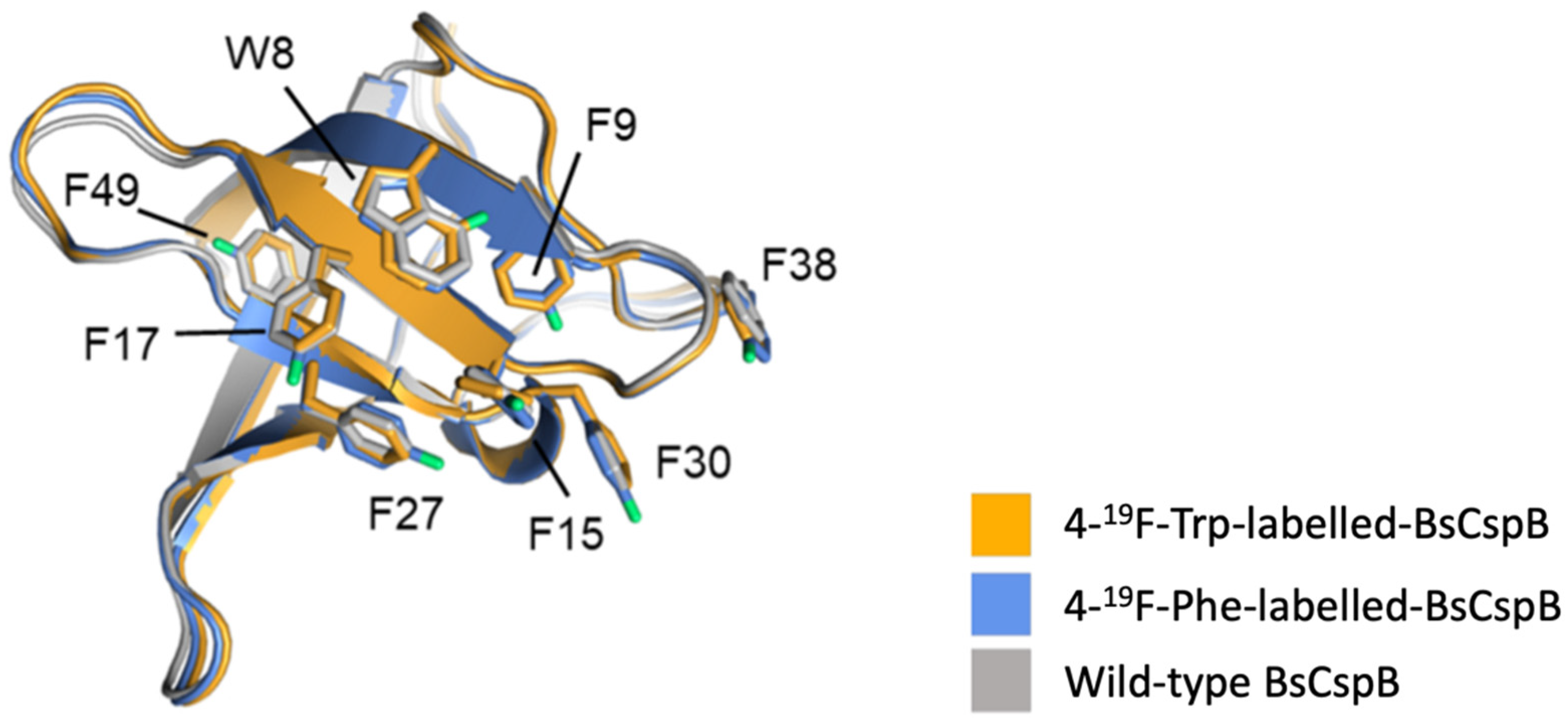

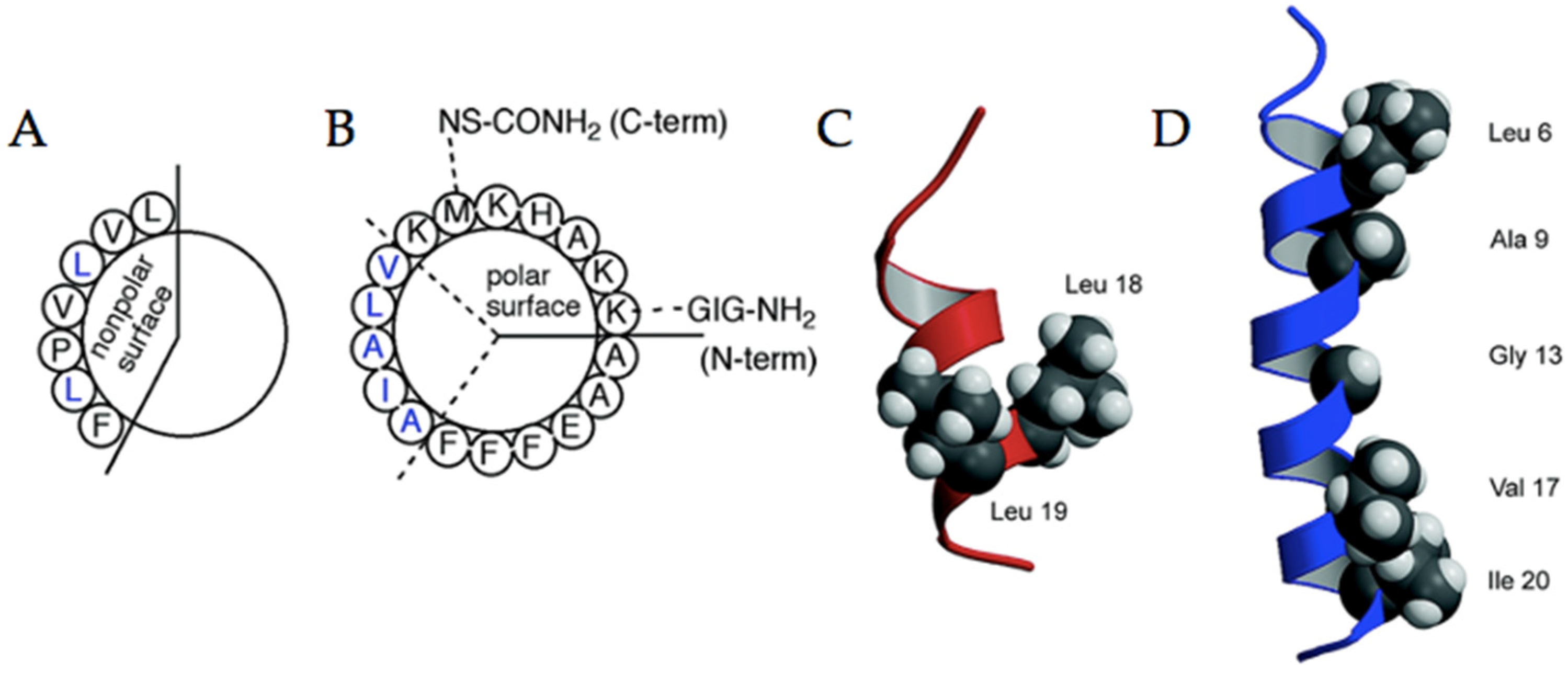

| Construct Name | Fluorinated Residue(s) | Number of Fluorines | Position of Fluorines | Synthetic Method | Secondary Structure | Sequence | References |
|---|---|---|---|---|---|---|---|
| KHfl | Leucine (L) | 6 per L (6 total) | Methyl groups, δ-carbons | SPPS | α-helix | YGGKAAAAKLAAKAAAAK | [32] |
| tfV3 | Valine (V) | 3 per V (3 total) | Methyl group, γ-carbon | SPPS | β-sheet | MKVIFLKDVKGKGKKGEIK NVADGYANNFLFKQGLAIE ATPANLKALEAQK | [33] |
| tfV21 | Valine (V) | 3 per V (3 total) | Methyl group, γ-carbon | SPPS | β-sheet | MKVIFLKDVKGKGKKGEIK NVADGYANNFLFKQGLAIE ATPANLKALEAQK | [33] |
| HH1 | Leucine (L) | 6 per L (12 total) | Methyl groups, δ-carbons | SPPS | β-hairpin | QRLTNCCNTLEG (cyclic) | [34] |
| HH2 | Leucine (L) | 6 per L (12 total) | Methyl groups, δ-carbons | SPPS | β-hairpin | L@VPAVTL (cyclic) | [34] |
| GB1-QfL | Leucine (L) | 4 per L (4 total) | Methyl groups, δ-carbons | SPPS | β-sheet | MGYKLALNGKTLKGETT TEAVDAATAEKVFKQYA NDNEGEWAYDDATLKT FLVTE | [35] |
| GB1-Hfl | Leucine (L) | 6 per L (6 total) | Methyl groups, δ-carbons | SPPS | β-sheet (globular protein) | MGYKLALNGKTLKGETTTE AVDAATAEKVFKQYANDN EGEWAYDDATLKTFLVTE | [35] |
| GB1-Pff | Phenylalanine (F) | 5 per F (5 total) | Aryl ring, all positions | SPPS | β-sheet (globular protein) | MGYKLALNGKTLKGETTTE AVDAATAEKVFKQYANDN EGEWAYDDATLKTFFVTE | [35] |
| Peptide 1 (n = 7) | Lysine (K) | 3 per TFA-modified K (21 total) | Modified acetyl group | SPPS | Intrinsically disordered protein | YGKTFAKKTFAKKTFAKKTFAKK TFAKKTFAKKTFAKK | [36] |
| Construct Name | Fluorinated Residue(s) | Number of Fluorines | Position of Fluorines | Synthetic Method | (Super) Secondary Structure(s) | Sequence | References |
|---|---|---|---|---|---|---|---|
