Phytoestrogens for the Management of Endometriosis: Findings and Issues
Abstract
1. Introduction
2. Estrogen as a Dominant Pathogenic Factor for Endometriosis
3. Phytoestrogens from Food Consumption
3.1. Categories and Contents of Phytoestrogens from Diet Sources
3.2. Bioavailability of Phytoestrogens
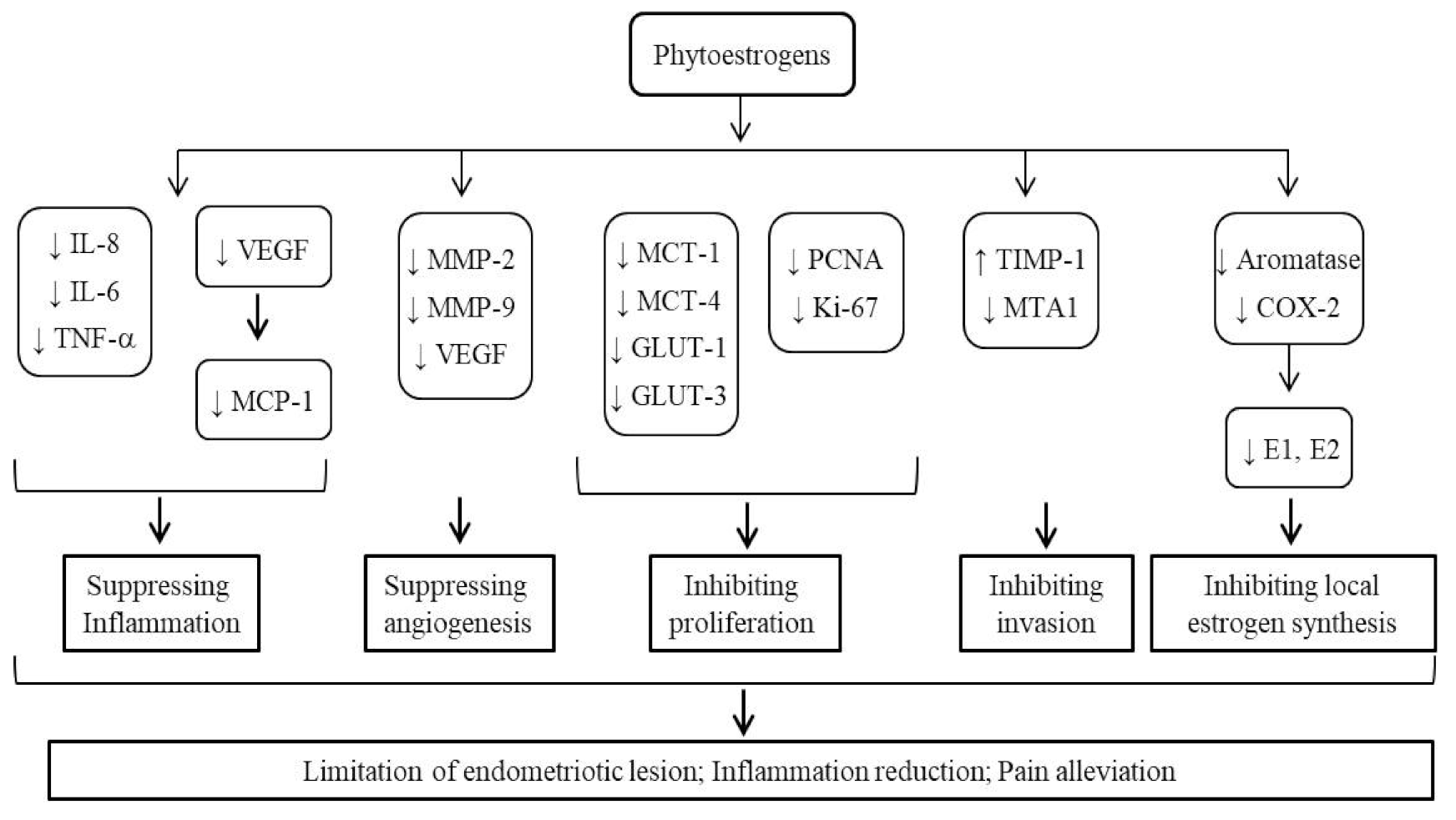
3.3. Bioactivities of Phytoestrogens
4. Uncertain Relationship between Phytoestrogen Intake and Endometriosis Risk
5. Phytoestrogens and Endometriosis
5.1. Resveratrol
5.1.1. Findings from In Vitro Studies
5.1.2. Findings from Rodent Study Models
5.1.3. Findings from Human Population
5.2. Isoflavones
5.2.1. Findings from In Vitro Studies
5.2.2. Findings from Rodent Study Models
5.2.3. Findings from Human Population
5.3. Puerarin
6. Critical Issues Concerning Studies of Phytoestrogens’ Effects on Endometriosis
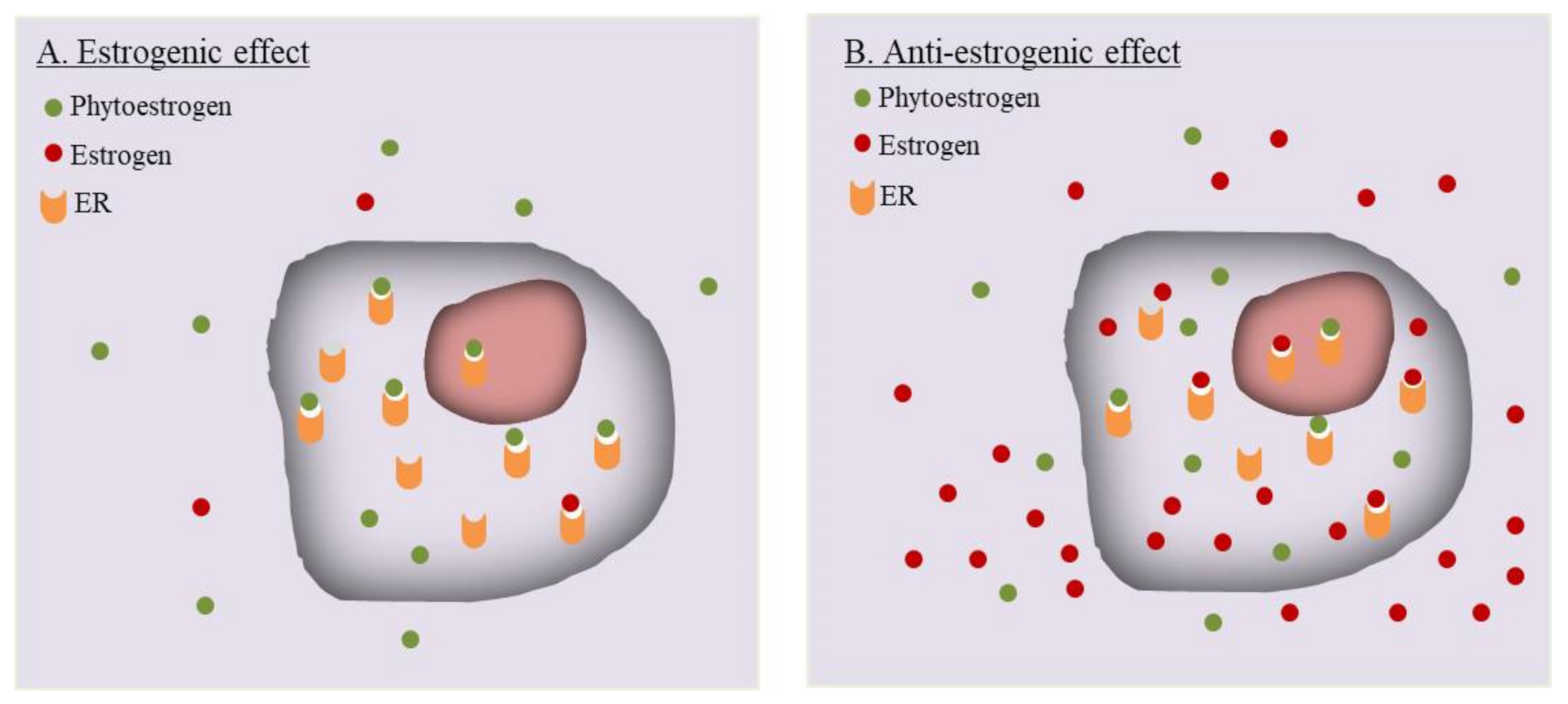
6.1. Estrogenic or Anti-Estrogenic? A Mechanistic Dilemma of Phytoestrogens
6.2. Dose-Dependent and Phytoestrogen Type-Specific Responses
6.3. Application of Appropriate Doses for Phytoestrogen Studies
6.4. Limitations of Current Animal Models
6.5. Proper Design of Intervention Studies on Phytoestrogens
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zondervan, K.T.; Becker, C.M.; Koga, K.; Missmer, S.A.; Taylor, R.N.; Viganò, P. Endometriosis. Nat. Rev. Dis. Primers 2018, 4, 9. [Google Scholar] [CrossRef] [PubMed]
- Bulun, S.E.; Yilmaz, B.D.; Sison, C.; Miyazaki, K.; Bernardi, L.; Liu, S.; Kohlmeier, A.; Yin, P.; Milad, M.; Wei, J. Endometriosis. Endocr. Rev. 2019, 40, 1048–1079. [Google Scholar] [CrossRef] [PubMed]
- Chantalat, E.; Valera, M.C.; Vaysse, C.; Noirrit, E.; Rusidze, M.; Weyl, A.; Vergriete, K.; Buscail, E.; Lluel, P.; Fontaine, C.; et al. Estrogen Receptors and Endometriosis. Int. J. Mol. Sci. 2020, 21, 2815. [Google Scholar] [CrossRef] [PubMed]
- Shafrir, A.L.; Farland, L.V.; Shah, D.K.; Harris, H.R.; Kvaskoff, M.; Zondervan, K.; Missmer, S.A. Risk for and consequences of endometriosis: A critical epidemiologic review. Best Pract. Res. Clin. Obstet. Gynaecol. 2018, 51, 1–15. [Google Scholar] [CrossRef]
- Zondervan, K.T.; Becker, C.M.; Missmer, S.A. Endometriosis. N. Engl. J. Med. 2020, 382, 1244–1256. [Google Scholar] [CrossRef]
- Greene, A.D.; Lang, S.A.; Kendziorski, J.A.; Sroga-Rios, J.M.; Herzog, T.J.; Burns, K.A. Endometriosis: Where are we and where are we going? Reproduction 2016, 152, R63–R78. [Google Scholar] [CrossRef]
- Kolahdouz-Mohammadi, R.; Shidfar, F.; Khodaverdi, S.; Arablou, T.; Heidari, S.; Rashidi, N.; Delbandi, A.A. Resveratrol treatment reduces expression of MCP-1, IL-6, IL-8 and RANTES in endometriotic stromal cells. J. Cell. Mol. Med. 2021, 25, 1116–1127. [Google Scholar] [CrossRef]
- Kim, H.S.; Kim, T.H.; Chung, H.H.; Song, Y.S. Risk and prognosis of ovarian cancer in women with endometriosis: A meta-analysis. Br. J. Cancer 2014, 110, 1878–1890. [Google Scholar] [CrossRef]
- Lee, A.W.; Templeman, C.; Stram, D.A.; Beesley, J.; Tyrer, J.; Berchuck, A.; Pharoah, P.P.; Chenevix-Trench, G.; Pearce, C.L. Evidence of a genetic link between endometriosis and ovarian cancer. Fertil. Steril. 2016, 105, 35–43. [Google Scholar] [CrossRef]
- Kajiyama, H.; Suzuki, S.; Yoshihara, M.; Tamauchi, S.; Yoshikawa, N.; Niimi, K.; Shibata, K.; Kikkawa, F. Endometriosis and cancer. Free Radic. Biol. Med. 2019, 133, 186–192. [Google Scholar] [CrossRef]
- Fuldeore, M.J.; Soliman, A.M. Prevalence and Symptomatic Burden of Diagnosed Endometriosis in the United States: National Estimates from a Cross-Sectional Survey of 59,411 Women. Gynecol. Obstet. Investig. 2017, 82, 453–461. [Google Scholar] [CrossRef]
- Rietjens, I.; Louisse, J.; Beekmann, K. The potential health effects of dietary phytoestrogens. Br. J. Pharmacol. 2017, 174, 1263–1280. [Google Scholar] [CrossRef]
- Rolla, E. Endometriosis: Advances and controversies in classification, pathogenesis, diagnosis, and treatment. F1000Research 2019, 8, 529. [Google Scholar] [CrossRef]
- Laganà, A.S.; Triolo, O.; Salmeri, F.M.; Granese, R.; Palmara, V.I.; Ban Frangež, H.; Vrtčnik Bokal, E.; Sofo, V. Natural Killer T cell subsets in eutopic and ectopic endometrium: A fresh look to a busy corner. Arch. Gynecol. Obstet. 2016, 293, 941–949. [Google Scholar] [CrossRef]
