Phytochemicals from Plant Foods as Potential Source of Antiviral Agents: An Overview
Abstract
1. Introduction
1.1. General Aspects of Viruses
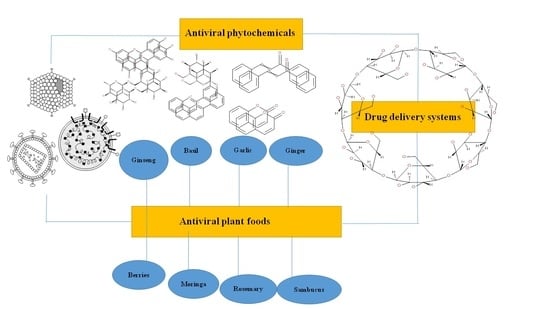
1.2. Focus of the Review
1.3. Extraction and Chemical Characterization of Plant Foods and Phytochemicals
1.4. Biological and Pharmacological Activities of Plant Foods and Phytochemicals
2. Methodology
Research Strategy
3. Viruses
3.1. Vicious Cycle of Viruses
3.2. Respiratory Syncytial Virus
3.3. Para-Influenza and Influenza Virus
3.4. Coronaviruses
3.5. Herpes
3.6. Rotavirus
3.7. Dengue Virus
3.8. Human Immunodeficiency Virus
| Plant | Family | Virus | Types of Extract | Antiviral Compounds | Ref. |
|---|---|---|---|---|---|
| Aegle marmelos | Rutaceae | Human coxsackie viruses B1-B6, nuclear polyhedrosis virus | Hexane, ethyl acetate and methanol and aqueous | Marmelide, seselin | [86] [87] |
| Allium cepa L. | Amaryllidaceae | SARS-COV, Newcastle disease virus | Ethanol | Quercetin, allicin, thiosulfinates | [88] [89] |
| Allium sativum L. | Amaryllidaceae | DENV, common cold virus, influenza virus A and B, HIV, HSV-1, HSV-2, Newcastle Disease Virus | Aqueous | Quercetin, allicin, thiosulfinates, ajoene | [90] [91] [89] [92] |
| Aloe vera | Liliaceae | HSV-1, HSV-2; | Gel | Unknown | [93] [94] |
| Artocarpus integrifolia | Moraceae | (SA-11) and human (HCR3) rotaviruses, HIV | - | Jacalin | [95] |
| Balanites aegyptiaca | Zygophyllaceae | VSV T2 | Hexane, 80% methanol and water | Unknown | [96] |
| Berberis vulgaris L. | Berberidaceae | Enterovirus 71, human cytomegalovirus (HCMV), CHIKV | - | Berbamine, berberine | [96] [97] [98] |
| Camellia sinensis | Theaceae | HBV, HCV, HSV-1, HIV-1, caliciviruses | - | Catechins, quercetin, gallic acid, theaflavins, theaflavin-3,3′-digallate, theaflavin digallate, Epigallocathechin-3-gallate, (-)-epicatechin gallate | [99] [100] [101] [102,103] |
| Capparis spinosa | Capparidaceae | HSV-2 HSV-1 | Methanol | Unknown Protein | [103] [104] |
| Carica papaya L. | Caricaceae | DENV | Aqueous | Unknown | [105] [106] [107] [108] |
| Cassine xylocarpa | Celastraceae | HIV-1 | - | Pentacyclic lupane-type triterpenoids | [109] [110] |
| Cistus incanus | Cistaceae | HIV-1, HIV-2 | - | ||
| Citrus aurantium L. | Rutaceae | DENV; HIV-1, HSV types 1 and 2, influenza, and yellow fever. | - | Polysaccharides, polyphenols | [111] [112] [112] |
| Curcuma longa | Zingiberaceae | HSV types 1 and 2 | - | Curcumin | [113] [114] |
| Diospyros kaki | Ebenacee | Influenza virus H3N2, H5N3, HSV-1, vesicular stomatitis virus, Sendai virus, poliovirus, coxsachievirus, adenovirus, rotavirus, feline calicivirus, mouse norovirus, Newcastle disease virus | Aqueous | Licocoumarone, licoflavonol, glyasperin D, luteolin, vitexin, apigenin-7-O-glucoside; tannins | [115] [116] |
| Euphorbia hirta | Euforbiacee | HIV-1, HIV-2, SIV mac 251 | Aqueous | Flavonoids | [117] |
| Euphorbia spinidens | Euforbiacee | HSV type 1 | Methanol | Unknown | [118] |
| Ficus carica | Moraceae | HSV-1 HSV-1, ECV-11 and ADV influenza virus | Latex | Unknown | [119] |
| Glycyrrhiza glabra | Fabaceae | HCV, HSV, CVB3, DHV, H5N1, Influenza virus, HRSV, | Aqueous, methanolic and ethanolic | Glycyrrhizin, 18 β-glycyrrhetinic acid, liquiritigenin, licochalcone A and E, glabridin | [120] [121] |
| Glycyrrhiza uralensis | Fabaceae | HCV; Rotavirus diarrhea | Methanol | Glycycoumarin, glycyrin, glycyrol, liquiritigenin, isoliquiritigenin, licochalcone A and glabridin | [122] [123] |
| Hyssopus officinalis | Lamiaceae | HSV type 1 and 2 | - | Unknown | [124] |
| Lycium barbarum | Solanaceae | NDVs | - | Polysaccharides | [125] [126] |
| Melissa officinalis | Lamiaceae | HSV-1, HSV-2 HIV, influenza virus | Aqueous | Essential oils | [127] |
| Mentha pulegium | Lamiaceae | HSV type 1 | Methanol | Unknown | [128] |
| Moringa peregrina | Moringaceae | HSV type 1 | Aqueous | Unknown | [129] |
| Moringa oleifera | Moringaceae | HIV, HSV, HBV, EBV, FMDV and NDV. | - | Flavonoids and phenolic acids | [130] [131] |
| Myristica fragrans | Myristicaceae | Human rotavirus | Unknown | [132] | |
| Olea oleuropaea | Oleaceae | NDV | Aqueous | Unknown | [133] |
| Panax ginseng | Araliaceae | RSV, influenza virus, HIV, HSV, hepatitis virus, enterovirus, coxsackievirus, norovirus, rotavirus rhinovirus, | - | Epigallocatechin gallate, theaflavin digallate, genistein, hesperidin, neohesperidin, diosmin, pectic polysaccharides; ginsenosides | [132,134] |
| Panax notoginseng | Araliaceae | Influenza A virus | Aqueous | Unknown | [135] |
| Phyllanthus acidus | Phyllanthaceae | HBV | - | Highly oxygenated norbisabolane sesquiterpenoids, phyllanthacidoid acid methyl ester | [136] |
| Phyllanthus emblica | Phyllanthaceae | Influenza A virus strain H3N2 HBV | - | Highly oxygenated norbisabolane sesquiterpenoids Sesquiterpenoid glycoside dimers | [136,137] |
| Piper longum | Piperaceae | HBV | - | Longumosides A and B, and two amide alkaloids | [138] |
| Prunella vulgaris | Lamiaceae | HIV-1 Ebola virus | Aqueous | Unknown | [139] |
| Psidium guajava | Myrtaceae | H1N1 viruses | Aqueous | Tannins and polyphenols | [140] [141] |
| Quercus persica | Fagaceae | HSV-1 | Aqueous | Unknown | [142] |
| Salacia reticulata | Celastraceae | H1N1 viruses | Aqueous | Unknown | [143] |
| Sanguisorba minor | Rosaceae | VSV, HSV-1 HIV | Methanol/Water | Unknown | [144] [145] |
| Solanum nigrum | Solanaceae | HCV | Methanol | Unknown | [146] |
| Spondias lutea | Anacardiaceae | Human rotavirus, HSV type 1 | - | Hydrolysable tannins, O-glycosylated flavonoids, phenolic acids, and a carbohydrate | [147] |
| Taraxacum officinale | Asteraceae | HCV Influenza virus type A, H1N1 | Methanol | Unknown | [148] |
| Thymus vulgaris | Lamiaceae | HIV-1 | Methanol | Unknown | [149] |
| Thymus carmanicus | Lamiaceae | HIV-1 | Aqueous | Unknown | [150] |
| Thymus daenensis | Lamiaceae | HIV-1 | Methanol | Unknown | [149] |
| Thymus kotschyanus | Lamiaceae | HIV-1 | Aqueous | Unknown | [151] |
| Viola diffusa | Violaceae | HBV | - | Friedelolactones | [152] |
| Zataria multiflora | Labiate | HSV-1 | - | Rosmarinic acid | [153] |
| Zingiber officinale | Zingiberaceae | Chikungunya virus (CHIKV) | Aqueous | Unknown | [154] |
| Phytochemicals or Extracts | Potential Antiviral Activity | Delivery System Strategy | Reference |
|---|---|---|---|
| Myricetin | HIV, RLV, influenza | Self-nanoemulsifying drug delivery systems SNEDDS, nanogel, mixed micelles, cocrystal, nanoencapsulation, nanosuspension | [155] |
| Apigenin | Influenza A, HCV, Enterovirus 71, FMDV, ASFV. | Solid dispersion, W/O/Wemulsion, O/W microemulsion, mixed micelles, phospholipid phytosome, self-microemulsifying drug delivery systems (SMEDDS) pellets | [156,157,158] |
| Baicalin | DENV, RSV, HIV, Hepatitis B virus (HBV), influenza virus, NDV, enterovirus 71. | Liposome, mixed micelles, polymeric micelles, SNEDDS, SMEDDS, nanoparticles, nanocrystals, inclusion complex solid dispersion | [159,160] |
| Catechins | HBV, HSV, EBV, Adenovirus, HIV, HCV, Influenza virus, DENV, JEV, TBEV Zika Virus (ZIKV), CHIKV, HTLV-1, Rotavirus, Enterovirus EV71, EBOV, PRRSV, VHSV, IHNV, SVCV, GCRV. | Microparticles, calcium pectinate gel particles, chitosan nanoparticles, Nanoparticles of Polylactic Acid–polyethylene Glycol, poly(lactic-co-glycolic acid) nanoparticles, gold nanoparticles, colloidal complexes, liposomes, nanoemulsions | [161,162] |
| Hydroalcoholic extracts from Forsythiae fructus rich in Forsythoside A; Phillyrin; Calceolarioside; Rengynic acid | Influenza, RSV | Chito-oligosaccharides | [163] |
| Extracts from Forsythiae fructus rich in 3,5-dicaffeoylquinic acid, 3,4-dicaffeoylquinic acid, neochlorogenic acid, chlorogenic acid, cryptochlorogenic acid, isoforsythoside, forsythoside A, forsythoside B | Influenza, RSV, HIV, NDV | Chito-oligosaccharides | [164] |
| Andrographolide | DENV, CHIKV, HBV, HCV, HSV1, EBV, HIV HPV16, pseudovirus, influenza. | Microspheres; nanosuspension and self-nanodispersion; nanoparticles, SMEDDS, inclusion complex | [165,165] |
| Anthocyanin-rich extracts (from Mulberry) and purified compounds (i.e., delphinidin, cyanidin, and pelargonidin) | Influenza virus, HSV type 1 and 2, RV, Adenovirus 36, BT2, T4 and simian rotavirus SA-11, HAV, FCV-F9 and MNV-1, HCV, WNV, DENV, and ZIKV. | Micro/nanoencapsulation systems, nano/micro-gels, spray-drying and freeze-drying, electrohydrodynamic encapsulation, emulsification and liposomal encapsulation | [166,167,168] [169] |
| Curcumin | ZIKV, CHIKV, norovirus, Influenza, DENV type 2, RSV, HBV, HCV, HPV, HIV, CMV, EV71. | Nanoparticles, solid dispersion, SNEDDS, SMEDDS, lipid carrier, copolymeric and mixed micelles, exosomes | [170,171] |
| Water extract from Panax ginseng root and punicalagin | HSV types 1 and 2 | Hydroxypropyl methylcellulose (HPMC) hydrogel | [172,173] |
| Water extract from Panax ginseng root and punicalagin | Influenza A virus (strain A/PR/8) | Ultra-sonication-assisted silver nanoparticles | [172,173] |
| Hydroalcoholic extract from Elderberry and Eucalyptus | Influenza virus | Oil-in-water (o/w) and water-in-oil (w/o) emulsions | [174,175] |
| Hydroalcoholic extract from Elderberry and Eucalyptus | Common cold | Encapsulation (BerryPharma® brand) | [174,175] |
| Coumestrol | HSV types 1 and 2 | Lipid nanoemulsion based on dioleylphosphocholine | [176] |
| Quercetin | JEV, influenza A, EBV, MAYV, RV, HCV | Nanocrystal, nanoparticles phytosome, nanoliposome, mixed micelles, SNEDDS, nanocarrier, nanoemulsion, nanosuspension | [177,178] |
| Naringenin | DENV, HCV | SNEDDS, solid dispersion, nanoparticles, liposome, nanosuspension, cyclodextrin complex | [178,179,180] |
| Resveratrol | HIV/AIDS | Polymeric nanoparticles, solid-liquid nanoparticles, self-emulsifying methods, nanosponges, liposomes, emulsion-liposome blends, lipid-core nanocapsules, active lipospheres | [179,180] |
| Silibinin | Hepatitis C virus infection | Encapsulation based on phytoliposomes | [181] |
4. The Healing Potential of Medicinal Plants
4.1. Herbs Remedies for viRAL Diseases: Historical Notes and General Aspects
4.2. Plant Extracts
5. Phytochemicals
5.1. Flavonoids
5.1.1. Anthocyanins
5.1.2. Chalcone and Dihydrochalcones
5.1.3. Flavones, Flavanones, Flavonols, Isoflavonoids and Derivatives
5.1.4. Flavans and Neoflavonoids
5.2. Coumarins and Arylcoumarins
5.3. Resveratrol (Stilbenes)
5.4. Other Phenolic Compounds
5.4.1. Lignans
5.4.2. Tannins
5.4.3. Hydroxyphenylpropenes
5.4.4. Curcumin
5.5. Terpenoids and Carotenoids
5.5.1. Terpenoids
5.5.2. Carotenoids
5.6. Alkaloids
5.7. Organosulfur Compounds
5.8. Poly-Acetylene and Polysaccharides
5.8.1. Poly-Acetylene
5.8.2. Polysaccharides
5.9. Antraquinones and Gingerol
5.10. Salicylic Acid and Chlorophyllin
6. Plant Foods in the Preparation of Antiviral Drugs
6.1. Ginseng
6.2. Basil and Oregano
6.3. Fennel
6.4. Garlic
6.5. Ginger
6.6. Peppermint
6.7. Sambucus
6.8. Licorice
6.9. Rosemary
6.10. Moringa
6.11. Pomegranate
6.12. Blackberry and Other Berries
7. Drug Delivery Strategies of Phytochemicals with Antiviral Activity
8. Conclusions
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wagner, E.K.; Hewlett, M.J.; Bloom, D.C.; Camerini, D. Basic Virology; Blackwell Science: Malden, MA, USA, 1999; Volume 3. [Google Scholar]
- Weber, I.T.; Agniswamy, J. HIV-1 Protease: Structural Perspectives on Drug Resistance. Viruses 2009, 1, 1110–1136. [Google Scholar] [CrossRef]
- Orhan, I.; Deliorman-Orhan, D.; Özçelik, B. Antiviral activity and cytotoxicity of the lipophilic extracts of various edible plants and their fatty acids. Food Chem. 2009, 115, 701–705. [Google Scholar] [CrossRef]
- Atanasov, A.G.; Zotchev, S.B.; Dirsch, V.M.; Supuran, C.T. Natural products in drug discovery: Advances and opportunities. Nat. Rev. Drug Discov. 2021, 20, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Casado, A. The Health Potential of Fruits and Vegetables Phytochemicals: Notable Examples. Crit. Rev. Food Sci. Nutr. 2016, 56, 1097–1107. [Google Scholar] [CrossRef]
- Putnik, P.; Barba, F.J.; Lucini, L.; Rocchetti, G.; Montesano, D. Conventional, non-conventional extraction techniques and new strategies for the recovery of bioactive compounds from plant material for human nutrition. Food Res. Int. 2019, 123, 516–517. [Google Scholar] [CrossRef]
- Putnik, P.; Lorenzo, J.M.; Barba, F.J.; Roohinejad, S.; Režek Jambrak, A.; Granato, D.; Montesano, D.; Bursać Kovačević, D. Novel food processing and extraction technologies of high-added value compounds from plant materials. Foods 2018, 7, 106. [Google Scholar] [CrossRef] [PubMed]
- Rocchetti, G.; Blasi, F.; Montesano, D.; Ghisoni, S.; Marcotullio, M.C.; Sabatini, S.; Cossignani, L.; Lucini, L. Impact of conventional/non-conventional extraction methods on the untargeted phenolic profile of Moringa oleifera leaves. Food Res. Int. 2019, 115, 319–327. [Google Scholar] [CrossRef] [PubMed]
- Rocchetti, G.; Pagnossa, J.P.; Blasi, F.; Cossignani, L.; Piccoli, R.H.; Zengin, G.; Montesano, D.; Cocconcelli, P.S.; Lucini, L. Phenolic profiling and in vitro bioactivity of Moringa oleifera leaves as affected by different extraction solvents. Food Res. Int. 2020, 127, 108712. [Google Scholar] [CrossRef]
- Chuanasa, T.; Phromjai, J.; Lipipun, V.; Likhitwitayawuid, K.; Suzuki, M.; Pramyothin, P.; Hattori, M.; Shiraki, K. Anti-herpes simplex virus (HSV-1) activity of oxyresveratrol derived from Thai medicinal plant: Mechanism of action and therapeutic efficacy on cutaneous HSV-1 infection in mice. Antiviral. Res. 2008, 80, 62–70. [Google Scholar] [CrossRef]
- Li, H.-B.; Chen, F. Isolation and purification of baicalein, wogonin and oroxylin A from the medicinal plant Scutellaria baicalensis by high-speed counter-current chromatography. J. Chromatogr. 2005, 1074, 107–110. [Google Scholar] [CrossRef]
- Roychoudhury, S.; Das, A.; Sengupta, P.; Dutta, S.; Roychoudhury, S.; Choudhury, A.P.; Ahmed, A.; Bhattacharjee, S.; Slama, P. Viral Pandemics of the Last Four Decades: Pathophysiology, Health Impacts and Perspectives. Int. J. Env. Res. Public Health 2020, 17, 9411. [Google Scholar] [CrossRef] [PubMed]