| Tfl-GCN4-p1d | Leucine (L) | 3 per Leucine (12 total) | Methyl group, δ-carbon | Residue-specific ncAA incorporation | α-helical coiled-coil | RMKQLEDKVEELLSKNYHLENEVARLKKLVGER | [53] |
| Hfl-GCN4-p1d | Leucine (L) | 6 per Leucine (24 total) | Methyl group, δ-carbon | Residue-specific ncAA incorporation | α-helical coiled-coil | RMKQLEDKVEELLSKNYHLENEVARLKKLVGER | [53] |
| TFL-Protein A1 | Leucine (L) | 3 per Leucine (24 total) | Methyl group, δ-carbon | Residue-specific ncAA incorporation | α-helical coiled-coil | MRGSHHHHHHGSASGDLENE VAQLEREVR SLEDEAAELEQKVSRLKNEIEDLKAEIGDLNNTSGIRRPAAKLN | [54] |
| HFL-Protein A1 | Leucine (L) | 6 per Leucine (48 total) | Methyl group, δ-carbon | Residue-specific ncAA incorporation | α-helical coiled-coil | MRGSHHHHHHGSASGDLENE VAQLEREVR SLEDEAAELEQKVSRLKNEIEDLKAEIGDLNNTSGIRRPAAKLN | [55] |
| C+TFL | Leucine (L) | 3 per Leucine (21 total) | Methyl group, δ-carbon | Residue-specific ncAAincorporation | α-helical coiled-coil | MRGSHHHHHHGSIEGRAPQMLRELQETNAALQDVRELLRQQVKEITFLKNTSKL | [56] |
| Q+TFL | Leucine (L) | 3 per Leucine (21 total) | Methyl group, δ-carbon | Residue-specific ncAA incorporation | α-helical coiled-coil | MRGSHHHHHHGSIEGRVKEITFLKNTAPQMLRELQETNAALQDVRELLRQQSKL | [56] |
| Peptide 2 (GCN4 modified coiled-coil region) | Leucine (L) Valine (V) | 3 per L or V (21 total) | Methyl group, δ-carbon | SPPS | α-helical coiled-coil | HNRMKQLEDKVEELLSKNASLEYEVARLKKLVGE | [57] |
| (flpProGly)7 collagen triple helix | Proline (P) | 1 per P (7 total) | Pyrrolidine ring, 4′-carbon | SPPS | Proline Type II Collagen triple helix | PhPGPhPGPhPGPhPGPhPGPhPGPhPG | [58] |
| (GlyProHyp)10 collagen model peptide | Phenylalanine (F) | 5 per F (10 total) | Aryl ring, all positions | SPPS | Proline Type II Collagen triple helix | FGPhPGPhPGPhPGPhPGPhPGPhPGPhPGPhPGPhPGPhPF | [58] |