- Laganà, A.S.; Salmeri, F.M.; Vitale, S.G.; Triolo, O.; Götte, M. Stem Cell Trafficking During Endometriosis: May Epigenetics Play a Pivotal Role? Reprod. Sci. 2018, 25, 978–979. [Google Scholar] [CrossRef]
- Smuc, T.; Pucelj, M.R.; Sinkovec, J.; Husen, B.; Thole, H.; Lanisnik Rizner, T. Expression analysis of the genes involved in estradiol and progesterone action in human ovarian endometriosis. Gynecol. Endocrinol. 2007, 23, 105–111. [Google Scholar] [CrossRef]
- Mori, T.; Ito, F.; Koshiba, A.; Kataoka, H.; Takaoka, O.; Okimura, H.; Khan, K.N.; Kitawaki, J. Local estrogen formation and its regulation in endometriosis. Reprod. Med. Biol. 2019, 18, 305–311. [Google Scholar] [CrossRef]
- Huhtinen, K.; Desai, R.; Ståhle, M.; Salminen, A.; Handelsman, D.J.; Perheentupa, A.; Poutanen, M. Endometrial and endometriotic concentrations of estrone and estradiol are determined by local metabolism rather than circulating levels. J. Clin. Endocrinol. Metab. 2012, 97, 4228–4235. [Google Scholar] [CrossRef]
- Gonçalves, H.F.; Zendron, C.; Cavalcante, F.S.; Aiceles, V.; Oliveira, M.A.; Manaia, J.H.; Babinski, M.A.; Ramos, C.F. Leptin, its receptor and aromatase expression in deep infiltrating endometriosis. J. Ovarian Res. 2015, 8, 53. [Google Scholar] [CrossRef]
- Xu, Z.; Zhang, L.; Yu, Q.; Zhang, Y.; Yan, L.; Chen, Z.J. The estrogen-regulated lncRNA H19/miR-216a-5p axis alters stromal cell invasion and migration via ACTA2 in endometriosis. Mol. Hum. Reprod. 2019, 25, 550–561. [Google Scholar] [CrossRef]
- Qi, S.; Yan, L.; Liu, Z.; Mu, Y.L.; Li, M.; Zhao, X.; Chen, Z.J.; Zhang, H. Melatonin inhibits 17β-estradiol-induced migration, invasion and epithelial-mesenchymal transition in normal and endometriotic endometrial epithelial cells. Reprod. Biol. Endocrinol. 2018, 16, 62. [Google Scholar] [CrossRef]
- Hudelist, G.; Keckstein, J.; Czerwenka, K.; Lass, H.; Walter, I.; Auer, M.; Wieser, F.; Wenzl, R.; Kubista, E.; Singer, C.F. Estrogen receptor beta and matrix metalloproteinase 1 are coexpressed in uterine endometrium and endometriotic lesions of patients with endometriosis. Fertil. Steril. 2005, 84 (Suppl. 2), 1249–1256. [Google Scholar] [CrossRef]
- Bukulmez, O.; Hardy, D.B.; Carr, B.R.; Word, R.A.; Mendelson, C.R. Inflammatory status influences aromatase and steroid receptor expression in endometriosis. Endocrinology 2008, 149, 1190–1204. [Google Scholar] [CrossRef]
- Tang, Z.R.; Zhang, R.; Lian, Z.X.; Deng, S.L.; Yu, K. Estrogen-Receptor Expression and Function in Female Reproductive Disease. Cells 2019, 8, 1123. [Google Scholar] [CrossRef]
- Xue, Q.; Lin, Z.; Cheng, Y.H.; Huang, C.C.; Marsh, E.; Yin, P.; Milad, M.P.; Confino, E.; Reierstad, S.; Innes, J.; et al. Promoter methylation regulates estrogen receptor 2 in human endometrium and endometriosis. Biol. Reprod. 2007, 77, 681–687. [Google Scholar] [CrossRef]
- Tamura, M.; Deb, S.; Sebastian, S.; Okamura, K.; Bulun, S.E. Estrogen up-regulates cyclooxygenase-2 via estrogen receptor in human uterine microvascular endothelial cells. Fertil. Steril. 2004, 81, 1351–1356. [Google Scholar] [CrossRef]
- Jiang, L.; Yan, Y.; Liu, Z.; Wang, Y. Inflammation and endometriosis. Front. Biosci. 2016, 21, 941–948. [Google Scholar] [CrossRef]
- Dull, A.M.; Moga, M.A.; Dimienescu, O.G.; Sechel, G.; Burtea, V.; Anastasiu, C.V. Therapeutic Approaches of Resveratrol on Endometriosis via Anti-Inflammatory and Anti-Angiogenic Pathways. Molecules 2019, 24, 667. [Google Scholar] [CrossRef] [PubMed]
- Park, G.Y.; Christman, J.W. Involvement of cyclooxygenase-2 and prostaglandins in the molecular pathogenesis of inflammatory lung diseases. Am. J. Physiol. Lung Cell. Mol. Physiol. 2006, 290, L797–L805. [Google Scholar] [CrossRef]
- Harris, R.C.; Zhang, M.Z. Cyclooxygenase metabolites in the kidney. Compr. Physiol. 2011, 1, 1729–1758. [Google Scholar] [CrossRef] [PubMed]
- Zidan, H.E.; Rezk, N.A.; Alnemr, A.A.; Abd El Ghany, A.M. COX-2 gene promoter DNA methylation status in eutopic and ectopic endometrium of Egyptian women with endometriosis. J. Reprod. Immunol. 2015, 112, 63–67. [Google Scholar] [CrossRef]