- Gasparini, R.; Amicizia, D.; Lai, P.L.; Panatto, D. Clinical and socioeconomic impact of seasonal and pandemic influenza in adults and the elderly. Hum. Vaccin. Immunother. 2012, 8, 21–28. [Google Scholar] [CrossRef]
- Nováková, L.; Pavlík, J.; Chrenková, L.; Martinec, O.; Červený, L. Current antiviral drugs and their analysis in biological materials–Part II: Antivirals against hepatitis and HIV viruses. J. Pharm. Biomed. Anal. 2018, 147, 378–399. [Google Scholar] [CrossRef] [PubMed]
- WHO. Infection Prevention and Control of Epidemic-and Pandemic-Prone Acute Respiratory Infections in Health Care; World Health Organization: Geneva, Switzerland, 2014. [Google Scholar]
- GBD 2015 Disease and Injury Incidence and Prevalence Collaborators. Global, regional, and national incidence, prevalence and years lived with disability for 310 diseases and injuries, 1990-2015: A systematic analysis for the global burden of disease study 2015. Lancet 2016, 388, 1448–1449. [Google Scholar]
- Maheshwari, A.; Thuluvath, P.J. Management of acute hepatitis C. Clin. Liver Dis. 2010, 14, 169–176. [Google Scholar] [CrossRef] [PubMed]
- Cos, P.; Berghe, D.V.; Bruyne, T.D.; Vlietinck, A. Plant substances as antiviral agents: An update (1997-2001). Curr. Org. Chem. 2003, 7, 1163–1180. [Google Scholar] [CrossRef]
- Watkins, T.; Resch, W.; Irlbeck, D.; Swanstrom, R. Selection of high-level resistance to human immunodeficiency virus type 1 protease inhibitors. Antimicrob. Agents Chemother. 2003, 47, 759–769. [Google Scholar] [CrossRef][Green Version]
- Murata, Y.; Falsey, A. Respiratory syncytial virus infection in adults. Antivir. Ther. 2007, 12, 659–670. [Google Scholar] [CrossRef]
- Wennergren, G.; Kristjansson, S. Relationship between respiratory syncytial virus bronchiolitis and future obstructive airway diseases. Eur. Respir. J. 2001, 18, 1044–1058. [Google Scholar] [CrossRef] [PubMed]
- Piedimonte, G.; Perez, M.K. Respiratory syncytial virus infection and bronchiolitis. Pediatr. Rev. 2014, 35, 519. [Google Scholar] [CrossRef]
- Boukhvalova, M.S.; Yim, K.C.; Blanco, J. Cotton rat model for testing vaccines and antivirals against respiratory syncytial virus. Antivir. Chem. Chemother 2018, 26. [Google Scholar] [CrossRef]
- Soto, J.A.; Gálvez, N.M.; Pacheco, G.A.; Bueno, S.M.; Kalergis, A.M. Antibody development for preventing the human respiratory syncytial virus pathology. Mol. Med. 2020, 26, 1–10. [Google Scholar] [CrossRef]
- Branche, A.R.; Falsey, A.R. In Respiratory Viral Infections: Parainfluenza Virus Infection. Semin. Respir. Crit. Care Med. 2016, 538. [Google Scholar] [CrossRef] [PubMed]
- Vachon, M.-L.; Dionne, N.; Leblanc, É.; Moisan, D.; Bergeron, M.G.; Boivin, G. Human parainfluenza type 4 infections, Canada. Emerg. Infect. Dis. 2006, 12, 1755. [Google Scholar] [CrossRef] [PubMed]
- Fouchier, R.A.; Rimmelzwaan, G.F.; Kuiken, T.; Osterhaus, A.D. Newer respiratory virus infections: Human metapneumovirus, avian influenza virus, and human coronaviruses. Curr. Opin Infect. Dis. 2005, 18, 141–146. [Google Scholar] [CrossRef] [PubMed]
- De Jong, J.C.; Claas, E.C.; Osterhaus, A.D.; Webster, R.G.; Lim, W.L. A pandemic warning? Nature 1997, 389, 554. [Google Scholar] [CrossRef]
- Claas, E.C.; Osterhaus, A.D.; Van Beek, R.; De Jong, J.C.; Rimmelzwaan, G.F.; Senne, D.A.; Krauss, S.; Shortridge, K.F.; Webster, R.G. Human influenza A H5N1 virus related to a highly pathogenic avian influenza virus. Lancet 1998, 351, 472–477. [Google Scholar] [CrossRef]
- Perlman, S.; Netland, J. Coronaviruses post-SARS: Update on replication and pathogenesis. Nat. Rev. Microbiol. 2009, 7, 439–450. [Google Scholar] [CrossRef]
- Hu, B.; Zeng, L.-P.; Yang, X.-L.; Ge, X.-Y.; Zhang, W.; Li, B.; Xie, J.-Z.; Shen, X.-R.; Zhang, Y.-Z.; Wang, N.; et al. Discovery of a rich gene pool of bat SARS-related coronaviruses provides new insights into the origin of SARS coronavirus. PLoS Pathog. 2017, 13, e1006698. [Google Scholar] [CrossRef]
- Cui, J.; Li, F.; Shi, Z.-L. Origin and evolution of pathogenic coronaviruses. Nat. Rev. Microbiol. 2019, 17, 181–192. [Google Scholar] [CrossRef]
- Ou, X.; Liu, Y.; Lei, X.; Li, P.; Mi, D.; Ren, L.; Guo, L.; Guo, R.; Chen, T.; Hu, J.; et al. Characterization of spike glycoprotein of SARS-CoV-2 on virus entry and its immune cross-reactivity with SARS-CoV. Nat. Commun. 2020, 11, 1620. [Google Scholar] [CrossRef]
- Wang, J.-T.; Chang, S.-C. Severe acute respiratory syndrome. Curr. Opin. Infect. Dis. 2004, 17, 143–148. [Google Scholar] [CrossRef] [PubMed]
- Gu, J.; Korteweg, C. Pathology and pathogenesis of severe acute respiratory syndrome. Am. J. Pathol. 2007, 170, 1136–1147. [Google Scholar] [CrossRef]
- Guan, Y.; Zheng, B.; He, Y.; Liu, X.; Zhuang, Z.; Cheung, C.; Luo, S.; Li, P.; Zhang, L.; Guan, Y. Isolation and characterization of viruses related to the SARS coronavirus from animals in southern China. Science 2003, 302, 276–278. [Google Scholar] [CrossRef] [PubMed]
- Yu, D.; Li, H.; Xu, R. Prevalence of IgG antibody to SARS-associated coronavirus in animal traders--Guangdong Province, China, 2003. MMWR Morb. Mortal. Wkly. Rep. 2003, 52, 986. [Google Scholar]
- Seto, W.; Tsang, D.; Yung, R.; Ching, T.; Ng, T.; Ho, M.; Ho, L.; Peiris, J.; Advisors of Expert SARS group of Hospital Authority. Effectiveness of precautions against droplets and contact in prevention of nosocomial transmission of severe acute respiratory syndrome (SARS). Lancet 2003, 361, 1519–1520. [Google Scholar] [CrossRef]
- Hijawi, B.; Abdallat, M.; Sayaydeh, A.; Alqasrawi, S.; Haddadin, A.; Jaarour, N.; El Sheikh, S.; Alsanouri, T. Novel coronavirus infections in Jordan, April 2012: Epidemiological findings from a retrospective investigation. EMHJ-East. Mediterr. Health J. 2013, 19 (Suppl. 1), S12–S18. [Google Scholar] [CrossRef]
- Zaki, A.M.; Van Boheemen, S.; Bestebroer, T.M.; Osterhaus, A.D.; Fouchier, R.A. Isolation of a novel coronavirus from a man with pneumonia in Saudi Arabia. N. Engl. J. Med. 2012, 367, 18141820. [Google Scholar] [CrossRef] [PubMed]
- WHO. Middle East Respiratory Syndrome Coronavirus (MERS-CoV). MERS Monthly Summary November 2019; World Health Organization: Geneva, Switzerland, 2019. [Google Scholar]
- WHO. Middle East Respiratory Syndrome Coronavirus (MERS-CoV). 2020. Available online: https://www.who.int/en/news-room/fact-sheets/detail/middle-east-respiratory-syndrome-coronavirus-(mers-cov) (accessed on 2 June 2020).
- Chan, J.F.; Lau, S.K.; To, K.K.; Cheng, V.C.; Woo, P.C.; Yuen, K.Y. Middle East respiratory syndrome coronavirus: Another zoonotic betacoronavirus causing SARS-like disease. Clin. Microbiol. Rev. 2015, 28, 465–522. [Google Scholar] [CrossRef]
- Arabi, Y.M.; Balkhy, H.H.; Hayden, F.G.; Bouchama, A.; Luke, T.; Baillie, J.K.; Al-Omari, A.; Hajeer, A.H.; Senga, M.; Denison, M.R.; et al. Middle East Respiratory Syndrome. N. Engl. J. Med. 2017, 376, 584–594. [Google Scholar] [CrossRef]
- WHO. Coronavirus Disease (COVID-19), Situation Report–134. 2020. Available online: https://www.who.int/docs/default-source/coronaviruse/situation-reports/20200602-covid-19-sitrep-134.pdf?sfvrsn=cc95e5d5_2 (accessed on 3 June 2020).
- WHO. Pneumonia of Unknown Cause in China. 2020. Available online: https://www.who.int/csr/don/05-january-2020-pneumonia-of-unkown-cause-china/en/ (accessed on 29 January 2021).
- Docea, A.O.; Tsatsakis, A.; Albulescu, D.; Cristea, O.; Zlatian, O.; Vinceti, M.; Moschos, S.A.; Tsoukalas, D.; Goumenou, M.; Drakoulis, N.; et al. A new threat from an old enemy: Re-emergence of coronavirus (Review). Int. J. Mol. Med. 2020, 45, 1631–1643. [Google Scholar] [CrossRef] [PubMed]
- Prompetchara, E.; Ketloy, C.; Palaga, T. Immune responses in COVID-19 and potential vaccines: Lessons learned from SARS and MERS epidemic. Asian Pac. J. Allergy Immunol. 2020, 38, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Pang, J.; Wang, M.X.; Ang, I.Y.H.; Tan, S.H.X.; Lewis, R.F.; Chen, J.I.; Gutierrez, R.A.; Gwee, S.X.W.; Chua, P.E.Y.; Yang, Q.; et al. Potential Rapid Diagnostics, Vaccine and Therapeutics for 2019 Novel Coronavirus (2019-nCoV): A Systematic Review. J. Clin. Med. 2020, 9. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Wang, X.; Cao, D.; Sun, R.; Li, C.; Li, G. Rapid review for the anti-coronavirus effect of remdesivir. Drug Discov. Ther. 2020, 14, 73–76. [Google Scholar] [CrossRef] [PubMed]
- Du, Y.X.; Chen, X.P. Favipiravir: Pharmacokinetics and Concerns About Clinical Trials for 2019-nCoV Infection. Clin. Pharmacol. Ther. 2020. [Google Scholar] [CrossRef]
- Zhang, C.; Wu, Z.; Li, J.-W.; Zhao, H.; Wang, G.-Q. Cytokine release syndrome in severe COVID-19: Interleukin-6 receptor antagonist tocilizumab may be the key to reduce mortality. Int. J. Antimicrob. Agents 2020, 55, 105954. [Google Scholar] [CrossRef] [PubMed]
- Fu, B.; Xu, X.; Wei, H. Why tocilizumab could be an effective treatment for severe COVID-19? J. Transl. Med. 2020, 18, 164. [Google Scholar] [CrossRef]
- Toniati, P.; Piva, S.; Cattalini, M.; Garrafa, E.; Regola, F.; Castelli, F.; Franceschini, F.; Airò, P.; Bazzani, C.; Beindorf, E.A.; et al. Tocilizumab for the treatment of severe COVID-19 pneumonia with hyperinflammatory syndrome and acute respiratory failure: A single center study of 100 patients in Brescia, Italy. Autoimmun. Rev. 2020, 19, 102568. [Google Scholar] [CrossRef]
- Roizman, B. Herpesviridae. In Fields’ Virology, 4th ed.; Howley, R.M.K.D., Ed.; Lippincott Press: New York, NY, USA, 2020. [Google Scholar]
- Roizman, B.; Knipe, D. The replication of herpes simplex virus. In Fields’ Virology; Howley, R.M.K.D., Ed.; Lippincott Press: New York, NY, USA, 2020. [Google Scholar]
- Groves, M.J. Genital herpes: A review. Am. Fam. Physician 2016, 93, 928–934. [Google Scholar]
- Jaishankar, D.; Shukla, D. Genital Herpes: Insights into Sexually Transmitted Infectious Disease. Microb. Cell 2016, 3, 438–450. [Google Scholar] [CrossRef]
- Koh, Y.; Matsumi, S.; Das, D.; Amano, M.; Davis, D.A.; Li, J.; Leschenko, S.; Baldridge, A.; Shioda, T.; Yarchoan, R.; et al. Potent Inhibition of HIV-1 Replication by Novel Non-peptidyl Small Molecule Inhibitors of Protease Dimerization. J. Biol. Chem. 2007, 282, 28709–28720. [Google Scholar] [CrossRef]
- Bishop, R.F. Natural history of human rotavirus infections. Viral Infect. Gastrointest. Tract 1994, 131–167. [Google Scholar]
- Haffejee, I.E. The epidemiology of rotavirus infections: A global perspective. J. Pediatr. Gastroenterol. Nutr. 1995, 20, 275–286. [Google Scholar] [CrossRef]
- Tate, J.E.; Burton, A.H.; Boschi-Pinto, C.; Parashar, U.D.; World Health Organization–Coordinated Global Rotavirus Surveillance Network; Agocs, M.; Serhan, F.; De Oliveira, L.; Mwenda, J.M.; Mihigo, R. Global, regional, and national estimates of rotavirus mortality in children<5 years of age, 2000–2013. Clin. Infect. Dis. 2016, 62 (Suppl. 2), 96–105. [Google Scholar] [CrossRef]
- Hagbom, M.; Istrate, C.; Engblom, D.; Karlsson, T.; Rodriguez-Diaz, J.; Buesa, J.; Taylor, J.A.; Loitto, V.M.; Magnusson, K.E.; Ahlman, H.; et al. Rotavirus stimulates release of serotonin (5-HT) from human enterochromaffin cells and activates brain structures involved in nausea and vomiting. PLoS Pathog. 2011, 7, e1002115. [Google Scholar] [CrossRef] [PubMed]
- Leung, A.K.; Robson, W.L. Acute gastroenteritis in children: Role of anti-emetic medication for gastroenteritis-related vomiting. Paediatr. Drugs 2007, 9, 175–184. [Google Scholar] [CrossRef] [PubMed]
- Heaton, P.M.; Ciarlet, M. Vaccines: The pentavalent rotavirus vaccine: Discovery to licensure and beyond. Clin. Infect. Dis. 2007, 45, 1618–1624. [Google Scholar] [CrossRef] [PubMed]
- Tate, J.E.; Burton, A.H.; Boschi-Pinto, C.; Steele, A.D.; Duque, J.; Parashar, U.D. 2008 estimate of worldwide rotavirus-associated mortality in children younger than 5 years before the introduction of universal rotavirus vaccination programmes: A systematic review and meta-analysis. Lancet Infect. Dis. 2012, 12, 136–141. [Google Scholar] [CrossRef]
- Martella, V.; Bányai, K.; Matthijnssens, J.; Buonavoglia, C.; Ciarlet, M. Zoonotic aspects of rotaviruses. Vet. Microbiol. 2010, 140, 246–255. [Google Scholar] [CrossRef]
- Guzmán, M.G.; Kouri, G. Dengue: An update. Lancet Infect. Dis. 2002, 2, 33–42. [Google Scholar] [CrossRef]
- Pang, X.; Zhang, R.; Cheng, G. Progress towards understanding the pathogenesis of dengue hemorrhagic fever. Virol. Sin. 2017, 32, 16–22. [Google Scholar] [CrossRef]
- Carlos, C.C.; Oishi, K.; Cinco, M.T.; Mapua, C.A.; Inoue, S.; Cruz, D.J.M.; Pancho, M.A.M.; Tanig, C.Z.; Matias, R.R.; Morita, K. Comparison of clinical features and hematologic abnormalities between dengue fever and dengue hemorrhagic fever among children in the Philippines. Am. J. Trop. Med. Hyg. 2005, 73, 435–440. [Google Scholar] [CrossRef]
- Tricou, V.; Minh, N.N.; Van, T.P.; Lee, S.J.; Farrar, J.; Wills, B.; Tran, H.T.; Simmons, C.P. A Randomized Controlled Trial of Chloroquine for the Treatment of Dengue in Vietnamese Adults. PLoS Negl. Trop. Dis. 2010, 4, e785. [Google Scholar] [CrossRef]
- Navarro-Sanchez, E.; Altmeyer, R.; Amara, A.; Schwartz, O.; Fieschi, F.; Virelizier, J.L.; Arenzana-Seisdedos, F.; Desprès, P. Dendritic-cell-specific ICAM3-grabbing non-integrin is essential for the productive infection of human dendritic cells by mosquito-cell-derived dengue viruses. EMBO Rep. 2003, 4, 723–728. [Google Scholar] [CrossRef]
- Nguyen, N.M.; Tran, C.N.B.; Phung, L.K.; Duong, K.T.H.; Huynh, H.L.A.; Farrar, J.; Nguyen, Q.T.H.; Tran, H.T.; Nguyen, C.V.V.; Merson, L. A randomized, double-blind placebo controlled trial of balapiravir, a polymerase inhibitor, in adult dengue patients. J. Infect. Dis. 2013, 207, 1442–1450. [Google Scholar] [CrossRef]
- Gottlieb, G.S.; Raugi, D.N.; Smith, R.A. 90-90-90 for HIV-2? Ending the HIV-2 epidemic by enhancing care and clinical management of patients infected with HIV-2. Lancet HIV 2018, 5, 390–399. [Google Scholar] [CrossRef]
- ICTV, International Committee on Taxonomy of Viruses, 61. Retroviridae; National Institutes of Health: Bethesda, MD, USA, 2002.