| 19F-Trp-labelled-BsCspB | Tryptophan (W) | 1 per W (1 total) | Aryl ring, 4-, 5-, or 6-carbon | Residue-specific ncAA incorporation | β-barrel | MLEGKVKWFNSEKGFGFIEVEGQDDVFVHFSAIQGEGFKTLEEGQAVSFEIVEGNRGPQAANVTKEA | [59] |
| 19F-Phe-labelled-BsCspB | Phenylalanine (F) | 1 per F (7 total) | Aryl ring, 2-ortho, 3-meta, or 4-para-position | Residue-specific ncAA incorporation | β-barrel | MLEGKVKWFNSEKGFGFIEVEGQDDVFVHFSAIQGEGFKTLEEGQAVSFEIVEGNRGPQAANVTKEA | [59] |
| pFF-EC | Phenylalanine (F) | 1 per F (2 total) | Aryl ring, 4-para-position | Residue-specific ncAA incorporation | Block Copolymer: Disordered elastin-like polypeptide connected to coiled coil | MRGSHHHHHHHGSKPIAASA[(VPGVG)2VPGFG(VPGVG)2]5VPLEGSELAATATATATATATAACGDLAPQMLRELQETNAALQDVRELLRQQVKEITFLKNTVMESDASGLQAATATATATATATAVDLQPS | [60] |
| pFF-CE | Phenylalanine (F) | 1 per F (2 total) | Aryl ring, 4-para-position | Residue-specific ncAA incorporation | Block Copolymer: Disordered elastin-like polypeptide connected to coiled coil | MRGSHHHHHHHGSACELAATATATATATATAACGDLAPQMLRELQETNAALQDVRELLRQQVKEITFLKNTVMESDASGLQAATATATATATATAVDKPIAASA[(VPGVG)2VPGFG(VPGVG)2]5LEGSGTGAKLN | [60] |
| pFF-ECE | Phenylalanine (F) | 1 per F (3 total) | Aryl ring, 4-para-position | Residue-specific ncAA incorporation | Block Copolymer: Disordered elastin-like polypeptide connected to coiled coil | MRGSHHHHHHHGSKPIAASA[(VPGVG)2VPGFG(VPGVG)2]5LEGSELAATATATATATATAACGDLAPQMLRELQETNAALQDVRELLRQQVKEITFLKNTVMESDASGLQAATATATATATATAVDKPIAASA[(VPGVG)2VPGFG(VPGVG)2]5LEGSGTGAKLN | [60] |
| Construct Name | Fluorinated Residue(s) | Number of Fluorines | Position of Fluorines | Synthetic Method | Assembly Morphology | Sequence | References |
|---|---|---|---|---|---|---|---|