- Cho, S.; Park, S.H.; Choi, Y.S.; Seo, S.K.; Kim, H.Y.; Park, K.H.; Cho, D.J.; Lee, B.S. Expression of cyclooxygenase-2 in eutopic endometrium and ovarian endometriotic tissue in women with severe endometriosis. Gynecol. Obstet. Investig. 2010, 69, 93–100. [Google Scholar] [CrossRef]
- Bulun, S.E.; Lin, Z.; Imir, G.; Amin, S.; Demura, M.; Yilmaz, B.; Martin, R.; Utsunomiya, H.; Thung, S.; Gurates, B.; et al. Regulation of aromatase expression in estrogen-responsive breast and uterine disease: From bench to treatment. Pharmacol. Rev. 2005, 57, 359–383. [Google Scholar] [CrossRef]
- Ayubi, E.; Safiri, S. Dietary isoflavone intake and all-cause mortality in breast cancer survivors: The Breast Cancer Family Registry-methodological issues. Cancer 2017, 123, 3638–3639. [Google Scholar] [CrossRef]
- Wang, T.; Wang, Y.; Zhuang, X.; Luan, F.; Zhao, C.; Cordeiro, M. Interaction of Coumarin Phytoestrogens with ER(α) and ER(β): A Molecular Dynamics Simulation Study. Molecules 2020, 25, 1165. [Google Scholar] [CrossRef]
- Riboli, E.; Kaaks, R. The EPIC Project: Rationale and study design. European Prospective Investigation into Cancer and Nutrition. Int. J. Epidemiol. 1997, 26 (Suppl. 1), S6–S14. [Google Scholar] [CrossRef]
- Riboli, E.; Hunt, K.J.; Slimani, N.; Ferrari, P.; Norat, T.; Fahey, M.; Charrondière, U.R.; Hémon, B.; Casagrande, C.; Vignat, J.; et al. European Prospective Investigation into Cancer and Nutrition (EPIC): Study populations and data collection. Public Health Nutr. 2002, 5, 1113–1124. [Google Scholar] [CrossRef]
- Zamora-Ros, R.; Knaze, V.; Luján-Barroso, L.; Kuhnle, G.G.; Mulligan, A.A.; Touillaud, M.; Slimani, N.; Romieu, I.; Powell, N.; Tumino, R.; et al. Dietary intakes and food sources of phytoestrogens in the European Prospective Investigation into Cancer and Nutrition (EPIC) 24-hour dietary recall cohort. Eur. J. Clin. Nutr. 2012, 66, 932–941. [Google Scholar] [CrossRef]
- Bedell, S.; Nachtigall, M.; Naftolin, F. The pros and cons of plant estrogens for menopause. J. Steroid Biochem. Mol. Biol. 2014, 139, 225–236. [Google Scholar] [CrossRef]
- Wang, H.J.; Murphy, P.A. Isoflavone Composition of American and Japanese Soybeans in Iowa: Effects of Variety, Crop Year, and Location. J. Agric. Food Chem. 1993, 42, 1674–1677. [Google Scholar] [CrossRef]
- Viggiani, M.T.; Polimeno, L.; Di Leo, A.; Barone, M. Phytoestrogens: Dietary Intake, Bioavailability, and Protective Mechanisms against Colorectal Neoproliferative Lesions. Nutrients 2019, 11, 1709. [Google Scholar] [CrossRef]
- Arai, Y.; Uehara, M.; Sato, Y.; Kimira, M.; Eboshida, A.; Adlercreutz, H.; Watanabe, S. Comparison of isoflavones among dietary intake, plasma concentration and urinary excretion for accurate estimation of phytoestrogen intake. J. Epidemiol. 2000, 10, 127–135. [Google Scholar] [CrossRef]
- Setchell, K.D.; Brown, N.M.; Desai, P.; Zimmer-Nechemias, L.; Wolfe, B.E.; Brashear, W.T.; Kirschner, A.S.; Cassidy, A.; Heubi, J.E. Bioavailability of pure isoflavones in healthy humans and analysis of commercial soy isoflavone supplements. J. Nutr. 2001, 131, 1362s–1375s. [Google Scholar] [CrossRef] [PubMed]
- Glazier, M.G.; Bowman, M.A. A review of the evidence for the use of phytoestrogens as a replacement for traditional estrogen replacement therapy. Arch. Intern. Med. 2001, 161, 1161–1172. [Google Scholar] [CrossRef]
- DeCosse, J.J.; Ngoi, S.S.; Jacobson, J.S.; Cennerazzo, W.J. Gender and colorectal cancer. Eur. J. Cancer Prev. 1993, 2, 105–115. [Google Scholar] [CrossRef]
- Dominguez-Lopez, I.; Yago-Aragon, M.; Salas-Huetos, A.; Tresserra-Rimbau, A.; Hurtado-Barroso, S. Effects of Dietary Phytoestrogens on Hormones throughout a Human Lifespan: A Review. Nutrients 2020, 12, 2456. [Google Scholar] [CrossRef]
- Kuiper, G.G.; Lemmen, J.G.; Carlsson, B.; Corton, J.C.; Safe, S.H.; van der Saag, P.T.; van der Burg, B.; Gustafsson, J.A. Interaction of estrogenic chemicals and phytoestrogens with estrogen receptor beta. Endocrinology 1998, 139, 4252–4263. [Google Scholar] [CrossRef]
- Kostelac, D.; Rechkemmer, G.; Briviba, K. Phytoestrogens modulate binding response of estrogen receptors alpha and beta to the estrogen response element. J. Agric. Food Chem. 2003, 51, 7632–7635. [Google Scholar] [CrossRef]