- Cleghorn, F.; Reitz, M.; Popovic, M.; Gallo, R. Human immunodeficiency viruses. In Bennett’s Principles and Practice of Infectious Diseases, 6th ed.; Churchill Caurehill Livingston: Philadelphia, PA, USA, 2005; pp. 2119–2133. [Google Scholar]
- Weiss, R.; Dalgleish, A.; Loveday, C.; DH, P. Human Immunodeficiency Viruses. In Principles and Practice of Clinical Virology, 5th ed.; Zuckerman, A.J., Banatvala, J.E., Pattison, J.R., Griffiths, P.D., Schoub, B.D., Eds.; John Wiley & Sons Ltd.: Hoboken, NJ, USA, 2004; pp. 721–757. [Google Scholar]
- Redfield, M.M. Heart failure with preserved ejection fraction. N. Engl. J. Med. 2016, 375, 1868–1877. [Google Scholar] [CrossRef]
- Grulich, A.E.; van Leeuwen, M.T.; Falster, M.O.; Vajdic, C.M. Incidence of cancers in people with HIV/AIDS compared with immunosuppressed transplant recipients: A meta-analysis. Lancet 2007, 370, 59–67. [Google Scholar] [CrossRef]
- Engels, E.A.; Biggar, R.J.; Hall, H.I.; Cross, H.; Crutchfield, A.; Finch, J.L.; Grigg, R.; Hylton, T.; Pawlish, K.S.; McNeel, T.S.; et al. Cancer risk in people infected with human immunodeficiency virus in the United States. Int. J. Cancer 2008, 123, 187–194. [Google Scholar] [CrossRef] [PubMed]
- Gallo, R.C. The end or the beginning of the drive to an HIV-preventive vaccine: A view from over 20 years. Lancet 2005, 366, 1894–1898. [Google Scholar] [CrossRef]
- Stadeli, K.M.; Richman, D.D. Rates of emergence of HIV drug resistance in resource-limited settings: A systematic review. Antivir. Ther. 2013, 18, 115. [Google Scholar] [CrossRef]
- Stankov, M.; Behrens, G. HIV-therapy associated lipodystrophy: Experimental and clinical evidence for the pathogenesis and treatment. Endocr. Metab. Immune Disord. Drug Targets 2007, 7, 237–249. [Google Scholar] [CrossRef]
- Lu, D.Y.; Wu, H.Y.; Yarla, N.S.; Xu, B.; Ding, J.; Lu, T.R. HAART in HIV/AIDS Treatments: Future Trends. Infect. Disord. Drug Targets 2018, 18, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Hima Bindu, A.; Naga Anusha, P. Adverse effects of highly active anti-retroviral therapy (HAART). J. Antivir. Antiretrovir. 2011, 3. [Google Scholar] [CrossRef]
- Badam, L.; Bedekar, S.; Sonavane, K.B.; Joshi, S.P. In vitro antiviral activity of bael (Aegle marmelos Corr) upon. J. Commun. Dis. 2002, 34, 88. [Google Scholar]
- Somu, C.; Karuppiah, H.; Sundaram, J. Antiviral activity of seselin from Aegle marmelos against nuclear polyhedrosis virus infection in the larvae of silkworm, Bombyx mori. J. Ethnopharmacol. 2019, 245, 112155. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Li, J.; Luo, C.; Liu, H.; Xu, W.; Chen, G.; Liew, O.W.; Zhu, W.; Puah, C.M.; Shen, X. Binding interaction of quercetin-3-β-galactoside and its synthetic derivatives with SARS-CoV 3CLpro: Structure–activity relationship studies reveal salient pharmacophore features. Bioorganic Med. Chem. 2006, 14, 8295–8306. [Google Scholar] [CrossRef]
- Harazem, R.; Abd El Rahman, S.; El Kenawy, A. Evaluation of Antiviral Activity of Allium Cepa and Allium Sativum Extracts Against Newcastle Disease Virus. Alex. J. Vet. Sci. 2019, 61, 108–118. [Google Scholar] [CrossRef]
- Hall, A.; Troupin, A.; Londono-Renteria, B.; Colpitts, T.M. Garlic organosulfur compounds reduce inflammation and oxidative stress during dengue virus infection. Viruses 2017, 9, 159. [Google Scholar] [CrossRef] [PubMed]
- Josling, P. Preventing the common cold with a garlic supplement: A double-blind, placebo-controlled survey. Adv. Ther. 2001, 18, 189–193. [Google Scholar] [CrossRef]
- Bayan, L.; Koulivand, P.H.; Gorji, A. Garlic: A review of potential therapeutic effects. Avicenna J. Phytomedicine 2014, 4, 1. [Google Scholar]
- Rezazadeh, F.; Moshaverinia, M.; Motamedifar, M.; Alyaseri, M. Assessment of anti HSV-1 activity of Aloe vera gel extract: An in vitro study. J. Dent. 2016, 17, 49. [Google Scholar]
- Zandi, K.; Zadeh, M.A.; Sartavi, K.; Rastian, Z. Antiviral activity of Aloe vera against herpes simplex virus type 2: An in vitro study. Afr. J. Biotechnol. 2007, 6, 15. [Google Scholar]
- Favero, J.; Corbeau, P.; Nicolas, M.; Benkirane, M.; Travé, G.; Dixon, J.F.; Aucouturier, P.; Rasheed, S.; Parker, J.W.; Liautard, J.P. Inhibition of human immunodeficiency virus infection by the lectin jacalin and by a derived peptide showing a sequence similarity with gp120. Eur. J. Immunol. 1993, 23, 179–185. [Google Scholar] [CrossRef]
- Maregesi, S.M.; Pieters, L.; Ngassapa, O.D.; Apers, S.; Vingerhoets, R.; Cos, P.; Berghe, D.A.V.; Vlietinck, A.J. Screening of some Tanzanian medicinal plants from Bunda district for antibacterial, antifungal and antiviral activities. J. Ethnopharmacol. 2008, 119, 58–66. [Google Scholar] [CrossRef] [PubMed]
- Varghese, F.S.; Kaukinen, P.; Gläsker, S.; Bespalov, M.; Hanski, L.; Wennerberg, K.; Kümmerer, B.M.; Ahola, T. Discovery of berberine, abamectin and ivermectin as antivirals against chikungunya and other alphaviruses. Antivir. Res. 2016, 126, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Luganini, A.; Mercorelli, B.; Messa, L.; Palù, G.; Gribaudo, G.; Loregian, A. The isoquinoline alkaloid berberine inhibits human cytomegalovirus replication by interfering with the viral Immediate Early-2 (IE2) protein transactivating activity. Antivir. Res. 2019, 164, 52–60. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, P.; Sahuc, M.-E.; Rouillé, Y.; Rivière, C.; Bonneau, N.; Vandeputte, A.; Brodin, P.; Goswami, M.; Bandyopadhyay, T.; Dubuisson, J. Theaflavins, polyphenols of black tea, inhibit entry of hepatitis C virus in cell culture. PLoS ONE 2018, 13, e0198226. [Google Scholar] [CrossRef]
- Ohba, M.; Oka, T.; Ando, T.; Arahata, S.; Ikegaya, A.; Takagi, H.; Ogo, N.; Zhu, C.; Owada, K.; Kawamori, F. Antiviral effect of theaflavins against caliciviruses. J. Antibiot. 2017, 70, 443–447. [Google Scholar] [CrossRef]
- De Oliveira, A.; Prince, D.; Lo, C.-Y.; Lee, L.H.; Chu, T.-C. Antiviral activity of theaflavin digallate against herpes simplex virus type 1. Antivir. Res. 2015, 118, 56–67. [Google Scholar] [CrossRef]
- Yang, J.; Li, L.; Tan, S.; Jin, H.; Qiu, J.; Mao, Q.; Li, R.; Xia, C.; Jiang, Z.-H.; Jiang, S. A natural theaflavins preparation inhibits HIV-1 infection by targeting the entry step: Potential applications for preventing HIV-1 infection. Fitoterapia 2012, 83, 348–355. [Google Scholar] [CrossRef] [PubMed]
- Zu, M.; Yang, F.; Zhou, W.; Liu, A.; Du, G.; Zheng, L. In vitro anti-influenza virus and anti-inflammatory activities of theaflavin derivatives. Antivir. Res. 2012, 94, 217–224. [Google Scholar] [CrossRef] [PubMed]
- Lam, S.-K.; Ng, T.-B. A protein with antiproliferative, antifungal and HIV-1 reverse transcriptase inhibitory activities from caper (Capparis spinosa) seeds. Phytomedicine 2009, 16, 444–450. [Google Scholar] [CrossRef]
- Sharma, N.; Mishra, K.P.; Chanda, S.; Bhardwaj, V.; Tanwar, H.; Ganju, L.; Kumar, B.; Singh, S.B. Evaluation of anti-dengue activity of Carica papaya aqueous leaf extract and its role in platelet augmentation. Arch. Virol. 2019, 164, 1095–1110. [Google Scholar] [CrossRef]
- Ahmad, N.; Fazal, H.; Ayaz, M.; Abbasi, B.H.; Mohammad, I.; Fazal, L. Dengue fever treatment with Carica papaya leaves extracts. Asian Pac. J. Trop. Biomed. 2011, 1, 330–333. [Google Scholar] [CrossRef]
- Abd Kadir, S.L.; Yaakob, H.; Zulkifli, R.M. Potential anti-dengue medicinal plants: A review. J. Nat. Med. 2013, 67, 677–689. [Google Scholar] [CrossRef]
- Radhakrishnan, N.; Lam, K.; Norhaizan, M. Molecular docking analysis of Carica papaya Linn constituents as antiviral agent. Int. Food Res. J. 2017, 24, 1819. [Google Scholar]
- Callies, O.; Bedoya, L.M.; Beltrán, M.; Muñoz, A.; Calderón, P.O.N.; Osorio, A.A.; Jiménez, I.A.; Alcami, J.; Bazzocchi, I.L. Isolation, structural modification, and HIV inhibition of pentacyclic lupane-type triterpenoids from Cassine xylocarpa and Maytenus cuzcoina. J. Nat. Prod. 2015, 78, 1045–1055. [Google Scholar] [CrossRef]
- Osorio, A.A.; Muñóz, A.; Torres-Romero, D.; Bedoya, L.M.; Perestelo, N.R.; Jiménez, I.A.; Alcamí, J.; Bazzocchi, I.L. Olean-18-ene triterpenoids from Celastraceae species inhibit HIV replication targeting NF-kB and Sp1 dependent transcription. Eur. J. Med. Chem. 2012, 52, 295–303. [Google Scholar] [CrossRef] [PubMed]
- Barreca, D.; Gattuso, G.; Bellocco, E.; Calderaro, A.; Trombetta, D.; Smeriglio, A.; Laganà, G.; Daglia, M.; Meneghini, S.; Nabavi, S.M. Flavanones: Citrus phytochemical with health-promoting properties. BioFactors 2017, 43, 495–506. [Google Scholar] [CrossRef]
- Frabasile, S.; Koishi, A.C.; Kuczera, D.; Silveira, G.F.; Verri Jr, W.A.; Dos Santos, C.N.D.; Bordignon, J. The citrus flavanone naringenin impairs dengue virus replication in human cells. Sci. Rep. 2017, 7, 41864. [Google Scholar] [CrossRef]
- Flores, D.; Lee, L.; Adams, S. Inhibition of Curcumin-Treated Herpes Simplex Virus 1 and 2 in Vero Cells. Adv. Microbiol. 2016, 6, 276–287. [Google Scholar] [CrossRef]
- Praditya, D.; Kirchhoff, L.; Brüning, J.; Rachmawati, H.; Steinmann, J.; Steinmann, E. Anti-infective Properties of the Golden Spice Curcumin. Front. Microbiol. 2019, 10. [Google Scholar] [CrossRef] [PubMed]
- Ueda, K.; Kawabata, R.; Irie, T.; Nakai, Y.; Tohya, Y.; Sakaguchi, T. Inactivation of Pathogenic Viruses by Plant-Derived Tannins: Strong Effects of Extracts from Persimmon (Diospyros kaki) on a Broad Range of Viruses. PLoS ONE 2013, 8, e55343. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, J.L.S.; Lopes, R.; Oliveira, D.; Costa, S.; Miranda, M.M.F.S.; Romanos, M.T.V.; Santos, N.; Wigg, M. In vitro anti-rotavirus activity of some medicinal plants used in Brazil against diarrhea. J. Ethnopharmacol. 2005, 99, 403–407. [Google Scholar] [CrossRef] [PubMed]
- Gyuris, A.; Szlávik, L.; Minarovits, J.; Andrea, V.; Molnar, J.; Hohmann, J. Antiviral activities of extracts of Euphorbia hirta L. against HIV-1, HIV-2 and SIVmac251. In Vivo 2008, 23, 429–432. [Google Scholar]
- Moradi, M.-T.; Rafieian-Kopaei, M.; Karimi, A. A review study on the effect of Iranian herbal medicines against in vitro replication of herpes simplex virus. Avicenna J. Phytomedicine 2016, 6, 506. [Google Scholar]
- Lazreg Aref, H.; Gaaliche, B.; Fekih, A.; Mars, M.; Aouni, M.; Pierre Chaumon, J.; Said, K. In vitro cytotoxic and antiviral activities of Ficus carica latex extracts. Nat. Prod. Res. 2011, 25, 310–319. [Google Scholar] [CrossRef] [PubMed]
- Ashraf, A.; Ashraf, M.M.; Rafiqe, A.; Aslam, B.; Galani, S.; Zafar, S.; Asad, F.; Asghar, R.D.; Akram, S.; Ahmed, H. In vivo antiviral potential of Glycyrrhiza glabra extract against Newcastle disease virus. Pak. J. Pharm. Sci. 2017, 30. [Google Scholar]
- Wang, L.; Yang, R.; Yuan, B.; Liu, Y.; Liu, C. The antiviral and antimicrobial activities of licorice, a widely-used Chinese herb. Acta Pharmaceutica Sinica B 2015, 5. [Google Scholar] [CrossRef]
- Adianti, M.; Aoki, C.; Komoto, M.; Deng, L.; Shoji, I.; Wahyuni, T.S.; Lusida, M.I.; Fuchino, H.; Kawahara, N.; Hotta, H. Anti-hepatitis C virus compounds obtained from Glycyrrhiza uralensis and other Glycyrrhiza species. Microbiol. Immunol. 2014, 58, 180–187. [Google Scholar] [CrossRef]
- Alfajaro, M.M.; Kim, H.-J.; Park, J.-G.; Ryu, E.-H.; Kim, J.-Y.; Jeong, Y.-J.; Kim, D.-S.; Hosmillo, M.; Son, K.-Y.; Lee, J.-H. Anti-rotaviral effects of Glycyrrhiza uralensis extract in piglets with rotavirus diarrhea. Virol. J. 2012, 9, 310. [Google Scholar] [CrossRef] [PubMed]
- Allahverdiyev, A.M.; Bagirova, M.; Yaman, S.; Koc, R.C.; Abamor, E.S.; Ates, S.C.; Baydar, S.Y.; Elcicek, S.; Oztel, O.N. Development of new antiherpetic drugs based on plant compounds. In Fighting Multidrug Resistance with Herbal Extracts, Essential Oils and Their Components; Elsevier: Amsterdam, The Netherlands, 2013; pp. 245–259. [Google Scholar]
- Wang, J.; Hu, Y.; Wang, D.; Zhang, F.; Zhao, X.; Abula, S.; Fan, Y.; Guo, L. Lycium barbarum polysaccharide inhibits the infectivity of Newcastle disease virus to chicken embryo fibroblast. Int. J. Biol. Macromol. 2010, 46, 212–216. [Google Scholar] [CrossRef] [PubMed]
- Cheng, J.; Zhou, Z.-W.; Sheng, H.-P.; He, L.-J.; Fan, X.-W.; He, Z.-X.; Sun, T.; Zhang, X.; Zhao, R.; Gu, L.; et al. An evidence-based update on the pharmacological activities and possible molecular targets of Lycium barbarum polysaccharides. Drug Des. Dev. Ther. 2015, 9, 33–78. [Google Scholar] [CrossRef]
- Astani, A.; Reichling, J.; Schnitzler, P. Melissa officinalis extract inhibits attachment of herpes simplex virus in vitro. Chemotherapy 2012, 58, 70–77. [Google Scholar] [CrossRef]
- Parsania, M.; Rezaee, M.-B.; Monavari, S.H.; Jaimand, K.; Mousavi-Jazayeri, S.M.; Razazian, M.; Nadjarha, M.H. Antiviral screening of four plant extracts against acyclovir resistant herpes simplex virus type-1. Pak. J. Pharm. Sci. 2017, 30 (Suppl. 4), 1407–1411. [Google Scholar]
- Xu, H.-B.; Ma, Y.-B.; Huang, X.-Y.; Geng, C.-A.; Wang, H.; Zhao, Y.; Yang, T.-H.; Chen, X.-L.; Yang, C.-Y.; Zhang, X.-M. Bioactivity-guided isolation of anti-hepatitis B virus active sesquiterpenoids from the traditional Chinese medicine: Rhizomes of Cyperus rotundus. J. Ethnopharmacol. 2015, 171, 131–140. [Google Scholar] [CrossRef]