| C7K2 | C-terminus | 13 per endcap (13 total) | C-terminal endcap | SPPS | Ribbon-like/cylindrical peptide amphiphiles | KKAAVV-CO-CF2-CF2- CF2-CF2-CF2-CF3 | [103,104] |
| C7E2 | C-terminus | 11 per endcap (11 total) | C-terminal endcap | SPPS | Ribbon-like/cylindrical peptide amphiphiles | EEAAVV-CO-CF2-CF2- CF2-CF2-CF2-CF3 | [103,104] |
| C8K2 | C-terminus | 15 per endcap (15 total) | C-terminal endcap | SPPS | Ribbon-like/cylindrical peptide amphiphiles | KKAAVV-CO-CF2-CF2- CF2-CF2-CF2-CF2-CF3 | [103] |
| C8E2 | C-terminus | 13 per endcap (13 total) | C-terminal endcap | SPPS | Ribbon-like/cylindrical peptide amphiphiles | EEAAVV-CO-CF2-CF2- CF2-CF2-CF2-CF2-CF3 | [103] |
| C7E3 | C-terminus | 11 per endcap (11 total) | C-terminal endcap | SPPS | Ribbon-like/cylindrical peptide amphiphiles | EEEAAVV-CO-CF2-CF2- CF2-CF2-CF2-CF3 | [103] |
| Compound 1 | Lysine (K) | 6 per FMBA-modified K (6 total) | Modified side chain aryl ring (3′ and 5′ positions) | SPPS | Linear Peptide | CDQVDFMBAKC | [105] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Monkovic, J.M.; Gibson, H.; Sun, J.W.; Montclare, J.K. Fluorinated Protein and Peptide Materials for Biomedical Applications. Pharmaceuticals 2022, 15, 1201. https://doi.org/10.3390/ph15101201
Monkovic JM, Gibson H, Sun JW, Montclare JK. Fluorinated Protein and Peptide Materials for Biomedical Applications. Pharmaceuticals. 2022; 15(10):1201. https://doi.org/10.3390/ph15101201
Chicago/Turabian StyleMonkovic, Julia M., Halle Gibson, Jonathan W. Sun, and Jin Kim Montclare. 2022. "Fluorinated Protein and Peptide Materials for Biomedical Applications" Pharmaceuticals 15, no. 10: 1201. https://doi.org/10.3390/ph15101201
APA StyleMonkovic, J. M., Gibson, H., Sun, J. W., & Montclare, J. K. (2022). Fluorinated Protein and Peptide Materials for Biomedical Applications. Pharmaceuticals, 15(10), 1201. https://doi.org/10.3390/ph15101201