- Křížová, L.; Dadáková, K.; Kašparovská, J.; Kašparovský, T. Isoflavones. Molecules 2019, 24, 1076. [Google Scholar] [CrossRef]
- Morito, K.; Hirose, T.; Kinjo, J.; Hirakawa, T.; Okawa, M.; Nohara, T.; Ogawa, S.; Inoue, S.; Muramatsu, M.; Masamune, Y. Interaction of phytoestrogens with estrogen receptors alpha and beta. Biol. Pharm. Bull. 2001, 24, 351–356. [Google Scholar] [CrossRef]
- Saczko, J.; Michel, O.; Chwiłkowska, A.; Sawicka, E.; Mączyńska, J.; Kulbacka, J. Estrogen Receptors in Cell Membranes: Regulation and Signaling. Adv. Anat. Embryol. Cell Biol. 2017, 227, 93–105. [Google Scholar] [CrossRef] [PubMed]
- Scherbakov, A.M.; Andreeva, O.E. Apigenin Inhibits Growth of Breast Cancer Cells: The Role of ERα and HER2/neu. Acta Nat. 2015, 7, 133–139. [Google Scholar] [CrossRef]
- Edmunds, K.M.; Holloway, A.C.; Crankshaw, D.J.; Agarwal, S.K.; Foster, W.G. The effects of dietary phytoestrogens on aromatase activity in human endometrial stromal cells. Reprod. Nutr. Dev. 2005, 45, 709–720. [Google Scholar] [CrossRef]
- Arispe, S.A.; Adams, B.; Adams, T.E. Effect of phytoestrogens on basal and GnRH-induced gonadotropin secretion. J. Endocrinol. 2013, 219, 243–250. [Google Scholar] [CrossRef][Green Version]
- Youseflu, S.; Jahanian Sadatmahalleh, S.H.; Mottaghi, A.; Kazemnejad, A. Dietary Phytoestrogen Intake and The Risk of Endometriosis in Iranian Women: A Case-Control Study. Int. J. Fertil. Steril. 2020, 13, 296–300. [Google Scholar] [CrossRef]
- Tsuchiya, M.; Miura, T.; Hanaoka, T.; Iwasaki, M.; Sasaki, H.; Tanaka, T.; Nakao, H.; Katoh, T.; Ikenoue, T.; Kabuto, M.; et al. Effect of soy isoflavones on endometriosis: Interaction with estrogen receptor 2 gene polymorphism. Epidemiology 2007, 18, 402–408. [Google Scholar] [CrossRef]
- Chandrareddy, A.; Muneyyirci-Delale, O.; McFarlane, S.I.; Murad, O.M. Adverse effects of phytoestrogens on reproductive health: A report of three cases. Complementary Ther. Clin. Pract. 2008, 14, 132–135. [Google Scholar] [CrossRef]
- Mumford, S.L.; Weck, J.; Kannan, K.; Buck Louis, G.M. Urinary Phytoestrogen Concentrations Are Not Associated with Incident Endometriosis in Premenopausal Women. J. Nutr. 2017, 147, 227–234. [Google Scholar] [CrossRef]
- Lampe, J.W. Isoflavonoid and lignan phytoestrogens as dietary biomarkers. J. Nutr. 2003, 133 (Suppl. 3), 956s–964s. [Google Scholar] [CrossRef]
- Lampe, J.W.; Gustafson, D.R.; Hutchins, A.M.; Martini, M.C.; Li, S.; Wähälä, K.; Grandits, G.A.; Potter, J.D.; Slavin, J.L. Urinary isoflavonoid and lignan excretion on a Western diet: Relation to soy, vegetable, and fruit intake. Cancer Epidemiol. Biomark. Prev. 1999, 8, 699–707. [Google Scholar]
- Rauf, A.; Imran, M.; Butt, M.S.; Nadeem, M.; Peters, D.G.; Mubarak, M.S. Resveratrol as an anti-cancer agent: A review. Crit. Rev. Food Sci. Nutr. 2018, 58, 1428–1447. [Google Scholar] [CrossRef]
- Soleas, G.J.; Diamandis, E.P.; Goldberg, D.M. Wine as a biological fluid: History, production, and role in disease prevention. J. Clin. Lab. Anal. 1997, 11, 287–313. [Google Scholar] [CrossRef]
- Nakata, R.; Takahashi, S.; Inoue, H. Recent advances in the study on resveratrol. Biol. Pharm. Bull. 2012, 35, 273–279. [Google Scholar] [CrossRef]
- Arablou, T.; Delbandi, A.A.; Khodaverdi, S.; Arefi, S.; Kolahdouz-Mohammadi, R.; Heidari, S.; Mohammadi, T.; Aryaeian, N. Resveratrol reduces the expression of insulin-like growth factor-1 and hepatocyte growth factor in stromal cells of women with endometriosis compared with nonendometriotic women. Phytother. Res. 2019, 33, 1044–1054. [Google Scholar] [CrossRef] [PubMed]
- Villanueva, J.A.; Sokalska, A.; Cress, A.B.; Ortega, I.; Bruner-Tran, K.L.; Osteen, K.G.; Duleba, A.J. Resveratrol potentiates effect of simvastatin on inhibition of mevalonate pathway in human endometrial stromal cells. J. Clin. Endocrinol. Metab. 2013, 98, E455–E462. [Google Scholar] [CrossRef] [PubMed]
- Bruner-Tran, K.L.; Osteen, K.G.; Taylor, H.S.; Sokalska, A.; Haines, K.; Duleba, A.J. Resveratrol inhibits development of experimental endometriosis in vivo and reduces endometrial stromal cell invasiveness in vitro. Biol. Reprod. 2011, 84, 106–112. [Google Scholar] [CrossRef]