- Biswas, D.; Nandy, S.; Mukherjee, A.; Pandey, D.; Dey, A. Moringa oleifera Lam. and derived phytochemicals as promising antiviral agents: A review. South. Afr. J. Bot. 2019. [Google Scholar] [CrossRef]
- Sangar, V.; Samant, L.; Pawar, S.; Vaidya, S.; Chowdhary, A. In silico approach to combat HIV using phytoconstituents of Moringa oleifera Lam. J. Chem. Pharm. Res. 2015, 7, 997–1021. [Google Scholar]
- Abdel-Daim, M.M.; Abo-El-Sooud, K.; Aleya, L.; Bungau, S.G.; Najda, A.; Saluja, R. Alleviation of drugs and chemicals toxicity: Biomedical value of antioxidants. Oxid. Med. Cell. Longev. 2018, 2018, 6276438. [Google Scholar] [CrossRef]
- Salih, R.; Odisho, S.; Al-Shammari, A.; Ibrahim, O. Antiviral effects of olea europaea leaves extract and interferon-beta on gene expression of newcastle disease virus. Adv. Anim Vet. Sci. 2017, 5, 436–445. [Google Scholar] [CrossRef]
- Im, K.; Kim, J.; Min, H. Ginseng, the natural effectual antiviral: Protective effects of Korean Red Ginseng against viral infection. J. Ginseng Res. 2015, 40. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.-G.; Jin, Y.-H.; Lee, H.; Oh, T.; Yim, N.-H.; Cho, W.-K.; Ma, J. Protective Effect of Panax notoginseng Root Water Extract against Influenza A Virus Infection by Enhancing Antiviral Interferon-Mediated Immune Responses and Natural Killer Cell Activity. Front. Immunol. 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Lv, J.-J.; Yu, S.; Wang, Y.-F.; Wang, D.; Zhu, H.-T.; Cheng, R.-R.; Yang, C.-R.; Xu, M.; Zhang, Y.-J. Anti-hepatitis B virus norbisabolane sesquiterpenoids from Phyllanthus acidus and the establishment of their absolute configurations using theoretical calculations. J. Org. Chem. 2014, 79, 5432–5447. [Google Scholar] [CrossRef]
- Lv, J.-j.; Yu, S.; Xin, Y.; Cheng, R.-R.; Zhu, H.-T.; Yang, C.-R.; Zhang, Y.-J. Anti-viral and cytotoxic norbisabolane sesquiterpenoid glycosides from Phyllanthus emblica and their absolute configurations. Phytochemistry 2015, 117, 123–134. [Google Scholar] [CrossRef]
- Jiang, Z.Y.; Liu, W.F.; Zhang, X.M.; Luo, J.; Ma, Y.B.; Chen, J.J. Anti-HBV active constituents from Piper longum. Bioorg. Med. Chem. Lett. 2013, 23, 2123–2127. [Google Scholar] [CrossRef]
- Oh, C.; Price, J.; Brindley, M.A.; Widrlechner, M.P.; Qu, L.; McCoy, J.-A.; Murphy, P.; Hauck, C.; Maury, W. Inhibition of HIV-1 infection by aqueous extracts of Prunella vulgaris L. Virol. J. 2011, 8, 188. [Google Scholar] [CrossRef]
- Sriwilaijaroen, N.; Fukumoto, S.; Kumagai, K.; Hiramatsu, H.; Odagiri, T.; Tashiro, M.; Suzuki, Y. Antiviral effects of Psidium guajava Linn.(guava) tea on the growth of clinical isolated H1N1 viruses: Its role in viral hemagglutination and neuraminidase inhibition. Antivir. Res. 2012, 94, 139–146. [Google Scholar] [CrossRef]
- Naseer, S.; Hussain, S.; Naeem, N.; Pervaiz, M.; Rahman, M. The phytochemistry and medicinal value of Psidium guajava (guava). Clin. Phytoscience 2018, 4, 1–8. [Google Scholar] [CrossRef]
- Karimi, A.; Rafieian-Kopaei, M.; Moradi, M.-T.; Alidadi, S. Anti–Herpes Simplex Virus Type-1 Activity and Phenolic Content of Crude Ethanol Extract and Four Corresponding Fractions of Quercus brantii L Acorn. J. Evid. Based Complementary Altern. Med. 2017, 22, 455–461. [Google Scholar] [CrossRef]
- Romero-Pérez, G.A.; Egashira, M.; Harada, Y.; Tsuruta, T.; Oda, Y.; Ueda, F.; Tsukahara, T.; Tsukamoto, Y.; Inoue, R. Orally administered Salacia reticulata extract reduces H1N1 influenza clinical symptoms in murine lung tissues putatively due to enhanced natural killer cell activity. Front. Immunol. 2016, 7, 115. [Google Scholar] [CrossRef]
- Zhao, Z.; He, X.; Zhang, Q.; Wei, X.; Huang, L.; Fang, J.C.; Wang, X.; Zhao, M.; Bai, Y.; Zheng, X. Traditional Uses, Chemical Constituents and Biological Activities of Plants from the Genus Sanguisorba L. Am. J. Chin. Med. 2017, 45, 199–224. [Google Scholar] [CrossRef]
- Karkanis, A.C.; Fernandes, Â.; Vaz, J.; Petropoulos, S.; Georgiou, E.; Ciric, A.; Sokovic, M.; Oludemi, T.; Barros, L.; Ferreira, I.C. Chemical composition and bioactive properties of Sanguisorba minor Scop. under Mediterranean growing conditions. Food Funct. 2019, 10, 1340–1351. [Google Scholar] [CrossRef]
- Javed, T.; Ashfaq, U.; Riaz, S.; Rehman, S.; Riazuddin, S. In-vitro antiviral activity of Solanum nigrum against Hepatitis C Virus. Virol. J. 2011, 8. [Google Scholar] [CrossRef] [PubMed]
- Siqueira, E.; Lima, T.L.C.; Boff, L.; Lima, S.G.M.; Lourenço, E.M.G.; Ferreira, É.G.; Barbosa, E.G.; Machado, P.R.L.; Farias, K.J.S.; Ferreira, L.S.; et al. Antiviral Potential of Spondias mombin L. Leaves Extract Against Herpes Simplex Virus Type-1 Replication Using In Vitro and In Silico Approaches. Planta Med. 2020, 86, 505–515. [Google Scholar] [CrossRef] [PubMed]
- Rehman, S.; Bushra, I.; Fatima, N.; Syed, A.; Riazuddin, S. Therapeutic potential of Taraxacum officinale against HCV NS5B polymerase: In-vitro and In silico study. Biomed. Pharmacother. 2016, 83, 881–891. [Google Scholar] [CrossRef] [PubMed]
- Soleimani Farsani, M.; Behbahani, M.; Isfahani, H.Z. The Effect of Root, Shoot and Seed Extracts of The Iranian Thymus, L. (Family: Lamiaceae) Species on HIV-1 Replication and CD4 Expression. Cell J. 2016, 18, 255–261. [Google Scholar] [CrossRef] [PubMed]
- He, W.; Han, H.; Wang, W.; Gao, B. Anti-influenza virus effect of aqueous extracts from dandelion. Virol. J. 2011, 8, 1–11. [Google Scholar] [CrossRef]
- Bedoya, L.; Abad, M.; Sánchez-Palomino, S.; Alcami, J.; Bermejo, P. Ellagitannins from Tuberaria lignosa as entry inhibitors of HIV. Phytomedicine 2010, 17, 69–74. [Google Scholar] [CrossRef]
- Dai, J.-J.; Tao, H.-M.; Min, Q.-X.; Zhu, Q.-H. Anti-hepatitis B virus activities of friedelolactones from Viola diffusa Ging. Phytomedicine 2015, 22, 724–729. [Google Scholar] [CrossRef]
- Arabzadeh, A.M.; Ansari-Dogaheh, M.; Sharififar, F.; Shakibaie, M.; Heidarbeigi, M. Anti herpes simplex-1 activity of a standard extract of Zataria multiflora Boiss. Pak. J. Biol. Sci. PJBS 2013, 16, 180–184. [Google Scholar] [CrossRef]
- Kaushik, S.; Jangra, G.; Kundu, V.; Yadav, J.P.; Kaushik, S. Anti-viral activity of Zingiber officinale (Ginger) ingredients against the Chikungunya virus. Virus Dis. 2020, 1. [Google Scholar] [CrossRef]
- Semwal, D.K.; Semwal, R.B.; Combrinck, S.; Viljoen, A. Myricetin: A Dietary Molecule with Diverse Biological Activities. Nutrients 2016, 8, 90. [Google Scholar] [CrossRef] [PubMed]
- Qian, S.; Fan, W.; Qian, P.; Zhang, D.; Wei, Y.; Chen, H.; Li, X. Apigenin Restricts FMDV Infection and Inhibits Viral IRES Driven Translational Activity. Viruses 2015, 7, 1613–1626. [Google Scholar] [CrossRef]
- Zhang, W.; Qiao, H.; Lv, Y.; Wang, J.; Chen, X.; Hou, Y.; Tan, R.; Li, E. Apigenin Inhibits Enterovirus-71 Infection by Disrupting Viral RNA Association with trans-Acting Factors. PLoS ONE 2014, 9, e110429. [Google Scholar] [CrossRef]
- Ali, F.; Rahul; Naz, F.; Jyoti, S.; Siddique, Y.H. Health functionality of apigenin: A review. Int. J. Food Prop. 2017, 20, 1197–1238. [Google Scholar] [CrossRef]
- Wei, Y.; Guo, J.; Zheng, X.; Wu, J.; Zhou, Y.; Yu, Y.; Ye, Y.; Zhang, L.; Zhao, L. Preparation, pharmacokinetics and biodistribution of baicalin-loaded liposomes. Int. J. Nanomed. 2014, 9, 3623. [Google Scholar] [CrossRef]
- Moghaddam, E.; Teoh, B.-T.; Sam, S.-S.; Lani, R.; Hassandarvish, P.; Chik, Z.; Yueh, A.; Abu Bakar, S.; Zandi, K. Baicalin, a metabolite of baicalein with antiviral activity against dengue virus. Sci. Rep. 2014, 4, 5452. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Xu, Z.; Zheng, W. A Review of the Antiviral Role of Green Tea Catechins. Molecules 2017, 22, 1337. [Google Scholar] [CrossRef]
- Rodrigues, C.; Ascencao, K.; Silva, F.; Sarmento, B.; Oliveira, M.; Andrade, J. Drug-Delivery Systems of Green Tea Catechins for Improved Stability and Bioavailability. Curr. Med. Chem. 2013, 20. [Google Scholar] [CrossRef]
- Dong, Z.; Lu, X.; Tong, X.; Dong, Y.; Tang, L.; Liu, M. Forsythiae Fructus: A Review on its Phytochemistry, Quality Control, Pharmacology and Pharmacokinetics. Molecules 2017, 22, 1466. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.; Yin, A.; Shan, J.; Wang, S.; Cai, B.; Di, L. Study on the rationality for antiviral activity of Flos Lonicerae Japonicae-Fructus Forsythiae herb couple preparations improved by chito-oligosaccharide via integral pharmacokinetics. Molecules 2017, 22, 654. [Google Scholar] [CrossRef] [PubMed]
- Panraksa, P.; Ramphan, S.; Khongwichit, S.; Smith, D.R. Activity of andrographolide against dengue virus. Antivir. Res. 2017, 139, 69–78. [Google Scholar] [CrossRef]
- Kim, H.; Chung, M.S. Antiviral Activities of Mulberry (Morus alba) Juice and Seed against Influenza Viruses. Evid. Based Complementary Altern. Med. 2018, 2018, 2606583. [Google Scholar] [CrossRef]
- Mohammadi Pour, P.; Fakhri, S.; Asgary, S.; Farzaei, M.H.; Echeverría, J. The Signaling Pathways, and Therapeutic Targets of Antiviral Agents: Focusing on the Antiviral Approaches and Clinical Perspectives of Anthocyanins in the Management of Viral Diseases. Front. Pharmacol. 2019, 10. [Google Scholar] [CrossRef] [PubMed]
- Sharif, N.; Khoshnoudi-Nia, S.; Jafari, S.M. Nano/microencapsulation of anthocyanins; a systematic review and meta-analysis. Food Res. Int. 2020, 132, 109077. [Google Scholar] [CrossRef]
- Kannan, S.; Kolandaivel, P. The inhibitory performance of flavonoid cyanidin-3-sambubiocide against H274Y mutation in H1N1 influenza virus. J. Biomol. Struct. Dyn. 2018, 36, 4255–4269. [Google Scholar] [CrossRef]
- Loutfy, S.A.; Elberry, M.H.; Farroh, K.Y.; Mohamed, H.T.; Mohamed, A.A.; Mohamed, E.B.; Faraag, A.H.I.; Mousa, S.A. Antiviral Activity of Chitosan Nanoparticles Encapsulating Curcumin Against Hepatitis C Virus Genotype 4a in Human Hepatoma Cell Lines. Int. J. Nanomed. 2020, 15, 2699. [Google Scholar] [CrossRef]
- Nabila, N.; Suada, N.K.; Denis, D.; Yohan, B.; Adi, A.C.; Veterini, A.S.; Anindya, A.L.; Sasmono, R.T.; Rachmawati, H. Antiviral action of curcumin encapsulated in nanoemulsion against four serotypes of dengue virus. Pharm. Nanotechnol. 2020, 8, 54–62. [Google Scholar] [CrossRef]
- Sreekanth, T.; Nagajyothi, P.; Muthuraman, P.; Enkhtaivan, G.; Vattikuti, S.; Tettey, C.; Kim, D.H.; Shim, J.; Yoo, K. Ultra-sonication-assisted silver nanoparticles using Panax ginseng root extract and their anti-cancer and antiviral activities. J. Photochem. Photobiol. B Biol. 2018, 188, 6–11. [Google Scholar] [CrossRef]
- Houston, D.M.; Robins, B.; Bugert, J.J.; Denyer, S.P.; Heard, C.M. In vitro permeation and biological activity of punicalagin and zinc (II) across skin and mucous membranes prone to Herpes simplex virus infection. Eur. J. Pharm. Sci. 2017, 96, 99–106. [Google Scholar] [CrossRef]
- Tiralongo, E.; Wee, S.S.; Lea, R.A. Elderberry Supplementation Reduces Cold Duration and Symptoms in Air-Travellers: A Randomized, Double-Blind Placebo-Controlled Clinical Trial. Nutrients 2016, 8, 182. [Google Scholar] [CrossRef]
- Sadatrasul, M.; Fiezi, N.; Ghasemian, N.; Shenagari, M.; Saber, E.; Jazaeri, E.O.; Abdoli, A.; Jamali, A. Oil-in-water emulsion formulated with eucalyptus leaves extract inhibit influenza virus binding and replication in vitro. AIMS Microbiol. 2017, 3, 899–907. [Google Scholar] [CrossRef]
- Argenta, D.F.; Bidone, J.; Koester, L.S.; Bassani, V.L.; Simões, C.M.O.; Teixeira, H.F. Topical Delivery of Coumestrol from Lipid Nanoemulsions Thickened with Hydroxyethylcellulose for Antiherpes Treatment. AAPS Pharm. Sci. Tech. 2018, 19, 192–200. [Google Scholar] [CrossRef]
- Kapoor, R.; Sharma, B.; Kanwar, S. Antiviral Phytochemicals: An Overview. Biochem. Physiol. 2017, 06. [Google Scholar] [CrossRef]
- Ben-Shabat, S.; Yarmolinsky, L.; Porat, D.; Dahan, A. Antiviral effect of phytochemicals from medicinal plants: Applications and drug delivery strategies. Drug Deliv. Transl. Res. 2020, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Singh, G.; Pai, R.S. Recent advances of resveratrol in nanostructured based delivery systems and in the management of HIV/AIDS. J. Control. Release 2014, 194, 178–188. [Google Scholar] [CrossRef]
- Kerdudo, A.; Dingas, A.; Fernandez, X.; Faure, C. Encapsulation of rutin and naringenin in multilamellar vesicles for optimum antioxidant activity. Food Chem. 2014, 159, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Ripoli, M.; Angelico, R.; Sacco, P.; Ceglie, A.; Mangia, A. Phytoliposome-Based Silibinin Delivery System as a Promising Strategy to Prevent Hepatitis C Virus Infection. J. Biomed. Nanotechnol. 2016, 12, 770–780. [Google Scholar] [CrossRef]
- De Clercq, E. Recent highlights in the development of new antiviral drugs. Curr. Opin. Microbiol. 2005, 8, 552–560. [Google Scholar] [CrossRef]
- Kovačević, D.B.; Maras, M.; Barba, F.J.; Granato, D.; Roohinejad, S.; Mallikarjunan, K.; Montesano, D.; Lorenzo, J.M.; Putnik, P. Innovative technologies for the recovery of phytochemicals from Stevia rebaudiana Bertoni leaves: A review. Food Chem. 2018, 268, 513–521. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Z.; Li, S.; He, J.; Thirumdas, R.; Montesano, D.; Barba, F.J. Enzyme-assisted extraction of polyphenol from edible lotus (Nelumbo nucifera) rhizome knot: Ultra-filtration performance and HPLC-MS2 profile. Food Res. Int. 2018, 111, 291–298. [Google Scholar] [CrossRef] [PubMed]
- Chantrill, B.; Coulthard, C.; Dickinson, L.; Inkley, G.; Morris, W.; Pyle, A. The action of plant extracts on a bacteriophage of Pseudomonas pyocyanea and on influenza A virus. Microbiology 1952, 6, 74–84. [Google Scholar] [CrossRef] [PubMed]
- Zandi, K.; Teoh, B.-T.; Sam, S.-S.; Wong, P.-F.; Mustafa, M.R.; AbuBakar, S. Antiviral activity of four types of bioflavonoid against dengue virus type-2. Virol. J. 2011, 8, 560. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.; Son, M.; Ryu, E.; Shin, Y.S.; Kim, J.G.; Kang, B.W.; Sung, G.-H.; Cho, H.; Kang, H. Quercetin-induced apoptosis prevents EBV infection. Oncotarget 2015, 6, 12603. [Google Scholar] [CrossRef] [PubMed]
- Neznanov, N.; Kondratova, A.; Chumakov, K.M.; Neznanova, L.; Kondratov, R.; Banerjee, A.K.; Gudkov, A.V. Quercetinase pirin makes poliovirus replication resistant to flavonoid quercetin. DNA Cell Biol. 2008, 27, 191–198. [Google Scholar] [CrossRef]
- Wu, W.; Li, R.; Li, X.; He, J.; Jiang, S.; Liu, S.; Yang, J. Quercetin as an antiviral agent inhibits influenza A virus (IAV) entry. Viruses 2016, 8, 6. [Google Scholar] [CrossRef]
- McCutcheon, A.; Roberts, T.; Gibbons, E.; Ellis, S.; Babiuk, L.; Hancock, R.; Towers, G. Antiviral screening of British Columbian medicinal plants. J. Ethnopharmacol. 1995, 49, 101–110. [Google Scholar] [CrossRef]
- Jassim, S.A.A.; Naji, M.A. Novel antiviral agents: A medicinal plant perspective. J. Appl. Microbiol. 2003, 95, 412–427. [Google Scholar] [CrossRef]
- Clarke, R.W. Forces and structures of the herpes simplex virus (HSV) entry mechanism. ACS Infect. Dis. 2015, 1, 403–415. [Google Scholar] [CrossRef]
- Ryan, K.J.; Ray, C.G. Medical microbiology. McGraw Hill 2004, 4, 370. [Google Scholar]
- Corey, L.; Adams, H.G.; Brown, Z.A.; Holmes, K.K. Genital herpes simplex virus infections: Clinical manifestations, course, and complications. Ann. Intern. Med. 1983, 98, 958–972. [Google Scholar] [CrossRef]
- Ma, L.; Yao, L. Antiviral Effects of Plant-Derived Essential Oils and Their Components: An Updated Review. Molecules 2020, 25, 2627. [Google Scholar] [CrossRef]
- Arbab, A.H.; Parvez, M.K.; Al-Dosari, M.S.; Al-Rehaily, A.J. In vitro evaluation of novel antiviral activities of 60 medicinal plants extracts against hepatitis B virus. Exp. Ther. Med. 2017, 14, 626–634. [Google Scholar] [CrossRef]
- International Symposium on Medicinal Plants, A.-., 1993, in Philadelphia; U.S.A The Morris Arboretum and the World Health Organization (WHO): Geneva, Switzerland, 1993.
- Wenzel, U.; Kuntz, S.; Daniel, H. Flavonoids with epidermal growth factor-receptor tyrosine kinase inhibitory activity stimulate PEPT1-mediated cefixime uptake into human intestinal epithelial cells. J. Pharmacol. Exp. Ther. 2001, 299, 351–357. [Google Scholar]
- Guven, H.; Arici, A.; Simsek, O. Flavonoids in our foods: A short review. J. Basic Clin. Health Sci. 2019, 3, 96–106. [Google Scholar] [CrossRef]
- Airoldi, C.; La Ferla, B.; D’Orazio, G.; Ciaramelli, C.; Palmioli, A. Flavonoids in the treatment of Alzheimer’s and other neurodegenerative diseases. Curr. Med. Chem. 2018, 25, 3228–3246. [Google Scholar] [CrossRef] [PubMed]
- Siasos, G.; Tousoulis, D.; Tsigkou, V.; Kokkou, E.; Oikonomou, E.; Vavuranakis, M.; Basdra, E.; Papavassiliou, A.; Stefanadis, C. Flavonoids in atherosclerosis: An overview of their mechanisms of action. Curr. Med. Chem. 2013, 20, 2641–2660. [Google Scholar] [CrossRef] [PubMed]
- Panche, A.; Diwan, A.; Chandra, S. Flavonoids: An overview. J. Nutr. Sci. 2016, 5. [Google Scholar] [CrossRef]
- Qiu, T.; Wu, D.; Yang, L.; Ye, H.; Wang, Q.; Cao, Z.; Tang, K. Exploring the Mechanism of Flavonoids Through Systematic Bioinformatics Analysis. Front. Pharmacol. 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Pojer, E.; Mattivi, F.; Johnson, D.; Stockley, C.S. The Case for Anthocyanin Consumption to Promote Human Health: A Review. Compr. Rev. Food Sci. Food Saf. 2013, 12, 483–508. [Google Scholar] [CrossRef]
- Khoo, H.E.; Azlan, A.; Tang, S.T.; Lim, S.M. Anthocyanidins and anthocyanins: Colored pigments as food, pharmaceutical ingredients, and the potential health benefits. Food Nutr. Res. 2017, 61, 1361779. [Google Scholar] [CrossRef]
- Behl, T.; Kaur, I.; Bungau, S.; Kumar, A.; Uddin, M.S.; Kumar, C.; Pal, G.; Sahil; Shrivastava, K.; Zengin, G.; et al. The dual impact of ACE2 in COVID-19 and ironical actions in geriatrics and pediatrics with possible therapeutic solutions. Life Sci. 2020, 257, 118075. [Google Scholar] [CrossRef]
- Wu, H.-Y.; Yang, K.-M.; Chiang, P.-Y. Roselle Anthocyanins: Antioxidant Properties and Stability to Heat and pH. Molecules 2018, 23, 1357. [Google Scholar] [CrossRef]
- Amin, F.U.; Shah, S.A.; Badshah, H.; Khan, M.; Kim, M.O. Anthocyanins encapsulated by PLGA@PEG nanoparticles potentially improved its free radical scavenging capabilities via p38/JNK pathway against Aβ1–42-induced oxidative stress. J. Nanobiotechnol. 2017, 15, 12. [Google Scholar] [CrossRef]
- Ahsan, S.M.; Rao, C.M.; Ahmad, M.F. Nanoparticle-Protein Interaction: The Significance and Role of Protein Corona. Adv. Exp. Med. Biol. 2018, 1048, 175–198. [Google Scholar] [CrossRef]
- Rammohan, A.; Reddy, J.S.; Sravya, G.; Rao, C.N.; Zyryanov, G.V. Chalcone synthesis, properties and medicinal applications: A review. Environ. Chem. Lett. 2020, 18, 433–458. [Google Scholar] [CrossRef]
- Onyilagha, J.C.; Malhotra, B.; Elder, M.; French, C.J.; Towers, G.N. Comparative studies of inhibitory activities of chalcones on tomato ringspot virus (ToRSV). Can. J. Plant. Pathol. 1997, 19, 133–137. [Google Scholar] [CrossRef]
- Deng, J.; Kelley, J.A.; Barchi, J.J.; Sanchez, T.; Dayam, R.; Pommier, Y.; Neamati, N. Mining the NCI antiviral compounds for HIV-1 integrase inhibitors. Bioorg. Med. Chem. 2006, 14, 3785–3792. [Google Scholar] [CrossRef]
- Nathan, B.; Bayley, J.; Waters, L.; Post, F.A. Cobicistat: A Novel Pharmacoenhancer for Co-Formulation with HIV Protease and Integrase Inhibitors. Infect. Dis. Ther. 2013, 2, 111–122. [Google Scholar] [CrossRef]
- Mateeva, N.; Eyunni, S.V.K.; Redda, K.K.; Ononuju, U.; Hansberry, T.D.; Aikens, C.; Nag, A. Functional evaluation of synthetic flavonoids and chalcones for potential antiviral and anticancer properties. Bioorg. Med. Chem. Lett. 2017, 27, 2350–2356. [Google Scholar] [CrossRef]
- Phrutivorapongkul, A.; Lipipun, V.; Ruangrungsi, N.; Kirtikara, K.; Nishikawa, K.; Maruyama, S.; Watanabe, T.; Ishikawa, T. Studies on the chemical constituents of stem bark of Millettia leucantha: Isolation of new chalcones with cytotoxic, anti-herpes simplex virus and anti-inflammatory activities. Chem. Pharm. Bull. 2003, 51, 187–190. [Google Scholar] [CrossRef]
- Wang, Q.; Ding, Z.-H.; Liu, J.-K.; Zheng, Y.-T. Xanthohumol, a novel anti-HIV-1 agent purified from Hops Humulus lupulus. Antivir. Res. 2004, 64, 189–194. [Google Scholar] [CrossRef]
- Buckwold, V.E.; Wilson, R.J.; Nalca, A.; Beer, B.B.; Voss, T.G.; Turpin, J.A.; Buckheit, R.W., III; Wei, J.; Wenzel-Mathers, M.; Walton, E.M. Antiviral activity of hop constituents against a series of DNA and RNA viruses. Antiviral Res. 2004, 61, 57–62. [Google Scholar] [CrossRef]
- Mishra, L.; Itokawa, H.; Bastow, K.F.; Tachibana, Y.; Nakanishi, Y.; Kilgore, N.; Lee, K.-H.; Sinha, R. Anti-HIV and cytotoxic activities of Ru (II)/Ru (III) polypyridyl complexes containing 2, 6-(2′-benzimidazolyl)-pyridine/chalcone as co-ligand. Biorg. Med. Chem. 2001, 9, 1667–1671. [Google Scholar] [CrossRef]
- Likhitwitayawuid, K.; Chaiwiriya, S.; Sritularak, B.; Lipipun, V. Antiherpetic flavones from the heartwood of Artocarpus gomezianus. Chem. Biodivers. 2006, 3, 1138–1143. [Google Scholar] [CrossRef] [PubMed]
- Prendergast, P.T.U.P. Patent No 6555523. US Patent 6555523. 2003, 2003. [Google Scholar]
- Moriarty, R.; Surve, B.; Naithani, R.; Chandersekera, S.; Tiwari, V.; Shukla, D. Synthesis and Antiviral Activity of Abyssinone II Analogs, Papers, 233rd; ACS National Meeting: Chicago, IL, USA, 2007; pp. 25–29. [Google Scholar]
- Vlietinck, A.; De Bruyne, T.; Vanden Berghe, D. Plant substances as antiviral agents. Curr. Org. Chem. 1997, 1, 307–344. [Google Scholar] [CrossRef]
- Zakaryan, H.; Arabyan, E.; Oo, A.; Zandi, K. Flavonoids: Promising natural compounds against viral infections. Arch. Virol. 2017, 162, 2539–2551. [Google Scholar] [CrossRef] [PubMed]
- Yarmolinsky, L.; Huleihel, M.; Zaccai, M.; Ben-Shabat, S. Potent antiviral flavone glycosides from Ficus benjamina leaves. Fitoterapia 2012, 83, 362–367. [Google Scholar] [CrossRef]
- Schwarz, S.; Sauter, D.; Wang, K.; Zhang, R.; Sun, B.; Karioti, A.; Bilia, A.R.; Efferth, T.; Schwarz, W. Kaempferol derivatives as antiviral drugs against the 3a channel protein of coronavirus. Planta Med. 2014, 80, 177–182. [Google Scholar] [CrossRef] [PubMed]
- Seo, D.J.; Jeon, S.B.; Oh, H.; Lee, B.-H.; Lee, S.-Y.; Oh, S.H.; Jung, J.Y.; Choi, C. Comparison of the antiviral activity of flavonoids against murine norovirus and feline calicivirus. Food Control. 2016, 60, 25–30. [Google Scholar] [CrossRef]
- Ortega, J.T.; Suárez, A.I.; Serrano, M.L.; Baptista, J.; Pujol, F.H.; Rangel, H.R. The role of the glycosyl moiety of myricetin derivatives in anti-HIV-1 activity in vitro. AIDS Res. Ther. 2017, 14, 57. [Google Scholar] [CrossRef]
- Takatsuki, A.; Yamaguchi, I.; Tamura, G.; Misato, T.; Arima, K. Correlation between the anti-animal and anti-plant-virus activities of several antibiotics. J. Antibiot. 1969, 22, 442–445. [Google Scholar] [CrossRef][Green Version]
- Pallag, A.; Bungau, S.; Tit, D.M.; Jurca, T.; Sirbu, V.; Honiges, A.; Horhogea, C. Comparative Study of Polyphenols, Flavonoids and Chlorophylls in Equisetum arvense L. Populations. Rev. Chim. 2016, 67, 530–533. [Google Scholar]
- De Oliveira, A.; Adams, S.D.; Lee, L.H.; Murray, S.R.; Hsu, S.D.; Hammond, J.R.; Dickinson, D.; Chen, P.; Chu, T.-C. Inhibition of herpes simplex virus type 1 with the modified green tea polyphenol palmitoyl-epigallocatechin gallate. Food Chem. Toxicol. 2013, 52, 207–215. [Google Scholar] [CrossRef]
- Li, Y.; Leung, K.-T.; Yao, F.; Ooi, L.S.; Ooi, V.E. Antiviral flavans from the leaves of Pithecellobium c lypearia. J. Nat. Prod. 2006, 69, 833–835. [Google Scholar] [CrossRef] [PubMed]
- Charles, L.; Laure, F.; Raharivelomanana, P.; Bianchini, J.P. Sheath liquid interface for the coupling of normal-phase liquid chromatography with electrospray mass spectrometry and its application to the analysis of neoflavonoids. J. Mass Spectrom. 2005, 40, 75–82. [Google Scholar] [CrossRef] [PubMed]
- Hassan, M.Z.; Osman, H.; Ali, M.A.; Ahsan, M.J. Therapeutic potential of coumarins as antiviral agents. Eur. J. Med. Chem. 2016, 123, 236–255. [Google Scholar] [CrossRef] [PubMed]
- Mishra, S.; Pandey, A.; Manvati, S. Coumarin: An emerging antiviral agent. Heliyon 2020, 6, e03217. [Google Scholar] [CrossRef]
- Abba, Y.; Hassim, H.; Hamzah, H.; Noordin, M.M. Antiviral Activity of Resveratrol against Human and Animal Viruses. Adv. Virol. 2015, 2015, 184241. [Google Scholar] [CrossRef] [PubMed]
- Shankar, S.; Singh, G.; Srivastava, R.K. Chemoprevention by resveratrol: Molecular mechanisms and therapeutic potential. Front. Biosci. 2007, 12, 4839–4854. [Google Scholar] [CrossRef]
- Ranjbar, A.; Jamshidi, M.; Torabi, S. Molecular modelling of the antiviral action of Resveratrol derivatives against the activity of two novel SARS CoV-2 and 2019-nCoV receptors. Eur. Rev. Med. Pharmacol. Sci. 2020, 24, 7834–7844. [Google Scholar] [CrossRef]
- Ramdani, L.; Bachari, K. Potential therapeutic effects of Resveratrol against SARS-CoV-2. Acta Virol. 2020, 64, 276–280. [Google Scholar] [CrossRef]
- Huang, H.; Liao, D.; Zhou, G.; Zhu, Z.; Cui, Y.; Pu, R. Antiviral activities of resveratrol against rotavirus in vitro and in vivo. Phytomedicine 2020, 77, 153230. [Google Scholar] [CrossRef]
- Konigheim, B.; Goleniowski, M.; Contigiani, M. Cytotoxicity and antiviral activity of a lignan extracted from Larrea divaricata. Drug Des. Rev. Online 2005, 2, 81–83. [Google Scholar] [CrossRef]
- Rietjens, I.M.; Louisse, J.; Beekmann, K. The potential health effects of dietary phytoestrogens. Br. J. Pharmacol. 2017, 174, 1263–1280. [Google Scholar] [CrossRef]
- Cui, Q.; Du, R.; Liu, M.; Rong, L. Lignans and their derivatives from plants as antivirals. Molecules 2020, 25, 183. [Google Scholar] [CrossRef]
- Arbenz, A.; Avérous, L. Chemical modification of tannins to elaborate aromatic biobased macromolecular architectures. Green Chem. 2015, 17, 2626–2646. [Google Scholar] [CrossRef]
- Vilhelmova-Ilieva, N.; Galabov, A.S.; Mileva, M. Tannins as Antiviral Agents, Tannins-Structural Properties, Biological Properties and Current Knowledge, Alfredo Aires, IntechOpen, doi:10.5772/intechopen.86490. 2019. Available online: https://www.intechopen.com/books/tannins-structural-properties-biological-properties-and-current-knowledge/tannins-as-antiviral-agents (accessed on 20 February 2021).