- Amaya, S.C.; Savaris, R.F.; Filipovic, C.J.; Wise, J.D.; Hestermann, E.; Young, S.L.; Lessey, B.A. Resveratrol and endometrium: A closer look at an active ingredient of red wine using in vivo and in vitro models. Reprod. Sci. 2014, 21, 1362–1369. [Google Scholar] [CrossRef]
- Rudzitis-Auth, J.; Menger, M.D.; Laschke, M.W. Resveratrol is a potent inhibitor of vascularization and cell proliferation in experimental endometriosis. Hum. Reprod. 2013, 28, 1339–1347. [Google Scholar] [CrossRef]
- Ricci, A.G.; Olivares, C.N.; Bilotas, M.A.; Bastón, J.I.; Singla, J.J.; Meresman, G.F.; Barañao, R.I. Natural therapies assessment for the treatment of endometriosis. Hum. Reprod. 2013, 28, 178–188. [Google Scholar] [CrossRef]
- Kong, X.; Xu, X.; Zhou, L.; Zhu, M.; Yao, S.; Ding, Y.; Liu, T.; Wang, Y.; Zhang, Y.; Li, R.; et al. MTA1, a Target of Resveratrol, Promotes Epithelial-Mesenchymal Transition of Endometriosis via ZEB2. Mol. Ther. Methods Clin. Dev. 2020, 19, 295–306. [Google Scholar] [CrossRef]
- Yavuz, S.; Aydin, N.E.; Celik, O.; Yilmaz, E.; Ozerol, E.; Tanbek, K. Resveratrol successfully treats experimental endometriosis through modulation of oxidative stress and lipid peroxidation. J. Cancer Res. Ther. 2014, 10, 324–329. [Google Scholar] [CrossRef] [PubMed]
- Ergenoğlu, A.M.; Yeniel, A.Ö.; Erbaş, O.; Aktuğ, H.; Yildirim, N.; Ulukuş, M.; Taskiran, D. Regression of endometrial implants by resveratrol in an experimentally induced endometriosis model in rats. Reprod. Sci. 2013, 20, 1230–1236. [Google Scholar] [CrossRef] [PubMed]
- Tekin, Y.B.; Guven, S.; Kirbas, A.; Kalkan, Y.; Tumkaya, L.; Guvendag Guven, E.S. Is resveratrol a potential substitute for leuprolide acetate in experimental endometriosis? Eur. J. Obstet. Gynecol. Reprod. Biol. 2015, 184, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Cenksoy, P.O.; Oktem, M.; Erdem, O.; Karakaya, C.; Cenksoy, C.; Erdem, A.; Guner, H.; Karabacak, O. A potential novel treatment strategy: Inhibition of angiogenesis and inflammation by resveratrol for regression of endometriosis in an experimental rat model. Gynecol. Endocrinol. 2015, 31, 219–224. [Google Scholar] [CrossRef]
- Bahrami, A.; Ayen, E.; Razi, M.; Behfar, M. Effects of atorvastatin and resveratrol against the experimental endometriosis; evidence for glucose and monocarboxylate transporters, neoangiogenesis. Life Sci. 2021, 272, 119230. [Google Scholar] [CrossRef]
- Maia, H., Jr.; Haddad, C.; Pinheiro, N.; Casoy, J. Advantages of the association of resveratrol with oral contraceptives for management of endometriosis-related pain. Int. J. Women’s Health 2012, 4, 543–549. [Google Scholar] [CrossRef]
- Mendes da Silva, D.; Gross, L.A.; Neto, E.P.G.; Lessey, B.A.; Savaris, R.F. The Use of Resveratrol as an Adjuvant Treatment of Pain in Endometriosis: A Randomized Clinical Trial. J. Endocr. Soc. 2017, 1, 359–369. [Google Scholar] [CrossRef]
- Kodarahmian, M.; Amidi, F.; Moini, A.; Kashani, L.; Shabani Nashtaei, M.; Pazhohan, A.; Bahramrezai, M.; Berenjian, S.; Sobhani, A. The modulating effects of Resveratrol on the expression of MMP-2 and MMP-9 in endometriosis women: A randomized exploratory trial. Gynecol. Endocrinol. Off. J. Int. Soc. Gynecol. Endocrinol. 2019, 35, 719–726. [Google Scholar] [CrossRef]
- Khodarahmian, M.; Amidi, F.; Moini, A.; Kashani, L.; Salahi, E.; Danaii-Mehrabad, S.; Nashtaei, M.S.; Mojtahedi, M.F.; Esfandyari, S.; Sobhani, A. A randomized exploratory trial to assess the effects of resveratrol on VEGF and TNF-α 2 expression in endometriosis women. J. Reprod. Immunol. 2021, 143, 103248. [Google Scholar] [CrossRef]
- Zaheer, K.; Humayoun Akhtar, M. An updated review of dietary isoflavones: Nutrition, processing, bioavailability and impacts on human health. Crit. Rev. Food Sci. Nutr. 2017, 57, 1280–1293. [Google Scholar] [CrossRef]
- Ho, H.M.; Chen, R.Y.; Leung, L.K.; Chan, F.L.; Huang, Y.; Chen, Z.Y. Difference in flavonoid and isoflavone profile between soybean and soy leaf. Biomed. Pharmacother. 2002, 56, 289–295. [Google Scholar] [CrossRef]
- Zhang, X.; Gao, Y.T.; Yang, G.; Li, H.; Cai, Q.; Xiang, Y.B.; Ji, B.T.; Franke, A.A.; Zheng, W.; Shu, X.O. Urinary isoflavonoids and risk of coronary heart disease. Int. J. Epidemiol. 2012, 41, 1367–1375. [Google Scholar] [CrossRef]
- Wei, P.; Liu, M.; Chen, Y.; Chen, D.C. Systematic review of soy isoflavone supplements on osteoporosis in women. Asian Pac. J. Trop. Med. 2012, 5, 243–248. [Google Scholar] [CrossRef]
- Neese, S.L.; Bandara, S.B.; Doerge, D.R.; Helferich, W.G.; Korol, D.L.; Schantz, S.L. Effects of multiple daily genistein treatments on delayed alternation and a differential reinforcement of low rates of responding task in middle-aged rats. Neurotoxicol. Teratol. 2012, 34, 187–195. [Google Scholar] [CrossRef]
- Salsano, S.; Pérez-Debén, S.; Quiñonero, A.; González-Martín, R.; Domínguez, F. Phytoestrogen exposure alters endometrial stromal cells and interferes with decidualization signaling. Fertil. Steril. 2019, 112, 947–958. [Google Scholar] [CrossRef]
- Srisomboon, Y.; Poonyachoti, S.; Deachapunya, C. Soy isoflavones enhance β-defensin synthesis and secretion in endometrial epithelial cells with exposure to TLR3 agonist polyinosinic-polycytidylic acid. Am. J. Reprod. Immunol. 2017, 78, e12694. [Google Scholar] [CrossRef]
- Ronis, M.J.; Gomez-Acevedo, H.; Blackburn, M.L.; Cleves, M.A.; Singhal, R.; Badger, T.M. Uterine responses to feeding soy protein isolate and treatment with 17bera-estradiol differ in ovariectomized female rats. Toxicol. Appl. Pharmacol. 2016, 297, 68–80. [Google Scholar] [CrossRef]
- Takaoka, O.; Mori, T.; Ito, F.; Okimura, H.; Kataoka, H.; Tanaka, Y.; Koshiba, A.; Kusuki, I.; Shigehiro, S.; Amami, T.; et al. Daidzein-rich isoflavone aglycones inhibit cell growth and inflammation in endometriosis. J. Steroid Biochem. Mol. Biol. 2018, 181, 125–132. [Google Scholar] [CrossRef]
- Sutrisno, S.; Aprina, H.; Simanungkalit, H.M.; Andriyani, A.; Barlianto, W.; Sujuti, H.; Santoso, S.; Dwijayasa, P.M.; Wahyuni, E.S.; Mustofa, E. Genistein modulates the estrogen receptor and suppresses angiogenesis and inflammation in the murine model of peritoneal endometriosis. J. Tradit. Complementary Med. 2018, 8, 278–281. [Google Scholar] [CrossRef]
- Yavuz, E.; Oktem, M.; Esinler, I.; Toru, S.A.; Zeyneloglu, H.B. Genistein causes regression of endometriotic implants in the rat model. Fertil. Steril. 2007, 88, 1129–1134. [Google Scholar] [CrossRef]
- Cotroneo, M.S.; Lamartiniere, C.A. Pharmacologic, but not dietary, genistein supports endometriosis in a rat model. Toxicol. Sci. 2001, 61, 68–75. [Google Scholar] [CrossRef] [PubMed]
- Duncan, A.M.; Underhill, K.E.; Xu, X.; Lavalleur, J.; Phipps, W.R.; Kurzer, M.S. Modest hormonal effects of soy isoflavones in postmenopausal women. J. Clin. Endocrinol. Metab. 1999, 84, 3479–3484. [Google Scholar] [CrossRef] [PubMed]
- Murray, M.J.; Meyer, W.R.; Lessey, B.A.; Oi, R.H.; DeWire, R.E.; Fritz, M.A. Soy protein isolate with isoflavones does not prevent estradiol-induced endometrial hyperplasia in postmenopausal women: A pilot trial. Menopause 2003, 10, 456–464. [Google Scholar] [CrossRef] [PubMed]
- Hale, G.E.; Hughes, C.L.; Robboy, S.J.; Agarwal, S.K.; Bievre, M. A double-blind randomized study on the effects of red clover isoflavones on the endometrium. Menopause 2001, 8, 338–346. [Google Scholar] [CrossRef]
- Quaas, A.M.; Kono, N.; Mack, W.J.; Hodis, H.N.; Felix, J.C.; Paulson, R.J.; Shoupe, D. Effect of isoflavone soy protein supplementation on endometrial thickness, hyperplasia, and endometrial cancer risk in postmenopausal women: A randomized controlled trial. Menopause 2013, 20, 840–844. [Google Scholar] [CrossRef]
- Unfer, V.; Casini, M.L.; Costabile, L.; Mignosa, M.; Gerli, S.; Di Renzo, G.C. Endometrial effects of long-term treatment with phytoestrogens: A randomized, double-blind, placebo-controlled study. Fertil. Steril. 2004, 82, 145–148. [Google Scholar] [CrossRef]
- Chen, X.; Yu, J.; Shi, J. Management of Diabetes Mellitus with Puerarin, a Natural Isoflavone From Pueraria lobata. Am. J. Chin. Med. 2018, 46, 1771–1789. [Google Scholar] [CrossRef]
- Wang, D.; Liu, Y.; Han, J.; Zai, D.; Ji, M.; Cheng, W.; Xu, L.; Yang, L.; He, M.; Ni, J.; et al. Puerarin suppresses invasion and vascularization of endometriosis tissue stimulated by 17β-estradiol. PLoS ONE 2011, 6, e25011. [Google Scholar] [CrossRef]