- Saha, R.K.; Takahashi, T.; Kurebayashi, Y.; Fukushima, K.; Minami, A.; Kinbara, N.; Ichitani, M.; Sagesaka, Y.M.; Suzuki, T. Antiviral effect of strictinin on influenza virus replication. Antiviral. Res. 2010, 88, 10–18. [Google Scholar] [CrossRef]
- Wynn, G.H.; Zapor, M.J.; Smith, B.H.; Wortmann, G.; Oesterheld, J.R.; Armstrong, S.C.; Cozza, K.L. Antiretrovirals, Part 1: Overview, History, and Focus on Protease Inhibitors. Psychosomatics 2004, 45, 262–270. [Google Scholar] [CrossRef]
- Rathinavel, T.; Palanisamy, M.; Palanisamy, S.; Subramanian, A.; Thangaswamy, S. Phytochemical 6-Gingerol–A promising Drug of choice for COVID-19. Int. J. Adv. Sci. Eng. 2020, 6, 1482–1489. [Google Scholar] [CrossRef]
- Yang, M.; Lee, G.; Si, J.; Lee, S.-J.; You, H.J.; Ko, G. Curcumin shows antiviral properties against norovirus. Molecules 2016, 21, 1401. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Zhong, C.; Wang, Q.; Chen, W.; Yuan, Y. Curcumin is an APE1 redox inhibitor and exhibits an antiviral activity against KSHV replication and pathogenesis. Antivir. Res. 2019, 167, 98–103. [Google Scholar] [CrossRef] [PubMed]
- Prasad, S.; Tyagi, A.K. Curcumin and its analogues: A potential natural compound against HIV infection and AIDS. Food Funct. 2015, 6, 3412–3419. [Google Scholar] [CrossRef] [PubMed]
- Montesano, D.; Rocchetti, G.; Putnik, P.; Lucini, L. Bioactive profile of pumpkin: An overview on terpenoids and their health-promoting properties. Curr. Opin. Food Sci. 2018, 22, 81–87. [Google Scholar] [CrossRef]
- Borbone, N.; Borrelli, F.; Montesano, D.; Izzo, A.A.; Marino, S.D.; Capasso, R.; Zollo, F. Identification of a new sesquiterpene polyol ester from Celastrus paniculatus. Planta Med. 2007, 73, 792–804. [Google Scholar] [CrossRef]
- Borrelli, F.; Borbone, N.; Capasso, R.; Montesano, D.; De Marino, S.; Aviello, G.; Aprea, G.; Masone, S.; Izzo, A.A. Potent relaxant effect of a Celastrus paniculatus extract in the rat and human ileum. J. Ethnopharmacol. 2009, 122, 434–438. [Google Scholar] [CrossRef] [PubMed]
- Xiao, S.; Tian, Z.; Wang, Y.; Si, L.; Zhang, L.; Zhou, D. Recent progress in the antiviral activity and mechanism study of pentacyclic triterpenoids and their derivatives. Med. Res. Rev. 2018, 38, 951–976. [Google Scholar] [CrossRef]
- Yang, W.; Chen, X.; Li, Y.; Guo, S.; Wang, Z.; Yu, X. Advances in pharmacological activities of terpenoids. Nat. Prod. Commun. 2020, 15, 1934578X20903555. [Google Scholar] [CrossRef]
- Mapelli-Brahm, P.; Barba, F.J.; Remize, F.; Garcia, C.; Fessard, A.; Mousavi Khaneghah, A.; Sant’Ana, A.S.; Lorenzo, J.M.; Montesano, D.; Meléndez-Martínez, A.J. The impact of fermentation processes on the production, retention and bioavailability of carotenoids: An overview. Trends Food Sci. Technol. 2020, 99, 389–401. [Google Scholar] [CrossRef]
- Hammond, B., Jr.; Renz, L. Carotenoids. Adv. Nutr. 2013, 4, 474–476. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Concepcion, M.; Avalos, J.; Bonet, M.L.; Boronat, A.; Gomez-Gomez, L.; Hornero-Mendez, D.; Limon, M.C.; Meléndez-Martínez, A.J.; Olmedilla-Alonso, B.; Palou, A.; et al. A global perspective on carotenoids: Metabolism, biotechnology, and benefits for nutrition and health. Prog Lipid Res. 2018, 70, 62–93. [Google Scholar] [CrossRef]
- Pang, R.; Tao, J.-Y.; Zhang, S.-L.; Zhao, L.; Yue, X.; Wang, Y.-F.; Ye, P.; Dong, J.-H.; Zhu, Y.; Wu, J.-G. In vitro antiviral activity of lutein against hepatitis B virus. Phytother. Res. 2010, 24, 1627–1630. [Google Scholar] [CrossRef] [PubMed]
- Azimova, S. Natural Compounds: Plant. Sources, Structure and Properties; Springer: New York, NY, USA, 2013. [Google Scholar]
- Aniszewski, T. Chapter 1: Definition, typology, and occurrence of alkaloids. In Alkaloids—Secrets of Life: Alkaloid Chemistry, Biological Significance, Applications and Ecological Role, 2nd ed.; Elsevier BV: Amsterdam, The Netherlands, 2015. [Google Scholar]
- Houghton, P.J. Chemistry and biological activity of natural and semi-synthetic chromone alkaloids. In Studies in Natural Products Chemistry; Elsevier: Amsterdam, The Netherlands, 2000; Volume 21, pp. 123–155. [Google Scholar]
- Wen, C.-C.; Kuo, Y.-H.; Jan, J.-T.; Liang, P.-H.; Wang, S.-Y.; Liu, H.-G.; Lee, C.-K.; Chang, S.-T.; Kuo, C.-J.; Lee, S.-S. Specific plant terpenoids and lignoids possess potent antiviral activities against severe acute respiratory syndrome coronavirus. J. Med. Chem. 2007, 50, 4087–4095. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Zhang, X.; Liu, P.; Shen, X.; Lan, T.; Li, W.; Jiang, Q.; Xie, X.; Huang, H. Effects of berberine on matrix accumulation and NF-kappa B signal pathway in alloxan-induced diabetic mice with renal injury. Eur. J. Pharmacol. 2010, 638, 150–155. [Google Scholar] [CrossRef]
- Suryavanshi, S.; Kulkarni, Y. NF-κβ: A potential target in the management of vascular complications of diabetes. Front. Pharmacol. 2017. [Google Scholar] [CrossRef] [PubMed]
- Yan, Y.Q.; Fu, Y.J.; Wu, S.; Qin, H.Q.; Zhen, X.; Song, B.M.; Weng, Y.S.; Wang, P.C.; Chen, X.Y.; Jiang, Z.Y. Anti-influenza activity of berberine improves prognosis by reducing viral replication in mice. Phytother. Res. 2018, 32, 2560–2567. [Google Scholar] [CrossRef] [PubMed]
- Thawabteh, A.; Juma, S.; Bader, M.; Karaman, D.; Scrano, L.; Bufo, S.A.; Karaman, R. The Biological Activity of Natural Alkaloids against Herbivores, Cancerous Cells and Pathogens. Toxins 2019, 11, 656. [Google Scholar] [CrossRef]
- Chiu, F.-L.; Lin, J.-K. Tomatidine inhibits iNOS and COX-2 through suppression of NF-κB and JNK pathways in LPS-stimulated mouse macrophages. FEBS Lett. 2008, 582, 2407–2412. [Google Scholar] [CrossRef]
- Ebert, S.M.; Dyle, M.C.; Bullard, S.A.; Dierdorff, J.M.; Murry, D.J.; Fox, D.K.; Bongers, K.S.; Lira, V.A.; Meyerholz, D.K.; Talley, J.J. Identification and small molecule inhibition of an activating transcription factor 4 (ATF4)-dependent pathway to age-related skeletal muscle weakness and atrophy. J. Biol. Chem. 2015, 290, 25497–25511. [Google Scholar] [CrossRef]
- Yan, K.-H.; Lee, L.-M.; Yan, S.-H.; Huang, H.-C.; Li, C.-C.; Lin, H.-T.; Chen, P.-S. Tomatidine inhibits invasion of human lung adenocarcinoma cell A549 by reducing matrix metalloproteinases expression. Chem. Biol. Interact. 2013, 203, 580–587. [Google Scholar] [CrossRef]
- Diosa-Toro, M.; Troost, B.; Van De Pol, D.; Heberle, A.M.; Urcuqui-Inchima, S.; Thedieck, K.; Smit, J.M. Tomatidine, a novel antiviral compound towards dengue virus. Antivir. Res. 2019, 161, 90–99. [Google Scholar] [CrossRef] [PubMed]
- Troost, B.; Mulder, L.M.; Diosa-Toro, M.; van de Pol, D.; Rodenhuis-Zybert, I.A.; Smit, J.M. Tomatidine, a natural steroidal alkaloid shows antiviral activity towards chikungunya virus in vitro. Sci. Rep. 2020, 10, 6364. [Google Scholar] [CrossRef]
- Sharma, N. Efficacy of garlic and onion against virus. Int. J. Res. Pharm. Sci 2019, 10, 3578–3586. [Google Scholar] [CrossRef]
- Hudson, B.S. Polyacetylene: Myth and Reality. Materials 2018, 11, 242. [Google Scholar] [CrossRef]
- Naithani, R.; Mehta, R.G.; Shukla, D.; Chandersekera, S.N.; Moriarty, R.M. Antiviral Activity of Phytochemicals: A Current Perspective. In Dietary Components and Immune Function; Watson, R.R., Zibadi, S., Preedy, V.R., Eds.; Humana Press: Totowa, NJ, USA, 2010; pp. 421–468. [Google Scholar]
- Ahmadi, A.; Zorofchian Moghadamtousi, S.; Abubakar, S.; Zandi, K. Antiviral Potential of Algae Polysaccharides Isolated from Marine Sources: A Review. BioMed Res. Int. 2015, 2015, 825203. [Google Scholar] [CrossRef] [PubMed]
- Sansone, C.; Brunet, C.; Noonan, D.M.; Albini, A. Marine Algal Antioxidants as Potential Vectors for Controlling Viral Diseases. Antioxidants 2020, 9, 392. [Google Scholar] [CrossRef]
- Huleihel, M.; Ishanu, V.; Tal, J.; Arad, S.M. Antiviral effect of red microalgal polysaccharides on Herpes simplex and Varicella zoster viruses. J. Appl. Phycol. 2001, 13, 127–134. [Google Scholar] [CrossRef]
- Malik, E.M.; Müller, C.E. Anthraquinones as pharmacological tools and drugs. Med. Res. Rev. 2016, 36, 705–748. [Google Scholar] [CrossRef]
- Duval, J.; Pecher, V.; Poujol, M.; Lesellier, E. Research advances for the extraction, analysis and uses of anthraquinones: A review. Ind. Crops Prod. 2016, 94, 812–833. [Google Scholar] [CrossRef]
- Singh, A. Herbal Drugs as Therapeutic Agents; CRC Press: Boca Raton, FL, USA, 2014. [Google Scholar]
- Ho, T.-Y.; Wu, S.-L.; Chen, J.-C.; Li, C.-C.; Hsiang, C.-Y. Emodin blocks the SARS coronavirus spike protein and angiotensin-converting enzyme 2 interaction. Antivir. Res. 2007, 74, 92–101. [Google Scholar] [CrossRef]
- Chrubasik, S.; Pittler, M.; Roufogalis, B. Zingiberis rhizoma: A comprehensive review on the ginger effect and efficacy profiles. Phytomedicine 2005, 12, 684–701. [Google Scholar] [CrossRef]
- Ding, P.; Ding, Y. Stories of Salicylic Acid: A Plant Defense Hormone. Trends Plant. Sci. 2020, 25, 549–565. [Google Scholar] [CrossRef] [PubMed]
- Singh, D.P.; Moore, C.A.; Gilliland, A.; Carr, J.P. Activation of multiple antiviral defence mechanisms by salicylic acid. Mol. Plant. Pathol. 2004, 5, 57–63. [Google Scholar] [CrossRef]
- Campos, L.; Granell, P.; Tárraga, S.; López-Gresa, P.; Conejero, V.; Bellés, J.M.; Rodrigo, I.; Lisón, P. Salicylic acid and gentisic acid induce RNA silencing-related genes and plant resistance to RNA pathogens. Plant. Physiol. Biochem. 2014, 77, 35–43. [Google Scholar] [CrossRef]
- Botelho, M.; Orlandi, J.; De Melo, F.; Mantovani, M.; Linhares, R.; Nozawa, C. Chlorophyllin protects HEp-2 cells from nuclear fragmentation induced by poliovirus. Lett. Appl. Microbiol. 2004, 39, 174–177. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.S.; Ko, E.-J.; Hwang, H.S.; Lee, Y.-N.; Kwon, Y.-M.; Kim, M.-C.; Kang, S.-M. Antiviral activity of ginseng extract against respiratory syncytial virus infection. Int. J. Mol. Med. 2014, 34, 183–190. [Google Scholar] [CrossRef]
- Cho, A.; Roh, Y.S.; Uyangaa, E.; Park, S.; Kim, J.W.; Lim, K.H.; Kwon, J.; Eo, S.K.; Lim, C.W.; Kim, B. Protective effects of red ginseng extract against vaginal herpes simplex virus infection. J. Ginseng Res. 2013, 37, 210. [Google Scholar] [CrossRef]
- Saleem, S.; Muhammad, G.; Hussain, M.A.; Altaf, M.; Bukhari, S.N.A. Withania somnifera L.: Insights into the phytochemical profile, therapeutic potential, clinical trials, and future prospective. Iran. J. Basic Med. Sci. 2020, 23, 1501–1526. [Google Scholar] [CrossRef]
- Chiang, L.C.; Ng, L.T.; Cheng, P.W.; Chiang, W.; Lin, C.C. Antiviral activities of extracts and selected pure constituents of Ocimum basilicum. Clin. Exp. Pharmacol. Physiol. 2005, 32, 811–816. [Google Scholar] [CrossRef]
- Gilling, D.H.; Kitajima, M.; Torrey, J.; Bright, K.R. Antiviral efficacy and mechanisms of action of oregano essential oil and its primary component carvacrol against murine norovirus. J. Appl. Microbiol. 2014, 116, 1149–1163. [Google Scholar] [CrossRef]
- Kubala, J. Impressive Herbs with Antiviral Activity. Nutr. Oct. 2019, 21, 2019. [Google Scholar]
- Pilau, M.R.; Alves, S.H.; Weiblen, R.; Arenhart, S.; Cueto, A.P.; Lovato, L.T. Antiviral activity of the Lippia graveolens (Mexican oregano) essential oil and its main compound carvacrol against human and animal viruses. Braz. J. Microbiol. 2011, 42, 1616–1624. [Google Scholar] [CrossRef] [PubMed]
- Badgujar, S.B.; Patel, V.V.; Bandivdekar, A.H. Foeniculum vulgare Mill: A review of its botany, phytochemistry, pharmacology, contemporary application, and toxicology. BioMed Res. Int. 2014, 2014. [Google Scholar] [CrossRef]
- Orhan, İ.E.; Özçelik, B.; Kartal, M.; Kan, Y. Antimicrobial and antiviral effects of essential oils from selected Umbelliferae and Labiatae plants and individual essential oil components. Turk. J. Biol. 2012, 36, 239–246. [Google Scholar]
- Dehghani, F.; Merat, A.; Panjehshahin, M.R.; Handjani, F. Healing effect of garlic extract on warts and corns. Int. J. Dermatol. 2005, 44, 612–615. [Google Scholar] [CrossRef]
- Thuy, B.T.P.; My, T.T.A.; Hai, N.T.T.; Hieu, L.T.; Hoa, T.T.; Thi Phuong Loan, H.; Triet, N.T.; Anh, T.T.V.; Quy, P.T.; Tat, P.V.; et al. Investigation into SARS-CoV-2 Resistance of Compounds in Garlic Essential Oil. ACS Omega 2020, 5, 8312–8320. [Google Scholar] [CrossRef] [PubMed]
- Kabir, M.T.; Uddin, M.S.; Hossain, M.F.; Abdulhakim, J.A.; Alam, M.A.; Ashraf, G.M.; Bungau, S.; Bin-Jumah, M.N.; Abdel-Daim, M.M.; Aleya, L. nCOVID-19 Pandemic from Molecular Pathogenesis to Potential Investigational Therapeutics. Front. Cell Dev. Biol. 2020, 8, 616. [Google Scholar] [CrossRef]
- Ahmed, I.; Aslam, A.; Mustafa, G.; Masood, S.; Ali, M.A.; Nawaz, M. Anti-avian influenza virus H9N2 activity of aqueous extracts of Zingiber officinalis (Ginger) and Allium sativum (Garlic) in chick embryos. Pak. J. Pharm. Sci. 2017, 30, 1341–1344. [Google Scholar]
- Schnitzler, P.; Koch, C.; Reichling, J. Susceptibility of drug-resistant clinical herpes simplex virus type 1 strains to essential oils of ginger, thyme, hyssop, and sandalwood. Antimicrob. Agents Chemother. 2007, 51, 1859–1862. [Google Scholar] [CrossRef]
- Li, Y.; Liu, Y.; Ma, A.; Bao, Y.; Wang, M.; Sun, Z. In vitro antiviral, anti-inflammatory, and antioxidant activities of the ethanol extract of Mentha piperita L. Food Sci. Biotechnol. 2017, 26, 1675–1683. [Google Scholar] [CrossRef] [PubMed]
- McKay, D.L.; Blumberg, J.B. A review of the bioactivity and potential health benefits of peppermint tea (Mentha piperita L.). Phytother. Res. 2006, 20, 619–633. [Google Scholar] [CrossRef] [PubMed]
- Kinoshita, E.; Hayashi, K.; Katayama, H.; Hayashi, T.; Obata, A. Anti-influenza virus effects of elderberry juice and its fractions. Biosci. Biotechnol. Biochem. 2012, 12011. [Google Scholar] [CrossRef]
- Hawkins, J.; Baker, C.; Cherry, L.; Dunne, E. Black elderberry (Sambucus nigra) supplementation effectively treats upper respiratory symptoms: A meta-analysis of randomized, controlled clinical trials. Complement. Ther. Med. 2019, 42, 361–365. [Google Scholar] [CrossRef]
- Cinatl, J.; Morgenstern, B.; Bauer, G.; Chandra, P.; Rabenau, H.; Doerr, H.W. Glycyrrhizin, an active component of liquorice roots, and replication of SARS-associated coronavirus. Lancet 2003, 361, 2045–2046. [Google Scholar] [CrossRef]
- Michaelis, M.; Geiler, J.; Naczk, P.; Sithisarn, P.; Ogbomo, H.; Altenbrandt, B.; Leutz, A.; Doerr, H.W.; Cinatl, J., Jr. Glycyrrhizin inhibits highly pathogenic H5N1 influenza A virus-induced pro-inflammatory cytokine and chemokine expression in human macrophages. Med. Microbiol. Immunol. 2010, 199, 291–297. [Google Scholar] [CrossRef]
- Marin, R.-C.; Behl, T.; Negrut, N.; Bungau, S. Management of Antiretroviral Therapy with Boosted Protease Inhibitors—Darunavir/Ritonavir or Darunavir/Cobicistat. Biomedicines 2021, 9, 313. [Google Scholar] [CrossRef] [PubMed]
- Fukuchi, K.; Okudaira, N.; Adachi, K.; Odai-Ide, R.; Watanabe, S.; Ohno, H.; Yamamoto, M.; Kanamoto, T.; Terakubo, S.; Nakashima, H. Antiviral and antitumor activity of licorice root extracts. In Vivo 2016, 30, 777–785. [Google Scholar] [CrossRef]
- Yan, W.; Zhang, C.; Li, B.; Xu, X.; Liang, M.; Gu, S.; Chu, F.; Xu, B.; Ren, J.; Wang, P.; et al. A Series of Oleanolic Acid Derivatives as Anti-Hepatitis B Virus Agents: Design, Synthesis, and in Vitro and in Vivo Biological Evaluation. Molecules 2016, 21, 402. [Google Scholar] [CrossRef]
- Mukherjee, H.; Ojha, D.; Bag, P.; Chandel, H.; Bhattacharyya, S.; Chatterjee, T.; Mukherjee, P.; Chakraborti, S.; Debprasad, C. Anti-herpes virus activities of Achyranthes aspera: An Indian ethnomedicine, and its triterpene acid. Microbiol. Res. 2012, 168. [Google Scholar] [CrossRef]