- Cheng, W.; Chen, L.; Yang, S.; Han, J.; Zhai, D.; Ni, J.; Yu, C.; Cai, Z. Puerarin suppresses proliferation of endometriotic stromal cells partly via the MAPK signaling pathway induced by 17ß-estradiol-BSA. PLoS ONE 2012, 7, e45529. [Google Scholar] [CrossRef]
- Chen, Y.; Chen, C.; Shi, S.; Han, J.; Wang, J.; Hu, J.; Liu, Y.; Cai, Z.; Yu, C. Endometriotic implants regress in rat models treated with puerarin by decreasing estradiol level. Reprod. Sci. 2011, 18, 886–891. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Woo, J.H.; Kim, H.M.; Oh, M.S.; Jang, D.S.; Choi, J.H. Anti-Endometriotic Effects of Pueraria Flower Extract in Human Endometriotic Cells and Mice. Nutrients 2017, 9, 212. [Google Scholar] [CrossRef]
- Yu, J.; Zhao, L.; Zhang, D.; Zhai, D.; Shen, W.; Bai, L.; Liu, Y.; Cai, Z.; Li, J.; Yu, C. The effects and possible mechanisms of puerarin to treat endometriosis model rats. Evid. Based Complement. Altern. Med. 2015, 2015, 269138. [Google Scholar] [CrossRef]
- Ferrero, S.; Remorgida, V.; Maganza, C.; Venturini, P.L.; Salvatore, S.; Papaleo, E.; Candiani, M.; Leone Roberti Maggiore, U. Aromatase and endometriosis: Estrogens play a role. Ann. N. Y. Acad. Sci. 2014, 1317, 17–23. [Google Scholar] [CrossRef]
- Baker, M.E.; Lathe, R. The promiscuous estrogen receptor: Evolution of physiological estrogens and response to phytochemicals and endocrine disruptors. J. Steroid Biochem. Mol. Biol. 2018, 184, 29–37. [Google Scholar] [CrossRef]
- Ji, M.; Liu, Y.; Yang, S.; Zhai, D.; Zhang, D.; Bai, L.; Wang, Z.; Yu, J.; Yu, C.; Cai, Z. Puerarin suppresses proliferation of endometriotic stromal cells in part via differential recruitment of nuclear receptor coregulators to estrogen receptor-α. J. Steroid Biochem. Mol. Biol. 2013, 138, 421–426. [Google Scholar] [CrossRef]
- Taguchi, A.; Wada-Hiraike, O.; Kawana, K.; Koga, K.; Yamashita, A.; Shirane, A.; Urata, Y.; Kozuma, S.; Osuga, Y.; Fujii, T. Resveratrol suppresses inflammatory responses in endometrial stromal cells derived from endometriosis: A possible role of the sirtuin 1 pathway. J. Obstet. Gynaecol. Res. 2014, 40, 770–778. [Google Scholar] [CrossRef]
- Sampson, J.A. Peritoneal endometriosis due to the menstrual dissemination of endometrial tissue into the peritoneal cavity. Am. J. Obstet. Gynecol. 1927, 14, 93–94. [Google Scholar] [CrossRef]
- Taguchi, A.; Koga, K.; Kawana, K.; Makabe, T.; Sue, F.; Miyashita, M.; Yoshida, M.; Urata, Y.; Izumi, G.; Tkamura, M.; et al. Resveratrol Enhances Apoptosis in Endometriotic Stromal Cells. Am. J. Reprod. Immunol. 2016, 75, 486–492. [Google Scholar] [CrossRef]
- Reagan-Shaw, S.; Nihal, M.; Ahmad, N. Dose translation from animal to human studies revisited. FASEB J. 2008, 22, 659–661. [Google Scholar] [CrossRef]
- Bruner-Tran, K.L.; Mokshagundam, S.; Herington, J.L.; Ding, T.; Osteen, K.G. Rodent Models of Experimental Endometriosis: Identifying Mechanisms of Disease and Therapeutic Targets. Curr. Women’s Health Rev. 2018, 14, 173–188. [Google Scholar] [CrossRef]
| Study Subjects | Doses/Routs | Findings | References | |
|---|---|---|---|---|
| Isoflavones, lignan, coumestrol, etc. | Infertility women (15–45 y); 78 laparoscopically confirmed endometriosis women and 78 normal controls. | Intake levels estimated based on questionnaire | Intake of phytoestrogen, isoflavones, lignan, coumestrol inversely correlated with endometriosis risk. | [55] |
| Genistein, daidzein | Infertility women (20–45 y); 79 with early or advance stage, 59 normal controls. | Estimated by urine concentration | Urine levels were inversely associated with the risk of advanced, not early stage endometriosis. | [56] |
| Genistein, daidzein, equol, etc. | Women with laparoscopy or laparotomy (18–44 y), 189 with endometriosis, 283 without; Women without operation (18–44 y), 14 with endometriosis, 113 without. | Estimated by urine concentration | Neither comparison found significant difference in urinary concentrations between groups with or without endometriosis. | [58] |
| Phytoestrogens from soy products | 3 women with high intake of soy products. | High intake | High intake was associated with uterine bleeding and endometrial pathology; Withdrawal of soy from diet led to symptoms alleviation. | [57] |
| Resveratrol | Human eutopic ESCs from healthy donors; Oophorectomized mouse endometriosis model, with human endometrial tissues. | 10–30 μmol/L; 6 mg/day, gavage, 10–12 and 18–20 days | Reduced invasiveness of ESCs in a concentration-dependent manner; reduced number and size of endometriotic implants. | [66] |
| Resveratrol | Human ectopic ESCs from ovarian endometriosis cysts; mouse endometriosis model by autotransplantation. | 25 and 50 μmol/L; 25 mg/kg/day, peritoneal injection, 4 weeks. | Suppressing proliferation, migration, and invasion in ESC culture; Inhabiting ectopic tissues’ growth, MTA1 and ZEB2 expression in vivo. | [70] |
| Resveratrol | Human endometrial epithelial cells from endometriosis; mouse endometriotic model, autotransplantion. | 25–100 μmol/L; 10 and 25 mg/kg/day, peritoneal injection, 4 weeks. | Reduced the cell proliferation and increase apoptosis; Reduced the number, volume, vascular density of endometriotic lesions. | [69] |
| Resveratrol | 34 women (18–37 y) with severe endometriosis | 400 mg, twice a day for 12–14 weeks | Decreased VEGF and TNF-α expression in eutopic endometrium. | [79] |
| Genistein, daidzein | Human endometrial stromal cells. | 10, 20, 50, and 100 μmol/L. | Inhibited cell proliferation in dose dependent manner, decreased PRL secretion, and induced in vitro decidualization, | [85] |
| Isoflavone aglycones | Primary cultures from ovarian endometrioma; mouse endometriosis model, allotransplantation. | 0.2–20 μmol/L, dietary supplements. | Inhibited cell proliferation; Inhibited expression of IL-6, IL-8, COX-2, aromatase, TNF-α-induced IκB phosphorylation, p65 transfer to nuclei; reduced serum glucocorticoid-regulated kinase and PGE2 levels. Decreased the number, weight, and Ki-67 activity in endometriosis-like lesions. | [88] |
| Isoflavones | 18 postmenopausal women | 7.1 ± 1.1, 65 ± 11, 132 ± 22 mg/day, oral administration, 93 days. | Have no significant influence on either the plasma levels of estrogen, androgen, gonadotropin, sex hormone binding globulin (SHBG), prolactin, insulin, cortisol, and thyroid hormone, or the vaginal cytology or endometrial biopsy. | [92] |
| isoflavones | Healthy postmenopausal women with intact uterus | 150 mg/day for 5 years. | Increased incidence of endometrial hyperplasia. | [96] |
| Puerarin | Primary culture of stromal cells from ectopic endometrium of premenopausal endometriosis patients. | 10−9 mol/L. | Reversed estrogenic activities: increasing MMP-9 expression, decreasing TIMP-1 expression, promoting invasiveness, and vascularization. | [98] |
| Puerarin | Primary culture of stromal cells from human ovary endometriotic cysts. | 10−9 mol/L. | Suppressed the cell proliferation-induced by E2, partly via impeding a rapid, non-genomic, membrane-initiated ERK pathway. | [99] |
| Puerarin | Rat endometriotic model, autotransplantation. | 60, 200 or 600 mg/kg/day, gavage, 4 weeks. | Inhibited proliferation of ectopic endometrium. Suppressed aromatase expression, reduced local estrogen biosynthesis. | [100] |
| pueraria flower extract | Human endometriotic 11Z and 12Z cells; mouse endometriotic model, allotransplantation. | 25–100 μg/mL; 150 and 300 mg/kg/day, oral administration, 5 weeks. | Suppressed adhesion of immortalized human endometriotic cells; reduced MMP-2 and MMP-9 expression. Activated ERK1/2 pathway. Suppressed endometriotic lesion formation. | [101] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cai, X.; Liu, M.; Zhang, B.; Zhao, S.-J.; Jiang, S.-W. Phytoestrogens for the Management of Endometriosis: Findings and Issues. Pharmaceuticals 2021, 14, 569. https://doi.org/10.3390/ph14060569
Cai X, Liu M, Zhang B, Zhao S-J, Jiang S-W. Phytoestrogens for the Management of Endometriosis: Findings and Issues. Pharmaceuticals. 2021; 14(6):569. https://doi.org/10.3390/ph14060569
Chicago/Turabian StyleCai, Xia, Min Liu, Bing Zhang, Shao-Jie Zhao, and Shi-Wen Jiang. 2021. "Phytoestrogens for the Management of Endometriosis: Findings and Issues" Pharmaceuticals 14, no. 6: 569. https://doi.org/10.3390/ph14060569
APA StyleCai, X., Liu, M., Zhang, B., Zhao, S.-J., & Jiang, S.-W. (2021). Phytoestrogens for the Management of Endometriosis: Findings and Issues. Pharmaceuticals, 14(6), 569. https://doi.org/10.3390/ph14060569