- Battistini, R.; Rossini, I.; Ercolini, C.; Goria, M.; Callipo, M.R.; Maurella, C.; Pavoni, E.; Serracca, L. Antiviral Activity of Essential Oils Against Hepatitis A Virus in Soft Fruits. Food Environ. Virol. 2019, 11, 90–95. [Google Scholar] [CrossRef] [PubMed]
- Domenico, M.; Lina, C.; Francesca, B. Sustainable crops for food security: Moringa (Moringa oleifera Lam.). Encycl. Food Secur. Sustain. 2019, 1, 409–415. [Google Scholar]
- Falowo, A.B.; Mukumbo, F.E.; Idamokoro, E.M.; Lorenzo, J.M.; Afolayan, A.J.; Muchenje, V. Multi-functional application of Moringa oleifera Lam. in nutrition and animal food products: A review. Food Res. Int. 2018, 106, 317–334. [Google Scholar] [CrossRef] [PubMed]
- Anwar, F.; Latif, S.; Ashraf, M.; Gilani, A.H. Moringa oleifera: A food plant with multiple medicinal uses. Phytother. Res. 2007, 21, 17–25. [Google Scholar] [CrossRef]
- Nandave, M.; Ojha, S.K.; Joshi, S.; Kumari, S.; Arya, D.S. Moringa oleifera leaf extract prevents isoproterenol-induced myocardial damage in rats: Evidence for an antioxidant, antiperoxidative, and cardioprotective intervention. J. Med. Food 2009, 12, 47–55. [Google Scholar] [CrossRef]
- Parvathy, M.; Umamaheshwari, A. Cytotoxic effect of Moringa oleifera leaf extracts on human multiple myeloma cell lines. Trends Med. Res. 2007, 2, 44–50. [Google Scholar]
- Muangnoi, C.; Chingsuwanrote, P.; Praengamthanachoti, P.; Svasti, S.; Tuntipopipat, S. Moringa oleifera pod inhibits inflammatory mediator production by lipopolysaccharide-stimulated RAW 264.7 murine macrophage cell lines. Inflammation 2012, 35, 445–455. [Google Scholar] [CrossRef]
- Ndong, M.; Uehara, M.; Katsumata, S.-i.; Suzuki, K. Effects of oral administration of Moringa oleifera Lam on glucose tolerance in Goto-Kakizaki and Wistar rats. J. Clin. Biochem. Nutr. 2007, 40, 229–233. [Google Scholar] [CrossRef] [PubMed]
- Pari, L.; Kumar, N.A. Hepatoprotective activity of Moringa oleifera on antitubercular drug-induced liver damage in rats. J. Med. Food 2002, 5, 171–177. [Google Scholar] [CrossRef]
- Nworu, C.; Okoye, E.; Ezeifeka, G.; Esimone, C. Extracts of Moringa oleifera Lam. showing inhibitory activity against early steps in the infectivity of HIV-1 lentiviral particles in a viral vector-based screening. Afr. J. Biotechnol. 2013, 12. [Google Scholar] [CrossRef]
- Feustel, S.; Ayón-Pérez, F.; Sandoval-Rodriguez, A.; Rodríguez-Echevarría, R.; Contreras-Salinas, H.; Armendáriz-Borunda, J.; Sánchez-Orozco, L. Protective effects of Moringa oleifera on HBV genotypes C and H transiently transfected Huh7 cells. J. Immunol. Res. 2017, 2017. [Google Scholar] [CrossRef]
- Nasr-Eldin, M.A.; Abdelhamid, A.; Baraka, D. Antibiofilm and Antiviral Potential of Leaf Extracts from Moringa oleifera and Rosemary (Rosmarinus officinalis Lam.). Egypt. J. Microbiol. 2017, 52, 129–139. [Google Scholar] [CrossRef]
- Karimi, M.; Sadeghi, R.; Kokini, J. Pomegranate as a promising opportunity in medicine and nanotechnology. Trends Food Sci. Technol. 2017, 69, 59–73. [Google Scholar] [CrossRef]
- Viuda-Martos, M.; Fernández-López, J.; Pérez-Álvarez, J.A. Pomegranate and its Many Functional Components as Related to Human Health: A Review. Compr. Rev. Food Sci. Food Saf. 2010, 9, 635–654. [Google Scholar] [CrossRef] [PubMed]
- Arunkumar, J.; Rajarajan, S. Study on antiviral activities, drug-likeness and molecular docking of bioactive compounds of Punica granatum L. to Herpes simplex virus-2 (HSV-2). Microb. Pathog. 2018, 118, 301–309. [Google Scholar] [CrossRef] [PubMed]
- Moradi, M.-T.; Karimi, A.; Shahrani, M.; Hashemi, L.; Ghaffari-Goosheh, M.-S. Anti-Influenza Virus Activity and Phenolic Content of Pomegranate (Punica granatum L.) Peel Extract and Fractions. Avicenna. J. Med. Biotechnol. 2019, 11, 285–291. [Google Scholar]
- Houston, D.M.J.; Bugert, J.J.; Denyer, S.P.; Heard, C.M. Potentiated virucidal activity of pomegranate rind extract (PRE) and punicalagin against Herpes simplex virus (HSV) when co-administered with zinc (II) ions, and antiviral activity of PRE against HSV and aciclovir-resistant HSV. PLoS ONE 2017, 12, e0179291. [Google Scholar] [CrossRef]
- Kaume, L.; Howard, L.R.; Devareddy, L. The Blackberry Fruit: A Review on Its Composition and Chemistry, Metabolism and and bioavailability, and health benefits. J. Agric. Food. Chem. 2012, 13, 5716–5727. [Google Scholar] [CrossRef]
- Tomas, M.; Rocchetti, G.; Ghisoni, S.; Giuberti, G.; Capanoglu, E.; Lucini, L. Effect of different soluble dietary fibres on the phenolic profile of blackberry puree subjected to in vitro gastrointestinal digestion and large intestine fermentation. Food Res. Int. 2020, 130, 108954. [Google Scholar] [CrossRef]
- Danaher, R.J.; Wang, C.; Dai, J.; Mumper, R.J.; Miller, C.S. Antiviral effects of blackberry extract against herpes simplex virus type 1. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontology 2011, 112, 31–35. [Google Scholar] [CrossRef][Green Version]
- Nikolaeva-Glomb, L.; Mukova, L.; Nikolova, N.; Badjakov, I.; Dincheva, I.; Kondakova, V.; Doumanova, L.; Galabov, A. In Vitro Antiviral Activity of a Series of Wild Berry Fruit Extracts against Representatives of Picorna-, Orthomyxo- and Paramyxoviridae. Nat. Prod. Commun. 2014, 9, 51–54. [Google Scholar] [CrossRef] [PubMed]
- Le Douce, V.; Janossy, A.; Hallay, H.; Ali, S.; Riclet, R.; Rohr, O.; Schwartz, C. Achieving a cure for HIV infection: Do we have reasons to be optimistic? J. Antimicrob. Chemother. 2012, 67, 1063–1074. [Google Scholar] [CrossRef] [PubMed]
- Vieira da Silva, B.; Barreira, J.C.M.; Oliveira, M.B.P.P. Natural phytochemicals and probiotics as bioactive ingredients for functional foods: Extraction, biochemistry and protected-delivery technologies. Trends Food Sci. Technol. 2016, 50, 144–158. [Google Scholar] [CrossRef]
- Wang, S.; Su, R.; Nie, S.; Sun, M.; Zhang, J.; Wu, D.; Moustaid-Moussa, N. Application of nanotechnology in improving bioavailability and bioactivity of diet-derived phytochemicals. J. Nutr. Biochem. 2014, 25, 363–376. [Google Scholar] [CrossRef]
- Armendáriz-Barragán, B.; Zafar, N.; Badri, W.; Galindo-Rodríguez, S.A.; Kabbaj, D.; Fessi, H.; Elaissari, A. Plant extracts: From encapsulation to application. Expert Opin. Drug Deliv. 2016, 13, 1165–1175. [Google Scholar] [CrossRef] [PubMed]
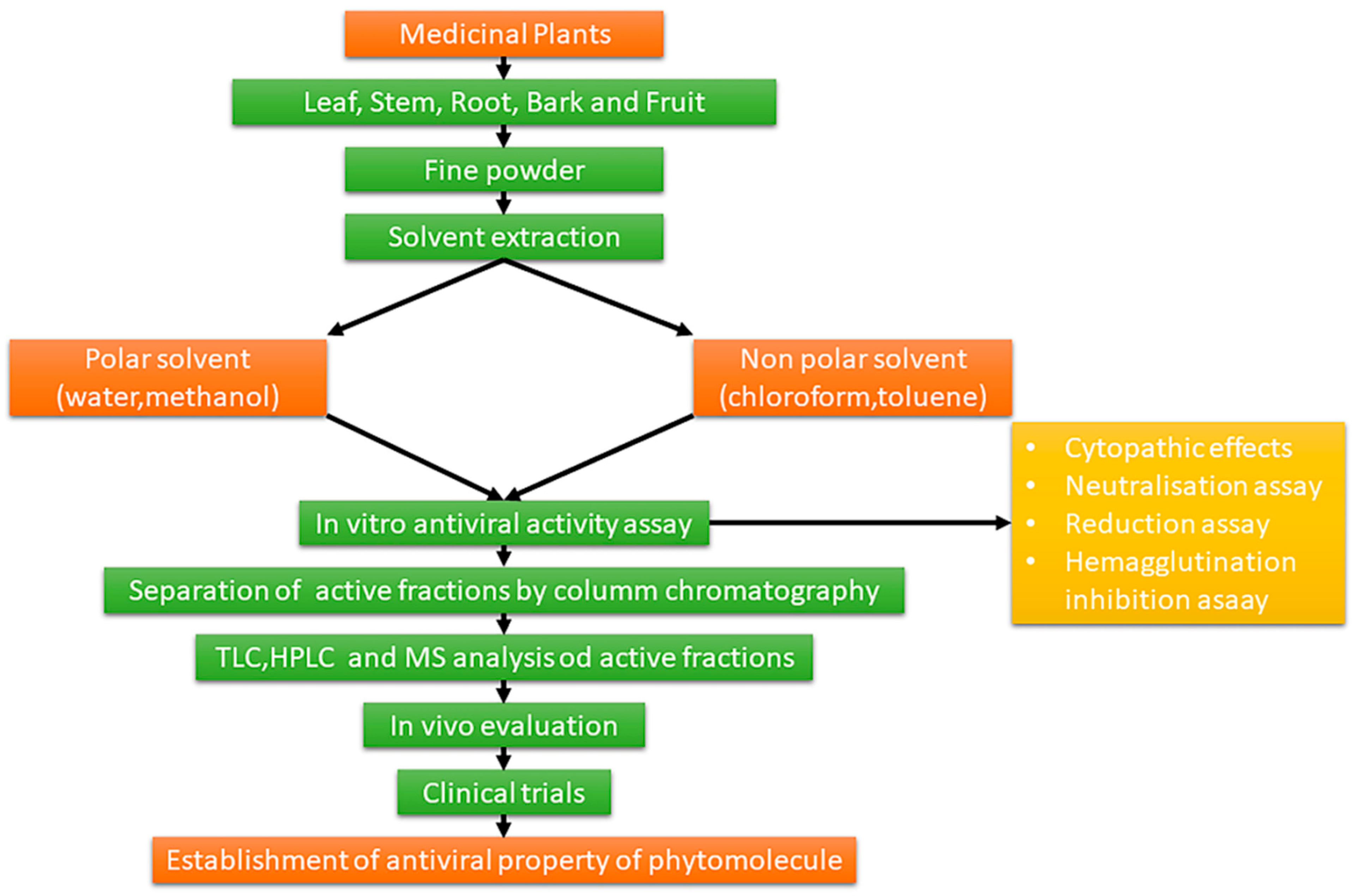
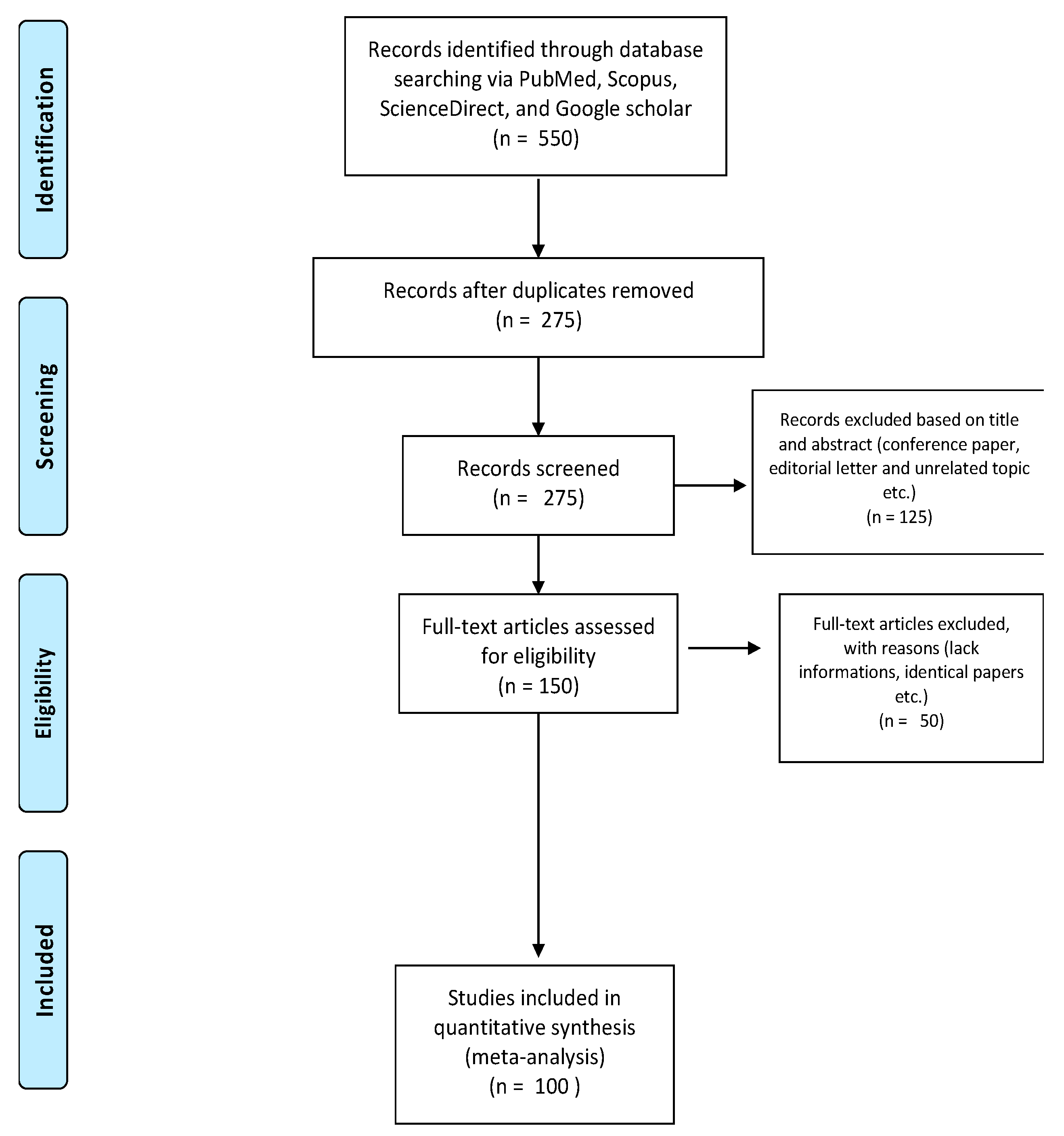
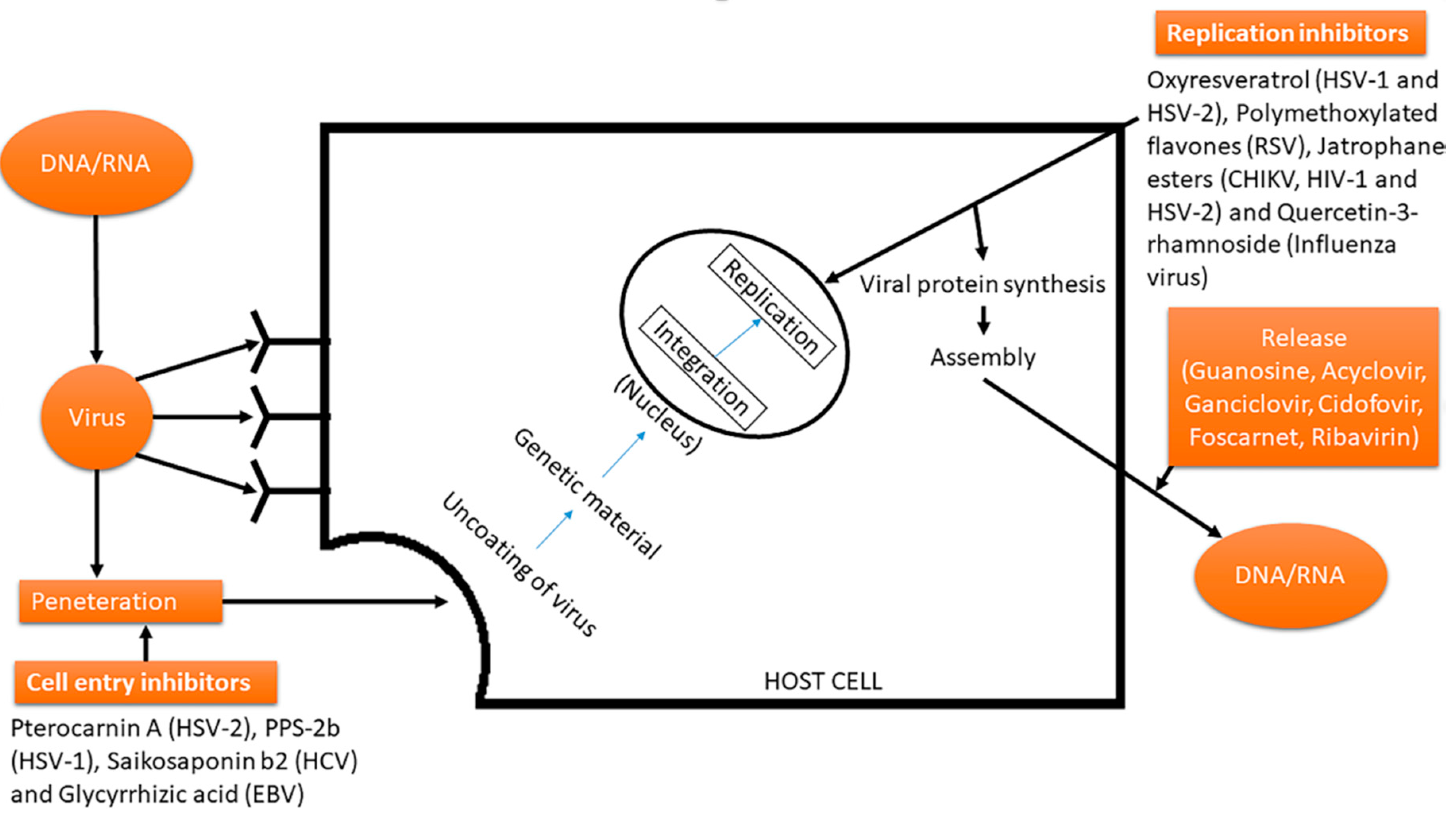

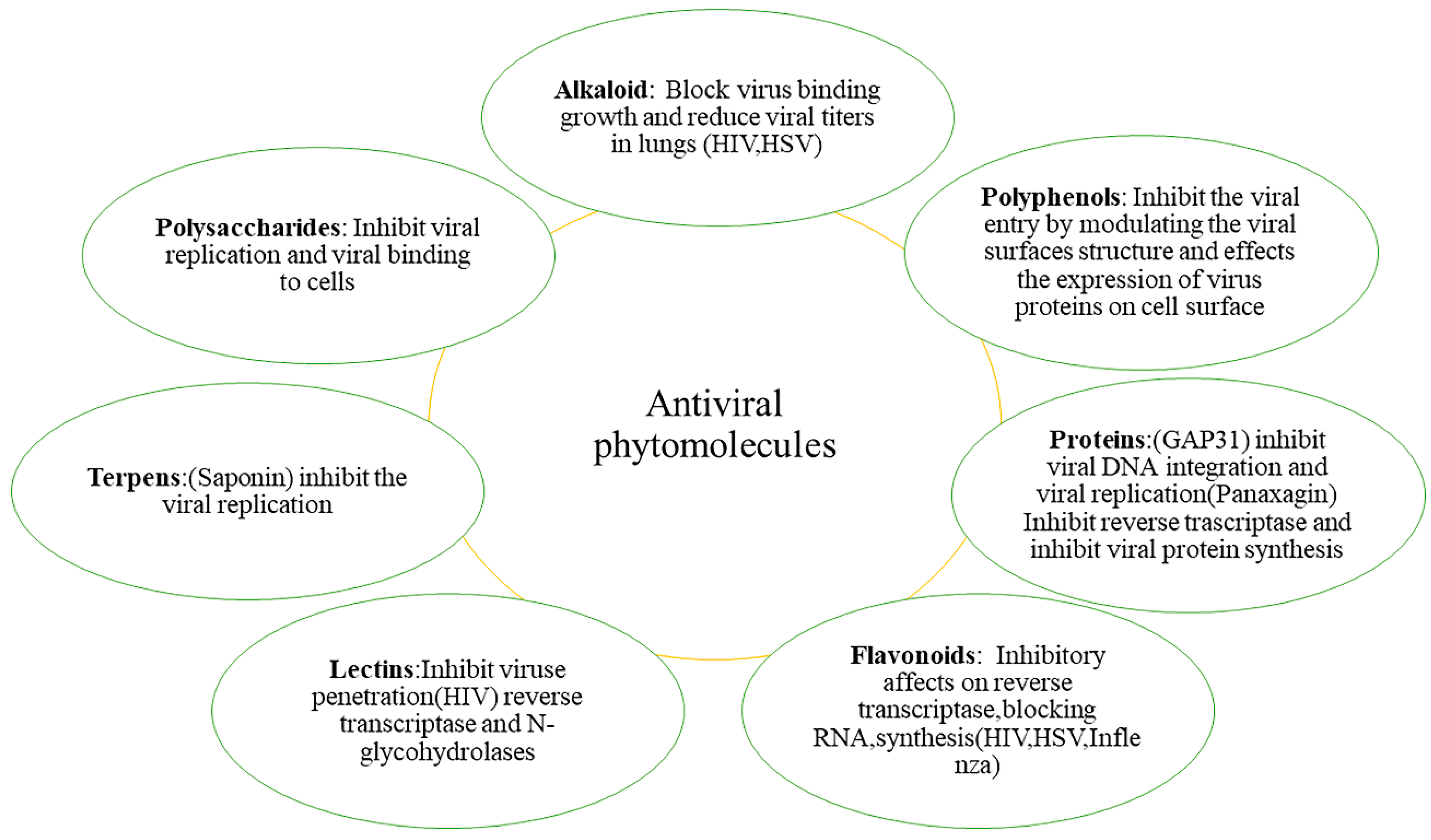
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Behl, T.; Rocchetti, G.; Chadha, S.; Zengin, G.; Bungau, S.; Kumar, A.; Mehta, V.; Uddin, M.S.; Khullar, G.; Setia, D.; et al. Phytochemicals from Plant Foods as Potential Source of Antiviral Agents: An Overview. Pharmaceuticals 2021, 14, 381. https://doi.org/10.3390/ph14040381
Behl T, Rocchetti G, Chadha S, Zengin G, Bungau S, Kumar A, Mehta V, Uddin MS, Khullar G, Setia D, et al. Phytochemicals from Plant Foods as Potential Source of Antiviral Agents: An Overview. Pharmaceuticals. 2021; 14(4):381. https://doi.org/10.3390/ph14040381
Chicago/Turabian StyleBehl, Tapan, Gabriele Rocchetti, Swati Chadha, Gokhan Zengin, Simona Bungau, Arun Kumar, Vineet Mehta, Md Sahab Uddin, Gaurav Khullar, Dhruv Setia, and et al. 2021. "Phytochemicals from Plant Foods as Potential Source of Antiviral Agents: An Overview" Pharmaceuticals 14, no. 4: 381. https://doi.org/10.3390/ph14040381
APA StyleBehl, T., Rocchetti, G., Chadha, S., Zengin, G., Bungau, S., Kumar, A., Mehta, V., Uddin, M. S., Khullar, G., Setia, D., Arora, S., Sinan, K. I., Ak, G., Putnik, P., Gallo, M., & Montesano, D. (2021). Phytochemicals from Plant Foods as Potential Source of Antiviral Agents: An Overview. Pharmaceuticals, 14(4), 381. https://doi.org/10.3390/ph14040381